Abstract
Inflammatory bowel disease (IBD) and intestinal lymphoma are intestinal disorders in dogs, both causing similar chronic digestive signs, although with a different prognosis and different treatment requirements. Differentiation between these 2 conditions is based on histopathologic evaluation of intestinal biopsies. However, an accurate diagnosis is often difficult based on histology alone, especially when only endoscopic biopsies are available to differentiate IBD from enteropathy-associated T-cell lymphoma (EATL) type 2, a small cell lymphoma. The purpose of this study was to evaluate the utility of histopathology; immunohistochemistry (IHC) for CD3, CD20, and Ki-67; and polymerase chain reaction (PCR) for antigen receptor rearrangement (T-cell clonality) in the differential diagnosis of severe IBD vs intestinal lymphoma. Endoscopic biopsies from 32 dogs with severe IBD or intestinal lymphoma were evaluated. The original diagnosis was based on microscopic examination of hematoxylin and eosin (HE)–stained sections alone followed by a second evaluation using morphology in association with IHC for CD3 and CD20 and a third evaluation using PCR for clonality. Our results show that, in contrast to feline intestinal lymphomas, 6 of 8 canine small intestinal lymphomas were EATL type 1 (large cell) lymphomas. EATL type 2 was uncommon. Regardless, in dogs, intraepithelial lymphocytes were not an important diagnostic feature to differentiate IBD from EATL as confirmed by PCR. EATL type 1 had a significantly higher Ki-67 index than did EATL type 2 or IBD cases. Based on the results of this study, a stepwise diagnostic approach using histology as the first step, followed by immunophenotyping and determining the Ki67 index and finally PCR for clonality, improves the accuracy of distinguishing intestinal lymphoma from IBD in dogs.
Keywords
Inflammatory bowel disease (IBD) and intestinal lymphoma are common intestinal disorders, causing chronic persistent or intermittent gastrointestinal clinical signs such as diarrhea, vomiting, or weight loss in dogs. Unfortunately, the clinical presentation of the 2 entities is often similar, and diagnostic tests addressing those clinical signs, including fecal examination or deworming, blood tests, imaging, and correction of dietary errors, often remain inconclusive. 53,26,37
Morphologic assessment of the small intestine by endoscopic or surgical biopsy remains the ultimate diagnostic tool to differentiate IBD from intestinal lymphoma. 26,38 While endoscopy is less invasive and has minimal side effects for the patient in most cases, examination is limited to the mucosa, and even some deep mucosal lesions may escape detection in superficial biopsies. 14,35,57 Occasionally, an intense inflammatory infiltrate induced by the tumor may mask neoplastic cells, resulting in an inaccurate diagnosis of IBD. 37 While surgical biopsies are guided by gross examination of the intestinal tract, and the ileocecal junction as a primary location of intestinal lymphoma is easily accessible, selection of endoscopic biopsy sites is limited to the upper portion of the small intestine. Last, the quality of endoscopic biopsies can be dramatically affected by collection techniques, which prompted a recent publication of standards for collecting endoscopic intestinal biopsies and their microscopic interpretation. 13,55 Moreover, substantial inconsistencies in interpretation of intestinal biopsy specimens among pathologists were identified in some studies, and a standardization of microscopic interpretations of intestinal biopsies was proposed. 56,58
Based on recent advances in the classification of lymphomas in cats, the most common primary intestinal lymphoma is enteropathy-associated T-cell lymphoma (EATL) type 2, a small cell lymphoma in which the cells might be morphologically indistinguishable from IBD. 14,39,43,54,59 Differentiation of IBD from EATL type 2 may be further complicated by the potential transformation of IBD to lymphoma, as suggested by some. 12,24,28 Other less common intestinal lymphomas in cats include EATL type 1, a large cell lymphoma, or B-cell lymphomas, either diffuse large B-cell lymphomas or mucosa-associated lymphoid tissue lymphomas. Similar to cats, a predominance of intestinal T-cell lymphomas has also been reported in dogs, 21,38,40,44 which is in contrast to canine multicentric lymphomas as well as human gastrointestinal lymphomas that are more commonly of B-cell origin.1,19,23,25,32,36,42,46,52,59 Subclassification of canine intestinal T-cell lymphomas into EATL type 1 or 2 has not been reported. One study reported that 75% of canine intestinal lymphomas were T-cell lymphomas that exhibited epitheliotropism, which may suggest a high incidence of EATL type 2. 11
Immunohistochemistry (IHC) and clonality testing by polymerase chain reaction (PCR) for antigen receptor rearrangement (PARR) have been used in combination with histopathology to enhance our ability to accurately differentiate between IBD and intestinal lymphoma in dogs, cats, and humans.7,8,11,22,24,29,31,33,39,48,51 However, the incidence of EATL type 1 and 2 in dogs remains unclear, as does the diagnostic utility of IHC and PARR for differentiating IBD from intestinal lymphoma in dogs. One study evaluated this method for the diagnosis of 12 canine intestinal lymphomas and found a sensitivity of only 66.7%. Detailed studies to further differentiate canine intestinal lymphomas and to assess the utility of IHC and PARR for differentiating IBD from lymphoma are lacking.
