Abstract
Clostridium difficile is commonly associated with diarrhea and colitis in humans and other mammals, including horses. To this date, the epidemiologic, microbiologic, clinical, and diagnostic aspects of C. difficile–associated disease (CDAD) in horses have been thoroughly described. However, reports describing the enteric pathology of this disease in horses are limited. This study presents a comprehensive description of the pathologic characteristics of CDAD in 21 horses and discusses the criteria for the diagnosis of the disease. Case selection was based on C. difficile A/B toxins detection (enzyme-linked immunosorbent assay) in intestinal content samples accompanied by compatible gross and microscopic enteric lesions. Grossly, multifocal, segmental, or diffuse hemorrhage; congestion; and/or marked gelatinous edema of the intestinal wall with abundant bloody or green watery contents were observed. Histologically, the most common lesion was severe necrotizing or necrohemorrhagic enteritis, colitis, or typhlocolitis, with mucosal and/or submucosal thrombosis and marked submucosal edema. The pathology of CDAD in horses is similar to that caused by other equine enteric pathogens; therefore, a definitive diagnosis requires detection of C. difficile A/B toxins in the intestinal contents.
Clostridium difficile is an anaerobic, Gram-positive, spore-forming bacillus commonly associated with diarrhea and colitis in humans and other mammals. 16,28 The essential virulence factors of C. difficile are toxin A (TcdA) and toxin B (TcdB). 17 There has been recent controversy about which of the 2 toxins is essential for virulence. However, recently published information based on experimental hamster infection models suggests that both TcdA and TcdB are important for the manifestation of C. difficile–associated disease (CDAD). 18,19
C. difficile is the most commonly identified cause of antibiotic-associated and nosocomial diarrhea in humans. 2,32 Horses are susceptible as adults or foals and may develop CDAD as early as the first few days of life. 1,13,16,20 In adult horses, the disease has been reported most commonly after antibiotic therapy and during hospitalization but also in horses in their home stable without prior antibiotic treatment. 4 –7,20,38 In foals, the disease may present in the form of outbreaks or sporadic cases. 5,14,21,22,34,36 Besides antibiotic therapy and hospitalization, other possible risk factors that may alter the intestinal flora predisposing horses to CDAD include stress, change of diet, transportation, starvation, nasogastric intubation, and surgical or medical treatment. 5 The carrier rate of C. difficile is low in asymptomatic foals and adult horses, with most studies showing a rate of isolation from feces of clinically normal adults and foals ranging from 0% to 4.3% and 0% to 3%, respectively. 5,7,14,16,20,22,36 A horse can be infected with vegetative cells of C. difficile or its spores from other infected horses, a contaminated environment, or human beings. It is also possible that C. difficile present in low levels in the gastrointestinal tract of subclinical carriers may proliferate when predisposing factors appear. 5 A presumptive diagnosis of equine CDAD can be based on the clinical history, clinical symptoms, and compatible gross and microscopic lesions in the small and/or large intestines, but confirmation requires toxin detection in intestinal contents and/or feces. 5,9,16
Numerous authors have thoroughly discussed the epidemiologic, microbiologic, clinical, and diagnostic aspects of CDAD in horses, 1,5,6,9,11,16,20,22 –24,26,29,31,35 –38 but studies describing the pathology of this disease are only limited to a single adult horse and 4 foals. 13,23 In this retrospective study, we thoroughly describe the pathology of CDAD confirmed by TcdA and/or TcdB toxin detection in 21 horses and discuss the current diagnostic criteria for this disease.
Materials and Methods
Case Selection and Clinical History
Twenty-one cases of CDAD were selected from the archives (1997–2012) of the San Bernardino branch of the California Animal Health and Food Safety Laboratory system. All horses fulfilled the following 2 conditions: (1) they had a positive result for C. difficile TcdA/TcdB in the small and/or large intestinal contents, and (2) they had a clinical history of acute onset of colic, diarrhea, and toxemia or sudden death, coupled with pathologic evidence of intestinal disease.
Gross and Microscopic Pathology
Necropsy was performed on all horses within 6 to 24 hours of death, and samples of lung, liver, kidney, spleen, stomach, small and large intestine, and adrenal gland in all cases, as well as skeletal muscle and brain in most cases, were collected and fixed in 10% buffered formalin, pH 7.2, for 24 to 72 hours. All tissues were processed by standard histologic techniques for the production of 4-μm-thick sections, which were stained with hematoxylin and eosin. Selected sections of the small intestine, cecum, and colon were also stained with Gram stain.
