Abstract
Human enteropathy-associated T-cell lymphoma (EATL) is considered to be derived from intraepithelial lymphocytes (IELs); however, the origin of canine intestinal T-cell lymphoma (ITCL) remains unclear. Histological, immunohistochemical, and clonality examinations were performed using endoscopically collected canine duodenum samples of mucosal lesions of chronic enteropathy (CE; 73 cases) and ITCL without transmural neoplastic mass lesions (64 cases). Histopathological examinations revealed the intraepithelial accumulation of lymphocytes (called “intraepithelial lymphocytosis”) in 54/73 CE cases (74%) and the epitheliotropism of neoplastic lymphocytes in 63/64 ITCL cases (98%). Immunohistochemically, IELs in CE with intraepithelial lymphocytosis (IEL+CE) were diffusely immunopositive for CD3, with scattered immunopositivity for CD5, CD8, CD20, and granzyme B (GRB). The percentage of CD8+ in CD3+ IELs was significantly lower in IEL+CE than in CE without intraepithelial lymphocytosis (IEL−CE). Double-labeling immunohistochemistry revealed a high percentage of GRB expression in CD8− IEL among IEL+CE. Among 64 ITCL cases, CD3 was immunopositive in 64 (100%), CD5 in 22 (34%), CD8 in 8 (13%), CD20 in 12 (19%), CD30 in 13 (20%), and GRB in 49 (77%). In CD3+ cells, Ki67 immunopositivity was highest in ITCL, intermediate in IEL+CE, and lower in IEL−CE. A clonal TCR gene rearrangement was detected in 1/19 IEL−CE cases (5%), 15/54 IEL+CE (28%), and 38/58 ITCL (66%). These results indicate that the immunophenotype of canine ITCL (CD8−GRB+) is similar to that of the increased IELs in CE. The high proliferative activity and clonality of T cells in IEL+CE suggest that canine ITCL originates from these IELs, similar to human EATL.
Keywords
In the World Health Organization (WHO) classification system of human hematopoietic tumors, primary intestinal T-cell lymphoma is classified as enteropathy-associated T-cell lymphoma (EATL; previously known as EATL type 1), monomorphic epitheliotropic intestinal T-cell lymphoma (MEITL; previously known as EATL type 2), indolent T-cell lymphoproliferative disorder of the gastrointestinal tract, and intestinal T-cell lymphoma–not otherwise specified. 33 EATL is considered to be derived from intraepithelial lymphocytes (IELs), the number of which increases in celiac disease. 6,33
In dogs, intestinal T-cell lymphoma (ITCL) may be broadly divided into large cell lymphoma (LCL) and small cell lymphoma (SCL) based on nuclear sizes. 3,15,16,38 Canine ITCL is also considered to be derived from IELs and arises in a background of chronic enteropathy (CE). 36,38 The neoplastic lymphocytes of canine ITCL often show epitheliotropism, and an increase in IELs, which is a characteristic histological feature of celiac disease, has been reported in a subset of canine CE. 3,17 However, the relationship between ITCL and CE in dogs remains unclear, particularly the pathogenesis of ITCL.
Human celiac disease is an autoimmune inflammatory enteropathy against gliadin and other related prolamins, and celiac disease with a poor therapeutic response to a gluten-free diet is called refractory celiac disease (RCD). 14 RCD is classified into types 1 and 2 based on the immunophenotype of IELs and results of a clonality analysis. 28 Type 1 RCD is characterized by an increase in CD8+ IELs and a polyclonal TCR gene rearrangement, while type 2 RCD is characterized by an increase in CD8− IELs and a monoclonal TCR gene rearrangement. Type 2 RCD is more likely to progress to EATL and have a poor prognosis. 7,27,33 In dogs, a monoclonal T-cell population has been reported in a subset of CE; 17,21 however, limited information is currently available on the immunophenotypes of IELs.
In our previous study on canine transmural, mass-forming gastrointestinal lymphoma using full-thickness biopsy samples, we revealed that most cases of canine LCL expressed granzyme B (GRB) without surface CD8 expression, 15 which is similar to the characteristic immunophenotype of human EATL. 33 We also revealed that canine ITCL includes various histological subtypes, with each one potentially having a different cell nature; therefore, mass-forming ITCL appears to be too complex to investigate the involvement of IELs in the pathogenesis of ITCL. On the other hand, non-mass-forming ITCL is generally diagnosed by endoscopic biopsy. The histological lesions of mucosal ITCL resemble those of CE, and thus, a clear distinction is sometimes difficult. 3,20,35 Since mucosal ITCL may occur continuously from CE, the use of endoscopic samples may be suitable for examining the relationship between ITCL and CE. The aim of the present study was to characterize the immunophenotype of IELs, particularly CD8 expression, by immunohistochemistry using endoscopic biopsy samples, and examine clonal TCR gene rearrangements in canine CE, as a way to infer the cell of origin in canine ITCL.
Materials and Methods
Case Selection
Intestinal tissue samples were endoscopically collected from dogs with a history of chronic gastrointestinal disease presented at the Japan Small Animal Medical Center (JSAMC) and Veterinary Medical Center of the University of Tokyo (VMC) between 2013 and 2021. Endoscopic samples from the duodenum of 137 dogs with a histological diagnosis of CE (lymphoplasmacytic enteritis; n = 73) or ITCL (n = 64) were included. In all cases, an intestinal mass was excluded by ultrasonographic or radiographic examination. Histopathological criteria are described in the histopathology section below. Regarding CE and ITCL, cases were selected when the main lesion was in the duodenum. Immunohistochemical inclusion criteria for ITCL included the clear membranous or cytoplasmic expression of CD3. Histopathological diagnoses and clonality analyses were performed at the Laboratories of Veterinary Pathology and Veterinary Internal Medicine, respectively, at the Graduate School of Agricultural and Life Sciences, The University of Tokyo. The age, sex, and breed of CE and ITCL cases are summarized in Supplemental Table S1.