The Ki-67 index, an indicator for the growth fraction of a cell population, has been used as a prognostic marker in canine and human lymphoma. 5,18,27,34,41 It was found useful for distinguishing between benign and malignant lymphoproliferative disorders in humans. 6 The Ki-67 index in T cells in the lamina propria in intestinal tissue from children with active IBD is very low compared with that of intestinal lymphoma. 15 There are few studies concerning Ki-67 expression in canine intestinal T-cell lymphoma, reporting a wide range of immunoexpression in 3.5% to 52.6% of neoplastic cells. 40 However, the Ki-67 index has not been studied as a marker for differentiating IBD from intestinal lymphoma in dogs.
The goal of this study was to evaluate the utility of histopathology, immunophenotyping, Ki-67 index, and PARR to more accurately diagnose intestinal lymphoma in dogs and to differentiate it from IBD.
Materials and Methods
Case Selection and Clinical History
Thirty-two dogs that had been presented to the Gastroenterology and Endoscopy Unit of the Veterinary Medical Teaching Hospital (Complutense University of Madrid) between 1999 and 2011 were included in this retrospective study. All dogs had a history of chronic gastrointestinal disease. Only endoscopic samples from the duodenum of dogs with a morphological diagnosis of either severe IBD (lymphoplasmacytic enteritis) or intestinal lymphoma were included. Dogs with parasitic disease or systemic disease affecting the intestinal tract or dogs with exocrine pancreatic insufficiency were excluded. Only endoscopic biopsies that were evaluated as “adequate” according to the recently published World Health Organization (WHO) criteria were included in the study. 57,58 History, physical examination, blood tests (hematology and blood biochemical), abdominal ultrasound, upper endoscopy, and endoscopic biopsy had been performed in all cases.
Histopathology, Immunohistochemistry, and PARR Testing
Multiple biopsy samples (8–10) were obtained by endoscopy of the duodenum of the 32 dogs. All samples were fixed in formalin, routinely processed, and embedded in paraffin. Serial sections from paraffin tissue blocks were cut for routine hematoxylin and eosin (HE) staining, IHC, and PARR.
IHC for Ki-67, CD3, and CD20 was performed, using a streptavidin-biotin-peroxidase complex method according to published methods. 10,34 Only cells with membranous and cytoplasmic expression of CD3 or CD20 were evaluated as positive, and labeling of over 50% of target cells was judged as positive. The Ki-67 index was determined by counting positive and negative lymphocyte nuclei in 10 representative fields of each slide (40× magnification) using computer image analysis. The Ki-67 index (KI) was defined as the percentage of Ki-67–positive lymphoid cells in 10 representative fields.
