Abstract
For decades, the advancement of cancer research has relied on in vivo models for examining key processes in cancer pathogenesis, including neoplastic transformation, progression, and response to therapy. These studies, which have traditionally relied on rodent models, have engendered a vast body of scientific literature. Recently, experimental cancer researchers have embraced many new and alternative model systems, including the zebrafish (
The zebrafish (
Although zebrafish are relative newcomers to the field of comparative cancer research, these small animals have emerged as a powerful tool for investigators. As vertebrate organisms, they share many features of embryonic development and adult anatomy that are critical for comparative animal models. 64,71 In the world of cancer research, this has important implications ranging from conserved signaling pathways and transcriptional regulatory mechanisms, to organogenesis and tissue differentiation, to cellular transformation and tumor initiation. Zebrafish cancer models have been produced by a variety of methods, leading to a broad spectrum of cancer susceptibilities in animals of multiple age groups. Flexibility in genetic manipulation, adaptability to current tools for cancer analysis, and capacity to define cellular and molecular interactions in vivo provide the zebrafish with its own very unique set of advantages for the investigation of cancer biology.
Mechanisms for Developing Cancer Models in Zebrafish
Zebrafish cancer models have been developed by a number of mechanisms (Table 1). Although initial studies relied on chemical carcinogens for tumor induction, 105 cancer modeling in zebrafish has expanded to include numerous mutant and transgenic lines. In this setting, chemical carcinogens are often useful for identifying tumor-prone zebrafish lines from forward genetic screens 97,98 or in combination with genetic manipulation in defining a tumor susceptibility phenotype. 39 Although the use of forward genetic screens to develop cancer models is not typical in traditional animal models, multiple zebrafish cancer models have been successfully derived by this method. 4,5,32,73,97,98 In some cases, this approach has identified novel genes with a previously unknown or poorly defined link to cancer susceptibility. 4,97,98
Methods Used to Generate Cancer Models in Zebrafish.
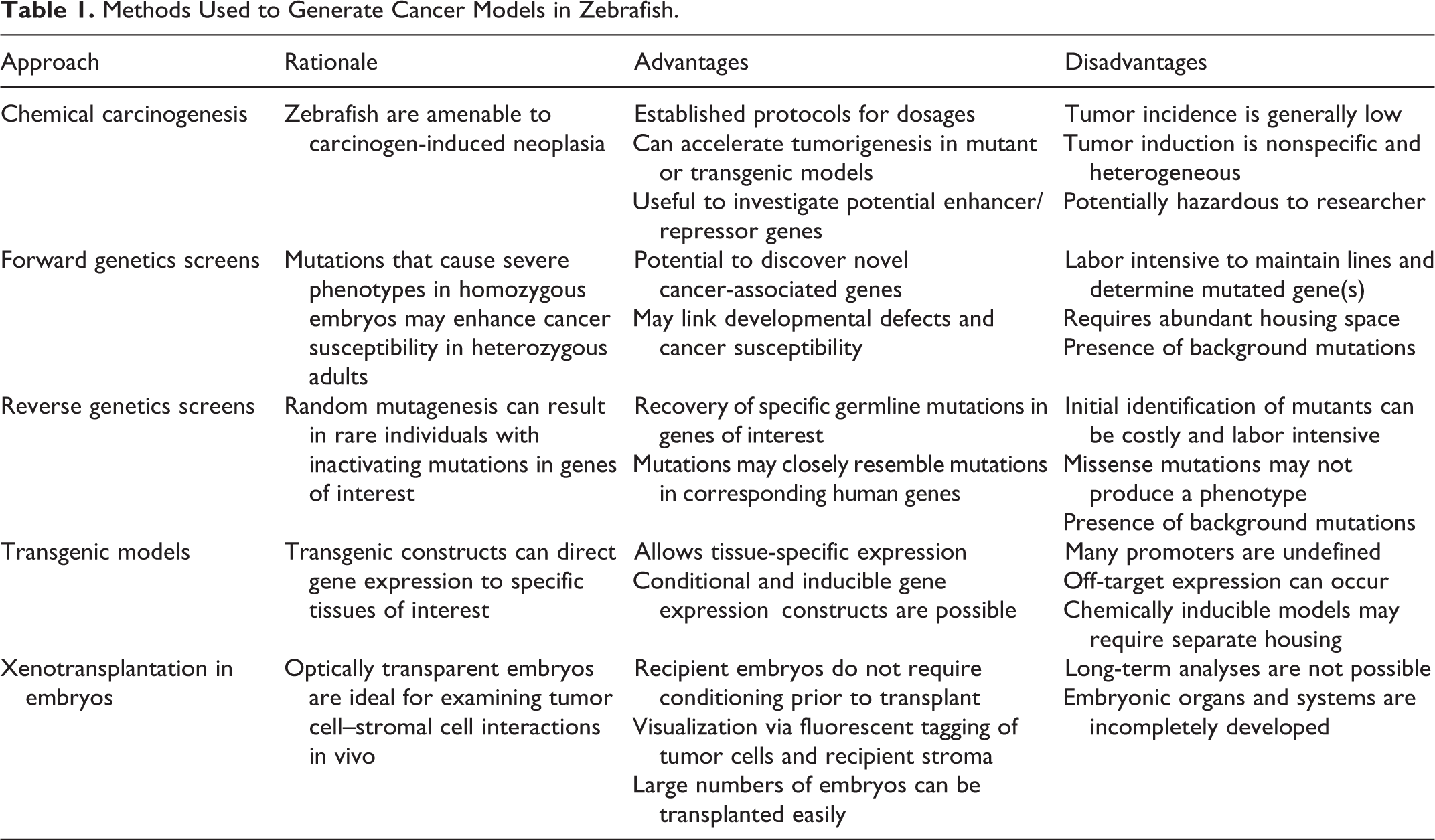
Multiple chemical carcinogens have been studied in fish species to determine their efficacy in fish, to model human carcinogen exposure, and to assess environmental toxins. 7,11,56,104,105 The most common routes of carcinogen exposure in zebrafish are aqueous and dietary. 11,39,51,56,73,104,105 Additional methods used in fish species include embryo injection and topical application. 11,104 In comparison, exposure methods used in rodent models include introduction in feed or water, gastric lavage, inhalation, injection, or topical application. 43,44 Although the routes of exposure may differ in carcinogenicity studies with fish versus mammalian species, the liver is a primary target tissue for many carcinogens in both fish and rodents. 11,43,44,104
