Abstract
The accumulation of intraepithelial lymphocytes (IELs) is a histopathological feature of canine chronic enteropathy (CE), and IELs are considered the cells of origin of intestinal T-cell lymphoma (ITCL). However, the pathogenic mechanism of IEL activation in CE remains unclear. This study hypothesized that the expression of proinflammatory cytokines, associated with cytotoxic T/NK-cell activation, is upregulated in CE and ITCL, and examined the expression of IFN-γ, IL-2, IL-12p35, IL-12p40, IL-15, and IL-21 and the downstream signal transducers and activators of transcription (STAT) pathway in the duodenal mucosa of dogs without lesions (n = 11; NC), with IEL–CE (n = 19; CE without intraepithelial lymphocytosis), IEL+CE (n = 29; CE with intraepithelial lymphocytosis), and with ITCL (n = 60). Quantitative polymerase chain reaction (PCR) revealed that IFN-γ and IL-21 were higher in IEL+CE than in IEL–CE or NC. Western blot revealed upregulation of STAT1 and STAT3 in IEL+CE. Double-labeling immunohistochemistry revealed a positive correlation between the Ki67 index of CD3+ T-cells and IFN-γ expression levels. Immunohistochemistry revealed a higher ratio of p-STAT1-positive villi in IEL+CE and ITCL than IEL–CE and NC, which positively correlated with IFN-γ expression levels. Among the 60 ITCL cases, neoplastic lymphocytes were immunopositive for p-STAT1 in 28 cases and p-STAT3 in 29 cases. These results suggest that IFN-γ and IL-21 contribute to the pathogenesis of IEL+CE, and IFN-γ may be involved in T-cell activation and mucosal injury in CE. STAT1 and STAT3 activation in ITCL cells suggests a role for the upregulation of the STAT pathway in the pathogenesis of ITCL.
Keywords
Chronic enteropathy (CE) is a common gastrointestinal disorder in dogs that causes chronic diarrhea and vomiting, as well as protein-losing enteropathy in a subset of cases.3,18,48 However, its etiology and pathogenic mechanisms remain unclear. Histologically, CE is characterized by lymphoplasmacytic and/or eosinophilic infiltration, and sometimes by increases in intraepithelial lymphocytes (IELs).8,25 Since canine CE involves various inflammatory cells, such as CD4+ T-cells, CD8+ T-cells, plasma cells, and macrophages, 19 a more detailed understanding of the cytokine interactions between these inflammatory cells is essential for elucidating the pathogenesis of this disease. Previous studies on canine duodenal CE reported decreases in interleukin (IL)-17, IL-13, and IL-33 mRNA expression in German shepherd dogs,24,45 while others found no significant difference in cytokine mRNA expression patterns between dogs with and without CE.35,39 Therefore, canine CE is currently considered to be a complex group of diseases involving multiple factors, such as breed, the type of cellular infiltration in the lamina propria, and disease progression.
Intestinal T-cell lymphoma (ITCL) also needs to be considered in dogs with chronic gastrointestinal symptoms.8,50 Canine ITCLs are broadly divided into large cell lymphoma (LCL) and small cell lymphoma (SCL) based on the nuclear size of neoplastic lymphocytes. ITCL cases diagnosed by endoscopic biopsies are generally characterized by epitheliotropism of neoplastic lymphocytes.25,29 Since CE cases also have increased IELs,8,25 the histopathological features of CE and SCL in endoscopic biopsies may sometimes appear similar. However, the role of increased IELs in the pathogenesis of canine CE and ITCL remains unclear.
Human celiac disease is an autoimmune inflammatory enteropathy against gliadin and other related prolamins that is histologically characterized by increased IELs. 21 Celiac disease that responds poorly to a gluten-free diet is called refractory celiac disease and is classified into types 1 and 2 based on the immunophenotype of IELs and the results of clonality analysis. 44 Type 2 refractory celiac disease is more likely to progress to enteropathy-associated T-cell lymphoma (EATL) and has a poor prognosis.13,43 In celiac disease, gluten-specific CD4+ T-cells bind to HLA-DQ2 or HLA-DQ8 and secrete interferon-γ (IFN-γ) and other cytokines, including IL-21 and IL-2.27,46 IL-15 is also produced by enterocytes and macrophages stimulated by gliadin peptides. 31 These proinflammatory cytokines activate IELs and enhance innate immune responses and cellular injury by IELs. Activated IELs induce apoptosis of the epithelium via granzyme B-induced cytotoxicity, resulting in villous atrophy. 1
We previously classified canine CE cases as CE with intraepithelial lymphocytosis (IEL+CE) or CE without intraepithelial lymphocytosis (IEL–CE). 25 Histologically, villous atrophy and inflammatory cell infiltration were shown to be more severe in IEL+CE cases than in IEL–CE cases. In IEL+CE, the IELs were of the CD3+CD8–granzyme B+ immunophenotype, and the T-cell receptor clonality positivity rate was higher than that in IEL–CE, suggesting that canine ITCL originate from these IELs. The histopathological features, immunophenotype of IELs, and clonality results of canine IEL+CE and ITCL are similar to those of celiac disease and EATL, suggesting a similar pathogenesis. However, limited information is currently available on the cytokine expression pattern underlying T-cell activation in canine IEL+CE. Therefore, we hypothesized that similar to human celiac disease, the expression of proinflammatory cytokines associated with cytotoxic T/NK-cell activation is upregulated in canine CE. We investigated the expression of IFN-γ, IL-2, IL-12p35, IL-12p40, IL-15, and IL-21, all of which are known to directly act on and activate cytotoxic T/NK-cells, in CE and ITCL. We then focused on IFN-γ and IL-21 and analyzed downstream signal transducers and activators of transcription (STAT) and proliferative activity in T-cells. In addition, we explored possible mechanisms underlying mucosal injury in IEL+CE and ITCL, that is, expression of phospho-STAT1 (p-STAT1) and cleaved caspase-3, an apoptosis-related factor whose cleavage is facilitated by granzyme B, 20 in enterocytes.
Materials and Methods
Case Selection
Cases of canine CE diagnosed by endoscopic biopsy (IEL+CE, n = 29; IEL–CE, n = 19) were selected from the database of the Japan Small Animal Medical Center from 2019 to 2022, while cases of endoscopically diagnosed canine ITCL (SCL, n = 30; LCL, n = 30) were selected from the database of Japan Small Animal Medical Center and the Veterinary Medical Center of the University of Tokyo from 2013 to 2022. The inclusion criteria were as follows: (1) there was a histopathological diagnosis of CE, SCL, or LCL in the duodenum; (2) the main lesion was in the duodenum; (3) an intestinal mass was excluded by ultrasonographic or radiographic examination; (4) for CE cases, both formalin-fixed paraffin-embedded and fresh-frozen duodenal tissues were available; and (5) for ITCL cases, formalin-fixed paraffin-embedded duodenal tissues were available and clear membranous or cytoplasmic CD3 expression was present in neoplastic cells. Thirty-eight CE and all of ITCL cases were identical to the cases included in our previous study. 25
Normal duodenal mucosal tissue samples were also collected from 11 dogs that underwent endoscopy for evaluation of primary gastroesophageal disease. No clinical signs of intestinal disease were observed (such as diarrhea or hypoalbuminemia) and histological examination revealed normal duodenal mucosa. Of the 11 normal controls, 6 were included in quantitative PCR analysis and 5 were included in western blot analysis. The clinical information of all cases is summarized in Supplemental Tables S1 and S2.
