Abstract
Leptomeningeal gliomatosis (LG) is characterized by extensive dissemination of neoplastic glial cells in the subarachnoid space either without an intraparenchymal glioma (primary LG or PLG) or secondary to an intraparenchymal glioma (secondary LG or SLG). Given the low frequency of LG in human and veterinary medicine, specific diagnostic criteria are lacking. Here, we describe 14 cases of canine LG that were retrospectively identified from 6 academic institutions. The mean age of affected dogs was 7.3 years and over 90% of patients were brachycephalic. Clinical signs were variable and progressive. Relevant magnetic resonance image findings in 7/14 dogs included meningeal enhancement of affected areas and/or intraparenchymal masses. All affected dogs were euthanized because of the poor prognosis. Gross changes were reported in 12/14 cases and consisted mainly of gelatinous leptomeningeal thickening in the brain (6/12 cases) or spinal cord (2/12 cases) and 1 or multiple, gelatinous, gray to red intraparenchymal masses in the brain (6/12 cases). Histologically, all leptomeningeal neoplasms and intraparenchymal gliomas were morphologically consistent with oligodendrogliomas. Widespread nuclear immunolabeling for OLIG2 was observed in all neoplasms. The absence of an intraparenchymal glioma was consistent with PLG in 3 cases. The remaining 11 cases were diagnosed as SLG.
Glioma is one of the most frequently diagnosed primary central nervous system (CNS) neoplasms of dogs.7,11 Oligodendrogliomas and astrocytomas, the 2 most common types of canine glioma, are typically solitary, well demarcated, and affect the rostral portions of the telencephalon, with fewer cases in the brainstem, cerebellum, and spinal cord.11,21 Rare subtypes of glioma with widespread infiltration through the neuroparenchyma and leptomeninges have also been described, including diffusely infiltrating gliomas (historically referred to as gliomatosis cerebri) and leptomeningeal gliomatosis (LG), respectively.5,6,12,14,22 LG has been described in human and veterinary medicine,12,29 and is characterized by dissemination of neoplastic glial cells within the subarachnoid space either without an intraparenchymal glioma (primary LG or PLG) or secondary to an intraparenchymal glioma (secondary LG or SLG).9,29
PLG is rare in human medicine, with fewer than 100 documented cases since its first description in 1923.25,26,29 The cell of origin of PLG remains unknown, but neoplasms are thought to arise from heterotopic glial cell nests found in the subarachnoid space. 26 The age of affected patients varies (median age = 35 years), with no apparent gender predilection. 29 Clinical signs are insidious and almost invariably lead to death a few months after their onset. 9 A clinical diagnosis is difficult due to the often subtle meningeal lesions, and although PLG can be suspected by magnetic resonance imaging (MRI), surgical biopsy is often necessary for diagnostic confirmation. 9 Although most cases consist of extensive neoplastic infiltration along the surface of the brain and/or spinal cord (hence, the occasional use of the term “diffuse leptomeningeal gliomatosis” to refer to PLG),13,26 solitary meningeal lesions are also reported.10,17 Early PLG is characterized by the involvement of supratentorial locations, with a predilection for the skull base. 29 However, lesions can progress and involve the spinal cord.4,26
SLG is reported in 1% to nearly 30% of human patients with an intraparenchymal glioma.26,30 Leptomeningeal involvement occurs following the dissemination of neoplastic glial cells from the main tumor via cerebrospinal fluid (drop metastasis).1,19,30 The median age of affected patients is 45 years and leptomeningeal spread is most commonly associated with high-grade gliomas.2,18
Given the low frequency of PLG and SLG in humans, specific diagnostic criteria and therapeutic protocols are still lacking, and the condition is not listed in the 2021 World Health Organization classification of tumors of the CNS.2,9,28 In fact, subsets of LG have been reclassified as a morphologically similar but distinct entity referred to as diffuse leptomeningeal glioneuronal tumor (DLGT), a rare neuronal and mixed neuronal-glial neoplasm that affects mainly children.24,28 Although a definitive diagnosis of DLGT is difficult, its characteristic molecular profile (KIAA1549-BRAF fusion combined with isolated 1p deletion or 1p/19q codeletion) supports its differentiation from LG. 28
In veterinary medicine, PLG is equally rare and has been described mainly in dogs,5,6,12,14 with additional single case reports in a cat and a cow.8,31 In addition, leptomeningeal spread is an uncommon but well known neoplastic cell infiltration pattern of canine glioma, 11 occurring locally as a component of the secondary structures of glioma (together with perineuronal satellitosis, perivascular satellitosis, and tumor invasion along white matter tracts) 11 or via drop metastasis to distant sites. 3 However, SLG has been rarely described as a distinct entity.12,16 Since much of our understanding of canine glioma has been heavily based on classification and grading schemes designed for human gliomas, it is likely that the lack of diagnostic and classification criteria of these neoplasms in human neuropathology has impaired our diagnostic approach to LG in veterinary medicine.
