Abstract
Clostridium perfringens type C is an important cause of enteritis and enterocolitis in foals and occasionally in adult horses. The disease is a classic enterotoxemia, and the enteric lesions and systemic effects are caused primarily by beta toxin, 1 of 2 major toxins produced by C. perfringens type C. Until now, only sporadic cases of C. perfringens type C equine enterotoxemia have been reported. We present a comprehensive description of the lesions in 8 confirmed cases of type C enterotoxemia in foals and adult horses. Grossly, multifocal to segmental hemorrhage and thickening of the intestinal wall were most common in the small intestine, although the colon and cecum were also frequently affected. All horses had variable amounts of fluid, often hemorrhagic intestinal contents. The most characteristic microscopic lesion was necrotizing or necrohemorrhagic enteritis, with mucosal and/or submucosal thrombosis. Numerous gram-positive rods were occasionally seen in affected mucosa. A definitive diagnosis of C. perfringens type C enterotoxemia in all 8 cases was based on the clinical history, gross and histologic lesions, and detection of the beta toxin in intestinal contents.
Enteritis, enterocolitis, and colitis, manifested clinically by diarrhea and colic, are important causes of morbidity and mortality in foals and adult horses. These conditions have been associated with various causes, including Clostridium spp, Salmonella spp, Ehrlichia risticii, Aeromonas spp, Lawsonia intracellularis, cantharidin toxicosis, and larval cyathostomiasis. 31 Together with Salmonella spp, clostridia, including Clostridium perfringens and Clostridium difficile, are the most important agents of equine enterocolitis.
The first reports associating clostridia with enteric disease in foals were published in the 1930s, 13,15 but in the past few decades, C. perfringens and C. difficile have been increasingly reported as relevant pathogens involved in equine enteritis and enterocolitis. 2,6,7,18,21,26,31,32
C. perfringens is classified into 5 types (A, B, C, D, and E) based on the production of 1 or more of 4 so-called major toxins: alpha (CPA), beta (CPB), epsilon (ETX), and iota (ITX). Two other major toxins, enterotoxin (CPE) and beta 2 (CPB2), can be produced by all types of C. perfringens but are not used in its classification. Although C. perfringens types A, B, and C have been associated with enterocolitis in foals, type C is the most commonly reported clostridial enteric pathogen in foals in North America. 2,7,10 C. perfringens type C produces major toxins CPA and CPB. The former is a lecithinase (phospholipase C), which is considered the main virulence factor in C. perfringens type A–associated myonecrosis in humans and animals. However, the contribution of CPA to the virulence of type C isolates is negligible. This was demonstrated by inoculation of rabbits and mice with C. perfringens type C CPA-null mutants (type C isolates genetically modified to produce all toxins except CPA), with no changes observed in the virulence of the organism. 20 CPB, however, is a necrotizing toxin that forms membrane pores in susceptible cells. It is considered responsible for the intestinal necrosis and systemic alterations in type C infections of several animal species, including horses. The importance of CPB toxin was corroborated by lack of virulence in animals experimentally inoculated with CPB-null mutants of type C isolates. 20
Disease caused by C. perfringens type C in many mammalian species, including humans, originates when type C strains proliferate and produce toxins in the intestine. 29 C. perfringens type C causes severe intestinal damage, but death is thought to result mainly from absorption of toxins from the intestine into the circulation. 23,24,28 Therefore, type C infections are considered true enterotoxemias (ie, diseases produced by toxins generated in the intestine but absorbed into the systemic circulation and acting on organs distant from the gastrointestinal tract).