Histopathology
Six endoscopic samples from the proximal and six from the distal duodenum (a total of 12 samples) were collected from each of the CE and ITCL cases. Mucosal samples were placed with the villous side facing up on filter paper before fixation to create a vertical section. The samples were fixed in 10% neutral buffered formalin solution, processed routinely, and embedded in paraffin wax. Four-micrometer-thick paraffin sections were deparaffinized, rehydrated through a graded series of alcohol, and stained with hematoxylin and eosin. A histopathological diagnosis was made based on the consensus of 3 veterinary pathologists (KK, KU, and JKC), including Japanese College of Veterinary Pathologists diplomates (KU and JKC). In this study, cases with lymphoplasmacytic infiltration accompanied by architectural changes including erosion, villous atrophy, and/or fibrosis were diagnosed as CE. Histopathological evaluations, including scoring of villous atrophy, were based on histopathological standards for the intestines established by the World Small Animal Veterinary Association Gastrointestinal Standardization Group. 14 Cases with more than 30 IELs per 100 enterocytes in the villous epithelium were considered to have intraepithelial lymphocytosis, 25 and cases in which intraepithelial lymphocytosis was observed in at least 3 out of 12 mucosal samples collected from the duodenum were classified as IEL+CE (n = 29). CE cases without intraepithelial lymphocytosis were classified as IEL–CE (n = 19). The diagnosis of ITCL was based on a dense monomorphic population of neoplastic lymphocytes with an abnormal cell size and distorted villous architecture.8,49 In each case, the nuclear size of neoplastic cells was evaluated relative to red blood cells (RBCs) as small (1-1.5 RBC), medium (1.5-2 RBC), or large (>2 RBC). ITCL cases were further classified into LCL (n = 30) and SCL (n = 30) according to their nuclear size as previously described: LCL, neoplastic cells are mainly large (>2 RBC); SCL, neoplastic cells are small to medium (<2 RBC).25,29
Total RNA and Protein Extraction
At least 2 mucosal samples were endoscopically collected from different sites of the duodenum and placed into a Tissue-Tek container, which was then filled with Tissue-Tek OCT compound gel (Sakura Finetek, Tokyo, Japan) and stored at –80°C until used. Samples were cut out from the compound gel and briefly washed with phosphate-buffered saline before total RNA and protein extraction. Total RNA was extracted from 2 mucosal samples in different tubes using an RNeasy Plus Mini Kit (Qiagen, Venlo, The Netherlands). Genomic DNA was removed from the samples using gDNA Eliminator Spin Columns (Qiagen). Total RNA concentrations were assessed by measuring spectrophotometric absorbance at 260 nm using the NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, Waltham, MA). The purity of RNA was also assessed by the ratio of absorbance at 260/280 nm (A260/A280), and A260/A280 of all samples was confirmed to be higher than 1.8. The quality of RNA samples was evaluated by electrophoresis on 1% degenerated agarose gels using RNA High for Easy Electrophoresis (BioDynamics Laboratory, Tokyo, Japan). Samples that clearly showed both 18S and 28S bands were used for subsequent reverse transcription.
For protein extraction, 2 mucosal samples from each case were placed in the same tube and homogenized in buffer containing 10 mM Tris-HCl (pH 7.8), 1% NP-40, 0.1% sodium dodecyl sulfate (SDS), 150 mM NaCl, 1 mM EDTA (pH 8.0), 2 mM Na3VO4, 10 mM NaF, proteinase inhibitor cocktail, alkaline phosphatase inhibitor, and distilled water. Lysates were incubated on ice for 30 minutes and centrifuged at 12,000 × g at 4°C for 10 min. Supernatants were collected, and protein concentrations were measured using the DC protein assay (Bio-Rad, Hercules, CA) according to the manufacturer’s instructions.
Reverse Transcription and Quantitative PCR Analysis
cDNA was synthesized from 700 ng of total RNA per 20 µL reaction volume using the PrimeScript RT reagent Kit with gDNA Eraser (Perfect Real Time, Takara Bio, Shiga, Japan), and all cDNA samples were then diluted 4-fold. Relative quantification was performed to analyze the expression levels of IFN-γ, IL-2, IL-12p35, IL-12p40, IL-15, IL-21, and 5 housekeeping genes: glyceraldehyde 3-phosphate dehydrogenase (GAPDH), succinate dehydrogenase complex flavoprotein subunit A (SDHA), TATA-binding protein (TBP), ribosomal protein S18 (RPS18), and ribosomal protein L32 (RPL32). Information on the primers used for cytokine- and housekeeping gene-encoding mRNA is shown in Table 1. Reaction efficiency was assessed for each primer set using 5-fold dilutions of the PCR amplicons of each cytokine and the reference gene and ranged between 90 and 110% (Supplemental Figure S1). The stability of the housekeeping genes was evaluated using NormFinder (https://moma.dk/normfinder-software), and GAPDH and SDHA were considered to be suitable reference genes.
Primer sequences used for quantitative polymerase chain reaction.

Abbreviations: IFN-γ, interferon-γ; IL, interleukin; GAPDH, glyceraldehyde 3-phosphate dehydrogenase; SDHA, succinate dehydrogenase complex flavoprotein subunit A; TBP, TATA-binding protein; RPL32, ribosomal protein L32; RPS18, ribosomal protein S18.
Quantitative PCR was performed using TB Green Premix Ex Taq II (Tli RNaseH Plus), ROX plus (Takara Bio), and the StepOnePlus Real-Time PCR System (Thermo Fisher Scientific) in a final reaction volume of 20 μL. In each reaction, 2 μL of a cDNA sample was used in the analysis of IFN-γ, IL-12p35, IL-12p40, IL-15, and housekeeping genes, while 6 µL of a cDNA sample was used for IL-2 and IL-21 because the mRNA expression levels of these cytokines were very low. Amplification conditions were 95°C for 60 seconds, followed by 40 cycles of 95°C for 5 seconds and 60°C for 30 s. A melting curve analysis was performed following every run to ensure a single amplified product for every reaction (1 cycle of 95°C 15 seconds, 60°C 30 seconds, 95°C 15 seconds; continuous acquisition mode from 60°C to 95°C with ramp rate of 0.3°C per second). All samples were examined in duplicate. Nuclease-free water and nonreverse transcription controls were used as negative controls. A sample with a known cycle threshold (Ct) value was included in all sample runs to control for run-to-run Ct variations. Gene expression in each case was quantified by averaging the Ct values of two duplicate measurements and then normalized by the expression ratio of each target gene to the geometric mean of the two reference genes (GAPDH and SDHA). The geometric mean between two different mucosal samples was used as a representative value. Relative expression levels were calculated by setting the average expression level of the normal control to 1.
RNA In Situ Hybridization
Colorimetric RNA in situ hybridization (ISH) was performed using RNAscope 2.5 HD Reagent Kit-Red (Advanced Cell Diagnostics, Newark, CA) according to the manufacturer’s instructions. Formalin-fixed paraffin-embedded tissues of 10 recent cases in each group (LCL, SCL, IEL+CE, IEL–CE, and normal control) submitted between 2019 and 2022 were included. Inclusion criteria were only the age of the specimens and, among included cases, the oldest specimen was collected 3 years 6 months before the experiment. An ISH probe targeting the mRNA sequence between nucleotides 2 and 1221 of canine IFN-γ (Accession NM_001003174) was used in the hybridization step. Probes for the canine peptidylprolyl isomerase B gene and the bacterial dihydrodipicolinate reductase gene served as positive and negative controls, respectively.
