Abstract
Canine splenic hemangiosarcoma has a high metastatic rate and short survival time. Currently, the main prognostic parameters are tumor stage and therapy, while data on histologic parameters, such as grade and Ki-67 expression, are scarce. The aims of this study were to compare two methods of assessment of Ki-67, verify their prognostic impact, and define a threshold value based on survival. Thirty-one cases of histologically diagnosed canine splenic hemangiosarcoma, which were treated with splenectomy and had full staging and follow-up information, were collected. Three were stage I, 17 stage II, and 11 stage III. The mean mitotic count (MC) was 23.9 (standard deviation [SD]: 22.1) and the median was 15 (range, 1–93). Immunohistochemistry for Ki-67 was performed, the Ki-67 labeling index (Ki-67LI) was assessed as a percentage of positive neoplastic nuclei per ≥500 cell, and the Ki-67 count (KI-67C) was defined as the average number of positive nuclei using a 1 cm2 optical grid performed in 5, 40× fields. The mean Ki-67LI and Ki-67C were 56.4% (SD: 38.7) and 27.2 (SD: 12.9) and medians were 51% (range, 8.2–55.2) and 26 (range, 5.5–148), respectively. Using a cut-off of 56% and 9, respectively, Kaplan-Meier survival curves showed an association of overall survival with Ki-67LI and MC. In addition to clinical stage, Ki-67LI maintained its prognostic value on multivariate analysis, supporting the role of Ki-67LI as an independent prognostic parameter. Based on these results, we propose a diagnostically applicable cut-off value of 56% for Ki-67LI as a prognostic parameter for canine splenic hemangiosarcoma.
Hemangiosarcoma (HSA) is a malignant neoplasm that has vascular endothelial differentiation. It occurs in several domestic animal species, with the dog being the most frequently affected. 21 HSA can develop in any tissue or organ containing vascular structures, 10 and in the dog, it arises most commonly in the spleen, heart, liver, and skin.19,21
Splenic HSA arises in 24 out of 100,000 dogs and is estimated to represent 7% of all canine malignancies. 7 More than half of the affected dogs die within the first year of diagnosis, and tumor-related mortality is extremely high. 1 Several breeds, including golden retriever, Labrador retriever, boxer, German shepherd, and other large-breed dogs, have an increased incidence of HSAs.6,7,25 Canine splenic HSA is a highly malignant neoplasm with a high metastatic rate and short survival time,4,18 representing one of the most aggressive cancers in dogs.3,24,27
Clinically, splenic HSA can be categorized by a 3-stage classification scheme. Stage I refers to tumors confined to the spleen and less than 5 cm in diameter with no regional lymph node involvement and no evidence of distant metastasis, stage II refers to ruptured splenic tumors or tumors greater than 5 cm in diameter with or without regional lymph node involvement, and stage III refers to tumors with distant metastases. 15
Currently, tumor stage and therapy are the only reliable prognostic parameters for splenic HSA in dogs. Dogs with stage II and III HSA have a shorter median survival time (MST) compared with those with stage I.10,12,17,24 Batschinski et al observed a significant difference in the MST based on therapy, with values of 66 days for surgery alone and 274 days for surgery followed by chemotherapy. Histological parameters of canine splenic HSA with a clear prognostic impact are lacking. Mitotic count (MC) and overall survival have been associated with canine splenic HSA treated with surgery and chemotherapy, 14 and a histologic grading system for canine HSA originating from any site was first proposed in 1996, 16 but this grading system provided inconsistent results and has never been widely applied.14,16
Ki-67 is a nuclear protein widely used for the evaluation of cell proliferation, expressed only in proliferating cells and in all active phases of the cell cycle, including early division. Increased Ki-67 is generally associated with high-grade tumors and a poorer prognosis.20,22,23 Despite its importance in veterinary oncology, only one study assessed the Ki-67 labeling index (Ki-67LI) in 47 canine splenic HSA and found no association with the metastatic status. 20 In contrast, an association was identified between Ki-67LI and tumor stage, with tumor stages II and III showing a higher Ki-67LI when compared with stage I. A longer survival time was associated with a lower Ki-67LI. 20 Nevertheless, a prognostically significant cut-off value for Ki-67LI has not been identified. 20
The aims of this study were as follows:
To compare the assessment of Ki-67 immunohistochemical reactivity using 2 different methods: Ki-67 count (Ki-67C) using an optical grid and Ki-67LI assessed in ≥500 cells;
Verify the prognostic impact of Ki-67 expression in canine splenic HSA;
Define a prognostically relevant Ki-67 cut-off value for splenic HSA.
Two different methods for the assessment of Ki-67 were chosen to compare the results with a previously investigated HSA case series, 20 as well as with data available for other canine tumors.22,23
Materials and Methods
Case Selection
Canine splenic HSA cases were retrospectively collected from the cases of a veterinary oncological referral practice, over a period of 7 years (2015–2022) based on the following inclusion criteria: (1) histologic diagnosis of splenic hemangiosarcoma, (2) treatment with splenectomy, (3) staged at the time of presentation or referral, (4) available formalin-fixed paraffin-embedded tissue, and (5) available tumor-related survival time (from the day of surgery until tumor-related death).