Because CPB is highly susceptible to the action of trypsin, 12,14 neonatal animals are particularly susceptible to type C infections, due to the low level of intestinal trypsin in the first days of life and the presence of trypsin inhibitors in colostrum. Type C disease also occurs in animals and humans that ingest food with trypsin inhibitors and in patients with pancreatic disease. 12
In horses, a presumptive diagnosis of C. perfringens type C enterotoxemia can be based on clinical history (acute onset of diarrhea, colic, or sudden death) and gross and microscopic lesions (necrotizing enteritis or enterocolitis). This presumptive diagnosis can be reinforced by isolation of numerous C. perfringens type C from the small or large intestine. However, because C. perfringens type C can be isolated from some healthy horses, definitive diagnosis should be based on detection of CPB in intestinal contents. 24 CPB can be detected in enteric content by assays in mice or guinea pigs (less common nowadays) or by in vitro methods based on enzyme immunoassays such as ELISA. 24
Published descriptions of equine enterotoxemia caused by C. perfringens type C, confirmed by CPB detection, have been limited to case reports. 6,9,16,18,22 A few reports 7,10,27,32 have described a larger number of animals, but the diagnosis was based on pathology and isolation of C. perfringens type C without toxin detection. In this retrospective study, we describe the pathologic and other laboratory findings in 8 horses with C. perfringens type C enterotoxemia that was confirmed by CPB detection in intestinal contents.
Materials and Methods
Case Selection and Clinical History
Eight cases were selected from the archives of the San Bernardino Branch of the California Animal Health and Food Safety Laboratory System. All cases fulfilled the following 2 conditions: (1) equine carcasses received for necropsy between 2003 and 2010 with a history of gastrointestinal disease and death within 48 hours or sudden death without clinical gastrointestinal disease and (2) positive ELISA test for C. perfringens CPB in intestinal contents.
Table 1 presents age, breed, sex, clinical history, and distribution of lesions in the gastrointestinal tract. Briefly, the 5 male and 3 female horses included 6 foals (n = 4, ≤ 16 days old) and 2 adults distributed among crossbred and 4 different breeds. Most horses had acute illness, followed by death or euthanasia within 2 days of the onset of clinical signs. One foal was found dead without any clinical signs observed. Only 1 horse (No. 4) had received antibiotics (drug not reported) 1 day before onset of clinical signs. The 4 youngest foals (2 to 16 days old) had been nursed by their dams from birth until onset of disease and had not received any additional feed or supplement. The remaining 4 horses (5 months to 6 years of age) had been on a diet of alfalfa hay and a mixture of grains for at least 1 month before onset of disease. No major dietary changes or stress factors were reported in any of the 8 cases.
Signalment, History, and Lesion Distribution in the Intestinal Tract in 8 Horses With Clostridium perfringens Type C Enterotoxemia
Gross and Microscopic Pathology
A necropsy was performed on all horses within 6 to 24 hours of death, and samples of lung, liver, kidney, spleen, stomach, and small and large intestine in all cases, as well as brain, adrenal gland, and skeletal muscle in most cases, were collected and fixed by immersion in 10% buffered formalin, pH 7.2, for 24 to 72 hours. All tissues were processed by standard histologic techniques for the production of 4-μm-thick sections stained with hematoxylin and eosin. Selected sections from the small intestine, cecum, and colon were also stained with Hucker and Conn’s Gram and phosphotungstic acid–hematoxylin.
Bacteriology
Small and large intestinal contents or swabs collected from grossly affected areas of the intestine of each horse were inoculated onto prereduced anaerobically sterilized Brucella Blood Agar (Anaerobic Systems, Morgan Hill, California), prereduced anaerobically sterilized phenylethyl alcohol sheep blood agar (Anaerobic Systems), and egg yolk agar (Anaerobic Systems) and incubated anaerobically at 37°C for 48 hours. Small and large intestinal content from each horse was inoculated onto cycloserine-cefoxitin-fructose agar (Veterinary Media Services, University of California, Davis, Davis, California) and incubated anaerobically at 37°C for 48 hours. Samples from the same specimens were also inoculated into cycloserine-cefoxitin-fructose tube (Veterinary Media Services) and incubated aerobically and anaerobically at 37°C for 48 to 72 hours. All isolates were identified by conventional biochemical techniques. Samples of small and/or large intestinal contents from each animal, as well as individual or pooled samples of liver, spleen, kidney, joint fluid or blood, were inoculated onto Columbia 5% sheep blood agar (Hardy Diagnostics, Santa Maria, California) and MacConkey agar plates (Hardy Diagnostics) and incubated aerobically at 37°C for 48 hours. A real-time polymerase chain reaction (PCR) to detect a fragment of the Salmonella-specific invA gene was performed on small and large intestinal content samples from 3 horses (Nos. 6–8) as described. 4 Briefly, enriched overnight cultures were centrifuged and processed for real-time PCR using the sediment as template. C. perfringens isolates were typed by a multiplex PCR technique to amplify segments specific for the genes encoding CPA, CPB, ETX, ITX, CPE, and CPB2, as described. 1 Briefly, bacteria were grown on brain-heart infusion agar plates (Hardy Diagnostics), incubated anaerobically overnight at 37°C and then processed for multiplex PCR analysis using colony lysates as templates. The multiplex PCR products were then separated in 2% agarose gels, stained with ethidium bromide, and examined by ultraviolet transillumination.