Double-labeling by RNA ISH for IFN-γ and immunohistochemistry for CD3 was performed to identify the types of cells expressing IFN-γ. After IFN-γ ISH probes were visualized, sections were blocked with 8% skim milk in Tris-buffered saline at 37°C for 40 min and incubated with an anti-CD3 antibody at 37°C for 40 min. After washing with Tris-buffered saline, a secondary antibody was applied and incubated at 37°C for 40 min. Labeled complexes were visualized with the diaminobenzidine chromogen and 0.03% hydrogen peroxidase in Tris-HCl buffer and then counterstained with hematoxylin. CD3 labeling was verified by single-labeling immunohistochemistry for CD3.
ISH labeling of the duodenal mucosa was examined at a medium magnification (10× objective) to select areas with the strongest positive hybridization signals. Within these areas, 5 fields were photographed at high magnification (40× objective, 0.104 mm2). The ratio of IFN-γ-positive areas in each image was quantified and averaged using ImageJ software (National Institutes of Health, Bethesda, MD). Details on quantification methods are summarized in Supplemental Figure S2.
Western Blot Analysis
Information on the primary antibodies used for western blotting and immunohistochemistry is summarized in Table 2. A western blot analysis using anti-dog IFN-γ, anti-dog IL-21, anti-p-STAT1, anti-p-STAT3, and anti-β-actin antibodies was performed to measure protein expression levels. Thirty-micrograms samples were dissolved in Laemmli Sample Buffer (Bio-Rad), which included dithiothreitol, and were then boiled for 5 min. Proteins dissolved in Laemmli Sample Buffer were separated using a 5% to 20% gradient polyacrylamide gel (ATTO, Tokyo, Japan) and transferred to a polyvinylidene fluoride membrane (Immobilon-P Transfer Membrane; Millipore). Membranes were blocked with 150 mM NaCl, 10 mM Tris-Base, and 40 mM Tris-HCl buffer (pH 7.8) containing 1% skim milk and 0.05% Tween-20 and were incubated with one of the primary antibodies at 4°C overnight and then with horseradish peroxidase-conjugated donkey anti-rabbit IgG (1:10000; Cytiva, Tokyo, Japan) at room temperature for 1 hour. Membranes were visualized with the Amersham ECL western blotting detection reagent (Cytiva) using Molecular Imager ChemiDox XRS+ with Image Lab software (Bio-Rad). Antibody stripping was performed using Restore Western Blot Stripping Buffer (Thermo Fisher Scientific). One identical sample was included in each membrane to control for variations. Protein expression in each case was quantified using Image Lab software (Bio-Rad) and normalized to the expression ratio of each protein to β-actin. Relative expression levels were calculated by setting the average expression level of the normal control to 1.
Primary antibodies used for western blotting and immunohistochemistry.
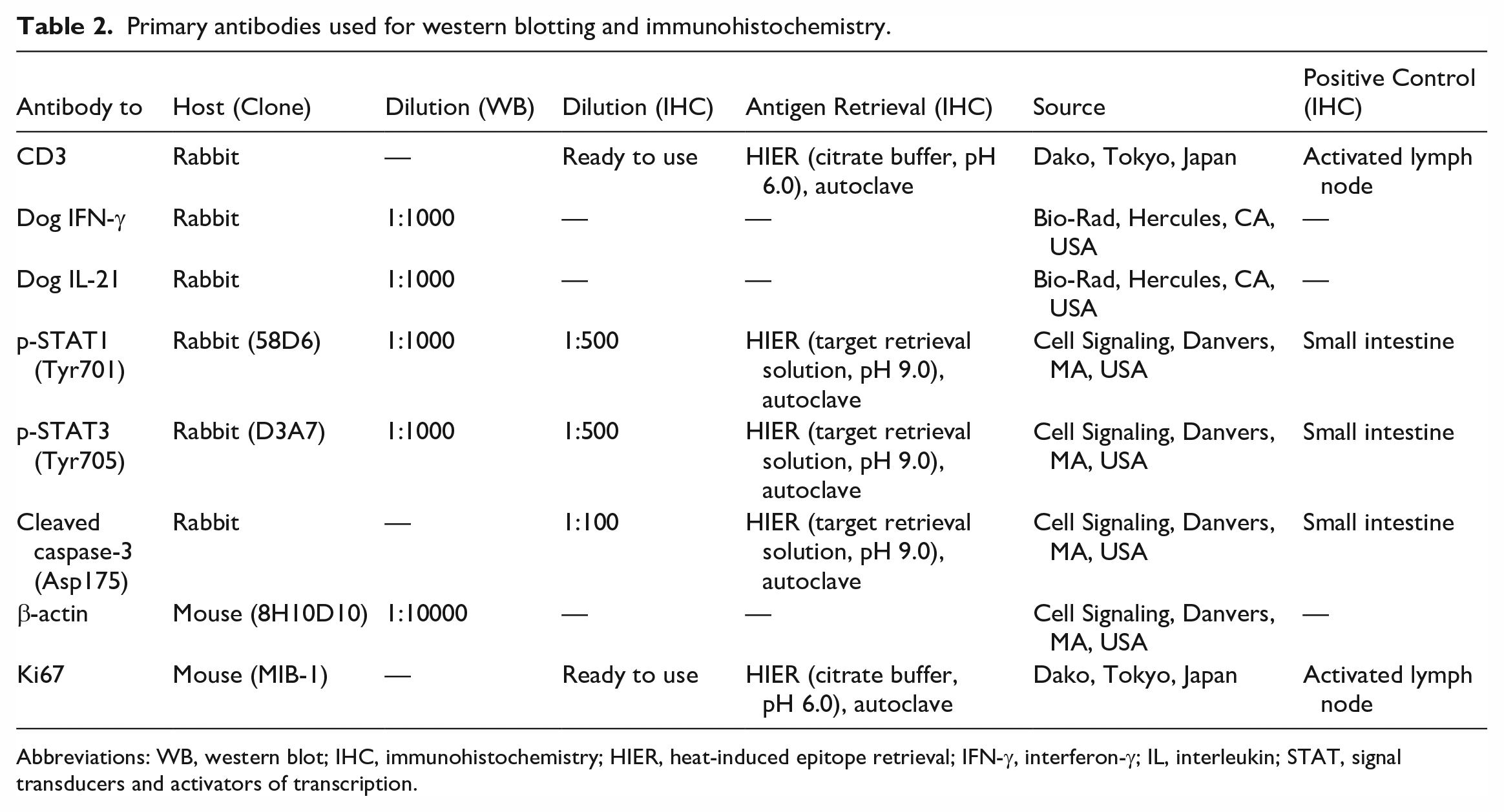
Abbreviations: WB, western blot; IHC, immunohistochemistry; HIER, heat-induced epitope retrieval; IFN-γ, interferon-γ; IL, interleukin; STAT, signal transducers and activators of transcription.
