Abstract
Canine thyroid carcinomas are relatively common malignant endocrine neoplasms in dogs derived from either thyroid follicular cells (forming follicular thyroid carcinomas) or medullary cells (parafollicular, C-cells; forming medullary thyroid carcinomas). Older and recent clinical studies often fail to discriminate between compact cellular (solid) follicular thyroid carcinomas and medullary thyroid carcinomas, which may skew conclusions. The compact subtype of follicular thyroid carcinomas appears to be the least differentiated subtype of follicular thyroid carcinomas and needs to be differentiated from medullary thyroid carcinomas. This review includes information on the signalment, presentation, etiopathogenesis, classification, histologic and immunohistochemical diagnosis, clinical management, and biochemical and genetic derangements of canine follicular and medullary carcinomas, and their correlates with human medicine.
Canine thyroid carcinomas (CTCs) are the most common endocrine malignancy in dogs, tend to grow rapidly, often invade local tissues and vasculature, and approximately one third of dogs with CTCs have metastasis at diagnosis.9,10,12,26,30,38,44,46–48,51 CTCs arise from 1 of the 2 main epithelial constituents of the thyroid gland—thyroid follicular cells (thyrocytes), which produce colloid, thyroid hormones (serum T3 and T4), and thyroglobulin, or medullary cells (C-cells, parafollicular cells), which produce calcitonin.29,30,41,42,46–48 Malignant neoplastic transformation of these cells yields follicular thyroid carcinomas (FTCs), which are then classified based on histologic patterns, and medullary thyroid carcinomas (MTCs), respectively.12,26,30,44,47 FTCs are considered to be more common; historically, MTCs accounted for less than 5% of all canine thyroid neoplasms, but recent literature suggests MTCs may be more prevalent with incidences ranging between 16% and 36%.4,9–12,26,27,30,40,45–47,49,50,53 Currently, the most common treatment modality is surgical excision, which may be accompanied by a multitude of other modalities.10–13,25,28,30,37,50 Histologic examination is required for the confirmation of malignancy (namely local invasion or intravascular tumor emboli), while the use of immunohistochemistry (IHC) is required for determining the cell of origin (FTC vs MTC).10,12,45,51 Beyond the individual patient, routinely and consistently confirming the cell of origin and subsequent appropriate histologic subtyping provides 2 main benefits. First, this allows for more accurate comparisons between types of CTCs, as would be needed for prospective and retrospective analyses on survival and prognosis or experimental treatment modalities. Second, the current trend in human medicine is for highly customized, molecular-based treatment regimens (known as personalized health care or precision medicine) which are based on specific diagnoses, like the presence of specific genetic mutations or overexpression of certain molecules which contribute to tumorigenesis.1,3,7,39,55,57 In veterinary medicine, the ability to provide a more definitive diagnosis for CTCs would, in the future, allow for more customizable treatment regimens for each patient.
Similar to dogs, thyroid cancer is the most common endocrine malignancy in humans and is a leading cause of death among endocrine cancers, usually due to invasion into surrounding tissues or metastasis.42,56 However, the most common mutations in human thyroid carcinomas are considered rare in dogs, although some similarities have been identified, like increased expression of AKT2 and PIK3CA in canine FTCs, and increased expression of RET in canine MTCs.1,11,22,55,56 In addition, epidermal growth factor expression has been identified in thyroid carcinomas of both human and dogs; human neoplasms specifically express human epidermal growth factor 2. 59 The identification and selection for these similar genetic and molecular alterations could allow for further development of canine models that are even more aligned to the human disease. Canine FTCs (including follicular, compact, mixed, and papillary subtypes) have similar histologic and biologic behavior compared with differentiated human thyroid carcinomas, lending support to the idea of dogs being used as an animal model for human thyroid cancer.11,15,22,36,55,60 Furthermore, canine MTC is similar to human MTC concerning morphology, cytochemical, and IHC features.2,7,11,14,18,27
As veterinary medicine tends to lag behind human medicine, the investigation into more specific diagnoses, including genetic or biochemical derangements, is critical to further advance our understanding of tumorigenesis of CTCs and the subsequent development of effective, customized treatments.
This review will cover signalment, presentation, etiopathogenesis, classification, histologic and immunohistochemical diagnosis, clinical management, and biochemical and genetic derangements of canine follicular and medullary carcinomas, and their correlates with human medicine with an emphasis on differences between canine compact FTCs and MTCs, where applicable. Figure 1 presents key takeaways from this review regarding the differences between FTCs and MTCs, whereas Table 1 compares and contrasts key features of human and dog thyroid carcinomas.

Graphical abstract highlighting key takeaways from this review. FTCs, follicular thyroid carcinomas; MTCs, medullary thyroid carcinomas.
Selected comparisons of thyroid neoplasia between dogs and humans.
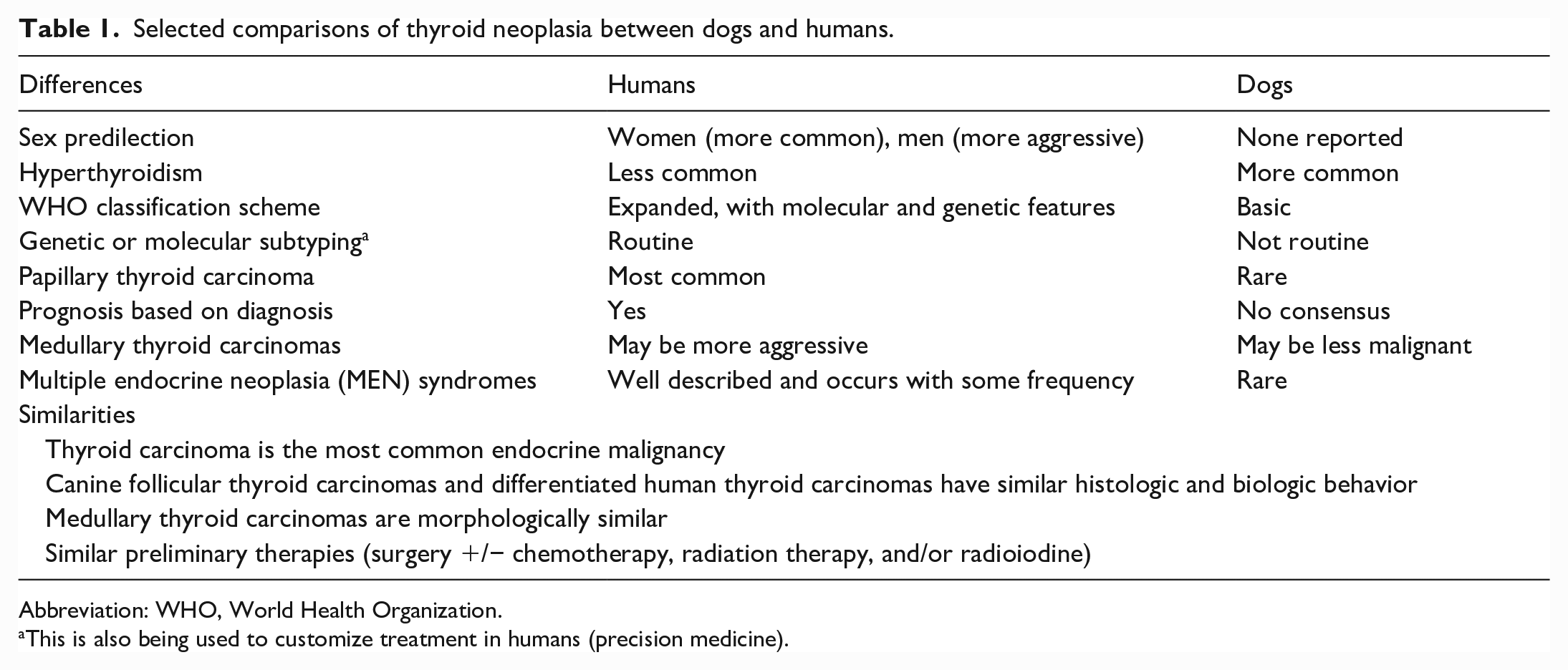
Abbreviation: WHO, World Health Organization.