Another approach for generating cancer models in animals is through genetic manipulation. As embryonic stem cell technology is yet to be achieved in zebrafish, the generation of zebrafish lines with mutations in genes of interest has most often relied on reverse genetic screens of mutagenized zebrafish. This approach typically involves chemical or retroviral mutagenesis strategies to generate a large population of zebrafish, with each individual carrying a unique collection of randomly distributed mutations.
3,74
The offspring of each uniquely mutagenized individual can be screened for mutations in genes of interest and are used to establish a mutant line.
3,103
Mutant zebrafish lines that have been established by this method harbor mutations in important cancer susceptibility genes, including zebrafish orthologues of the human tumor suppressor genes
Creation of transgenic zebrafish lines is also possible, since 1-cell stage zebrafish embryos can be injected with plasmid or bacterial artificial chromosome (BAC) gene constructs with relative ease. 18,115 A number of technologies have been developed to increase the rate of early and efficient transgene integration. 107,108 Transgenic lines have typically relied on zebrafish promoters driving expression of human or mouse genes, 59,60,87,88 although in some cases, the zebrafish orthologue has been used succesfully. 78 The adaptation of conditional or inducible strategies for gene expression in zebrafish has greatly expanded the possibilities for genetic manipulation in this species. 37,57,63
Xenotransplantation of human or mouse cancer cells into zebrafish embryos has also proven to be a useful tool in cancer research (reviewed by Amatruda and Patton 2 ). As zebrafish embryos do not have a fully developed immune system, conditioning of recipients is not required in these studies. 80 Because of the optical transparency of zebrafish embryos and the availability of multiple zebrafish lines that express fluorescent proteins in normal tissues, xenotransplantation experiments can allow detailed in vivo examination of cell-cell and cell-stromal interactions within the context of neoplastic cell survival, migration, and invasion. 80 Xenotransplanted zebrafish embryos also offer a high-throughput system for evaluating novel drug therapies in vivo. 54 Although further discussion of this methodology is beyond the scope of this article, the zebrafish model clearly offers a unique and diverse spectrum of approaches for the study of cancer.