Histopathology
Endoscopic tissue samples submitted from JSAMC were collected from the stomach, proximal duodenum, distal duodenum, ileum, and colon. Endoscopic tissue samples submitted from VMC were collected from the duodenum and the stomach, jejunum, ileum, and/or colon. Mucosal tissue samples were placed with the villous side facing upwards on filter paper to make a vertical section. A minimum of 6 tissue specimens were collected per region. All samples were fixed in 10% neutral buffered formalin solution, processed routinely, and embedded in paraffin wax. Four-micrometer-thick paraffin sections were deparaffinized, rehydrated through a graded series of alcohol, and stained with hematoxylin and eosin. A histopathological diagnosis was performed by 2 veterinary pathologists accredited by the Japanese College of Veterinary Pathologists (KU and JKC).
A histological diagnosis of CE was based on the histopathological standards for the intestines established by the World Small Animal Veterinary Association Gastrointestinal Standardization Group. 8 The diagnosis of ITCL was based on a dense monomorphic population of neoplastic lymphocytes with an abnormal cell size or distorted architecture. 3,37 To evaluate histological features, the following criteria were assessed 8 : intraepithelial lymphocytosis (ie, more than 30 IELs per 100 enterocytes in the villous epithelium), epitheliotropism of lymphomas (ie, formation of intraepithelial nests [≥5 clustered lymphocytes] within lymphoma lesions), villous atrophy, increase in number of lamina propria lymphocytes and plasma cells, diffuse infiltration of neutrophils (ie, more than 5 neutrophils per high power field [HPF] = 0.196 mm2]) and eosinophils (ie, more than 5 eosinophils per HPF) in the lamina propria, crypt dilation, and lacteal dilation. In each criterion, the most severe lesion was selected for evaluation. CE cases were further classified into CE with intraepithelial lymphocytosis (IEL+CE) and CE without intraepithelial lymphocytosis (IEL−CE) based on the histological evaluation. ITCL cases were also further classified into LCL and SCL according to their nuclear size relative to red blood cells (RBCs), as previously described: LCL, >2 RBC; SCL, <2 RBC. 15,16
Immunohistochemistry
Details on the primary antibodies and antigen retrieval procedures used are summarized in Table 1. Following antigen retrieval, endogenous peroxidase was inactivated with 3% hydrogen peroxide in methanol at room temperature for 5 minutes. Sections were blocked with 8% skim milk in Tris-buffered saline (TBS) at 37 °C for 40 minutes, and then incubated with primary antibodies at 4 °C overnight. After washing with TBS, each secondary antibody was applied. In the Dako Envision+ System, sections were incubated with an anti-rabbit or anti-mouse hydrogen peroxidase-labeled secondary antibody polymer at 37 °C for 40 minutes. In the Ventana OptiView DAB Universal Kit (Ventana Medical Systems), sections were incubated with Linker-HQ at 37 °C for 8 minutes, and then with Multimer-HRP at 37 °C for 8 minutes. To detect CD8, 29 sections were incubated with biotin-conjugated anti-rat immunoglobulin G (1:500; Dako) at 37 °C for 40 minutes, and then with horseradish peroxidase-conjugated streptavidin (1:400; Dako) at 37 °C for 40 minutes. Labeled complexes were visualized with the 3,3′-diaminobenzidine (DAB) chromogen and 0.03% hydrogen peroxidase in Tris-HCl buffer and then counterstained with hematoxylin. Canine activated lymph nodes or normal intestines were used as positive and negative tissue controls, respectively, and negative antibody controls were performed by applying an irrelevant antibody and buffer instead of the primary antibodies. 26
Methods used for immunohistochemistry.
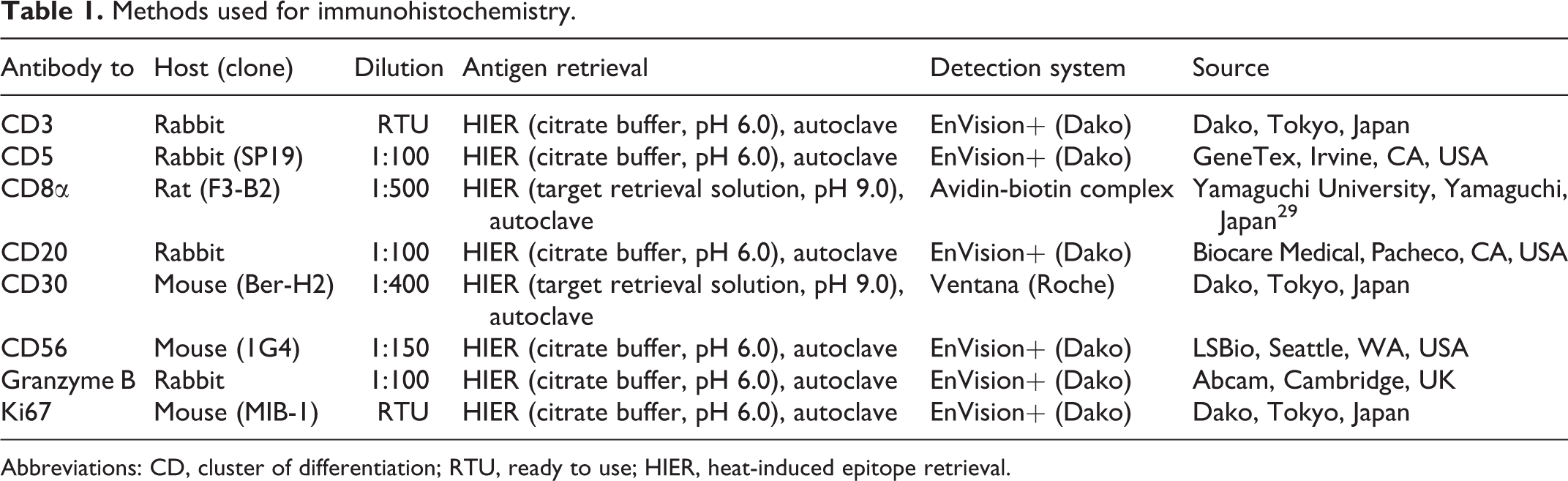
Abbreviations: CD, cluster of differentiation; RTU, ready to use; HIER, heat-induced epitope retrieval.
