Abstract
Equine parvovirus-hepatitis (EqPV-H) is the causative agent of Theiler’s disease, or severe acute hepatic necrosis, in horses. However, it is poorly understood whether EqPV-H is associated with other histologic findings in horses with clinical liver disease. The objective of this study was to examine the prevalence and severity of EqPV-H infections in diagnostic liver samples. Archived formalin-fixed, paraffin-embedded (FFPE) liver samples (n = 98) from Cornell University and University of California, Davis, collected between 2007 and 2022 were evaluated for 15 individual histologic features and by EqPV-H in situ hybridization. EqPV-H was detected in 48% (n = 47) of samples. The most common histologic features of EqPV-H-positive samples included individual hepatocyte death (n = 40, 85%), lobular infiltrates (n = 38, 80%), portal infiltrates (n = 35, 74%), and ductular reaction (n = 33, 70%). Centrilobular necrosis, portal infiltrate, and individual hepatocyte death were positively associated with high viral load. Neutrophil infiltrates, bridging fibrosis, and portal edema were negatively associated with a high viral load. Only 4 of 49 tested samples were positive for equine hepacivirus by polymerase chain reaction (PCR), but the PCR assay was unreliable for FFPE tissues. In summary, this study demonstrates that EqPV-H is common in a variety of liver pathologies and should be considered as a differential diagnosis in cases of hepatitis other than Theiler’s disease.
Theiler’s disease, also known as serum hepatitis, was first described in 1918 by Sir Arnold Theiler in South Africa. 31 For almost 100 years, the cause of Theiler’s disease proved elusive. In 2018, deep sequencing of samples from a horse that died of Theiler’s disease identified a novel parvovirus, named equine parvovirus-hepatitis (EqPV-H). 6 Since that discovery, EqPV-H has been found in serum samples from horses around the world,1,2,6,11,12,13,14,16,17,18,24,33,34,32,37,40 with a seroprevalence between 15% and 34.7% and DNA prevalence between 7.1% and 37%. Despite this apparently high prevalence, very little is known about naturally occurring diseases associated with EqPV-H.
We and others have demonstrated widespread viral nucleic acid (NA) in naturally occurring cases of Theiler’s disease and scattered positive hybridization in cases of mild to moderate hepatitis in experimentally inoculated horses.32,37,39 While subclinical and mild hepatitis are the most common outcomes of experimental infections, it is unknown whether mild to moderate disease is observed under natural conditions as well. A recent study examined liver tissues from horses and donkeys in Austria with various histopathologic abnormalities, including cirrhosis, circulatory disorders, toxic, metabolic, inflammatory, and neoplastic liver diseases. 42 Out of the 84 cases examined, EqPV-H DNA was only detected in 2 cases by real-time polymerase chain reaction (PCR) of DNA extracted from formalin-fixed, paraffin-embedded (FFPE) sections. Interestingly, both cases involved metastatic neoplasia spread to the liver. With a 9% underlying prevalence of EqPV-H infection in Austria, 2 the authors concluded there was no evidence that EqPV-H was a significant cause of liver diseases aside from Theiler’s disease. However, we observed histopathological evidence of hepatitis associated with EqPV-H infection in a large cohort of racehorses at New York State racetracks with features that mimicked those described in experimental infections. 8 Therefore, we hypothesized that EqPV-H could still be an important cause of clinical hepatitis. In addition to EqPV-H, equine hepacivirus (EqHV), the closest relative to hepatitis C virus (HCV) in humans, is also known to cause acute hepatitis and suspected to cause chronic hepatitis in horses.4,9,19,23,25,30,35 A secondary objective was to assess whether EqHV contributes to clinical hepatitis cases.
Materials and Methods
Liver Samples
Archived FFPE liver samples from Cornell University and University of California (UC), Davis, collected between 2007–2022 and 2010–2021, respectively, were used for analysis. Liver tissue samples were collected either ante-mortem by percutaneous biopsy or during postmortem examination within 24 hours of euthanasia or death. A power analysis (https://clincalc.com/stats/samplesize.aspx) with alpha 0.05 and 80% power determined that a sample number of 7 per group was required to statistically differentiate whether at least 66% of cases could be attributed to EqPV-H infection versus the underlying background prevalence estimate of 15% of 100 horses. 6 For this calculation, we used the seroprevalence data as the estimate of population prevalence, rather than serum viremia, as a more stringent estimate since serum viremia can fluctuate below the limit of detection of the test in the chronic phase, but the liver could remain positive by in situ hybridization (ISH). Sufficient samples from each of the following initial diagnostic categories that were hypothesized to be related to parvoviral infection were included: (1) centrilobular to massive hepatic necrosis, also known as Theiler’s disease; (2) lobular hepatitis; and (3) portal hepatitis. Samples from the following initial diagnostic categories that were hypothesized to be unrelated to EqPV-H infection were also included: (1) cholangitis/cholangiohepatitis, (2) bridging fibrosis, and (3) toxic hepatopathy. Because insufficient samples were available from horses with pyogranulomatous hepatitis, lipidosis, vacuolar hepatopathy, or neoplasia, they were not included for formal analysis.
Histopathology, ISH, and Immunohistochemistry
Diagnostic records were reviewed by a board-certified veterinary anatomic pathologist (M.C.J.). To provide consistency between pathologists and institutions, the initial diagnosis for each submission was confirmed by M.C.J. and summarized into 1 or more of 12 categories based on the emphasis of the initial morphologic diagnosis or pathologist comment. All initial reports were written by board-certified veterinary anatomic pathologists. Two diagnoses were assigned to cases where each diagnosis was of similar severity. The 12 diagnosis categories included hepatic necrosis, megalocytosis, bridging fibrosis, lobular hepatitis, portal hepatitis, pyogranulomatous hepatitis, toxic hepatopathy, cholangiohepatitis, cholangitis, lipidosis, vacuolar hepatopathy, and neoplasia. Cases were assigned an initial diagnosis of toxic hepatopathy if the original comment or morphologic diagnosis emphasized toxic injury as the primary etiologic differential. Cases describing megalocytosis in the absence of fibrosis or ductular reaction and that reported a lower likelihood of pyrrolizidine alkaloid toxicity were assigned to the megalocytosis category.
Previous studies have described the histologic features of either experimental or naturally occurring EqPV-H infections with subclinical disease.8,32 To assess whether other lesions could be associated with EqPV-H infection in horses with clinical liver disease, we assessed each case for 15 individual histologic features (Table 1, Supplemental Table S1). To facilitate a consistent categorization, independent histologic examination was performed on archived FFPE blocks of liver tissues by a board-certified veterinary anatomic pathologist (M.C.J.). Tissue sections (4-µm-thick) were analyzed after staining with hematoxylin and eosin. Features were designated as either present (+) or absent (−).
Criteria description for the 15 histologic features.