Bacteriology
Small and large intestinal contents or swabs collected from grossly affected areas of the small and/or large intestine of each horse were inoculated onto prereduced Brucella blood agar (Anaerobic Systems, Morgan Hill, CA), prereduced phenylethyl alcohol sheep blood agar (Anaerobic Systems), and egg yolk agar (Anaerobic Systems) and incubated anaerobically at 37°C for 48 hours. Small and large intestinal content from each horse was inoculated onto cycloserine-cefoxitin-fructose agar (Veterinary Media Services, University of California, Davis) and incubated anaerobically at 37°C for 48 hours. Samples from the same specimens were also cultured for C. difficile into cycloserine-cefoxitin-fructose broth (Veterinary Media Services, University of California, Davis) and incubated aerobically and anaerobically at 37°C for 48 to 72 hours. Suspect colonies were identified by standard biochemical techniques. Samples of small and/or large intestinal contents from each animal, as well as individual or pooled samples of liver, spleen, kidney, joint fluid, or blood, were inoculated onto Columbia 5% sheep blood agar (Hardy Diagnostics, Santa Maria, CA) and MacConkey agar plates (Hardy Diagnostics) and incubated aerobically at 37°C for 48 hours. In addition, small and large intestinal contents from 26 Thoroughbred racehorses that had been euthanized due to catastrophic leg injuries but were otherwise in good health were collected within 24 hours of death and used as controls to monitor the prevalence of C. difficile adult healthy carriers. These samples were also subjected to C. difficile culture as described above.
TcdA and TcdB Enzyme-Linked Immunosorbent Assay
TcdA and TcdB were investigated in small and/or large intestinal content of the 21 horses with intestinal disease and of the 26 control racehorses with no intestinal disease, using a commercial capture enzyme-linked immunosorbent assay (ELISA) kit, following the manufacturer’s instructions (Techlab, Blacksburg, VA). This kit has antibodies against TcdA and TcdB in the same well. A mixture of purified TcdA and TcdB was used in the positive control wells; toxins were replaced by buffer in negative control wells.
Clostridium perfringens Toxins Enzyme-Linked Immunosorbent Assay
