Abstract
Over the course of an approximately 11-month period, an outdoor, freshwater, mixed species, recirculating, display system at a public aquarium experienced intermittent mortalities of channel catfish (Ictalurus punctatus) and blue catfish (I. furcatus). Catfish acutely presented for abnormal buoyancy, coelomic distention, and protein-rich coelomic effusion. Gross lesions typically involved massive coelomic distension with protein-rich effusion, generalized edema, and gastric hemorrhage and edema. Microscopically, primary lesions included renal tubular necrosis, gastric edema with mucosal hemorrhages, and generalized edema. Aerobic culture and virus isolation could not recover a consistent infectious agent. Intracoelomic injection of coelomic effusion and aspirated retrobulbar fluid from a catfish into naïve zebrafish (bioassay) produced peracute mortality in 3 of 4 fish and nervous signs in the fourth compared with 2 saline-injected control zebrafish that had - no mortality or clinical signs. Kidney tissue and coelomic effusion were submitted for gas chromatography tandem mass spectrometry by multiple reaction monitoring against laboratory standards, which detected the presence of multiple pyrethroid toxins, including bioallethrin, bifenthrin, trans-permethrin, phenothrin, and deltamethrin. Detection of multiple pyrethroids presumably reflects multiple exposures with several products. As such, the contributions of each pyrethroid toward clinical presentation, lesion development, and disease pathogenesis cannot be determined, but they are suspected to have collectively resulted in disrupted osmoregulation and fluid overload due to renal injury. Pesticide-induced toxicoses involving aquarium fish are rarely reported with this being the first description of pyrethroid-induced lesions and mortality in public aquarium-held fish.
Keywords
Pyrethroid insecticides are synthetic analogs of pyrethrins, which naturally occur in chrysanthemum flowers. The mechanism of action of pyrethroid insecticides involves blocking sodium channels within the nervous system resulting in overactivation of neurons. 14 Commercial products containing pyrethroid and pyrethrins have largely replaced the use of organophosphates and organochlorines in insecticides due to their comparatively lower toxicity in humans and other animals. Regulated by the Environmental Protection Agency, these insecticides still cause toxicosis in a variety of species ranging from invertebrates to fish, birds, and mammals.4,18 Invertebrates and fish are particularly sensitive to toxicosis compared with other animal groups. 18
Catfish aquaculture represents the largest, most important sector of aquaculture in the United States with a total economic impact of over US$2 billion dollars. 11 Channel catfish (Ictalurus punctatus), blue catfish (I. furcatus), and their hybrid cross are grown in the industry with channel and hybrid catfish making up the majority of production. 11 Infectious diseases and water quality–related issues result in significantly lost production performance and fish mortality in the industry. 11 The vast majority of catfish research centers around optimizing growth performance and minimizing disease impacts for food production and economic purposes.11,16,24 Reports of disease investigations involving catfish from public or private aquarium facilities are limited.
In addition to infectious diseases, catfish may experience a variety of toxicoses in US-based aquaculture settings. Water quality–related toxicoses, such as ammonia poisoning and nitrite toxicity, are relatively common in the ponds.8,11,40 Visceral toxicosis of catfish is caused by Clostridium botulinum type E neurotoxin and results in large-scale mortalities during outbreaks.15,23 Plumb and Richberg reported increased serum levels of endrin, toxaphene, and dichlorodiphenyltrichloroethane (DDT) during a continuous winter mortality event resulting from aerial pesticide exposure the previous summer. 35 Microcystins and mycotoxins are widely studied in channel catfish with occasionally conflicting reports on growth performance impacts and mortality.20,26,28-31,37,50 Examples of other compounds investigated for toxicity under natural or experimental conditions in channel catfish include copper sulfate, cyclopiazonic acid, potassium permanganate, and methyl mercury.17,19,22,39
This report details the clinical findings, pathology, and diagnostic investigation into a prolonged mortality event involving channel and blue catfish exposed to pyrethroid insecticides in an outdoor, recirculating display system at a public aquarium.