C. perfringens Toxins ELISA
Samples of small and/or large intestinal contents from all horses were tested for CPA, CPB, and ETX via a commercial capture ELISA kit (BIO-X, Brussels, Belgium), following the manufacturer’s instructions. Briefly, the test used 96-well plates sensitized by specific monoclonal antibodies for CPA, CPB, or ETX. Samples were added to wells, and plates were incubated for 60 minutes at room temperature, followed by washing and incubation for 60 minutes with peroxidase-labeled anti-CPA, anti-CPB, or anti-ETX polyclonal antibodies. Plates were washed again, and a mixture of chromogen-substrate (hydrogen peroxide and tetramethyl benzidine) was added. The enzymatic reaction was stopped by acidification with phosphoric acid. Optical densities were read using an ELISA reader with a 450-nm filter. Purified CPA, CPB, or ETX was used in positive control wells; toxins were replaced by buffer in negative control wells. Results were calculated according to the manufacturer’s instructions.
C. difficile Toxins ELISA
Samples of small and/or large intestinal contents from all horses were tested for toxins A and B of C. difficile using a commercial ELISA kit (Techlab, Blacksburg, Virginia) according to manufacturer’s instructions. This kit has antibodies against toxin A and B in the same well. A mixture of purified toxin A and B was used in the positive control wells; toxins were replaced by buffer in negative control wells.
C. perfringens Immunohistochemistry
Paraffin sections, 4 μm in thickness, of ileum and/or colon from 2 of the horses with moderate or large numbers of gram-positive rods on the mucosal surface were processed by an indirect immunoperoxidase technique for C. perfringens, using the Dako EnVision Kit (Dako, Carpenteria, California) according to the manufacturer’s instructions. The primary antibody was rabbit polyclonal anti– C. perfringens (GenWay Bio, San Diego, California). Small intestine from a goat inoculated experimentally with C. perfringens type C was used as positive control tissue. Small intestine from a goat that was culturally negative for C. perfringens was used as a negative control tissue. Additional negative controls consisted of serial tissue sections of the test tissue, incubated with normal rabbit serum instead of the specific antibodies.
Results
Gross Pathology
Gross lesions ( Table 1) were seen in the small intestine of 7 of 8 horses (all but No. 7), in the colon of 3 (Nos. 2, 3, and 7), and in the cecum of 3 (Nos. 1, 2, and 3). Small intestinal lesions were segmental (Fig. 1) in 4 horses (Nos. 1, 3, 6, and 8), with one or more lesions of the jejunum and/or ileum well demarcated and up to 3 m long. Lesions were diffuse (Fig. 2) in 2 horses (Nos. 1 and 5), in which the affected area was more extensive, involving most of the mid and aboral jejunum and/or ileum and usually sparing the duodenum and oral jejunum. The most common small intestinal lesions were intense mucosal and serosal hyperemia and hemorrhage; moderate transmural thickening; dull and dark red ulcerated mucosa (Fig. 3) with or without multifocal, tan to yellowish pseudomembranes (Fig. 4); and abundant brown to red, foul-smelling, fluid intestinal contents. A gelatinous, clear, transmural edema was associated multifocally with areas of severe ulceration and necrosis in 2 horses (Nos. 1 and 2). In horse No. 4, the only gross abnormality was diffuse reddening of the jejunal/ileal mucosa and serosa with abundant watery, red-brown contents (Figs. 5, 6). Lesions in the colon and cecum resembled those in the small intestine; the distribution was multifocal in 3 horses (Nos. 1, 3, and 7), locally extensive to diffuse in 1 horse (No. 2), and not described in the other 4 horses (Nos. 4, 5, 6, and 8). Colonic contents were described as abundant, clear to clear green, and fluid (Nos. 2, 5, 7, and 8); brown or red and fluid (Nos. 1 and 3); and pasty yellow or normal looking (No. 4). Mesenteric lymph nodes were moderately enlarged and edematous and/or hemorrhagic in 1 case (No. 2). Gross lesions other than those in the gastrointestinal tract included serous or serosanguineous pericardial effusion (Nos. 1, 2, 3, and 8), pulmonary edema and congestion (No. 2), and multifocal petechiae and ecchymoses of endocardium and serosal surfaces, including the peritoneum, pleura, and epicardium (Nos. 1, 2, and 6).