Immunohistochemistry
Immunohistochemistry for CD3, p-STAT1, p-STAT3, and cleaved caspase-3 was conducted using formalin-fixed paraffin-embedded tissues in the same manner as in our previous study. 25 Canine lymph nodes or normal small intestines were used as positive and negative tissue controls. Negative antibody controls were performed by applying an irrelevant antibody and buffer instead of the primary antibodies. The immunohistochemical labeling of villi for p-STAT1 and cleaved caspase-3 was evaluated in SCL, CE, and normal control cases by calculating the ratio of villi with 5 or more immunopositive enterocytes out of all villi. Regarding p-STAT1, cells with nuclear labeling were counted. All LCL, 5 SCL, and 1 IEL–CE cases were excluded from the immunohistochemical evaluation of villi because of severe atrophy and the loss of villi. The immunohistochemical labeling of neoplastic lymphocytes was considered to be positive when more than 30% of neoplastic cells were immunopositive.
For CE cases and normal controls, double-labeling immunohistochemistry for Ki67/CD3 was conducted and the Ki67 index was calculated as the mean ratio of Ki67+ nuclei in CD3+ cells. Detailed protocols are summarized in our previous study. 25 Canine lymph nodes or normal intestines were used as positive and negative tissue controls. Negative antibody controls were performed using buffer instead of the primary antibodies.
Statistical Analysis
The transcription level of each gene and the results of RNA ISH and immunohistochemistry were compared between groups using the Steel-Dwass test. The percentage of cases with detectable IL-21 mRNA expression levels was compared using Fisher’s exact test. Correlations were examined using Spearman’s rank correlation test. P values < .05 were considered to be significant. All statistical analyses were conducted using Statcel4 software (OMS publishing, Tokyo, Japan).
Results
Quantitative PCR
The mRNA level of IFN-γ was significantly higher in IEL+CE than in IEL–CE (P < .01) or normal controls (P < .01; Fig. 1a). The mRNA level of IL-21 was below detectable levels in all normal controls and 10/12 IEL–CE cases, whereas it was detectable in 16/17 IEL+CE cases (Fig. 1b). The percentage of cases with detectable IL-21 expression was significantly higher in IEL+CE than in IEL–CE (94% vs 17%; P < .01) or normal controls (94% vs 0%; P < .01). The mRNA levels of IL-12p35 (P < .01; Fig. 1c) and IL-12p40 (P < .05; Fig. 1d) were significantly higher in IEL+CE than in normal controls, but did not significantly differ between IEL+CE and IEL–CE. No significant differences were observed in the mRNA expression levels of IL-2 (Fig. 1e) and IL-15 (Fig. 1f) between IEL+CE, IEL–CE, and normal controls.
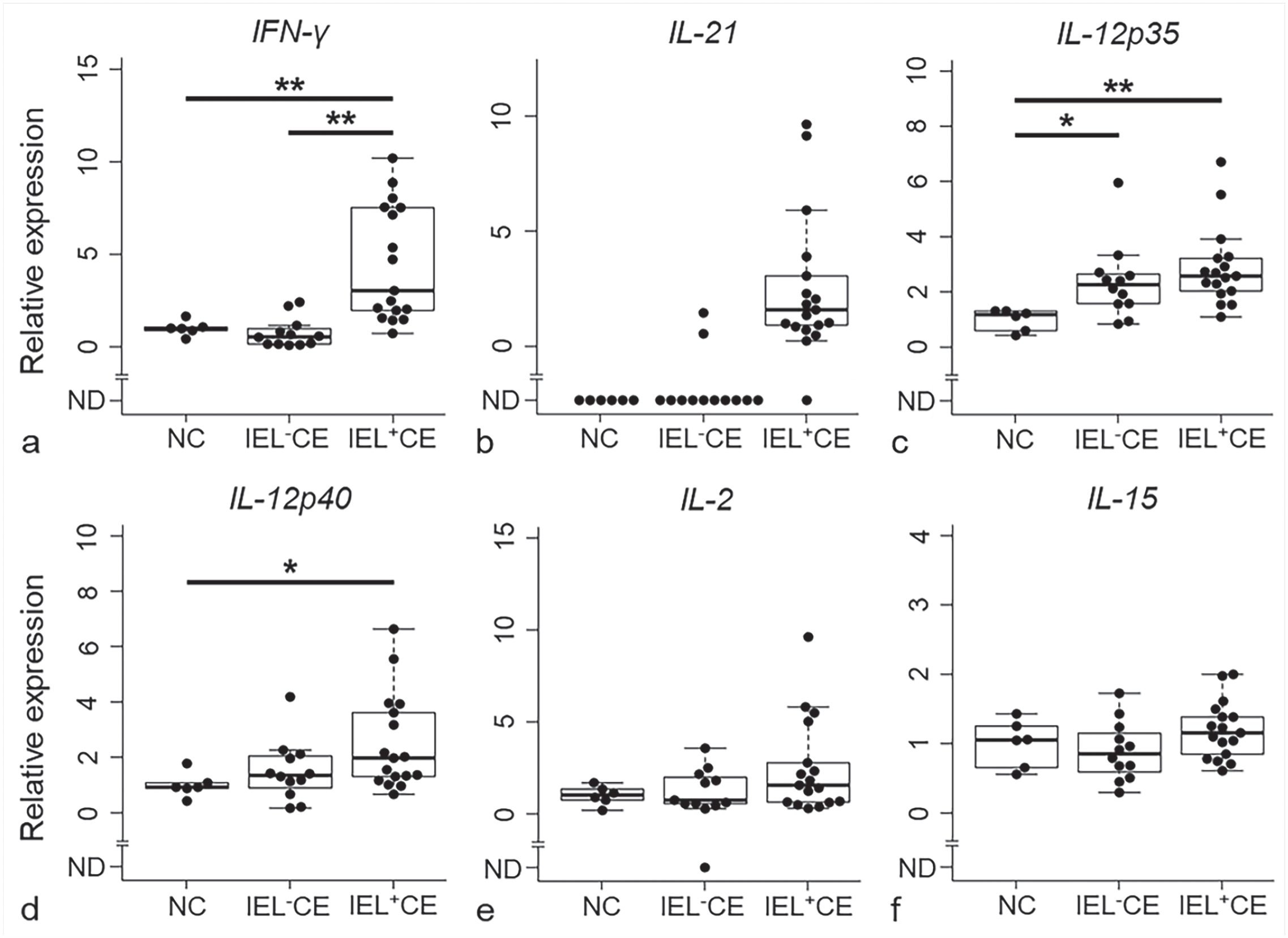
Relative mRNA transcription levels of (a) IFN-γ, (b) IL-21, (c) IL-12p35, (d) IL-12p40, (e) IL-2, and (f) IL-15 in the duodenal mucosa of dogs with chronic enteropathy with intraepithelial lymphocytosis (IEL+CE; n = 17) and without intraepithelial lymphocytosis (IEL-CE; n = 12) and normal controls (NC; n = 6). ND, not detected; *, P < .05; **, P < .01. Horizontal bars indicate maximum, upper quartile, median, lower quartile, and minimum. Boxes indicate interquartile ranges. The data exceeding 1.5 times the interquartile range are shown as outliers. IFN-γ, interferon-γ; IL, interleukin; IEL, intraepithelial lymphocytes; CE, chronic enteropathy; NC, normal controls.