For PARR testing, DNA extraction was performed on five 6-μm-thick serial sections from formalin-fixed, paraffin-embedded tissues. The rearrangement of the TCRγ variable region was assessed by amplifying the complementary determining region 3 (CDR3) as previously described with slight modifications. 8 All PCR reactions were run in duplicate. Postamplification analysis of the products was performed on the QIAxcel Advanced Instrument (Qiagen, Valencia, CA), which allows high-resolution capillary electrophoresis of amplicons and provides electropherograms and gel images with the QIAxcel Screengel Software. A result was considered clonal or positive, as well as most consistent with a neoplastic cell population, when 1 or 2 sharp gel bands or sharp electropherogram peaks, 3 times the size of background peaks, appeared in duplicate samples within a target size range of 80 to 100 bp. 8 Polyclonal amplification, consistent with a nonneoplastic process, such as inflammation, was identified by the presence of a broad smeared band or multiple small peaks. Additional details on the IHC and PARR methodology are provided as supplemental materials.
Case Evaluation
Histologic sections of small intestine from each dog were evaluated by 2 pathologists together (MK, AR), without knowledge of the case history, and diagnosis was by consensus. A diagnosis of IBD, intestinal lymphoma, or lymphoma-suspect was made based on the microscopic appearance of the most severely affected section. Briefly, diagnosis of IBD (lymphocytic-plasmacytic enteritis) was based on the presence of persistent clinical signs, lasting more than 3 weeks, and a diffuse lymphoplasmacytic inflammatory infiltrate as the main histologic finding in the endoscopic biopsies. IBD diagnosis was only considered once all other possible causes of enteritis/infiltrates were investigated and excluded. 13,14,28,29 A diagnosis of lymphoma was based on a dense monomorphic population of neoplastic lymphocytes with signs of malignancy, such as invasion of the muscularis mucosae, architecture distortion, or abnormal cell size. The diagnosis of lymphoma-suspect was based on the presence of a dense focal or patchy monomorphic mucosal infiltrate and/or the presence of intraepithelial lymphocytes forming plaques or nests in 1 or more intestinal villi.
Without prior knowledge of these results, HE sections were then studied in conjunction with IHC for CD3 and CD20 by the same pathologists, and a second diagnosis was made. Briefly, IBD was diagnosed based on mixed inmunophenotype with CD3+ and CD20+ cells, T-cell lymphoma based on large numbers of CD3+ cells with scarce CD20+ cells corresponding to the inflammatory background, B-cell lymphoma based on numerous CD20+ cells with sparse CD3+ cells corresponding to the inflammatory background, or lymphoma-suspect based on focal dense areas of CD3+ cells in the villi and/or CD3+ intraepithelial plaques. The diagnoses were revised based on uniformity of cell labeling, phenotype in association with cell localization, and cell size as well as growth pattern. The number of cases that were reclassified after immunophenotyping was recorded. Each case was analyzed a third time, without knowledge of previous diagnosis, by combining histomorphology (HE), IHC, and PARR. The differences in diagnosis at each step were compared.
In addition, morphologic features of crypt distension (normal/mild/moderate/marked), lacteal dilation (normal/mild/moderate/marked), villous stunting (normal/mild/moderate/marked), mucosal fibrosis (normal/mild/moderate/marked), presence of increased intraepithelial lymphocytes (more than 20–30 per 40× stretch of villous epithelium), 13 the distribution of the lymphocytic infiltrate (focal, multifocal or diffuse), and monomorphism versus polymorphism of lymphocytes based on nuclear size and morphology were recorded for each case. Furthermore, intraepithelial lymphocytes were assessed in the entire sample and defined as “diffuse” or “involving one to several villi.” 34 The infiltrates of T cells within the epithelium were recorded as either surface or crypt infiltrates or both, and intraepithelial infiltrates were divided into single cells, nests, or plaques. Nests of lymphocytes were defined as ≥5 clustered intraepithelial lymphocytes, while plaques were defined as ≥5 adjacent epithelial cells overrun by lymphocytes. 33 In the lymphoma group, the number of mitotic figures per ten 400× fields was counted, and cell size (large vs small, >1.5 blood cell size vs <1.5 blood cell size) was evaluated. 49 The lymphoma cases were classified according to the WHO classification of lymphoid neoplasms. 3,47
Statistical Analysis
Data were analyzed by using SPSS statistical software (SPSS, Inc., an IBM Company, Chicago, IL). A “Descriptive statistics” procedure was used for quantitative variables and a “frequencies” procedure for qualitative variables. 16 The Pearson χ 1 test was used to determine independency between quantitative variables. One-way analysis of variance (ANOVA) was used for mean comparison of Ki-67 among groups. P ≤ .05 was considered statistically significant.