ISH for EqPV-H was performed on liver samples. RNAScope probe V-EqPV-H-VP1 (Catalog no. 559991; Advanced Cell Diagnostics, Inc., Newark, CA) against the EqPV-H VP1 gene was applied to FFPE liver biopsy samples, exactly as previously described.32,37 Each case was assigned a score for ISH (Table 2). For immunohistochemistry (IHC) of select samples, 4-µm-thick paraffin sections were submitted to the New York State Animal Health Diagnostic Center for CD3, IBA1, and CD20 immunolabeling. Briefly, sections were immunolabeled for CD3 (ready-to-use, PA0553; Leica, Deer Park, IL), IBA1 (1/3000 dilution, 019-19741; Wako, Richmond, VA), or CD20 (1/400 dilution, PA5-16701; ThermoFisher/Invitrogen, Waltham, MA). The immunolabeling procedure was performed using a Leica Bond Max automated IHC stainer. Leica Bond Polymer Refine Detection (DS9800, Leica) with 3,3’-diaminobenzidine (DAB) was used as the chromogen. Slides were counterstained with hematoxylin (DS9390; Leica). The control tissues were equine lymph node, spleen, and tonsil and labeled as expected. Negative controls were isotype-matched mouse monoclonal antibodies or irrelevant rabbit polyclonal antibodies.
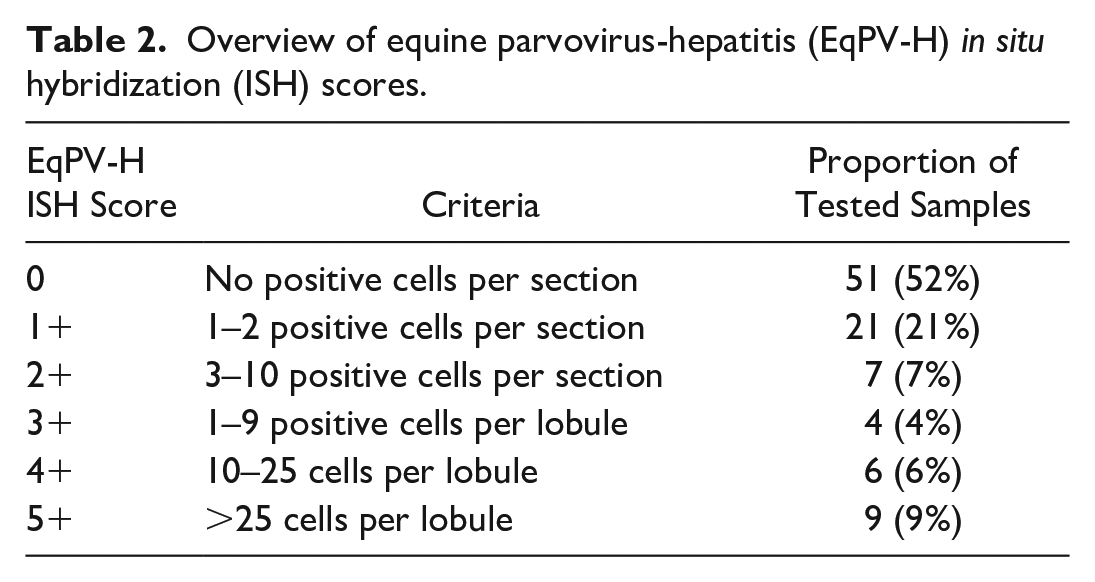
Overview of equine parvovirus-hepatitis (EqPV-H) in situ hybridization (ISH) scores.
Detection of EqHV by Reverse Transcription-Quantitative PCR
Four 25-µm scrolls of FFPE liver tissue per sample were submitted to the New York State Animal Health Diagnostic Center for RNA extraction and EqHV reverse transcription-quantitative polymerase chain reaction (RT-qPCR). Adequate tissues for EqHV PCR were only available from the Cornell University cases. An additional 17 FFPE biopsies with known EqHV RT-qPCR status from serum and/or frozen or RNAlater preserved livers were submitted as controls to estimate FFPE EqHV RT-qPCR sensitivity, as formalin is known to damage RNA. RNA extraction was performed using the RecoverAll Total Nucleic Acid Isolation Kit for FFPE (AM1975, Invitrogen). RT-qPCR was performed as previously described using primers QANTI-5UF1 and R1. 34
Statistical analysis
Prior experimental infections have shown that horses can remain weakly positive for EqPV-H by ISH without hepatitis, where EqPV-H appears to be in a quiescent state. 32 An ISH score of 3+ or greater, indicating ≥1 positive cell per lobule, is interpreted to correlate with active infection where the virus is contributing to pathology. Therefore, 2 dependent variables were analyzed: (1) EqPV-H ISH positive versus negative and (2) EqPV-H ISH ≥3+ versus <3+. First, the prevalence of EqPV-H-positive cases among diagnoses with at least 7 cases were compared to the estimated population prevalence of 15/100 by the hypothesis test for 2 populations. 6 Because the 15% population prevalence is based on a small sample set of only 100 horses, we applied a more stringent analysis by using a proxy group of 100 healthy horses and 15 positives, instead of comparing our data to a set population prevalence of 15%. Second, chi-square analysis was used to assess whether individual histologic features are associated with an increased risk of liver being EqPV-H positive or ≥ 3+ using ISH. These features were then built into a decision tree using partition predictive modeling to develop an algorithm for recommending EqPV-H diagnostic testing on clinical samples. In addition, the age, sex, and breed distribution of positive and negative horses were compared by t-test and chi-square analysis. Analyses were performed in JMP Pro version 14.0 (SAS Institute Inc., Cary, NC). Significance was set at P < .05.
Results
Study Population and Histopathologic Assessment
A total of 98 FFPE liver samples were collected from the archives of the Cornell University College of Veterinary Medicine (n = 53, 54%) and the UC Davis College of Veterinary Medicine (n = 45, 46%). Samples from Cornell University and UC Davis were collected between 2007–2022 and 2010–2021, respectively. Samples included biopsies (n = 69, 70%) and tissues collected from postmortem examinations (n = 29, 30%). The most common breeds in this sample set were Thoroughbreds (n = 20), Quarter Horses (n = 19), and Arabians (n = 8) (Supplemental Tables S2 and S3). There were 43 mares, 39 geldings, 10 stallions, and 6 horses of unreported sex. The ages of horses ranged from 3 months to 33 years, with an average age of 14 years.
The initial diagnosis for each submission was summarized into one or more of the 12 categories, based on the emphasis of the initial morphologic diagnosis or pathologist comment, and included hepatic necrosis (n = 15), megalocytosis (n = 4), bridging fibrosis (n = 10), lobular hepatitis (n = 17), portal hepatitis (n = 15), pyogranulomatous hepatitis (n = 2), toxic hepatopathy (n = 22), cholangiohepatitis (n = 12), cholangitis (n = 12), lipidosis (n = 2), vacuolar hepatopathy (n = 2), and neoplasia (n = 1) (Supplemental Tables S2 and S3). Cases of hepatic necrosis were characterized predominantly by acute centrilobular to massive hepatocyte necrosis with variable degrees of portal and lobular inflammatory cell infiltrates and were diagnosed as Theiler’s disease at the time of the initial diagnosis. Cases designated as toxic hepatopathies had evidence of individual cell death, vacuolar change in hepatocytes, anisokaryosis and/or anisocytosis, ductular reaction, variable degrees of fibrosis, and portal infiltrates of inflammatory cells. Many of the cases in the toxic hepatopathy category listed pyrrolizidine alkaloid toxicity as a top differential diagnosis. The single case of neoplasia was diagnosed as likely lymphoma by the original pathologist.
Each histologic section was examined for 15 individual histologic features by a board-certified veterinary anatomic pathologist (M.C.J.) (Table 1). The most common histologic features for the entire study population were individual hepatocyte death (n = 76, 78%), lobular infiltrates (n = 75, 76%), portal infiltrates (n = 71, 72%), and ductular reaction (n = 77, 79%) (Supplemental Table S1).
