Abstract
Swine are increasingly studied as animal models of human disease. The anatomy, size, longevity, physiology, immune system, and metabolism of swine are more like humans than traditional rodent models. In addition, the size of swine is preferred for surgical placement and testing of medical devices destined for humans. These features make swine useful for biomedical, pharmacological, and toxicological research. With recent advances in gene-editing technologies, genetic modifications can readily and efficiently be made in swine to study genetic disorders. In addition, gene-edited swine tissues are necessary for studies testing and validating xenotransplantation into humans to meet the critical shortfall of viable organs versus need. Underlying all of these biomedical applications, the knowledge of husbandry, background diseases and lesions, and biosecurity needs are important for productive, efficient, and reproducible research when using swine as a human disease model for basic research, preclinical testing, and translational studies.
Swine were domesticated over 10,000 years ago in the Near East and have continued to be a species closely linked with human existence. 32 In fact, during the Shang dynasty (approximately 1600–1050 years before the common era), the symbol for home or house was depicted as a pig with a roof over it. 144
The first well-documented experiments involving pigs were performed more than 2000 years ago, by Erasistratus, Galen, and Aristotle.54,147 From these investigations, a base understanding of the anatomic and physiologic relationships between the heart, blood flow, vasculature, and lungs was established. Galen also studied the nervous system in pigs and demonstrated the necessity of the recurrent laryngeal nerve for vocalization. He also showed that transection of the spinal cord between the third and fourth vertebra resulted in the cessation of respiration in the pig.54,147
Swine have been studied for many years, but the popularity of swine in research has dramatically increased in recent years for several reasons. First, the pig is closer in size and has an increased lifespan more comparable to humans than most other research species, making it a useful model for many preclinical and translational studies. These factors are especially important for studies in trauma, medical imaging (diagnostic, therapeutic, monitoring), cancer, and surgical interventions, to name a few.71,103,119,130 Second, traditional rodent models do not always produce relevant phenotypes for studying some conditions or for preclinical testing and safety assessments. Swine are a suitable large animal model that can fill in this gap in model biology. Examples of this include cystic fibrosis (CF) and Duchenne muscular dystrophy.57,72,115 Third, recent development of new gene editing techniques have accelerated the efficiency and speed of generating swine models using targeted gene-editing.143,150 Fourth, limitations in swine-specific expertise, reagents, and facilities had been a concern for investigators, but these hurdles are being removed making swine models more attractive and feasible for translational studies. 10 Finally, swine have preferential advantages over other traditional large animal models. The use of nonhuman primates (NHPs) is increasingly restricted by ethical issues, costs, and increased regulations. 10 Use of canine species has also been on the decline in recent years. For example, dogs are considered a sensitive species and special approvals are required before their use as subjects in funded research by the United States Veterans Administration. 85 With the looming concerns of using NHP and dog models, swine have become an appropriate option for large animal studies. In addition, pigs are increasingly being used in pharmacological studies to examine pharmacokinetics, oral bioavailability, skin penetrance, and toxicology of new drugs and entities, the discussion of which is beyond the scope of this article.7,46,110,132
Numerous swine breeds are available, 130 and similar to dog breeds, were often created to serve specific purposes. Agricultural breeds such as the Yorkshire, Duroc, and Landrace were bred for feed efficiency and high fecundity. Agricultural breeds have also been bred for meat quality, which has resulted in selecting for or against some cytochromes which may affect toxicology studies. 19 There are over 30 breeding groups of minipigs in the world. 116 Those discussed here are most commonly represented in the literature. The most popular minipig breeds used in research in the United States are the Göttingen, Hanford, Sinclair, and Yucatan. Both the Sinclair and Yucatan have mini or micro lines that have been selected for decreased size to facilitate toxicology studies with smaller amounts of test article requirements and to control the need for larger housing units. The Göttingen and the micro-Yucatan are the smallest of the minipig breeds. The Hanford is most similar in size to a human and is therefore well-suited to surgical studies that require any subsequent length of survival. The Göttingen is also available in Europe and Asia, but these are separate breeding colonies and genetic divergence has occurred. In Japan, the Micromini breed is popular, and in China, there is an inbred breed called Bama. Apart from the Bama breed, all other breeds mentioned here are considered outbred. The terminology of breed versus strain in pigs has not yet become stringently defined. In mice, a strain is defined as a line that is genetically identical resulting from repeated brother × sister matings (typically 20 generations). In pigs, this has only been pursued in a few breeds (e.g. Bama, Wuzhishan) with the Wuzhishan breed having both inbred and outbred lines that have been developed.80,142,153 Additional resources about pig breeds and resources are available for further reading.40,116,130
