Abstract
Cutaneous and subcutaneous mast cell tumors (MCTs) are common canine neoplasms characterized by variable biological behavior. Tumor-associated macrophages (TAMs) and tumor-infiltrating lymphocytes (TILs) can be effective prognostic markers in numerous human neoplasms and are increasingly investigated in dogs. The aim of this study was to characterize immune cells in canine MCTs and their relationship with histological location (cutaneous, subcutaneous) and histologic nodal metastatic status (HN0-3). Thirty-eight MCTs (26 cutaneous, 12 subcutaneous) from 33 dogs with known sentinel lymph node (SLN) metastatic status were immunolabeled for Iba1 (macrophages), CD20 (B cells), CD3 (T cells), and Foxp3 (regulatory T cells). Semiquantitative scoring of interstitial and perivascular CD3+, CD20+, and Foxp3+ cells and morphological evaluation of Iba1+ cells were performed. For each marker, the percent immunopositive area was evaluated by image analysis. All MCTs were diffusely infiltrated by Iba1+ cells and variably infiltrated by CD20+, CD3+, and rare Foxp3+ cells. Stellate/spindle Iba1+ cells were associated with HN2 and HN3 SLNs. Perivascular Foxp3+ cells, CD3+ cells, and percent CD3+ areas were increased in subcutaneous MCTs. Interstitial CD3+ cells were increased in cutaneous MCTs with HN0 SLNs. No differences in CD20+ cells were identified between cutaneous and subcutaneous MCTs and among SLN classes. MCTs were markedly infiltrated by TAMs and variably infiltrated by TILs. Stellate/spindle morphology of TAMs associated with HN2 and HN3 SLNs is suggestive of a pro-tumoral (M2) phenotype. Cutaneous and subcutaneous MCTs have different tumor-immune microenvironments, and T-cell infiltration might contribute to prevention of nodal metastatic spread of cutaneous MCTs.
Keywords
Cutaneous and subcutaneous mast cell tumors (MCTs) are common neoplasms in dogs. 2 Numerous clinical and pathologic features have been recognized as having prognostic utility in canine MCT, including tumor site and size, presence of satellite nodules, occurrence of paraneoplastic signs (eg, Darier’s sign, vomiting, gastro-intestinal hemorrhages), clinical stage, lymph node (LN) and visceral organ metastatic status, histological grade, mitotic count, proliferation assessed through immunohistochemistry or histochemistry (Ki67 and AgNOR, respectively), and immunohistochemical c-KIT pattern.2,6,14,23,27,38,40,46,47,49 Despite the plethora of prognostic parameters available, the accurate prognostication of canine cutaneous and subcutaneous MCTs still represents a significant challenge.2,40
Based on mast cells burden, MCT-draining LNs are histologically classified as non-metastatic HN0, pre-metastatic HN1, early metastatic HN2, and overtly metastatic HN3. 48 Sentinel lymph nodes (SLNs) are defined as the first nodes receiving lymphatic drainage from a tumor and are the primary site of metastasis of neoplasms spreading by the lymphatic circulation. 22 It has been reported that in dogs with MCTs, from 28% to 63% of SLNs do not correspond to regional LNs.9,15 The identification of SLNs is, therefore, paramount to detect metastatic disease that would be otherwise missed by sampling a non-draining lymphocenter.9,15,51
Over the last 20 years, the complex interactions between tumors and the immune system have been extensively investigated in humans, leading to important advances in prognostic assessment and treatment of several malignancies.19,24 Immune system cells continuously patrol the body to identify and eliminate neoplastic cells that, in turn, develop several mechanisms to suppress the immune response favoring the formation of a pro-tumoral microenvironment. This complex interaction between tumors and the immune system, referred to as immunoediting, can result in either the elimination of the tumor or the uncontrolled proliferation of neoplastic cells after failure of cytotoxic immune mechanisms.21,23,25
This process is carried out by different populations of tumor-infiltrating immune cells that exert antitumoral or pro-tumoral activities depending on the milieu of the tumor microenvironment. Among them are tumor-infiltrating lymphocytes (TILs), which represent a heterogeneous group of cells comprising CD8+ T lymphocytes with direct cytotoxic antineoplastic activity, Foxp3+ regulatory T cells (Tregs) capable of immune-suppressive pro-tumoral response, and interleukin (IL)-10-producing regulatory B cells with a still undetermined role as they seem associated with a positive prognosis in certain malignancies and a negative prognosis in others.21,35
Tumor-associated macrophages (TAMs) are another intra-tumoral population that, under the driving effects of local cytokines in the tumor microenvironment, can acquire different phenotypes. Classically activated M1 macrophages display a strong antitumoral response, while alternatively activated M2 macrophages promote tumoral development and growth, stimulate angiogenesis, and suppress T-cell antitumoral activities.5,56,57
Although TILs and TAMs have been investigated in several canine malignancies, such as mammary gland tumors,3,4,20,32,33,37 prostatic carcinoma, 26 colorectal carcinoma, 50 pulmonary adenocarcinoma, 34 nasal carcinoma, 42 oral squamous cell carcinoma, 34 soft tissue sarcomas, 10 intestinal lymphoma,17,43 and melanocytic tumors,29 –31,54 often with promising prognostic meaning, very few data exist in the literature regarding canine MCTs.7,34 In one study, only rare Foxp3+ cells were reported in canine MCTs, while another study found higher Iba1+ areas in high-grade cutaneous MCTs compared with low-grade cutaneous MCTs, with no differences in T-cell infiltration.7,34 Given the gap of knowledge in canine cutaneous and subcutaneous MCTs, and the promising results obtained with other canine neoplasms, the aim of this study was to investigate the presence of infiltrating T and B cells, Tregs, and TAMs within canine MCTs and to evaluate their potential association with tumor histological location (cutaneous vs subcutaneous) and SLN metastatic status.
Materials and Methods
Case Selection
Cases of canine cutaneous and subcutaneous MCTs diagnosed from January 2017 to June 2020 were retrieved from the database of the Pathology Service of the Veterinary Teaching Hospital of Lodi (University of Milan). All selected tumors were first occurrences and treatment naive, and for each case distant metastases were not detected through cytological examination of blood, liver, and spleen. 39 All tumors were excised with a curative intent surgery (2–3 cm or proportional lateral margins, and at least one deep fascial plane). For each tumor, SNLs were detected through lymphoscintigraphy prior to surgical guided excision according to previously published criteria. 9 Dogs with multiple concurrent MCTs at clinical presentation were included only when each tumor was associated with a distinct draining lymphocenter. Ulcerated MCTs and muco-cutaneous and mucosal MCTs were excluded from the study to avoid potential confounding effects resulting from ulcer-driven inflammation or from the peculiar mucosal immune response. For each case, data on breed, sex, age of the patient, anatomical site of the tumor, and anatomical site and metastatic status of SLNs were recorded (Supplemental Table S1).
Histopathology
For the present retrospective investigation, paraffin blocks of the tumors and SLNs of cases included in the study were retrieved from the archive of the pathology unit of the Veterinary Teaching Hospital. Excised surgical samples had been fixed in 10% neutral-buffered formalin. For each tumor, blocks of all tangential margins of the surgical specimen, of a complete longitudinal section of the mass, and of SLNs that had been longitudinally cut along the median sagittal plane were available.