Results
Clinical Data
The age of the dogs in this study was 7.65 ± 3.00 years (mean ± SD). Twenty dogs (62.5%) were male and 12 (37.5%) were female. The most represented breeds were Rottweiler (n = 5), Yorkshire terrier (n = 4), boxer (n = 3), cocker spaniel (n = 3), and gos d'Atura catalán (n = 2).
Clinical signs at presentation were related to the gastrointestinal tract and included weight loss (96.9%), chronic diarrhea of small bowel origin (81.3%), and chronic vomiting (53.1%). A remarkable number of cases also had varying degrees of decreased appetite (53.1%).
Case Evaluation
All 32 cases were diagnosed by histopathology, immunophenotyping, and PARR. Based on morphologic evaluation alone, 7 cases were classified as intestinal lymphoma (21.9%), 12 cases as IBD (37.5%), and 13 as lymphoma-suspect (40.6%). When IHC for CD3 and CD20 was analyzed in conjunction with the microscopic findings, 8 cases were classified as T-cell lymphoma (25%; Fig. 2), 15 cases as IBD (46.9%; Fig. 1), and 9 cases as lymphoma-suspect (28.1%). There were no B-cell lymphomas. IHC allowed reclassification of 6 of the 13 cases originally classified as lymphoma-suspect; 5 were thought to be IBD and 1 a T-cell lymphoma. However, 2 cases originally diagnosed as IBD