EqPV-H Detection by ISH
ISH was performed on each FFPE liver tissue sample to detect EqPV-H NA. In the study population (n = 98), 47 (48%) samples had positive hybridization for EqPV-H NA. The age of positive horses ranged from 3 months to 33 years with an average age of 12.6 years. EqPV-H-positive horses included 21 mares (44.7%), 17 geldings (36%), 6 stallions (12.7%), and 3 horses of unreported sex. Positive horses included 10 Thoroughbreds (23%), 14 Quarter Horses (32%), and 20 other breeds (45%). The distribution of age (P = .061), sex (P = .64), and breed (P = .92) was not significantly different between EqPV-H-positive and EqPV-H-negative horses. Scoring of the hybridization was used to assess the severity of infection in each individual sample (Table 2, Supplemental Tables S1 and S2). In samples with 3+ or greater, there was evidence that EqPV-H was involved in the pathology. For example, hybridization was found in dead hepatocytes or in clusters of inflammatory cells. Eleven of the 13 samples with severe centrilobular necrosis had dense, widespread hybridization (Fig. 1a). In areas of necrosis, hybridization was present but reduced compared to regions with identifiable cells (Supplemental Figure S1). All 9 of the samples with the highest hybridization scores were cases with centrilobular necrosis that had been diagnosed as Theiler’s disease (Table 2, Supplemental Table S2). The 2 cases that had been diagnosed as Theiler’s disease but were negative for hybridization were unique in that they had centrilobular necrosis but also had other features that were different from EqPV-H-positive cases. Specifically, case 48 had no individual apoptotic or necrotic hepatocytes in periportal regions and no significant inflammation, and case 50 had severe, diffuse canalicular cholestasis with no individual apoptotic or necrotic hepatocytes and minimal inflammation (Supplemental Table S3). Conversely, all EqPV-H-positive cases with centrilobular necrosis had individual apoptotic or necrotic hepatocytes in periportal regions and moderate inflammation in portal tracts and lobules.
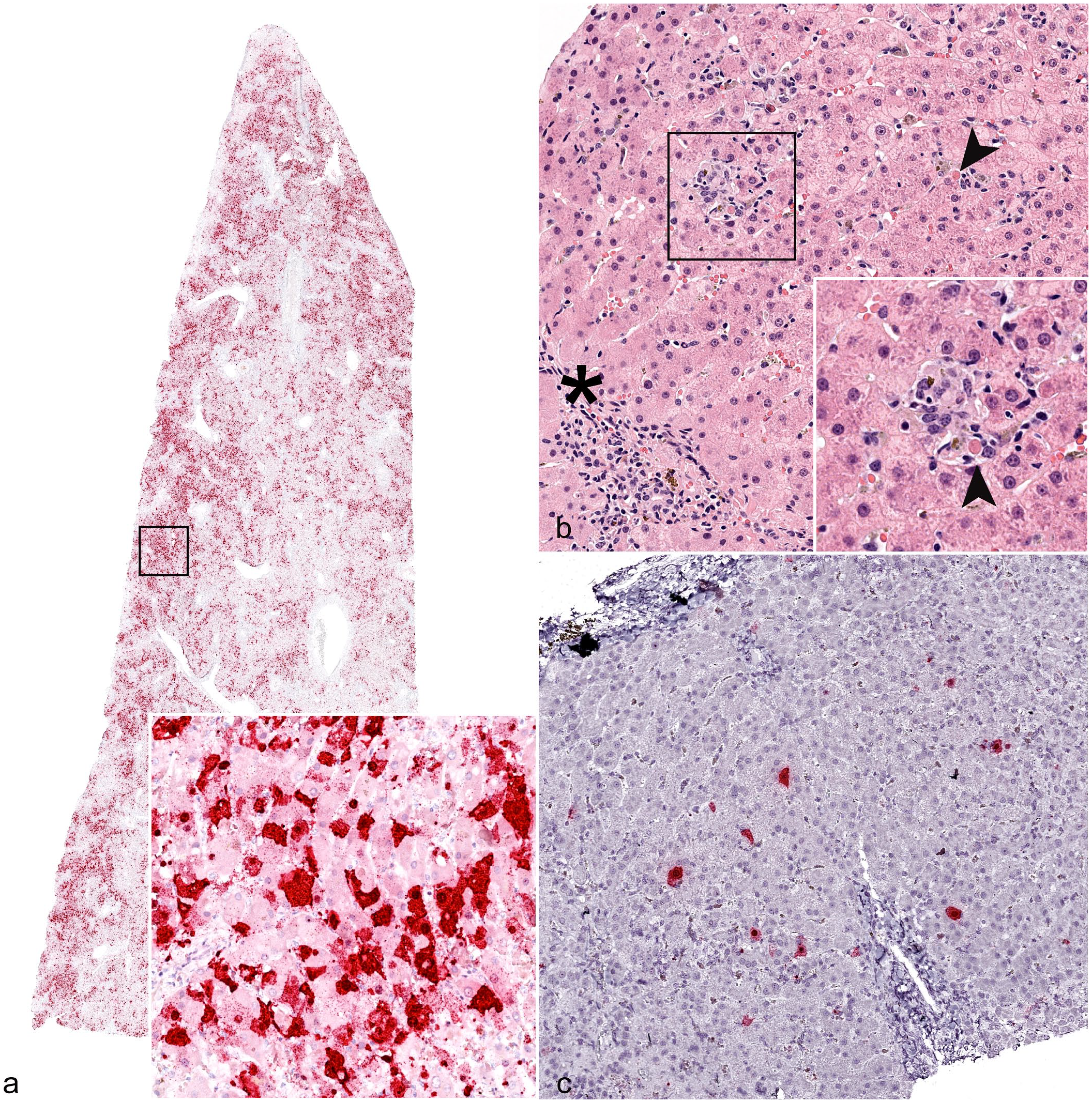
Histology and in situ hybridization (ISH) for equine parvovirus-hepatitis (EqPV-H) nucleic acid (NA), liver, horse. (a) Widespread hybridization of hepatocytes in a case of Theiler’s disease given a 5+ ISH score. Inset: higher magnification of area highlighted by black box, showing hepatocytes with positive hybridization. Case 46. EqPV-H ISH. (b) Representative histology of equine parvoviral hepatitis with multifocal lobular infiltrates of macrophages and small lymphocytes occasionally associated with individual dead hepatocytes (arrowheads) and increased mononuclear inflammatory cells in the portal stroma (asterisk). Inset: higher magnification of area highlighted by black box, showing inflammatory cells and an individual dead hepatocyte (arrowhead). Case 36. Hematoxylin and eosin. (c) Scattered hybridization of hepatocytes in the case from (b), which was assigned a 4+ ISH score. Case 36. EqPV-H ISH.
Robust hybridization for EqPV-H NA in all cases was present in the cytoplasm and nucleus of hepatocytes (Fig. 1), although rare hybridization puncta were observed in unidentified cells within portal tracts and sinusoids as well. All horses that were known to be serum qPCR-positive for EqPV-H at the time of sample submission were also positive by ISH (Supplemental Table S2). The prevalence of EqPV-H detection in each category with at least 7 cases was compared to the population prevalence of 15/100 to determine whether specific diagnoses were associated with EqPV-H. 6 We found that horses with a diagnosis of cholangitis, lobular hepatitis, toxic hepatopathy, and hepatic necrosis were more likely to be infected with EqPV-H than a healthy horse (Fig. 2). All except one of the cases assigned an initial diagnosis of “toxic hepatopathy” had pyrrolizidine alkaloid toxicity listed as a primary etiologic differential diagnosis in the original report, based on the presence of megalocytosis combined with other features. To our knowledge, none of the cases in this study had follow-up testing for pyrrolizidine alkaloids in liver tissue.