Double-labeling immunohistochemistry for Ki67/CD3 was conducted to detect Ki67+ T cells. In CE cases, double-labeling immunohistochemistry for CD8/CD3 and CD8/GRB was also performed to clarify the immunophenotype of IELs. After labeled complexes of Ki67 or CD8 were visualized with the DAB chromogen and 0.03% hydrogen peroxidase in Tris-HCl buffer, sections were blocked with 8% skim milk in TBS at 37 °C for 40 minutes and incubated with the anti-CD3 or anti-GRB antibody at 4 °C overnight. After washing with TBS, sections were incubated with an alkaline phosphatase-conjugated anti-rabbit secondary antibody polymer (N-Histofine Simple Stain AP (R); Nichirei Biosciences), the immunoreaction was visualized using the N-Histofine New Fuchsin substrate (Nichirei Biosciences), and sections were counterstained with hematoxylin. Canine-activated lymph nodes or normal intestines were used as positive and negative tissue controls, respectively, and negative antibody controls were performed using buffer instead of the primary antibodies. 26
The immunohistochemical staining of IELs for CD3, CD8, CD20, CD30, CD56, and GRB was evaluated in IEL+CE cases using a semiquantitative assessment (ie, 0%, 5%, 25%, 50%, 75%, 95%, and 100%). The immunohistochemical staining of ITCL was considered to be positive when more than 30% of neoplastic cells were positive. CD3/Ki67 double labeling was examined at a medium magnification to select areas with the strongest infiltration of CD3+ cells in CE cases and those with representative lymphoma lesions in ITCL cases. Within these areas, 5 fields at a high magnification (40× objective, 0.0625 mm2) were photographed. The number of Ki67+ nuclei in each image was manually assessed in CD3+ cells using ImageJ software (National Institutes of Health, Bethesda, MD), and the Ki67 index was calculated as the mean percentage of Ki67+ nuclei in CD3+ cells. CD3/CD8 double labeling was also examined in the same manner, and 10 fields at high magnification were photographed. In each image, the number of CD8+ IELs was manually assessed among CD3+ IELs and the mean percentage of CD8+ IELs was calculated. In 12 cases with a limited number of IELs (ie, less than 50 IELs per field at a high magnification), all CD8+ and CD3+ IELs on the slide were counted. IEL+CE cases were further classified into CD8+IEL+CE and CD8−IEL+CE based on the percentage of CD8+ IELs: CD8+IEL+CE, >50%; CD8−IEL+CE, <50%. 28
Clonality Analysis
Tissue collection and a clonality analysis were performed as previously described. 13 Briefly, tissue samples were endoscopically collected from the duodenum and stored at −20 °C until used. Genomic DNA was extracted from frozen samples using the DNeasy Blood and Tissue Kit (Qiagen). Forward primers were labeled with fluorescent dyes at their 5′ ends. All reactions were conducted in duplicate. A GeneScan analysis was performed on the ABI 3130xl Genetic Analyzer (Applied Biosystems) as a multiplexed run. A clonal T-cell population was defined by the presence of distinct peaks of fluorescence intensity that were at least 2-fold higher than other background peaks. Monoclonal or biclonal peaks indicated a positive result.
Statistical Analysis
The mean percentage of CD8+ IELs between IEL−CE and IEL+CE cases were compared using the Student’s t test. Variances were compared with the F test. Histological features between IEL−CE and IEL+CE cases and the mean Ki67 index between CD8+IEL+CE and CD8−IEL+CE cases were compared using the Mann-Whitney U test. The Steel-Dwass test was employed to compare the mean Ki67 index between IEL−CE, IEL+CE, SCL, and LCL cases. The frequencies of a clonal TCR gene rearrangement were compared with Fisher’s exact test. P values <.05 were considered to be significant. All statistical analyses were conducted with Statcel4 software (OMS publishing Inc).