Clostridium perfringens alpha (CPA), beta (CPB), and epsilon (ETX) toxins were investigated in small and/or large intestinal content of 13 horses using a commercial capture enzyme-linked immunosorbent assay (ELISA) kit following the manufacturer’s instructions (BIO-X, Brussels, Belgium). Purified CPA, CPB, or ETX was used in positive control wells; toxins were replaced by buffer in negative control wells.
Results
Clinical History
The clinical history, including clinical signs, antibiotic administration, and/or hospitalization record prior to the development of disease, is presented in Table 1. Breeds represented in this study were Thoroughbred (n = 6), Quarter horse (n = 6), Paint (n = 3), Warmblood (n = 2), Lucitano (n = 1), Andalusian (n = 1), Azteca (n = 1), and Gypsy Vanner (n = 1). The age of the horses ranged from 2 days to 9 years, with 4 horses being younger than 1 month of age, 5 horses between 1 month and 1 year, and 12 horses older than a year. There were 6 females, 6 were castrated males, and 9 were entire males. Fifteen horses had a history of antibiotic therapy and/or hospitalization before the development of CDAD; in 3 horses, previous administration of antibiotics was unknown but the animals had not been hospitalized, and 3 foals had not been given antibiotics or been hospitalized.
Clinical History of 21 Horses With Confirmed Clostridium difficile Enteric Infection.
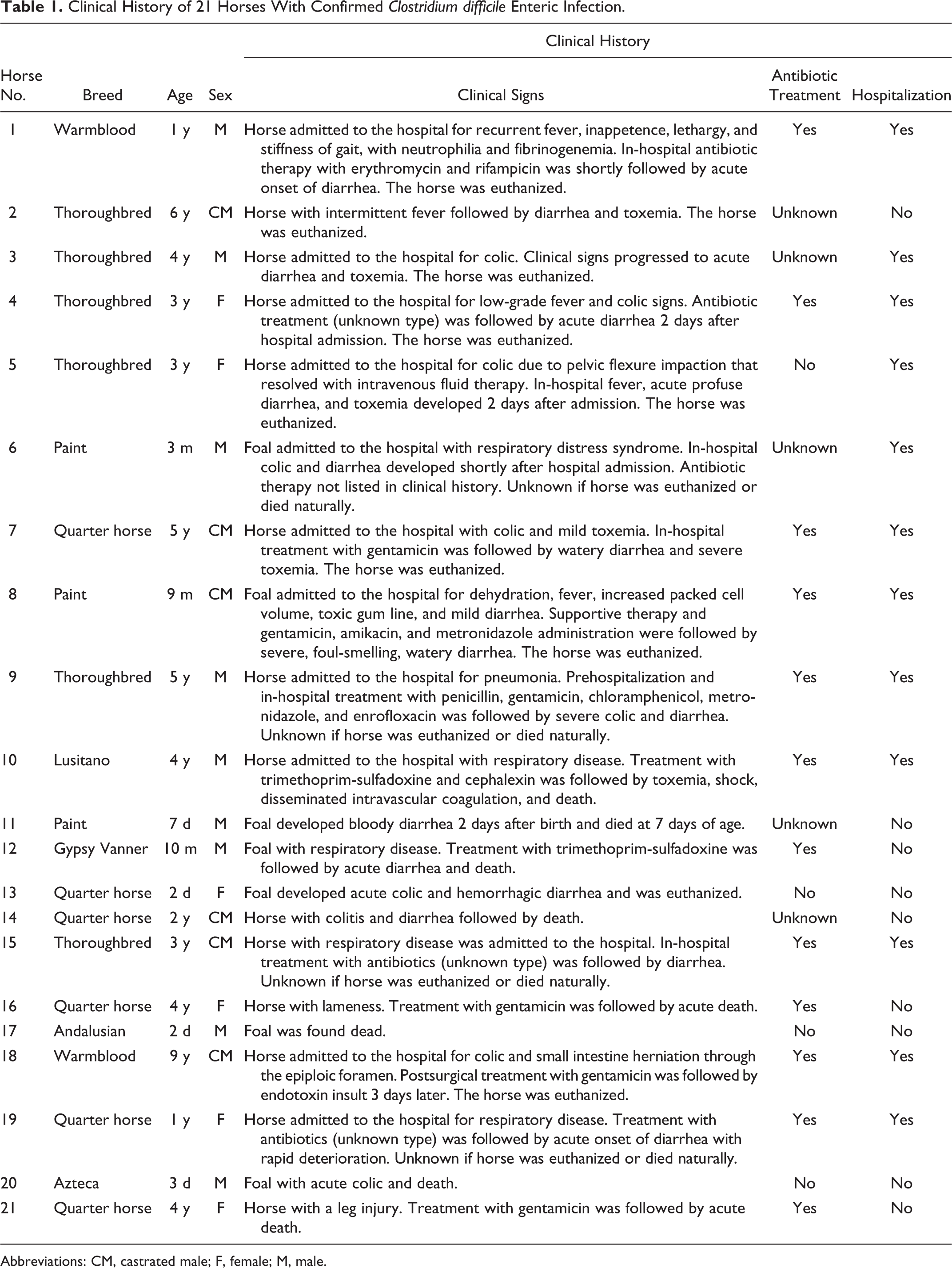
Abbreviations: CM, castrated male; F, female; M, male.
Toxin Detection and Bacteriology
Table 2 summarizes the most important bacteriological results from the 21 horses included in this study. TcdA and TcdB were detected by ELISA in small and/or large intestinal content of all 21 horses. Nineteen of these 21 horses were positive for C. difficile culture in enriched C. difficile culture media and/or routine anaerobic culture, 1 was negative to both cultures, and in 1 horse, the intestinal content was not cultured in enriched C. difficile culture media and the microorganism did not grow in the routine anaerobic culture. In addition, neither C. difficile nor its toxins, TcdA and TcdB, were detected in the small or large intestinal content samples from the 26 control racehorses that were euthanized due to catastrophic leg injuries. The C. perfringens toxin ELISA was negative for CPB and ETX in