Four-micrometer-thick sections were stained with hematoxylin and eosin and the histological location (cutaneous/subcutaneous) of the tumors were assessed via light microscopy (Fig. 1a, b). MCTs were diagnosed as cutaneous when occupying the superficial to deep dermis, growing among, distorting, and often effacing cutaneous adnexa with only limited extension into the subcutis. MCTs were diagnosed as subcutaneous when the bulk of the tumor was located in the subcutis, with only limited involvement of the deep dermis and no involvement of cutaneous adnexa. 40 Cutaneous MCTs were graded according to the 3-tier (grades I, II, III) and 2-tier (low grade/high grade) grading systems.14,27 Subcutaneous MCTs were classified as circumscribed, infiltrative, or combined (mixed) according to the criteria indicated by Thompson et al. 40 SLN sections were stained with Giemsa and classified under light microscopy according to the criteria indicated by Weishaar et al. 48
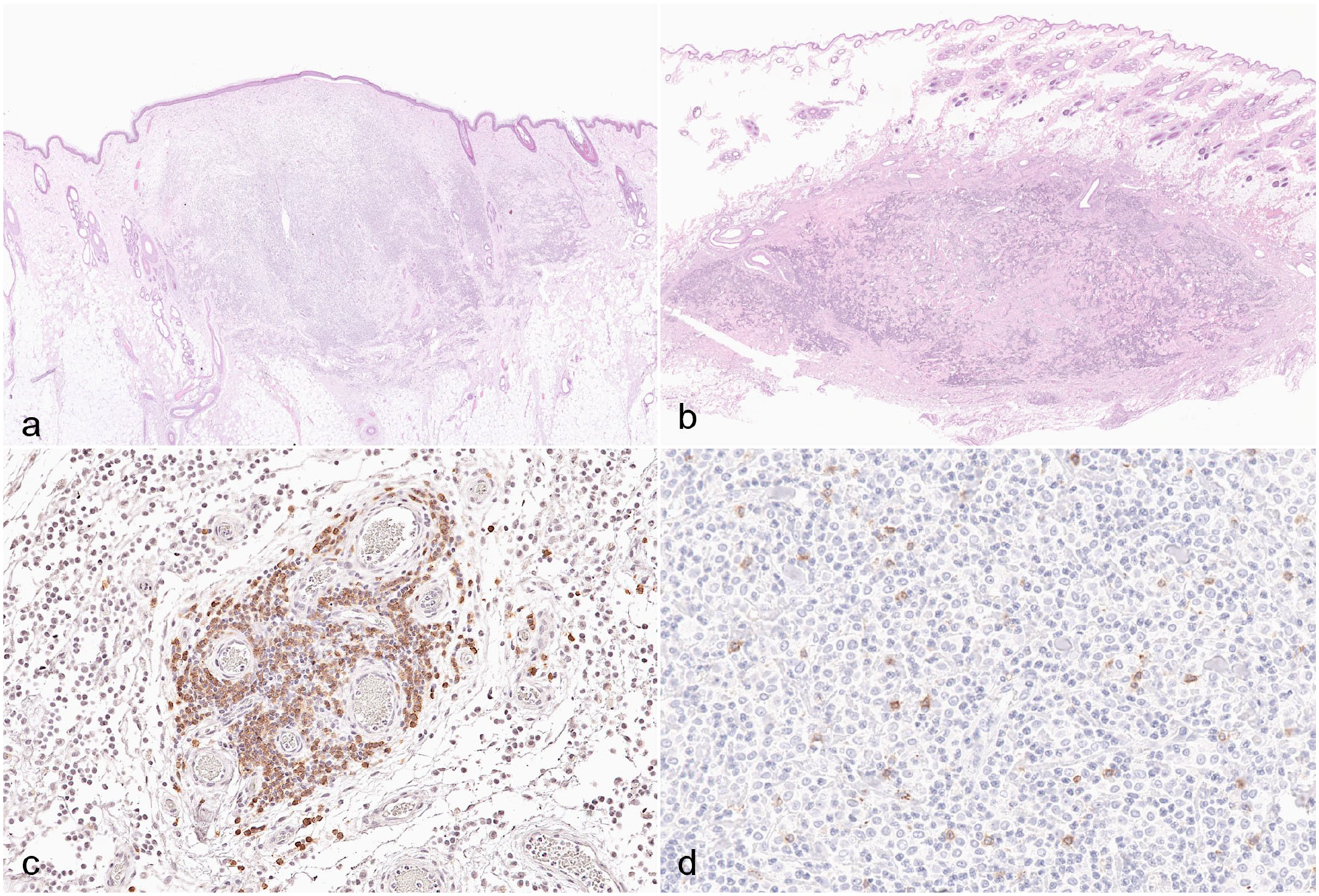
Mast cell tumors (MCTs), dog. (a) Cutaneous MCT. Neoplastic mast cells expand the superficial and deep dermis, replacing the adnexa and slightly raising the overlying epidermis. Hematoxylin and eosin (HE). (b) Subcutaneous MCT. Neoplastic cells expand the subcutaneous adipose tissue without involving the cutaneous adnexa. HE. (c) Perivascular immunolabeling pattern characterized by aggregates of lymphocytes arranged around blood vessels. CD20 immunohistochemistry (IHC). (d) Interstitial immunolabeling pattern characterized by individualized positive lymphocytes among neoplastic mast cells, eosinophils, and scant collagen fibers. CD3 IHC.
To perform statistical analyses, MCTs were classified into different classes according to histological location (cutaneous or subcutaneous) and SLN metastatic status according to the Weishaar classification. 48 When more than 1 SLN was examined for a single MCT, the SLN with the highest Weishaar class was considered as the SLN metastatic status.
Immunohistochemistry
Serial, 4-µm-thick sections of tumors underwent deparaffinization and heat-induced epitope retrieval in a water bath for 30 minutes at 100°C (Dewax and HIER Buffer H, Thermo Scientific Lab Vision, cat. No. TA-999-DHBH). The slides were rinsed in phosphate-buffered saline and placed in an autostainer (Lab Vision Autostainer 480S-2D Thermo Fisher Scientific) after application of PapPen (Liquid Daido Sangyo Co. Ltd.). Endogenous peroxidase activity was blocked by incubating sections with 3% hydrogen peroxide for 10 minutes. Slides were rinsed, incubated with phosphate-buffered saline containing 10% normal goat serum (CD3, CD20, and Iba1) or normal rabbit serum (Foxp3) for 30 minutes at room temperature to prevent nonspecific background and then incubated for 1 hour at room temperature with primary antibodies (Table 1). Sections were subsequently rinsed with phosphate-buffered saline and incubated with a biotinylated secondary antibody (goat anti-rabbit, Vector Laboratories, USA, cat. No. BA-1000 for CD3, CD20, and Iba1 and rabbit anti-rat, Vector Laboratories, USA, cat. No. BA-4001 for Foxp3) and labeled through avidin-biotin-peroxidase procedure (VECTASTAIN Elite ABC-Peroxidase Kit Standard, Vector Laboratories, USA, cat. No.PK-6100). The immunoreaction was visualized with 3,3′-diaminobenzidine substrate (DAB, Peroxidase DAB Substrate Kit, Vector Laboratories, USA, cat. No. SK-4105). Sections were counterstained with Mayer’s hematoxylin, dehydrated in a graded alcohol series, and coverslipped with resinous mounting medium. Adequate positive controls (canine lymph nodes) and negative controls (omission of primary antibodies) were included in each immunolabeling assay.
Primary antibodies used for immunohistochemistry.

All slides were digitalized in 40× high-resolution mode (0.23 µm × pixel) through the NanoZoomer-XR C12000 Digital slide scanner (Hamamatsu, Shizuoka, Japan) at the Unitech Nolimits facility (UNIMI, Milan). Digital slides were visualized using the NDP.view2 Viewing software (Hamamatsu, U12388-01).
CD3, CD20, and Foxp3 Evaluations
CD3, CD20, and Foxp3 intra-tumoral positive cells were evaluated according to a newly designed semiquantitative scoring system (Table 2), considering 2 distinct immunohistochemical patterns: perivascular aggregates and interstitial infiltrates, that is, lymphocytes infiltrating among neoplastic cells (Fig. 1c, d). This scoring system was developed to classify the evaluated cases according to similar perivascular and/or interstitial infiltrates. Perivascular aggregates were evaluated considering their number and diameter. For the number, perivascular aggregates were manually counted within the tumor, and the number was divided by the tumor area measured by outlining MCTs borders on NDP.view2 software. For the diameter, the maximum diameter of each perivascular aggregate was manually measured on NDP.view2 software, and the mean maximum diameter was obtained by dividing the sum of the maximum diameters by the number of perivascular aggregates. Interstitial infiltrates were evaluated considering the extent of the infiltration (percent of tumor with immune cell infiltration estimated via light microscopy) and the mean number of cells per field at 20× (each field corresponding to 0.612 mm2), obtained after counting the positive cells in 10, 20× hot-spot fields (ie, fields with the highest density of positive cells) and dividing the total number of cells by 10. The perivascular score (number score × diameter score) and the interstitial score (extent score × number of cells score) were calculated. The perivascular score and the interstitial score were then summed, obtaining the total score for each tumor.
Semiquantitative score system for CD3+, CD20+, and Foxp3+ cells evaluation.
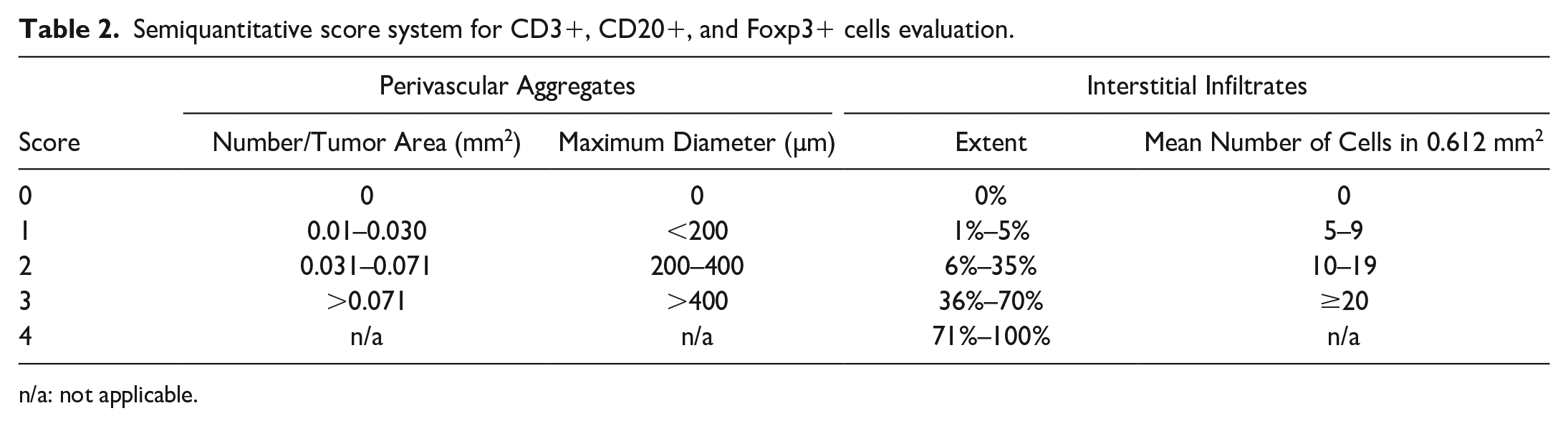
n/a: not applicable.