Methods
Case History
Adult channel catfish and blue catfish were obtained from a commercial aquaculture facility and quarantined for an 8-week period prior to being moved into a display system. The catfish were displayed in a 90,000 gallon, outdoor, freshwater, mixed species recirculating system with alligator gar, longnose gar, spotted gar, largemouth bass, smallmouth bass, white crappie, black crappie, freshwater drum, sunfish, bluegill, yellow-bellied sliders, red-eared sliders, Alabama red-bellied slider, Buffalo suckers, and an alligator snapping turtle. Intermittent, acute presentations involving 12 channel and blue catfish occurred during a period spanning late June 2021 to early December 2021 and May 2022. Reported clinical signs included abnormal behavior and buoyancy, inability to orient correctly in the water column, acute coelomic distension, cutaneous abrasions, exophthalmia, hyphema, increased respiratory effort, lethargy, and facial swelling. Ultrasound examination commonly illustrated the presence of abundant coelomic effusion not seen at previous health assessments.
Four catfish throughout the mortality event received necropsies according to standard methods followed by histological examination and ancillary diagnostics. 33 Case 1 was an adult, female channel catfish that presented for abnormal buoyancy and severe coelomic distension due to effusion. The fish was treated with dexamethasone SP at 1 mg/kg and intravenous enrofloxacin without response. Case 2 was an adult, male channel catfish that presented for an approximately 3.0 × 3.0 cm abrasion on the left lateral body wall extending from the dorsal fin to the anal fin. The only other reported gross finding was the presence of coelomic effusion. Case 3 was an adult, blue catfish that presented due to upside-down orientation in the water; fluid-distended coelom; exophthalmia, anisocoria, and ocular hemorrhage; cutaneous abrasions; and unilateral facial swelling and erythema. Case 4 was an adult, male blue catfish that initially presented for cutaneous abrasions on the dorsum just caudal to the dorsal fin and bilaterally on the dorsal aspect of the pectoral fins. The fish was treated with 6.6 mg/kg ceftiofur. Three weeks later, the fish acutely presented with abnormal buoyancy, lethargy, nonresponsiveness, massive coelomic distension, generalized edema, and vent prolapse. All fish either expired naturally or were euthanized via an overdose of buffered tricaine methanesulfonate (MS-222, Syndel Inc.) within 24 hours of coelomic effusion onset.
Following the final case (case 4), clinicians and animal care staff were notified about pesticides being regularly applied to landscaping adjacent to the affected system. These pesticides were applied without approval from the veterinarians or animal care staff and were not on a list of approved chemicals provided by the veterinary staff to the landscape management company. Review of limited records from the landscape management company from April to June 2022 showed one or more applications of various insecticides and herbicides with active ingredients, including bifenthrin, acephate, glyphosate, and s-metolachlor. New catfish added to the system since cessation of pesticide applications and discontinuing service with the landscape management company in July 2022 have not had any significant health concerns or mortalities.
Sample Collection and Processing
Representative fluid and tissue samples from each fish were either frozen at –20°C or preserved in 10% neutral-buffered formalin. Formalin-fixed samples were processed for histology using standard methods involving progressive tissue dehydration, paraffin embedding, sectioning at 5 µm thickness, and staining with hematoxylin and eosin. 45 Select, unstained slides were stained with Giemsa or Gram stains. Aerobic cultures were performed on chilled coelomic effusion and frozen-thawed kidney tissue streaked onto tryptic soy agar plates infused with 5% sheep’s blood and incubated at 25°C and 37°C. Kidney and coelomic effusion fluid from case 4 were inoculated onto 3 cell lines for virus isolation (channel catfish ovary, brown bullhead, and bluegill fry-2).
Toxicologic Analysis
Coelomic effusion and retrobulbar fluid aspirated at necropsy from case 4 were used for a zebrafish bioassay to investigate the presence of a toxin. 6 Briefly, 6 zebrafish were inoculated intracoelomically with 10 µL of coelomic effusion fluid, retrobulbar fluid, or saline and monitored for behavioral abnormalities and mortality. All fish were euthanized via an overdose of buffered MS-222 following 10 minutes of observation regardless of mortality status. Zebrafish were preserved in 10% neutral-buffered formalin and processed for histology as described above.
Kidney tissue and coelomic effusion from case 4 were submitted to the Michigan State University Veterinary Diagnostic Laboratory for gas chromatography mass spectrometry (GC-MS) general toxin screen, which was performed in accordance with procedures from this laboratory, as described elsewhere.3,4 Both sample types initially screened negative for a variety of pesticides, herbicides, illicit drugs, pharmaceuticals, and industrial compounds. The increased sensitivity of fish species to various pesticides may result in clinically significant pesticide concentrations below the detection limit of the initial GC-MS screen; therefore, a subsequent gas chromatography tandem mass spectrometry (GC-MS/MS) analysis using multiple reaction monitoring was utilized. A synthetic pyrethroid pesticide reference material containing 18 pyrethroid compounds (GC Multiresidue Pesticide Standard #6 100 μg/mL in toluene) was obtained from Restek (Bellefonte, PA). Analytes included tefluthrin, transfluthrin, antraquinone, bioallethrin, resmethrin, tetramethrin, bifenthrin, phenothrin, lambda-cyhalothrin, acinathrin, permethrin (cis and trans), cyfluthrin, cypermethrin, flucythrinate, fevalerate, fluvalinate, and deltamethrin. Kidney and coelomic fluid extracts were compared with neat standards prepared in toluene at concentrations of 1, 10, 100, 1,000, 10,000, and 100,000 ng/mL.