the 13 horses in which the intestinal contents were tested and positive for CPA in 4 of these 13 samples. All CPA-positive samples were from foals younger than 7 days. Routine anaerobic cultures from small intestine, colon, and/or cecum yielded C. perfringens in 4 cases and Clostridium bifermentans and Clostridium sp in 1 case each. The remaining samples produced no growth or yielded a variety of nonpathogenic mixed anaerobic flora. Samples from small intestine, cecum, and/or colon from all 21 horses were negative for Salmonella sp by culture. Aerobic cultures performed on small intestine, colon, and/or cecum samples from 15 horses yielded a variety of microorganisms, including Escherichia coli, Enterococcus sp, Streptococcus equi subsp zooepidemicus, Aeromonas hydrophila, and mixed flora or were reported as no growth after 48 hours.
Microbiological Test Results From 21 Horses Affected With Clostridium difficile Enteric Infection That Were Positive for TcdA and TcdB (ELISA) in the Intestinal Contents.
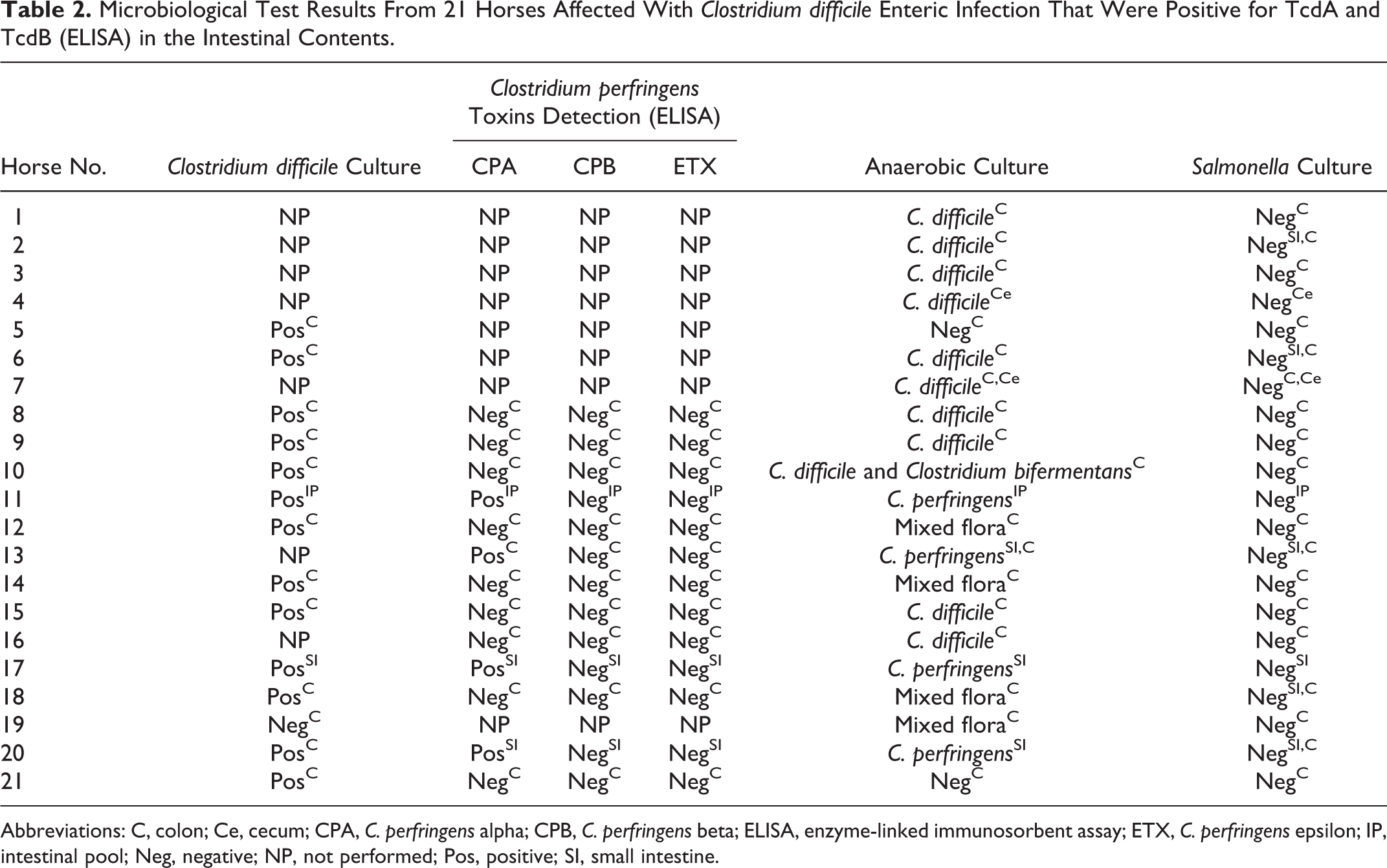
Abbreviations: C, colon; Ce, cecum; CPA, C. perfringens alpha; CPB, C. perfringens beta; ELISA, enzyme-linked immunosorbent assay; ETX, C. perfringens epsilon; IP, intestinal pool; Neg, negative; NP, not performed; Pos, positive; SI, small intestine.
Gross Pathology
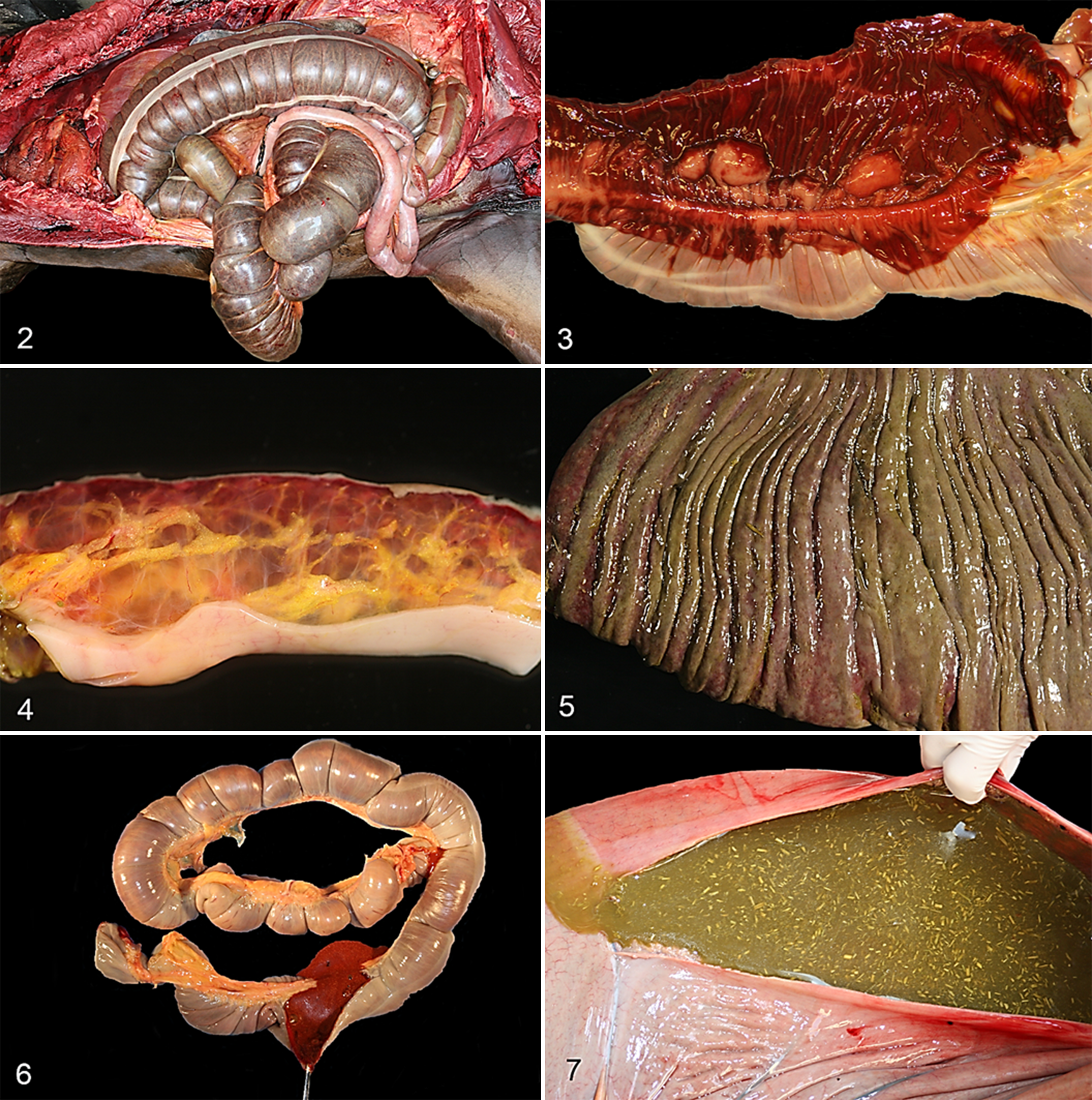
The distribution of the lesions is summarized in Fig. 1. All 4 horses younger than 1 month had lesions in the small intestine, and 3 of these also had lesions in the colon and 1 in the cecum; 5 horses between 1 month and 1 year of age had lesions in the colon, but the small intestine and cecum were not involved; and 12 horses older than 1 year had lesions in the colon and frequently also in the cecum, but small intestine lesions were not recorded. The external appearance of the small and large intestines is characterized by a multifocally hemorrhagic or diffusely darker serosal surface (Fig. 2) as the result of diffuse mucosal congestion and/or hemorrhage (Fig. 3). The wall of the small intestine can be normal thickness or thickened; the mucosa is often diffusely reddened and may have an overlying, multifocal, tan to orange pseudomembrane. The wall of the affected colon and/or cecum is usually moderately to markedly thickened by