Here, we describe the neuropathology and diagnostic immunohistochemistry (IHC) of 14 cases of canine PLG and SLG diagnosed at 6 academic institutions from 1995 to 2022. In addition, we propose tentative guidelines to diagnose and differentiate LG from local secondary leptomeningeal spread of an intraparenchymal glioma.
Material and Methods
The electronic web-based archives of the Athens Veterinary Diagnostic Laboratory, Department of Pathology, University of Georgia (Athens, GA); Department of Anatomic Pathology, Schwarzman Animal Medical Center (New York, NY); Department of Biomedical Sciences, Section of Anatomic Pathology, College of Veterinary Medicine, Cornell University (Ithaca, NY); Department of Pathobiology, School of Veterinary Medicine, University of Pennsylvania (Philadelphia, PA); and the Institute of Pathology, Department of Pathobiology, University of Veterinary Medicine, (Vienna, Austria) were searched for cases of LG diagnosed between 2010 and 2022 using the key words “dog,” “meningeal gliomatosis,” and “leptomeningeal gliomatosis.” The same search was conducted in the archives of the Department of Pathology, Microbiology and Immunology, University of California, Davis (Davis, CA) for cases diagnosed between 1995 and 2022.
Clinical forms, autopsy reports, and archived hematoxylin and eosin tissue sections from retrieved cases were reviewed and cases were included in the study if an extensive leptomeningeal glioma with or without an intraparenchymal glioma in the brain and/or spinal cord was reported. In the brain, “extensive leptomeningeal glioma” was defined as leptomeningeal involvement of 2 or more of the following locations: (1) telencephalon, (2) brainstem, and (3) cerebellum. In the spinal cord, cases were included if leptomeningeal gliomas involved 1 or more of the following segments: (1) cervical, (2) thoracic, (3) lumbar, or (4) sacral. Selected cases were further reviewed and classified as either PLG (extensive leptomeningeal glioma without intraparenchymal glioma) or SLG (extensive leptomeningeal glioma with 1 or multiple intraparenchymal gliomas). A tumor grade was assigned for the leptomeningeal glioma and intraparenchymal glioma (when present), according to the revised classification system for canine glioma. 11 Briefly, gliomas (oligodendrogliomas, astrocytomas, or undefined gliomas) were classified as low-grade (low or moderate cellularity, low anisocytosis and anisokaryosis, absent or low mitotic activity, and no geographic necrosis or microvascular proliferation) or high-grade (moderate or high cellularity, prominent anisocytosis and anisokaryosis, high mitotic activity, geographic necrosis with or without palisading of neoplastic cells, and/or microvascular proliferation). The mitotic count was assessed by counting the number of mitoses in 2.37 mm2 (10 consecutive FN22/400X fields). 15
Archived paraffin-embedded tissue sections were immunolabeled for OLIG2 (rabbit monoclonal, 1:400 dilution at 60 minutes, GeneTex, Irvine, California, GTX62440) and GFAP (glial fibrillary acidic protein) (mouse monoclonal, 1:4000 dilution at 60 minutes, Biogen, Cambridge, MA, MU020-UC) for diagnostic confirmation based on previously published protocols. 11 In brief, an automated stainer (Nemesis 3600, BioCare Medical, Concord, CA) was utilized. Antigen retrieval was achieved using Antigen Retrieval Citra Solution 10X (BioGenex) at a dilution of 1:10 for 15 minutes at 110°C. Biotinylated rabbit (OLIG2) and mouse (GFAP) secondary antibodies (Vector Laboratories, Burlingame, CA) were used to detect the immunolabeling, and the immunoreaction was visualized using 3,3-diaminobenzidine (DAB) (BioCare Medical, Concord, CA) substrate for 12 minutes, and slides were counterstained with hematoxylin. Positive and negative control tissues consisted of normal canine brain.