Analyte separation was achieved using an Agilent Technologies 7890A GC system coupled with an Agilent 7000 GC-MS triple quadrupole detector (Agilent, Santa Clara, CA). The GC separation was conducted with a Phenomenex Zebron ZB-Multiresidue column (45 m × 250 μm × 0.25 μm) with a 4 μL injection into an inlet operated in splitless mode. An Agilent 5062-3587 split/splitless glass cylinder with single taper liner was used with the glass wool removed. The inlet had a pressure of 13.4 psi with an initial temperature of 255°C held for 20 minutes and then ramped at 200°C/min to 280°C. Oven temperature began at 80°C and was held for 0.5 minutes, and then increased sequentially at (1) 40°C/min to 210°C and held for 5 minutes, (2) 10°C/min to 235°C and held for 5 minutes, (3) 10°C/min to 275°C and held for 5 minutes, and (4) 10°C/min to 300°C and held for 20 minutes for a total run time of 56 minutes. Helium carrier gas flow was 1.7 mL/min. The triple quadrupole mass analyzer was operated in negative electron ionization mode at 70 eV with a source temperature of 230°C. Nitrogen gas was used as the collision gas at 1.5 mL/min and helium was used as a quench gas at 2.25 mL/min. Multiple reaction monitoring was initiated after a 6-minute solvent delay. Data acquisition and processing was performed with MassHunter software (Agilent, Santa Clara, CA). Multiple reaction monitoring transitions and retention times for the 1000 ng/mL synthetic pyrethroid pesticide standard are listed in Supplemental Table S1. Fluvalinate was excluded from analysis due to poor chromatographic performance. Control samples from clinically healthy, adult, laboratory reared channel and blue catfish were found to be negative for synthetic pyrethroid pesticides to a reporting limit of 10 ppb.
Results
Clinical Pathologic Findings
For case 1, evaluation of the coelomic fluid identified the presence of leukocytes and a total protein of 4 g/dL. For case 3, skin scrape and gill clip did not illustrate the presence of significant pathology or infectious agents. In-house bloodwork revealed a packed cell volume of 15% and a total protein of 1.8 g/dL. Total protein of the coelomic effusion was 1.7 g/dL. Blood chemistry revealed a markedly elevated creatinine, slight hypoglycemia, hypophosphatemia, hyponatremia, hypochloremia, hyperalbuminemia, hyperbilirubinemia, hypocholesterolemia, elevated alanine aminotransferase, and decreased alkaline phosphatase. 38 Coelomic fluid electrolytes were comparable with peripheral blood values. A complete blood count demonstrated a neutrophilia with toxic change.
Gross and Microscopic Findings
At necropsy, the prominent finding across cases consistently involved massive coelomic distension due to abundant, straw-colored, transparent effusion which clotted on the table (Fig. 1a, b). Locally extensive to generalized edema contributed to exophthalmia, facial swelling, and vent prolapse (Fig. 1a). Other external findings included cutaneous abrasions and hyphema. Internally, the stomach wall was expanded by clear, translucent fluid and covered by mucosal petechial hemorrhages (Fig. 1c). Two fish (cases 1 and 4) each had small, light red nodular masses embedded within the mesentery. Other internal findings included swollen mottled kidneys (Fig. 1d), gall bladder distension, and cerebral and pericardial effusion. There were no significant findings on skin scrapes or gill clips.