Results
Histology
Chronic enteropathy
CE was histologically characterized by mild to severe increase in number of lamina propria lymphocytes and plasma cells (mild: 23/73 cases, 32%; moderate: 39/73 cases, 53%; marked: 11/73 cases, 15%) with villous atrophy (intact to mild: 10/73 cases, 14%; moderate: 38/73 cases, 52%; marked: 25/73 cases, 34%; Fig. 1a). Intraepithelial lymphocytosis was frequently observed at the tips of villi (54/73 cases, 74%; Fig. 1b). The infiltration of neutrophils and eosinophils was observed in 33/73 cases (45%) and 22/73 cases (30%), respectively. Crypt dilation was detected in 39/73 cases (53%) and lacteal dilation in 25/73 cases (34%). The severities of villous stunting (P < .05) and lymphoplasmacytic infiltration (P < .005) were significantly greater in IEL+CE cases than in IEL−CE cases.
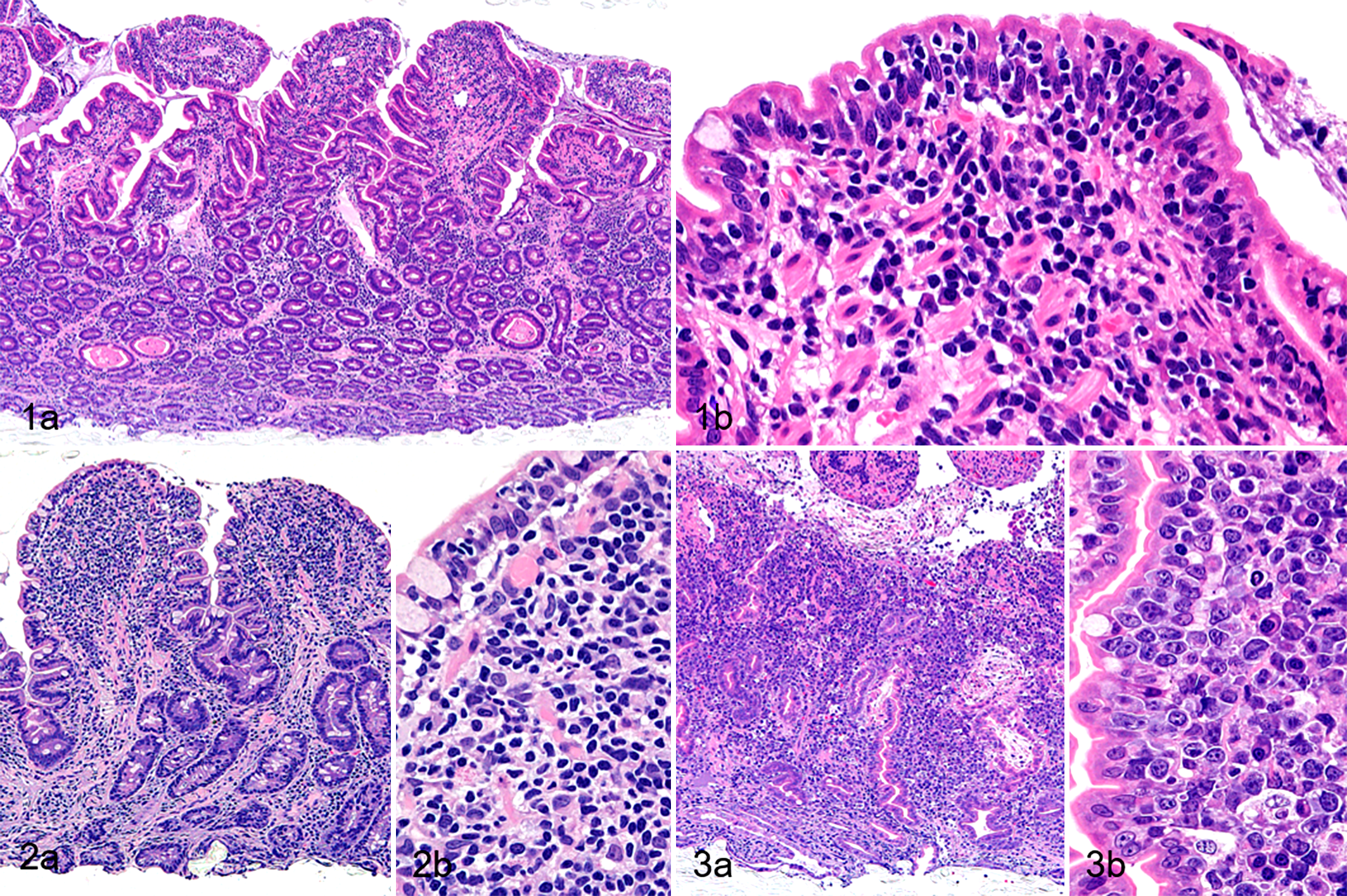
Duodenum, dog. Hematoxylin and eosin.
Intestinal T-cell lymphoma
In 56/64 ITCL cases, not only the duodenum but also other regions of the gastrointestinal tract were affected by lymphoma, including the stomach (6/57 cases, 11%), jejunum (14/14 cases, 100%), ileum (53/57 cases, 93%), and colon (12/53 cases, 23%). The neoplastic cells of SCL (n = 33) were characterized by round and monomorphic nuclei with dense chromatin (Fig. 2a and b), and those of LCL (n = 31) by large cells with pleomorphic, round to ovoid nuclei containing scattered chromatin and distinct nucleoli (Fig. 3a and b). In 10/31 cases (32%) of LCL, a moderate number of eosinophilic granules (which expressed cytotoxic granule proteins) were observed in the cytoplasm of neoplastic cells. ITCL was characterized by moderate to marked villous atrophy (intact to mild: 1/64 cases, 2%; moderate: 16/64 cases, 25%; severe: 47/64 cases, 73%) and epitheliotropism of neoplastic lymphocytes was detected in all cases, except for 1 SCL case. All cases showed mild to moderate increase in number of plasma cells. The infiltration of neutrophils and eosinophils was noted in 44/64 cases (69%) and 29/64 cases (45%), respectively. Crypt dilation was observed in 40/64 cases (63%) and lacteal dilation in 20/64 cases (31%).
