Abstract
Canine pigmented viral plaques (PVPs) are proliferative epidermal lesions caused by canine papillomaviruses (CPVs). Although the lesions are benign, neoplastic transformation has been reported. Cases reported in the literature are few and mainly focused on genome sequencing. The aim of this study was to collect data on the epidemiology, clinicopathological features, and genotyping of PVPs. Fifty-five canine PVPs were retrospectively retrieved and histologically evaluated. Follow-up was available for 33 cases. The median age was 6.5 years and pugs were the most represented breed (25%). There were 4 clinical presentations: a single lesion (24%), multiple lesions (75%) in one (41%) or different sites (34%), and generalized lesions all over the body (24%). The abdomen and axillae were the most common sites. In single lesions, no recurrence was observed after conventional surgery, whereas different medical treatments reported for multiple lesions were not successful. Spontaneous regression was reported in 3 cases. Neoplasia in contiguity with PVPs was seen in 5 of 55 lesions (9%), and 1 dog was euthanized due to invasive squamous cell carcinoma (SCC). The most useful histopathological features for diagnosis were scalloped profile, epidermal spikes, hypergranulosis, and hyperpigmentation. L1 immunolabeling was present in 14 of 16 cases (87%). Sequencing revealed that 10 of 16 cases were associated with CPV-9 (71%), 2 cases were associated with CPV-4 (14%), and 2 cases were associated with CPV-8 (14%). In conclusion, this represents a large cohort study on canine PVPs reporting data on clinicopathological features, therapy, outcome, and the type of CPV involved for the first time in Italy.
Keywords
Canine pigmented viral plaques (PVPs) are benign, proliferative epidermal lesions associated with canine papillomaviruses (CPVs) belonging to the genus Chipapillomavirus. To date, CPVs identified from pigmented plaques are CPV-3, 4, 5, 8, 9, 10, 11, 12, 14, 16, 18, and 24.13,20,22,32 The first report investigating a role of papillomavirus (PV) in the genesis of PVPs dates back to 1995. 23 It is noteworthy that similar cases had been published previously, but were considered to be idiopathic and hereditary and were named lentiginosis profusa and referred to as pigmented epidermal nevus. 33 Restriction enzyme analysis and molecular hybridization experiments recognized these lesions as papillomavirus-induced with features distinct from the more commonly encountered canine oral papillomatosis. 14 Subsequent genomic studies identified several CPV types as the causative agents not only from PVPs but also from squamous cell carcinoma (SCC), basal cell carcinoma (BCC), and trichoblastoma.5,17,15,21,25,30 The malignant potential of these documented CPVs has been hypothesized and other PV types involved in the genesis of viral plaques cannot be excluded. In fact, although viral plaques are considered benign lesions, neoplastic transformation to SCC with visceral metastasis, although rare, has been reported. In particular, CPV-3 has been found in PVPs that progressed to SCC over a 20-month to 5-year period in both immunocompetent and immunocompromised dogs. 21
Since 1995, 56 cases of viral plaques in dogs have been published in different parts of the world, whereas data are not yet available in Italy.9,12,14,31 To date, case reports and a few case series have been described in the literature, mainly focusing on the sequencing of the implicated CPV genome.11,15,16,36 The aim of this study is to provide data on the epidemiology and clinicopathological features of PVPs in dogs in a series of Italian cases. Data on genotyping, prognosis, and therapeutic approaches are presented as available.
Materials and Methods
Case Selection and Histology
Cases of PVPs were obtained retrospectively from biopsies submitted to the Department of Veterinary Sciences (Pathology section) of the University of Pisa (Italy) and MyLav Laboratory (Passirana di Rho, Italy) over a period of 9 years, from 2014 to 2022. The complete signalment, history, clinical signs, diagnosis, and anatomical location of the lesions were recorded for each dog from the histopathology submission forms. Further clinical data on treatment and outcome for at least a 6-month follow-up period were obtained for each case, when available, by administering a questionnaire to the referring veterinarians (Supplemental Material).
Formalin-fixed paraffin-embedded biopsy samples were routinely processed, and 5-μm-thick tissue sections were stained with hematoxylin and eosin to evaluate the main histopathological features of PVPs according to the guidelines of the Surgical Pathology of Tumours of Domestic Animals Series and the current scientific literature.7,8,24,28,29 Histopathologic features evaluated included epidermal spikes (long and thin surface digitation), scalloped surface, epidermal hyperplasia, hypergranulosis, orthokeratotic hyperkeratosis, hyperpigmentation, koilocytes (defined as keratinocytes with shrunken nuclei surrounded by a clear cytoplasmic halo), and dermal melanophages. Additional features commonly associated with PV infection, such as blue-gray cytoplasmic pallor and intranuclear inclusion bodies, were also examined. The histopathological diagnosis was then confirmed by 2 veterinary pathologists (MO, FAb) and 2 dermatologists with dermatopathology experience (FAl, LP).
Immunohistochemistry and Molecular Biology Analysis (Polymerase Chain Reaction and Sequencing)
In 16 cases, additional 5- and 10-μm-thick sections were used for immunohistochemistry (IHC) and molecular assays. To avoid cross-contamination, the microtome blade holder was covered with a new piece of adhesive tape and a new blade was used for each sample.