severe, gelatinous, clear, or sometimes hemorrhagic, submucosal edema (Fig. 4), and the mucosa may be diffusely dull, pale green or dark red and may have multifocal to coalescing ulcers with or without an overlying tan to yellowish or greenish pseudomembrane (Fig. 5). In the affected newborn foals, the small and/or large intestinal contents were composed of yellowish, light brown or red, foul-smelling, dense fluid (Fig. 6). In horses older than 1 month, the large intestinal contents varied from brown or red, foul-smelling, dense fluid to very abundant green, watery fluid with scant feed (Fig. 7). Gross lesions other than those in the gastrointestinal tract were compatible with those seen in endotoxic shock and/or disseminated intravascular coagulation (DIC), including serous or serosanguineous pericardial effusion, pulmonary edema and congestion, and multifocal subendocardial and subserosal petechiae and ecchymoses. Animals with a history of respiratory disease before the onset of C. difficile infection had preexistent pneumonia, pleuropneumonia, or pulmonary abscesses.
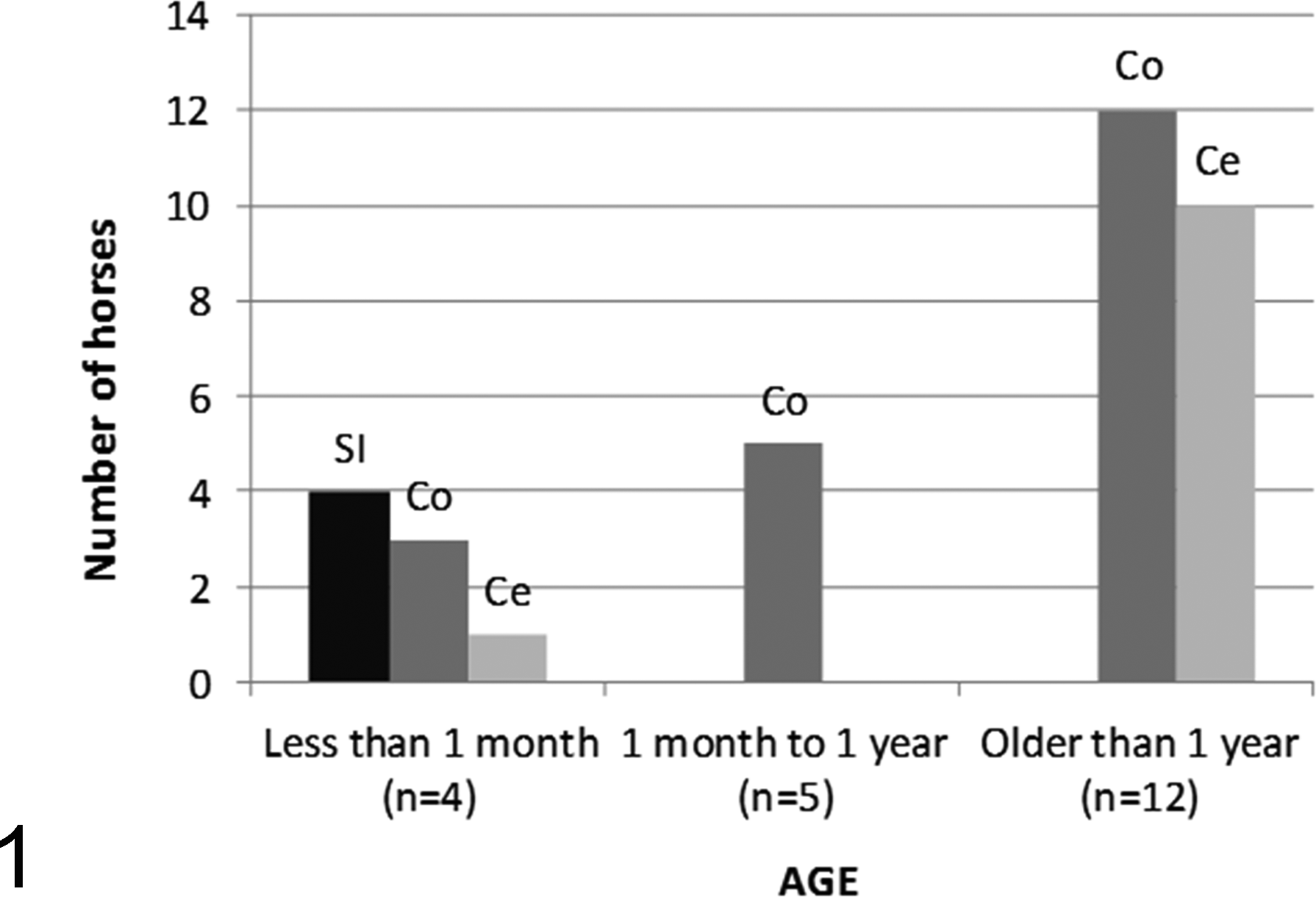
Anatomical distribution of the gross lesions by age from 21 horses with Clostridium difficile enteric disease. Ce, cecum; Co, colon; SI, small intestine.
Microscopic Pathology
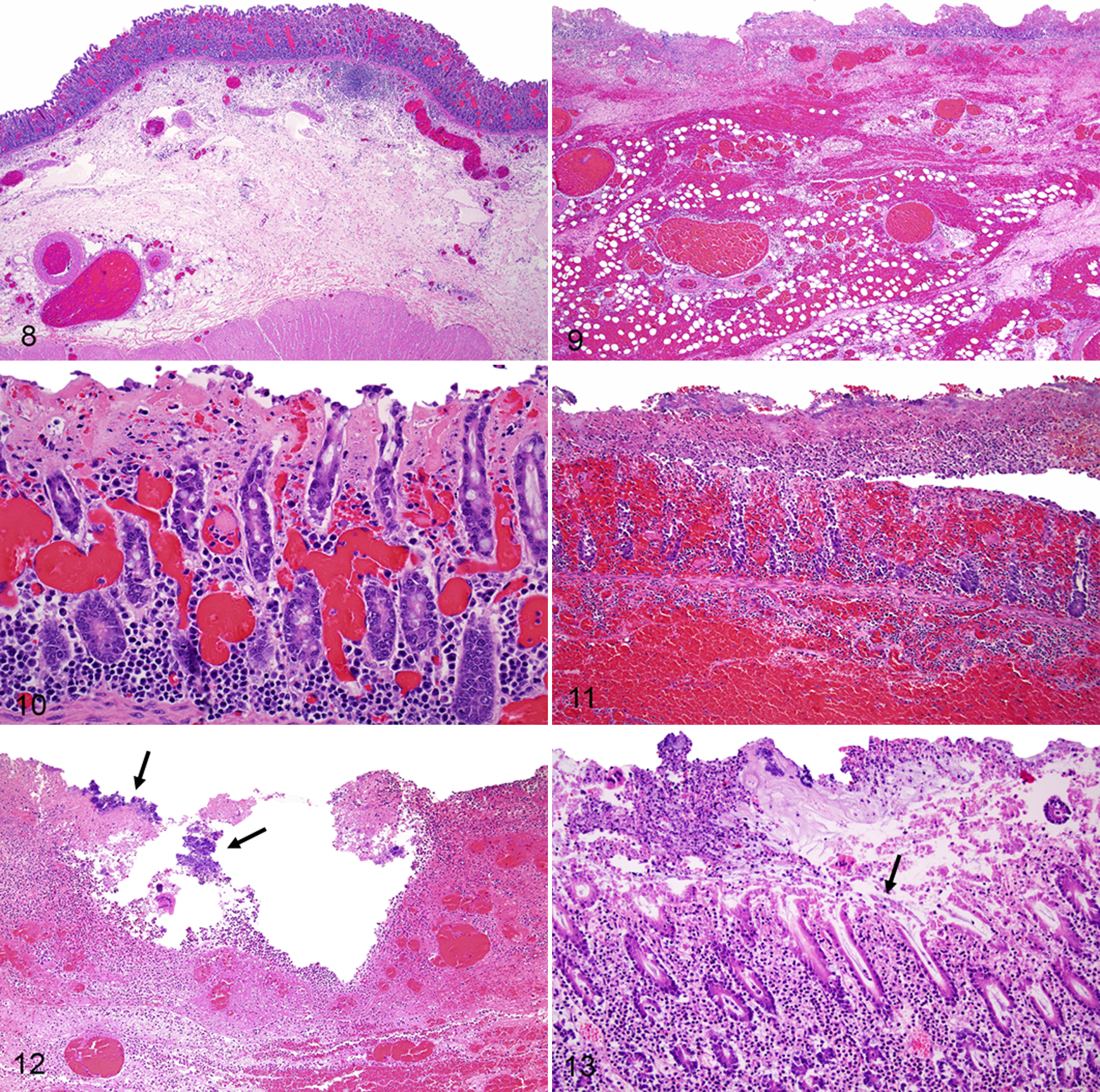
Histologically, necrotizing or necrohemorrhagic enteritis, colitis, and/or typhlitis were observed in the gastrointestinal tract of 20 horses; in 1 foal with markedly hemorrhagic small intestinal contents, the microscopic examination was hampered by severe tissue autolysis. The microscopic lesion distribution was consistent with the distribution of the gross lesions. The severity of the lesions ranged from moderate to severe, but in a few cases, the severity was difficult to determine due to advanced tissue autolysis. Histologically, the small and large intestines had a combination of mucosal necrosis, mucosal and/or submucosal thrombosis, mucosal and/or submucosal hemorrhage, submucosal edema, mucosal and/or submucosal congestion, mucosal and/or submucosal neutrophilic infiltration, fibrinonecrotic pseudomembranes, and occasionally numerous Gram-positive rods on the superficial mucosa. The mucosal coagulative necrosis was often full thickness, erosive, or