Results
We selected 14 cases of LG for inclusion in our study (Supplemental Tables S1 and S2). The age of affected dogs varied from 2 to 12 years (mean age = 7.3 years). Affected breeds included French bulldog (5/14 cases), boxer (3/14 cases), English bulldog (2/14 cases), American bulldog, Boston terrier, Cavalier King Charles spaniel, and mixed breed (1/14 case each). Neurologic signs varied according to the neurolocalization of the lesions. MRI findings (7/14 cases) were limited to the information provided in the clinical submission forms and consisted of meningeal enhancement of affected areas (5/7 cases) and hyperintense intraparenchymal masses (4/7 cases). Euthanasia was elected in all cases because of the poor prognosis associated with the progressive clinical signs and/or CNS lesions.
Gross neuropathologic changes were reported in 12/14 cases. Leptomeningeal changes were reported in 8/12 cases and consisted of gelatinous leptomeningeal thickening in the brain (6/12 cases) or spinal cord (2/12 cases) (Fig. 1a–c). One or multiple, gelatinous, gray to red intraparenchymal masses were grossly reported in the brain in 6/12 cases (Fig. 1d).
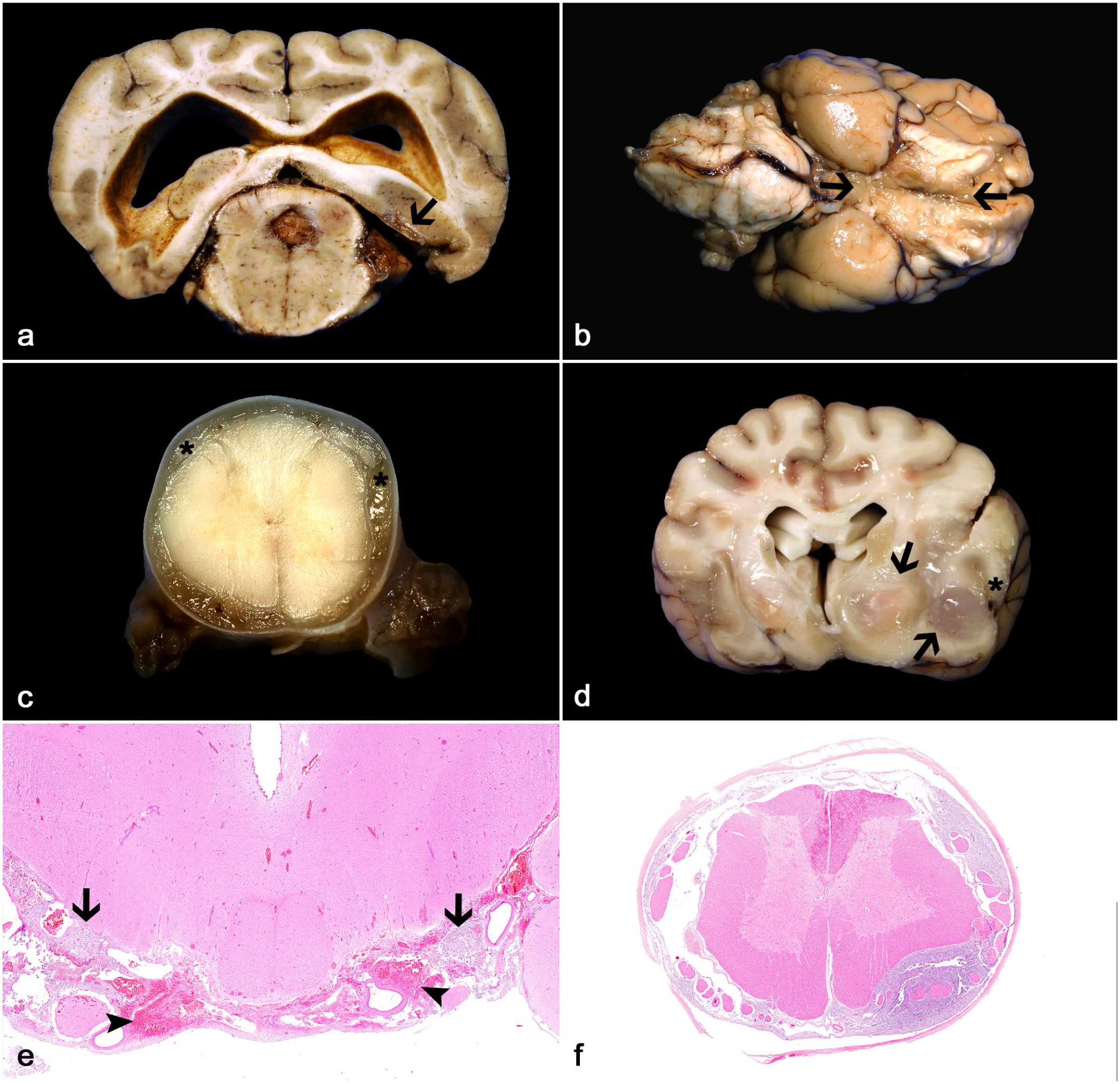
Leptomeningeal gliomatosis (LG) in dogs. (