Other considerations include the source of the swine. Many Yorkshire background pigs in the United States originate from farms that also breed animals for market. Although housing facilities for agricultural and research animals may be separate, animals from these vendors may be exposed to the typical swine diseases.45,155 Research facilities and equipment are also important considerations. For instance, swine surgeries or imaging may require dedicated facilities, equipment, and professional expertise for successful studies.118,130
Common Nutritional and Infectious Diseases of Swine That May Impact Biomedical Studies
Health challenges in commercially raised pigs often present a diagnostic dilemma and potential complication in research studies.45,155 Many common pathogens are endemic on commercial farms, so disease expression depends upon the interplay of host susceptibility (immunity, nutrition, and genetics), environmental factors (temperature, humidity, animal density, etc.), and the pathogen itself (dose and virulence). Given this interplay, disease expression is often variable within and among farms or vendors. From a diagnostic standpoint, detection of a pathogen may or may not equal disease, and it is up to the clinician, diagnostician, or both to align diagnostic test results with clinical and pathological observations.
Clinical signs, history, and epidemiologic observations are important components for generating a specific case definition in a population of pigs. The case definition is a critical step in the diagnostic process. All deviations from normal (physiological or behavioral) are important clues used to differentiate disease processes and should be characterized, quantified, and recorded as part of daily clinical observations. When coupled with gross lesions at necropsy, these data drive the case definition and inform sampling for laboratory testing to screen for specific etiological agents. In pigs, many pathogens commonly affect pigs at consistent periods in the production cycle (suckling phase, weaning phase, grower-finisher, and adult), and this can also help in formulating a case definition. Table 1 provides a list of some of the more common infectious agents detected in U.S. commercial swine, along with most impacted ages, typical clinical signs, and microscopic lesions. It is important to remember that microscopic lesions might be observed when reviewing samples from a diagnostic case or when scoring samples for a research investigation. 45
Common endemic infectious diseases in U.S. swine.
To further confound the issue, many available diagnostic tests (polymerase chain reaction [PCR], culture, enzyme-linked immunosorbent assay [ELISA], and some immunohistochemistry (IHC)) do not differentiate pathogenic strains of some endemic agents from nonpathogenic strains or attenuated vaccine strains. Modified live vaccines are common in swine production (porcine reproductive and respiratory syndrome virus [PRRSV], Lawsonia spp., Salmonella spp., and Escherichia coli), and their use and the time since administration must be accounted for when interpreting diagnostic test results. Finally, mixed infections with one or more endemic agents are common, if not the normal. Additional ancillary diagnostic techniques such as in situ hybridization and gene sequencing may be required to differentiate pathogens from nonpathogens.
When using commercial pigs in research, it is also important to have a relevant understanding of modern swine production. Domestic pigs raised in confinement are restricted to what is provided and many diets are prepared in small mills or mixed on site. In this way, there is opportunity for human error in diet preparation, and diet formulations are therefore not always equal to the “diet as fed.” Furthermore, commercial swine diets are generally formulated to minimize cost and use cereal grains as base ingredients with supplemental protein, vitamins, and minerals. As such, deficiencies and over-supplementation are possible. Much of the nutritional research for pigs is done in optimal environments, whereas pigs in endemically infected herds or under experimental conditions may have different metabolic demands and needs. Formulations are also based on the mean response in a population, and individual animals may show signs of deficiency while most of a population remains unaffected. It is good practice to save liver, serum, and feed samples in any research study involving pigs for potential future analysis should study results indicate the potential for underlying nutritional issues.