Before reviewing zebrafish models for human cancer, one should be aware of nomenclature conventions for zebrafish gene names and symbols, proteins, and genotypes. Guidelines for zebrafish nomenclature are available from the Zebrafish Information Network (ZFIN), an online database for zebrafish researchers.
106
In brief, zebrafish gene names and gene symbols are lowercase and italicized and are named after the mammalian orthologue whenever possible. The protein symbol matches the gene symbol, but is not italicized, and begins with an uppercase letter. For mutant zebrafish lines, the mutant allele is identified by a unique line designation code that indicates the institution of origin. Transgenic lines are generally identified by “
Models of Cutaneous Neoplasia
A basic understanding of zebrafish skin anatomy facilitates the discussion of zebrafish models of cutaneous neoplasia. The integument of fish includes a nonkeratinized stratified squamous epidermis and underlying dermis. 71,72 Teleost species (including zebrafish) have overlapping elasmoid scales that are covered by the epidermis in the trunk region. 72 Within the epidermis are keratinocytes, goblet cells, and alarm cells. 71 Pigmented cell populations, including melanocytes, iridiphores, and xanthophores, are predominantly located in the hypodermis between the dermal stratum compactum and underlying muscle layer. 40 As in mammals, pigmented cells in fish are of neural crest origin and are designated during embryogenesis. 13
The majority of zebrafish models for cutaneous neoplasia have focused on melanocyte-derived tumors. Multiple models for benign nevus and cutaneous melanoma exist and have been derived by targeted expression of human oncogenes in melanocytes (Table 2 and Fig. 1). Most of these models use the melanocyte-specific promoter
Zebrafish Models of Cutaneous Neoplasia.
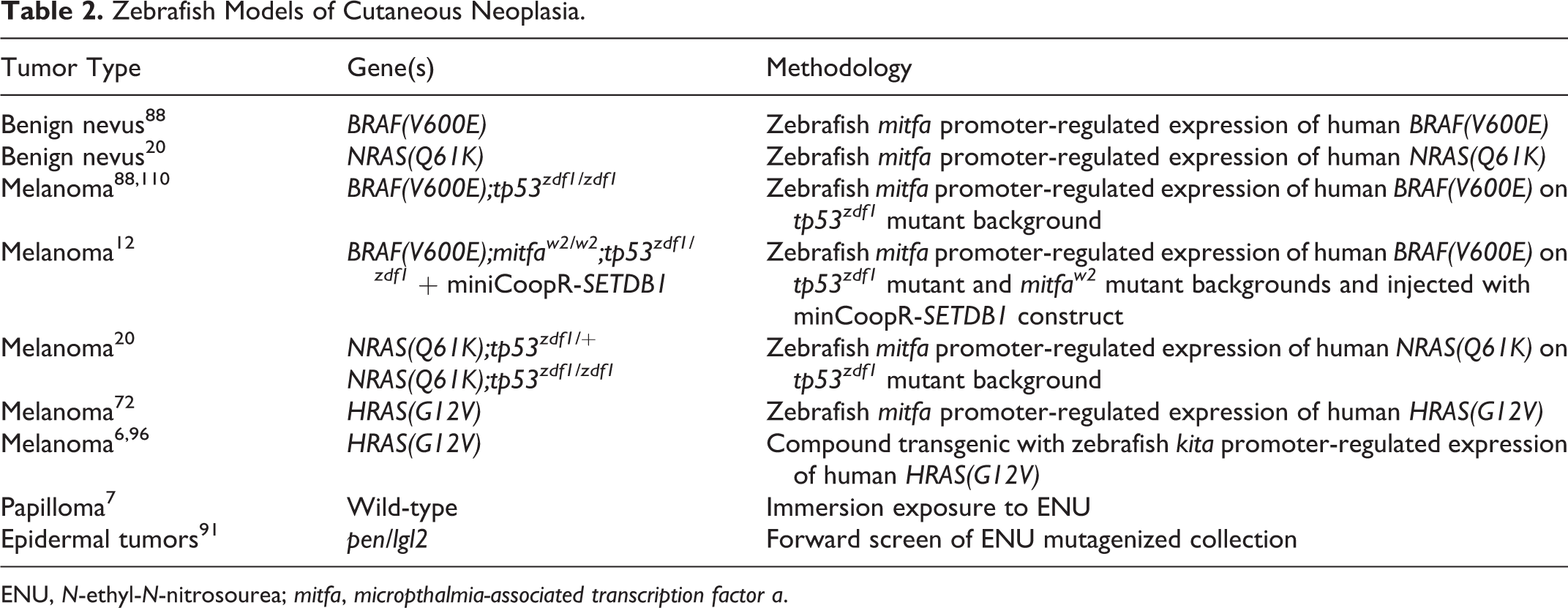
ENU,
Zebrafish models for melanoma demonstrated that expression of a mutant allele of human
Abnormal stripe patterning and irregular areas of cutaneous pigmentation (hyperpigmentation) are commonly observed in zebrafish models of melanoma. 20,72,88,96 Areas of aberrant pigmentation were observed in zebrafish embryos as early as 3 days postfertilization (dpf) in some models. 96 Benign nevi arise in adult zebrafish as flat, discrete to coalescing patches that are heavily pigmented, randomly distributed over the head and body, and confined to the epidermis. 20,88 In comparison, zebrafish harboring melanomas present with variably nodular masses that exhibit invasive behavior. 