Immunohistochemistry for the detection of PV was performed using a mix of mouse monoclonal antibodies raised against the major capsid proteins (L1) of bovine PV1 and human PV16 (ab-2417; Abcam, Cambridge, UK), which react with different PV types. Immunohistochemistry was performed using a standard protocol previously reported. 26 A canine oral papilloma and a squamous papilloma were used as positive and negative controls, respectively. Only the positive control showed a diffuse nuclear immunolabeling, as expected.
For the polymerase chain reaction (PCR) assay, DNA was extracted from formalin-fixed paraffin-embedded sections from each of the 16 cases (QIAamp DNA FFPE tissue kit and deparaffinization solution; Qiagen, Germany) according to the manufacturer’s instructions. DNA was eluted in 100 μL of elution buffer and stored at −20°C until analyses.
Samples were initially tested with the generic primers MY09/MY11, FAP59/FAP64, PapE1F/PapE1R, and CP4/CP5 (Supplemental Table S1). Subsequently, specific primer sets were designed for each CPV (Primer 3; NCBI) using the genomic sequence of the major capsid protein (L1) gene of CPV-3, 4, 5, 8, 9, 10, 11, 12, 14, 15, 16, 18, and 20 available in GenBank (Supplemental Table S2).
To ensure primer specificity and to avoid unspecific amplification of the host genome, the entire genome of Canis familiaris (Taxid: 9615) was examined. Efficiency of DNA extraction and DNA integrity were verified by PCR using a set of primers designed to amplify a 212 bp amplicon of the reference gene encoding ribosomal protein RPL15 of Canis familiaris (NG_030479.2). Polymerase chain reaction was performed in a thermocycler (iCycler; Bio-Rad, Italy) using a thermostable DNA polymerase (Wonder Taq Hot Start; EuroClone, Italy) following manufacturer’s instructions using a Tm of 60°C and using as template 2.5 μL of the extracted DNA. As a positive PCR control, a panel of positive CPV and Felis catus papillomavirus DNA samples from previous studies were tested using a set of broad PV primers available in the literature.21,26,34
Each PCR assay was also performed in the presence of negative controls (DNA extracted from a normal dog skin section and no template control), to determine the specificity of the reaction. All PCR products were analyzed by a 1% agarose gel electrophoresis in the presence of a 100-bp quantitative DNA marker (SharpMass 100; EuroClone).
All PCR-positive samples that yielded amplicons of the expected size were subjected to sequence analysis (BMR Genomics, Padova, Italy) using the Sanger method. 27 To confirm the CPV type, the obtained nucleotide sequences were subjected to BLAST analysis (https://blast.ncbi.nlm.nih.gov/Blast.cgi).
Results
Clinical Presentation, Therapy, and Follow-up
Fifty-nine cases with a histopathological diagnosis of canine PVPs were initially collected, of these 4 cases were excluded because they did not meet the inclusion criteria to be considered PVPs, as listed above. The signalment and clinical presentation collected for each dog from histopathology submission forms are reported in Supplemental Table S3.
The most common breeds in this study were pugs (n = 14), mixed breed (n = 8), miniature Pinscher (n = 5), English setter (n = 4), Dachshund (n = 4), English bulldog (n = 2), French bulldog (n = 2), Chihuahua (n = 2), Yorkshire terrier (n = 2), and Italian hound (n = 2). The following breeds were represented by single dogs: American Staffordshire, Epagneul Breton, Golden retriever, Jack Russell terrier, pitbull terrier, pointer, Shih-tzu, Staffordshire bull terrier, Weimaraner, and West Highland white terrier. The age of the dogs ranged from 5 months to 13 years (median: 6.5 years; interquartile range: 4–8 years), and 17% of the lesions (9 cases) were found in dogs younger than 2 years. No sex predilection was observed (31 females and 24 males).
The number and distribution of lesions was known for 54 animals. Thirteen cases had a single lesion (24%), whereas the remaining 41 dogs (76%) had multiple lesions. Of these, 10 were generalized over the body (24%) while the remaining (31, 76%) were multiple, either grouped in a localized body site (17, 55%) or in different sites (14, 45%) (Fig. 1a–d). Single lesions were distributed over the head (6), trunk (3), and limbs (3); the exact localization was not reported for 1 dog. The most common site of multiple grouped viral plaques was the abdomen (9 cases as single site, 6 cases with multiple sites including the abdomen). The limbs were involved in 16 cases (9 cases involving the axillae), head and neck in 4 cases, and the chest and the flank in the other 6 cases.