RNA ISH
IFN-γ expression was detected in IELs and lamina propria cells in IEL+CE cases (Fig. 2a), and these cells were immunopositive for CD3 in double labeled sections (Fig. 2b). Although expression levels were low in IEL–CE and normal controls, a few IELs and lamina propria cells expressed IFN-γ (Fig. 2c). One IEL–CE case (43) had increased IFN-γ expression in CD3+ cells of the lamina propria (Supplemental Figure S3). IFN-γ expression was also detected in CD3+ neoplastic cells in SCL (Figs. 2d, e) and LCL (Figs. 2f, g) cases. The ratio of IFN-γ positive areas was significantly higher in IEL+CE than in IEL–CE (P < .05) and normal controls (P < .01), and even higher in SCL and LCL (Supplemental Figure S4).
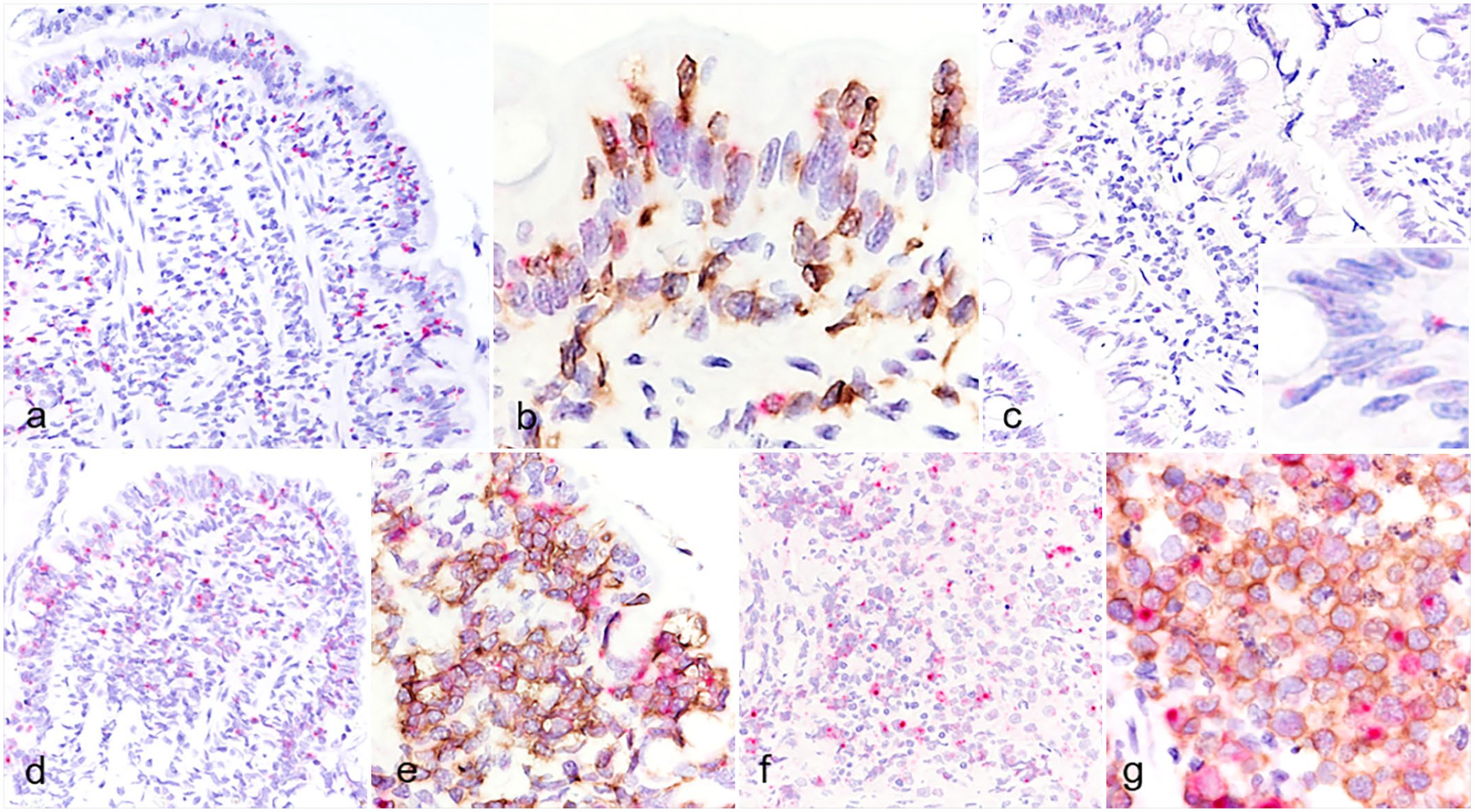
IFN-γ mRNA expression in the duodenal mucosa of dogs. (a) Chronic enteropathy with intraepithelial lymphocytosis (IEL+CE). Widely scattered positive signals (red) are in the epithelium and lamina propria. RNA in situ hybridization (ISH) for IFN-γ. Case 23. (b) IEL+CE. Positive hybridization signals for IFN-γ (red) are detected in CD3+ intraepithelial lymphocytes (IELs) and lamina propria lymphocytes (brown). Double labeling by RNA ISH for IFN-γ and immunohistochemistry (IHC) for CD3. Case 22. (c) Chronic enteropathy without intraepithelial lymphocytosis. Only a few positive signals (red) are present in the epithelium and lamina propria. Insets: higher magnification. RNA ISH for IFN-γ. Case 39. (d, e) Small cell lymphoma. Case 78. (d) Widely scattered positive signals (red) are in the epithelium and lamina propria. RNA ISH for IFN-γ. (e) Positive hybridization signals for IFN-γ (red) are present in CD3+ neoplastic lymphocytes (brown) clustered in the epithelium and lamina propria. Double labeling by RNA ISH for IFN-γ and IHC for CD3. (f, g) Large cell lymphoma. Case 111. (f) Widely scattered positive signals (red) are present in large neoplastic lymphocytes. RNA ISH for IFN-γ. (g) Positive hybridization signals for IFN-γ (red) are present in CD3+ large neoplastic lymphocytes (brown). Double labeling by RNA ISH for IFN-γ and IHC for CD3. IFN-γ, interferon-γ.
Western Blot Analysis
In the western blot analysis using the anti-dog IFN-γ antibody, a 20 to 25 kDa band was detected in protein extracts from the duodenal mucosa of dogs with CE and was considered to be glycosylated mature form of IFN-γ; however, the proprotein and nonglycosylated forms of IFN-γ were not detected (Supplemental Figure S5a). 34 No significant differences were observed in IFN-γ protein levels between IEL+CE, IEL–CE, and normal controls (Figs. 3a, d). Regarding the anti-dog IL-21 antibody, a band at 15 to 20 kDa was detected (Supplemental Figure S5b). IL-21 protein levels were lower in IEL+CE than in IEL–CE and normal controls (Figs. 3a, e).