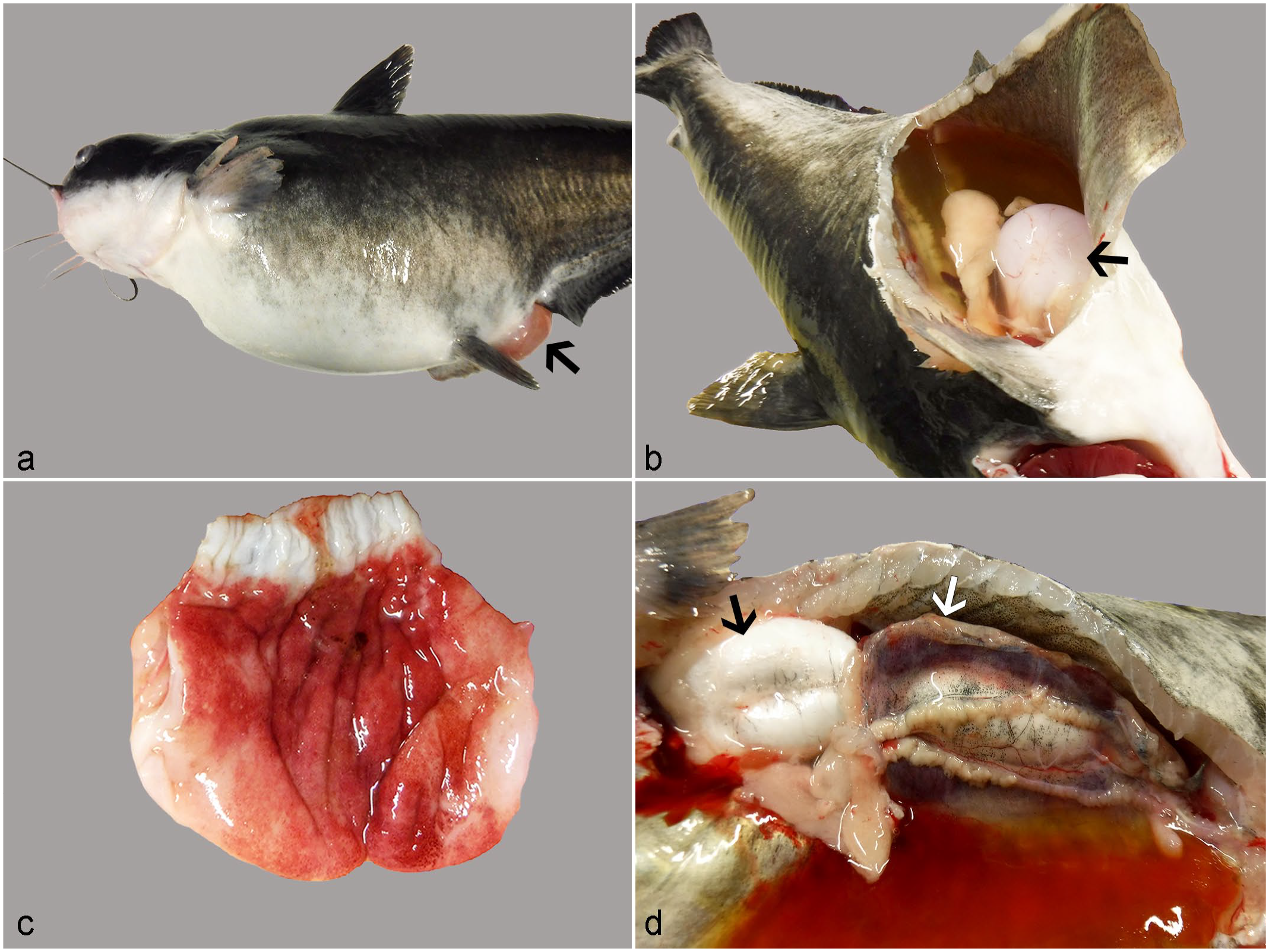
Gross lesions associated with pyrethroid toxicity in ictalurid catfish.