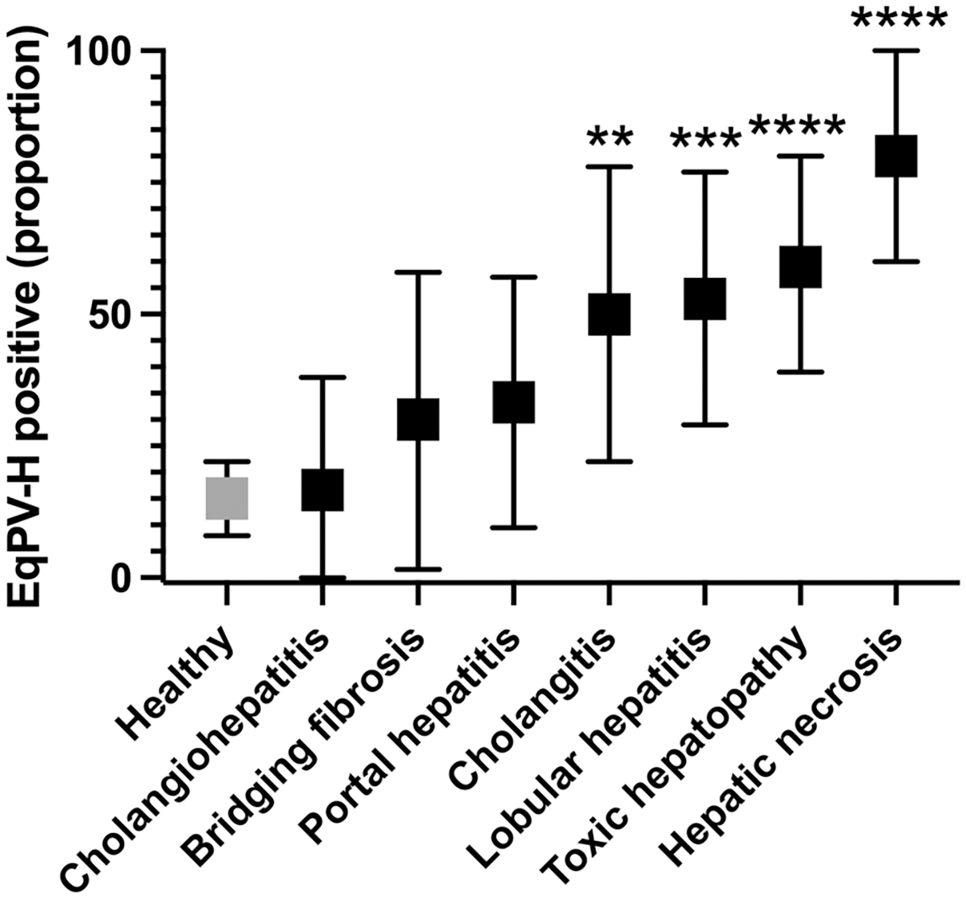
Prevalence of equine parvovirus-hepatitis (EqPV-H) infection in horses with 7 diagnoses compared to the healthy population. The healthy population was set at 15 positive horses out of 100, based on the work of Divers et al. 6 Population proportion and 95% confidence interval are shown. Significant difference from the healthy population according to the hypothesis test for two proportions is indicated as: *P < .05, **P < .01, ***P < .001, ****P < .0001.
The most common histologic features of EqPV-H-positive samples on hematoxylin and eosin–stained sections were individual hepatocyte death (n = 40, 85%), lobular infiltrates (n = 38, 80%), portal infiltrates (n = 35, 74.4%), and ductular reaction (n = 33, 70%) (Fig. 1b). Features significantly associated with EqPV-H infection were centrilobular necrosis and the absence of neutrophil infiltrates or ductular reaction (Table 3). Because EqPV-H can be detected in clinically normal horses, the analysis was repeated to determine if there was an association between individual histologic features and infection in cases with EqPV-H ISH scores of 3 and greater, which are more likely to indicate an active or currently pathologic infection.11,18,24,32,41 We found centrilobular necrosis, individual hepatocyte death, and portal infiltrate to be positively associated with EqPV-H 3+, while bridging fibrosis, portal edema, and neutrophil infiltrates were negatively associated with EqPV-H 3+ (Table 4). A partition-predictive model was developed to determine when EqPV-H infection is more likely based on features identified on histology (Fig. 3; Supplemental Figure S2). For this analysis, we targeted cases with EqPV-H 3+ as cases where EqPV-H was likely to be playing a significant role in the liver pathology. In essence, horses with centrilobular necrosis are highly likely to have an ISH score of 3+. If centrilobular necrosis is not present, the presence of individual hepatocyte death and lack of neutrophil infiltrates are most associated with ISH 3+.
Association between equine parvovirus-hepatitis (EqPV-H) detection by ISH and histologic features assessed by chi-square analysis.
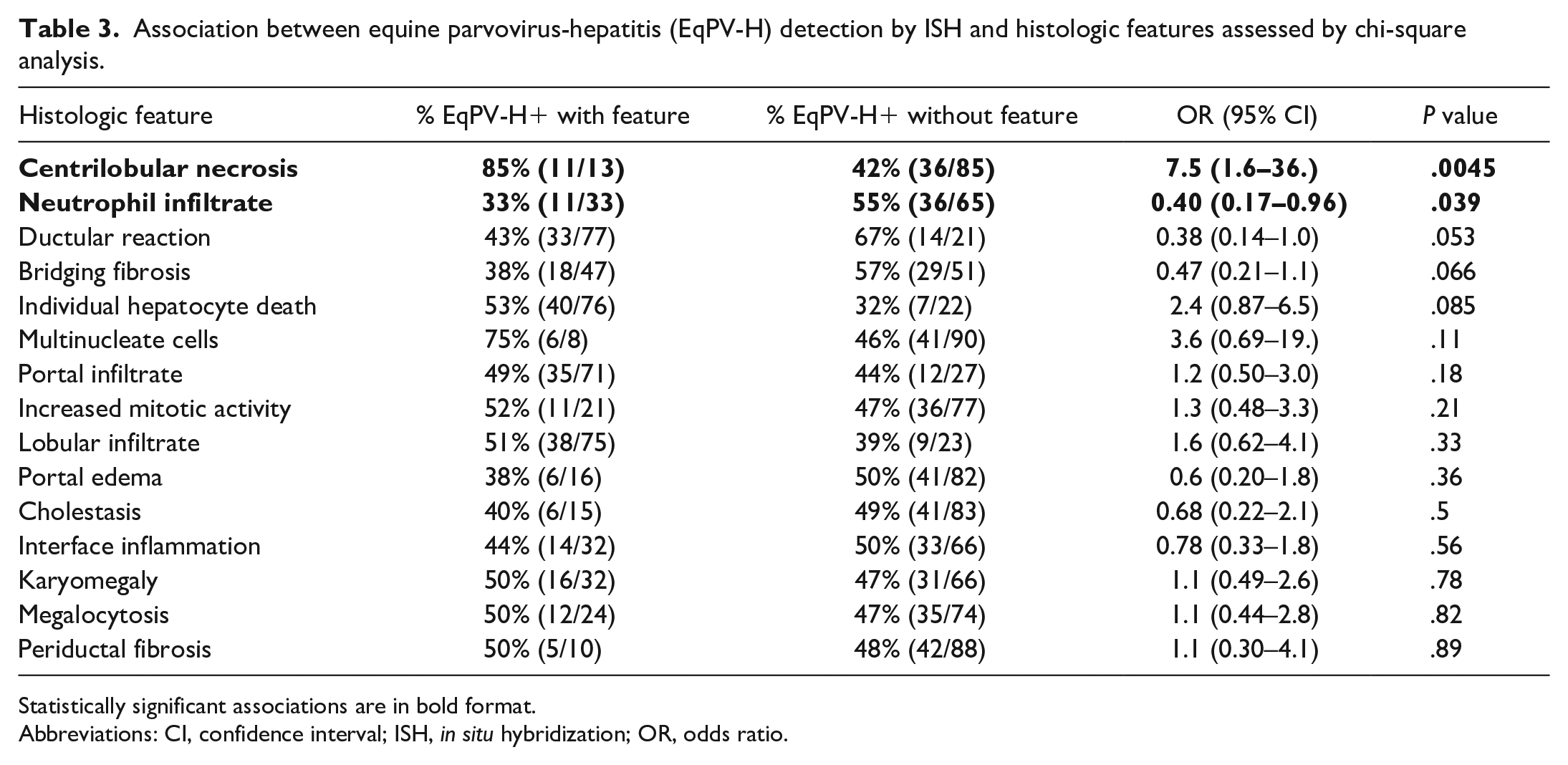
Statistically significant associations are in bold format.