20,72,88,96 Although the majority of zebrafish melanomas appear heavily pigmented grossly, neoplastic cells show variable degrees of pigmentation by histology. 20,72,88,96 Zebrafish melanomas are transplantable into sublethally irradiated recipients, 20,88 although benign nevi are not. 88 Additional characterization demonstrated that zebrafish melanoma cells are highly aneuploid, 88 are highly proliferative, 72,96 and exhibit immunoreactivity for tyrosinase, Melan-A, S100, and HMB-45, 96 markers that are useful in the diagnosis of human melanoma. 16
Zebrafish models of melanoma represent a powerful tool for multiple downstream experiments that delve more deeply into melanoma pathogenesis. Comparisons of microarray data from zebrafish and human melanoma samples have demonstrated similar patterns of gene expression and enrichment profiles, suggesting a similar biology among these tumors. 6,20 In a recent report, multiple candidate genes that are located in a recurrently amplified region of chromosome 1 in human melanoma were tested in a zebrafish melanoma model. 12 This study showed that overexpression of the histone methyltransferase SETDB1 significantly accelerated zebrafish melanoma formation and induced transcriptional dysregulation of multiple genes. 12 A zebrafish melanoma model has also revealed the importance of neural crest developmental pathways in melanoma formation. 110 Through a chemical screen for small-molecule inhibitors of neural crest development in zebrafish embryos, White et al 110 found that an inhibitor of neural crest progenitor cells could abrogate melanoma cell proliferation in vitro and in vivo.
Nonmelanoma models for cutaneous tumors in zebrafish are few (Table 2), although the convenience of this animal model for studying tumors of the skin is evident. In one study, adult zebrafish that were exposed to the mutagen
Models of Muscular, Adipocytic, and Vascular Neoplasia
Muscular and vascular tumors are described at low frequency in several zebrafish models with a diverse spectrum of cancer susceptibility.
48,98,105
However, there are only a small number of zebrafish models with a specific propensity for muscular, adipocytic, or vascular neoplasia (Table 3). Rhabdomyosarcoma and liposarcoma can be induced in transgenic zebrafish by targeting the Ras-Raf-MEK-ERK or PI(3)K-Akt pathways, respectively, whereas hemangiosarcoma in zebrafish is associated with haploinsufficiency for
Zebrafish Models of Muscular, Adipocytic, and Vascular Neoplasia.

