Abstract
We report a case of Becker muscular dystrophy in a 6-month-old, mixed-breed, castrated male pig detected with macroglossia at a meat inspection center. The pig presented a severely enlarged tongue extending outside its mouth. The tongue was firm and pale with discolored muscles. Histologically, there was severe fibrosis, fatty replacement, and myofiber necrosis, degeneration, and regeneration. Immunofluorescence showed focal and severely weak labeling for dystrophin at the sarcolemma of myocytes in the tongue. Analysis of dystrophin mRNA showed a 62 base pair insertion between exons 26 and 27. The insertion was derived from intron 26. Based on these findings, we diagnosed the case as Becker muscular dystrophy—the first known muscular dystrophy case induced by pseudoexon insertion in animals.
Keywords
Muscular dystrophy (MD) is a hereditary primary skeletal muscle disorder characterized by progressive muscle degeneration and gradual muscle fiber loss. 6 Dystrophinopathy is a type of MD induced by a pathogenic variant in the dystrophin gene. Dystrophinopathies encompass a spectrum of X-linked muscle diseases ranging from mild to severe. They include Duchenne muscular dystrophy (DMD), Becker muscular dystrophy (BMD), and DMD-associated dilated cardiomyopathy. 5 DMD is the most severe phenotype of the disease. BMD is characterized by late onset and ranges from mild to severe progressive MD. In meat inspection centers, porcine muscle degeneration characterized by excessive fatty replacement within muscles has been reported.4,13 Among these lesions, a case of human BMD-like myopathy characterized by decreased dystrophin labeling of the sarcolemma was reported. 8 Only one family of dystrophinopathies has been reported in pigs, with a spontaneously occurring substitution in exon 41 causing an amino acid change from arginine to tryptophan.7,14 We report a case of porcine BMD caused by dystrophin pseudoexon insertion derived from intron 26 and detected with macroglossia at a meat inspection center. To the best of our knowledge, this is the first case of MD by pseudoexon insertion in animals.
The case was a 6-month-old, mixed-breed, castrated male pig. No prior clinical history was available; however, the pig exhibited no clinical signs and had a mildly lean physique during the antemortem inspection at the meat inspection center. During the macroscopic inspection, the tongue was severely enlarged and extended outside the mouth (Fig. 1). On cut section, the tongue was firm and pale with discolored muscles (Fig. 2). The remaining muscles appeared normal, and no significant lesions were noted in other organs.
The tongue and skeletal muscle from the cervical and brachium regions were fixed in 10% neutral buffered formalin, and frozen skeletal muscle from the cervical region was used for molecular analysis of the dystrophin gene. The formalin-fixed samples were routinely processed and embedded in paraffin. Sections of 4 μm were stained with hematoxylin and eosin (HE). Immunofluorescence was performed on formalin-fixed paraffin-embedded samples. The indirect immunofluorescence antibody technique was applied using an anti-dystrophin rabbit polyclonal antibody (ab15277; 1:400 dilution; Abcam, Cambridge, Massachusetts) as the primary antibody. To unmask the epitope, the tissue sections were boiled in ImmunoSaver (Nisshin EM Co., Ltd., Tokyo, Japan) for 45 minutes. An Alexa Fluor 488-labeled anti-rabbit IgG goat polyclonal antibody (1:500 dilution; Life Technologies Corporation, Carlsbad, California) and Vectashield mounting medium with DAPI (Vector Laboratories, Burlingame, California) were used. Formalin-fixed paraffin-embedded muscle from a healthy 3-month-old male pig was used as control.
For molecular analysis of the dystrophin gene, genomic DNA and total RNA were extracted from the frozen skeletal muscle using the NucleoSpin Tissue Kit (Macherey-Nagel, Düren, Germany) and RNeasy Mini Kit (Qiagen, Hilden, Germany), respectively. We used the extracted total RNA for cDNA synthesis using the SuperScript IV First-Strand Synthesis System (Thermo Fisher Scientific, Waltham, Massachusetts). The purified genomic DNA and cDNA were used for subsequent PCR analyses. Supplemental Table S1 shows the primer pairs for the PCR reactions. PCR was performed under the following conditions: 94°C for 2 minutes, 40 cycles at 98°C for 10 seconds, 56°C for 30 seconds, and 68°C for 2 minutes. The PCR products were then sequenced.