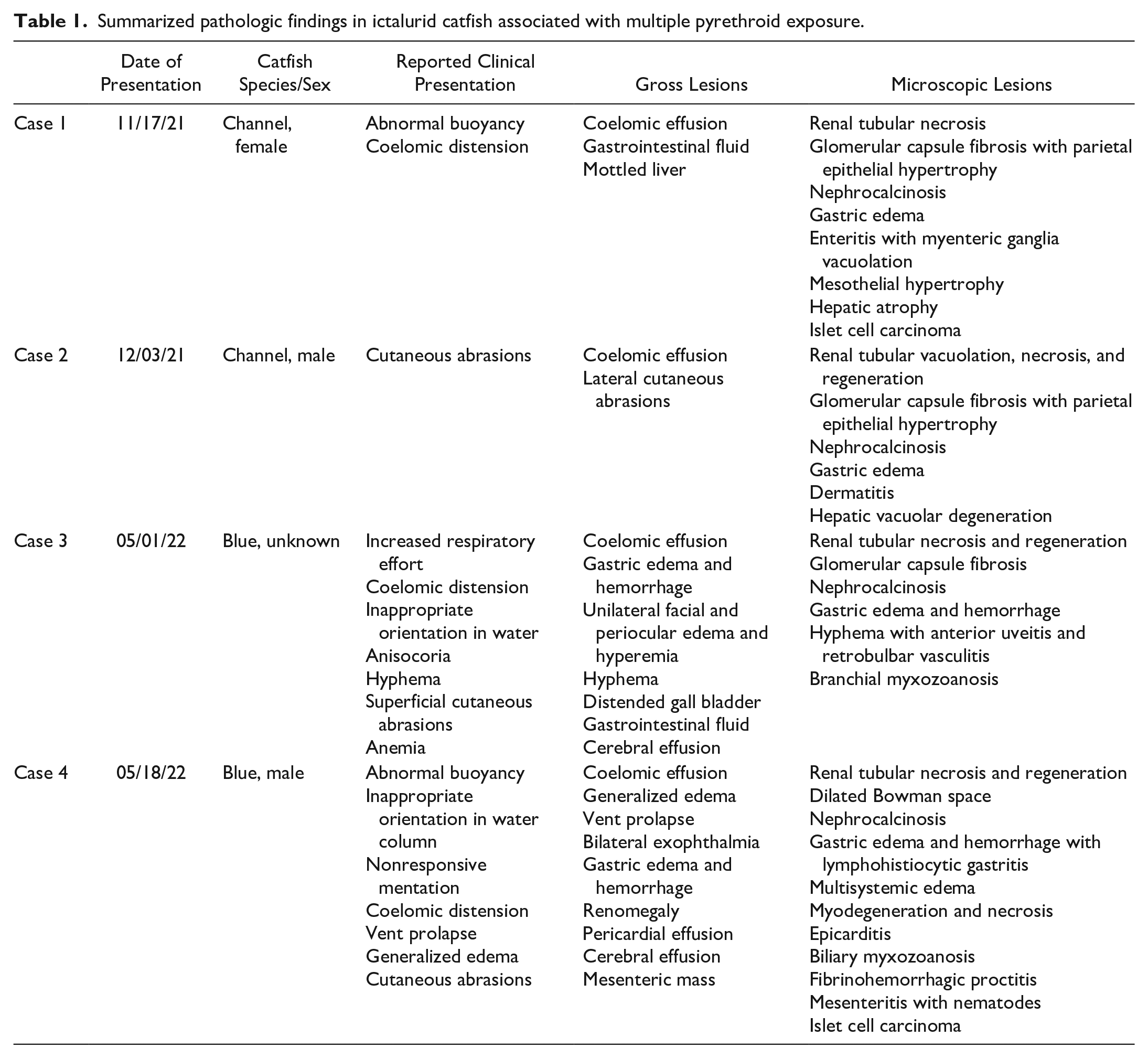
Microscopically, the main findings across animals involved moderate to severe, renal tubular necrosis and gastric edema with mucosal hemorrhage (Figs. 2a–f). Affected tubules were distended by amorphous, hypereosinophilic to mineralizing cellular debris (Fig. 2a, b). Renal epithelium lining approximately 50%–80% of proximal and, to a lesser extent, distal tubules exhibited hypertrophy, attenuation, and proliferation of small, basophilic cuboidal regenerative cells (Fig. 2b). Mildly increased numbers of immature, developing tubules were scattered throughout the posterior kidney (Fig. 2c). Within other portions of the nephron, Bowman’s spaces were frequently distended by clear space (Fig. 2d). In cases 1 and 2, Bowman’s capsules of multifocal glomeruli were thickened by fibrosis and hypertrophy of the parietal epithelium (Fig. 2e). Similar to glomeruli, collecting ducts and the mesonephric ducts were occasionally distended by clear space and low amounts of cell debris. The interstitial hematopoietic tissue was mild to moderately hyperplastic due to increased numbers of early erythroid precursors (Fig. 2a). The capsule and adjacent connective tissues were markedly expanded by edema (Fig. 2f). Other findings identified in one or more fish included myonecrosis, proctitis with vent prolapse, enteritis with edema, generalized edema, biliary myxozoanosis consistent with Myxidium sp., branchial myxozoanosis, epicarditis, mesenteritis with nematodes, ocular hemorrhage with edema, anterior uveitis and retinal degeneration, hepatic lipidosis, and erosive dermatitis. Mesenteric masses identified grossly in cases 1 and 4 were microscopically consistent with islet cell carcinomas with local and vascular invasion. Lesions are summarized in Table 1.