ENU,
The
Embryonal rhabdomyosarcoma developed in zebrafish that were injected at the 1-cell stage with a human
In a similar approach, the zebrafish
Although rhabdomyosarcoma and liposarcoma models were developed by transgenic approaches, a model for hemangiosarcoma in zebrafish was recently described in zebrafish with germline mutations in the zebrafish orthologue of the tumor suppressor gene
Zebrafish models for rhabdomyosarcoma and liposarcoma have provided interesting insights into the biology of these diseases in humans. On the basis of the finding that Ras activation resulted in a high incidence of rhabdomyosarcoma in zebrafish, Langenau et al 59 compared human and zebrafish rhabdomyosarcoma by microarray analysis. These studies revealed conserved gene signatures for rhabdomyosarcoma in both species and showed that Ras pathway activation is a common feature of human rhabdomyosarcoma. 59 Similarly, the discovery that Akt activation drives liposarcoma development in zebrafish led to the finding that the Akt pathway is activated in about a third of clinical liposarcoma specimens and that inhibition of the PI(3)K-Akt-mTOR pathway in human liposarcoma cell lines impairs viability. 38 These findings demonstrate that zebrafish can be a useful and relevant comparative animal model for human mesenchymal tumors.
Models of Intestinal, Hepatic, and Exocrine Pancreatic Neoplasia
Although the basic structures and functions of the intestine, liver, and exocrine pancreas are similar between zebrafish and mammals, some anatomical features in zebrafish are of note. As zebrafish are agastric, the esophagus leads directly into the intestine. 71 The intestine is a simple tubular structure that lacks distinguishable small and large intestinal segments, although differences can be observed in the density of goblet cells and morphology of the mucosal epithelium along its length. 71 The liver is composed of 3 lobes and a gallbladder. 71 Unlike mammals, hepatic cords, lobules, and portal triads are not clearly defined in the zebrafish liver, and Kupffer cells are absent. 71 The pancreas in zebrafish is anatomically distinct from the liver but does not form a discrete organ. 71 Clusters of exocrine pancreatic tissue are present along the intestinal tract; similar to mammals, pancreatic acini are composed of secretory cells containing bright eosinophilic granules. 71
Zebrafish that are heterozygous for a germline mutation in the tumor suppressor gene
Zebrafish Models of Intestinal, Pancreatic, and Hepatic Neoplasia.

DBP, dibenzo[a, l]pyrene; DMBA, 7,12-dimethylbenz[a]anthracene; ENU,
Intestinal hyperplasia, dysplasia, and neoplasia have been described at variable frequency in several other transgenic and mutant zebrafish lines,
61,73,98
as well as in zebrafish exposed to carcinogens.
105
Importantly, malignant intestinal neoplasia can develop in zebrafish infected with the nematode parasite
Zebrafish models for hepatic neoplasia have employed both genetic manipulation and carcinogen exposure. Carcinogens are relatively reliable for the induction of hepatic neoplasia and have been useful in defining conserved gene expression signatures in human and zebrafish hepatic tumors. 56,105 However, the lack of specificity for hepatic tumorigenesis in carcinogenesis studies and the desire for a stable and heritable cancer susceptibility phenotype have spurred the development of transgenic zebrafish models for hepatic cancer.