Staging, Treatment, and Follow-up
Staging tests included hematology (complete blood count), biochemistry, coagulation profile (prothrombin time, activated partial thromboplastin clotting time, and fibrinogen), urinalysis, and diagnostic imaging.
The diagnostic tests and imaging modality performed at the time of staging were at the discretion of the clinician. Imaging techniques included abdominal ultrasonography combined with 3-view thoracic radiographs and echocardiography or total body contrast-enhanced computed tomography. Cases subsequently underwent adjuvant treatment with anthracyclines or metronomic chemotherapy-based protocols. Dogs were usually restaged at the time of the fourth or fifth anthracyclines treatment or at the end of the protocol and then every 2 to 3 months if no signs of disease progression were identified earlier. Dogs receiving metronomic chemotherapy were usually restaged every 2 to 3 months. Procedures performed at the time of follow-up visits included hematology, biochemistry, and urinalysis with 3-view thoracic radiographs and abdominal ultrasound or total body computed tomography depending on the managing clinician and owners’ preference. For each dog, data collected included breed, age, sex, date of the diagnosis, date of death, disease-free time, and overall survival. Follow-up in the majority of the cases was based on recheck at the hospital. Follow-up information not available in the medical record was obtained through telephone calls to the referring veterinarians and/or the owners.
Histologic Evaluation
All hematoxylin and eosin-stained slides available were reviewed to select blocks containing neoplastic tissue with the highest cellularity and lowest amount of necrosis, hemorrhage, inflammation, and extramedullary hematopoiesis.
MC was assessed simultaneously by 2 experienced board-certified pathologists at a multiheaded microscope, selecting the areas with the highest cellularity and proliferative activity and by counting 10 consecutive high-power fields, equating to the standard area of 2.37 mm2. 13
Immunohistochemistry
Serial sections of 3 to 4 µm were obtained from selected paraffin blocks, air dried, dewaxed, and rehydrated. Endogenous peroxidase was blocked by immersion in 3% H2O2 in methanol for 30 minutes. Heat-induced antigen retrieval was used (pH 6.0 citrate buffer, in a microwave oven at 750 W for 4 cycles of 5 minutes each).
After cooling, sections were preincubated with blocking solution (10% normal goat serum with phosphate-buffered saline) for 30 minutes. Anti-Ki-67 primary antibody (mouse monoclonal, clone MIB-1, dilution 1:400, Dako, Glostrup, Denmark) was applied and incubated at 4°C overnight.
Sections were then incubated for 30 minutes at room temperature with anti-mouse biotin-conjugated secondary antibody (dilution 1:200, Dako, Glostrup, Denmark). The reaction was amplified by the avidin-biotin method (ABC kit elite; Vector, Burlingame, CA) and visualized by incubating with 3,30-diaminobenzidine for 2 minutes. Harris’ hematoxylin was used as counterstaining. Finally, the sections were dehydrated and cover slipped. Sections of canine small intestine were used as positive control. Negative controls included slides incubated with a nonspecific antibody or the omission of the primary antibody.
Immunohistochemistry Evaluation
Measurements of Ki-67-positive cells (Fig. 1) were performed in areas with the highest proliferation (selected by scanning the entire section at low magnification) using 2 distinct methods, by 2 distinct operators (one for each method):
Ki-67LI was defined as the percentage of Ki-67-positive cells out of at least 500 neoplastic cells for each case, as previously reported. 23 Cells were counted using the manual counting tool of ImageJ 1.48 analysis software on digital pictures taken at 40×. For each case, 5 nonoverlapping, consecutive images were taken in the areas of highest proliferation. The total number of neoplastic cells in the images that were evaluated when the number of cells counted reached 500 was included to avoid subjectivity.
Ki-67C was defined as the average number of positive cells in a 1 cm2 optical grid performed in 5 high-power fields (40×), as previously reported for other canine tumors.22,23 The total area assessed (equating the 5 grids) was 0.3125 mm2.