This is also being used to customize treatment in humans (precision medicine).
Signalment and Presentation
Dogs presenting with any form of thyroid carcinoma (FTC or MTC) have a mean and/or median age of 9 to 10 years, with increased risk associated with advancing age, and there is no defined sex predilection.4,9,24,30,47,48,51 Generally, regarding age, FTCs and MTCs are not separated in literature reports, but a few studies have shown that dogs with MTCs have a mean age of 9.6 years or median age of 9 years, with a range of 4 to 16 years.10,12,40 One study reported that differentiated FTCs have a median age of 10 years, with a range of 4 to 14 years. 10 An older study of 16 dogs with MTCs found males were 3 times more likely to have MTCs than females, but the same has not been corroborated in other studies. 40 In humans, older women have a higher risk of developing thyroid cancer, while men tend to experience more aggressive thyroid cancer.4,24,56
Breed predilection reports are conflicting and variable; however, breeds such as boxers, beagles, Cairn terriers, golden retrievers, old English Sheepdogs, Shetland collies, and Siberian huskies may have a higher risk for CTC.23,24,30,47,48 One study suggested poodles (miniature and toy) may have decreased the risk of developing CTC. 24
CTCs are most commonly unilateral tumors that arise near the larynx without a reported side predisposition (ie, right vs left thyroid gland); bilateral neoplasms are less common.27,30,40,43,47,48 In humans, there are also no reported predilections for the left or right thyroid gland. Canine thyroid neoplasms may form anywhere along the cranial neck (including the tongue) to the thoracic inlet or even in the ventral mediastinum, pericardium, heart base, and descending aorta due to ectopic thyroid parenchyma that was presumably entrapped during early development.30,48
The most common presentation for CTCs is a palpable ventral or ventrolateral cervical mass that may be associated with additional clinical signs or symptoms such as coughing, dyspnea, dysphagia, dysphonia, gagging, retching, regurgitation, Horner’s syndrome, and cranial vena caval syndrome due to either tumor compression (mass effect) or invasion.28,30,48 These clinical signs may result in either the death of the animal or election of humane euthanasia by the owners.
Clinically distinguishing between FTCs and MTCs can be a diagnostic challenge, but functional neoplasms may be identified by evaluating serum hormone levels. Dogs with FTCs tend to be euthyroid, but hypothyroidism and hyperthyroidism may occur. Mechanisms of hypothyroidism can be nonspecific and include complete bilateral destruction of the thyroid glands, suppression of pituitary-derived thyroid-stimulating hormone (thyrotropin), or the suppressive effects of nonspecific illnesses on circulating thyroid hormone concentrations (euthyroid sick syndrome), while mild hyperthyroidism may rarely occur with functional FTCs that secrete sufficient T3 and/or T4 thyroid hormones.12,30,43,47,48 Dogs with functional FTCs and concurrent hyperthyroidism are clinically similar but less severely affected than cats with clinical hyperthyroidism. 30 Dogs with CTCs tend to experience hyperthyroidism more commonly than humans. 27 For MTCs, humans and dogs may both have hypercalcitoninemia, but its use as a diagnostic marker is mostly restricted to human medicine.14,44 There are apparently no reports of nonfunctional MTCs (non-calcitonin secreting MTCs) in the veterinary literature, while this is rarely reported in humans based on serum calcitonin levels and pentagastrin stimulation testing.18,44 In these human cases, approximately half expressed calcitonin and carcinoembryonic antigen and most expressed chromogranin A by IHC. 18 MTCs may also be accompanied by low to normal levels of serum calcium.26,39,45 Alternatively, MTCs may be associated with hypercalcemia (given the proposed pathogenesis, highlighted below), but this is less diagnostic, because FTCs may cause humoral hypercalcemia of malignancy.47,48
Histopathologic Classification of CTCs
Histologic classification of CTCs follows the World Health Organization classification scheme which separates FTCs into well-differentiated (with subtypes), poorly differentiated, and undifferentiated neoplasms (with subtypes like carcinosarcoma [malignant mixed thyroid tumor]), while MTCs are considered a separate, single entity.10,26 Subtypes of well-differentiated FTCs are based on histologic patterns and include follicular, follicular-compact (also known as mixed), compact cellular (also known as compact or solid), and papillary.12,26,30,44,47,48 Confusingly, some veterinary references use “mixed” to describe carcinosarcoma (malignant mixed thyroid tumors) where malignant follicular cells are mixed with malignant mesenchymal components (eg, cartilage, bone, or both), while in human thyroid carcinomas there also exists a mixed carcinoma consisting of both neoplastic medullary cells and neoplastic follicular cells.3,8,46 For this review, the follicular-compact subtype will hereby be