Common nutritional issues in commercial swine include deficiencies in vitamins A, D, and E and deficiencies in iron and selenium. Marginal deficiency in vitamin A is common and may manifest as poor reproductive performance, weak born piglets, and increased susceptibility to infections. Sow’s milk is iron deficient, so piglets require supplementation within 5 days of birth. Piglets not receiving proper supplementation may develop microcytic, hypochromic anemia. Mulberry heart disease (MHD) is a nutritional cardiomyopathy that is often observed in well-conditioned, young growing pigs and is thought to occur due to excess free radicals. While MHD has been historically associated with vitamin E and selenium deficiency, this condition can manifest in pigs sufficient in both vitamin E and selenium. 86 In one study involving samples received at a veterinary diagnostic laboratory, liver selenium concentrations were adequate in all pigs with lesions of MHD and vitamin E concentrations were only deficient in 25% of pigs with MHD. 92 This discord has prompted investigation into other potential causes of MHD, such as viral coinfections, but evidence for coinfections has thus far been lacking. 117 It is important to note that nutrient requirements in pigs can be interdependent and, as an example, several dietary factors (iron, copper sulfur, etc.) can influence vitamin E requirements.47,48,120 Disorders in calcium and phosphorus homeostasis can occur secondary to diet formulation errors, including vitamin D deficiency, and can manifest in varying degrees of lameness and unexpected fractures.
While responsible animal research functions under the care of a clinical veterinarian, it is useful to include an experienced swine pathologist and diagnostic laboratory resources as part of the team. Awareness of background diseases optimizes biosecurity and management practices to prevent these conditions from confounding the reproducibility of research. Examples of potential background lesions that can confound research studies are shown in Fig. 1a–c.
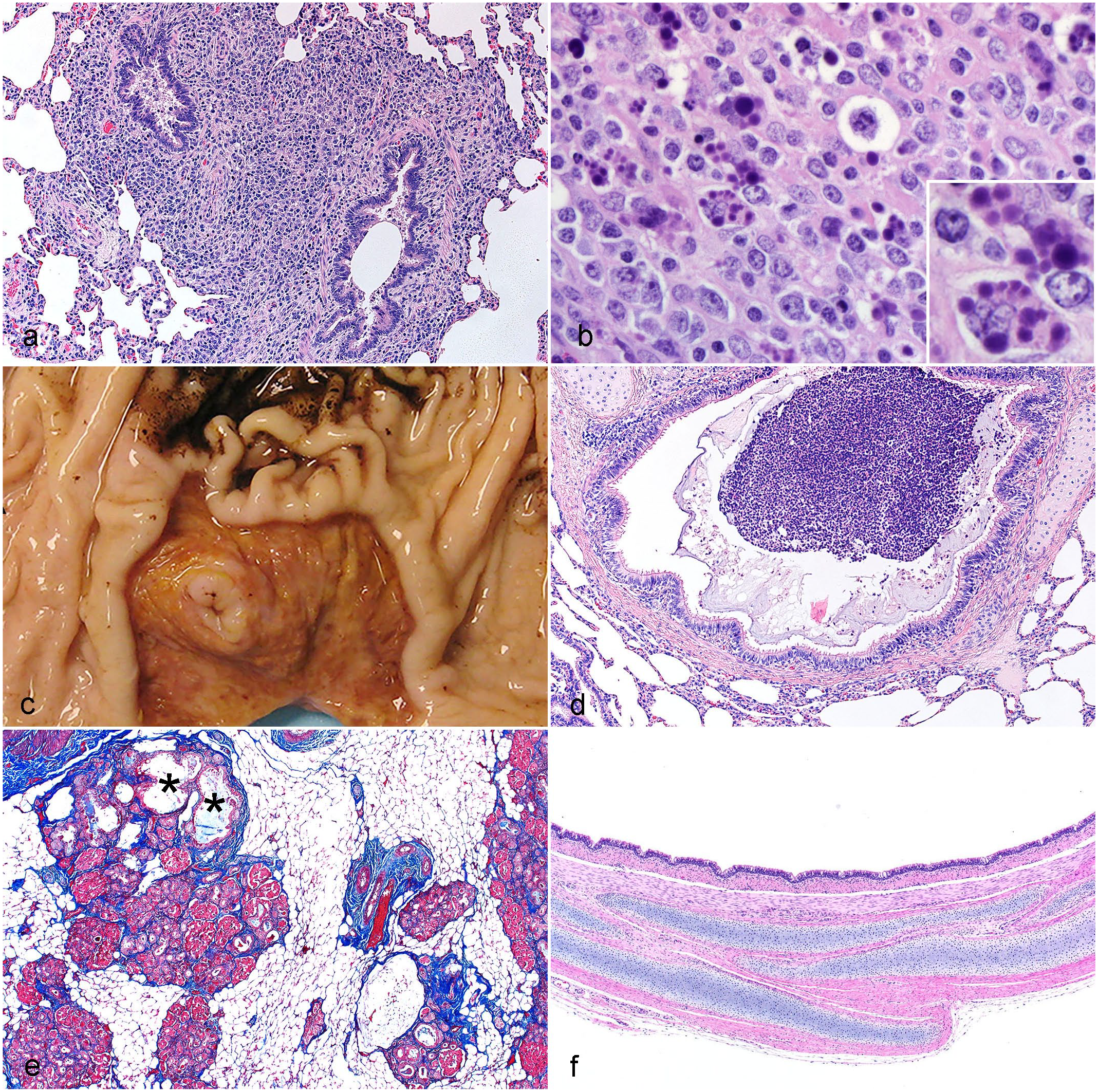
Examples of incidental and model-specific lesions in swine. (a and b) A 2-month-old pig with histiocytic effacement of the bronchus-associated lymphoid tissue in (a) lung and (b) gut-associated lymphoid tissue (GALT) in ileum caused by porcine circovirus-2 infection. Note the botryoid cytoplasmic inclusions in macrophages of the GALT (b, inset). Hematoxylin and eosin (HE). (c) Stomach from a 2-month-old pig with ulceration of the pars esophagea and hemorrhage