Iba1 Evaluation
Since all tumors had marked Iba1+ cell infiltration without relevant differences among cases, semiquantitative scoring was not performed. For each tumor, the prevalent morphology of Iba1+ cells was assessed in all the available tumor tissue, and tumors were classified as tumors with prevalent round Iba1+ cells, tumors with prevalent stellate/spindle Iba1+ cells, or as tumors with mixed morphologies of Iba1+ cells (absence of a predominant cell morphology). A prevalent morphology was considered when at least 80% of the macrophages had a round or spindle/stellate morphology.
Digital Image Analysis
For each marker (CD3, CD20, Foxp3, and Iba1), 6 hot-spot fields (ie, fields with the highest density of positive cells) at 40× magnification (each field corresponding to 0.153 mm2) were selected from each digital slide. All selected fields were evaluated using the ImageJ analysis software (http://rsb.info.nih.gov/ij/). 36 Each acquired image was split into 3 monochrome images using the “split channels” function. The channel with the highest contrast between DAB signal and background (blue channel) was selected, and a threshold value avoiding non-specific signals was selected and applied to each hot-spot field; the percentage of positive area per field was obtained (Supplemental Figure S1).
Statistical Analysis
Statistical analyses were performed to investigate possible differences in TILs and TAMs in MCTs according to tumor histological location and SLN metastatic status. Two different analyses were carried out. The first analysis compared non-metastatic and pre-metastatic MCTs (MCTs with HN0 and HN1 SLNs) with early and overtly metastatic MCTs (MCTs with HN2 and HN3 SLNs), the second analysis compared non-metastatic MCTs (MCTs with HN0 SLNs) with pre-metastatic, early, and overtly metastatic MCTs (MCTs with HN1, HN2, and HN3 SLNs). The analyses were performed considering cutaneous and subcutaneous MCTs together and then separately. A statistical analysis between non-metastatic (HN0) subcutaneous MCTs and pre-metastatic, early, and overtly metastatic (HN1, HN2, and HN3) subcutaneous MCTs was not performed since only 1 case of HN0 subcutaneous MCT was present in our data set. Data were analyzed using Graph Pad Prism version 9.0 (GraphPad Software, San Diego, CA, USA). Comparisons between the groups were performed using Mann-Whitney U test. Possible associations between macrophage morphology and MCT location and SLN metastatic status were evaluated using Fisher’s exact test. P-values <.05 were considered statistically significant.
Results
Cases
A total of 61 dogs with MCT were retrieved from the database of the Pathology Service, but only 33 dogs met the inclusion criteria and were included in the study. Dog breeds represented in the study included 8 Labrador retrievers (24%), 6 crossbreeds (18%), 3 English setters (9%), 2 pit bulls (6%), 2 golden retrievers (6%), 2 pugs (6%), 2 boxers (3%), and 1 (3%) of the following breeds: Italian Bracco, Dogo Argentino, Yorkshire terrier, Chihuahua, Dachshund, Bernese Mountain dog, bull terrier, and American Staffordshire. Fourteen dogs were spayed females, 1 dog was an intact female, 6 dogs were castrated males, and 12 dogs were intact males.
A total of 38 MCTs (26 cutaneous, 12 subcutaneous) and 66 SLNs from the 33 selected dogs were retrieved. Three dogs had multiple MCTs at clinical presentation, with distinct lymphocenters. All cutaneous MCTs were classified as grade II and low grade. One subcutaneous MCT was classified as well circumscribed, 6 as infiltrative, and 5 as combined. Twenty SLNs were classified as HN0, 24 as HN1, 18 as HN2, and 4 as HN3. MCTs were further classified according to the highest SLN metastatic status as 7 HN0 MCTs, 16 HN1 MCTs, 12 HN2 MCTs, and 3 HN3 MCTs.
Immunohistochemistry
Results of the semiquantitative (interstitial, perivascular, and total scores) and digital image analysis evaluation of TAMs and TILs according to MCT location and SLN metastatic status are summarized in Tables 3–5. Raw data of the analysis can be found in Supplemental Tables S2–S5.
Results of interstitial, perivascular, total score, and percent of positive area of CD3+, CD20+, Foxp3+, and Iba1+ cells in all (cutaneous and subcutaneous) MCTs.
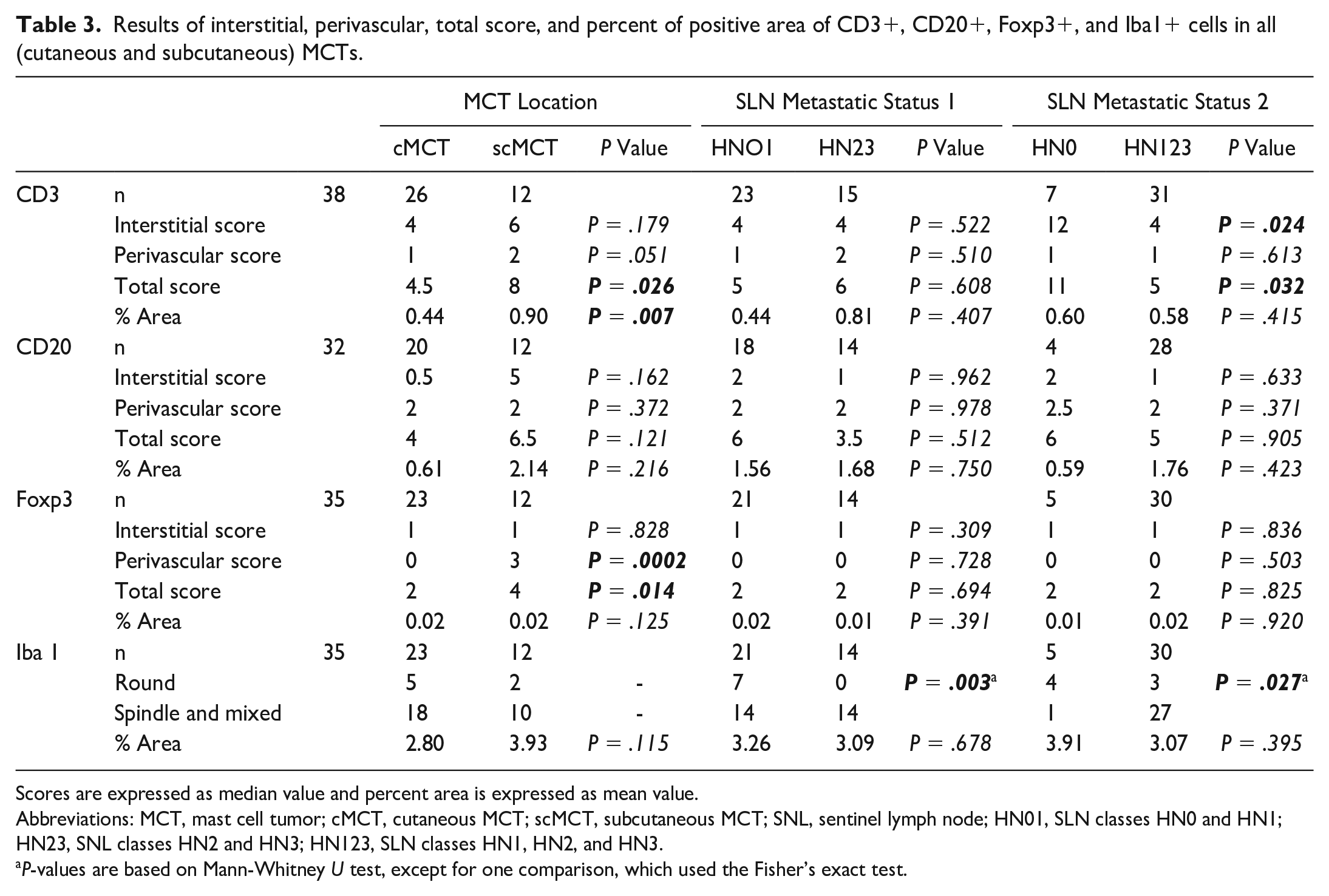
Scores are expressed as median value and percent area is expressed as mean value.