ulcerative and characterized by partial or complete loss of the epithelial lining with or without hypereosinophilia and usually accompanied by moderate to severe mucosal and/or submucosal hemorrhage, congestion, and edema (Figs. 8, 9). Vascular thrombosis was frequently present and involved venules, arterioles, and capillaries of the lamina propria and sometimes the small- to mid-size arteries and veins of the submucosa (Fig. 10). A diphtheritic pseudomembrane overlying the necrotic mucosa and composed of abundant fibrin mixed with cell debris, feed material, and mixed bacteria was sometimes present (Fig. 11). Mild to moderate mucosal and submucosal fibrino-neutrophilic infiltration with fewer plasma cells, lymphocytes, and macrophages was frequent, and occasionally, numerous clusters of short and thick bacilli (usually Gram-positive rods) could be observed in the lumen and/or along the superficial epithelial lining of the small and large intestinal mucosa (Fig. 12). The so-called volcano lesions characterized by patchy focal erosions on the colonic mucosa, through which fibrin and neutrophils exude, were rarely present (Fig. 13). Moderately to markedly dilated submucosal lymphatic vessels were frequently observed.
Discussion
The aim of this study was to comprehensively document the lesions of confirmed C. difficile infection in a series of 21 equine cases, including newborns, older foals, and adult horses, and to discuss the current diagnostic criteria of equine CDAD. Although a presumptive diagnosis was based on the clinical history and gross and microscopic lesions, confirmation of the diagnosis was established based on detection of TcdA and TcdB in the intestinal contents, as proposed by other authors. 16,34 In agreement with a previous report, 16 the distribution of the lesions throughout the intestinal tract in the current study seemed to be dependent on the age of the horses. Foals younger than 1 month had lesions in the small intestine and sometimes colon and cecum, whereas older foals and adults had a more aboral distribution, involving the large intestine but usually sparing the small intestine. However, due to the relatively small number of cases investigated, final conclusions cannot be drawn from this study; a different lesion distribution should therefore not necessarily rule out CDAD. Possible reasons for this age-related pattern of distribution are unknown, and elucidation of the cause would require additional research.