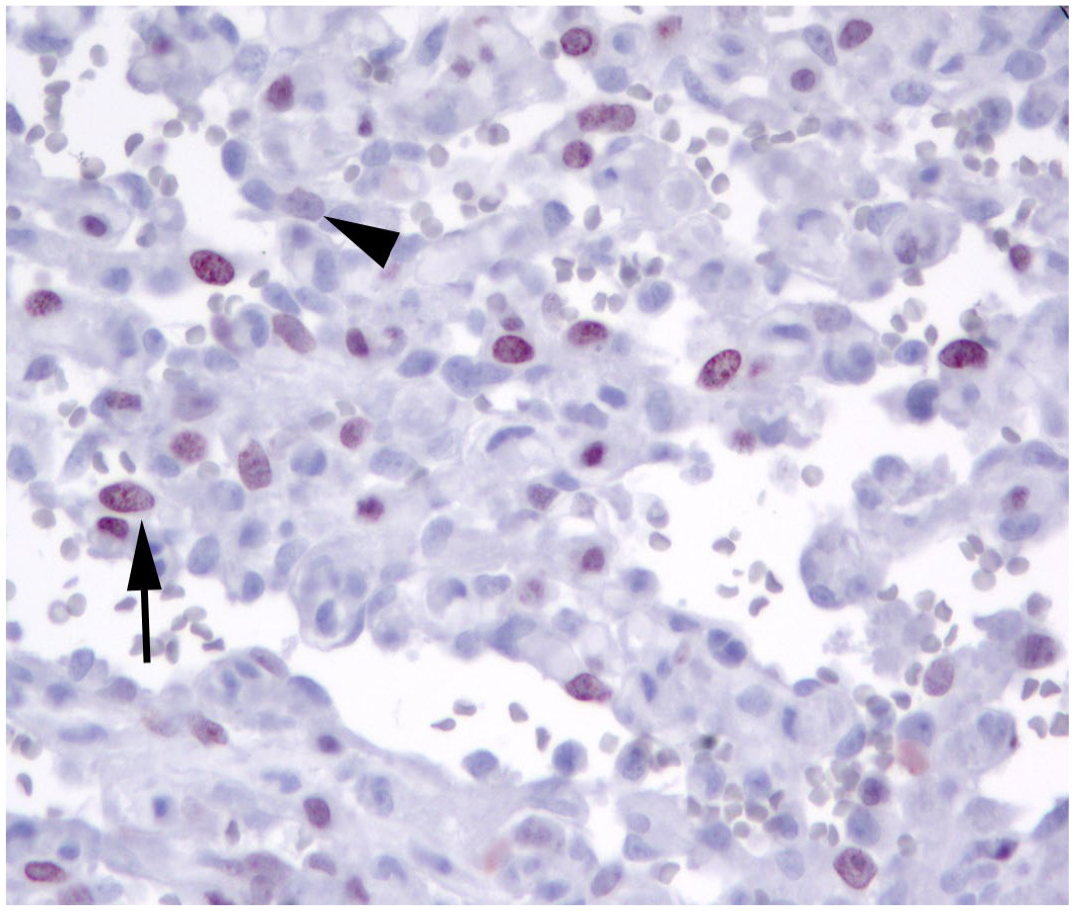
Dog, case 29. Splenic hemangiosarcoma, Ki-67 immunohistochemistry. Neoplastic cells have variable expression of Ki-67. Only cells with clearly visible nuclear labeling were considered positive (arrow), while cells with questionable labeling (arrowhead) were considered negative.
Statistical Analysis
For continuous variables, mean, median, standard deviation (SD), and range were identified. To quantify the linear correlation among continuous variables, Pearson’s correlation coefficient was used. Optimal cut-off values for MC, Ki-67LI, and Ki-67C were determined by receiver-operating characteristic curves. For each variable, the sensitivity-specificity threshold was the value closer to the top-left corner.
Tumor-related survival was defined as the time from the date of diagnosis to death due to HSA. Cases were censored if the dog was alive at the end of the study, had been lost to follow-up, or died due to causes unrelated to HSA. Survival probabilities and MSTs were estimated with the Kaplan-Meier product-limit estimated and were compared among groups by log-rank tests. The Cox proportional hazard model was used for the univariate analysis of the continuous variables (Ki-67LI, Ki-67C, and MC) and for the multivariate analysis; variables were included in the multivariate analysis only if they were significant at univariate analysis. Since the study population included only 3 stage I cases, stage I and II cases were grouped for all analyses including the clinical stage. For each test, differences were considered significant when P ≤ .05. Statistical analysis was performed using R (version 4.1.2 for Windows, Vienna, Austria).
Results
A total of 31 splenic HSAs in 31 dogs were included in the study. Ten dogs were crossbreeds, 6 were retrievers, 5 were boxer dogs, 4 were German shepherd dogs, and 1 dog represented each of the following breeds: Akita Inu, beagle, bulldog, Cocker Spaniel, Siberian husky, and pinscher. Sixteen dogs were female and 15 were male. Ages were available in 25 out of 31 cases and ranged between 6 and 14 years (mean = 10.5, median = 10, SD = 2.64).
At the time of diagnosis, 3 of 31 dogs were stage I (10%), 17 of 31 were stage II (55%), and 11 of 31 were stage III (36%). Twelve out of 31 tumors (39%) were metastatic at the time of diagnosis. Of these, 8 metastasized to the liver (67%), 1 to the regional lymph node (8%), 1 to the retroperitoneum (8%), and 2 dogs had multiorgan metastases to liver, kidneys, lungs, muscle, and/or subcutaneous tissue (17%). Metastases were confirmed histologically.