Several transgenic models for hepatocellular carcinoma (HCC) have been described, each using the liver-specific promoter
A transgenic approach has also been applied for developing 2 zebrafish models for exocrine pancreatic carcinogenesis (Table 4). In both models, an
Models of Hematopoietic and Lymphoid Neoplasia
A relatively large body of work is devoted to zebrafish models for hematopoietic and lymphoid neoplasia (Table 5), including the first report of a line of transgenic tumor-bearing zebrafish. 60 Several key anatomic and histologic differences between zebrafish and mammals are relevant for the study of these diseases. Unlike mammals, zebrafish bones do not have a medullary cavity containing hematopoietic tissue; instead, hematopoiesis occurs in the splenic stroma and renal interstitium. 71 Zebrafish lack lymph nodes but do possess a thymus with cortical and medullary organization that supports lymphocytic maturation. 71 Peripheral blood components include erythrocytes and thrombocytes (both nucleated in teleost species), 46,71 neutrophilic and eosinophilic granulocytes, 8,17,65 monocytes, 75 and B and T lymphocytes. 71,75 Lymphocytes are the predominant circulating leukocyte in zebrafish, representing 71% to 92% of the population. 75
Zebrafish Models of Hematopoietic, Lymphoid, and Small Round Blue Cell Neoplasia.

AML, acute myeloid leukemia; B-ALL, B-cell acute lymphocytic leukemia; ENU,
The majority of transgenic models for T-cell lymphoma/leukemia in zebrafish make use of the
More complex transgenic approaches have modeled the progression from T-LBL to T-ALL in zebrafish. Use of conditional promoters in compound transgenic zebrafish have shown that zebrafish develop T-LBL, characterized by thymic enlargement and local infiltration, which progresses to T-ALL, characterized by widely disseminated disease and an abundance of malignant cells in the kidney marrow.
27,28
These types of complex genetic manipulations have permitted investigation into the mechanisms of malignant progression in zebrafish and human lymphoid neoplasia. Overexpression of the antiapoptotic gene
Zebrafish are also useful in studying acute myeloid leukemia (AML), although there are fewer models for this disease (Table 5). Unlike models for T-cell malignancies, zebrafish models for AML have generally relied on the expression of human fusion genes in myeloid cells. Several of these models have demonstrated that expression of these fusion genes induces disturbances in the myeloid compartment, including redirection of progenitor cell fate from erythroid to myeloid lineage and expansion of progenitor and myeloid compartments in the marrow.
10,83,116
Transgenic zebrafish expressing the human fusion gene
Few other models for lymphoid or other small round cell tumors exist in zebrafish (Table 5). A single zebrafish model for B-cell acute lymphocytic leukemia (B-ALL) has been described, in which the human TEL-AML1 fusion was expressed under ubiquitous promoters. 94 Transgenic zebrafish developed lymphoblastic leukemia at low incidence, and neoplastic cells appeared to be arrested at a pre–B-cell differentiation stage. 94 In addition, a zebrafish model transgenic for the human EWS-FLI1 fusion was recently found to develop small round blue cell tumors at low incidence. 62
Models of Neural and Neuroendocrine Neoplasia
Tumors of the peripheral nervous system are not uncommon in zebrafish cancer models (Table 6). A number of mutant zebrafish lines develop malignant spindle cell tumors that are histologically reminiscent of malignant peripheral nerve sheath tumors (MPNSTs). 5,9,26,85,92 Curiously, these cancers arise with greatest frequency in zebrafish lines that harbor mutations in tumor suppressor genes (Table 7), suggesting that the cancer spectrum associated with these types of mutations may be more limited in zebrafish compared with humans. MPNSTs in these mutant lines typically arose in zebrafish older than 1 year. 5,9,85,92
Zebrafish Models of Neural and Neuroendocrine Neoplasia.
