Abstract
Pattern analysis of inflammatory skin diseases is a technique that offers a systematic approach to the histologic diagnosis of skin diseases. First introduced to human dermatopathology in the 1970s, it was widely adopted by veterinary pathologists for the histologic diagnosis of skin diseases in animals. As the inflammatory pattern reflects, to varying extents, aspects of the underlying disease pathogenesis, its use has contributed to the recognition of novel skin diseases in domestic animals, particularly in dogs and cats. Alternative diagnostic approaches used in human dermatopathology, such as “tissue-reaction pattern” and a purely “anatomic approach” have not been as widely used in veterinary pathology. However, veterinary pathologists often combine pattern analysis with anatomic and etiologic factors. This overview outlines the technique, introduces the patterns, and discusses advantages and limitations of pattern analysis in veterinary diagnostic dermatopathology. While molecular analytic techniques and image informatics will undoubtedly prove to be revolutionary in many areas of diagnostic pathology, it is recognized in both human and veterinary arenas that the light microscopic interpretation of hematoxylin and eosin-stained tissue sections will remain the mainstay of routine dermatopathology diagnosis for the foreseeable future.
Keywords
In a routine veterinary diagnostic pathology service, skin biopsies account for between 10% and 40% of tissue biopsies submitted, reflecting the high proportion of dermatology cases in companion animal practice and the relative ease of the skin biopsy procedure. Most samples are from dogs and cats. Many general pathologists find skin biopsies challenging as the discipline of dermatology has become more advanced and its practitioners more specialized. The situation is often compounded by a poor history, inadequate description of the clinical signs and skin lesions, and inappropriate biopsy site selection. In the absence of an obvious cause and with no better technique to characterize the inflammatory lesions, too often a diagnosis of “non-specific dermatitis” is rendered, which is disappointing to both the clinician and the pathologist.
The lack of a systematic approach to the histologic diagnosis of inflammatory skin diseases in humans led Bernard Ackerman to publish his seminal textbook
What is Pattern Analysis?
Pattern analysis is a 2-stage diagnostic technique. The approach is morphological, but the patterns reflect underlying pathomechanisms and sometimes the cause of the diseases. Bernard Ackerman was inspired by the way field ornithologists and amateurs identify birds visually (Fig. 1). Using the same logic tree, whereby an unknown bird is assigned to its major group based on its silhouette, a skin lesion is first assigned into one or more of the 8 major patterns, also based on its silhouette. This is assessed at scanning magnification at 20 to 40× (Fig. 1). The second stage, which is akin to using binoculars to check for finer details that characterize an individual bird species (such as presence of eye rings, shape of and size of beak, coloration, and patterning of feathers), is carried out with increasing magnification (Fig. 1). Here, the major patterns are further subdivided based chiefly on analysis of the inflammatory infiltrates (ie, type and combination of inflammatory cells) and changes in anatomical structures (ie, epidermis, adnexa, dermis, and panniculus/subcutis) (Fig. 1).