Abbreviations: MCT, mast cell tumor; cMCT, cutaneous MCT; scMCT, subcutaneous MCT; SNL, sentinel lymph node; HN01, SLN classes HN0 and HN1; HN23, SNL classes HN2 and HN3; HN123, SLN classes HN1, HN2, and HN3.
P-values are based on Mann-Whitney U test, except for one comparison, which used the Fisher’s exact test.
Results of interstitial; perivascular; total score; and percent of positive area of CD3+, CD20+, Foxp3+, and Iba1 + cells in cutaneous mast cell tumors.
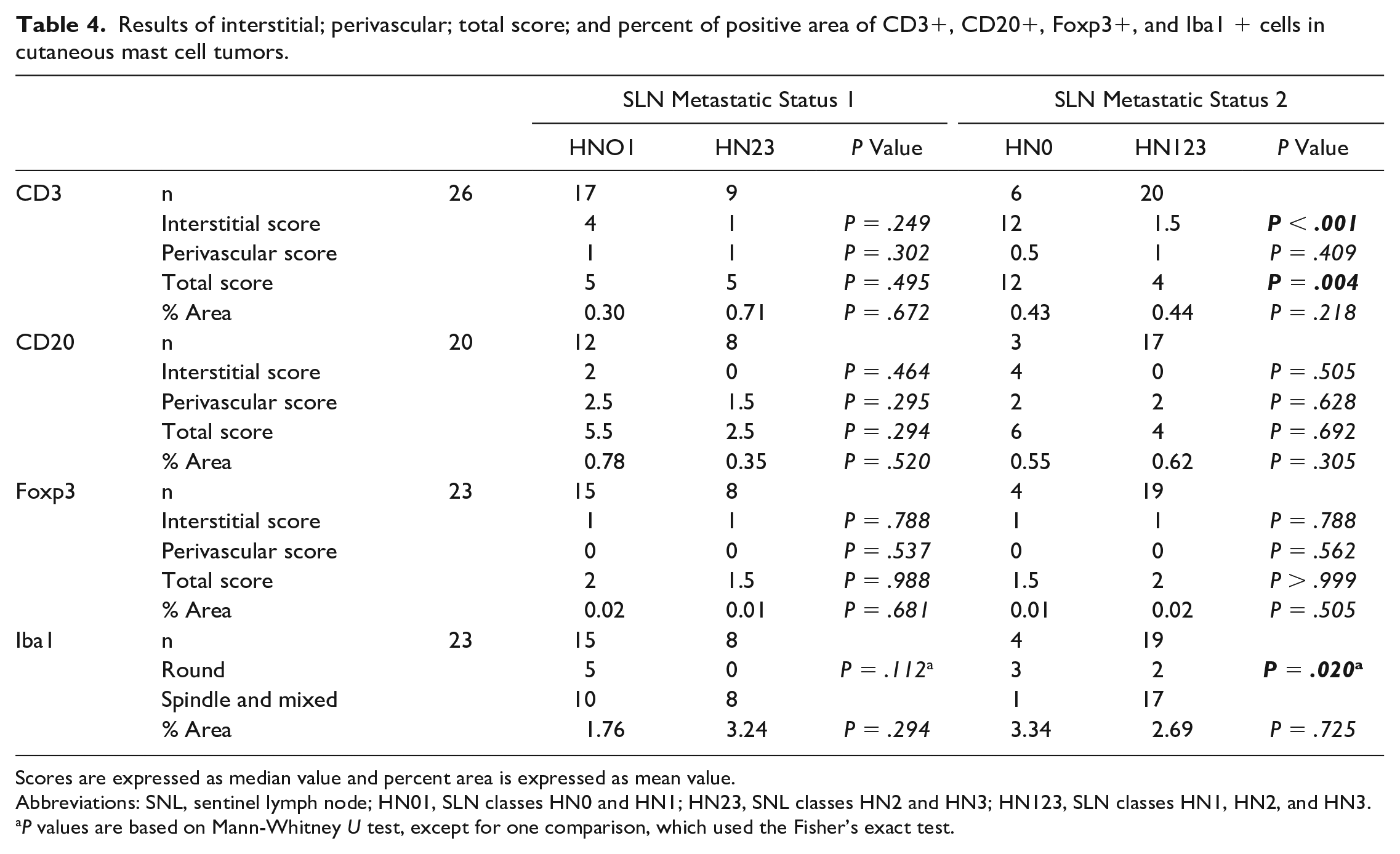
Scores are expressed as median value and percent area is expressed as mean value.
Abbreviations: SNL, sentinel lymph node; HN01, SLN classes HN0 and HN1; HN23, SNL classes HN2 and HN3; HN123, SLN classes HN1, HN2, and HN3.
P values are based on Mann-Whitney U test, except for one comparison, which used the Fisher’s exact test.
Results of interstitial; perivascular; total score; and percent of positive area of CD3+, CD20+, Foxp3+, and Iba1+ cells in subcutaneous mast cell tumors.
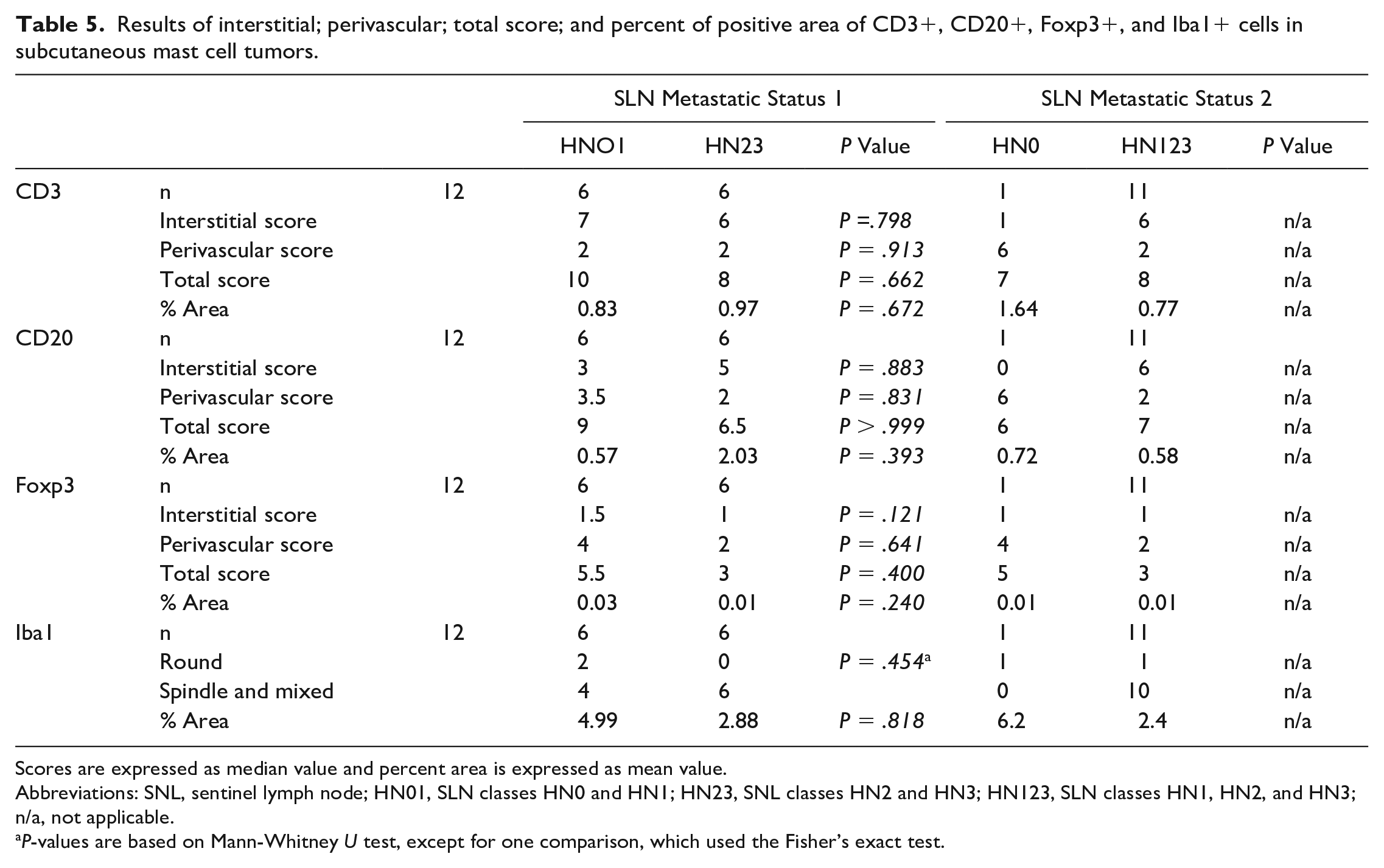
Scores are expressed as median value and percent area is expressed as mean value.