Histologically, all leptomeningeal (cases 1–14) and intraparenchymal neoplasms (cases 4–14) were morphologically and immunohistochemically consistent with oligodendrogliomas. Regardless of the diagnosis (PLG or SLG), leptomeningeal neoplasms were characterized by subarachnoid expansion and effacement by sheets of neoplastic glial cells supported by a fine network of branching capillaries and a faintly basophilic mucinous stroma (Fig. 1e, f). Low-grade leptomeningeal neoplasms (6/14 cases) had low to moderate cellularity and were characterized by neoplastic cells with round and scant, often retracted cytoplasm (fried-egg or honeycomb appearance) and round to oval nuclei with dense to coarse chromatin with one occasionally evident nucleolus (Fig. 2a, b). No mitoses were observed. High-grade leptomeningeal neoplasms (8/14 cases) had moderate to increased cellularity and were composed of neoplastic cells with round, eosinophilic or retracted cytoplasm and round to oval nuclei with moderate pleomorphism (Fig. 2c, d). Mitotic activity was observed in 5/8 high-grade leptomeningeal gliomas and ranged from 3 to 7 mitoses in 2.37 mm2. High-grade neoplasms also had occasional areas of microvascular proliferation (5/8 cases) or geographic necrosis (1/8 cases) (Fig. 2e, f).