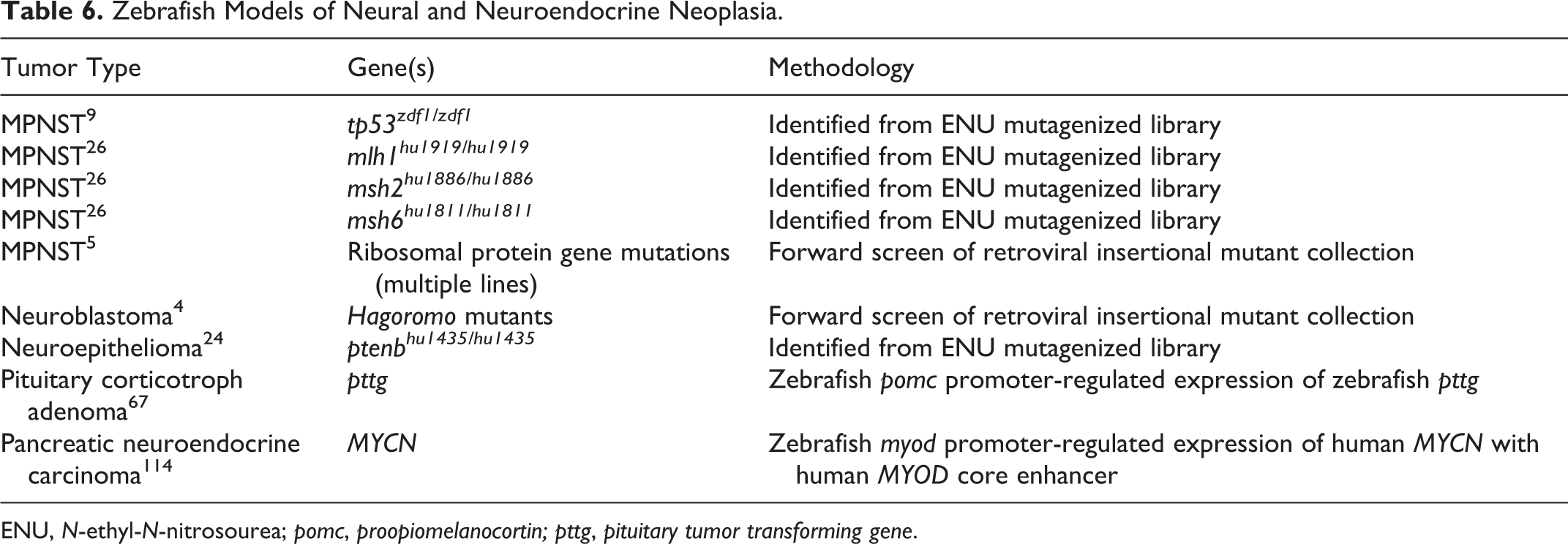
ENU,
Histologic features of these tumors are similar between mutant models. Neoplastic cells are typically spindloid to variably epithelioid with oval to tapered nuclei, and they are arranged in fascicles, stacks, and whorls (Fig. 5). 5,9,26,85 These highly invasive tumors can arise in any of 3 distinct sites: the retrobulbar region, the coelom, and the flank or tail. 9,26,85,99 Interestingly, MPNSTs in zebrafish are highly aneuploid and show nonrandom regions of chromosomal amplifications and losses, even when tumors are derived from different mutant zebrafish lines. 118
Classification of these tumors as MPNSTs has been somewhat controversial, as these tumors reportedly do not express S100 or PGP9.5,
85
markers that have been used for identification of human MPNSTs.
30,41
However, the report that zebrafish MPNSTs do express
In a forward genetic study, 4 mutant zebrafish lines (designated as
There are very few models for tumors of neuroendocrine origin in zebrafish (Table 6). A single zebrafish model for pituitary corticotroph adenoma has been described.
67
In this model, overexpression of the
A model for pancreatic neuroendocrine carcinoma in zebrafish has also been described, although a stable transgenic line has not yet been established (Table 6). Zebrafish that overexpressed
Much remains to be explored in the development of transgenic or mutant zebrafish models for tumors of neural and neuroendocrine origin. The development of reporter lines defining functional promoter regions for the neural-specific genes
Models of Gonadal Neoplasia
There are a small number of zebrafish models for reproductive tumorigenesis, all of which show gonadal tumor susceptibility in males (Table 8). Gonadal tumors in male zebrafish have also been observed at variable frequency in several studies reporting a spectrum of tumor susceptibility. 73,98,105 In contrast, ovarian tumors are rarely reported in transgenic, mutant, or carcinogen-exposed female zebrafish. 73,77
Zebrafish Models of Gonadal Neoplasia.