evidenced as black, coffee ground-like material on adjacent stomach mucosa. (d and e) Two-month-old pig model of cystic fibrosis (CF). (d) The airway is obstructed with mucopurulent secretions and (e) pancreas is composed of remnant lobules sitting in a backdrop of white adipose tissue. The cystic (*) and fibrotic regions of the diseased CF pancreas (e) were the original basis for naming the condition cystic fibrosis.5,127 HE and Masson’s trichrome, respectively. (f) Newborn swine model of ectodermal dysplasia. 91 Note that the tracheal wall has normal structure except for the absence of submucosal glands.
Medical Device Testing
Medical devices are an omnipresent feature of modern medical care. The potential locations and functions of device implants combined with their future applications appear limitless, and their mode of therapy delivery is broad and will most certainly expand to human diseases in ways we have yet to fathom. In contrast to the pharmaceutical industry in which progress is often painstakingly incremental, medical device innovation and improvement is very fast-paced and preclinical testing must be concluded before first use in humans. Preclinical studies are performed primarily in large animal species, such as pigs, sheep, goats, and decreasingly in dogs. 85 These studies are sandwiched between bench tests to ensure engineering performance and device longevity on one hand, and clinical trials to confirm safety and ensure efficacy on the other. Preclinical testing is a scientific necessity and a regulatory requirement to reveal if a device performs to its intended use and has the anticipated biological and physiological effects. The pathology portion of well-executed and well-documented preclinical research plays an important role in accelerating the approval process as it provides sound scientific evidence that strengthens other study endpoints to substantiate claims.
When preclinical testing is needed to achieve regulatory approval for an implanted or temporarily inserted medical device,6,31 several factors need to be considered in view of the proposed animal model. These factors include similarities to human anatomy and physiology, comparable organ sizes allowing treatment with or implantation of actual size medical devices as they are intended for human clinical implant (i.e., the “final finished product” with the intended clinical design), reproducibility based on low genetic variation among individuals, and an understanding of unavoidable background lesions including the impact of long-term healing effects.