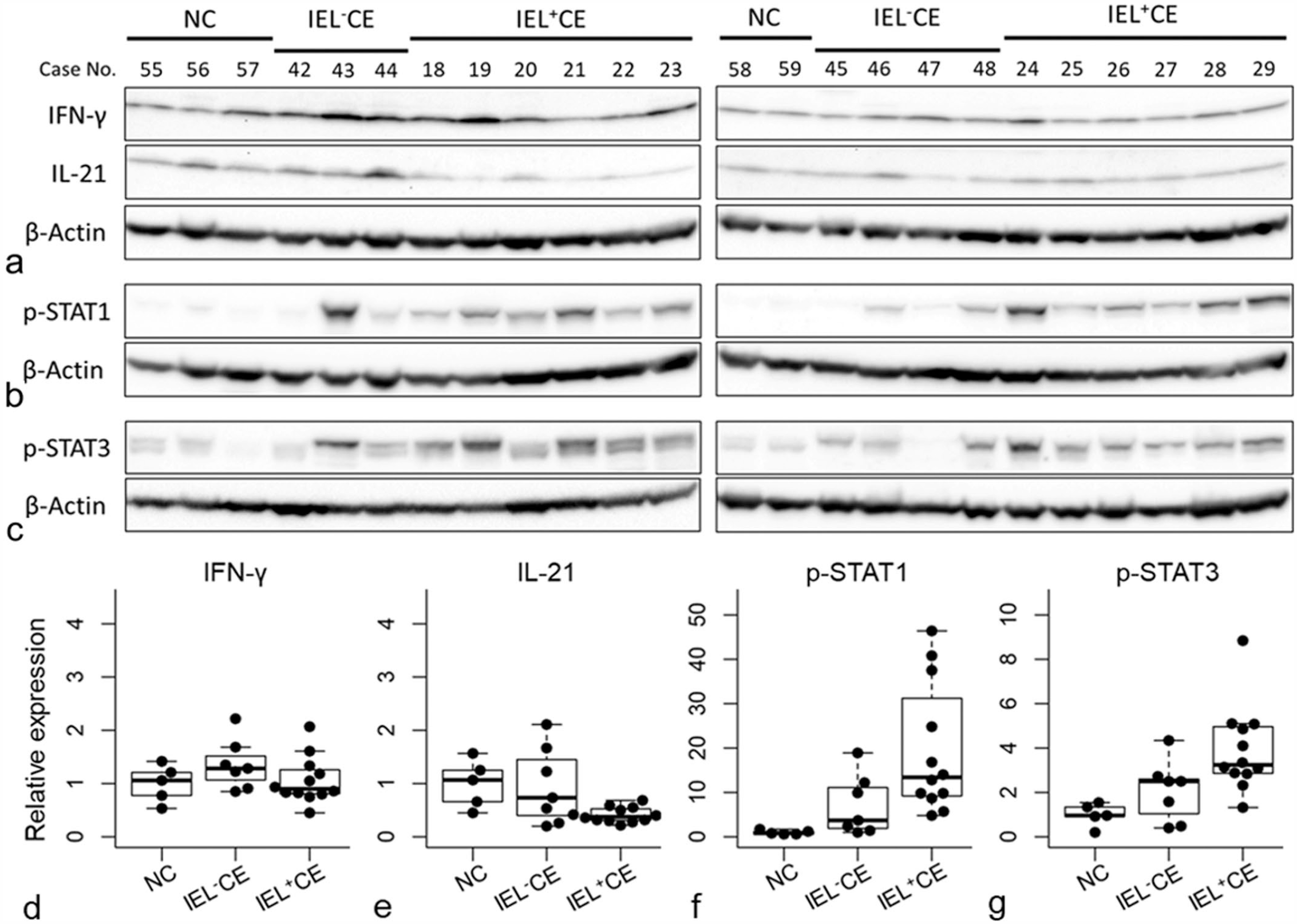
Western blot analysis of (a) IFN-γ, IL-21, (b) p-STAT1, and (c) p-STAT3 in the duodenal mucosa of dogs with chronic enteropathy with intraepithelial lymphocytosis (IEL+CE; n = 12) and without intraepithelial lymphocytosis (IEL–CE; n = 7) and normal controls (NC; n = 5). (d-g) Relative protein expression levels of (d) IFN-γ, (e) IL-21, (f) p-STAT1, and (g) p-STAT3. Horizontal bars indicate maximum, upper quartile, median, lower quartile, and minimum. Boxes indicate interquartile ranges. The data exceeding 1.5 times the interquartile range are shown as outliers. IFN-γ, interferon-γ; IL, interleukin; STAT, signal transducers and activators of transcription; IEL, intraepithelial lymphocytes; CE, chronic enteropathy; NC, normal controls.
To analyze the downstream signaling of IFN-γ and IL-21, western blotting for p-STAT1 and p-STAT3 was performed. Using an anti-p-STAT1 antibody, a 75 to 100 kDa band was detected in protein extracts from the duodenal mucosa of dogs with CE (Supplemental Figure S5c). p-STAT1 was higher in both IEL+CE and IEL–CE than in normal controls (Fig. 3b, f). Increases in p-STAT1 levels were greater in IEL+CE than in IEL–CE. In the western blot analysis using the anti-p-STAT3 antibody, 2 bands (isoform of STAT3) at approximately 75 kDa were detected in protein extracts from the canine duodenal mucosa (Supplemental Figure S5d). p-STAT3 was higher in IEL+CE and IEL–CE than in normal controls (Fig. 3c, g). IEL+CE showed an increase in p-STAT3 levels that was equal to or higher than that in IEL–CE.
Immunohistochemistry
The Ki67 index in CE cases and normal controls positively correlated with the mRNA expression level of IFN-γ assessed with quantitative PCR (n = 35, r = 0.38, P < .05; Fig. 4h) and RNA ISH (n = 30, r = 0.49, P < .01; Supplemental Figure S6).