Immunohistochemistry
Chronic enteropathy
IELs in IEL+CE were diffusely positive for CD3 (median: 100%; range: 95% to 100%; Fig. 4) and scattered cells were positive for CD5 (median: 25%; range: 0% to 95%; Fig. 5), CD8 (median: 50%; range: 5% to 95%; Fig. 6), CD20 (median: 5%; range: 0% to 95%; Fig. 7), and GRB (median: 25%; range: 1% to 95%; Fig. 8). IELs were negative for CD30 and CD56; however, some large lymphocytes in the lamina propria were positive for CD30 in 2 IEL+CE cases (Fig. 9). In double-labeling immunohistochemistry for CD8/CD3, the mean percentage of CD8+ IELs among CD3+ IELs was significantly lower in IEL+CE cases than in IEL−CE cases (45.3% vs 60.3%; P < .02; Figs. 10, 11). GRB was positive not only in CD8+ IELs but also in CD8− IELs by double-labeling immunohistochemistry for CD8/GRB (Fig. 12).
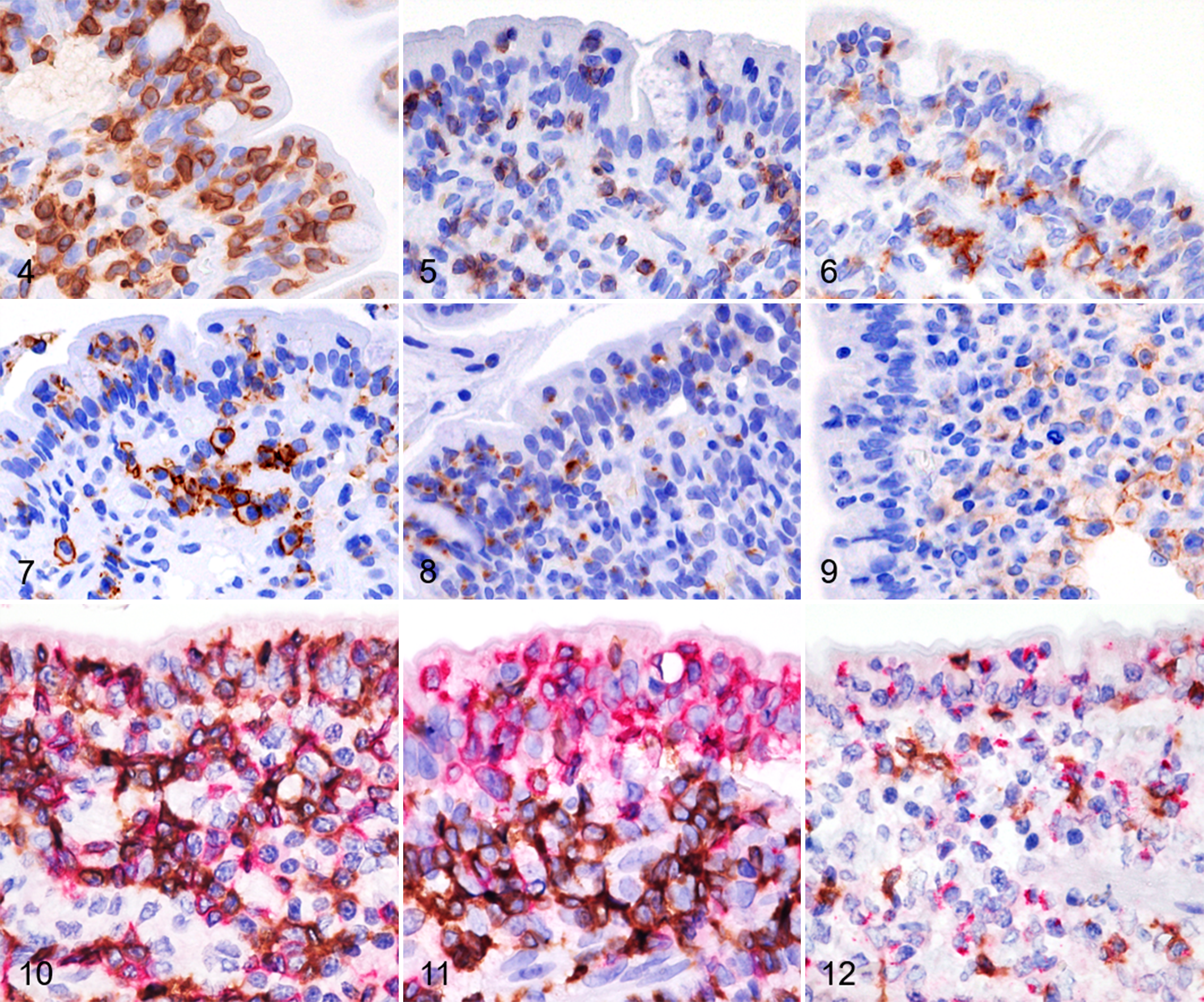
Chronic enteropathy with intraepithelial lymphocytosis (IEL+CE), duodenum, dog. Immunohistochemistry.