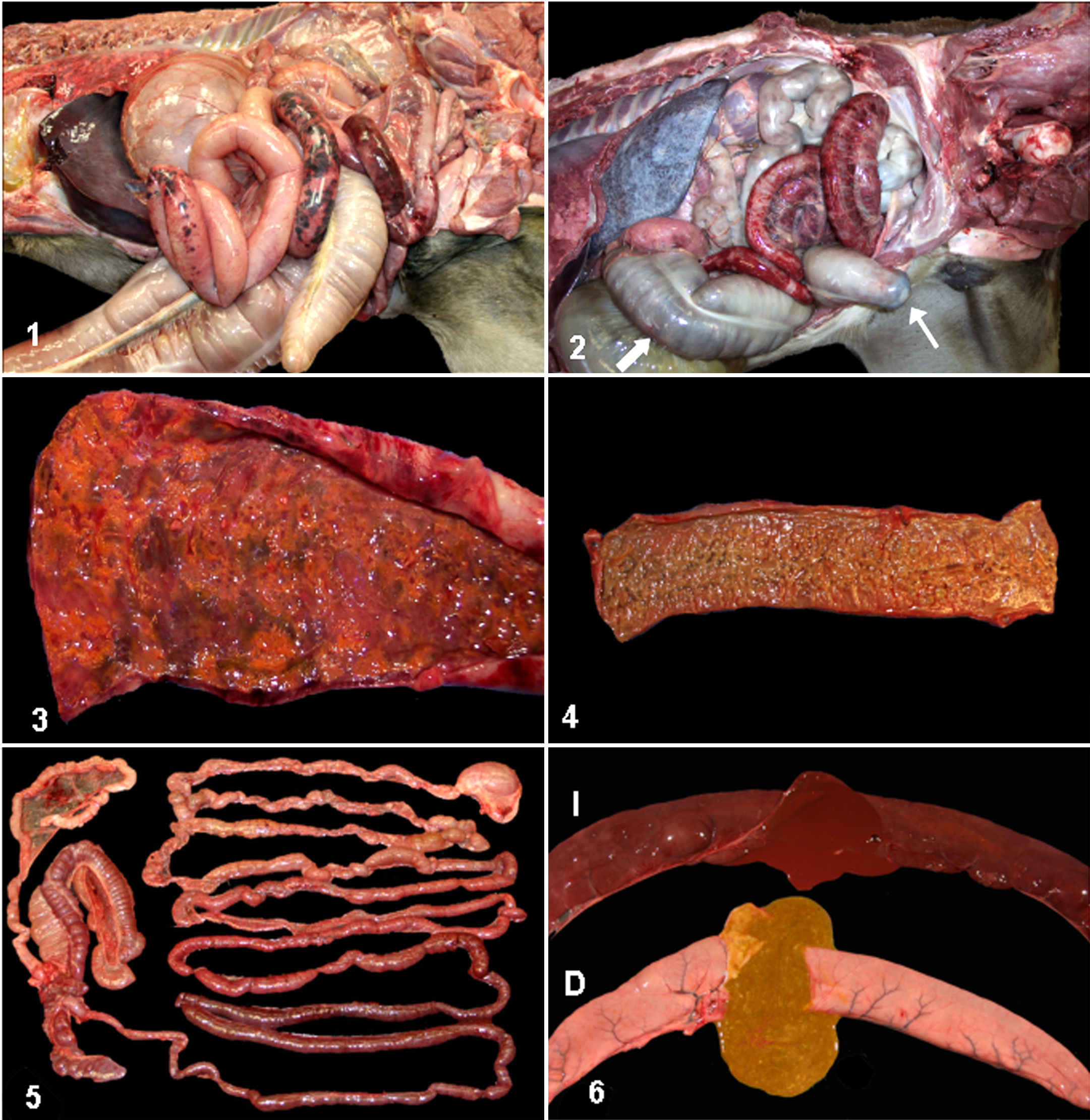
Abdominal cavity, horse No. 6. Segments of small intestine have transmural, multifocal, necrohemorrhagic enteritis visible from the serosal surface.