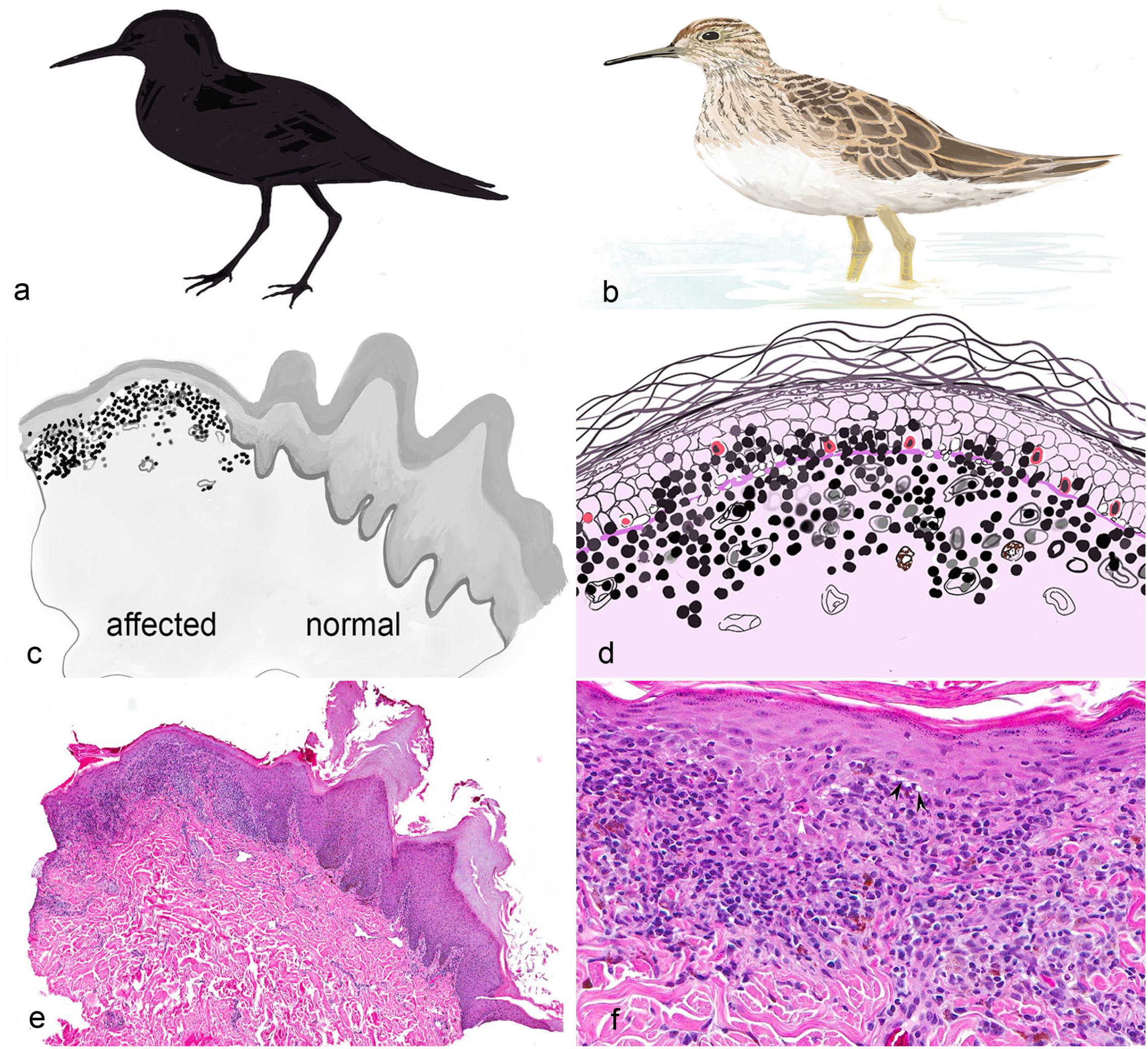
Pattern analysis can be compared to bird watching. Recognizing the silhouette of the bird (a) is the equivalent to identifying the low-magnification histologic pattern (c and e). The hematoxylin and eosin (HE) example (e) is a scanning magnification image of nasal planum lesions in a dog with nasal-predominant chronic cutaneous lupus, commonly known as discoid lupus erythematosus (DLE). The epidermis to the right of center (c and e) is normal bosselated nasal pad skin with papillated epidermis, thick stratum corneum, and heavy pigmentation. The epidermis to the left is atrophic (important feature of DLE) with the characteristic thick lichenoid band of inflammation at the dermo-epidermal junction. Identifying specifics of the birds coloring and so on at close range (b) can be compared to identifying detailed microscopic features at higher magnification (d and f), that allow differentiation of individual diseases. In (f), a higher-magnification view of (e), there are characteristic lesions of DLE including loss of definition of the dermo-epidermal junction, lichenoid band, vacuolar change, apoptosis, and pigmentary incontinence (HE).
The original Ackerman patterns have undergone continued adaptive modifications because his methodology has been applied to veterinary dermatopathology. This technique is an aid to histological diagnosis, not a rigid classification. The 8 major patterns we find most useful are perivascular dermatitis, vasculitis, cytotoxic (interface) dermatitis, intraepidermal vesicular and pustular dermatitis, subepidermal vesicular and pustular dermatitis, folliculitis/furunculosis, nodular dermatitis, and panniculitis. A ninth pattern, originally named endocrinopathy and subsequently atrophic dermatitis, is no longer used. These noninflammatory skin diseases are now categorized as noninflammatory alopecias or degenerative skin diseases. The current edition of a standard veterinary pathology textbook, “Pathologic Basis of Veterinary Disease” uses the term “inflammatory reaction patterns,” 7 of which are essentially the same as the patterns presented in this paper. 32 The exception is the pattern “panniculitis” which is used in the sense of morphologic diagnosis. 32
Advantages of Pattern Analysis
The major advantage of the pattern analysis technique is the ability to rapidly narrow down diagnostic possibilities. Pattern analysis also provides a straightforward template that is useful for pathology and dermatology resident training. Instead of learning by rote the microscopic lesions of hundreds of individual diseases in a non-systematic fashion, even a neophyte can identify the major pattern and thus reduce the remaining options. The pattern analysis technique emphasizes the importance of low-magnification examination prior to proceeding to high-magnification evaluation of samples. Those in teaching positions are well aware of the tendency for novices to move too quickly to higher magnification.