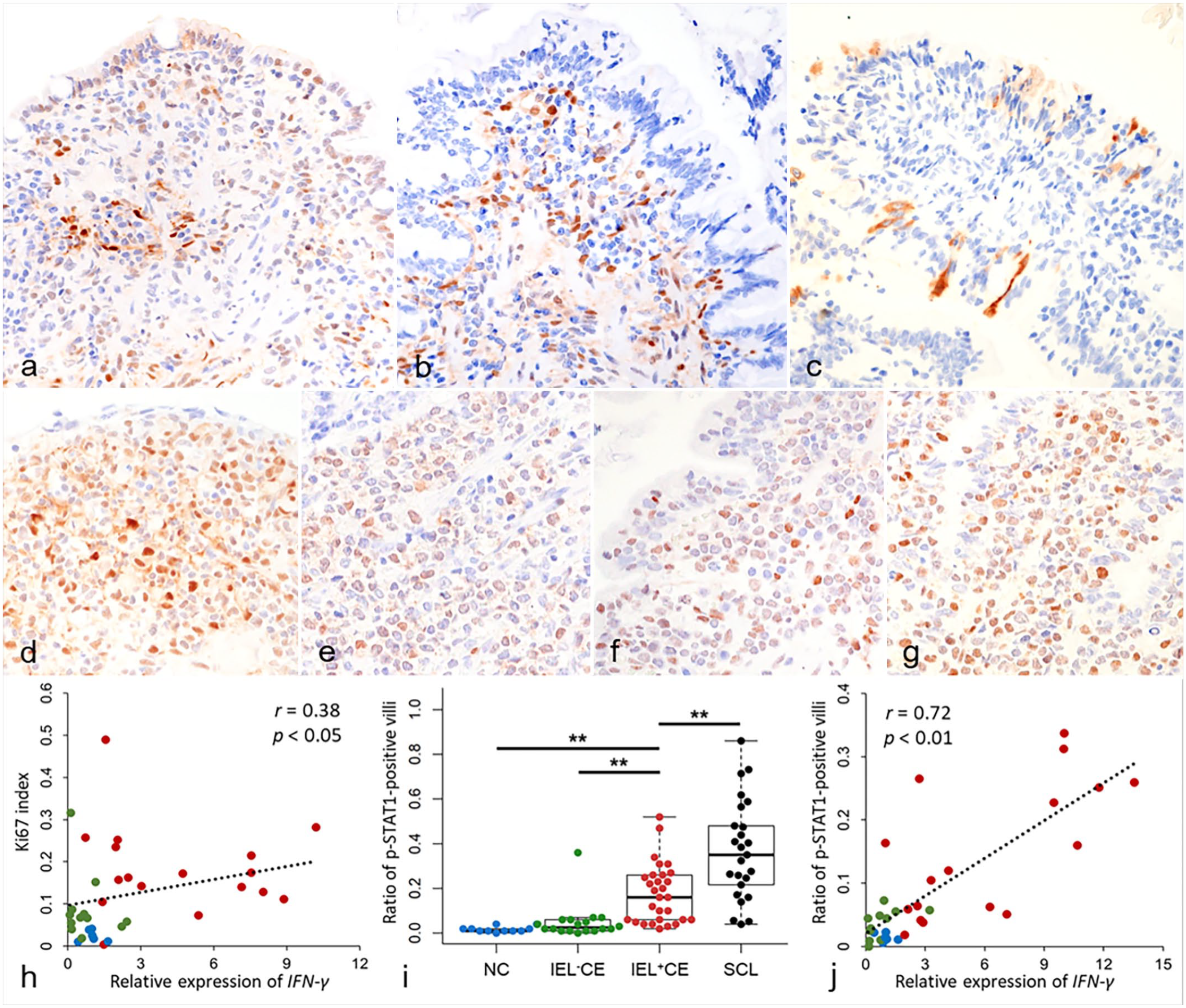
(a-g) Duodenum, dog. Immunohistochemistry. (a-c) Chronic enteropathy with intraepithelial lymphocytosis (IEL+CE). (a) Nuclear and cytoplasmic immunolabeling for p-STAT1 in enterocytes and lamina propria cells. Case 2. (b) Nuclear immunolabeling for p-STAT3 in lamina propria cells. Case 22. (c) Cytoplasmic immunolabeling for cleaved caspase-3 in enterocytes. Case 1. (d) Small cell lymphoma (SCL). Diffuse nuclear immunolabeling for p-STAT1 in neoplastic lymphocytes. Case 83. (e) Large cell lymphoma (LCL). Diffuse nuclear immunolabeling for p-STAT1 in neoplastic lymphocytes. Case 111. (f) SCL. Diffuse nuclear immunolabeling for p-STAT3 in neoplastic lymphocytes. Case 71. (g) LCL. Diffuse nuclear immunolabeling for p-STAT3 in neoplastic lymphocytes. Case 115. (h) A positive correlation was observed between the relative expression of IFN-γ by quantitative PCR and the Ki67 index, normal controls (NC) n = 6, blue; chronic enteropathy without intraepithelial lymphocytosis (IEL–CE): n = 12, green; IEL+CE: n = 17, red. (i) The ratio of p-STAT1-positive villi in NC (n = 11; blue), IEL–CE (n = 18; green), IEL+CE (n = 29; red), and SCL (n = 25; black). **, P < .01. Horizontal bars indicate maximum, upper quartile, median, lower quartile, and minimum. Boxes indicate interquartile ranges. The data exceeding 1.5 times the interquartile range are shown as outliers. (j) A positive correlation was observed between the relative expression of IFN-γ by quantitative PCR and the ratio of p-STAT1-positive villi (NC: n = 6, blue; IEL–CE: n = 11, green; IEL+CE: n = 17, red). IEL, intraepithelial lymphocytes; CE, chronic enteropathy; STAT, signal transducers and activators of transcription; SCL, Small cell lymphoma; NC, normal controls; IFN-γ, interferon-γ; IL, interleukin.
In the normal and CE duodenal mucosa, p-STAT1 was immunopositive in enterocytes and lamina propria cells at the tip of the villi, whereas IELs were negative (Fig. 4a). p-STAT3 was immunopositive in lamina propria cells and a small number of enterocytes and villi (Fig. 4b). Less than 5% of IELs were immunopositive for p-STAT3 in 7/29 (24%) IEL+CE, whereas those in the other CE and normal controls did not. Cleaved caspase-3 labeling was present in the cytoplasm of scattered enterocytes (Fig. 4c). In ITCL, neoplastic lymphocytes were immunopositive for p-STAT1 in 9/30 (30%) SCL (Fig. 4d) and 19/30 (63%) LCL cases (Fig. 4e) and immunopositive for p-STAT3 in 12/30 (40%) SCL (Fig. 4f) and 17/30 (57%) LCL cases (Fig. 4g). Cleaved caspase-3 was negative in neoplastic lymphocytes.
The ratio of p-STAT1-positive villi was significantly higher in IEL+CE than in IEL–CE (P < .01) and normal controls (P < .01), and even higher in SCL (P < .01; Fig. 4i). The ratio of p-STAT1-positive villi was positively correlated with the mRNA expression level of IFN-γ assessed with quantitative PCR (n = 34, r = 0.72, P < .01; Fig. 4j) and RNA ISH (n = 35, r = 0.73, P < .01; Supplemental Figure S7a). In addition, the ratio of p-STAT1-positive villi was significantly higher in cases with moderate (n = 39, P < .01) or severe (n = 26, P < .01) villous atrophy than in cases with intact or mildly atrophied villi (n = 18; Supplemental Figure S7b). No significant differences were observed in the ratio of cleaved caspase-3-positive villi between SCL, IEL+CE, IEL–CE, and normal controls (Supplemental Figure S8).
Discussion
In this study, IFN-γ and IL-21 mRNA expression levels were significantly higher and the downstream signals p-STAT1 and p-STAT3 were stronger in IEL+CE than IEL–CE and normal controls. These results suggest that these two cytokines play a role in the pathogenesis of IEL+CE in dogs. IFN-γ and IL-21 are both proinflammatory cytokines that stimulate, recruit, and/or induce the proliferation of immune cells,23,42 which may explain the more severe inflammation observed in IEL+CE. 25 Focusing on their actions of activating cytotoxic T/NK-cells and positive correlation with Ki67 index of T-cells,37,41,42,52 these cytokines are considered to induce activation of IELs. However, we were unable to validate these results by western blot analyses of IFN-γ and IL-21, which may be due to the low absolute expression levels of cytokines, extremely low ratio of cytokines to total protein, and/or inhibition of mRNA-to-protein translation in IEL+CE. Nevertheless, the western blot analysis results do not necessarily rule out the involvement of IFN-γ and IL-21. More detailed analysis, such as absolute quantification by enzyme-linked immunosorbent assay (ELISA) or analysis of mononuclear cells isolated from duodenal tissues, would be necessary to verify the protein expression.