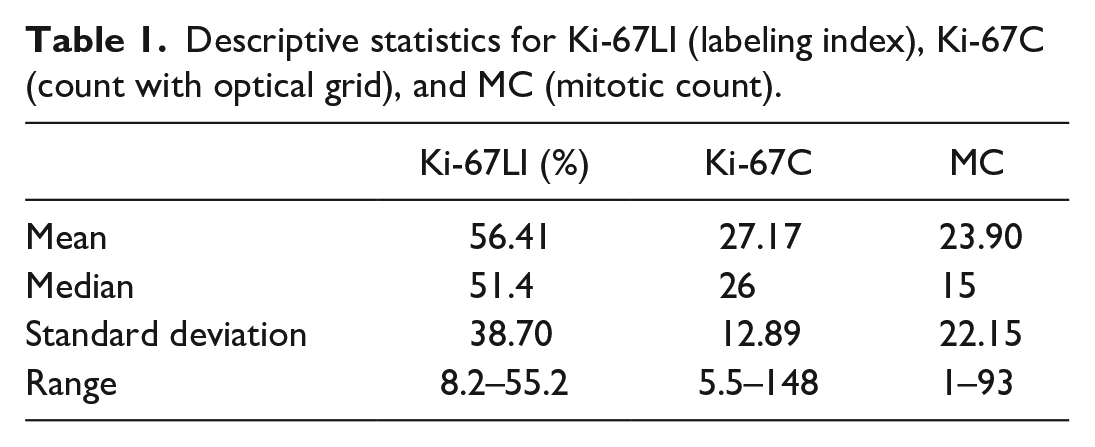
All cases underwent splenectomy, and 21 of 31 received adjuvant chemotherapy (68%). Twenty-one dogs out of 31 died due to tumor-related causes (68%). Seven cases were censored: 3 died for causes unrelated to hemangiosarcoma (10%), 3 were still alive at the end of the study, and 1 was lost to follow-up. MC, Ki-67LI, and Ki-67C descriptive statistics are reported in Table 1.
Descriptive statistics for Ki-67LI (labeling index), Ki-67C (count with optical grid), and MC (mitotic count).
A statistically significant correlation was observed between Ki-67C and Ki-67LI (P = .00344, Pearson’s correlation coefficient = 0.509, 95% confidence interval (CI) = 0.189–0.732), but not between Ki-67LI and MC (P = .121, Pearson’s correlation coefficient = 0.284, 95% CI = 0.0778–0.58) or Ki-67C and MC (P = .066, Pearson’s correlation coefficient = 0.334, 95% CI = 0.0228–0.616).
To determine optimal cut-off value for Ki-67C, Ki-67LI, and MC, analyses of the corresponding receiver-operating characteristics curves were performed comparing the variables with survival at 2, 4, and 6 months; results are reported in Table 2. The highest area under the curve value was obtained at 6-month survival for all the tested variables, and the cut-offs selected based on the best combination of specificity and sensitivity were 9 for MC, 24 for Ki-67C, and 56% for Ki-67LI.
Area under the curve (AUC) assessed by receiver-operating characteristic (ROC) curve analysis.
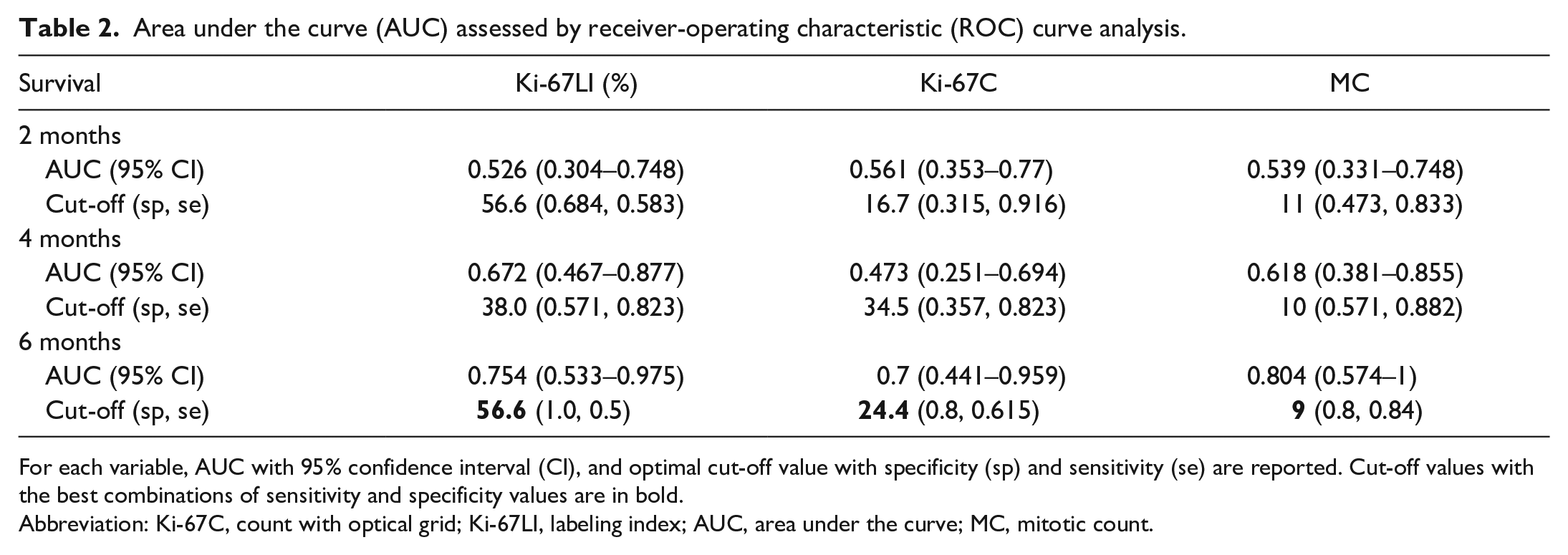
For each variable, AUC with 95% confidence interval (CI), and optimal cut-off value with specificity (sp) and sensitivity (se) are reported. Cut-off values with the best combinations of sensitivity and specificity values are in bold.