referred to as the “mixed” subtype, and the compact-cellular will be referred to as the “compact” subtype. The World Health Organization classification scheme for CTCs is superficially similar to the scheme for human thyroid neoplasms, but the human scheme incorporates pertinent molecular and genetic features; contains more specific subtypes, variants, and categories of thyroid neoplasms (eg, Hürthle [oncocytic] cell tumor, mucoepidermoid carcinoma); and is more regularly updated (most recently in 2017, 4th edition), as compared to the scheme in domestic animals (originally from the 1970s with the most current, 2nd edition released in 2008).3,26 In addition, differentiated thyroid carcinomas in humans appear to be restricted to papillary thyroid carcinoma (derived from follicular cells), FTC (derived from follicular cells), MTC (derived from medullary cells), and Hürthle cell carcinoma (thought to be derived from metabolically altered follicular cells with increased numbers of mitochondria).1,3,56
Follicular, mixed, and compact subtypes are commonly diagnosed in dogs. 42 Of these, mixed FTCs are reported to be the most common and are characterized by approximately equal proportions of follicular and compact neoplastic growth, with neoplastic follicles that may be smaller and contain less colloid than would be observed in the follicular pattern.26,47,48 Few studies indicate that the compact type may be more common than other subtypes, and some have suggested this is the least differentiated subtype out of the well-differentiated FTCs, while mixed FTCs and follicular FTCs represent intermediate and the most differentiated subtypes, respectively.11,42 In contrast, papillary thyroid carcinoma predominates in humans (estimated 75%–90% of all differentiated thyroid carcinomas).1,2,7,12,37,60
A diagnostic challenge arises with differentiating compact FTCs from MTCs with routine hematoxylin and eosin light microscopy and typically requires ancillary diagnostic modalities (Fig. 2).12,26,27,40,43,47,48 Table 2 summarizes histologic features that may assist with distinguishing compact FTCs and MTCs on hematoxylin and eosin-stained slides. Compact FTCs are composed of aggregates to solid sheets of polyhedral cells with finely granulated to vacuolated eosinophilic cytoplasm, which gives little to no attempt at follicle formation and/or colloid secretion.26,47,48 MTCs are similarly composed of polyhedral to spindle-shaped cells with lightly eosinophilic to amphophilic, finely granular cytoplasm, and an oval to elongate vesicular nucleus.26,41,46–48,58 Histologic features that support the diagnosis of FTC include numerous variably sized follicles with periodic acid-Schiff-positive colloid; eosinophilic cytoplasm; greater pleomorphism than what is seen in adenomas; and hemorrhage into follicles. In contrast, histologic features that support the diagnosis of MTC include intrathyroidal and/or extracapsular invasion; occasional ducts and acini; production of amyloid derived from secreted proteins; solid growth with neuroendocrine packeting; amphophilic cytoplasm; prominent stroma; few to no follicles; and palisading cells along the periphery of lobules.27,44,46,48 MTCs may also have variable histologic patterns of growth, including tubular (follicular), papillary, small cell, giant cell, clear cell, oncocytic, and mixed variants; however, this distinction is not often pursued or well described in the current diagnostic setting. 26 Amyloid production is rarely observed in canine MTCs and is more commonly found in human or bull medullary carcinomas.26,41,44,47,48 Differentiation between MTC and compact FTC is further confounded by the presence of either entrapped, “normal” (non-neoplastic) thyroid follicles surrounded by neoplastic cells (Fig. 3) or the formation of medullary cell follicles, which are neoplastic medullary cells surrounding free colloid following the destruction and loss of thyroid follicular cells.47,48 MTCs tend to entrap thyroid follicles more than FTCs tend to entrap medullary cells. 48
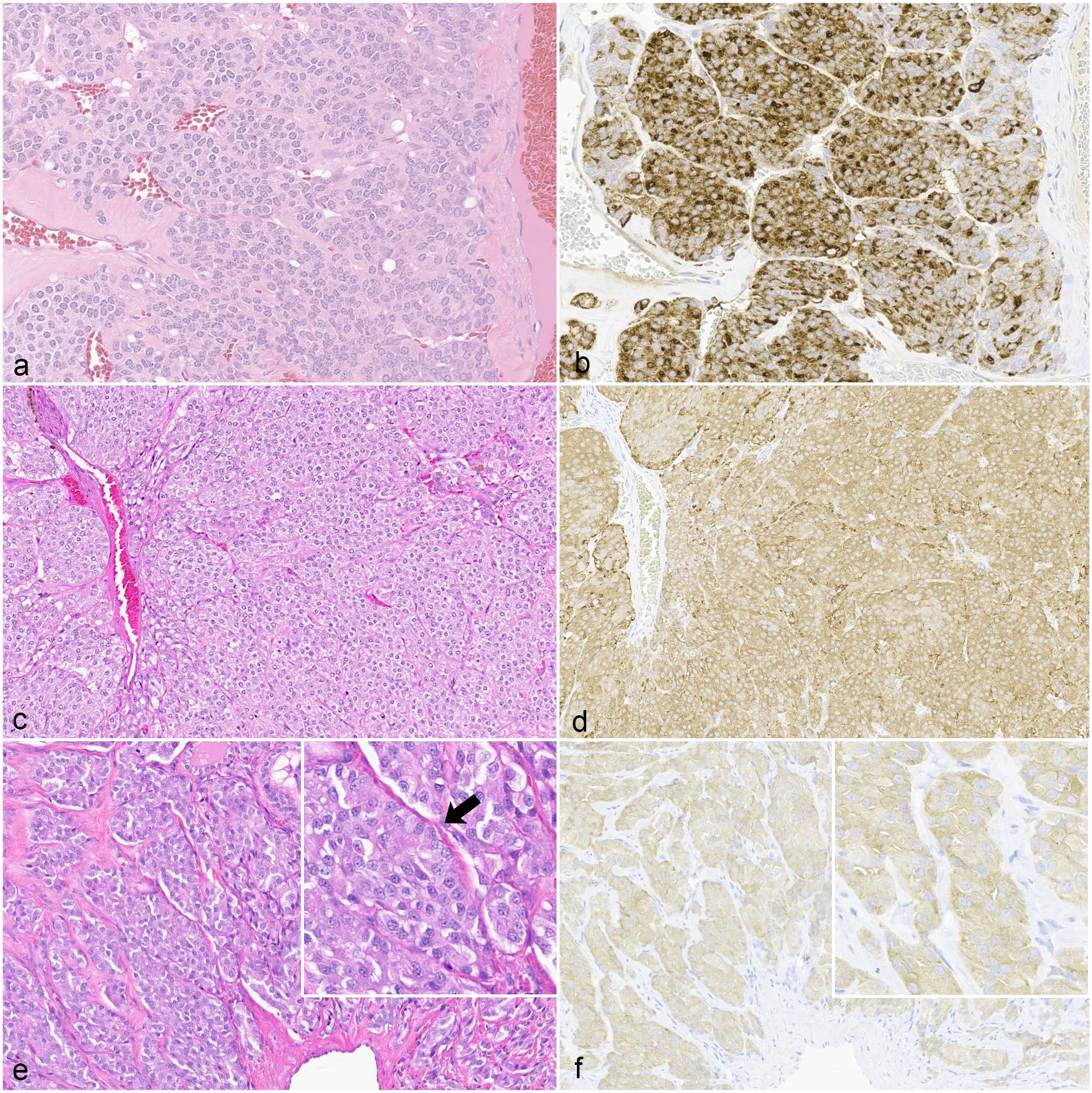
Histologic similarity between canine compact cellular (solid) follicular thyroid carcinomas (FTCs) and medullary thyroid carcinomas (MTCs). (a) Compact FTC with polyhedral cells that form aggregates with eosinophilic cytoplasm, few vacuoles, and no identifiable colloid or colloid follicles. Hematoxylin and eosin (HE). (b) Compact FTC. The majority of the neoplastic cells have moderate to strong cytoplasmic immunoreactivity for thyroglobulin. Immunohistochemistry (IHC) for thyroglobulin. (c) MTC with polyhedral cells forming packets. HE. (d) MTC. Neoplastic cells have moderate to strong cytoplasmic immunoreactivity for synaptophysin. Granular cytoplasmic immunoreactivity is identifiable in some regions. IHC for synaptophysin. (e) MTC with polyhedral cells forming packets with a prominent stroma. Cells subtly palisade along basement membranes (arrow; inset). HE. (f) MTC. Neoplastic cells have faint to lesser amounts of moderate cytoplasmic immunoreactivity for calcitonin. IHC for calcitonin IHC.