Histologically, the myofibers of the tongue were multifocally replaced by abundant fibrous and adipose tissue (Fig. 3). The remaining myocytes were swollen and rounded with variable diameters and they had loss of cross striations, vacuolar changes, and hyalinization of the sarcoplasm. Moreover, myofiber necrosis, endomysial macrophage infiltration, regeneration with internalized nuclei, and myofiber splitting were noted (Fig. 4). In the skeletal muscle sampled from the cervical and brachial regions, mild fibrosis and slight myofiber degeneration were observed. Immunofluorescence analysis using an anti-dystrophin antibody revealed discontinuous and severely weak labeling at the sarcolemma of myofibers of the muscle from the tongue, and reduced discontinuous labeling of myofibers from the cervical and brachial regions (Fig. 5). In contrast, dystrophin labeling of the sarcolemmal membrane in the control skeletal muscle was intense and continuous (Fig. 6).
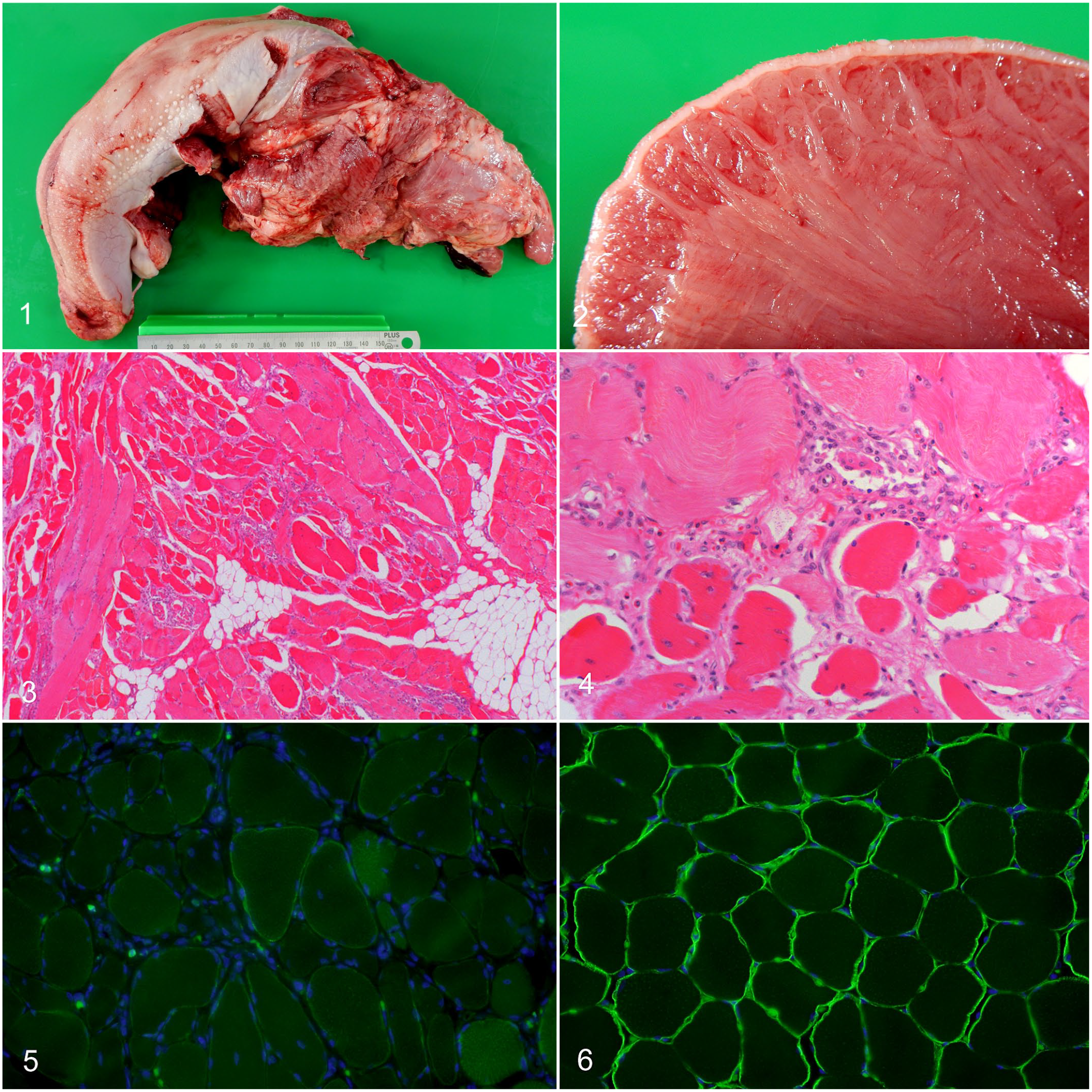
Becker muscular dystrophy (BMD), tongue, pig.