Second, the technique eliminates the supremely unhelpful diagnosis of
Board-certified veterinary dermatologists are instructed in the pattern approach and expect it to be used in their biopsy reports. Even when a specific disease diagnosis cannot be made; knowing that the lesion falls into a particular pattern, allows the clinician to make more informed judgments on how to proceed. The exception to this is the perivascular pattern, which without any additional special features, reflects generic skin inflammation.
Pattern analysis has been a powerful tool in the analysis of the hundreds of new veterinary skin diseases that have been recognized in the past 40 years. The pattern approach is also very useful when pathologists, whether trainees or experts, are faced with a novel lesion. Its application has led to greater understanding of disease pathogenesis by applying knowledge gained from well-characterized diseases, typically human diseases, with similar patterns. As a result, the technique has advanced understanding of new diseases by grouping together conditions that may have a common or related pathogenesis. It is even valid to compare across species, albeit with caution. In the future, pattern analysis may prove useful in the development of algorithms for computer-assisted diagnosis.
Patterns of Inflammation
Perivascular Dermatitis
Perivascular dermatitis (Fig. 2a) is universally present in inflamed skin as all inflammatory processes start with a perivascular infiltrate due to leukocyte migration through the vascular walls. It reflects the stereotypic response to injury and is the least diagnostic of the 8 patterns. Features that assist narrowing down the list of differential diagnoses are (1) types and combinations of inflammatory cells, (2) identifying which vascular plexus is affected; for example, in dogs and cats involvement of all 3 vascular plexii often points toward cutaneous involvement of a systemic process, (3) epidermal changes such as diffuse parakeratosis and epidermal pallor, lesions virtually diagnostic for superficial necrolytic dermatitis in dogs, and (4) dermal changes. When the inflammatory cells are more diffuse but not sufficiently solid to qualify as nodular and/or diffuse patterns, the modifying term “interstitial” is often applied.