There are no strict regulatory guidelines on the species or the number of animals to be used to reach approval for a medical device, let alone the details of how the pathology shall be conducted. 31 If researchers have established that an animal study is necessary, regulatory bodies expect that a specific and detailed scientific justification will be provided for the animal species and number of animals. 31 One approach is to model the study according to previous work that used a certain animal species in support of a highly similar, successfully approved device. Another approach is to adopt the methods of a preclinical study that was previously conducted in that species and published in peer-reviewed literature. 31 Overall, the animal species and its physiological attributes should provide “a test system that offers a best attempt at simulating the clinical setting.”31,34 A comprehensive understanding of the benefits and limitations of different species and the setting in which the study will be conducted is of unquestionable importance. Despite these aspirations, it is noteworthy that preclinical safety or efficacy recommendations for devices are usually based on low animal numbers, on abbreviated implant-duration times, and on implant data from healthy animals. Animal testing is both expensive and time consuming, and while necessary, does not entirely replicate the complexities that influence the phenotypic and molecular diversity observed in human diseases. However, clinical trials in humans also have inherent limitations and are even more costly and time-consuming than animal studies. Thus, animal models that have similar anatomy and biology, like swine, are useful for certain preclinical studies in testing medical devices. 112 Therefore, project leaders should prudently complete their risk assessment before conducting and explaining animal studies.
In addition to the physiologic and pathologic challenges in extrapolating animal data to humans, 154 the medical device world is prone to carelessly use the term “animal models” thus fostering the belief that there is a repository of animal disease models that can be used during device development. 112 In contrast, there is a low incidence of naturally occurring pathologies described in large animals that can be applied as actual disease models during device development. In the pig, for example, human intervention by way of selective breeding has eliminated genes that increase disease susceptibility. Moreover, most domestic farm pigs are slaughtered at a young age (less than 6-months-old), precluding the detection of late onset diseases such as cancer. 40 Genetically engineered pigs are vital for gaining a proper understanding of disease mechanisms 95 but have yet to find their way into medical device innovation, testing, and approval. 112 Put simply, current animal models used in device research or regulatory submissions are imperfect anatomical and physiological proxies to the human species. They are predominantly implant or surgery stand-ins, with the main intent of the normal, healthy animal recipient being of adequate size to accommodate human-tailored devices for an extended period of time to show evidence of safety.
Swine are the major large animal species used in the preclinical testing of medical devices. The U.S. Food and Drug Administration (FDA) recognizes swine as a suitable model for biocompatibility testing and the safety assessment of implanted devices in a variety of organs and systems.56,130 Coronary stents,84,93,94 abdominal aortic aneurysm grafts, 154 prosthetic heart valves, 133 abdominal meshes, 53 and neurovascular devices 121 are medical devices that are commonly tested in swine. In addition, temporarily inserted energy-based devices for renal denervation 108 or for cardiac,124,126 lung, 114 or liver ablations 50 are usually evaluated in swine.
Juvenile farm swine are the default model for nonsurvival surgical training classes. 130 The haptics of performing surgery cannot be replaced by mannequins or simulators, so a pig or pig tissue is commonly used in training for interventional catheter techniques and complex trauma and endoscopic procedures. Apart from implanter training, prototype devices are also widely tested in farm pigs via acute nonsurvival procedures.
For survival studies that extend beyond a few weeks, the rapid growth rate of commercial farm pigs usually precludes its participation; this is not only because handling them at body weights beyond 100 kg is a challenge but also that their rapid growth impacts the sizing ratio between the implant location and the device, particularly for implantable cardiac or vascular devices. Fully grown miniature swine offer the same physiological advantages as the farm pig model, but at the equivalent maturity they are much smaller and therefore, the preferred selection for long-term studies. Currently, a widely used miniature swine breed is the Yucatan minipig. Although available from various vendors, their phenotype and disposition remain relatively consistent. 87 One disadvantage in the use of Yucatan pigs is their high cost. Since they are specifically bred for research in relatively small numbers, the cost per animal is typically 3 times higher than for a regular farm swine, and 1.5 to 2 times higher than for a purpose-bred hound.