Small intestine, horse No. 3. Transmural necrohemorrhagic enteritis. The mucosa is diffusely necrotic, and there is loss of mucosal/submucosal boundary, submucosal emphysema, and transmural hemorrhage. HE. See Figure 11 for box.
Microscopic Pathology
Histologically, necrotizing or necrohemorrhagic lesions were observed in the gastrointestinal tract of all the horses; lesion distribution was generally consistent with the distribution of the gross lesions. Histologic sections of the small and/or large intestine had at least 2, but usually 3 or more, microscopic lesions, including (from most to least commonly present) mucosal necrosis (8 of 8), mucosal and/or submucosal thrombosis (8 of 8), mucosal and/or submucosal hemorrhage (5 of 8), submucosal edema (5 of 8), mucosal and/or submucosal congestion (4 of 8), mucosal and/or submucosal leukocytic infiltration (4 of 8), fibrinonecrotic pseudomembranes (3 of 8), numerous gram-positive rods on the superficial mucosa (2 of 8), and fibrinoid necrosis of submucosal blood vessels (1 of 8). Severity of the lesions was similar in the small and large intestine when both portions of the gastrointestinal tract were affected ( Table 2).
Histologic Lesions in the Small or Large Intestine of 8 Horses With Clostridium perfringens Type C Enterotoxemia

–, not observed; +, mild; ++, moderate; +++, marked/severe; S, small numbers or not observed; M, moderate numbers; L, large numbers; NP, not performed.
a Immunohistochemistry.
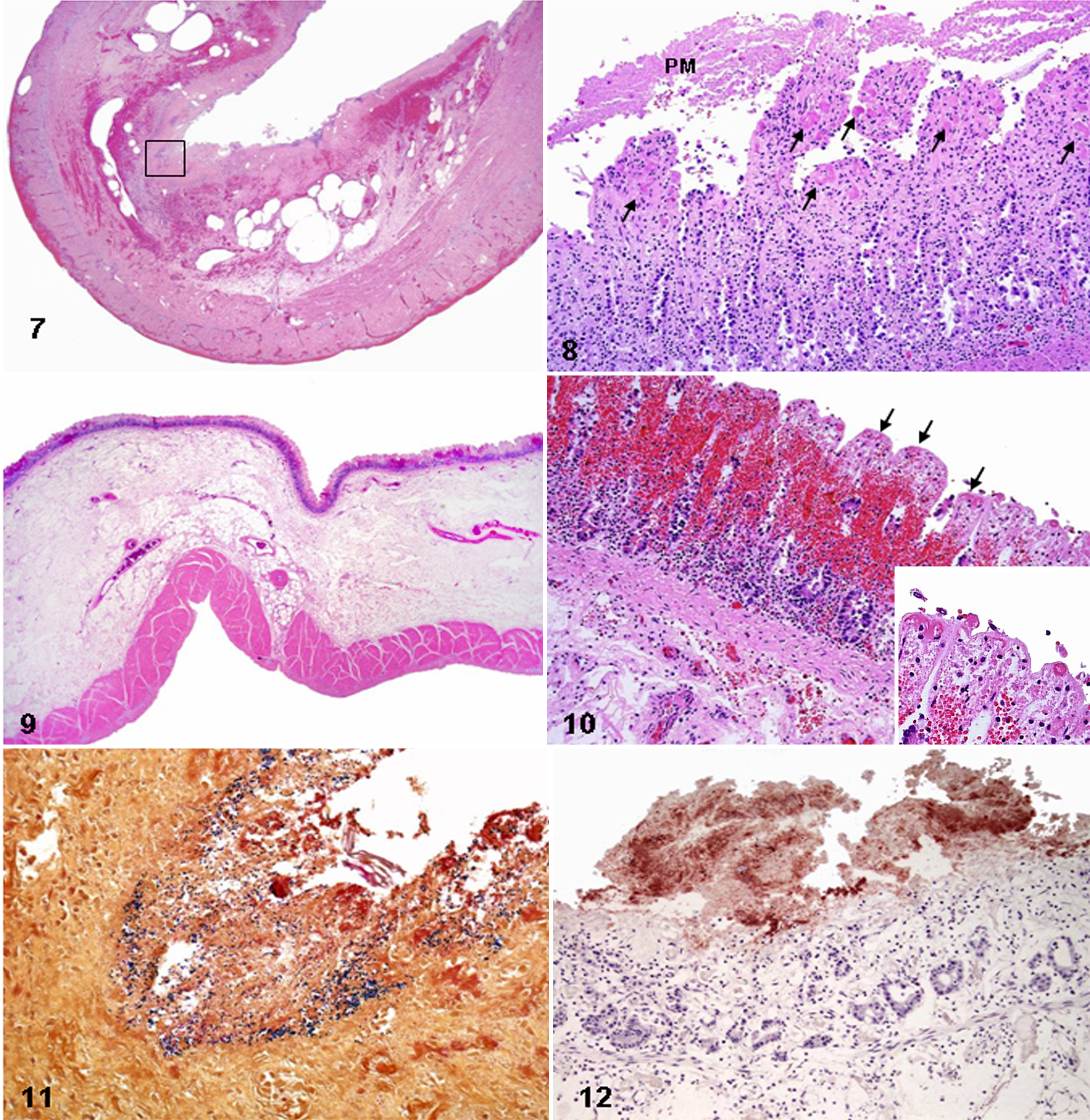
Mucosal necrosis was always coagulative, often full thickness, and characterized by mucosal hypereosinophilia, loss of the mucosal epithelial lining, attenuation or collapse of the villi and crypts, and moderate to severe hemorrhage and congestion (Figs. 7 –10). Fibrin thrombi (Figs. 8, 10), stained with phosphotungstic acid–hematoxylin, were present in small