Abbreviations: CI, confidence interval; ISH, in situ hybridization; OR, odds ratio.
Association between equine parvovirus-hepatitis (EqPV-H) 3+ ISH samples and histologic features assessed by chi-square analysis.
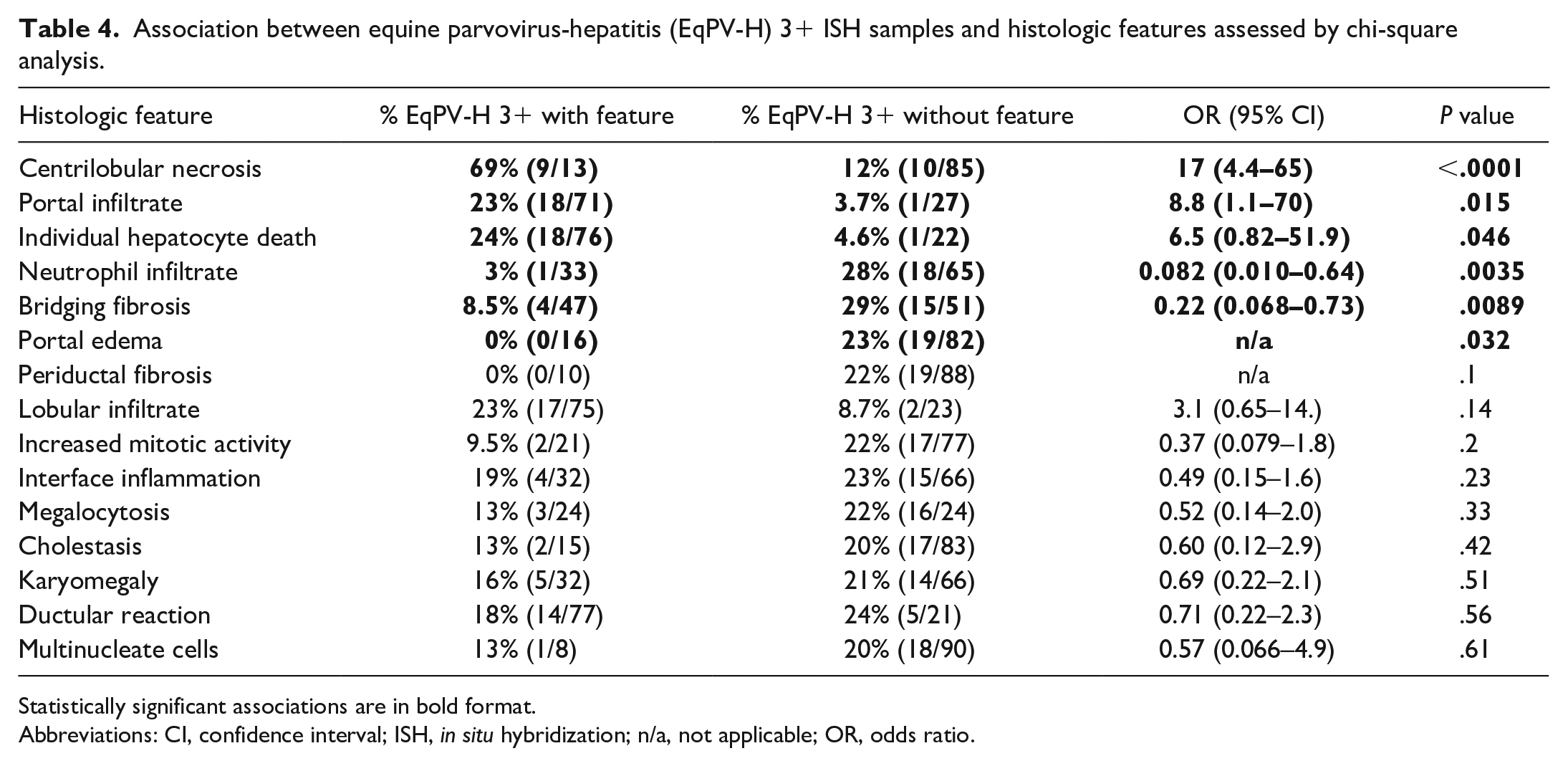
Statistically significant associations are in bold format.
Abbreviations: CI, confidence interval; ISH, in situ hybridization; n/a, not applicable; OR, odds ratio.
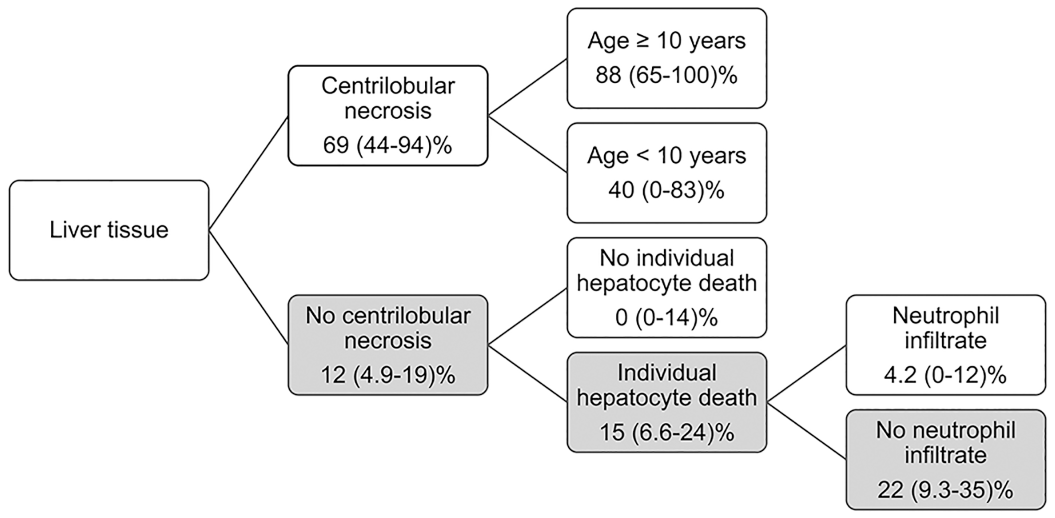
A partition-predictive model used to develop recommendations for equine parvovirus-hepatitis (EqPV-H) testing. Percentages show estimated proportion and 95% confidence interval of the proportion of those cases that will have an EqPV-H in situ hybridization (ISH) score of 3+ or above. Horses with centrilobular necrosis are highly likely to have ISH 3+. If centrilobular necrosis is not present, the features in gray support that EqPV-H ISH testing could be useful. Summary clinical decision tree is shown in Figure 5, and a complete analysis is shown in Supplemental Figure S2.