Abbreviations: SNL, sentinel lymph node; HN01, SLN classes HN0 and HN1; HN23, SNL classes HN2 and HN3; HN123, SLN classes HN1, HN2, and HN3; n/a, not applicable.
P-values are based on Mann-Whitney U test, except for one comparison, which used the Fisher’s exact test.
MCTs (both cutaneous and subcutaneous) were diffusely infiltrated by numerous Iba1+ cells, followed by CD20+ cells, CD3+ cells, and only rare Foxp3+ cells. CD20+ cells were the predominant perivascular population with few positive cells in the interstitium, while CD3+ cells were less numerous than CD20+ cells in perivascular aggregates and were the predominant interstitial TIL population (Fig. 2a–d).
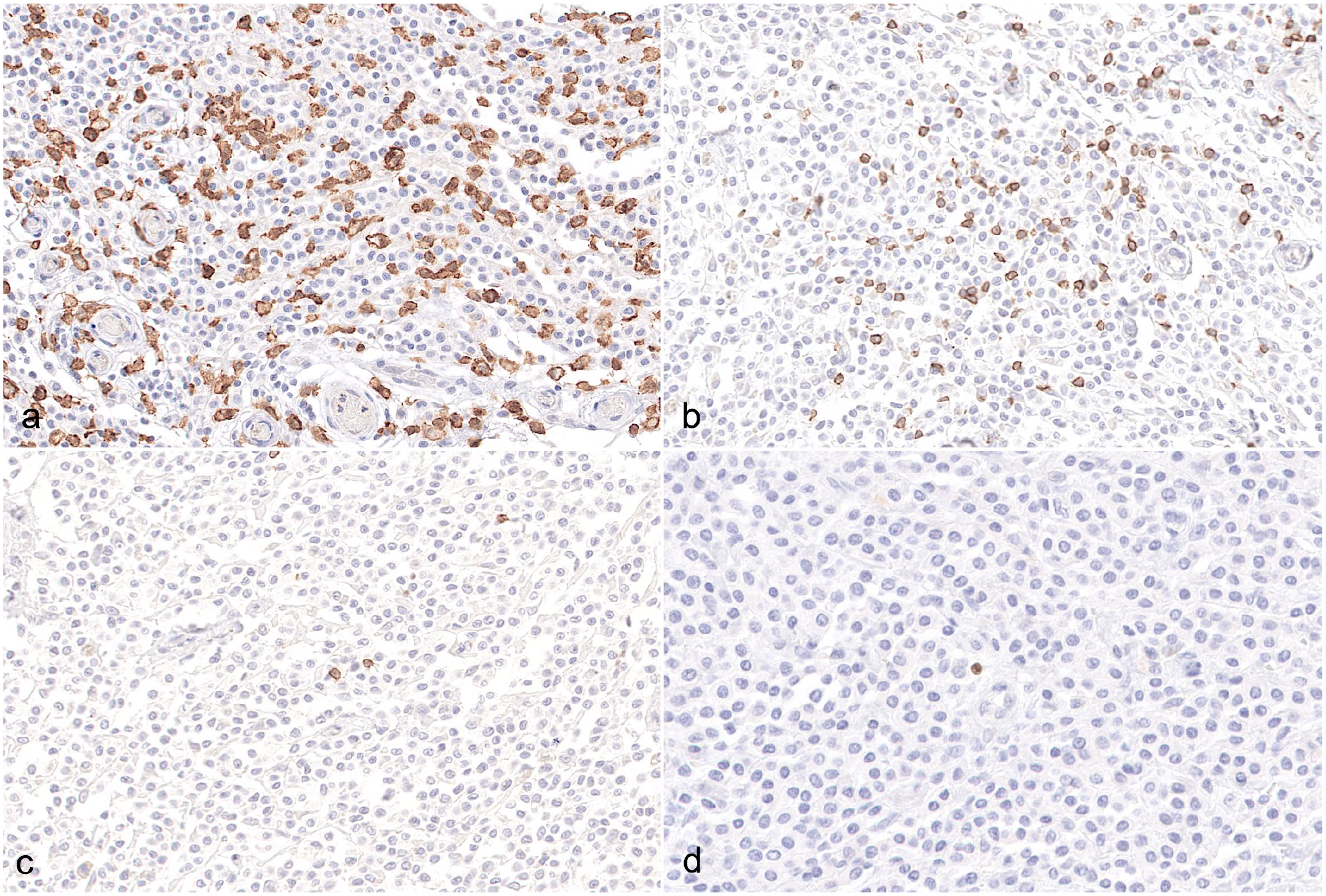
Canine cutaneous mast cell tumor (MCT). (a) Tumor-associated macrophages are the predominant population of infiltrating leukocytes in canine MCTs with numerous individual and aggregated macrophages among neoplastic cells. Iba1 immunohistochemistry (IHC). (b) Among the tumor-infiltrating lymphocytes (TILs), CD3+ T cells are the most common cell population among the neoplastic mast cells. CD3 IHC. (c) Scattered individualized B cells are observed among neoplastic mast cells. CD20 IHC. (d) Regulatory T cells are the least common cell population of TILs, with rare positive cells admixed with the neoplastic cells. Foxp3 IHC.
CD3
All 38 MCTs were immunolabeled for CD3 and analyzed. The tumors had variable numbers (7 score 0, 15 score 1, 10 score 2, and 6 score 3) and diameters (7 score 0, 21 score 1, 8 score 2, and 2 score 3) of perivascular aggregates and variable numbers (2 score 0, 16 score 1, 9 score 2, 11 score 3, and 0 score 4) and extents (2 score 0, 11 score 1, 6 score 2, 6 score 3, and 13 score 4) of CD3+ interstitial infiltrates.
Comparing cutaneous and subcutaneous MCTs, subcutaneous MCTs had higher total scores (P = .0269) and higher percent positive areas (P = .0074) compared with cutaneous MCTs, while no statistically significant differences in perivascular scores (P = .0518) or interstitial scores (P = .179) were identified (Figs. 3a, b, 4a, b).
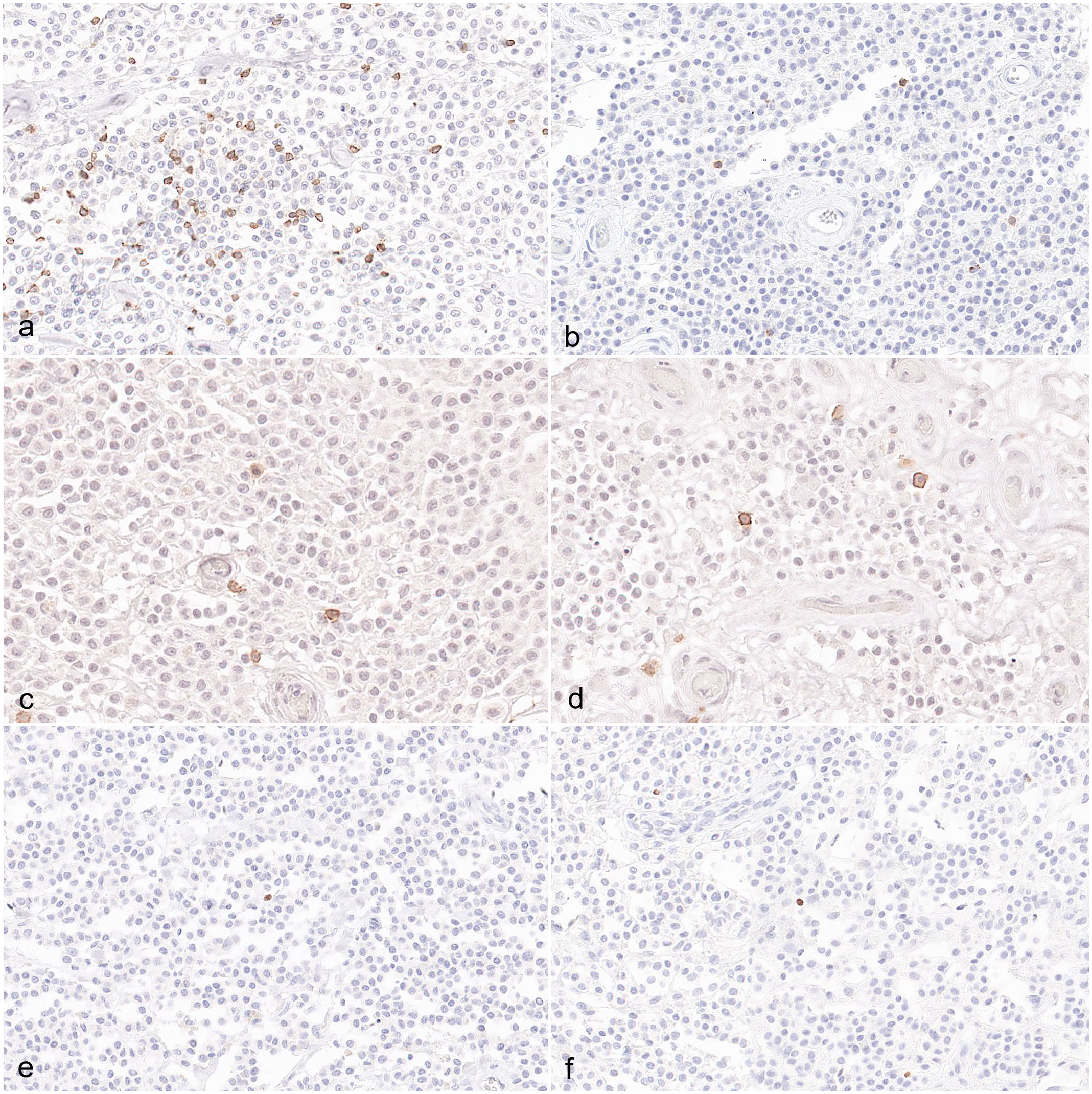
Canine mast cell tumors (MCTs), interstitial infiltrates, and immunohistochemistry for (a, b) CD3, (c, d) CD20, and (e, f) Foxp3. (a) Cutaneous MCTs with HN0 had increased CD3+ T-cell infiltration compared with (b) cutaneous MCTs of other sentinel lymph node grade. There were no differences between (c) cutaneous and (d) subcutaneous MCTs regarding CD20+ interstitial B cells. There were no differences between (e) cutaneous and (f) subcutaneous MCTs regarding Foxp3+ interstitial regulatory T cells infiltration.