Intestinal T-cell lymphoma
Neoplastic lymphocytes were diffusely positive for CD3 in all cases (Fig. 13). CD5 was positive in 11/33 SCL (33%) and 11/31 LCL (35%) cases (Fig. 14), CD8 in 7/33 SCL (21%) and 1/31 LCL (3%) cases (Fig. 15), CD20 in 8/33 SCL (24%) and 4/31 LCL (13%) cases (Fig. 16), CD30 in 2/33 SCL (6%) and 11/31 LCL (35%) cases (Fig. 17), and GRB in 24/33 SCL (73%) and 25/31 LCL (81%) cases (Fig. 18).
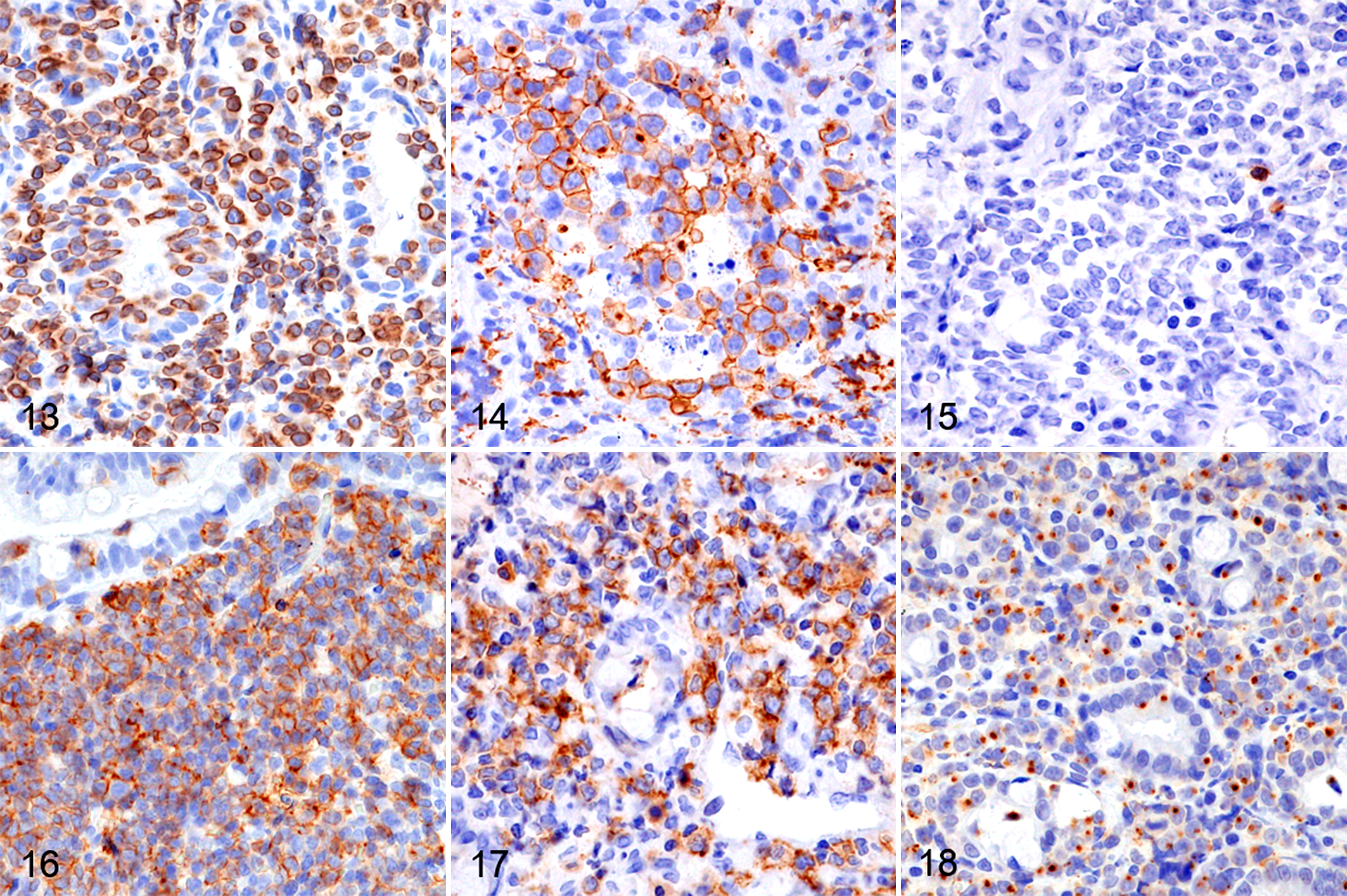
Large cell lymphoma, duodenum, dog. Immunohistochemistry.
The mean Ki67 index was 10.2% (range: 2.4% to 31.6%) in IEL−CE cases, 19.5% (range: 4.2% to 55.6%) in IEL+CE cases, 26.3% (range: 6.0% to 65.6%) in SCL cases, and 48.8% (range: 17.6% to 81.9%) in LCL cases. A significant difference in the Ki67 index was observed between the combination of IEL−CE versus IEL+CE (10.2% vs 19.5%; P < .01) and SCL versus LCL (26.3% vs 48.8%; P < .01), but not between IEL+CE and SCL. Among IEL+CE cases, no significant difference was noted between CD8+IEL+CE (n = 21) and CD8−IEL+CE (n = 33; 17.6% vs 20.7%; P = .23).