Abbreviation: Ki-67C, count with optical grid; Ki-67LI, labeling index; AUC, area under the curve; MC, mitotic count.
These cut-off values were used to assess differences in survival times using Kaplan-Meier survival curves. Cases with Ki-67LI above 56% had significantly shorter survival compared with those that had lower Ki-67LI (P = .00721, log-rank test). Cases with MC above 9 had significantly shorter survival compared with those that had lower MC (P = .00281, log-rank test). Kaplan-Meier curves (Fig. 2) showed no statistically significant relation between Ki-67C and survival (P = .293, log-rank test). Results are reported in Table 3.
Survival analysis of MC (mitotic count), Ki-67C (count with optical grid), Ki-67LI (labeling index), clinical stage, and treatment (log-rank test).
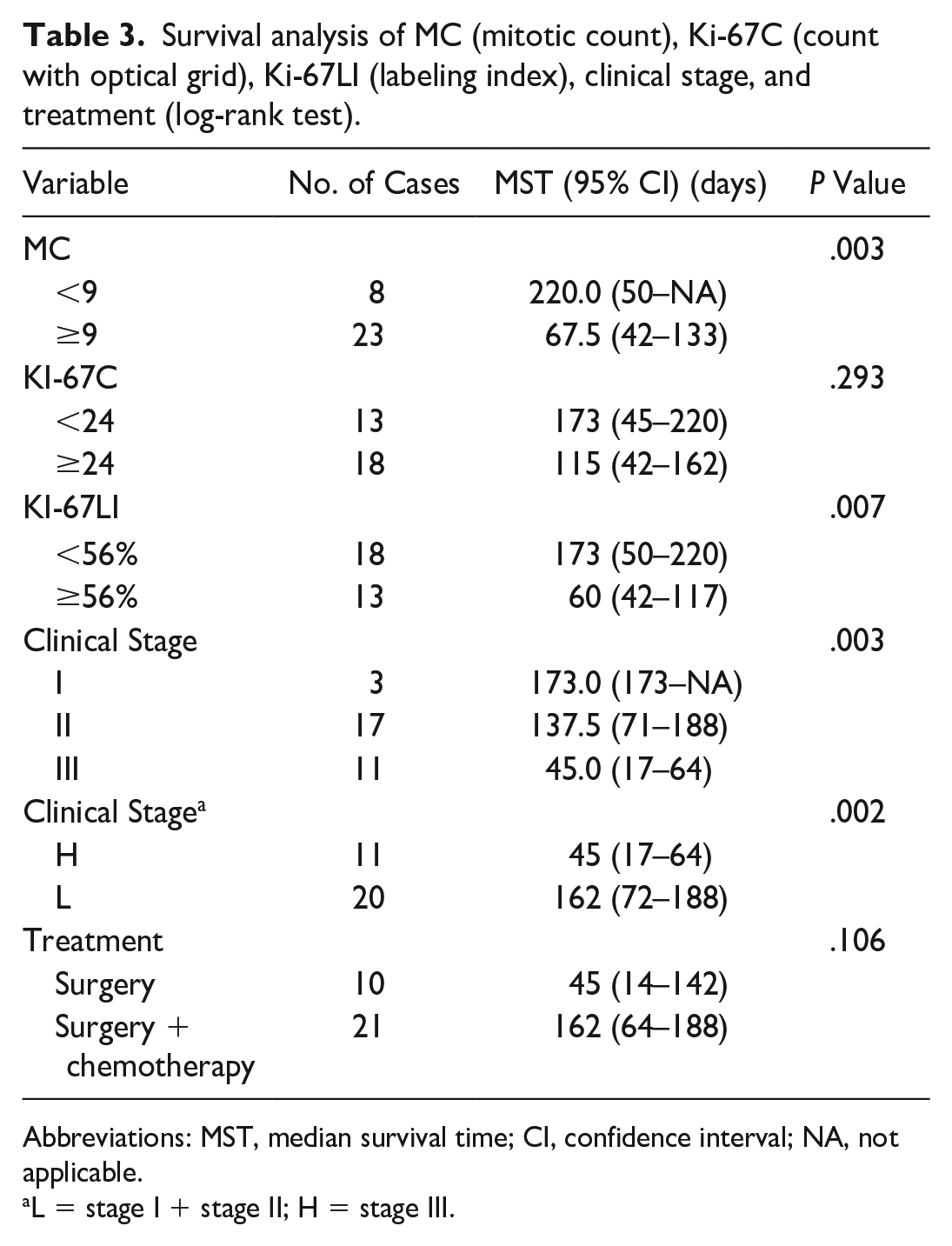
Abbreviations: MST, median survival time; CI, confidence interval; NA, not applicable.