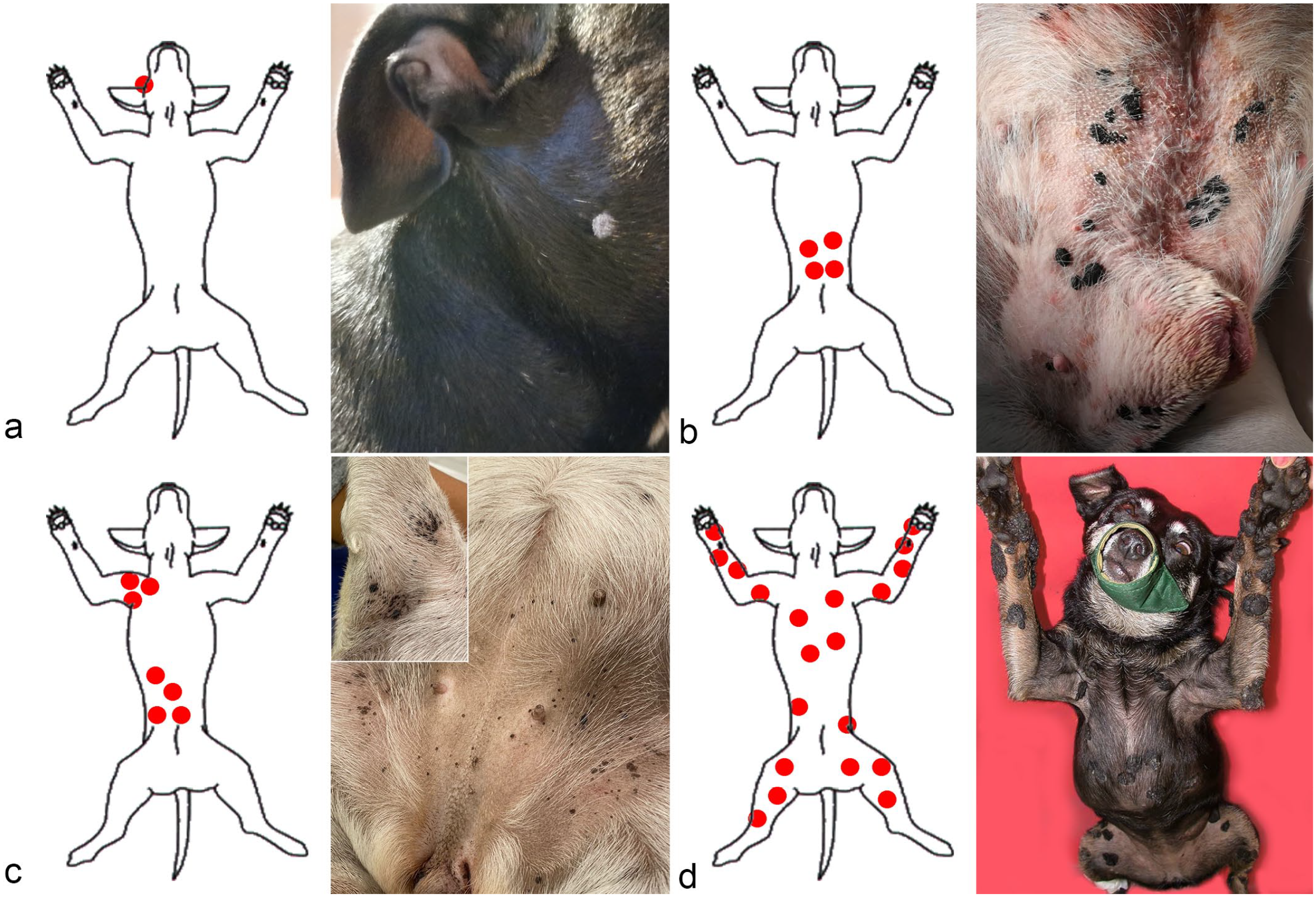
Dog. Schematic and clinical picture of different clinical presentations. (a) Single lesion. Staffordshire bull terrier with a hyperkeratotic plaque on the cheek (case 25) (Courtesy of Dr Barbara Vieri). (b) Multiple grouped lesions in one anatomical site. English bulldog with multiple grouped plaques in the ventral abdomen (case 21) (Courtesy of Dr Francesca Bonaga). (c) Multiple grouped lesions in more than 1 anatomical site. Pug with multiple grouped plaques on the abdomen and right forelimb (inset) (case 41) (Courtesy of Dr Martina Nava). (d) Generalized lesions. Mixed-breed dog with multiple and often coalescing plaques all over the body (case 51) (Courtesy of Dr Federico Leone).
In 41 cases, a description of the macroscopic features of the lesions was reported by the referring veterinarian, which included macules (13 dogs, 31%), plaques (11 dogs, 27%), and verruca (pedunculated/papillated; 10 dogs, 25%). Three dogs were reported to have both macules and plaques. Pigmentation was reported in 34 cases (83%) (Fig. 2a–d).