TCR and IgH Gene Rearrangement Clonality
A clonality analysis of 73 CE, 28 SCL, and 30 LCL cases was conducted. A clonal TCR gene rearrangement was detected in 14/73 CE cases (19%), 18/28 SCL cases (64%), and 17/30 LCL cases (57%). The clonal rearrangement of both the TCR gene and IgH gene was detected in 2/73 CE cases (3%), 2/28 SCL cases (7%), and 1/30 LCL cases (3%). A clonal IgH gene rearrangement was detected in 5/73 CE cases (7%) and 1/30 LCL cases (3%). Among CE cases, a clonal TCR gene rearrangement with or without a clonal IgH gene rearrangement was detected more frequently in IEL+CE (15/54 cases, 28%) than in IEL−CE (1/19 cases, 5%; P < .04), whereas no significant difference was observed between CD8+IEL+CE (5/21 cases, 24%) and CD8−IEL+CE (10/33 cases, 30%; P = .42).
Discussion
Canine ITCL is classified into LCL and SCL and has been compared to human EATL and MEITL, respectively. 3,15,16,38 We previously revealed that canine ITCL was negative for CD56, which differs from human MEITL. 15,16 On the other hand, canine ITCL, particularly transmural LCL, is similar to human EATL characterized by the CD3+CD8−CD56−GRB+ immunophenotype. 15 In the present study on the mucosal lesions of canine ITCL, the majority of SCL and LCL showed the CD3+CD8−CD56−GRB+ immunophenotype. Histological features, such as epitheliotropism and the infiltration of various inflammatory cells, were also consistent with human EATL. 33 These results indicate that the neoplastic lymphocytes of canine ITCL and human EATL share similar properties.
Human EATL is derived from aberrant CD8− IELs in type 2 RCD. 6,33 Canine ITCL is also regarded as a neoplasm of IEL origin; 36,38 however, the immunophenotypes of IEL and ITCL have not been compared in detail in dogs. In the present study, canine IEL+CE showed a significant increase in the proportion of CD8− IELs, and these CD8− IELs expressed GRB, which was the same immunophenotype as most ITCL cases. Taken together with the epitheliotropism of canine ITCL, these results suggest that CD8− IELs in IEL+CE are the origin of canine ITCL (Fig. 19). The findings of phenotypic, transcriptional, and functional studies on IELs in human type 2 RCD and in vivo murine studies suggested that a subtype of innate lymphoid cells, which do not express T- or B-cell receptors, but produce effector cytokines that are also produced by cytotoxic T cells or T-helper cells, is the origin of type 2 RCD and human EATL. 6,9,30 However, innate lymphoid cells have not been identified in dogs and the detailed lineage of neoplastic lymphocytes remains unclear.
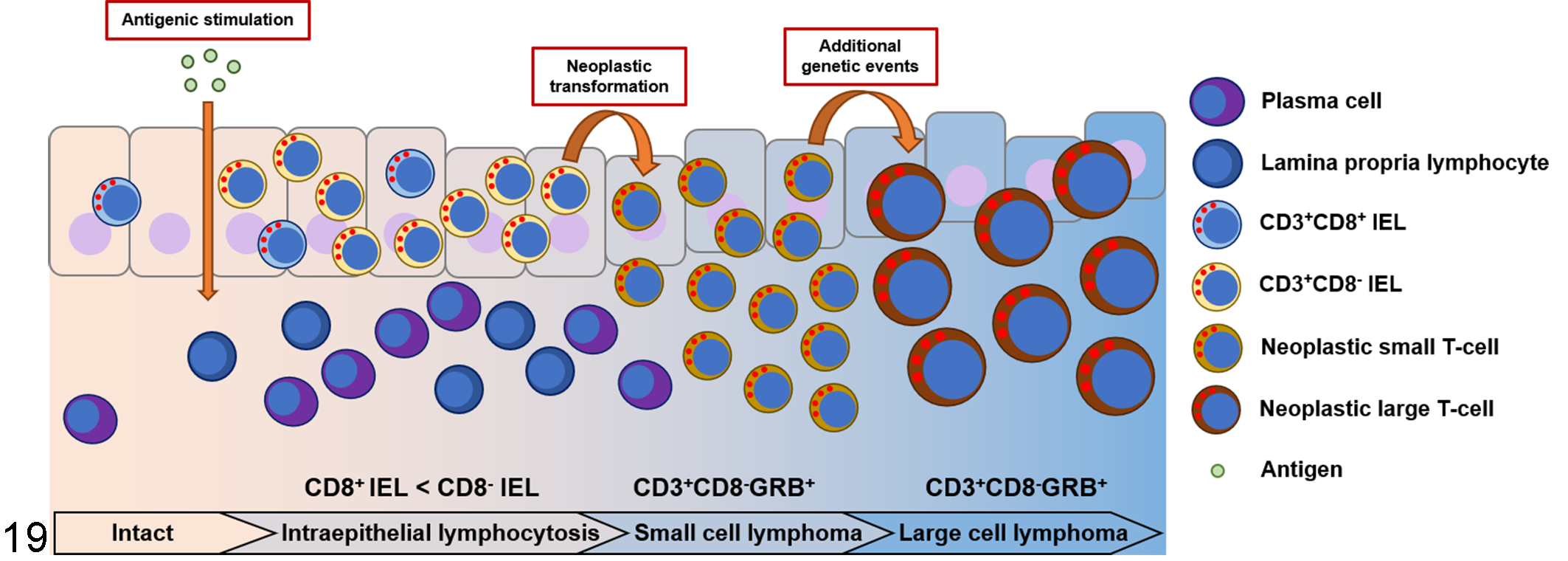
Schematic diagram of the proposed pathogenesis of canine intestinal T-cell lymphoma (ITCL). As shown by histological examination, a subset of chronic enteropathy cases shows an increase in intraepithelial lymphocytes (IELs; intraepithelial lymphocytosis). Increased number of IELs among CD8− T cells. A monoclonal TCR gene rearrangement is occasionally detected. Continuous inflammation may result in the genetic alteration and neoplastic proliferation of IELs. Neoplastic cells predominantly have the CD3+CD8−GRB+ immunophenotype, which is consistent with the immunophenotype of the increased IELs, regardless of the cell morphology. With the progression of small cell lymphoma, a subset of neoplastic cells may undergo additional genetic events and transform into large cell lymphoma.