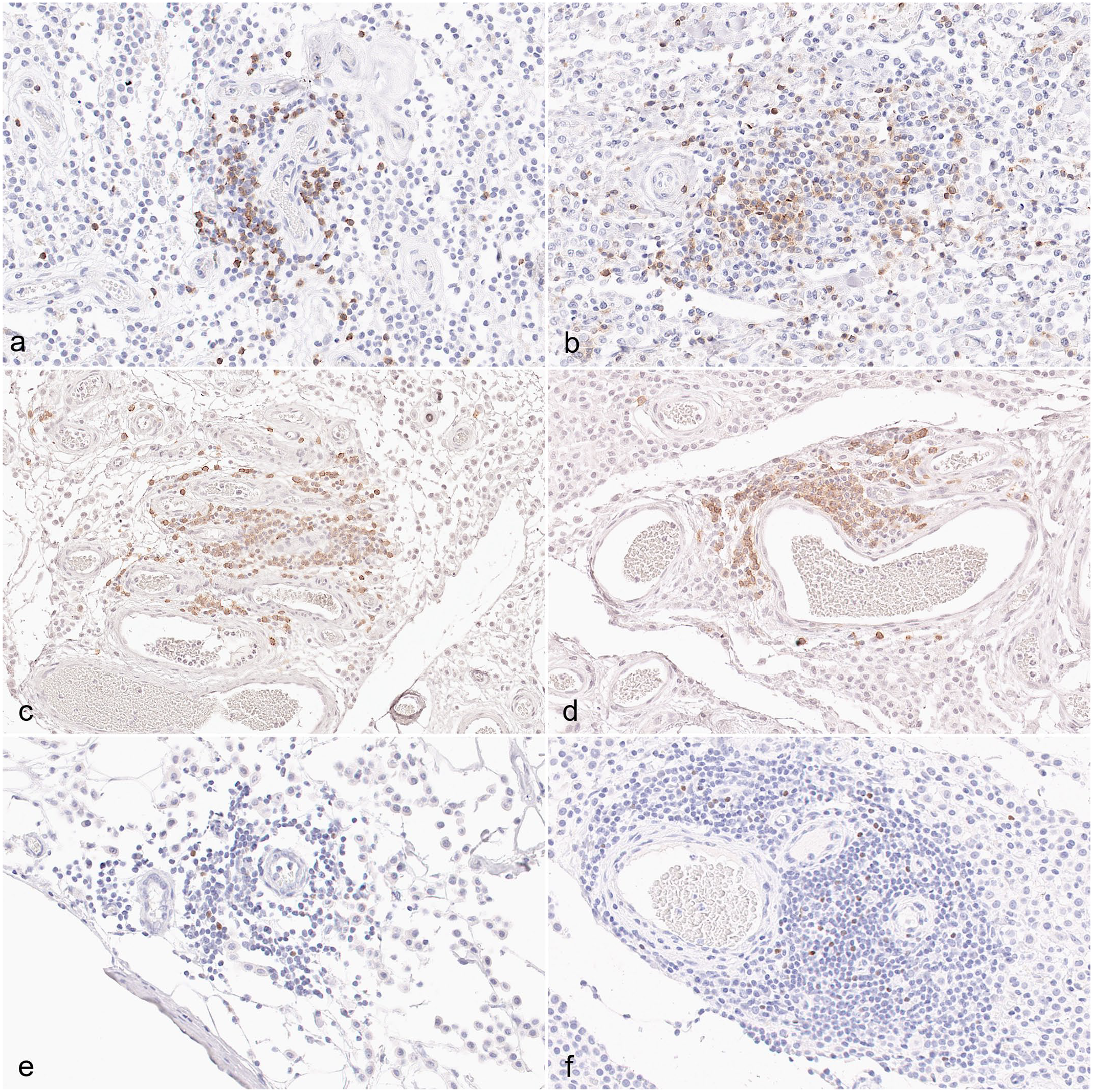
Canine mast cell tumors (MCTs), perivascular infiltrates, and immunohistochemistry for (a, b) CD3, (c, d) CD20, and (e, f) Foxp3. (a) Cutaneous MCTs had decreased perivascular CD3+ T-cell infiltration compared with (b) subcutaneous MCTs. There were no differences between (c) cutaneous and (d) subcutaneous MCTs regarding CD20+ perivascular B cells. (f) Subcutaneous MCTs had increased perivascular Foxp3+ regulatory T cells compared with (e) cutaneous MCTs.
Regarding SLNs metastatic status, no statistically significant differences were identified in the interstitial scores, perivascular scores, total scores, or percent positive areas between non-metastatic and pre-metastatic (HN0-HN1) and early and overtly metastatic (HN2-HN3) MCTs, either considering all tumors together or cutaneous and subcutaneous MCTs separately. When comparing non-metastatic (HN0) MCTs and pre-, early, and overtly metastatic (HN1, HN2, and HN3, respectively) MCTs, non-metastatic MCTs had higher interstitial scores (P = .0248) and higher total scores (P = .0323) than pre-, early, and overtly metastatic MCTs, while no differences in the perivascular scores or percent positive areas were detected, when considering all tumors together. Considering only cutaneous MCTs, statistically significant differences between non-metastatic and pre-, early metastatic, and overtly metastatic MCTs were identified in the interstitial scores (P < .001) and total scores (P = .0042), which were higher in non-metastatic MCTs than in pre-, early metastatic, and overtly metastatic MCTs, but not in perivascular scores or percent positive areas.
CD20
Thirty-two tumors (32/38) were successfully immunolabeled for CD20 and analyzed, while 6 cases were excluded due to poor immunohistochemical reactivity or artifacts hindering digital image analysis. Examined tumors had variable numbers (7 score 0, 7 score 1, 10 score 2, and 8 score 3) and diameters (7 score 0, 15 score 1, 6 score 2, and 4 score 3) of perivascular aggregates and variable numbers (12 score 0, 8 score 1, 5 score 2, 7 score 2, and 0 score 4) and extents (12 score 0, 5 score 1, 6 score 2, 1 score 3, and 8 score 4) of CD20+ interstitial infiltrates.
Comparing cutaneous and subcutaneous MCTs, no statistically significant differences in the interstitial scores, perivascular scores, total scores, or percent positive areas were identified (Figs. 3c, d, 4c, d).
Regarding SLNs metastatic status, no statistically significant differences were identified in the interstitial scores, perivascular scores, total scores, or percent positive areas between non-metastatic and pre-metastatic MCTs and early and overtly metastatic MCTs when considering either all MCTs together or cutaneous and subcutaneous MCTs separately. No statistically significant differences between non-metastatic and pre-, early, and overtly metastatic MCTs were identified in the interstitial scores, perivascular scores, total scores, or percent positive areas, when considering cutaneous and subcutaneous MCTs together or only cutaneous MCTs.
Foxp3
Thirty-five tumors (35/38) were successfully immunolabeled for Foxp3 and analyzed, while 3 cases were excluded due to poor immunohistochemical reactivity or artifacts hindering digital image analysis. The examined tumors had variable numbers (22 score 0, 5 score 1, 7 score 2, and 1 score 3) and diameters (21 score 0, 2 score 1, 8 score 2, and 4 score 3) of perivascular aggregates and variable numbers (7 score 0, 24 score 1, 3 score 2, 1 score 3, and 0 score 4) and extents (7 score 0, 16 score 1, 8 score 2, 3 score 3, and 1 score 4) of Foxp3+ interstitial cells.