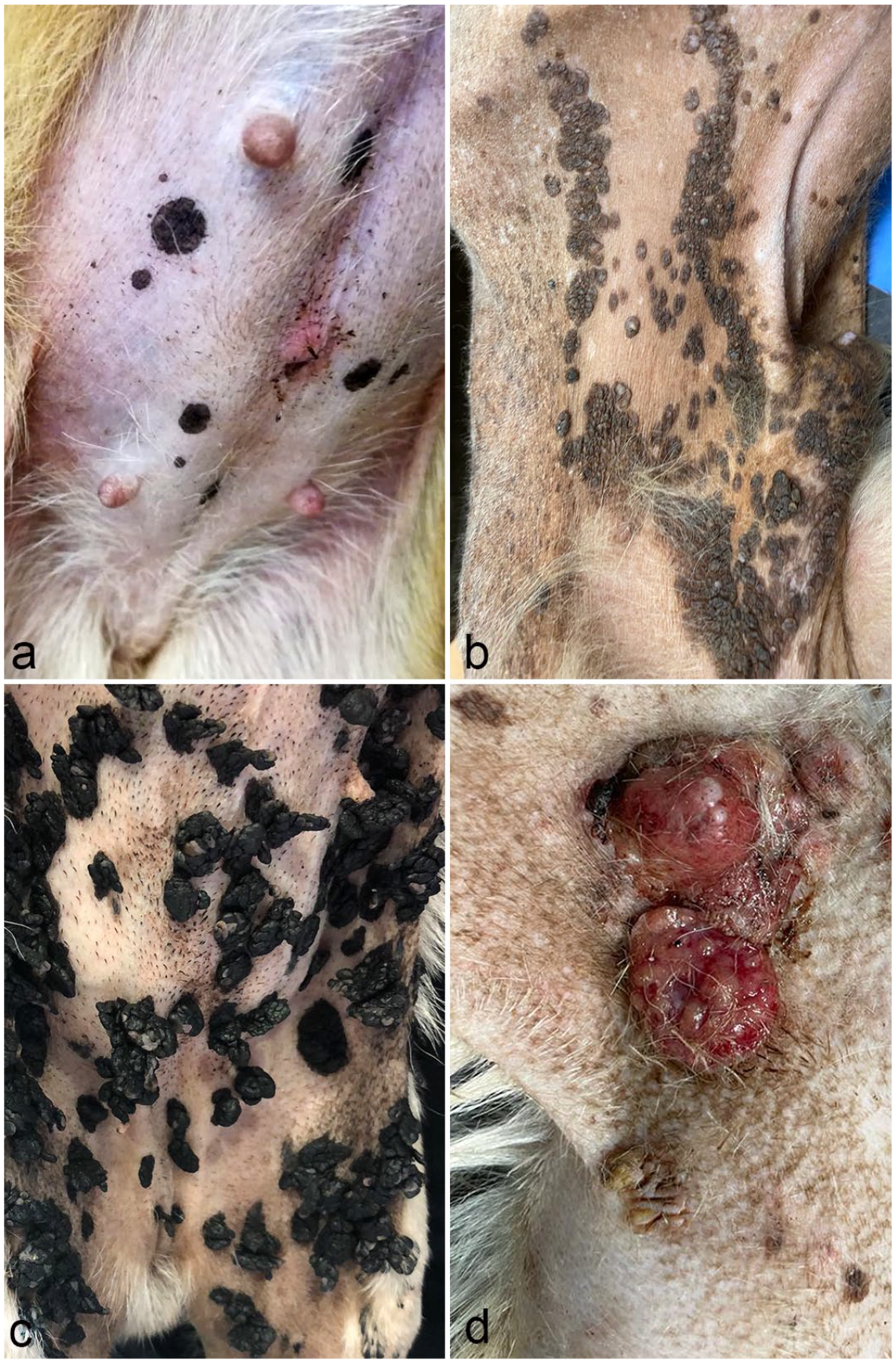
Dog. Various macroscopic features of viral plaques. (a) Multiple pigmented macules ranging from 3 mm to 1.5 cm on the abdomen of a Chihuahua (case 14) (Courtesy of Dr Marco Fornasarig). (b) Multifocal to coalescing pigmented plaques on the abdomen of a mixed breed dog (case 42) (Courtesy of Dr Fabiano Necci). (c) Numerous pigmented pedunculated warts on the abdomen and inner thighs of a pug (case 4) (Courtesy of Dr Federica Rossi). (d) Variably pigmented single and confluent plaques on the lateral chest, around an extensive ulcerated squamous cell carcinoma in an English setter (case 43) (Courtesy of Dr Marta Bianchi).
Complete follow-up questionnaires were obtained for 33 dogs. The duration of the lesions at biopsy ranged from a few weeks to years (in most cases the duration of the disease was approximate). Systemic signs were only reported in 2 dogs with hypothyroidism (case 26) and fecal incontinence (case 31), respectively. Concomitant cutaneous lesions were reported in 11 dogs, including inflammatory and neoplastic conditions (Supplemental Table S3). Atopic dermatitis and otitis were diagnosed in 4 dogs (cases 30, 34, 46, and 49), demodicosis was diagnosed in 2 dogs (confirmed histologically, cases 27 and 43), and solar/actinic dermatosis was diagnosed in 1 dog (case 42).
Skin neoplasms (6 cases) were seen alone or in combination in the same dog, which included a keratinizing BCC in 1 dog (case 11), SCCs in 2 dogs (cases 42 and 43), a SCC combined with a BCC in 1 dog (case 27), and multifocal trichoepitheliomas in 2 dogs (cases 21 and 44), one of which also had a pilomatricoma (case 44). All these dogs were adult, aged between 6 and 13 years (mean 10 years), and one of these was euthanized due to neoplastic invasion of the perianal area (case 21). Histologically, in 5 of the 6 cases (cases 11, 21, 27, 43, and 44) each of the aforementioned neoplasms were seen in contiguity with PVPs. In 1 dog (case 42), SCC was diagnosed on the dorsum associated with actinic dermatosis, and in this case, PVPs were diagnosed in a separate biopsy from the abdomen. Interestingly, 2 dogs with neoplasia in contiguity with PVPs had concomitant Demodex spp. infection.