Histologic tendencies that may allow discrimination between the compact subtype of follicular thyroid carcinomas and medullary thyroid carcinomas on H&E-stained slides, canine.

Abbreviations: H&E, hematoxylin and eosin; PAS, periodic acid-Schiff.
Colloid will be PAS-positive.
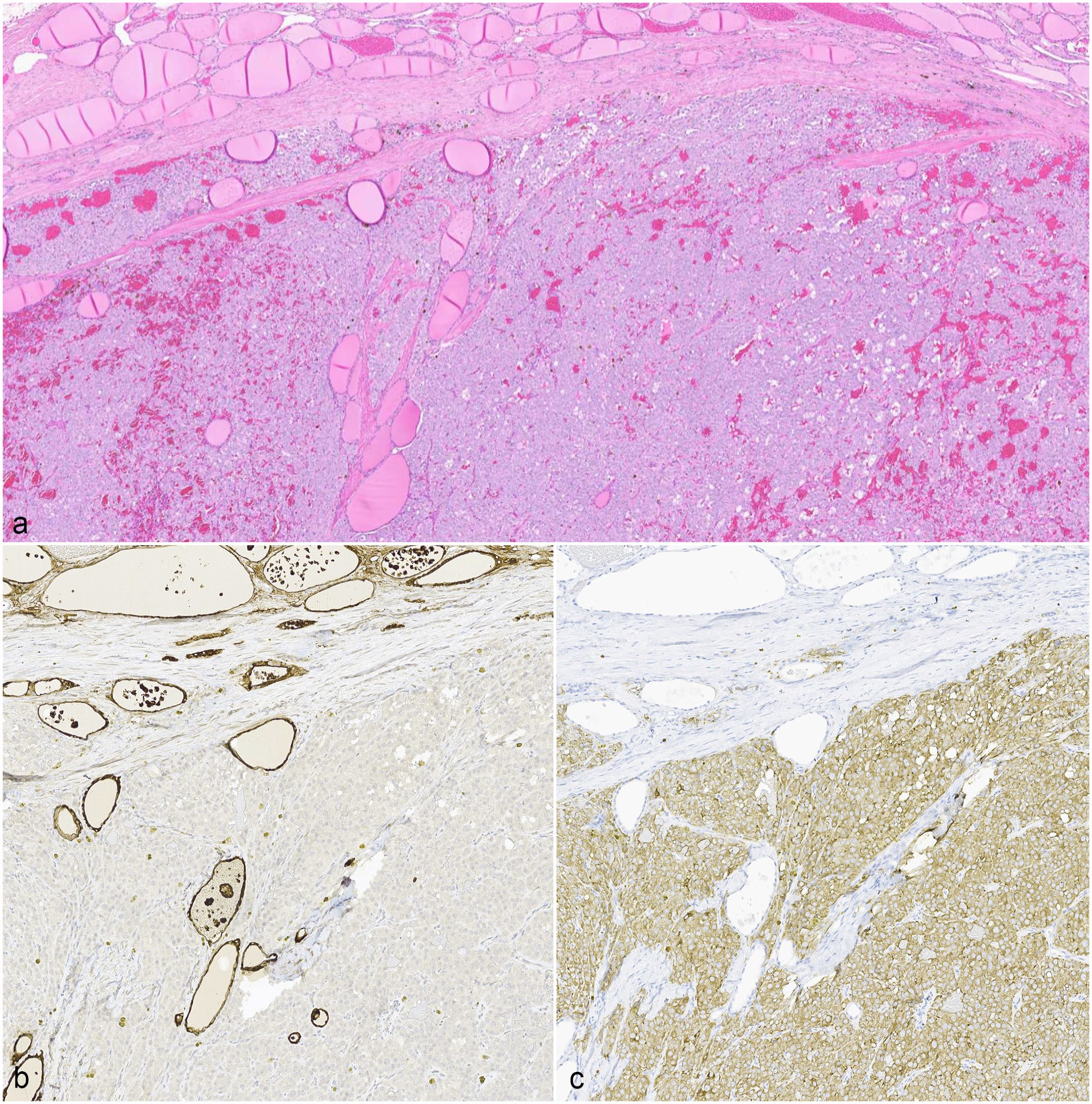
Example of entrapped thyroid follicles within a canine medullary thyroid carcinoma (MTC). (a) The neoplastic cells encompass the bottom half of the image along with a few entrapped thyroid follicles and hemorrhage. The non-neoplastic thyroid gland with numerous colloid follicles is in the top half of the image. Hematoxylin and eosin (HE). (b) Thyroglobulin immunohistochemistry (IHC) highlights the entrapped thyroid follicles. (c) The neoplastic cells, but not the thyroid follicles, have immunoreactivity to synaptophysin. IHC for synaptophysin.
Differentiation of CTCs by IHC, and Histologic and Immunohistochemical Differentials
As alluded to above, IHC examination is often required to distinguish compact FTCs from MTCs.10,12,27,37,51 Table 3 summarizes diagnostically useful IHC assays for thyroid or parathyroid neoplasia while Table 4 provides an overview of IHC labeling patterns for thyroglobulin, calcitonin, and neuroendocrine markers in FTCs and MTCs.
Selected discriminatory immunohistochemical markers for canine thyroid- or parathyroid-derived neoplasms.

Abbreviations: TTF-1, thyroid transcription factor-1; ND, not done; IHC, immunohistochemistry.
Neuroendocrine markers most commonly include synaptophysin or chromogranin A but may include protein gene product 9.5 and neuron-specific enolase.
PAX-8 may also be used to help confirm FTCs.
Napsin A and carcinoembryonic antigen may also be used to help confirm MTCs.
Additional IHCs used for this report included negative immunoreactivity for carcinoembryonic antigen and S100 protein and positive immunoreactivity cytokeratin AE1–AE3.
Immunohistochemical labeling patterns of canine well-differentiated follicular thyroid carcinomas and medullary thyroid carcinomas.