The enteric lesions seen in this study were similar to those described by other authors. 13,23 Multifocal, locally extensive or segmental, often hemorrhagic, necrotizing enteritis, colitis, or typhlitis accompanied by mucosal or submucosal thrombosis, marked mural edema, mild to moderate neutrophilic infiltration, and a superficial pseudomembrane were frequently present. The characteristic volcano lesions described in pigs with CDAD 15,29,30 were rarely observed in this study. One author describes the presence of vasculitis with fibrinoid degeneration of the blood vessels, 23 which was not seen in the intestinal sections examined here. It is possible that fibrinoid degeneration of blood vessels may be a sporadic lesion in horses with CDAD as it is in horses with C. perfringens type C enterotoxemia. 10 Although characteristic, the gross and microscopic changes described in this study are not pathognomonic for CDAD. Other causes of infectious equine enteric disease as C. perfringens type C enterotoxemia, salmonellosis, or ehrlichial enteritis (Potomac horse fever) may cause similar, if not identical, lesions. 8,10,15 The term colitis X was frequently used in the past and is occasionally used nowadays to describe a peracute, often fatal disease of undetermined etiology characterized by sudden onset of shock, dehydration, diarrhea, enterocolitis, and toxemia. 17,25 The gross and microscopic enteric lesions described for colitis X in the past 33 resemble those we now know can be caused by infectious organisms, such as C. difficile, C. perfringens type C, or Salmonella sp. Since the term colitis X does not reflect a specific disease or syndrome, we discourage its use to refer to cases of acute equine enterocolitis of undetermined etiology. A better understanding of the diverse and dynamic intestinal microflora, improvement and broadening of the current diagnostic techniques, and the systematic use of ancillary laboratory tests to aid in the detection of infectious pathogens and their toxins are required to reduce the still significant number of cases of equine enterocolitis of undetermined origin.