According to the questionnaires received (n = 33), therapeutic interventions and outcomes are listed in Supplemental Table S4. Therapy for viral plaques was surgical in 11 cases, medical in 4 cases, and not performed in 18 cases. In 8 cases, conventional surgery with clean margins was the only treatment for single lesions, while in the other 3 cases, en bloc surgery was performed for grouped or generalized lesions. Recurrence was not reported for single lesions following excision and tumor-related death was not reported. The survival times ranged from 5 months to 4 years. Of the 18 cases that did not undergo therapy for PVPs, all had multiple lesions and 5 of these had a generalized presentation.
Of the 33 dogs, 2 were lost to follow-up and 8 dogs died. Of the dogs that died, 1 dog was euthanized because of an invasive SCC arising in contiguity with PVP, 3 died for causes other than PVP (cardiomyopathy, tumor at the base of the heart with pulmonary metastasis, and a laryngeal tumor), and 3 died due to unknown causes.
Histopathology
All 55 lesions had epidermal hyperplasia with a scalloped profile, hypergranulosis, and orthokeratotic hyperkeratosis. Hyperpigmentation was present in 51 cases, and in 33 of these cases, there were associated dermal melanophages. Thirty-six lesions contained epidermal spikes between shallow cup-shaped depressions. Nineteen cases contained scattered koilocytes. Viral cytopathic effect (blue-gray cytoplasm) was seen in 1 case. Intranuclear inclusion bodies were not observed in any case (Fig. 3a–c).
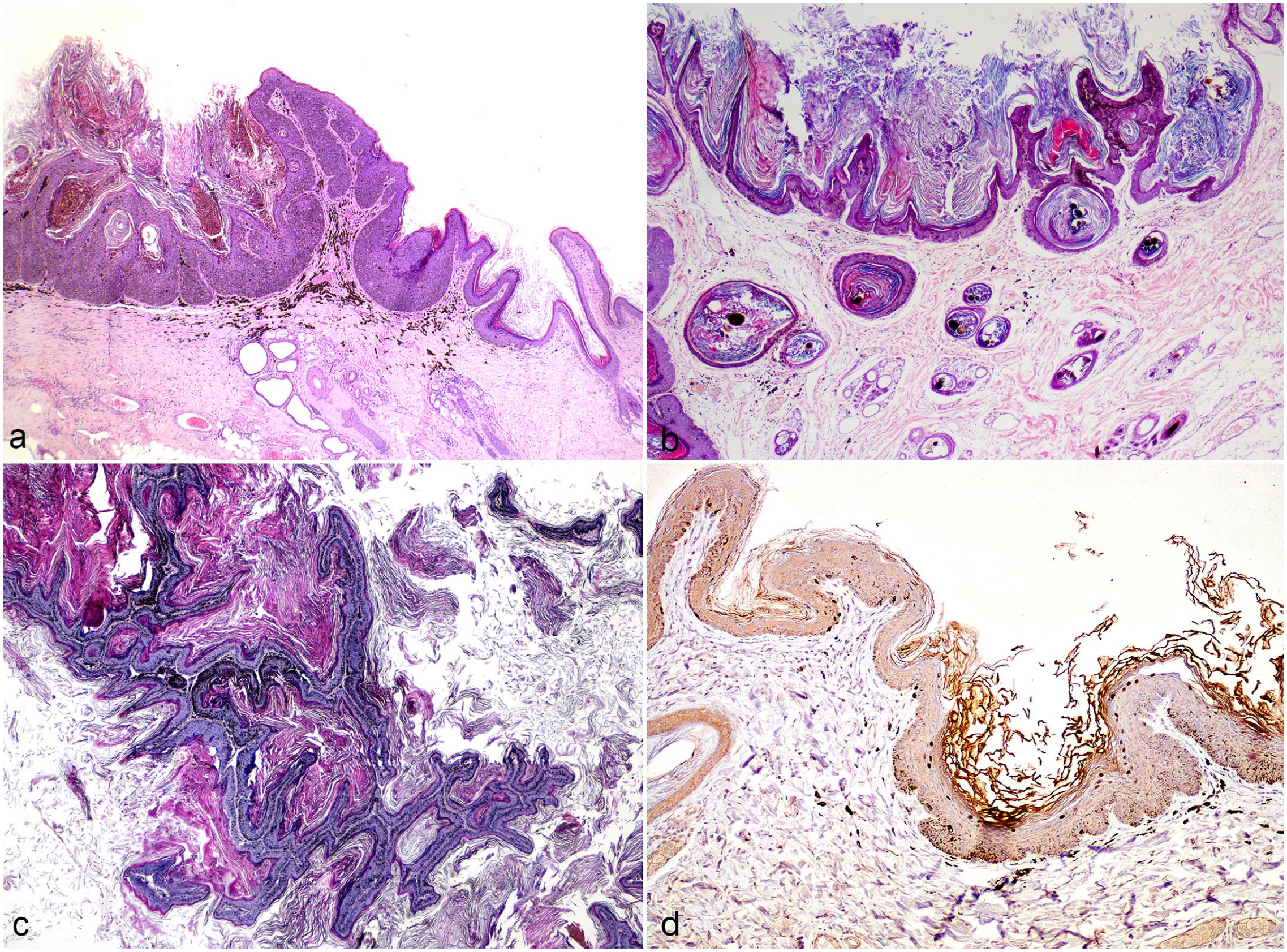
Dog. Histopathology and immunohistochemistry (IHC) of canine viral plaques. (a) Hyperpigmented and hyperkeratotic plaque with scalloped surface that involves the epidermis and follicular infundibula. Case 11. Hematoxylin and eosin (HE). (b) Pigmented plaques with scalloped appearance, epidermal spikes, and marked hyperkeratosis. Case 51. HE. (c) Pigmented plaque with filiform projections, mild epidermal hyperplasia, and marked hyperkeratosis. Case 46. HE. (d) Nuclear immunolabeling for L1 capsid protein in subcorneal keratinocytes. Case 11. IHC.