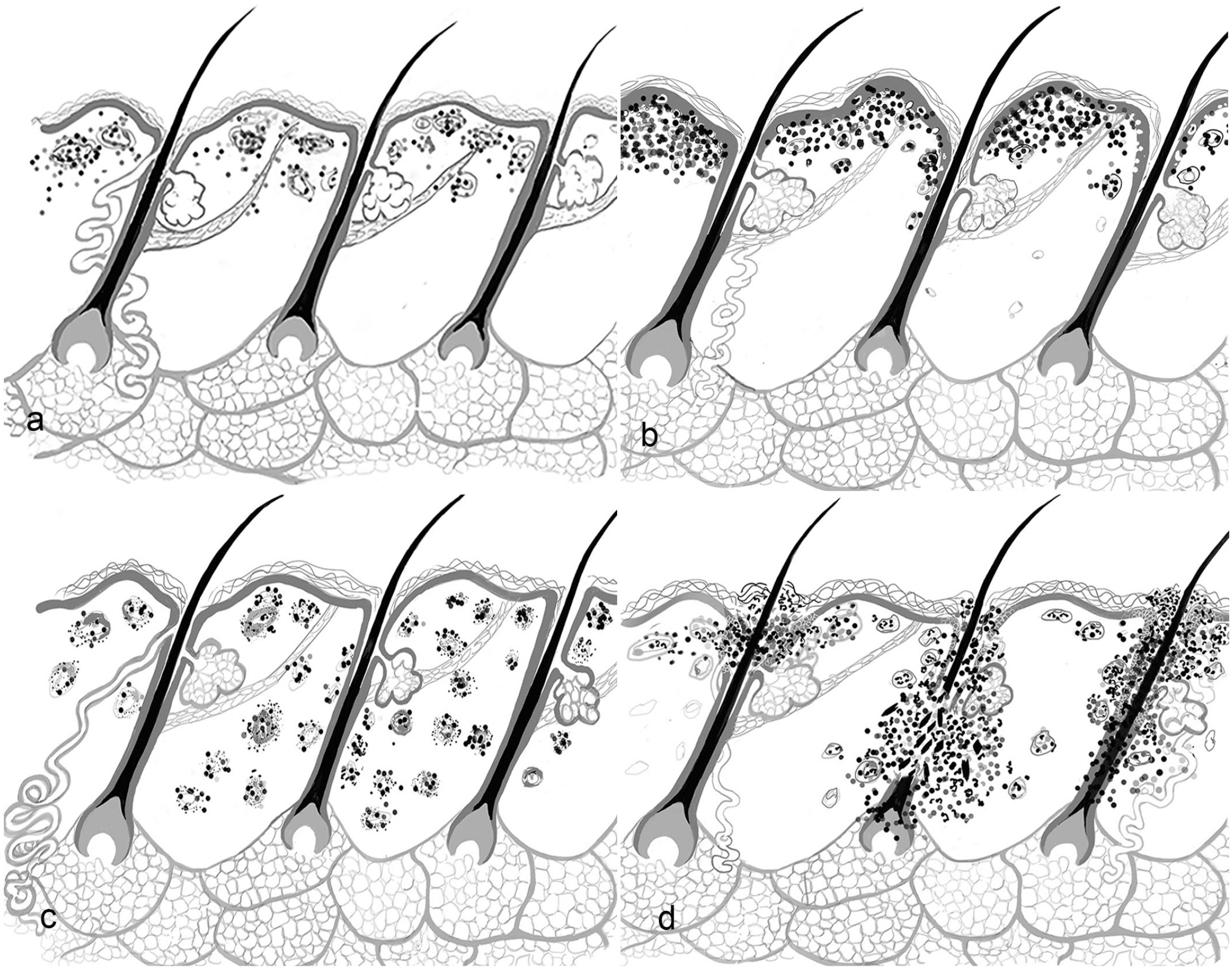
Schematics visualizing the major patterns of inflammatory diseases in domestic animals (drawings by: JA Yager): (a) perivascular dermatitis, (b) cytotoxic dermatitis, (c) vasculitis, and (d) folliculitis and furunculosis. Note for simplicity, the hair follicles are represented as simple type not compound as occur in many domestic animals (eg. dogs and cats).
Cytotoxic Dermatitis
Cytotoxic (interface) dermatitis (Fig. 2b), originally considered a variant of perivascular dermatitis, 1 is identified by an inflammatory reaction predominantly at the dermo-epidermal junction (DEJ) and the lower layers of the epidermis. The mononuclear cell-rich infiltrate (usually lymphocytic) may obscure the DEJ. The key to this pattern, however, is keratinocyte cell death. This is the central tenet of Pinkus’ lichenoid tissue reaction, which was renamed the “interface pattern” by Ackerman.1,32 The pattern is confirmed by the presence of apoptotic keratinocytes, evidence of vacuolar degeneration at and below the DEJ, and satellitosis (lymphocytes adjacent to apopototic keratinocytes). While the original emphasis was on apoptosis in the basal and suprabasal layers (interface cytotoxic dermatitis), in certain conditions such as erythema multiforme, apoptosis is present at all levels (panepidermal cytotoxic dermatitis). In chronic lesions, histologic changes may be subtle with evidence of prior damage only, such as loss of the normal columnar layer of basal keratinocytes (squamatization), basal lamina thickening (uncommon in animal lesions), and pigmentary incontinence (melanin released from damaged keratinocytes or melanocytes phagocytized by macrophages). Many autoimmune skin conditions manifest this pattern including the various forms of cutaneous lupus erythematosus and some cutaneous adverse drug reactions (drug-induced erythema multiforme). 29 Cytotoxic dermatitis is the pattern of virus-associated diseases, such as a rare instance of systemic parvovirus infection in puppies. 11 This often collectively called “erythema multiforme,” to the extent that the pattern is sometimes wrongly equated to the disease. Clinical correlates include depigmentation, erosions, ulcers, atrophy, scaling, and alopecia if follicles are involved.