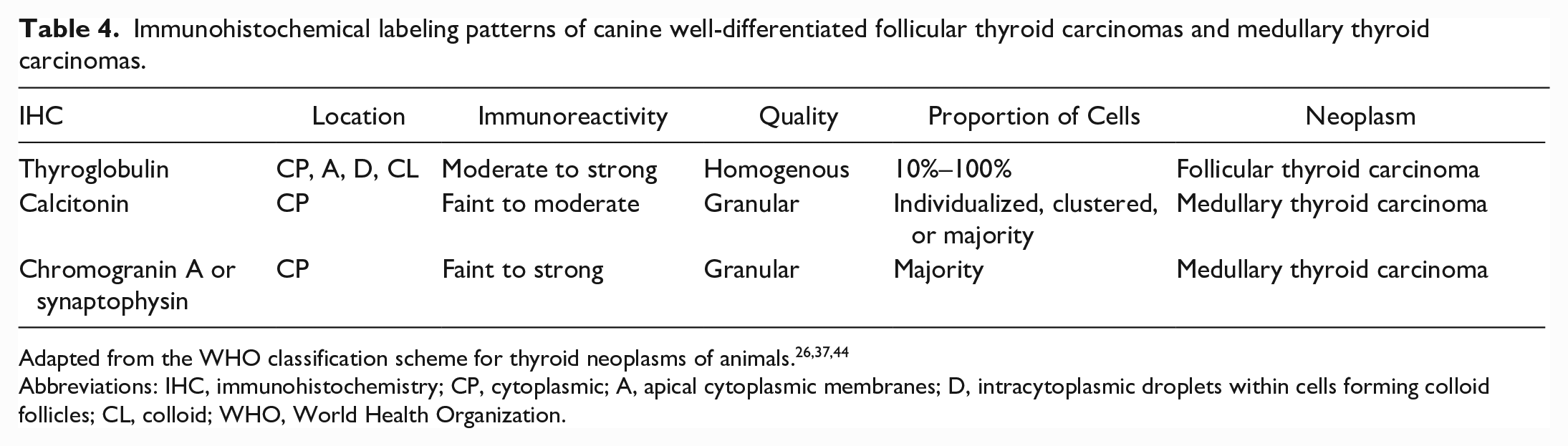
Abbreviations: IHC, immunohistochemistry; CP, cytoplasmic; A, apical cytoplasmic membranes; D, intracytoplasmic droplets within cells forming colloid follicles; CL, colloid; WHO, World Health Organization.
FTCs are diagnosed based on thyroglobulin immunoreactivity in 10% to 100% of the cells and are ideally concurrently negative for calcitonin and/or neuroendocrine IHC markers (such as chromogranin A+B, synaptophysin, protein gene product 9.5 [PGP9.5], and neuron-specific enolase).12,26,37,44,46 In contrast, MTCs may be diagnosed with variable amounts of cytoplasmic immunoreactivity for calcitonin or neuroendocrine markers (as above) without immunoreactivity for thyroglobulin.12,26,37,44,45 The range in thyroglobulin immunoreactivity may relate to histologic subtypes, as follicular and mixed subtypes appear to more consistently express thyroglobulin as compared to the compact subtype. 12 Less commonly, IHCs for thyroid transcription factor-1 (TTF-1) or PAX8 can be used to confirm FTCs while calcitonin gene-related peptide, napsin A, and carcinoembryonic antigen can be used to confirm MTCs; however, some of these are not readily available for routine diagnostic use.44–47 One study found that TTF-1 cannot be used as a discriminatory IHC marker, because TTF staining was detected in both FTCs and MTCs; they instead propose TTF may be used in conjunction with thyroglobulin IHC to improve the overall sensitivity of the IHC panel. 46 Another study also suggests calcitonin gene-related peptide may be a more sensitive marker than calcitonin for diagnosing MTCs. 52 An additional challenge may arise with either scant or faint IHC staining for either thyroglobulin or calcitonin. For thyroglobulin, this may be from altered physicochemical hormones or inactivity of neoplastic cells. In the case of calcitonin, this may result from preferential secretion and concurrent lack of hormone storage with highly functional MTCs.37,48 Conversely, inadequate or delayed fixation of CTCs may allow diffusion of thyroglobulin into the interstitium, contributing to increased background staining and potentially confounding thyroglobulin IHC interpretation. 46 In MTCs, the presence of entrapped, remnant, thyroglobulin-positive parenchyma may also interfere with accurate IHC interpretation. 48
In the past, MTCs were routinely underdiagnosed due to their histologic similarity to compact FTC and the lack of the confirmatory calcitonin IHC marker.10,12 Underdiagnosis of canine MTCs likely contributes to the lack of comparative prognostic studies with canine FTCs and, as previously mentioned, many current clinical studies fail to distinguish between these tumors. This lack of discrimination between FTCs and MTCs may perpetuate skewed data with lower MTC prevalence and may yield unreliable interpretations and conclusions from CTC studies due to the masking of differences between MTCs and FTCs.
Interestingly, bulls may get ultimobranchial thyroid neoplasms, which have a heterogeneous histologic pattern of more typical medullary cells mixed with undifferentiated cells that express both thyroglobulin and calcitonin (dual immunoreactivity); the ultimobranchial body is thought to be the origin of medullary cells in embryological development.26,48 There is speculation that a unique version of mixed thyroid carcinoma in humans (mixed medullary and follicular thyroid carcinoma) could also represent this entity.3,8,26,48 Ultimobranchial tumors should therefore be considered tumors of stem cells and may be a consideration for unusual, dual-positive canine tumors.26,37 Canine ultimobranchial tumors have not been previously reported, although incidental ultimobranchial cysts are relatively common.47,48
Recently, a calcitonin-negative (nonmedullary) primary neuroendocrine tumor of the thyroid in a dog was reported that had no immunolabeling for calcitonin, carcinoembryonic antigen, thyroglobulin, S100 protein, and had expression of synaptophysin and cytokeratin AE1-AE3, which is similar to the rare human tumor known as calcitonin-negative neuroendocrine tumor of the thyroid or “nonmedullary” thyroid tumor. 52 In this case, it was not possible to rule out a parathyroid tumor by IHC, but primary hyperparathyroidism was ruled out clinically and a parathyroid tumor was therefore considered unlikely. 52 Other differentials for human calcitonin-negative neuroendocrine tumors of the thyroid gland include paraganglioma, hyalinizing trabecular tumor, metastatic neuroendocrine tumor to the thyroid gland, and intrathyroidal parathyroid adenoma or tumor; some of these may also be viable differentials for dogs.8,52 For parathyroid tumors, primary hyperparathyroidism often aids in establishing an initial diagnosis with histopathology and IHC evaluation acting to confirm the diagnosis. 52 Neoplastic cells for canine parathyroid adenomas and carcinomas express parathyroid hormone, cytokeratins, neuroendocrine markers (eg, chromogranin A), and neurofilaments (such as S100).26,47,48 Literature examining cytokeratin immunohistochemical staining in parathyroid glands is sparse in veterinary medicine. Human parathyroid glands (normal and non-neoplastic) express cytokeratins, including cytokeratin 19, but the diagnostic value of this is considered low because human thyroid tumors may also express cytokeratin 19. 54
Therefore, in cases of solid neoplasms with thyroid carcinoma as a differential, a definitive diagnosis may be challenging without pertinent clinical history, such as the presence or absence of hyperthyroidism or hyperparathyroidism, and without a robust IHC panel. A robust IHC panel should include at least 2 FTC markers (thyroglobulin and TTF-1), 2 MTC markers (calcitonin and calcitonin gene-related peptide), 1 or more neuroendocrine markers (synaptophysin or chromogranin A), and parathyroid hormone, for example.