Comparing cutaneous and subcutaneous MCTs, subcutaneous MCTs had higher perivascular total scores (P = .0002) and total scores (P = .0136) compared with cutaneous MCTs, while no statistically significant differences in the interstitial scores or percent positive areas detected through digital image analysis were identified (Figs. 3e, f, 4e, f).
Regarding SLNs metastatic status, no statistically significant differences were identified in the interstitial scores, perivascular scores, total scores, or percent positive areas, between non-metastatic and pre-metastatic MCTs and early and overtly metastatic MCTs either considering all tumors together, or cutaneous and subcutaneous MCTs separately. No statistically significant differences between non-metastatic and pre-, early, and overtly metastatic MCTs were identified in the interstitial scores, perivascular scores, total scores, or percent positive areas, when considering cutaneous and subcutaneous MCTs together or only cutaneous MCTs.
Iba1
Thirty-five tumors (35/38) were successfully immunolabeled for Iba1 and analyzed, while 3 cases were excluded due to poor immunohistochemical reactivity or artifacts hindering digital image analysis. All tumors were diffusely (the total positive area ranging from 2% to 14%) infiltrated by Iba1+ cells with variable morphologies: 7 tumors were primarily infiltrated by round Iba1+ cells, 10 primarily by stellate/spindle Iba1+ cells, and 18 tumors by Iba1+ cells with mixed morphologies (Fig. 5a, b).
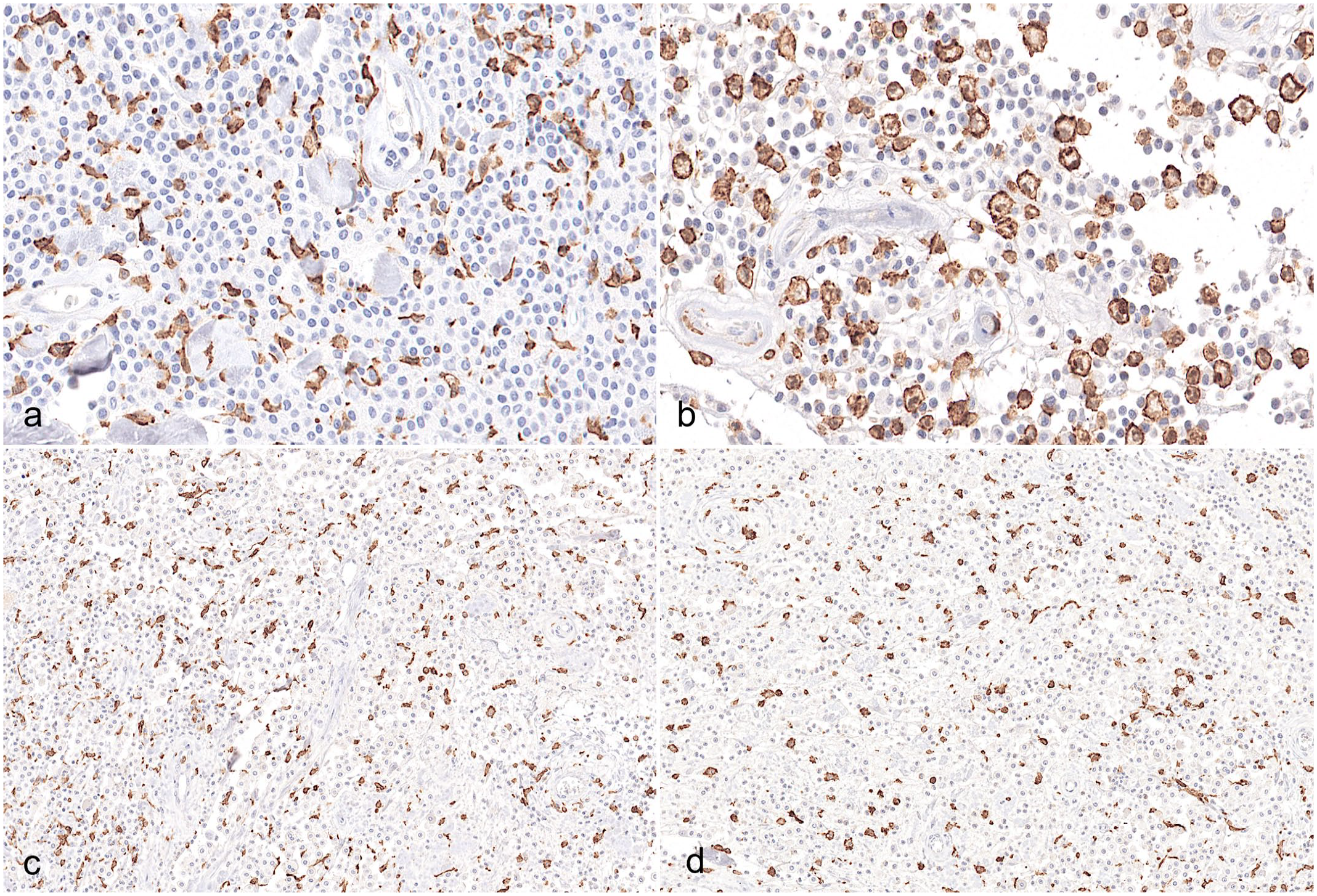
Canine mast cell tumors (MCTs), Iba1 immunohistochemistry. (a) Macrophages infiltrate among neoplastic cells with a predominant stellate/spindle morphology in a MCT associated with a HN2 sentinel lymph node (SNL). (b) Macrophages infiltrate among neoplastic mast cells with a predominant round morphology in a MCT associated with a HN0 SLN. There were no differences between (c) cutaneous and (d) subcutaneous MCTs regarding macrophagic infiltration.
No association between Iba1+ cells morphology and MCT location (cutaneous or subcutaneous MCTs) was identified. Comparing cutaneous and subcutaneous MCTs, no statistically significant differences in the percent positive areas were identified (Fig. 5d, e).
Regarding SLNs metastatic status, no statistically significant difference was identified in the percent positive areas between non-metastatic and pre-metastatic MCTs and early and overly metastatic MCTs when considering all tumors together, or cutaneous and subcutaneous MCTs separately. No statistically significant difference between non-metastatic and pre-, early, and overtly metastatic MCTs was identified in percent positive areas, either considering cutaneous and subcutaneous MCTs together or only cutaneous MCTs. MCTs with stellate/spindle Iba1+ cells or with mixed morphology Iba1+ cells had a statistically higher incidence of metastasis (HN2 and HN3 SLNs) compared with MCTs with primarily round Iba1+ cells (P = .0272). The association was even stronger when comparing non-metastatic MCTs to pre-, early, and overtly metastatic MCTs, with primarily round Iba1+ cells identified only in non-metastatic MCTs (P = .0031).
Discussion
The interaction between the immune system and tumors plays a central role in tumor development, growth, and metastatic spread and involves numerous molecular pathways and tumor-infiltrating immune cells with both pro- and antitumoral activities. 28 Tumor-infiltrating immune cells are of pivotal importance in this interaction with plenty of studies in human oncology highlighting their prognostic relevance and potential therapeutical application.5,28,35,41,53,54,56,57 In veterinary oncology, this field is mostly unexplored; immune cell infiltration has been evaluated in a limited set of canine malignancies, mainly mammary and melanocytic tumors, and only few data exist for MCTs.3,4,7,20,30 –34
All MCTs evaluated in the present study were infiltrated by immune cells, with Iba1+ TAMs being the main cellular population in both cutaneous and subcutaneous MCTs, followed by perivascular CD20+ B cells, perivascular and interstitial CD3+ T cells, and Foxp3+ Tregs.