Dystrophin mRNA was amplified by RT-PCR, and 2 amplicons were identified that spanned exons 22–31. Sequencing showed that 1 PCR product was normal-sized with a normal exon nucleotide sequence, whereas the second PCR product showed a 62-base pair (bp) insertion between exons 26 and 27 (Fig. 7). A BLAST search using the 62-bp sequence revealed a 100% match with intron 26 of the pig dystrophin gene on the Sus scrofa chromosome X genome (28449365-28449304 of GenBank NC_010461.5). In the present case, the intron 26 region was amplified from genomic DNA. Further, the 62-bp sequence was confirmed, and the 112-bp sequence around the insertion was a 100% match with the 2 pig DNA sequences on chromosome X (28449298-28449409 of NC_010461.5 and 152814-152925 of FP565156.7), among the 60 sequences in the GenBank database. Moreover, the inserted sequence was bracketed by 5′ and 3′ splice sites (AG<exon>GT), and a putative branch point and a polypyrimidine tract sequence were observed upstream of the 3′ splice site (Fig. 8).
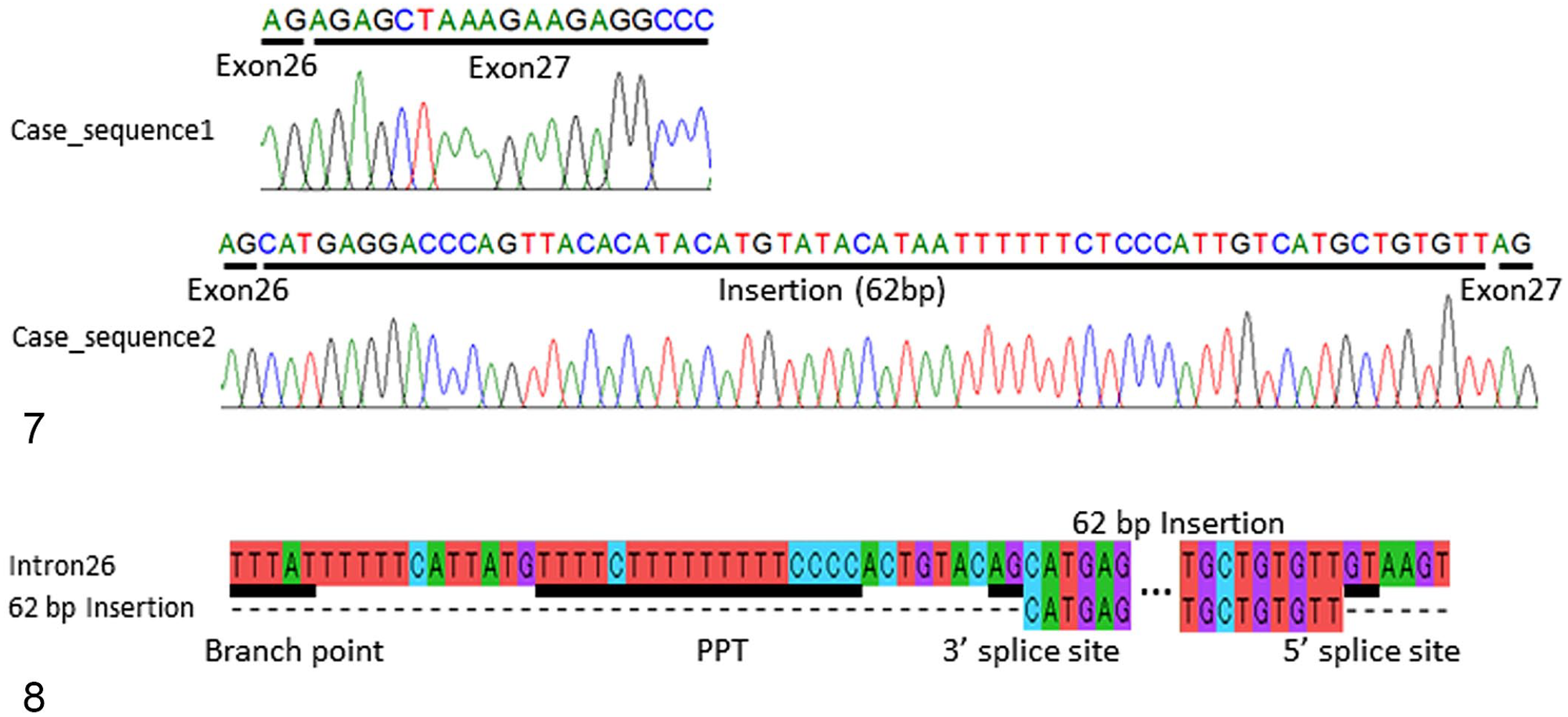
Molecular analysis of the dystrophin gene.