Differences in Biologic Behavior and Prognosis Between FTCs and MTCs and Treatment Modalities
Metastatic disease is frequently present at the time of diagnosis of CTCs, but some recent studies suggest that the progression of metastatic disease is slow. 20 The likelihood of metastasis increases with the increased size of the primary tumor, evidence of vascular invasion (eg, tumor thrombi in the cranial thyroid vein), and bilateral disease.10,25,30,38 A study on stereotactic body radiation therapy in the treatment of CTCs found the presence of metastasis was not a negative prognostic factor, while another study found no difference between post-thyroidectomized well-differentiated FTCs and MTCs in the overall survival, disease-free survival, time to metastasis, and time to recurrence.10,28 Reported rates of metastasis at diagnosis include a range from 14% to 26% for regional metastasis (regional lymph nodes), 20% to 38% for distant metastasis (pulmonary), and 18% to 95% for overall metastasis (combining regional and distant).20,25,38 However, prior studies largely do not distinguish between FTCs and MTCs. Regional lymph nodes may include the submandibular, medial retropharyngeal, and parotid lymph nodes. 30
FTCs and MTCs carry different metastatic tendencies, which may have undiscovered treatment implications. FTCs tend to metastasize to the lung via invasion of the cranial and caudal thyroid veins with intravascular neoplastic emboli, while MTCs tend to metastasize to the anterior cervical lymph nodes.26,30,47,48 The metastasis rates for FTCs have been associated with cellular differentiation, as poorly differentiated tumors have a higher rate of metastasis, while functional FTCs may have a lower metastatic rate.13,10,49 Distant metastases may uncommonly be found in the brain, bone, liver, kidney, adrenal gland, liver, heart, and other organs.28,30 Previously, there was speculation that the metastatic rate for MTCs may be lower than FTCs, but one study found that at diagnosis, there was no difference in the incidence of metastasis.10,12,48 Various antemortem imaging techniques may be used to detect metastatic disease, but computed tomography appears to be a more sensitive modality compared with older, different imaging techniques. 20 Scintigraphy may be useful in identifying well-differentiated neoplasms, but is less reliable for the detection of poorly differentiated lesions, such as those with pulmonary metastasis, due to their low uptake of iodine. 30
At this time, both canine MTCs and all FTC subtypes have similar treatment protocols and are given similar prognoses; however, an older study suggested that MTCs may have a less biologically aggressive behavior given that they tended to be well circumscribed and resectable.10,12 In contrast, for humans, there are described and accepted differences in prognosis based on the subtype or variant while some even consider MTCs as more aggressive with a less favorable prognosis than differentiated FTCs.12,17 Furthermore, human papillary thyroid carcinoma carries a favorable prognosis with a low incidence of metastasis; however, several described variants carry a worse prognosis.2,3,7,12,15
Currently, the main treatment modality is surgery with curative intent for mobile and well-circumscribed CTCs (complete thyroidectomy), but incisional biopsy with or without additional ancillary therapies may be pursued for invasive and non-resectable neoplasms, which is similar for humans.2,7,10–14,18,25,28,30,36,37,50,53,55 Ancillary therapies for dogs may include local external beam radiation therapy, chemotherapy (eg, toceranib phosphate [Palladia], doxorubicin, carboplatin, cisplatin, and adjunctive retinoic acid 9 cis [isotretinoin 9-cis; RA9-cis]), and/or radioactive iodine administration (131iodine).10–13,25,28,30,37,50 Only approximately 25% to 50% of canine cases are amenable to complete surgical resection due to local invasiveness and proximity of critical anatomic structures.25,28 For cases where surgical resection is not possible, radiation therapy and radioiodide therapy may provide median survival times of up to 24 and 30 months, respectively, while the reported median survival time of dogs with incompletely excised CTCs without ancillary therapies is 10 months.5,49 In humans, specific and/or additional treatments may include customized targeted therapies (eg, tyrosine kinase inhibitors, BRAF inhibitors, immune checkpoint inhibitors; singly or in combination), lymphadenectomy for patients with nodal metastasis of MTCs, and levothyroxine to suppress thyrotropin.2,7,14,18,36,53,55 One glaring difference between veterinary medicine and human medicine is that in human thyroid cancer, it is routine to pursue genetic subtyping for common mutations (eg, BRAFV600E) which then permits the use of a customized and highly targeted therapeutic regimen (also known as personalized health care or precision medicine) for the patient in question, especially in cases with advanced disease (eg, utilization of dabrafenib/trametinib combination therapy for BRAF-mutated cancer [Food and Drug Administration approved for anaplastic thyroid carcinoma]).1,3,6,7,14,22,55,56 Because of the inclusion of molecular and genetic derangements to the most current human World Health Organization thyroid neoplasm classification scheme, clinicians can more easily practice precision medicine. 3 In addition, other therapeutic targets being investigated in human and canine CTCs may include cancer-associated fibroblasts. 57 Unsurprisingly, veterinary medicine lags behind human medicine in these aspects, but this may be what the future of cancer treatment looks like for veterinary patients as well. The possible use and efficacy of checkpoint inhibitors in CTC are not yet reported, but in a study of dogs with pulmonary metastatic oral malignant melanoma treated with anti-programmed cell death-ligand 1 monoclonal antibody, overall survival time was increased. 33 This same study surveyed several malignant canine cancers for the presence of programmed cell death-ligand 1 expression, but CTCs were not included. 33
As mentioned earlier, the uptake of radioactive iodine appears to be related to the degree of neoplastic cell differentiation in dogs, with compact FTCs likely representing the least differentiated neoplasm within the well-differentiated FTC category.13,25,43 This is supported by a preliminary study which found canine follicular FTCs were associated with hyperthyroidism and increased uptake of scintigraphy agent 99mTc, mixed FTCs were associated with euthyroidism and normal uptake of 99mTc uptake, and compact FTCs were associated with hypothyroidism and decreased 99mTc uptake.13,25,43 CTCs do not need to be functional for abnormal scintigraphy studies, and there is speculation that neoplasms may be sensitive to radioactive iodine regardless of functional status, but consensus, even in human medicine, is lacking.16,30,36 There is no reported information regarding iodine uptake and efficacy of radioactive iodine administration on MTCs in dogs. A recent study describes the response of CTCs (primarily FTCs) to radioiodine, but they fail to describe methods used to exclude MTCs. 25 In human medicine, one study concluded that radioactive iodine treatment may be a valid, locally aimed adjuvant treatment modality in MTCs, even though these cells do not concentrate radioactive iodine, while another multicenter study did not corroborate the same and strongly opposed the use of radioactive iodine in MTCs.16,36 Mechanistically, for both species, it is thought that the organification (binding of iodine to thyroglobulin tyrosine residues) of radioactive iodine isotopes in adjacent thyroid follicular cells could destroy the adjacent medullary cells (“bystander effect”).16,34 Numerous publications on the role of radiation therapy in fixed CTCs suggest that the primary treatment focus should be on local control of the primary tumor rather than systemic treatments, based on their study results and that progression of metastatic disease is slow.19,25,38