L = stage I + stage II; H = stage III.
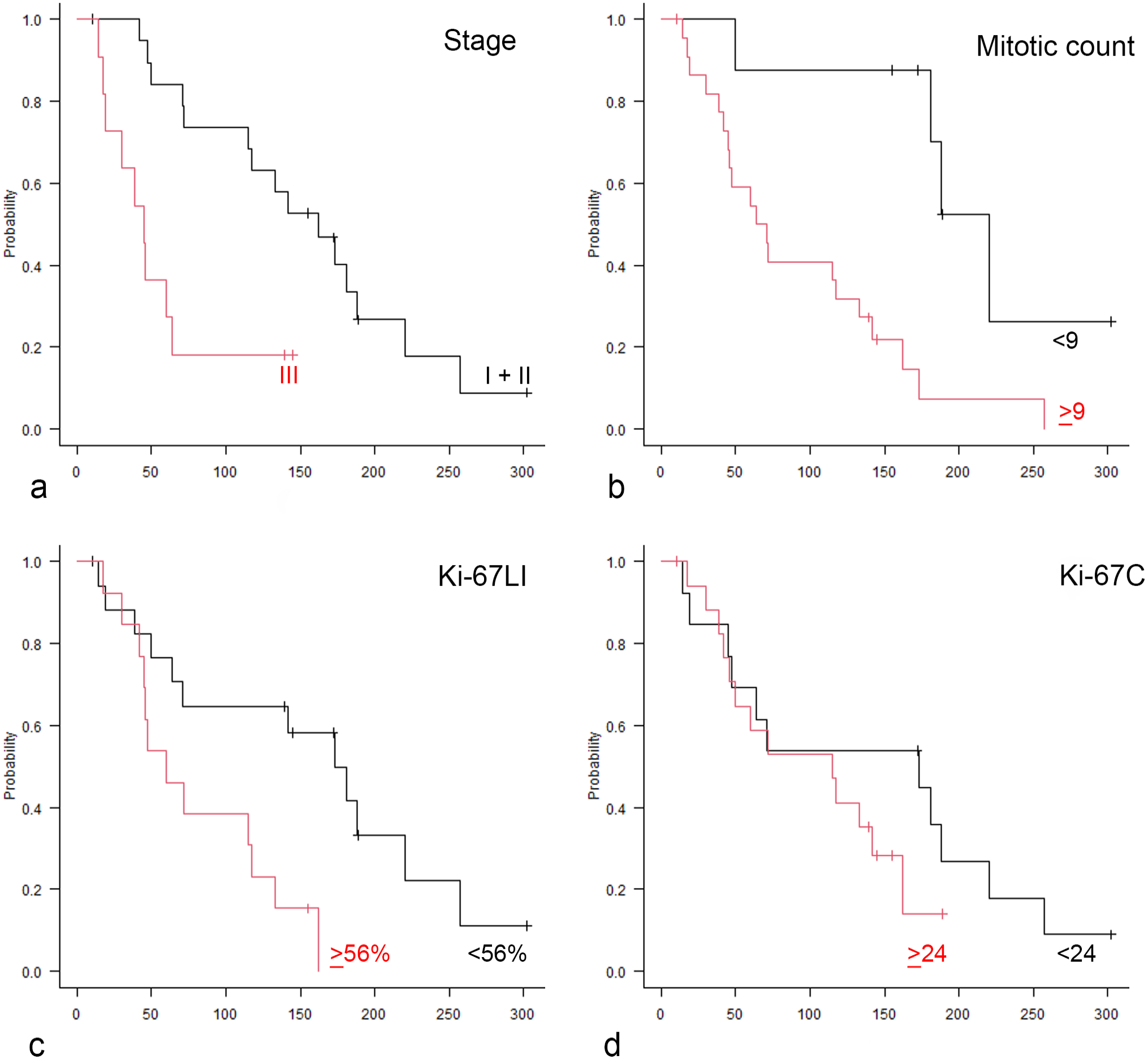
Kaplan-Meier curves comparing the survival probability of dogs with splenic hemangiosarcoma. X-axis represents overall survival expressed in days. (a) Red line, high-stage cases (stage III); black line, low-stage cases (stage I and II). (b) Red line, cases with mitotic counts higher than or equal to 9; black line, cases with mitotic counts lower than 9. (c) Red line, cases with Ki-67 labeling index (Ki-67LI) higher than or equal to 56%; black line, Ki-67LI lower than 56%. (d) Red line, cases with Ki-67 grid count (Ki-67C) higher than or equal to 24; black line, Ki-67C lower than 24.