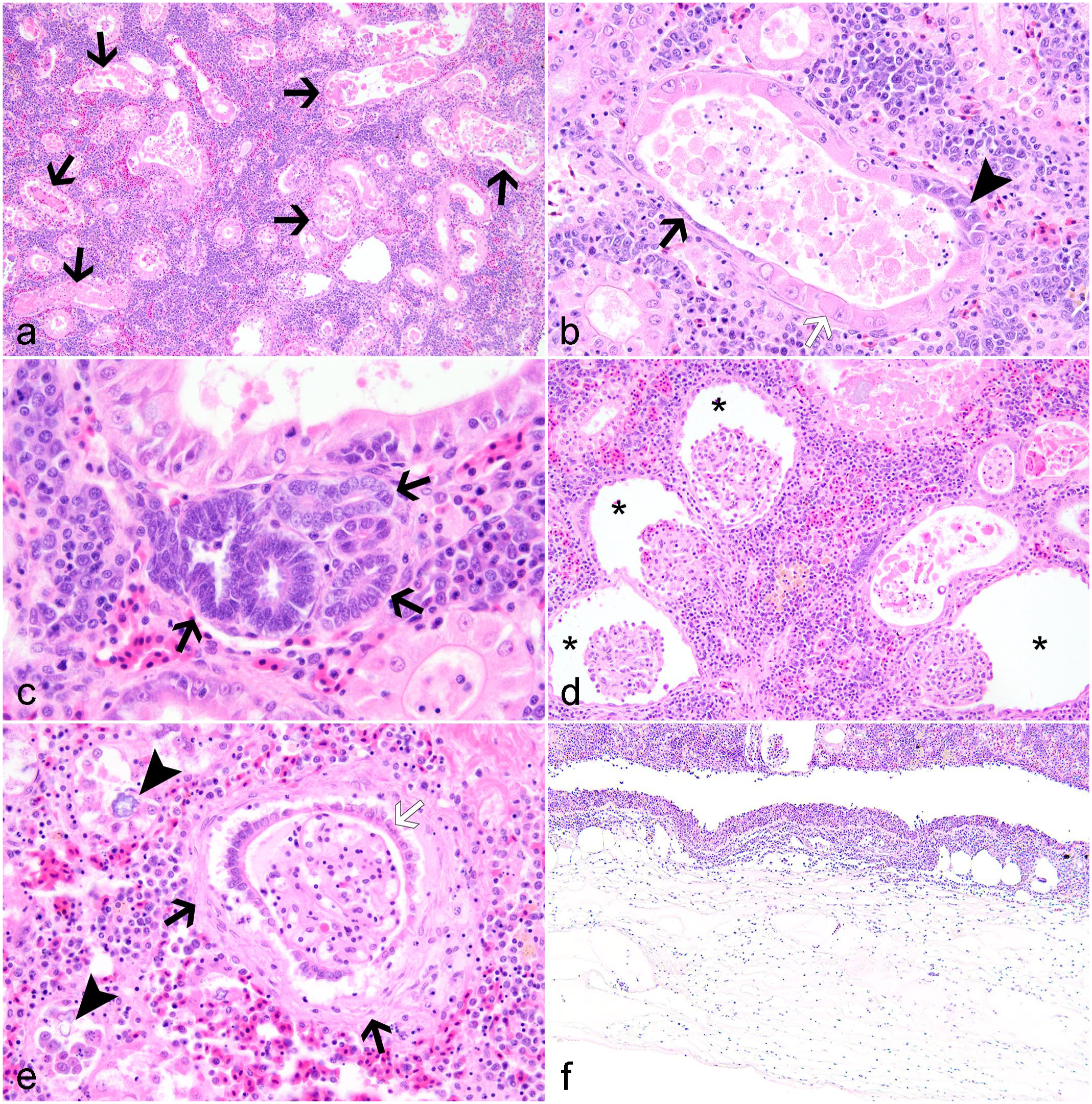
Microscopic lesions in the kidney associated with pyrethroid toxicity in ictalurid catfish. Hematoxylin and eosin.
Summarized pathologic findings in ictalurid catfish associated with multiple pyrethroid exposure.