The present report demonstrates muscular degeneration, reduced dystrophin expression, and a 62-bp insertion in the dystrophin mRNA in a 6-month-old, mixed-breed, castrated male pig. Based on the characteristic findings of BMD that include mild clinical signs and decreased dystrophin labeling, we diagnosed the present case as BMD. Moreover, residual wild-type dystrophin mRNA was detected, thereby explaining the milder signs of BMD rather than the more severe phenotype characteristic of DMD.
Reports on naturally occurring dystrophinopathies in domestic animals are few. Late-onset BMD is rare among dystrophinopathies and was only reported in several dogs1,9 and in 1 pig. 7 In humans, BMD is characterized by its late onset, wherein patients lose their ability to walk in their late teens or early twenties. 3 No clinical history was available before 6 months of age and disease onset was unknown; however, the present case exhibited no clinical signs at 6 months of age, and clinical presentation and histopathological and immunochemical findings are consistent with BMD in humans.
Deletions or duplications in the dystrophin gene are typical causes of BMD, with the remaining cases occurring presumably due to point mutations. 5 In addition, pseudoexon insertions—intronic point mutations that create novel splice sites,
resulting in the insertion of intronic sequence within the mRNA—are implicated in various dystrophinopathies including BMD in humans.10,18,20 This is the first known case of dystrophinopathy caused by pseudoexon insertion in animals. BMD due to an intronic splicing mutation in intron 26 has been reported in humans.2,17 The mutation observed in the present case—a 62-bp insertion—has not previously been reported; however, the clinical and laboratory findings presented by the cited literature in human patients with BMD (including mild clinical signs, decreased dystrophin labeling, and residual wild-type dystrophin mRNA)2,17 were comparable to the present case. Remarkably, in this case, the sequence of intron 26 was 100% homologous with 2 normal pig DNA sequences on chromosome X among the 60 sequences in the GenBank database. In this pig, the clinical manifestation of BMD was unnoticed (i.e., no behavioral signs such as abnormal gait or movements), and only the physical examination revealed a slightly lean body condition and macroglossia; thus, it is possible that BMD may occur undetected in pig populations.
In patients with DMD or BMD, some muscles (particularly the gastrocnemius muscle) are enlarged due to fat and connective tissue deposition, which is known as pseudohypertrophy. 11 Although the tongue may not be a frequent site of pseudohypertrophy, it has been reported in humans,12,19 dogs, 16 and cats. 15 Similarly, in the present case, macroglossia lesions were consistent with pseudohypertrophy. Furthermore, macroglossia is one of the characteristic findings of DMD and BMD.
In meat inspection centers, porcine muscle degeneration is characterized by excessive fatty replacement within muscles; 4 human BMD-like myopathies are occasionally observed. 8 Various dystrophin mutations might be latently present in pig populations, and some variants may be expressed as macroglossia.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221079669 – Supplemental material for Macroglossia in a pig diagnosed as Becker muscular dystrophy due to dystrophin pseudoexon insertion derived from intron 26
Supplemental material, sj-pdf-1-vet-10.1177_03009858221079669 for Macroglossia in a pig diagnosed as Becker muscular dystrophy due to dystrophin pseudoexon insertion derived from intron 26 by Naoyuki Aihara, Sho Kuroki, Ryosuke Inamuro, Yumiko Kamiya, Takanori Shiga, Yoh Kikuchihara, Emiko Ohmori, Michiko Noguchi and Junichi Kamiie in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was partially supported by a research project grant awarded by the Azabu University Research Services Division and The Ito Foundation research grant project.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