Prognostication of CTCs largely varies with gross and histologic characteristics, such as tumor size or volume, local invasiveness, evidence of distant metastases, or evidence of vascular invasion; however, some of these features may be controversial.10,12,30,51 One study concluded that some of these features had no significant effect on overall survival, disease-free survival, time to distant metastasis, or time to loco-regional occurrence, but macroscopic and histologic vascular invasion were independent negative predictors for disease-free survival. 10 Prognosis can range from good to excellent; for surgery alone, median survival times range from 7 to 8 months to over 36 months, while the median survival time for untreated dogs is 3 months.10,12,30,51 The reported median survival time for incompletely excised CTCs without ancillary therapy is 10 months and is largely due to disease secondary to local neoplastic invasion. 5 The control provided by radioiodide appears similar to other non-surgical treatment modalities, alleviates clinical signs, and prolongs survival, but does not significantly reduce tumor size; in people with lymph node and distant metastasis, radioactive iodine independently predicts a longer survival. 25 In humans, there are differences in prognosis based on the subtype or variant of thyroid neoplasm, which contrasts dogs. Other reported prognostic factors in humans include age, sex, tumor size, stage, histologic grade, vascular invasion, and extrathyroidal tumoral extension.10,21 However, overall, both human FTCs and MTCs are considered relatively indolent neoplasms with high 10-year survival rates in humans.11,15,22,36,55
Some of the aforementioned gross or clinical characteristics can be surmised from the clinician’s impression before and during surgery; however, the surgeon’s interpretation may be inaccurate, so histologic examination is critical for confirmation of the gross findings, cell of origin, and further evaluation of criteria of malignancy (eg, capsular invasion, vascular invasion, mitotic rate).10,12,51 Negative prognostic indicators may include both gross and histologic vascular invasion, increased (delayed) time to presentation, and increased tumor size (before thyroidectomy), while some papers maintain that histologic evidence of capsular or vascular invasion does not correlate with poor survival.10,30 However, some of the data was from dogs that were examined for necropsy or had inoperable tumors, and it may not accurately reflect the situation in dogs with operable tumors. 10 Studies on operable thyroid tumors found that bilateral disease and histologic grade of malignancy may be helpful as prognostic indicators, although a histologic grading scheme has not been previously published nor accepted. 10 Another paper suggested that completely excised mobile thyroid tumors and irradiated fixed thyroid carcinomas had good prognoses. 30 While the use of chemotherapy or surgery tends to improve the prognosis, there does not appear to be a difference in median survival time when chemotherapy and surgery are used in combination compared with surgery alone.13,20,30,38 Studies have attempted to correlate World Health Organization tumor, lymph node, and metastasis (TNM) staging system to prognosis in CTCs, but results are largely conflicting at this time.20,25,53
Ki-67, a cellular marker of proliferation, has been evaluated in canine FTCs and MTCs, and no significant differences were identified. In addition, Ki-67 labeling may be positively associated with local invasiveness and negatively associated with time to metastasis.10,51 In well-differentiated human FTCs, increased Ki-67 is associated with higher metastatic rates at diagnosis and decreased disease-free survival. 10 Ki-67 may assist in identifying poorly differentiated human thyroid neoplasms from well-differentiated or anaplastic thyroid carcinomas, and was used, along with mitotic figures and necrosis, in a recently proposed grading scheme for human MTCs that is thought to accurately predict overall survival.3,17 Unfortunately, there is controversy surrounding the routine use of Ki-67 in humans, as other studies have found no prognostic associations. 17
Etiopathogenesis
The etiopathogenesis of both canine FTCs and MTCs is largely unknown. For FTCs, iodine deficiency causing chronic thyroid follicular hyperplasia, prolonged lymphocytic thyroiditis with hypothyroidism, and sufficient regional irradiation have all been implicated.24,30 Some of these risk factors may also be found in human medicine, such as a low-iodine diet and radiation exposure. 56 In humans, there is also an association between chronic inflammation and the development of thyroid carcinoma. 56 Additional human risk factors include having a history of goiter or thyroid nodules, a family history of thyroid carcinoma, and obesity. 56
For iodine deficiency and prolonged lymphocytic thyroiditis, it is speculated that FTCs may retain thyroid-stimulating hormone sensitivity. Thyroid-stimulating hormone is the main regulator of thyrocyte differentiation and proliferation, so it may then act as a growth factor causing chronic overstimulation and subsequent neoplastic transformation. 43 In contrast, MTCs may be preceded by hypercalcemia (eg, primary hyperparathyroidism or hypercalcemia of malignancy).47,48 In bulls, a relationship between chronic dietary intake of excessive calcium and MTCs is proposed, but there are no reports evaluating this in dogs. 47
Recent studies have attempted to explore the expression of markers that may contribute to CTC development, including those markers found on cancer-associated fibroblasts, which are activated fibroblasts that promote cancer cell survival, invasion, and metastasis, induce angiogenesis, and attenuate sensitivity to chemotherapeutics.9,42 Markers investigated include insulin-like growth factor (IGF)-1, vascular endothelial growth factor (VEGF), fibroblast growth factor (FGF)-2, and their receptors (eg, IGF-1R), which are often expressed during tumor cell interactions with the stroma in the tumor microenvironment.9,42 More specifically, IGF-1, IGF-1R, and VEGF were more highly expressed in fibroblasts and endothelial cells of compact FTCs compared with healthy glands; IGF-1R was more highly expressed in fibroblasts of compact FTCs compared with mixed FTCs; FGF2 was more highly expressed in endothelial cells of compact FTCs than healthy glands or mixed FTCs. 42 These changes support the hypotheses that the stromal microenvironment has an effect on oncogenesis and that compact FTCs could represent the least differentiated of FTCs.13,25,30,42,43 One study found that a high percentage of neoplastic cells in 80% of examined FTCs and all examined MTCs expressed VEGF, which supports that it contributes to tumor progression. 9 Another study found several genes in both FTCs and MTCs that contribute to increased expression of the phosphatidylinositol-3-kinase (PI3K/AKT) signaling pathway, which is a pathway that promotes cell growth and survival. 11