Ancillary Diagnostic Findings
Giemsa and Gram stains did not illustrate the presence of infectious agents in histological sections of any cases. Aerobic culture yielded faint to light growth of Vibrio vulnificus from only 1 fish of the 3 tested (case 1). Similarly, cell culture failed to yield cytopathic effect on case 4.
Three of four zebrafish exposed to catfish fluids as part of the bioassay died almost immediately while the fourth exhibited severe hyperexcitability, convulsions, twitching, whirling, and tremors. The 2 saline-inoculated control zebrafish were unaffected. No lesions were identified histologically in any organs of catfish fluid or saline-inoculated zebrafish.
Initial screening of kidney tissue and coelomic effusion from case 4 did not detect the presence of organic toxicants within samples. Following repeat analysis with more sensitive and specific methodology, the pyrethroids bioallethrin, bifenthrin, phenothrin, trans-permethrin, and deltamethrin were detected in kidney tissue and/or coelomic effusion (Supplemental Table S2).
Discussion
Pyrethroid insecticides are highly toxic to a variety of fish species with effects primarily on the nervous, respiratory, and renal systems. 18 Pyrethroid uptake occurs readily across the gills given their lipophilic structure in an aqueous environment and is poorly metabolized in fish. 10 Fish possess limited capacity for metabolizing pyrethroids due to a lack of hydrolases, relying on oxidative reactions mediated by cytochrome P450 enzymes. 44 Pyrethroids may bioaccumulate in wild and cultured fish and act as endocrine disruptors.2,7,9 Factors influencing toxicity of pyrethroids in fish include fish species, age/size, biotransformation rates, water temperature, and structural features of the toxic molecule. 18 Thus, a combination of species sensitivity, limited biotransformation capability, and bioaccumulation over multiple exposures potentially contributed to the severity of the toxicity.
While descriptions of pathology associated with pyrethroid exposure are extremely limited in fish, the physiologic impacts of acute and chronic pyrethroid exposure have been studied in numerous fish species under experimental conditions. Previous work showed channel catfish fry were particularly sensitive to permethrin compared with largemouth bass fingerlings and mosquitofish (Gambusia affinis). 21 Transcriptomic sequencing of trunk kidney from deltamethrin-exposed channel catfish illustrated upregulation of genes related to apoptosis during acute stages of disease. 48 Similar to catfish, acute, sublethal exposure of goldfish to deltamethrin showed upregulation of apoptosis-related gene expression in the kidney along with evidence of immunotoxicity and gut microbiome alterations. 49 Bifenthrin- and deltamethrin-induced kidney lesions have been reported in common carp under experimental conditions.5,32 Developmental anomalies, altered behavioral, biochemical, and hematological parameters, and impaired inflammatory responses have been reported in a variety of fish exposed to bifenthrin.12,34,46 Lesions associated with phenothrin and bioallethrin are not well characterized in fish, although the 96-hour lethal concentration 50 for fish range from 17 to 200 µg/L and 9 to 90 µg/L, respectively.41,42 In contrast, the 96-hour lethal concentration 50 for bifenthrin and deltamethrin in fish is lower, ranging from 0.8 to 2.08 and 0.4 to 2.0 µg/L, respectively, classifying each of them as highly toxic to fish.27,43
Given the detection of multiple pyrethroids in samples, the exact relationships and effects of each pyrethroid on the clinical presentation, lesion development, and disease pathogenesis cannot be determined. Nontarget pyrethroid toxicosis of various organ systems in fish is increasingly attributed to oxidative stress, resulting in subsequent damage to DNA, organelles, and lipid membranes. Pyrethroids act by altering the activity and expression of several antioxidant enzymes in fish, including superoxide dismutase, glutathione peroxidase, glutathione reductase, catalase, and others, often leading to increased lipid peroxidation and DNA damage in multiple organ systems. 47 Free radical injury to renal tubular epithelial cells and circulating erythrocytes, which are particularly sensitive to free radicals, could have contributed to degeneration and cell death, which would explain the erythropoietic hyperplasia noted microscopically. 25 Exposure to pyrethroids in these fish presumptively involved multiple products and chronic, sublethal exposures resulting in bioaccumulation. While fish have various mechanisms and enzymes to combat free radicals, exhaustion of these physiologic defenses over multiple pyrethroid exposures could have predisposed to the acute presentations seen with these cases.