While swine are a preferred species for testing medical devices, it is important to mention other large animal species that might have advantages over swine. Sheep are preferentially used for the long-term implantation of subcutaneous electronic devices (cardiac- or neuro-stimulators, see example in Supplemental Figure S1) with or without absorbable antibacterial envelopes 66 as well as for the testing of complex lead systems designed for deep-brain stimulation, 26 or brain catheters that are used in focal brain ablation. 61 For central-nervous studies, sheep are preferred to swine due to their favorable skull anatomy. Sheep have also been the historical model for prosthetic mitral valve research, 111 as adolescent sheep show a detrimental tendency for leaflet calcification similar to that observed in children, and sheep continue to be used for catheter-delivered, suture-less mitral valves. 138 Skin implants in ovine candidates are favored to the porcine or canine model as sheep are less prone to pocket infections and can accommodate more concurrently implanted devices over their larger body surface; the sheep heart is also used for leadless cardiac pacing devices. 139 Dogs were the historical model for developing and refining cardiac leads for pacing, defibrillation, or both including recent magnetic resonance imaging (MRI)-safety experiments but are currently in the process of being replaced by pigs or sheep. This is usually the case unless the project has already matured toward a pivotal GLP study where dogs continue to be the preferred species for purposes of regulatory submission.
Several recent publications offer helpful approaches on how to perform the preclinical pathology analysis of medical devices33,106,122,141 or discuss their assessment via ISO standard 10993-6. 88 Without exaggeration, pathology data provide the most relevant measure of local device healing and its systemic consequences. This information is essential in determining the overall impact, both positive and negative, of a medical device on the body. As previously stated by Rouselle and Wicks, “Preparing and processing medical device implants for evaluation is a relatively high-risk and high-dollar process in which studies get made and endpoints can be lost with no second chance.” 105
Swine are currently center stage in the process of bringing incrementally improved or completely novel medical devices to market. At what time and to what degree computer modeling or human “digital twins” will substitute for a living animal in preclinical research, or whether genetic swine models of human disease will have matured into a practical, economical, and validated translational platform for testing device efficacy and safety cannot be predicted.
Swine Models for the Study of Genetic Diseases
Genetic disorders have been classified into 3 basic categories. The first category is monogenic disorders in which mutations of a specific gene causes the condition. An example of a monogenic disorder is CF with mutations in the cystic fibrosis transmembrane conductance regulator (CFTR). 128 The second category of genetic disease is chromosomal disorders where chromosomes are absent or duplicated. An example of a chromosomal disorder is trisomy 21 (Down syndrome), a condition with 3 copies of chromosome 21 instead of standard 2 copies. The third category is complex disorders in which multiple genes and/or environmental conditions contribute in a complex fashion to develop disease. For example, predisposition to cancer can involve germline and somatic gene mutations in combination with environmental factors like diet, smoking exposure, and so on. 8 As our understanding of gene regulation and expression continues to grow, the complexity of genetic diseases may not so easily fit into a discrete classification system.51,113 Monogenic disorders will be highlighted as a basic example of the usefulness of gene-targeted swine models.