were reclassified as lymphoma-suspect based on the combined HE and IHC results. All 7 cases originally diagnosed as lymphoma remained in the lymphoma group and were further classified as T-cell lymphomas. When PARR results were used in conjunction with HE and IHC results, all 9 cases that had been included in the lymphoma-suspect group were diagnosed as IBD, all 15 cases in the IBD group were diagnosed as IBD, and 8 cases of lymphoma were diagnosed as T-cell lymphoma (Tables 1, 2). All lymphomas in this study were T-cell lymphomas. The neoplastic lymphocytes were classified as large cells in 6 of these cases and as small cell in 2 cases, resulting in a diagnosis of EATL type 1 and type 2, respectively. 3,47 The average number of mitosis per 400× field was 13.8 for EATL type 1 and 7.5 for EATL type 2.
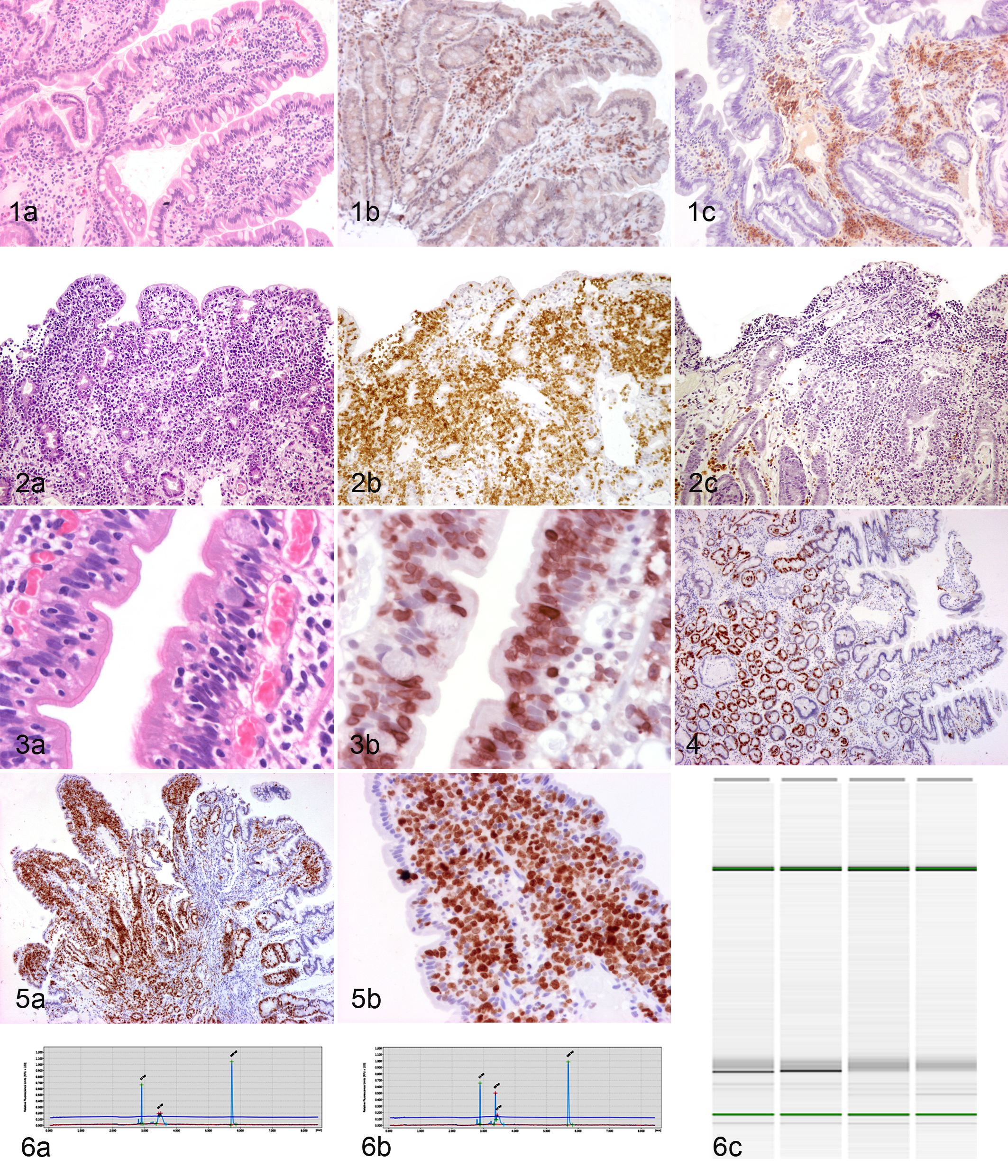
Inflammatory bowel disease, small intestine, dog. (a) Hematoxylin and eosin (HE), (b) immunohistochemistry (IHC) for CD3, and (c) immunohistochemistry for CD20. Notice the mixed immunophenotype, characteristic of an inflammatory process.
Comparison of Diagnoses Based on Morphologic Evaluation Alone (HE), in Combination With Immunophenotyping (IHC), and in Combination With Immunophenotyping and PARR.