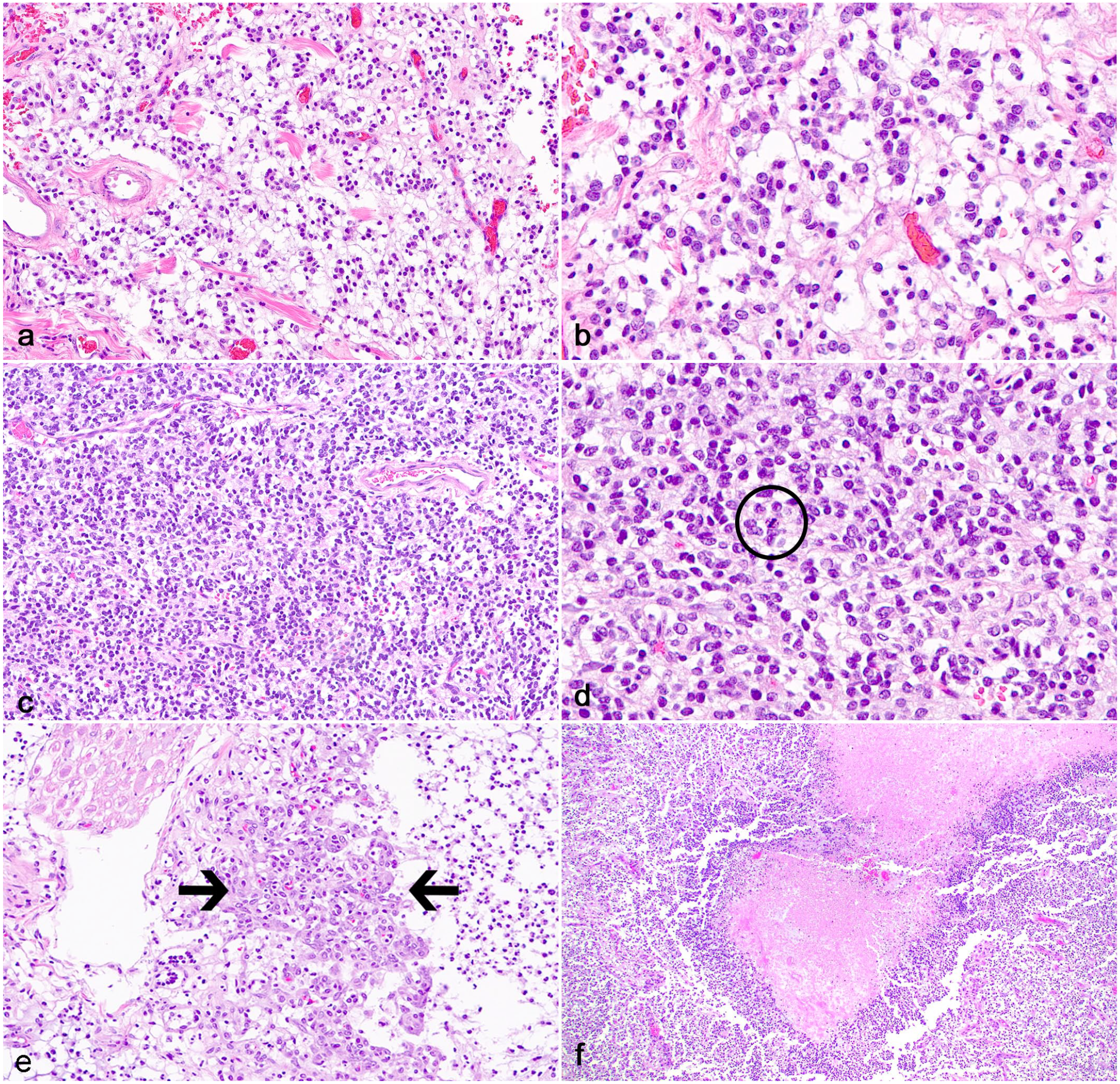
Leptomeningeal gliomatosis (LG) in dogs. (
Additional findings associated with the leptomeningeal neoplasms (regardless of tumor grade) included subdural telencephalic and spinal nerve entrapment by neoplastic cells (6/14 cases), neoplastic cell infiltration along the pia mater surrounding superficial cortical perivascular spaces and adjacent telencephalic gray or white matter (2/14 cases) (Fig. 3a, b), and neoplastic cell infiltration within ventricles and/or periventricular neuroparenchyma (2/14 cases) (Fig. 3c, d). All neoplasms had widespread, moderate to robust nuclear immunolabeling for OLIG2 (Fig. 3e, f) and rare (less than 5% of neoplastic cells) cytoplasmic immunolabeling for GFAP in 1 case (case 14).