While IFN-γ and IL-21 are known to promote the cytotoxicity of T/NK-cells, the involvement of granzyme B-mediated enterocyte damage by IELs in the pathogenesis of canine CE remains unclear because no significant difference was observed in the ratio of cleaved caspase-3-positive villi between IEL+CE, IEL–CE, and normal controls. In human inflammatory bowel disease, IFN-γ induces changes in epithelial paracellular permeability. 9 IFN-γ is believed to disrupt the barrier function of the epithelium through the internalization of tight junction transmembrane proteins, such as occludin and claudin-1. 7 In this study, IFN-γ was highly expressed in IELs and positively correlated with the ratio of p-STAT1-positive villi. Since STAT1 is considered to be a major downstream signaling molecule of IFN-γ, 23 we hypothesized that IFN-γ expressed by IELs acts on enterocytes by paracrine secretion and may disrupt epithelial barrier functions in the same manner as in human inflammatory bowel disease. STAT3 upregulation in villous epithelium has been reported in a previous study, 28 while only a small number of villi were immunopositive for p-STAT3 in this study. This discrepancy is possibly due to the difference in the clones of anti-p-STAT3 antibodies used for immunohistochemistry and/or the methods of immunohistochemistry evaluation. Previous study evaluated the number of p-STAT3-positive enterocytes, while the ratio of positive villi was evaluated in this study.
IL-12 is a heterodimeric proinflammatory cytokine composed of IL-12p35 and IL-12p40. It is mainly produced by macrophages and dendritic cells, and induces IFN-γ production in T/NK-cells.47,51 In this study, IL-12p35 and IL-12p40 expression levels were significantly higher in IEL+CE than in normal controls, suggesting that IL-12 is another factor inducing IFN-γ expression in T-cells in canine IEL+CE. However, no significant differences were observed in IL-12p35 or IL-12p40 expression levels between IEL+CE and IEL–CE, indicating that high IL-12 expression is not a specific cytokine expression pattern of IEL+CE. Furthermore, a recent study revealed that IL-12p35 and IL-12p40 were components of the other IL-12 family cytokines, IL-35 and IL-23, respectively. 51 While IL-23 exerts similar proinflammatory effects to those of IL-12, IL-35 exhibits anti-inflammatory activity. 10 Therefore, higher IL-12p35 expression in IEL+CE and IEL–CE may reflect an anti-inflammatory response via IL-35.
The present results indicate that cytokine expression patterns in canine CE differed slightly from those in celiac disease. In celiac disease, CD4+ T-cells secrete IFN-γ, IL-21, and IL-2 after antigen presentation by dendritic cells and induce the activation of cytotoxic T IELs.27,46 Increased IL-15 production in enterocytes and dendritic cells is also a characteristic of celiac disease. 31 In the present study, IFN-γ and IL-21 expression was also increased in canine IEL+CE. RNA ISH revealed IFN-γ expression in both IELs and lamina propria T-cells, which is consistent with previous findings in celiac disease.15,36 However, IL-2 and IL-15 expression remained unchanged in canine IEL+CE. Although IL-12 is considered to be less important in the pathogenesis of celiac disease,17,27 the expression of IL-12p35 and IL-12p40 increased in canine IEL+CE.
In humans, gluten is considered to induce an innate immune response via the production of IL-15 in the intestinal mucosa of all patients with and without celiac disease, and changes in adaptive mediators, including STAT1, STAT3, and IFN-γ, are specific for patients with celiac disease. 5 Histological and clinical features in dogs with food-responsive diarrhea are sometimes similar to those found in patients with celiac disease. 2 Gluten sensitivity was reported in Irish setter dogs with CE.4,22 Also, elevations in anti-gliadin and anti-tissue transglutaminase antibodies were reported in a subset of dogs with CE and ITCL. 30 Therefore, gluten intake is considered to be involved in the development of CE in certain breeds and a subset of dogs. However, the involvement of IL-15 in the pathogenesis of canine CE was not found in the present study or in a previous study. 16 Gliadin-independent mechanisms or physiological effects of gliadin that differ from those in humans may be involved in the pathogenesis of canine CE. However, it is important to note that cytokine expression patterns in celiac disease vary according to the clinical stage of the disease.6,26 A limitation of the present study is that we did not account for the stage or treatment status when selecting CE cases.
An IEL–CE case (43) showed markedly high IFN-γ expression in lamina propria T-cells, as well as elevated levels of p-STAT1 and p-STAT3. Since the overexpression of IFN-γ in lamina propria CD4+ T-cells in celiac disease is associated with the genetic background of HLA-DQ2 or HLA-DQ8,1,27 a specific genotype of dog leukocyte antigen (DLA) could be involved in the significant expression of IFN-γ observed in this case. In canine CE, specific DLA class II genotypes were reported in French bulldogs and German shepherd dogs with CE.33,38
In this study, neoplastic lymphocytes in ITCL expressed IFN-γ mRNA. In addition, the ratio of p-STAT1-positive villi was significantly higher in SCL. These results suggest that the mechanisms underlying severe mucosal injury in ITCL may be similar to those in IEL+CE, and this may be one of the reasons why the clinical course and histopathological features of canine CE and ITCL are similar. Furthermore, the results showing that neoplastic lymphocytes express the same cytokine as IELs in CE support our hypothesis that ITCL arises from activated IELs in IEL+CE. 25
The dysregulation of the JAK/STAT pathway has been implicated in various immunodeficiencies, autoimmune diseases, and tumorigenesis. In this study, neoplastic lymphocytes in 43/60 (72%) ITCL cases were immunopositive for p-STAT1 and/or p-STAT3, while the majority of IELs in CE were negative, suggesting the involvement of JAK/STAT pathway activation in the pathogenesis of canine ITCL. In humans, gain-of-function mutations in STAT3 have been reported in various T/NK-cell lymphomas11,12,32 and the upregulation of STAT1 may be associated with the survival of anaplastic LCL cells. 40 Highly recurrent gain-of-function mutations in JAK1 and STAT3 have also been reported in human EATL and increased IELs in type 2 refractory celiac disease. 11
In conclusion, the present results demonstrated an increase in IFN-γ and IL-21 mRNA expression in canine IEL+CE. An increase in IL-12p35 and IL-12p40 mRNA levels suggests the participation of IL-12 in the pathogenesis of IEL+CE, and these three cytokines may be involved in IEL activation. In canine IEL+CE and ITCL cases, IFN-γ was expressed in the IELs, lamina propria T-cells, and ITCL cells and is hypothesized to be associated with mucosal injury. STAT1 and STAT3 activation in ITCL cells suggests a role for the upregulation of the JAK/STAT pathway in the pathogenesis of canine ITCL.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231207017 – Supplemental material for Pro-inflammatory cytokine expression and the STAT1/3 pathway in canine chronic enteropathy and intestinal T-cell lymphoma
Supplemental material, sj-pdf-1-vet-10.1177_03009858231207017 for Pro-inflammatory cytokine expression and the STAT1/3 pathway in canine chronic enteropathy and intestinal T-cell lymphoma by Kazuhiro Kojima, James K. Chambers, Ko Nakashima and Kazuyuki Uchida in Veterinary Pathology
Footnotes
Author Contributions
KK designed and performed the experiments; JKC, KN, and KU contributed to the experimental design; KN performed endoscopic sample collection; KK, JKC, and KU performed histological evaluations; the manuscript was written by KK, JKC, and KU with a contribution from KN.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Grant-in-Aid from the Japan Society for the Promotion of Science (19H03122, 23KJ0671).
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