Immunohistochemistry
Immunohistochemistry was used to assess the expression of the L1 viral capsid protein. Samples showing a colorimetric reaction in the nucleus and/or cytoplasm in the cells of the subcorneal and corneal layers were considered positive. Overall, a positive immunolabeling was found in 14 of 16 cases (87%) (Table 1). The immunoreaction was mainly detected in the subcorneal layer and was nuclear; in 2 cases, cytoplasmic reaction was observed in the corneal layer (Fig. 3d).
Results of immunohistochemical and molecular analyses on 16 cases of canine pigmented viral plaques.
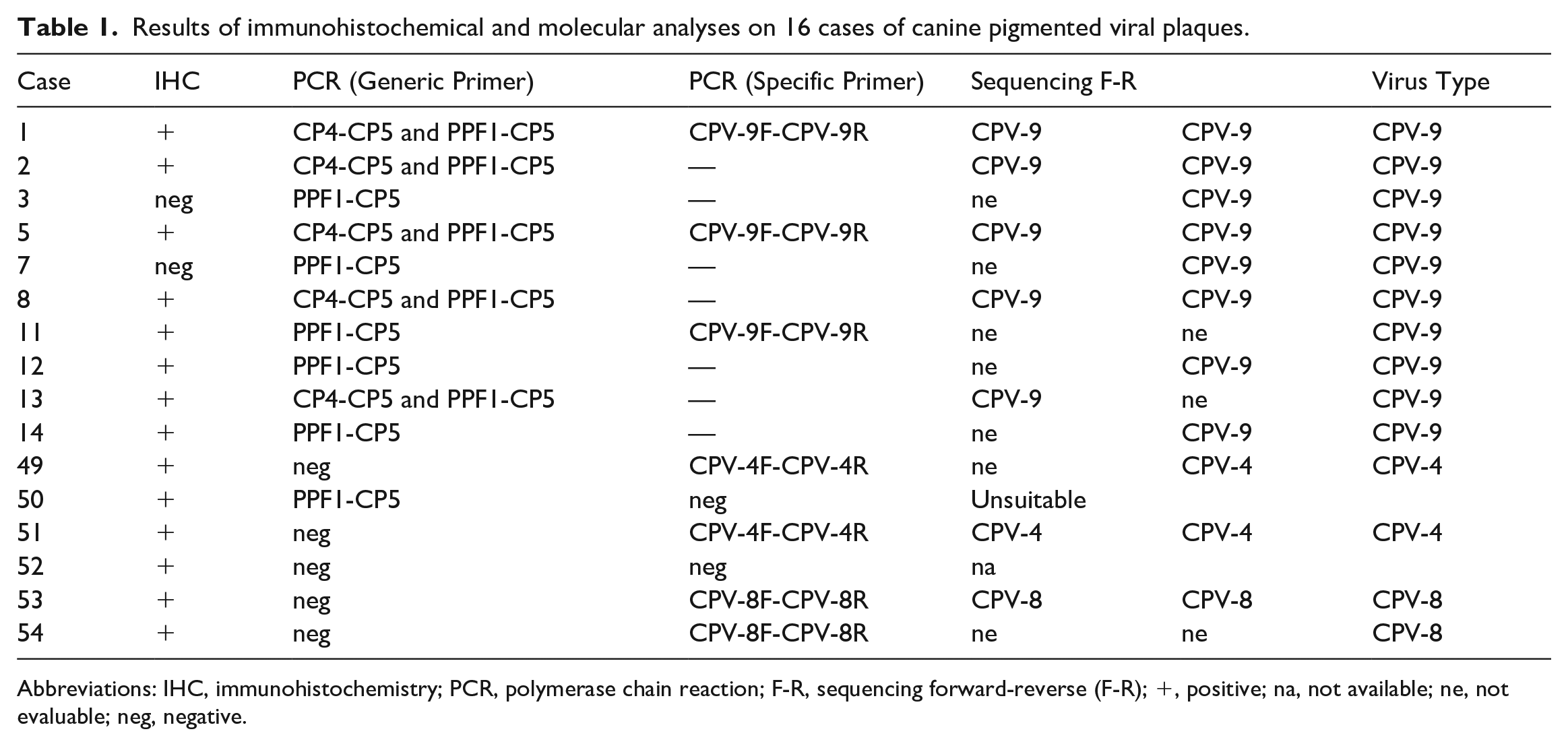
Abbreviations: IHC, immunohistochemistry; PCR, polymerase chain reaction; F-R, sequencing forward-reverse (F-R); +, positive; na, not available; ne, not evaluable; neg, negative.