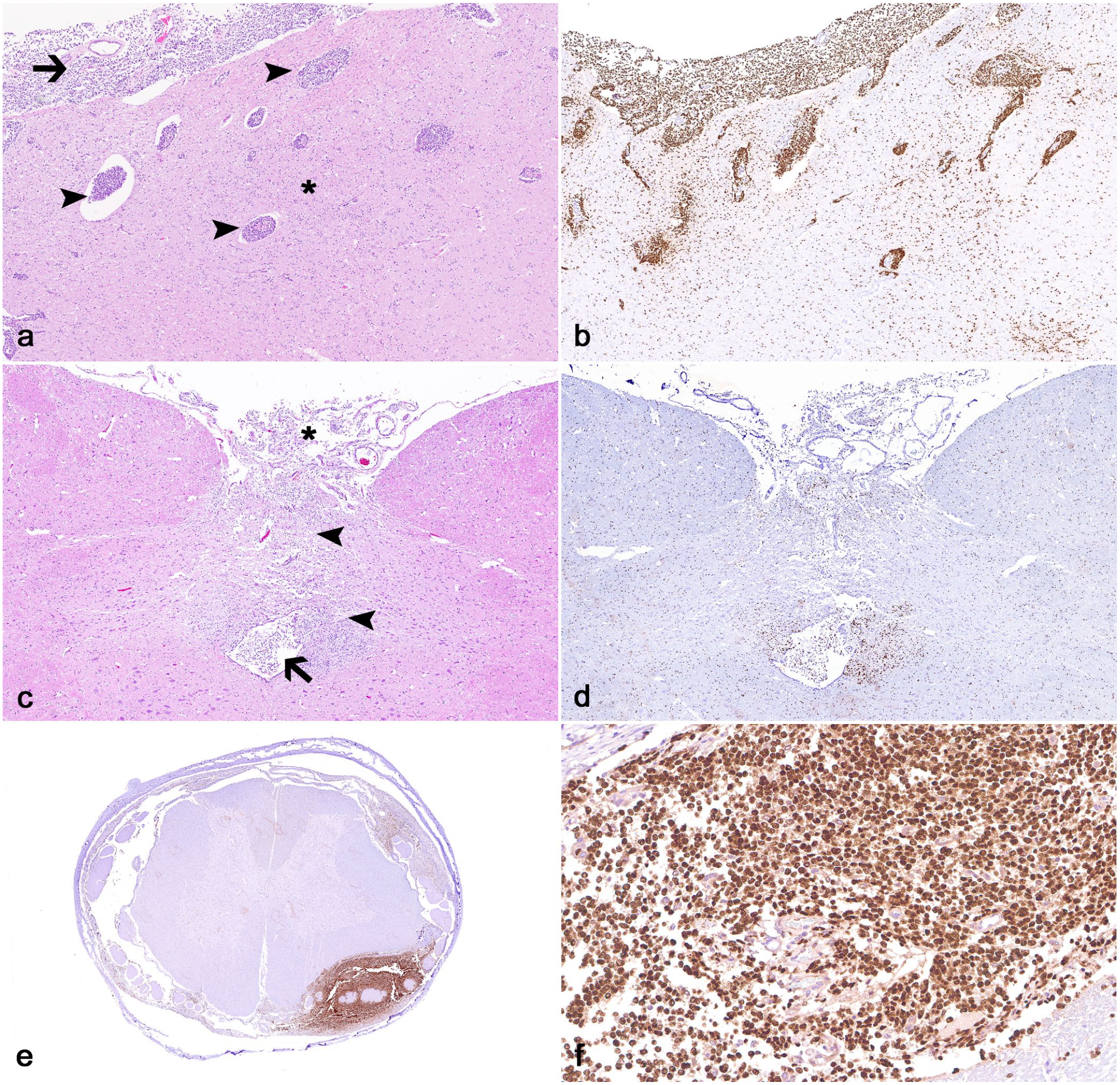
Leptomeningeal gliomatosis (LG) in dogs. (
Intraparenchymal gliomas were not reported grossly or histologically in 3/14 cases (cases 1–3) after examination of multiple sections of brain and spinal cord, allowing for the diagnosis of PLG. In 11/14 cases, 1 or more intraparenchymal oligodendrogliomas (10 high-grade tumors and 1 low-grade tumor) were reported concomitantly with the leptomeningeal lesions, consistent with SLG. All high-grade intraparenchymal oligodendrogliomas were densely cellular and had increased cellular and nuclear pleomorphism, with geographic necrosis (4/10 cases) and/or microvascular proliferation (4/10 cases). The mitotic count varied from 2 to 16 in 2.37 mm2. The low-grade intraparenchymal oligodendroglioma was sparsely to moderately cellular, with low cellular pleomorphism and no observable mitosis.