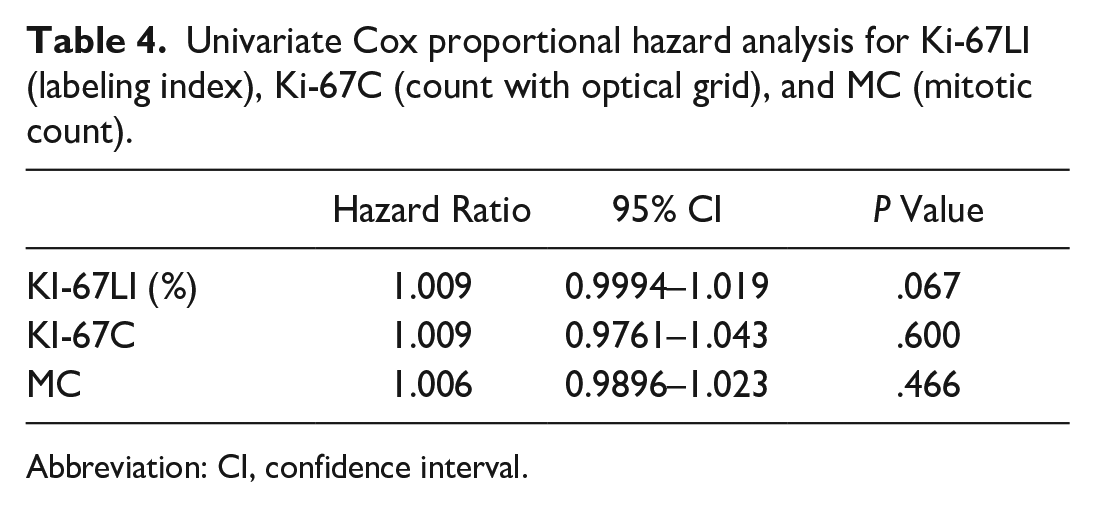
In addition to receiver-operating characteristic curve analyses, Ki-67C, Ki-67LI, and MC were analyzed as continuous variables in univariate Cox proportional hazards models, which did not show statistically significant relations with the survival times (Table 4).
Univariate Cox proportional hazard analysis for Ki-67LI (labeling index), Ki-67C (count with optical grid), and MC (mitotic count).
Abbreviation: CI, confidence interval.
The clinical variables, stage and type of treatment, were included in the survival analysis. Stage I and stage II cases were grouped together (low stage) and compared with stage III (high stage) because of the low number of stage I HSAs. Dogs with high-stage tumors had a significantly shorter MST than dogs with low-stage tumors (MST = 45 days, 95% CI = 17–64 and 162 days, 95% CI = 72–188, respectively). Cases undergoing surgery and chemotherapy had a longer MST (145 days, 95% CI = 64–181) compared with dogs that were treated with surgery alone (MST = 45 days, 95% CI = 14–142), excluding dogs that were still alive or died for causes unrelated to the disease.