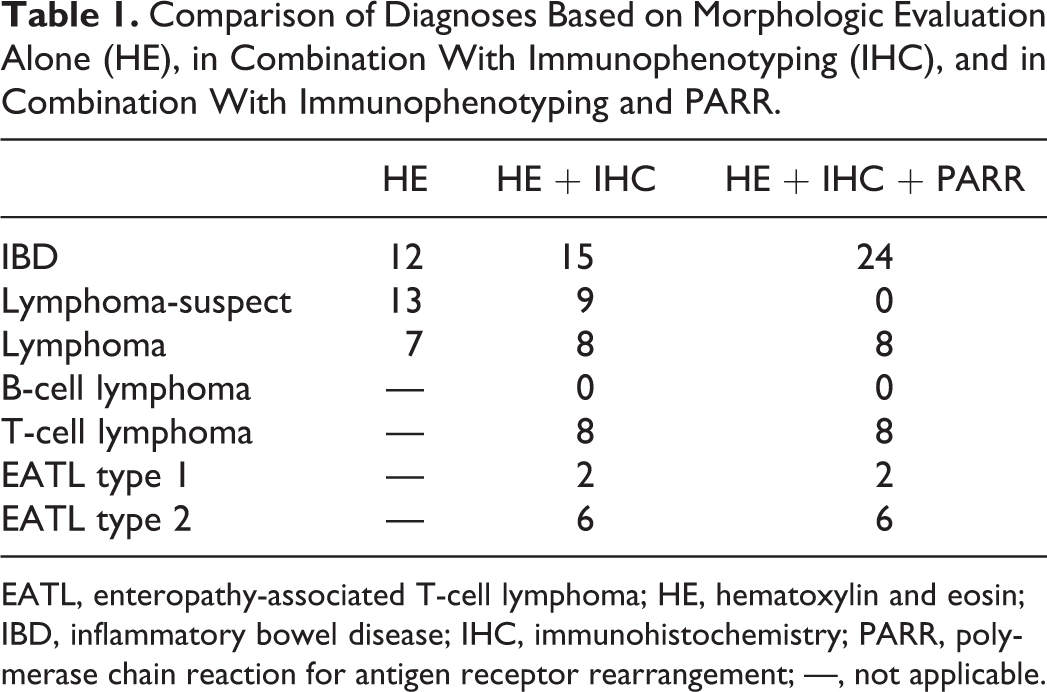
EATL, enteropathy-associated T-cell lymphoma; HE, hematoxylin and eosin; IBD, inflammatory bowel disease; IHC, immunohistochemistry; PARR, polymerase chain reaction for antigen receptor rearrangement; —, not applicable.
Changes of Diagnosis After Combining the Morphologic Evaluation With Immunophenotyping and With Immunophenotyping Plus PARR.

HE, hematoxylin and eosin; IBD, inflammatory bowel disease; IHC, immunohistochemistry; PARR, polymerase chain reaction for antigen receptor rearrangement.
PARR Testing
PARR testing was performed on all 32 cases. Six cases were determined to have a clonal population of T cells, while the rest of the samples were polyclonal. All clonal samples had been diagnosed as T-cell lymphoma based on HE and IHC results. However, 2 samples from the lymphoma group were polyclonal based on PARR testing, a result that was considered incorrect after further review of the morphology and IHC results, which supported the diagnoses of lymphoma (Fig. 6). When HE and IHC results were considered in combination with PARR, all the cases included in the IBD and lymphoma-suspect groups were classified as IBD.
Morphologic Features
All samples had a severe lymphocytic and variable plasmacytic infiltration within the lamina propria. The distribution of this severe infiltration was diffuse in 20 of 24 cases of IBD and 6 of 8 cases of lymphoma and multifocal in 4 of 24 and 2 of 8 cases of IBD and lymphoma, respectively. According to The World Small Animal Veterinary Association (WSAVA) standards, 13 villous stunting was found in most cases but was statistically more severe in lymphomas (P = .004). Epithelial injury was observed more frequently and was more severe in lymphomas than in IBD cases (P = .002). Crypt distension, lacteal dilation, and mucosal fibrosis appeared to be similar in cases of IBD and lymphoma.
Intraepithelial lymphocytes were always CD3 positive and CD20 negative and were therefore evaluated in CD3-labeled slides. Twenty-four cases (8/8 lymphoma, 16/24 IBD) had increased intraepithelial infiltrates, with most cases presenting with diffuse intraepithelial infiltrates within the surface epithelium. Intraepithelial infiltrates involving only 1 to several villi were observed in 2 of 8 cases of lymphoma and 4 of 24 cases of IBD. Intraepithelial infiltrates affecting both the surface and the crypts were significantly less common in IBD (2/24) than in lymphoma (5/8; P = .004) cases. Of the 24 cases that had intraepithelial infiltrates, 7 cases had only single intraepithelial lymphocytes (6/24 IBD, 1/8 lymphoma), whereas 5 had nests (3/24 IBD, 2/8 lymphoma), and 12 had plaques (7/24 IBD, 5/8 lymphoma; Fig. 3).