Megalocytosis and multinucleated hepatocytes, two histologic features not previously reported in cases of EqPV-H infection, were also detected in EqPV-H-positive cases. Of the 24 samples with megalocytosis, 12 (50%) were positive for EqPV-H on ISH (Fig. 4a, b), and 3 (12.5%) were given an ISH score of 3+ or above. Multinucleated hepatocytes were rarely seen in the study population; however, 6 of the 8 cases with multinucleated hepatocytes were positive for EqPV-H hybridization.
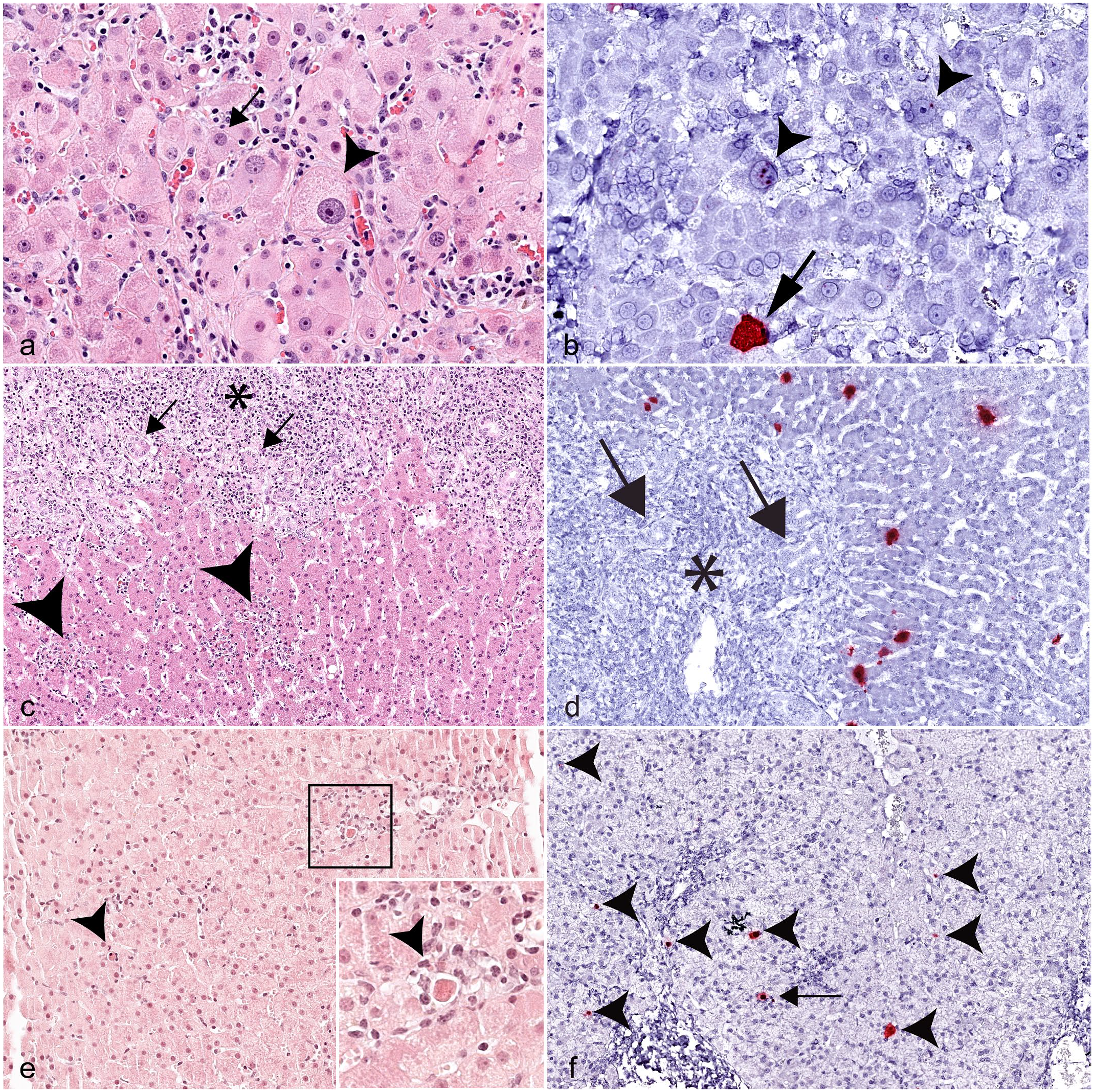
Histology and in situ hybridization (ISH) of equine parvovirus-hepatitis (EqPV-H)-positive cases, liver, horse. (a) Megalocytosis and karyomegaly (arrowhead) in an EqPV-H-positive case compared to surrounding hepatocytes (arrow). Case 35. Hematoxylin and eosin (HE). (b) Positive hybridization for EqPV-H nucleic acid in the nuclei of karyomegalic hepatocytes (arrowheads) and the cytoplasm of nearby hepatocytes (arrow). Case 35. EqPV-H ISH. (c) In addition to expansion of portal tracts by neoplastic round cells (asterisk) and increased numbers of bile ducts (arrows), focal aggregates of inflammatory cells and dead cells are scattered throughout the lobules (arrowheads). Case 34. HE. (d) Hybridization for EqPV-H nucleic acid (red) is limited to hepatocytes in lobules and is absent in neoplastic cells (asterisk) and bile duct epithelium (arrows). Case 34. EqPV-H ISH. (e) Multifocal random lobular hepatitis with individual cell death and inflammatory cells (arrowheads) in a foal. Inset: higher magnification of area highlighted by black box, showing inflammatory cells and a dead cell. Case 30. HE. (f) EqPV-H hybridization in hepatocytes (arrowheads), some of which are associated with inflammatory cells (arrow). Case 30. EqPV-H ISH.
Only one case in the study population (case 34) had histologic evidence of neoplasia in the liver, either primary or metastatic. Histologic evaluation of that liver sample showed widespread expansion of the portal tracts by round cells with frequent mitotic figures (Supplemental Figure S3). IHC revealed that this proliferative round cell population was positive for CD3 (T cells) and negative for IBA1 (macrophages) and CD20 (B cells), consistent with the initial diagnosis of lymphoma (Supplemental Figure S3). Within the portal regions, increased numbers of medium-sized bile ducts were noted, some of which contained luminal aggregates of neutrophils (Fig. 4c). Increased numbers of individual necrotic cells were found at the interface between the round cell population and hepatic lobules. Small clusters of neutrophils or small lymphocytes were found scattered throughout hepatic lobules and were often associated with shrunken, eosinophilic, individual necrotic cells or small lakes of bile (Fig. 4c). Only lobular hepatocytes, not the neoplastic cells or bile duct epithelium, were positive for hybridization of EqPV-H NA (Fig. 4d).