C. difficile was cultured from the small intestine, colon, and/or cecum in 19 of 21 horses positive for TcdA and TcdB, which shows some discrepancies between the culture and the toxin assay as others have described. 7,12,36 Negative C. difficile culture/toxin-positive cases should be considered true positives because of a reduced C. difficile recovery rate in aerobically stored fecal samples and culture sensitivity below 100%. 36,37 Positive C. difficile culture/toxin-negative cases should be considered suggestive of C. difficile enteric disease. However, since C. difficile can be isolated from a small percentage of healthy horses, isolation of this microorganism alone does not confirm the disease. Most authors report a nil or very low C. difficile carrier rate in feces of clinically normal adults and foals, ranging from 0% to 4.3% and 0% to 3%, respectively. 5,7,14,16,20,22,36 All of the control samples from 26 Thoroughbred racehorses euthanized due to catastrophic leg injury tested in this study were negative for both culture and toxin assay. The possibility of obtaining false-negative results in the toxin assay because of inactivation of the toxins during transport or storage highlights the importance of the bacterial culture to support the diagnosis of infection by C. difficile. Although rare, it is important to be aware that a low percentage of healthy or sick horses may harbor nontoxigenic strains of C. difficile. 3,34,36 However, in veterinary medicine, toxin genotyping of isolates is not routinely performed.
Other common and potentially fatal causes of infectious or noninfectious acute enterocolitis in horses were ruled out in our study, taking into account the clinical history, necropsy findings, and ancillary laboratory test results, including bacteriology and histopathology. C. perfringens type C enterotoxemia was ruled out based on negative C. perfringens CPB toxin ELISA results in the 4 foals younger than 1 month and in 10 horses older than 1 month. In 7 horses, C. perfringens toxins ELISA was not performed, but coinfection between C. difficile and C. perfringens type C has been reported only in newborn foals up to 7 days of age, 34 and since C. perfringens type C enterotoxemia is rare in older foals and adults, 10 a C. difficile/C. perfringens type C coinfection in these 7 horses older than 1 month is considered highly unlikely. Four foals younger than 1 month were positive for CPA, but the role of this toxin in the pathogenesis of equine colitis has not been well established. CPA can be found in the intestinal tract of many healthy horses (the authors’ personal observation), and it has been recently demonstrated in rabbits and mice that this toxin has negligible action in the intestinal virulence of C. perfringens type C isolates. 27 Salmonella sp were not isolated from any of the horses, and nonsteroidal anti-inflammatory drug (NSAID)–induced colitis, also known as right dorsal colitis, was not considered a possibility based on the clinical history and lesion distribution.
Our results are in agreement with several authors who have reported that prior antibiotic administration and/or hospitalization are significant predisposing factors for the development of CDAD. 3 –7,20 However, CDAD can occur without previous antibiotic administration or hospitalization. 3,34,38 Consequently, the lack of antimicrobial therapy and hospitalization cannot be used to rule out CDAD and, as other authors have stated, it is reasonable to suggest that fecal samples should be submitted for C. difficile toxin testing regardless of the history of antimicrobial use. 38 In foals less than a week of age, a possible synergism between C. perfringens type C and C. difficile has been proposed recently. 34 In that study, in addition to TcdA and TcdB, the authors identified CPB in the intestinal contents of affected animals. The 4 newborn foals included in this study were negative for CPB, and a coinfection between C. perfringens type C and C. difficile was therefore ruled out.
In summary, our results indicate that a presumptive diagnosis of CDAD can be made based on the clinical history and compatible gross/microscopic findings, but a definitive diagnosis requires a thorough laboratory workup to identify TcdA and TcdB toxins and to rule out other possible causes of acute enterocolitis, especially C. perfringens type C enterotoxemia and Salmonella sp. The retrospective study presented here may serve as hypothesis-generating information on which to base plans for future prospective studies on pathogenesis and diagnosis of CDAD in horses.
Footnotes
Acknowledgements
We thank Drs J. Moore, H. Kinde, D. Read, and M. Shahriar for their work in some of the carcasses included in this article; D. Paulson, P. Yant, R. Cazares, E. J. Hurley, and J. Beingesser for excellent technical assistance; and the Universidad Complutense de Madrid for providing author Rodriguez-Bertos with a postdoctoral Amo grant for his stay at UC Davis, CAHFS San Bernardino.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