PCR and Sequencing
In total, 15 out of 16 samples were positive by at least 1 set of CPV primers (Table 1). CPV DNA was only detected with generic primers in 8 samples, 4 samples were positive only with CPV-specific primers, and CPV DNA was amplified with both generic and specific primers in 3 samples.
Ten cases contained amplifiable CPV-9 DNA, 2 cases contained CPV-4 DNA, and 2 cases contained CPV-8 DNA, confirmed by sequence analysis. In 2 cases, no CPV DNA was amplified. Of the samples containing CPV-9 DNA, DNA was amplified using generic and/or specific primers, whereas CPV-4 and CPV-8 DNA was amplified using only the specific set of primers.
Discussion
The present study describes a series of 55 cases of canine PVPs.
Many breeds developed viral plaques; however, pugs represent over a quarter of the total population, followed by pinschers (9%), setters (7%), and Dachshunds (7%). Out of a total of 20,626 dogs submitted to this diagnostic laboratory over a period of about 15 years (2008–2022), 3% were miniature pinscher and 0.6% were pugs; the latter being almost 6 times less common than miniature pinscher in our laboratory database. These data may further support the predisposition of pugs to develop PVPs. The age of the dogs was variable, and patients less than 2 years of age were of different breeds.
The clinical presentation of multiple lesions, either grouped or generalized, was more common than the presentation of single lesions in the population studied. Almost one fifth of dogs with multiple lesions had PVPs diffuse all over the body, representing a major challenge to the therapeutic approach, as discussed below. All body sites can be affected by PVPs; however, the abdomen and the axillae represent a typical clinical presentation and were present in almost 62% of the cases and included different appearances: single, grouped, or generalized.
Neoplasia in contiguity with PVPs was observed (5 dogs, 9%) and represents the major concern in assessing prognosis. It is noteworthy that 3 of the hound dogs were included in the study, 2 English setters and 1 pointer had neoplastic diseases. Two of them also had concomitant demodicosis; although it was diagnosed histologically and not clinically. One of these dogs (case 27) had previously suffered from recurrent demodicosis in the peri-partum period, thus sustaining the suspicion of a concomitant immunodeficient state. The same was stated by Cavana et al for a case of generalized verrucosis due to CPV-9 and concomitant demodicosis. The authors theorized an inherited Demodex-specific T-cell defect, considering that lesion regression in PV infection is a T-cell-mediated event. 4 Immune suppression was suggested to be responsible for the malignant transformation in a fox terrier dog receiving chronic treatment with corticosteroids and cyclosporine, but immune compromise, although suspected, was not found in many other cases reported in the literature.3,9,24
To date, there is no consensus or standardized therapeutic protocols for the treatment of PVPs. Surgery has been reported to be curative and is recommended as the main choice for single and grouped lesions when the site allows an en bloc excision.5,19,24 As an alternative to conventional surgery, laser therapy has been successfully used to treat grouped and extensive lesions; however, new lesions can develop elsewhere on the body.10,20,29 Unfortunately, only one of the cases with a single lesion herein documented was treated with the CO2 laser. According to Knight et al, 10 the advantage of the laser was that the method only vaporizes tissue within the epidermal basal membrane avoiding scarring and promoting rapid healing, allowing it to be used on large areas.
Regarding medical therapy, various drugs have been documented in the literature, although none of them have proven to be effective, except for the use of topical glycate gel. None of the cases were treated with tiginalol glycate, which is currently only commercially available as an intratumoral treatment for canine mast cell tumors.6,9 Azithromycin was proposed as medical treatment for canine papillomatosis and was later used to treat PVPs in dogs without any effect on the size or number of lesions. Similarly, azithromycin was found to be uneffective in a single case in this series and it is reasonable that it could be abandoned, although a proper efficacy study in PVPs is lacking.9,20,35 Imiquimod was uneffective when used by both Hansen et al 9 and Munday et al 20 and lacked a therapeutic benefit in a dog in this case series. All other attempts to use drugs such as retinoids, interferon, and firocoxib, in addition to other drugs, were unsuccessful or with inconsistent results.9,20,23,29 To date, it appears that no medical therapy is able to cure multiple to generalized viral plaques; laser therapy is the only one that can benefit dogs without the side effects of a more aggressive conventional surgery. 10
Although considered rare, spontaneous regression was observed in 3 cases. 22 Lymphocytic inflammation in the underlying dermis was absent.
Among the histopathological features indicative of PVPs, the authors found the low magnification profile of the lesion, especially the scalloped configuration that is responsible for the formation of shallow cups to be very useful in the diagnosis of PVP. These features together with hypergranulosis were present in all cases and were often associated with another useful parameter, the presence of surface spikes often at the border of the shallow cups, except in a few cases (36; 65%). Hypergranulosis and the presence of large clumped keratohyalin granules were reported by Bradley et al 2 as important features to differentiated PVPs from seborrheic keratosis.