All MCTs were diffusely infiltrated by Iba1+ TAMs, which accounted for 2% to 14% of the tumoral area. This result is consistent with data in human oncology, where TAMs are the predominant infiltrating cell population of the tumoral mass, representing up to 50% of the mass in several tumor entities, such as lymphomas and glioblastomas.1,45 It is also comparable with previous findings in canine lymphomas and canine soft tissue sarcomas, where Iba1+ TAMs represented up to 63% of the cells.10,43 The morphology of Iba1+ TAMs is variable (round, stellate/spindle). There is evidence that the different morphologies of TAMs could reflect a different polarization, with intratumoral M1 macrophages tending to assume a round morphology, while M2 macrophages tending to appear as elongated or dendritic cells.8,18,44 Although in the present study, the percent of the area of Iba1+ TAMs was not correlated with MCT histologic location or SLN metastatic status, a predominance of spindle to stellate macrophages was statistically correlated with early and overtly metastatic SLNs, suggesting a potential pro-tumoral or tumor-permissive M2-oriented phenotype of these macrophages. On the other hand, round Iba1+ cells were statistically correlated with non-metastatic MCTs, potentially highlighting their antitumoral, metastasis-preventive M1-like function. TAMs have been correlated with poor prognostic outcome and several negative prognostic histopathologic features in different human and canine malignancies.4,5,30,37,43,57 In canine soft tissue sarcomas, increased TAM infiltration was linked to higher histological grade and higher neoplastic proliferative activity. 11 In canine mammary cancers, increased numbers of TAMs infiltrating both stroma and neoplastic lobules was correlated with negative prognostic features, such as vascular invasion, node metastasis, and increased angiogenesis, as well as shorter survival times.20,32 Concerning canine MCTs, few data exist in the literature. The percentage of Iba1-positive area was found to be increased in high-grade MCTs compared with low-grade MCTs, and correlated with ulceration, nuclear atypia, and mitotic count. 7 The presence of macrophages detected through MAC-387 immunohistochemistry in canine cutaneous MCTs was correlated with the presence of nuclear atypia, mitotic activity, high and II and III grades, and shorter survival times in one study. 13 In contrast to our results, 36% of the tumors in that study (18 out of 50) did not have macrophage infiltrates. 13 The difference could be related to a different specificity of MAC-387 compared with Iba1, which was used in the present study. Despite this evidence, TAMs’ pro-tumoral role has not been definitively proven in all canine cancers. One study evaluating TAMs in canine colonic adenocarcinomas and adenomas found higher levels of TAMs in neoplastic samples compared to healthy colon, but no correlation with the patients’ prognosis or histologic prognostic features, or with higher TAM counts in adenomas compared with adenocarcinomas. 50 Although not investigated, the higher macrophagic infiltration observed in benign compared with malignant tumors could be explained by the specific polarization assumed by the infiltrating macrophages, with adenomas being potentially associated with high numbers of antitumoral M1 macrophages and adenocarcinomas being infiltrated by fewer M1 macrophages and more M2 pro-tumoral macrophages. The importance of macrophagic polarization has been highlighted in several human malignancies, while it has only rarely been investigated in canine cancers.52,55,56 Canine benign mammary tumors were infiltrated by higher numbers of nitric oxide synthase 2+ M1 macrophages, while malignant tumors were infiltrated by higher numbers of CD206+ M2 macrophages. 20 MAC 387+ and MHC-II+ cells were also described in canine nasal carcinomas, but no correlations with prognosis or other histologic features were evaluated. 42 Further studies are thus needed in dogs to characterize TAM infiltrates and to better define the role of polarized M1 and M2 macrophages in canine cancers, including cutaneous and subcutaneous MCTs.
TILs are a major component of immune cell infiltrates in several cancers in both human and veterinary medicine.11,21,25,34 TILs are both B and T cells, the latter composed of a heterogeneous cell population, including CD8+ cells exerting cytotoxic activity, CD4+ helper cells involved in cytokine production and T- and B-cell activation, and Foxp3+ Tregs with immunosuppressive activity. 21 TILs have been investigated in numerous canine neoplasms through a combination of histopathology, immunohistochemistry, and cytofluorimetry.3,11,17,29,31,33,34,42,54 In a study on canine oral melanomas, cutaneous melanomas, and cutaneous melanocytomas, the authors reported a mild lymphocytic infiltration in cutaneous tumors and a more robust lymphocytic infiltration in oral tumors potentially related to a lower expression of tumor-associated antigens by neoplastic cells in cutaneous tumors and to the presence of a specific microenvironment in the oral mucosa favoring lymphocytic activation and infiltration in the oral tumors. 31 Based on immunohistochemical phenotyping of lymphocytes, CD3+ T cells were not correlated with clinical outcome, including local recurrence or metastasis, while CD20+ B cell infiltration was correlated with an increased risk of local recurrence, increased risk of metastases, and decreased survival times. 31 In contrast to these studies, in mammary tumors, CD3+ cells and CD4+ cells were increased in malignant compared with benign tumors and correlated with decreased survival time, while no correlation with survival time could be found in relation to CD20+ cell infiltrates.11,33 Through cytofluorimetric analysis, it was demonstrated that canine oral melanomas with higher survival rates had greater infiltration by CD8+ cells and canine complex mammary tumors with poorer prognoses had higher infiltration by CD4+ cells, thus highlighting an antitumoral response of cytotoxic cells in canine melanomas and a pro-tumoral or tumor-permissive response related to CD4+ helper cells in mammary tumors.11,54 While, to the authors’ knowledge, infiltrating B cells have not been previously investigated in canine MCTs, a study found no differences in T-cell infiltration comparing high- and low-grade cutaneous MCTs. 7 While CD20+ B cells were not correlated with tumor histological location and SLN metastatic status in our cases, we highlighted a statistically significant difference between cutaneous and subcutaneous tumors concerning CD3+ T cells. We speculate that the different anatomy of the dermis and subcutis with diverse venous and lymphatic vascular networks could impact on recruitment of leukocytes. Concerning SLN metastatic spread, cutaneous MCTs with HN0 SLN showed a statistically significant higher infiltration of interstitial CD3+ T cells compared with HN1, HN2, and HN3 MCTs, suggesting a potential role of CD3+ T cells in preventing metastatic spread of cutaneous MCTs.
Foxp3 cells exert immunosuppressive activity mainly through CTLA4/CD80-CD86 pathway activation and transforming growth factor beta (TGF-β) and IL-10 secretion and have been proven useful in prognostic determination of several human malignancies, such as lymphoma and colorectal cancers.16,41,53 In canine mammary carcinomas, intestinal lymphomas, oral malignant melanomas, melanocytic tumors, oral squamous cell carcinomas, and pulmonary carcinomas, intra-tumoral infiltrates of Foxp3+ cells were significantly correlated with decreased overall survival times.3,17,29,34 Interestingly, canine hepatocellular carcinomas and MCTs were associated with low numbers of Tregs.7,34 These data are consistent with what was observed in our study, in which Foxp3+ cells were present in very low numbers, and when present they were mainly arranged as perivascular infiltrates. Our results highlighted a difference between cutaneous and subcutaneous MCTs, further supporting an anatomical location-dependent lymphocytes recruitment. No correlation between intratumoral Foxp3+ cells and SLN metastatic status was identified, suggesting that Tregs do not play a major role in the immune system-neoplastic regulation in canine MCTs.
This study had some limitations. Among the 12 cases of subcutaneous MCTs, only 1 HN0 was present, preventing a comparison between this and other SLNs classes. Furthermore, only grade II/low-grade cutaneous MCTs and only 3 HN3 SLNs cases meeting the inclusion criteria were retrieved from the archives of the Veterinary Teaching Hospital, potentially hampering the investigation of MCTs with the highest metastatic potential. This is partly related to the fact that dogs with clinically enlarged lymph nodes in our hospital are either treated with neoadjuvant therapy or do not always undergo lymphoscintigraphy of the SLNs especially with a pre-surgical positive lymph node cytology, and partly related to the Veterinary Teaching Hospital caseload, which has always been characterized by a low number of grade III/high-grade MCTs and of overtly metastatic lymph nodes, as seen in previous studies.12,38 In conclusion, this study evaluated TAMs and TILs in canine grade II/low-grade cutaneous and subcutaneous MCTs through immunohistochemistry, highlighting the presence of numerous Iba1+ TAMs, various numbers of CD3+ T cells and CD20+ B cell, and rare Foxp3+ Tregs. The stellate/spindle morphology of Iba1+ cells was correlated with higher SLN metastatic classes (HN2 and HN3), suggesting a possible pro-tumoral (M2) phenotype. The presence of increased CD3+ T cell and Foxp3+ infiltrates in subcutaneous MCTs compared with cutaneous MCTs and of CD3+ cells in non-metastatic (HN0) cutaneous MCTs compared with pre-, early, and overtly metastatic (HN1, HN2, and HN3, respectively) MCTs supports the presence of a location-dependent tumor-immune microenvironment and suggests that increased intratumoral T-cell infiltration might contribute to SLN metastatic spread prevention in canine cutaneous grade II/low-grade MCTs. Further studies, enrolling a larger cohort of patients with MCTs of different grades and evaluating disease-free intervals and survival times with long follow-up periods, are needed to assess the prognostic role of TILs and TAMs in canine MCTs.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241244851 – Supplemental material for Tumor-associated macrophages and tumor-infiltrating lymphocytes in canine cutaneous and subcutaneous mast cell tumors
Supplemental material, sj-pdf-1-vet-10.1177_03009858241244851 for Tumor-associated macrophages and tumor-infiltrating lymphocytes in canine cutaneous and subcutaneous mast cell tumors by Luca Bertola, Benedetta Pellizzoni, Chiara Giudice, Valeria Grieco, Roberta Ferrari, Lavinia E. Chiti, Damiano Stefanello, Martina Manfredi, Donatella De Zani and Camilla Recordati in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