A historic advantage of mouse models over other species in research had been the ability to target genes using homologous recombination and embryonic stem cells. 41 This approach was not readily adaptable to other species, giving mice a favored status by investigators. However, new opportunities began to appear in the 1990s when Dolly the cloned sheep was generated using somatic cell nuclear transfer. 146 Since then, a variety of new approaches including gene-editing techniques have provided the ability to specifically target a gene and make these edits in a much faster and efficient manner.30,143,150 These newer technical advances have contributed to the growing number of useful swine models used to study human diseases. 68
Swine models of monogenetic diseases are often generated by targeting a gene of interest (mutation or excision) to prevent production of a functional protein (a “knockout”). However, in some situations, study of a genetic disorders may require further nuances to this approach to optimally study the model. For instance, mutations in CFTR-/- produced a knockout model of CF, one of the first reported swine models of a human genetic disease. 104 However, the CFTR-/- swine model exhibited a severe intestinal obstruction (meconium ileus) phenotype that required surgical correction to permit postnatal study. 127 One explanation for the enhanced severity of the swine model was that CFTR mutations in humans often produced a protein that had partial function (hypomorph), whereas the swine model was a null without functional protein.104,127,140 Efforts to target the gene corresponding to the most common mutation in humans (F508) produced a swine model with low level of protein function (~6% of wildtype). 89 This low level of functional protein was sufficient to predispose the lung to CF lung diseases, but insufficient to correct the severe intestinal obstruction that required surgical intervention. 89 To overcome the intestinal obstruction, another knockout model was produced that also had a CFTR transgene under the control of the fatty acid binding protein promotor for targeted intestinal expression of CFTR protein. This model mitigated the severe intestinal phenotype to allow for postnatal lung disease studies without the requirement of surgical intervention. 129
The CF swine model has arguably had some of the broadest impact in studying a monogenic disorder while targeting a wide variety of systems, organs, and tissues (Fig. 1d, e and Table 2). 128 The breadth of monogenic disorders studied in gene-edited swine continues to grow, and these specific models often identify niches for targeted studies to accelerate research and complement other models. For instance, a recently developed model of ectodermal dysplasia lacks submucosal glands in cartilaginous airways (Fig. 1f). 91 This model will be useful for studying the relative contributions of submucosal gland secretions to host defense in normal lung as well as CF models.
Selected examples of monogenic disorders studied in gene-edited swine.

The use of swine models for the study of genetic diseases has facilitated the development/validation of several diagnostic tools and research approaches that have helped advance the available resources for swine models in research. These resources include immunolabeling validation,29,74,75 prosection techniques, 76 allodynia/nociception assessments, 55 cognitive/memory assessments, 145 recording sleep/rest/behavior patterns, 55 and imaging modalities/techniques,118,119,125 to name a few.
Swine in Xenotransplantation
Organ transplantation in humans is the best therapy available for virtually any form of end-stage organ failure. It is a life-saving treatment for patients at imminent risk of death from heart, lung, or liver failure. Kidney transplantation provides a life-lengthening survival benefit for those with end-stage kidney disease on dialysis. Transplantation can even be considered “life-giving” in the case of uterine transplant. Despite the clear benefits, transplantation has always been limited by the availability of donor organs.
This has been especially true in the case of kidney transplantation. The increasing prevalence of end-stage kidney disease has created a situation where, until very recently, the number of patients added to transplant waiting lists has exceeded the number of transplants performed in a given year. 62 Thus, the wait has grown longer and longer. Sadly, many patients will either die or become too sick for transplantation before a donor kidney becomes available. Decades of efforts to increase both living and deceased organ donation have had a modest effect and have not been able to overcome this deficit. These ongoing issues have prompted a search for alternative sources for donor organs.
Xenotransplantation is the transplantation of an organ from one species to another. This contrasts with allotransplantation, in which the organ is transplanted between 2 individuals of the same species. The concept of using an animal as the donor organ source is not new. Very early experiments in transplantation often used animals as the donor source; 16 however, the lack of understanding of immunology doomed all of these experiments. Once clinical transplantation was shown to be feasible in 1954 in twins, 43 there was renewed interest in xenotransplantation. At this point, NHPs were thought to be the preferred source, owing to their close genetic relationship to humans.100,123 These clinical experiments also failed. It would take several decades of developing an understanding of immunology before significant improvement in outcomes was seen in preclinical models. During that time, it also became apparent that NHPs would not be a suitable source animal. The need for organs would quickly outstrip the ability of NHP populations to provide them, and there was increasing concerns about the ethical use of NHPs in medicine and research, which persists today. 10