In humans, type 2 RCD is regarded as a neoplastic T-cell disorder or a cryptic low-grade T-cell lymphoma because monoclonal populations of T cells are frequently detected in duodenal or jejunal biopsies. 1,2,4 Furthermore, the risk of developing EATL is higher. 7,27 Previous studies successfully detected clonal T-cell populations in canine CE cases. 17,21 In the present study, we detected clonal T-cell populations in canine CE and ITCL cases. A clonal TCR gene rearrangement was more frequently observed in IEL+CE cases than in IEL-CE cases, suggesting the monoclonal proliferation of IELs. Moreover, the increased proliferative activity of T cells in IEL+CE suggested neoplastic cell proliferation. Negative results for a clonal TCR gene rearrangement in ITCL may be explained by the absence of neoplastic lymphocytes in biopsy samples due to the patchy distribution of ITCL, a polyclonal background, the insufficient coverage of primers, primer site mutations, or chromosomal aberrations. 16
RCD with an increased number of CD8+ IELs, a finding also observed in uncomplicated celiac disease, is classified as type 1 RCD. 28 In contrast to type 2 RCD, the majority of type 1 RCD cases show a polyclonal TCR gene rearrangement and have a favorable prognosis with a low risk of developing EATL. 7,27,33 Progression from type 1 to type 2 RCD is uncommon. 6,7 In the present study, IEL+CE was divided into CD8+IEL+CE and CD8−IEL+CE based on the percentage of CD8+ IELs, as reported for types 1 and 2 RCD. 28 However, there were no significant differences in the Ki67 index or clonality results between CD8+IEL+CE and CD8−IEL+CE.
The lack of surface expression of CD5 and CD20 expression are uncommon phenotypes of T cells but are observed in a subset of T-cell lymphoma, including canine ITCL. 15,16,23 In the present study, CD5− or CD20+ IELs were also detected in a subset of canine IEL+CE; however, it remains unclear whether this is the normal immunophenotype of IELs or an immunophenotype suggestive of neoplastic proliferation. The lack of CD5 expression is also a phenotypic feature of IELs in type 2 RCD, while CD20 expression is uncommon. 33 In humans, CD20 expression has been detected in MEITL cases. 5,34
The expression of CD30 is typically observed in anaplastic large cell lymphoma (ALCL) in humans and dogs. 25,33 CD30+ intestinal ALCL has been reported in dogs. 15,32 In the present study, some cases of SCL (5%) and LCL (35%) were positive for CD30, indicating that a subset of canine ALCL is also derived from IELs in the background of CE. In humans, EATL with anaplastic morphology also expresses CD30, and type 2 RCD with CD30+ IELs was reported to have a worse prognosis or indicated transformation to EATL. 10
Human EATL is an aggressive disease with a poor prognosis, and there is currently no specific treatment. 12,22 Therefore, the validation of a suitable, spontaneous animal model is needed. The dog has often been proposed as a useful model for studying the etiology of or treatments for lymphoma. 11,19 Although ITCL is a common disease in older dogs, 20,38 its cause, phenotype, and genetic factors have not yet been elucidated in detail. In the present study, we described similarities and differences in histologic lesions, immunophenotype, and clonality results between canine ITCL and human EATL and also between canine CE and human RCD. The results obtained suggest that canine ITCL is derived from IELs that increase in number in CE. The CD8- immunophenotype of increased IELs was similar to that of type 2 RCD, suggesting similarities in the pathogenesis of canine ITCL and that of human EATL. The underlying pathogeneses of canine CE and ITCL remain unclear; however, the present results suggest the potential of canine ITCL as a model of human EATL.
There are 2 major differences between canine CE and human celiac disease. Human celiac disease is an autoimmune inflammatory enteropathy characterized by the production of autoantibodies, such as the anti-tissue transglutaminase antibody, 14 while elevations in autoantibodies in canine CE and ITCL have only been detected in a subset of dogs. 17 Furthermore, although the genetic backgrounds of HLA-DQ2 and HLA-DQ8 are known in celiac disease, 14,31 there is currently no reported genetic predisposition in canine CE. Therefore, molecular or genetic conditions appear to differ between canine ITCL and human EATL. In Japan, Shiba dogs were shown to be predisposed to CE and ITCL and may have some genetic predisposition. 18,24 To validate canine ITCL as a model of EATL, further studies on the molecular pathogenesis and genetic profile of neoplastic lymphocytes and IELs in IEL+CE are needed.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211057220 - Immunophenotyping of intraepithelial lymphocytes in canine chronic enteropathy and intestinal T-cell lymphoma using endoscopic samples
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211057220 for Immunophenotyping of intraepithelial lymphocytes in canine chronic enteropathy and intestinal T-cell lymphoma using endoscopic samples by Kazuhiro Kojima, James K. Chambers, Ko Nakashima, Yuko Goto-Koshino and Kazuyuki Uchida in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Grant-in-Aid from the Japan Society for the Promotion of Science (19H03122).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