veins, arterioles, and capillaries of the lamina propria and often also in the small to midsize arteries and veins of the submucosa. Vascular thrombosis was multifocal, and in some cases, a thorough search of multiple sections of the bowel was necessary to detect it. Three cases (Nos. 2, 3, and 6) had a diphtheritic pseudomembrane that was multifocally attached to the necrotic mucosa and composed of abundant fibrin mixed with cellular debris, feed material, and mixed bacteria. Infiltration of leukocytes into the lamina propria or submucosa was not a prominent feature, and in only 4 cases (Nos. 1, 2, 5, and 6), a mild to moderate number of neutrophils mixed with lymphocytes and plasma cells, as well as cellular debris, diffusely infiltrated the mucosa and submucosa. Numerous gram-positive rods (Fig. 11) were observed multifocally in the lumen and/or along the surface of the small and large intestinal mucosa in 2 horses (Nos. 2 and 3). These rod-shaped bacteria were strongly positive for C. perfringens by immunohistochemistry (Fig. 12). Dilated lymphatic vessels were observed in 4 horses (Nos. 1, 3, 5, and 6), commonly in the submucosa of the small intestine and rarely in the large intestine. One case (No. 6) had severe fibrinoid necrosis of the submucosal blood vessels. Microscopic lesions in organs other than the intestine were inconsistently present and included pulmonary congestion, hemorrhage, and edema (Nos. 1, 2, 5, 6, and 7); pulmonary thrombosis (Nos. 1 and 7); embolic pneumonia (No. 5); subepicardial and subendocardial hemorrhage (1, 2, and 6); lymphoid depletion (Nos. 5 and 7); periportal lymphohistiocytic and neutrophilic hepatitis (No. 1); congestion and hemorrhage of multiple visceral organs and serosal surfaces (Nos. 1, 2, and 6); mesenteric lymph node edema, congestion, and hemorrhage (Nos. 1 and 2); and acute renal tubular degeneration and necrosis (No. 1).