In 15 cases (8/8 lymphoma, 7/24 IBD), the lymphocytic infiltration was predominantly monomorphic. Monomorphic infiltrates in IBD cases were observed in the sections most severely affected, and this morphologic feature had originally led to a diagnosis of lymphoma-suspect in all 7 IBD cases. A diagnosis of IBD was finally reached based on PARR testing. Polymorphic infiltrates were identified in 17 cases of IBD.
Immunohistochemistry for Proliferation Index Ki-67
IHC for Ki-67 was performed on all 32 cases. All lymphomas expressed a significantly higher Ki-67 index (median ± SD: 52.9% ± 11.9%) than IBD (15.3% ± 10.0%) (P < .001) cases. Ki-67 immunolabeling in lymphomas ranged from 30% to 64% of neoplastic cells, while only 5 cases of IBD had a Ki-67 index higher than 20%, and only 2 had a Ki-67 index between 30% and 50% (Figs. 4 and 5).
Discussion
Similar to previous studies, clinical signs or the gross appearance of the duodenal surface as observed by endoscopy did not correspond to the degree of histopathologic changes and were insufficient for differentiating between intestinal lymphoma and severe IBD in this study. 14,28,37,43,45 While surgical biopsies provide the most diagnostic detail, histologic interpretation of duodenal tissues obtained by endoscopy is difficult, and ideally, sections throughout the small intestine are encouraged. 14,37,56 The examined morphologic features such as villous stunting, crypt distension, lacteal dilation, or mucosal fibrosis did not help differentiate between severe IBD and lymphoma. 13 While dogs with intestinal lymphoma would be expected to present with a monomorphic lymphocytic population, in many cases, intestinal lymphomas may occur in conjunction with lymphoplasmacytic inflammation. 37 Combining HE with IHC evaluation improved the ability to differentiate canine intestinal T-cell lymphomas from inflammation, similar to what has been reported in cats. 2,34,54 In our study, all lymphoma cases were diffuse T-cell lymphomas with a lymphoplasmacytic inflammatory background, which is similar to an earlier study. 38 Other studies identified between 63% and 75% of gastrointestinal lymphomas as having a T-cell phenotype, but these studies also examined the stomach, which is known to have a higher incidence of B-cell lymphomas in dogs and cats. 4,11,20,39,44
The only statistically significant morphologic parameter for differentiating IBD from lymphoma was the presence of intraepithelial infiltrations in both surface and crypt epithelium. In contrast to cats, a significant number of dogs with a diagnosis of IBD based on the combined HE, IHC, and PARR results exhibited marked intraepithelial infiltrates. The occurrence of intraepithelial plaques and nests that has been shown to be a strong indicator of EATL type 2 in cats was not predictive of intestinal lymphoma in dogs. Since EATL type 1 was much more commonly observed in dogs than EATL type 2, epitheliotropism may be a much less important diagnostic parameter in dogs. In humans, EATL type 1 is the predominant intestinal T-cell lymphoma, with an 80% to 90% prevalence, while in cats, EATL type 2 is the predominant intestinal T-cell lymphoma. 3,34,39,47 Our data are similar to a previous study that reported 3 cases of small cell, 4 of intermediate cell, and 4 of large cell T-cell lymphomas. 40
Even when combining morphologic evaluation with IHC and PARR, a certain diagnosis could not be reached in all cases. Some of the cases in our study that were diagnosed as IBD based on polyclonal PARR testing may represent early stages of EATL type 2. 7,9,47,50 A polyclonal PARR result of an intestinal lymphoma may be due to low numbers of neoplastic cells in a strong inflammatory background or low sensitivity of the multiplex PCRs covering insufficient antigen receptor rearrangements. A recent publication of new sets of primers for PARR for canine TCRG improved the test sensitivity in dogs by 25% compared with the primers used in this study 30 Considering that 2 cases diagnosed as lymphoma based on morphology and IHC results were polyclonal by PARR testing, a sensitivity of less than 75% for PARR also has to be assumed for our study. The sensitivity of PARR for detecting clonal cell proliferations was lower than what has been published for other types of canine lymphoma 8 but higher than previously reported by others (66.7%) for canine intestinal lymphoma. 22 The increase in sensitivity in our study may be due to the use of capillary electrophoresis rather than a polyacrylamide gel, leading to an increased resolution and thereby sensitivity. We performed PARR testing in duplicate to ensure that no single bands representing pseudoclones were misinterpreted as a clonal cell population. In contrast to previous studies, we also analyzed both not applicable native and denatured-reannealed products (heteroduplex analysis). 31 Denaturation and reannealing do not affect bands amplified from truly clonal cells but will produce a smear for pseudoclonal cell populations. Failure to eliminate pseudoclones may result in loss of specificity of up to 10%. 31,50 Integrating the newly developed primers into PARR testing combined with morphology and IHC will likely aid in making a more accurate diagnosis.