Hyperplasia and orthokeratotic hyperkeratosis were also present in all of the cases, but are also seen in other non-viral inflammatory skin conditions and are therefore not considered specific in themselves. Koilocytes represent a cytopathic effect of PV on keratinocytes in other papillomavirus-induced diseases in dogs and cats, but they are rarely reported as a key feature of canine PVPs. Similarly, in this study, they were seen in only 19 cases (34%). None of the cases showed viral nuclear inclusion bodies, and inclusions are also rarely reported in the literature. 5
Immunohistochemistry was able to detect the presence of viral L1 capsid in 87% of the cases tested, which is consistent with the literature, although in some case reports no immunolabeling was detected.5,10,18 In our study, we cannot exclude that the lack of positivity in 2 of the cases could be due to prolonged formalin fixation of the specimen. The use of different commercially available antibodies may explain differences between studies. Immunohistochemistry could be a useful tool to confirm papillomavirus infection, although clinical and histopathological features are quite straightforward for the diagnosis.
Analogous to a recent paper on inverted papillomas, specific primers for each CPV already known to be involved in canine PVPs were designed in this study and tested for the first time on these lesions. 26 In our study, CPV4 was not amplified by the MY09/11 primers, which is different from what has been reported previously. 18 MY09 and MY11 are degenerate primers used to amplify a wide range of PVs, whereas in our study CPV4 samples were positive only when using primers designed for the specific CPV-4 sequence, and the specificity of the result was also confirmed by sequence analysis. We suggest that a low viral load in the lesion could be the cause of the lack of sensitivity when using degenerate primers.
The discordance between the immunohistochemistry and PCR results in 2 samples (cases 50 and 52) could be due to the fact that the samples subjected to the different analyses included in this study were obtained from serially sectioned tissue blocks, so that the sections used for immunohistochemistry were different from those used for PCR. An alternate explanation could be a strain of CPV was not detectable with the tested primers, either due to a novel strain or because the primers were inefficient due to the few GenBank sequences available for each CPV studied (https://www.ncbi.nlm.nih.gov/nuccore). Unfortunately, we could not perform a positive control on the specific PCR panel and we cannot guarantee the sensitivity of our molecular assays, but we can assure the specificity of the positive result by the subsequent sequence analysis performed on the positive samples.
To the best of our knowledge, this is the first evidence of the presence of CPV-4, 8, and 9 associated with PVPs in Italy. Although follow-up data were only available in half of the cases for which molecular analyses were performed, according to the literature, none of the CPVs tested were associated with neoplastic transformation. Unfortunately, in the 6 cases with neoplasia, no molecular tests were performed to confirm which PV type was involved in the pathogenesis of this transformation.5,18,31 The literature reports that CPV-4 is unlikely to be associated with a worse prognosis as compared to the other CPVs, such as CPV-3, 12, 16, and 18, which are considered to be responsible for neoplastic transformation.1,17,15,25,30,37 In the only 2 cases reported as CPV-4 positive in our study, neoplasia was not reported at follow-up.
In conclusion, 4 clinical presentations were found in dogs with PVPs: single, multifocal in one or different anatomical sites, or generalized. The 2 cases with CPV-4 in a pug and a mixed breed showed a generalized presentation of the lesions, the same has been reported by other authors in pugs as well as other breeds.5,9,10 Histopathological examination and immunohistochemistry should be performed to confirm the clinical diagnosis and to document the viral origin. Molecular analysis is necessary to identify the type of CPV involved. Despite the large number of cases, medical therapy still remains to be investigated; surgery is the first choice for single or grouped lesions, and laser therapy seems to be promising. For the first time, CPV-4, 8, and 9 are reported to be responsible for canine PVPs in Italy.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231195762 – Supplemental material for Clinical, histopathological, and molecular characterization of canine pigmented viral plaques
Supplemental material, sj-pdf-1-vet-10.1177_03009858231195762 for Clinical, histopathological, and molecular characterization of canine pigmented viral plaques by Margherita Orlandi, Maurizio Mazzei, Francesco Albanese, Luca Pazzini, Martina Mei, Giulia Lazzarini, Mario Forzan, Maria Massaro, Marta Vascellari and Francesca Abramo in Veterinary Pathology
Footnotes
Acknowledgements
We are grateful to clinicians who provided history, biopsies, and clinical follow-up of the cases included in the study and to the pathologists who contributed with their cases: Anzolin E, Benali S, Bonaga F, Banco B, Brambilla P, Favaretto E, Franzoni P, Galvani A, Iussich S, Leone F, Minoli L, Necci F, Passalacqua R, Pennisi MG, Pota A, Previti A, Rostagni M, Tortorella G, Vieri B, Zanella D, and Zanna G and to the following Veterinary Clinics: “Anicura Clinica dell’Orologio,” “Animal Eco, “Azzano Decimo,” “Bagno a Ripoli,” “Baroni Buldrini,” “Bovianum,” “Camposampiero,” “Città di Catania,” “Corte Grande,” “CVT Quattro srl,” “Empolese,” “Europa srl,” “Feridoun Didomizio,” “Fiesole,” “Fuccelli e Palazzi,” “Julia,” “La Sala,” “Magenta,” “Monferrato,” “Novara,” ”Prato Nord,” “Pronto Soccorso Laudense,” “Roncalli,” “Sambuco,” “San Martino srl,” “San Minianimal,” “Sant’Anna,” “San Vitale,” “Sasive,” “7 Star srl,” “Sinigaglia,” “Stanga Montoleo,” “Val di Sambro,” “Villoresi,” and “Zani Amboini.”
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