There is a paucity of reported familial thyroid cancer in dogs. Some Dutch German longhaired pointers have 2 recessive deletion mutations within the thyroid peroxidase (thyroperoxidase) gene, which contributes to familial FTC in this breed. 60 Thyroid peroxidase is important in the production of thyroid hormones.47,48 Further investigation is needed to determine the presence of this derangement in CTCs of other breeds and species. Thyroid peroxidase mutations in humans have been previously associated with thyroid carcinoma, while inactivating thyroid peroxidase mutations in both humans and dogs have been shown to cause autosomal recessive congenital goitrous primary hypothyroidism. 60 In 2006, the first cases of familial MTCs in 3 related dogs were reported, but no RET mutations were identified, which contrasts hereditary MTCs (of which, 30% contain germline RET alterations) and multiple endocrine neoplasm (MEN) syndromes in people.7,11,14,17,18,24,29,35–37,48,55,60 MEN (multiple endocrine adenomatosis, MEA) in humans is a heritable syndrome with several subtypes which results in multiple neoplasms in several endocrine organs.24,41,48 MEN type 1 is associated with hyperplasia, adenomas, and/or carcinomas of the thyroid glands, adrenal cortex, and pituitary gland, while MEN type 2 is associated with pheochromocytomas and MTCs. 24 In veterinary species, only bulls have a similar and well-described MEN-like syndrome (type 2).24,48 MEN-like syndromes have been less commonly reported in other domestic animals, including dogs.24,48
Biochemical and Genetic Derangements and Applications
Altered growth factors, such as IGF-1, VEGF, and FGF-2, and their receptors are confirmed or thought to contribute to the progression of both human and canine thyroid cancers.9,44 In dogs, neoplastic cells as well as tumor-associated fibroblasts and/or endothelial cells of compact FTCs were found to have increased IHC expression of IGF-1, VEGF, FGF-2, and retinoic acid receptor α, which may contribute to tumor progression. In this study, mixed FTCs had intermediate expression levels that could further reflect the amount of differentiation present. 44 Another study has identified thyroglobulin and thyroid-stimulating hormone-receptor in tumor-associated fibroblasts and that proliferating cell nuclear antigen (a marker for cell mitosis) was expressed in neoplastic canine follicular cells and fibroblasts while TTF-1 was restricted only to neoplastic follicular cells. 43 The significance and implications of some of these findings remain unclear, and, similar for other cancers, growth factors and other peptides likely play a role in thyroid cancer pathogenesis. 43
In humans, IGF-1/IGF-1R is overexpressed in some types of thyroid carcinomas and is correlated with poor prognosis.31,42 VEGF contributes to tumor-induced angiogenesis controlled by neoplastic, stromal, and tumor-infiltrating cells, and one study correlated increased VEGF expression with poorer outcomes in compact FTCs. 42 FGF-2 is implicated in abnormal human thyroid growth as a mitogen and stimulator for endothelial cell growth but reports on FGF-2 levels in human thyroid carcinomas are contradictory and may depend on the degree of differentiation. 42
Both canine FTCs and MTCs tend to have overexpression of VEGF while MTCs tend to express higher levels of cyclooxygenase-2 and p-glycoprotein compared with FTCs. 9 These overexpressed molecules may be therapeutic targets in the future. For example, a preliminary study utilizing multitargeted tyrosine kinase inhibitor found promising results of remission in 4 of 15 dogs and stable disease in 8 of 15 dogs with thyroid carcinoma. 9 P-glycoprotein (ABCB1 gene) is an efflux pump involved in multidrug resistance and has been found in human chemotherapy-resistant MTC cell lines; targeting and inhibiting this molecule may improve chemotherapeutic efficacy in otherwise resistant tumors. 9 Cyclooxygenase-2 expression may be linked to tumor development, and studies in human thyroid cancer have found that increased cyclooxygenase-2 expression has a direct causal relationship with p-glycoprotein, meaning cyclooxygenase inhibitors may decrease p-glycoprotein expression and therefore improve tumor sensitivity to chemotherapeutic agents. 9
Another study found significant upregulation of VEGFR-1, VEGFR-2, PDPK1, AKT1, and AKT2 in canine FTCs and of EGFR, VEGFR-1, and PIK3CA in canine MTCs, which are all genes involved in the PI3K/AKT signaling pathway and could be candidates for therapeutic targets. 11 This pathway regulates numerous cell processes, including cell proliferation, differentiation, and survival. 39 In humans, copy number gains in receptor tyrosine kinase genes are associated with activation of the PI3K/AKT pathway and are involved in the pathogenesis of human FTC.19,32 This study’s conclusion was that activation of PI3K/AKT pathway could contribute to the pathogenesis of CTCs via promotion of cell proliferation, resistance to apoptosis (via derangement of cyclooxygenase-2 expression), and malignant transformation, but more research is still needed. 11 Interestingly, the most common mutations in human thyroid carcinomas (RAS [N, K, and H], PIK3CA, BRAF, PTEN, [follicular-derived carcinomas] and RET [MTCs]) are considered rare in dogs.1,11,14,23,55,56 One study corroborated patterns of expression of some of these genes (FTC expression of AKT2 and PIK3CA and MTC expression of RET) and provided additional differentially expressed genes on an mRNA level between normal canine thyroid glands, FTCs, MTCs, and a canine thyroid adenocarcinoma cell line. 23 This information may be used for selecting therapies, as encouraging results have been identified from human studies using PI3K/AKT signaling inhibitors as well as preliminary canine studies using toceranib phosphate (multitargeted tyrosine kinase inhibitor that targets VEGFR-2). 11
Epidermal growth factor-2 immunohistochemical expression has also been evaluated in canine CTCs, with nearly 50% of cases having moderate to strong expression, without apparent discrimination between FTCs and MTCs. 59 In human thyroid carcinomas, increased human epidermal growth factor-2 expression is correlated with worse prognostic indicators, but no correlation has been identified in CTCs. 59
These specific molecules, receptors, genes, and pathways are mentioned as they may represent therapeutic targets that may eventually play a role in precision medicine for canine patients. Furthermore, FTCs and MTCs in both human and canine patients appear to have genetic differences that contribute to tumorigenesis, which supports the argument of differentiating these tumors rather than consolidating them under the broad category of CTCs.
Conclusion
In conclusion, our understanding of CTCs is largely based on older literature, while new and old literature often fail to discriminate between FTCs and MTCs. In addition, the compact FTC subtype appears to represent the least differentiated subtype within the category of well-differentiated FTCs. There is exceptional histologic similarity between the compact subtype of FTCs and MTCs that warrants at least immunohistochemical investigation for a definitive diagnosis of either tumor. A continued lack of discrimination between these neoplasms may mask significant differences in biologic behavior, tumorigenesis, or optimal therapies leading to unreliable conclusions and/or inappropriate treatment protocols. While surgery is the treatment of choice for resectable FTCs and MTCs, unresectable neoplasms are often treated with ancillary modalities like chemotherapy, radioactive iodine, or radiation therapy. In human medicine, genotyping of neoplasms is often routine to help guide chemotherapy (precision medicine), and veterinary medicine will likely follow suit in the future. With this in mind, the importance of discriminating FTCs and MTCs becomes more obvious. This is also highlighted by the fact that some current studies have found differences in genetic mutations between FTCs and MTCs in both human and canine neoplasms.
Footnotes
Acknowledgements
The authors would like to thank the reviewers and editors of Veterinary Pathology for considering this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