Discussion
Here, we describe 14 cases of LG in dogs that were classified as PLG (3 cases) and SLG (11 cases). The age of affected dogs and the high frequency of brachycephalic breeds were similar to that described for gliomas. 11 Furthermore, leptomeningeal lesions were morphologically and immunohistochemically similar to intraparenchymal gliomas, regardless of the final diagnosis (PLG or SLG). A consensus on the diagnostic criteria of LG (especially SLG) is lacking in veterinary medicine. 2 Here, we propose objective guidelines for the diagnosis of PLG and SLG and provide information that allows for the differentiation between gliomas with local leptomeningeal spread and SLG.
The diagnostic confirmation of PLG depends on the presence of neoplastic glial cells that is restricted to the leptomeningeal subarachnoid space with no evidence of intraparenchymal involvement,5,8,12,27,31 as reported for cases 1–3 in our study. Case 3 had neoplastic cell infiltration around superficial cortical blood vessels and evidence of neoplastic cell invasion into the surrounding gray matter, which was highlighted by OLIG2 IHC. However, superficial perivascular invasion in PLG is not considered true parenchymal invasion but an extension of subarachnoid neoplastic cells around the underlying perivascular pia mater.5,27,31 Furthermore, minimal neuroparenchymal involvement with no distinct mass formation has been reported late in the course of the disease in humans, still allowing for a diagnosis of PLG. 27 The vicinities of areas with neuroparenchymal infiltration should be assessed in detail to rule out an adjacent intraparenchymal glioma with focal leptomeningeal spread or SLG in such cases.
SLG is characterized by extensive leptomeningeal involvement with a concurrent intraparenchymal glioma, as highlighted by cases 4–14 in our study. 2 SLG is thought to result mainly from dissemination of neoplastic glial cells from a glioma into the cerebrospinal fluid and subsequent leptomeningeal spread (drop metastasis),1,3,19,30 as evidenced by the presence of neoplastic cells within the cervical central canal and mesencephalic aqueduct in 2 of our cases.
Although SLG has been rarely described as a distinct entity in dogs,12,16 local leptomeningeal spread from an intraparenchymal glioma has been described as part of the secondary structures of glioma, along with perivascular and neuronal neoplastic satellitosis. 11 However, it is critical to note that secondary structures occur as a local spread of the intraparenchymal glioma and are not characterized by the disseminated infiltration pattern observed in SLG. No objective diagnostic criteria to differentiate these 2 leptomeningeal infiltration patterns of canine glioma have been proposed to date. In this study, SLG was characterized as leptomeningeal spread from an intraparenchymal glioma involving 2 or more of the major anatomic areas of the brain (telencephalon, brainstem, and cerebellum) and/or 1 or more segments of the spinal cord (cervical, thoracic, lumbar, and sacral). These criteria were based on the diagnostic definitions utilized to classify diffusely infiltrating gliomas (often referred to as gliomatosis cerebri), which reflect the widespread infiltration of neoplastic cells typical of those neoplasms and not local infiltration of neoplastic glial cells. 22
Similar to what has been reported in cases of canine PLG or SLG, brachycephalic dogs were affected in over 90% of our cases, reinforcing the trend widely reported for canine glioma. 11 The neurologic signs associated with PLG or SLG in our cases were progressive and invariably led to euthanasia of affected patients. MRI findings were reported in 7 cases (2 PLG and 5 SLG), but information was limited to what was provided in the clinical forms. Leptomeningeal lesions were characterized by meningeal enhancement on MRI, as reported elsewhere. 5 The symptoms of PLG in humans are typically nonspecific and associated with increased intracranial pressure and hydrocephalus caused by the obstruction of cerebrospinal fluid drainage in the subarachnoid space. 27 Therefore, a clinical diagnosis PLG is difficult in human and veterinary medicine, as clinical signs are nonspecific and meningeal enhancement can reflect not only a primary or metastatic meningeal neoplasm but also other types of lesions, such as inflammation, hemorrhage, and edema.5,6 Intraparenchymal gliomas, when present, can be more easily detected by MRI, as reported in a subset of SLG cases in our study and other investigations.5,6,12,14 A concurrent intraparenchymal mass was identified in 4 of the 5 SLG cases that had an MRI performed.