Pigs offered a suitable alternative to NHPs as the donor source. As pigs have been domesticated and bred for human consumption for millennia, we have a good understanding of the required husbandry, and there are fewer ethical concerns regarding their use. Pigs reach reproductive age relatively early, reproduce quickly, and produce large litters, allowing for the development of a large donor population. Pigs grow quickly and reach an appropriate size for donation to a human early in life, but also have relatively long lifespans, so there is the potential a donated organ could last a long time posttransplantation. Finally, pig renal function is like that of humans in most ways, with similar electrolyte concentrations, osmolality, and glomerular filtration rates.42,107
Despite these advantages of pigs as organ donors, crossing the species barrier introduces some new immunologic challenges. Pig cells express surface glycoproteins that are absent in humans and other primates. 60 These glycoproteins act as carbohydrate antigens, akin to ABO blood groups. The major antigen, the alpha-Gal antigen, 69 is expressed by human gut flora. 36 This exposure leads to innate antibody formation and immunologic memory against the antigen. 35 Therefore, pig-to-human xenotransplantation is a case of immunologically incompatible transplantation, and grafts suffer from hyperacute rejection within minutes of reperfusion. These challenges can at least be partially overcome by genetic manipulation of the donor pigs to delete alpha-1,3-galactosyltransferase, 58 which generates alpha-Gal, and this has been made considerably easier with the development of CRISPR-Cas9 techniques. 109
Work in pig-to-NHP models of xenotransplantation has shown that making these genetic modifications can improve the outcomes of xenotransplantation and prevent hyperacute rejection.25,59 Subsequent efforts over many years have led to the development of multigene-edited pigs in which all 3 major carbohydrate antigens have been knocked-out; 6 human regulatory genes have been inserted, and the growth hormone receptor gene has been knocked out (Table 3). However, as the genetic modifications of the donor pig have been fine-tuned toward the human immune system in anticipation of clinical translation, the donor pigs have become less-ideal donors for an NHP immune system, limiting progress in studying these preclinical models.4,70,149
Genetic modifications used in pig-to-human or pig-to-nonhuman primate xenotransplantation.

This has led to the development of a human preclinical research model, known as the Parsons model. In this approach, a recently deceased human, declared dead by brain-death criteria but deemed unsuitable for organ donation, is used as the recipient of pig organs. This allows testing of the donor organs in a human recipient, with a human immune system, without risk to a living human. Two groups have reported experiments using this approach and have demonstrated that no hyperacute rejection occurs, as predicted by prospective cross-matching; the kidney xenografts produce urine; and no zoonotic infection or chimerism was detected.82,99 The Parsons model is limited by the pathophysiology of brain death, and therefore no long-term outcomes will be achieved in this model. However, this remains a critical step toward clinical translation of xenotransplantation.
In January 2022, the FDA granted a compassionate use exception for use of a porcine heart for xenotransplantation into a living human. The heart functioned well until the patient expired for unclear reasons 60 days following transplantation. 38 This case is notable for both the success of the transplant, but also because the recipient was found to have detectable porcine cytomegalovirus about 3 weeks posttransplant. This highlights ongoing knowledge gaps that will need to be addressed for xenotransplantation to fully reach the clinic, including the biosafety of porcine-derived grafts, the function of a porcine kidney in a living human, long-term durability of the xenotransplant, and optimal immunosuppression strategies. These issues are also observed in human-to-human allotransplantation and are not inherently unique to cross-species transplantation. However, in the context of allotransplantation, we benefit from roughly 70 years of clinical experience. In contrast, xenotransplantation remains in its infancy, and ultimately these questions may only be answerable in a clinical trial in living humans.
In conclusion, swine have increasingly become a useful and complementary resource for biomedical research, device testing, and medical therapies through xenotransplantation. The use of good swine husbandry and biosecurity practices along with awareness of common background diseases helps to prevent confounding factors that cloud reproducible research.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231222235 – Supplemental material for Swine models in translational research and medicine
Supplemental material, sj-pdf-1-vet-10.1177_03009858231222235 for Swine models in translational research and medicine by David K. Meyerholz, Eric R. Burrough, Nicole Kirchhof, Douglas J. Anderson and Kristi L. Helke in Veterinary Pathology
Footnotes
Supplemental material for this article is available online.
Authors’ Note
Part of this material was presented as part of a half-day session at the 2022 ACVP meeting in Boston, MA, USA.
Author Contributions
All authors contributed to the planning, writing, and editing of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to acknowledge support from the NIH (grant nos. HL163556, HL152960, and HL09184), Cystic Fibrosis Foundation, United Therapeutics Corporation, Lung Biotechnology PBC, and Revivicor.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