Two foals, aged 3 and 6 months, were included in this study. Surprisingly, both cases had histologic evidence of hepatitis with individual hepatocyte death and were positive for EqPV-H NA hybridization (Fig. 4e, f).
EqHV Detection by RT-qPCR
Although this study was designed to focus on EqPV-H, EqHV is also known to cause hepatitis in horses with findings that overlap with those associated with EqPV-H infection, such as individual hepatocyte death and lobular and portal infiltrates.4,9,19,23,25,30,35 Therefore, we performed RT-qPCR for EqHV on RNA extracted from 49 Cornell University FFPE tissue samples.
Because formalin fixation can degrade RNA and might result in false negative samples, we tested the efficacy of our protocol using 15 control samples known to be EqHV positive. No control sample stored longer than 2 years in paraffin tested positive. Eight samples were from horses that had previous fresh or RNAlater preserved liver RT-qPCR-positive results and had been stored 1–5 years in paraffin blocks. Two of these were inconclusive, and the rest were negative. Seven samples were from horses that had positive serum RT-qPCR at the time of biopsy and had been stored in paraffin for less than 1–4 years. The 4-year-old sample was inconclusive, and the rest were positive. Two negative controls were included from serum RT-qPCR-negative, seronegative, naïve horses.
In our study population, 4 horses tested positive for EqHV, with 2 of those also testing positive for EqPV-H by ISH (cases 23 and 35, Supplemental Tables S2 and S3). Review of the EqPV-H ISH showed active pathology associated with EqPV-H hybridization in both cases. All 4 EqHV-positive cases had evidence of individual hepatocyte death and lobular infiltrates; 3 had ductular reaction; and 2 had evidence of fibrosis, portal infiltrates, karyomegaly, and neutrophil infiltrates. Case 35 was documented to have persistent EqHV infection for at least 11 months. The age range for EqHV-positive horses was between 12 and 32 years, with an average age of 20 years.
Discussion
In this study, we investigated the prevalence of EqPV-H in archived liver tissues of horses with various diagnosed liver disorders, with a special focus on inflammatory diseases. Specifically, we hypothesized that EqPV-H is associated with lobular hepatitis and designed our study to examine cases of Theiler’s disease and lobular hepatitis, while using other diseases as control groups given the background prevalence of infection. Our data demonstrated that EqPV-H is associated with a wide spectrum of histologic lesions, which are similar to those observed in experimental infections and associated with disease manifestations ranging from subclinical hepatitis to Theiler’s disease.
A recent study examined 84 equid liver samples submitted to the Institute of Pathology in Vienna, Austria, and found only 2 cases (2%) to be positive for EqPV-H using real-time PCR of FFPE-extracted DNA. 42 The prevalence in our study was much higher, with 48% EqPV-H-positive samples identified by ISH. A variety of factors could have played a role in this difference, with the technique first and foremost used to identify EqPV-H. We used ISH for the detection of EqPV-H based on the fact that ISH is considered more sensitive than PCR as it can still detect fragments of NA that would otherwise be too small to facilitate PCR, 20 and ISH has an increased sensitivity that allows single copies of NA to be visualized. 38 In our study, we identified 21 EqPV-H-positive samples with only rare hybridization (21/47; ~45%). These cases might represent subclinical or persistent infection, although early stages of infection cannot be excluded. Second, the case selection approach was targeted to identify cases with specific morphologic diagnoses hypothesized to be related or unrelated to EqPV-H infection, which could have increased the detection of positive samples. Third, regional differences in prevalence might also have played a role, although this seems less likely. In our study, the prevalence between samples from Cornell University in New York (19/44, 43%) and UC Davis in California (19/45, 42%) was similar when Theiler’s cases were excluded. Globally, seroprevalence is reported to be between 15% and 34.7%, and DNA prevalence between 7.1% and 37%.1,2,6,11,12,13,14,16,17,18,24,33,34,32,37,40 A recent study in Austria reported a seroprevalence of 30.1% and DNA prevalence of 8.9%. 2
Histologic features associated with experimental EqPV-H infection are much milder than those described for Theiler’s disease.3,6,10,24,33,34,32,37,40 Previous reports that included histopathology of EqPV-H-positive horses described lobular hepatitis with lymphocytic infiltrates, satellitosis, hepatocellular degeneration and necrosis, and portal infiltrates.3,6,8,10,24,32 In our present study, the most common histologic features in EqPV-H-positive cases were individual hepatocyte death, lobular infiltrates, portal infiltrates, and ductular reaction. With the exception of ductular reaction, these features have been described previously in cases of mild to moderate hepatitis in experimentally infected horses. 32 While these 4 features are relatively common and nonspecific, we determined based on this present study that centrilobular necrosis, individual hepatocyte death, and the absence of neutrophil infiltrates were determined to be the most discriminatory features to guide diagnostic pathologists and clinicians when weighing ancillary diagnostic test decisions. Several of the cases that had moderate hybridization scores were diagnosed as toxic hepatopathies, cholangiohepatitis, or bridging fibrosis, demonstrating the need for a broader interpretation of these diagnoses. Moreover, it is possible that these cases represent comorbidities where the virus takes advantage of tissue regeneration or relative immunosuppression.
Other uncommon histologic features that were seen in EqPV-H-positive samples included multinucleated hepatocytes, megalocytosis, and karyomegaly. Multinucleated hepatocytes with more than 2 nuclei in nonneoplastic disease are a rare phenomenon in domestic animals in general, and particularly in horses, 5 although there is a single report of Theiler’s disease in a Canadian horse with multinucleated hepatocytes. 28 In animals, neonatal giant cell hepatitis has been reported in cats, cattle, pigs, and foals. 5 For the latter, multinucleated hepatocytes have been described in stillborn foals and fetuses infected with leptospirosis.21,29 In humans, giant-cell hepatitis is well described and is also most commonly seen in neonates.26,36 In adult humans, hepatocyte multinucleation is a nonspecific feature and can occur as a result of many diseases, including viral hepatitis. 26 In this study, 6 of the 8 samples with multinucleated hepatocytes were positive for EqPV-H NA, although the association was not statistically significant. Additional features observed in EqPV-H samples in this study were megalocytosis and karyomegaly, which are also nonspecific just like multinucleation. Nonetheless, megalocytosis and karyomegaly in horses is commonly attributed to toxicity and, specifically, pyrrolizidine alkaloid toxicosis.5,27 Our findings suggest that these features might be less pathognomonic for toxicity than initially appreciated.
One of the unexpected findings in this study was the detection of EqPV-H in cases of hepatitis in foals. We were the first to report EqPV-H in foals as determined by PCR; however, there was no evidence of illness in this population, and there are no cases of Theiler’s disease reported in foals to date. 32 A recent study in Canada examining EqPV-H infections on a farm following a case of Theiler’s disease found that foals were more likely to be viremic and displayed higher viral loads than the mares on the same farm. 15 Importantly, none of the foals in that study had a clinical disease or significant elevations of liver values on serum biochemistry at the timepoint tested. In the present study, 2 foals aged 3 and 6 months had moderate hybridization for EqPV-H NA and mild-to-moderate hepatitis with lobular infiltrates, individual hepatocyte death, and ductular reaction. These findings suggest that EqPV-H should be considered a potential differential diagnosis for hepatitis in foals and warrant future follow-up studies.