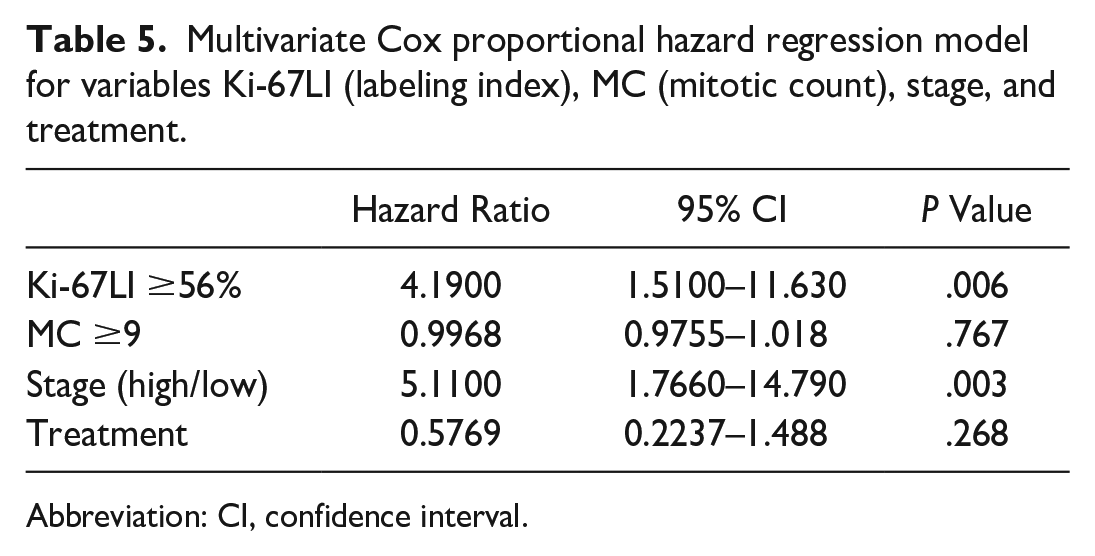
To analyze the impact of Ki-67LI on survival, a multivariate analysis was performed using the Cox proportional hazard regression model that included variables that were significant at univariate analysis: Ki-67LI, MC, clinical stage (low vs high), and treatment type (surgery vs surgery and chemotherapy). Of these, Ki-67LI and clinical stage remained significant on multivariate analysis. Specifically, Ki-67LI at its optimal cut-off value represented an independent prognostic variable (hazard ratio = 4.1380, 95% CI = 1.49–11.48, P = .006351). In addition, high-stage (stage III) cases had a larger instantaneous death risk than low-stage (stage I/II) cases, and clinical stage also represented an independent prognostic factor (hazard ratio = 5.38, 95% CI = 1.75–16.53, P = .003). By contrast, treatment type and MC were not statistically significant in the multivariate analysis. Results are summarized in Table 5.
Multivariate Cox proportional hazard regression model for variables Ki-67LI (labeling index), MC (mitotic count), stage, and treatment.
Abbreviation: CI, confidence interval.
Discussion
Canine HSA is one of the most aggressive cancers in dogs,3,24,27 and tumor stage and therapy are the only reliable prognostic parameters reported thus far,10,12,17,24 while histological features with a clear prognostic impact are lacking.
A histologic grading system for canine HSA (originating from any site) was first proposed in 1996. 16 This grading system takes into account the differentiation of tumor cells, cellular pleomorphism, percentage of necrosis, and MC (number/10 high-powered fields). 14 The prognostic significance of this grading scheme was suggested by univariate but not by multivariate analysis, and therefore, it has not been widely applied. 2 To the best of our knowledge, only one study in the literature evaluated Ki-67 expression in canine splenic HSA, showing a correlation of Ki-67LI with overall survival; however, prognostically relevant threshold values for Ki-67 were not provided. 20 Therefore, the main aims of our study were to evaluate the prognostic impact of Ki-67 expression assessed by 2 different methods and to define a Ki-67 cut-off that could be of prognostic relevance.
Metastases occurred in approximately one third of cases, with the liver being the most common site of metastases, as reported in the literature.5,9 In our study, the stage at diagnosis had a statistically significant correlation with tumor-related survival, representing one of the most important prognostic factors, in agreement with the literature.3,11,14,24,27
All cases in this study underwent splenectomy and 21 received adjuvant chemotherapy, including different chemotherapy protocols. Taking all cases into consideration, therapy was not associated with tumor-related survival. Nevertheless, as 2 out of 3 stage I cases were still alive at the end of the study, one of which was a small tumor treated with surgery alone, we hypothesize these cases may have produced a confounding effect. Indeed, by removing them from the analysis, the 2 treatment modalities were associated with survival. The cases with stage II or III HSA that underwent surgery followed by chemotherapy had a longer survival time than those treated with surgery alone, as demonstrated in previous studies.8,26
For the first time in this study, 2 different methods of assessment of Ki-67-positive cells were performed and compared. As expected, there was a statistically significant correlation between Ki-67C and Ki-67LI, indicating a linear relationship between these 2 continuous variables, but a correlation between MC and Ki-67LI or Ki-67C was not found.
The prognostic value of MC, Ki-67LI, and Ki-67C was assessed with the Kaplan-Meier survival curve and log-rank test using the cut-off derived from the receiver-operating characteristic curve analysis. This approach allowed demonstration of an association of tumor-related survival with Ki-67LI and MC using the cut-off of 56% and 9, respectively. Ki-67LI was also confirmed as an independent prognostic parameter on multivariate analysis, together with treatment and clinical stage. These results further support the previously reported prognostic impact of Ki-67 expression in canine splenic HSA 20 and provide a cut-off value of potential use in everyday diagnostic activity.
In summary, in this study, the prognostic role of Ki-67LI was demonstrated for canine splenic HSAs, providing a counting technique and a statistically valid cut-off value of practical use in a diagnostic setting. Further studies on larger caseloads may be useful to confirm and/or refine these results and to investigate the prognostic value of Ki-67.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