Bacteriology
C. perfringens was cultured from the intestinal contents from 7 of 8 horses ( Table 3). Of these, 4 isolates were identified as type C and 4 as type A. Mixed aerobic and anaerobic flora, without growth of C. perfringens, was obtained from the remaining horse. In addition, a variety of other aerobic and/or anaerobic microorganisms were isolated from all cases. No C. difficile was isolated from any horse. No Salmonella spp was isolated or detected by PCR in any horse.
Laboratory Test Results From 8 Horses With Clostridium perfringens Type C Enterotoxemia
PCR, polymerase chain reaction; SI, small intestine; R, rare; L, large; NP, not performed; Neg, negative; Pos, positive.
a Salmonella culture and/or PCR was negative in all samples of the small and/or large intestine analyzed.
C. perfringens and C. difficile Toxins ELISAs
The C. perfringens toxin ELISA results from the intestinal contents are summarized in Table 3. All 8 horses tested positive for CPB; 4 were positive for CPA; and none was positive for ETX. No toxins (A/B) of C. difficile were detected in the intestinal contents of any horse.
Discussion
The objective of this study was to document the lesions of confirmed type C enterotoxemia in a series of equine cases because previous reports have been based on individual cases, often with incomplete laboratory workup. 6,9,16,18 In all cases in this retrospective study, a presumptive diagnosis of C. perfringens type C enterotoxemia was based on gross and microscopic lesions of necrotizing enteritis and/or enterocolitis and confirmed by detection of CPB in the intestinal content. Detection of CPB is considered the gold standard for diagnosis of C. perfringens type C infections in humans and animals. 24 The “molecular Koch’s postulates” for C. perfringens type C infection have been fulfilled in several species, including rabbits and mice, 20 via elimination of virulence by removing the gene that encodes CPB from virulent type C isolates and via restoration of virulence by reinserting the gene. 20 In this case series, C. perfringens was isolated from the small and/or large intestine of 7 of 8 horses, and 4 of these 8 isolates were identified as type C. Failure to isolate C. perfringens type C in some of our cases does not preclude a diagnosis of type C enteritis, because detection of CPB was positive in all cases. C. perfringens type C can grow in the intestine in pockets, 23 with some segments devoid of this microorganism. It is therefore possible that in the culturally negative cases, the sample was collected from areas of the intestine where C. perfringens type C was not present or was present in numbers below the sensitivity of the culture method. C. perfringens type C is rarely isolated from the intestine of normal foals (our unpublished observation), which lends support to the idea that isolation of this microorganism from foals with necrotizing enteritis has at least presumptive diagnostic value. C. perfringens type A (CPE positive), but not type C, was isolated from the intestine of 4 horses in our study. C. perfringens type A is considered a normal inhabitant of the equine intestine, so isolation of this microorganism was probably an incidental finding. However, several authors 2,7,11 have suggested a pathogenic role for C. perfringens type A in equine enterocolitis, so a possible synergism between C. perfringens type A and C. perfringens type C cannot be ruled out. The 4 C. perfringens type A isolates from our horses carried the CPE gene. Enterotoxigenic C. perfringens type A has been associated with equine enterocolitis, 2 and it is possible that CPE acted synergistically with CPB to produce the lesions observed in this study. It is also possible that the CPE produced by type C isolates (1 of our type C isolates carried the CPE gene) was also implicated in the pathogenesis of enterocolitis in these cases. However, the presence of CPE in the intestinal content was not investigated, so this possibility remains only speculative. CPA was detected in 4 of the 8 horses in this study. Although all types of C. perfringens carry the gene that encodes this toxin, not all C. perfringens strains produce detectable concentrations of CPA. Because similar lesions were observed in horses with and without CPA in the intestinal content, it is unlikely that this toxin contributed much to lesion development. Moreover, it was recently demonstrated that this toxin has negligible action in the intestinal virulence of C. perfringens type C isolates. 20 However, as for CPE, we cannot completely rule out a synergistic action between CPA and CPB.