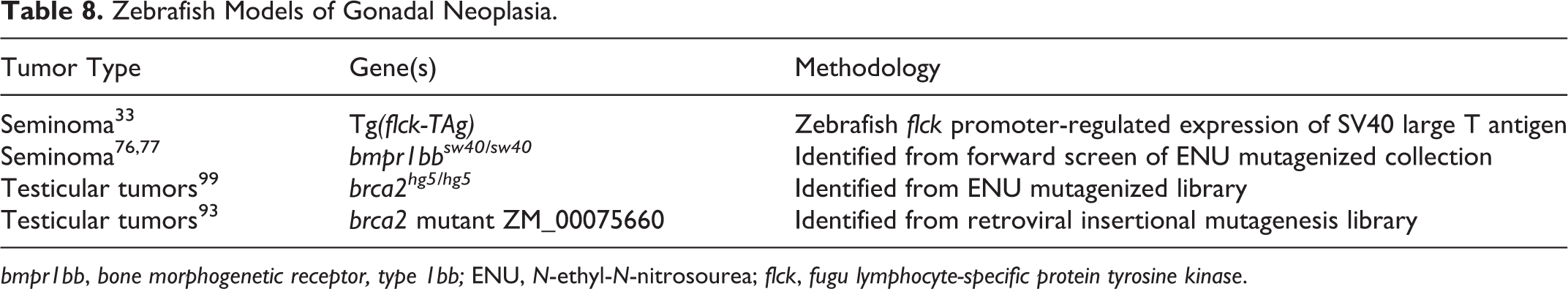
Specific susceptibility to testicular germ cell tumors (seminomas) has been described in a transgenic zebrafish model
33
and in a mutant zebrafish line.
77
In
Testicular tumors have also been reported in 2 different zebrafish lines with mutations in
Zebrafish Cancer Models: Future Directions
There are currently numerous undefined or underexplored areas for cancer research in zebrafish models, some of which have been noted in this review. Models for cancers of the brain, bone, and ovary remain to be developed; many other types of cancer models, including nonmelanoma cutaneous tumors, certain mesenchymal tumors, and endocrine tumors, are thus far quite limited. Although the absence of some tissue types in fish species, such as breast and prostate, limits the usefulness of zebrafish for investigating cancers of these organs, the zebrafish model has nonetheless become a powerful tool in comparative biomedical research. New methods for creating gene modifications or disruptions, such as zinc finger nucleases and transcription activator-like effector nucleases (TALENS), are being applied in zebrafish to generate targeted, heritable genetic mutations. 21,42,70,95 Recent technological advances for analyzing tumor development in zebrafish in vivo, including magnetic resonance imaging, 49 ultrasound biomicroscopy, 34 and high-throughput fluorescent macroscopic imaging, 101 reflect the development and adaptation of increasingly sophisticated systems tailored to zebrafish research.
The successful development of numerous zebrafish models for human cancer clearly demonstrates that mechanisms for neoplastic transformation and tumor progression are conserved across relatively wide evolutionary distances. Although it is significant that many features of cancer development in zebrafish faithfully recapitulate important aspects of their human counterparts, the usefulness of zebrafish as a cancer model extends far beyond mere phenotypic or genotypic replication of disease. Multiple studies have shown that judicious and creative use of zebrafish cancer models can lead to important insights into disease pathogenesis and therapy. Such approaches may range from small-molecule chemical screens to high-throughput investigations of potential enhancer/repressor genes to transplantation or xenotransplantation experiments. A key strength of the zebrafish model in cancer research is the ability to seamlessly transition between embryonic and adult animal studies, providing coordinated exploration of critical developmental, transformative, and pathological events in vivo. These attributes ensure that zebrafish models for human cancer are here to stay.
Footnotes
Acknowledgements
The author thanks Dr Dennis Hickstein, Dr Robert West, and Ms Lisa Embree for critical review of the manuscript and Dr Leonard Zon, Dr Charles Kaufman, Dr Colleen Albacker, Dr Narie Storer, Dr Steven Leach, Dr Shu Liu, Dr David Langenau, and Dr Jessica Blackburn for contributions of histologic images.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Intramural Research Program of the US National Institutes of Health, National Cancer Institute, Center for Cancer Research.