Ki-67 has been used as a marker of cellular proliferation in canine and human lymphoma, and the Ki-67 index correlates with the prognosis of canine and human lymphoma. 5,19,34,41 There are no previous studies investigating the utility of the Ki-67 index to differentiate canine IBD from intestinal lymphoma. In humans, lamina propria T-lymphocytes in IBD are normally not dividing, despite mucosal inflammation. 15 Furthermore, benign versus malignant lymphoid proliferations have been differentiated based on the Ki-67 index in lymph nodes. 6 We demonstrated that the Ki-67 index was significantly higher in canine lymphomas than in IBD. Lymphomas exhibited between 30% and 62% of Ki-67–positive cells, while most cases of IBD displayed a Ki-67 index lower than 25%. Previous studies indicated that canine T-cell lymphomas may have a higher Ki-67 index than B-cell lymphomas. 17,42 The mean Ki-67 index in the lymphoma group was higher than previously published values for intestinal lymphomas 40 but similar to multicentric T-cell lymphomas in other studies. 17,42 EATL type 2 lymphomas had the lowest Ki-67 index, and a similar index may be observed in IBD cases with large numbers of infiltrating T cells. In the current study, only 1 case of IBD had a high Ki-67 index (47.15%), similar to the Ki-67 index in lymphomas. The affected dog did not respond to treatment for several weeks and was euthanized. No follow-up necropsy was available, and it remains unclear whether this dog had intestinal lymphoma. Based on our current knowledge, Ki-67 should not be used as a sole parameter to differentiate IBD from intestinal lymphoma. However, integration of the Ki-67 index into a diagnostic panel composed of the histopathologic evaluation, immunophenotyping, and PARR testing can be useful. These tests should be performed in sequence, and test results should only be interpreted in context. Especially for dogs that have been diagnosed as lymphoma suspects or with IBD but do not respond appropriately to treatment, the histopathology should be reviewed in combination with immunophenotyping, PARR testing, and evaluation of the Ki-67 index. Immunophenotyping and determining the Ki-67 index are recommended in any case with a morphologic diagnosis of lymphoma, and PARR testing can be performed as a confirmatory test. Regardless, additional biopsies may be required throughout the course of the disease. This systematic assessment of canine endoscopic biopsies will increase diagnostic accuracy and will enable both proper prognosis and appropriate treatment.
Footnotes
Author Contribution
Conception or design: VC, ARB, FRF, AW, RM, TM, MK. Data acquisition, analysis, or interpretation: VC, ARB, FRF, AW, RM, TM, MK. Drafting the manuscript: VC, MK. All authors participated in critically revising the manuscript, gave final approval, and agree to be accountable for all aspects of work to ensure integrity and accuracy.
Acknowledgements
We thank Tom Wood and the DCPAH Histology and Immunohistochemistry Laboratory. We also thank the personnel of the DCPAH Virology Laboratory. We are grateful to the Complutense Veterinary Teaching Hospital Histology Laboratory for technical support and to Santiago Cano for his help with statistics.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Violeta Carrasco's stay in the DCPH (Michigan State University) was financed by the scholarship program of the Spanish Ministry of Education: University Teaching Training Program (Formación de Profesorado Universitario-FPU), code AP2007-01210.