Gross changes in our cases were similar to those described in human and canine cases of PLG and SLG.5,6,12 –14,16 Hydrocephalus and other secondary changes associated with the leptomeningeal or intraparenchymal gliomas were reported in a subset of our cases, consistent with what has been reported by other authors.5,14 In humans, PLG is thought to arise from heterotopic subarachnoid glial nests that occur mostly in the medulla oblongata, a hypothesis that is partially supported by the predilection of PLG for the leptomeninges of the skull base. 27 However, lesions also occur at sites other than the skull base. 23 Although a ventral brain lesion was reported in all of our PLG cases and also by other authors,5,12,14,16 not enough cases have been described to determine whether a true neuroanatomic predisposition exists for canine PLG. In humans, PLG can be solitary (nodular) or diffuse. While solitary lesions are characterized as focal thickening or mass in the leptomeninges,10,17 diffuse neoplasms affect a wide area of the CNS.13,26 In most of the reported cases of canine PLG and in the all 3 PLG cases in our study, lesions affected great extensions of the brainstem and/or spinal cord, consistent with diffuse PLG.5,12,14
Histologically, all leptomeningeal lesions in our study were morphologically and immunohistochemically consistent with oligodendrogliomas. IHC for OLIG2 and GFAP suffices for diagnostic confirmation in cases in which a glioma is already suspected based on routine examination. However, further IHC may be needed to rule out other leptomeningeal neoplasms, such as meningioma, lymphoma, metastatic choroid plexus carcinomas, or other metastatic carcinomas, in less typical cases.12,20 The current canine glioma classification guidelines were standardized for intraparenchymal tumors. However, given their overall similarities, the grading system likely applies to leptomeningeal neoplasms. 11 In our study, 2/3 PLG cases had morphologic features of a low-grade oligodendroglioma. For the SLG, 4/11 cases had morphologic features of a low-grade oligodendroglioma even when the concurrent intraparenchymal glioma was a high-grade oligodendroglioma. These findings indicate that morphologic tumor variations can occur between the intraparenchymal glioma and LG. However, it remains to be determined whether the infiltrative nature and different grades of these leptomeningeal lesions may result in major differences in biological behavior and clinical outcome.
Our findings confirm that LG is rare in dogs, particularly those cases in which a primary intraparenchymal glioma is not present (PLG). Primary or secondary leptomeningeal involvement was observed in adult dogs with a high frequency of brachycephalic breeds, similar to what has been extensively reported for canine glioma. 11 Diagnostic confirmation was achieved based on histology and IHC for OLIG2 and GFAP, regardless of the final diagnosis (PLG or SLG).
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231193104 – Supplemental material for Primary and secondary leptomeningeal gliomatosis in dogs
Supplemental material, sj-pdf-1-vet-10.1177_03009858231193104 for Primary and secondary leptomeningeal gliomatosis in dogs by Daniel R. Rissi, Vicente A. A. Reyes, Taryn A. Donovan, Molly E. Church, Elizabeth W. Howerth, Andrea Klang, Kevin D. Woolard and Andrew D. Miller in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