In addition, the pyrethroid acute mechanism of action involving blockage of sodium channels, as evidenced by immediate onset of mortality and central nervous system (CNS) signs in zebrafish used in the bioassay, could have targeted sodium-potassium pumps of renal tubular epithelial cells in the larger catfish and caused renal injury. Increased urine concentrations and excretion rates of sodium and potassium along with increased urine osmolality were reported in trout exposed to fenvalerate and attributed to disruption of sodium-potassium pumps resulting in loss of ion regulation. 1 This is consistent with the blood chemistry abnormalities noted in case 3 of this report, demonstrating ion loss and electrolyte imbalances. Regardless of the pathogenic mechanisms involved, it is evident that accumulation of these pyrethroids collectively leads to kidney injury with excessive fluid retention in the form of massive coelomic effusion, pericardial effusion, and generalized edema resulting from impaired water excretion and culminating in osmoregulatory failure. Skin lesions identified in multiple catfish presumptively reflect traumatic abrasions secondary to abnormal buoyancy and an inability to orient properly in the water column.
Despite being in a mixed species exhibit, only channel and blue catfish were affected as part of the mortality event. This is potentially related to the fact that these catfish lack dermal scales compared with other fish species in the system. Scaleless fish, such as Ictalurid catfish, are considered more susceptible to toxicoses broadly due to uptake of environmental contaminants across the skin. 38 This can include therapeutic and anesthetic agents such as formaldehyde, malachite green, and MS-222.13,33 As such, scaleless fish, including ictalurid catfish, may be more susceptible to pyrethroid-induced nephropathy compared with scaled fish, although experimental exposure challenges would be required to investigate and confirm species-specific sensitivities and pathologies.
The consistency and nature of the clinical presentations and pathology across cases, the rapid onset and nature of clinical signs in the zebrafish bioassay, results of ancillary testing for infectious agents and toxins, and documented pesticide exposure with resolution of mortalities following removal of exposure all point to a pyrethroid-induced toxic nephropathy as the main pathologic process involved in the mortality event. The cessation of mortalities following diagnosis and discontinued pesticide exposure precluded additional environmental and fish screening for pyrethroids to provide further support for the conclusions stated herein. That being said, environmental or opportunistic fish sampling at the time of diagnosis would have been helpful to confirm exposure to systems in unusual circumstances and presentations such as this report. In addition, further research under controlled exposures and conditions utilizing adult ictalurid catfish and individual pyrethroid compounds is required to investigate the pathology, pathogenesis, and toxicological properties and concentrations associated with pyrethroid exposure.
Pesticide-induced toxicoses involving aquarium fish are rarely reported with primary differential etiologies generally including organophosphates, chlorinated hydrocarbons, pyrethroids, and pyrethrins. 36 The lesions associated with repeated, inadvertent pyrethroid exposure have not been described in fish nor in public aquarium settings. These cases illustrate the dynamic nature of managing outdoor, semi-closed systems exposed to the elements and the importance of communication among the veterinary team, animal care team, facilities management, and outside contractors to prevent and diagnose exposure of collection animals to environmental contaminants.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231222226 – Supplemental material for Pyrethroid-associated nephrotoxicity in channel catfish, Ictalurus punctatus, and blue catfish, I. furcatus, at a public aquarium
Supplemental material, sj-pdf-1-vet-10.1177_03009858231222226 for Pyrethroid-associated nephrotoxicity in channel catfish, Ictalurus punctatus, and blue catfish, I. furcatus, at a public aquarium by Justin M. Stilwell, Sean M. Perry, Lora Petrie-Hanson, Rachel Sheffler, John P. Buchweitz and Alexa J. Delaune in Veterinary Pathology
Footnotes
Acknowledgements
The authors thank the microbiology and histology services at Mississippi State University College of Veterinary Medicine, Lori Ford, and Dr. Larry Hanson for their contributions in the diagnostic investigations of these cases. The authors also thank Drs. Lester Khoo, Brian Bosworth, and Geoff Waldbieser for providing control catfish tissues. The zebrafish bioassay was performed under an approved IACUC protocol (IACUC-22-087) through the Mississippi State University College of Veterinary Medicine according to previously published methods.
Supplemental Material for this article is available online.
Author Contributions
J.M.S. performed histopathologic examination on all cases and coordinated ancillary diagnostic testing; S.M.P. and A.J.D. performed the clinical management on all cases and provided clinical history; J.M.S., S.M.P., and A.J.D. performed the gross necropsies; L.P.H. performed the zebrafish bioassay; R.S. and J.P.B. performed toxicological analysis on tissue and fluid samples and interpreted the findings; the manuscript was written by J.M.S. with contributions from all coauthors.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