In this study, all but 2 of the cases that had been diagnosed as Theiler’s disease based on the clinical profile and histologic features were positive for EqPV-H in ISH. It has previously been reported that not all cases classified as Theiler’s disease are EqPV-H infected. 34 Here, EqPV-H-positive cases had individual necrotic/apoptotic cells in periportal regions and moderate inflammation that extended into lobules, suggesting that these features might be useful to diagnosticians for determining the likelihood of EqPV-H infection prior to ancillary testing.
Although we cannot definitively assign causation to EqPV-H in this study, an important utility of ISH over PCR testing is the ability to observe distribution, viral burden, and relationship of infected cells and pathologic lesions. These features, combined with data from experimental studies, strongly suggest that cases with ≥3+ ISH hybridizations likely had at least a component of liver injury directly due to EqPV-H. However, additional studies are necessary to determine whether these features are a direct result of EqPV-H infection or reflect comorbidities that facilitate or exacerbate EqPV-H infection.
In the author’s experience (J.E.T./M.C.J.), clinical cases of presumed chronic hepaciviral hepatitis are sometimes co-infected with a high viral load of EqPV-H. As with case 35, this might explain the presence of 3+ EqPV-H in cases of fibrosing hepatitis despite there being little evidence that EqPV-H itself causes fibrosis.34,32 This serves as a reminder that more than one viral agent could be involved in cases of hepatitis. A major limitation of this study was the poor reliability of EqHV PCR on FFPE samples, likely because RNA is more sensitive to degradation than DNA. Prospective studies are likely required to better assess the role of EqHV in equine hepatitis.
In summary, we found that EqPV-H is present in a broad range of liver pathologies and should be considered a differential diagnosis in cases of hepatitis other than Theiler’s disease. This should assist anatomic pathologists and clinicians in interpreting hepatitis in biopsies or necropsies in the context of EqPV-H-positive PCR or ISH results. In addition, we have developed a decision tree for recommending EqPV-H testing based on certain histologic features detected in liver tissue (Fig. 5). Based on these and other recent findings, we suggest altering the terminology used to describe EqPV-H-related disease from the restricted context of “Theiler’s disease” to the more encompassing term “equine parvoviral hepatitis,” with a spectrum of lesions ranging from mild hepatitis to fulminant hepatic necrosis.
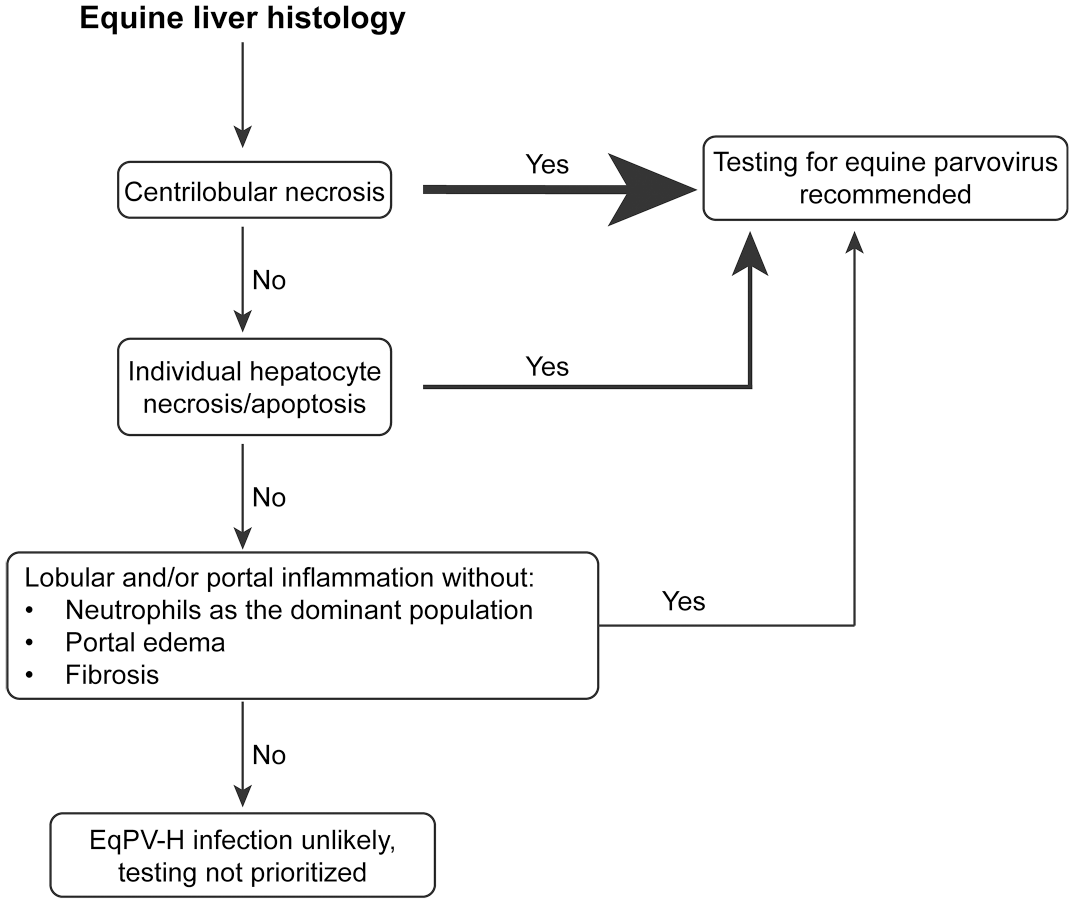
Decision tree to aid in recommendation to test for equine parvovirus-hepatitis (EqPV-H). The thickness of the arrow increases with the likelihood of a positive test result.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231214024 – Supplemental material for Naturally acquired equine parvovirus-hepatitis is associated with a wide range of hepatic lesions in horses
Supplemental material, sj-pdf-1-vet-10.1177_03009858231214024 for Naturally acquired equine parvovirus-hepatitis is associated with a wide range of hepatic lesions in horses by Mason C. Jager, Eunju Choi, Joy E. Tomlinson and Gerlinde Van de Walle in Veterinary Pathology
Footnotes
Acknowledgements
The authors would like to thank May Boggess of the Cornell Statistical Consulting Unit, the veterinary pathologists of the Cornell University Department of Population Medicine and Diagnostic Sciences, the staff of the New York State Animal Health Diagnostic Center, and the veterinary pathologists and staff of the University of California, Davis Veterinary Medical Teaching Hospital.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health (NIH) under the following awards: the Office of the Director under award number T32ODO011000 to M.C. Jager and the National Institute of Allergy and Infectious Diseases (NIAID) under award numbers K08AI163401 to M.C. Jager and K08AI141767 to J.E. Tomlinson; the Agriculture & Food Research Initiative Competitive Grant numbers 2020-67015-31297 to G.R. Van de Walle and 2022-67015-36343 to J.E. Tomlinson from the USDA National Institute of Food and Agriculture; and a Harry M. Zweig Memorial Fund for Equine Research grant to G.R. Van de Walle. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH and USDA.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