The most consistent gross and histologic finding in this series of cases was necrotizing enteritis with mucosal and/or submucosal thrombosis. It is noteworthy that the identification of mucosal necrosis, grossly or histologically, may be hampered by autolysis, in which case, histologic evidence of mucosal or submucosal thrombosis was the most important indicator of intestinal damage. Although neither mucosal necrosis nor thrombosis is specific for type C enterotoxemia in horses, the presence of both lesions narrows the differential diagnosis to a few conditions. Besides C. perfringens type C, agents that can produce acute onset of diarrhea or sudden death, with intestinal necrosis and mucosal and/or submucosal thrombosis in horses, include Salmonella spp and C. difficile. In our study, Salmonella spp was ruled out by negative culture and PCR test results. Neither C. difficile nor its toxins were detected in any horse in this study.
E. coli has been associated with enterocolitis in horses. However, the gross and histologic lesions in this study are very different from those described in E. coli–associated enterocolitis. 8 This difference in lesions, coupled with the fact that E. coli was isolated from the gastrointestinal tract in only 2 of our horses, strongly suggests that this microorganism was not responsible for the lesions described in this study.
C. cadaveris was associated with experimental colitis in horses treated with lincomycin. 25 That microorganism was ruled out in our study by negative anaerobic culture results. In addition, only 1 horse had received antibiotic treatment, a predisposing factor that was considered responsible for colitis in the cited study, 25 before onset of clinical signs.
Traditionally, type C infections have been reported in newborn animals of several species, including horses. 23,24,29 The reason for this age predisposition is thought to be the very low level of trypsin activity in the intestine of newborn animals, which allows CPB to remain active. This toxin is exquisitely sensitive to the action of trypsin and other proteases normally present in the intestinal content; therefore, intestinal trypsin is considered an important innate defense mechanism against type C infections. 14 However, malnutrition, trypsin deficiency, and diets rich in trypsin inhibitors, such as sweet potato or soy bean products, predispose people and animals to type C infections. 12 The sensitivity of CPB to trypsin explains why attempts to reproduce type C enterotoxemia in animal models by inoculating CPB toxin orally or intraduodenally have consistently failed or produced only low lethality. 28 In contrast, experimental mice and rabbits inoculated with CPB or type C cultures mixed with trypsin inhibitor developed clinical signs and/or lesions characteristic of type C infections. 20,30
Four of the 8 cases in this study were older foals or adult horses. The pathogenesis of type C enterotoxemia in horses after the first few weeks of life is poorly understood, but low intestinal trypsin activity, as in the neonates, could have been an important factor. Low intestinal trypsin activity may be a consequence of trypsin inhibitors in feed; acute pancreatic necrosis, pancreatitis, or exocrine pancreatic insufficiency; changes in the intestinal microflora; and administration of certain antibiotics. 3,5,17,19 Antimicrobials are known predisposing factors for colitis in horses, especially that produced by C. difficile. The role of previous antibiotic treatment in the pathogenesis of C. perfringens type C colitis is not known. Nevertheless, only 1 horse in our study had received antibiotics before the onset of intestinal disease, which rules out antimicrobial treatment as a predisposing factor in the majority of the cases. Importantly, type C enterotoxemia in the nonneonatal horse may not be as uncommon as once believed.
In summary, C. perfringens type C enterotoxemia occurs most commonly in foals but also in adult horses and should be included in the differential diagnosis in cases with a history of sudden death or acute onset of colic, with or without diarrhea. The hallmark lesions were necrotizing enteritis and mucosal/submucosal thrombosis. Although isolation of numerous C. perfringens type C from the intestinal tract of affected horses is highly suggestive of the disease, the gold standard for diagnosis of type C enterotoxemia is the detection of CPB toxin in the intestinal contents.
Footnotes
Acknowledgements
We thank J. Saputo, A. Curtis, E. Hurley, D. Paulson, P. Yant, R. Cazares, S. Fitisemanu, and M. Rhea for excellent technical assistance.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