The presence of a band of mononuclear inflammatory cells at the DEJ without evidence of keratinocyte damage is insufficient evidence of the cytotoxic pattern. Relying solely on a “lichenoid” band may lead to the misdiagnosis of certain inflammatory skin diseases as autoimmune conditions, particularly at mucocutaneous locations.
Cell-poor cytotoxic (interface) dermatitis refers to lesions with minimal or even absent inflammation. In human dermatology, these conditions exhibit certain clinical signs (poikilodermatas) and in veterinary medicine they are associated mostly with ischemic dermatopathies.
The suggestion moving forward is to focus on the keratinocyte damage and its location in the epithelium in this pattern, applying the term interface cytotoxic dermatitis for lesions with basal cell damage and panepidermal cytotoxic dermatitis with keratinocytes cytotoxicity throughout the thickness of the epithelium.
Vasculitis/Vasculopathy
Vasculitis (Fig. 2c) is an uncommon to rare pattern in veterinary skin biopsies. Classifications of vasculitis used in human medicine, where the emphasis is on large and medium-sized vessels, is not particularly useful in the skin, because all cutaneous vessels are, by definition, small vessels. On low magnification, lesions are difficult to distinguish from a perivascular pattern. Higher magnification is required to confirm vasculitis by evidence of vascular wall damage and inflammation targeting the vessel wall. Fibrinoid degeneration is the gold standard but is not a common lesion in small-vessel vasculitis in animals. Other lesions include leukocytoclasia (“nuclear dust”), thrombosis, particularly in infectious vasculitis, and direct and indirect evidence of vascular damage, such as loss of endothelial cells, protein-rich edema, microhemorrhage, or infarction. In rare instances, there may be inflammation of medium-sized vessels in the subcutis. Pathomechanisms of small-vessel vasculitis include (1) deposition of immune complexes, (2) direct or indirect damage by infectious pathogens, (3) endothelial damage secondary to vascular events such as thrombosis, and (4) idiopathic, including rare breed-associated vasculitis syndromes. Clinical lesions typically occur in areas supplied by end arteries (footpads, ear tips, and tail tip). Acute lesions include petechia, echymosis, erosion, ulcers, and infarcts. Chronic lesions include eschar formation and scarring.
Vasculopathy refers to changes that appear to be more degenerative than inflammatory, such as thickening and homogenization of the vessel wall with loss of nuclear staining. This may represent either the end stage of prior vascular inflammation or result from low-grade vascular damage over an extended period. The lesion occurs most frequently in ischemic dermatopathies, such as canine dermatomyositis. 17 The resultant atrophic lesions also affect adnexal epithelia leading to follicular atrophy and subsequent alopecia. The dermis may contain increased mucin and deposition of fine fibrillar collagen. Owing to the presence of vacuolar degeneration and occasional basal cell apoptosis in the epidemis, such lesions are sometimes described as cell-poor variants of the cytotoxic pattern.
Folliculitis and Furunculosis
Folliculitis and furunculosis (Fig. 2d) is a common pattern and is readily identified at low magnification. The silhouette of the lesion reflects the part of the follicle affected. When the follicle is destroyed and the hair shaft is released into the tissue (furunculosis), the low-magnification pattern appears nodular. The sebaceous glands may be involved by extension or independently. Owing to the close relationship of sebaceous glands to the hair follicle embryologically, morphologically, and functionally, it is implied that sebaceous gland inflammation is included in this pattern. The term “sebaceous adenitis” refers to lesions where the gland or its duct is the target organ, but there may be conditions where both follicles and sebaceous glands are equally targeted, such as alopecia in Norwegian puffin dogs. 5 Subdivision of the pattern is based on (1) the anatomic portion of hair follicle affected and (2) the nature of the inflammatory response based on predominant inflammatory cell type. 16 The classification is based initially on anatomic features of the follicle, from top to bottom and from the outside to the inside. Luminal folliculitis is characterized by inflammatory cells within the lumen of the follicle. This common form of folliculitis is usually associated with infectious agents including bacteria, parasites, and fungi. Mural folliculitis is a term coined for veterinary use and not found in the human dermatology literature. It affects the wall of the follicle. When the anagen bulb is preferentially affected, the lesion is termed “bulbitis.” Modifying terms such as cytotoxic (interface), mucinotic, necrotizing, pustular, and infiltrative may be applied to mural folliculitis. Cytotoxic mural folliculitis usually indicates an immune-mediated disorder, for example, “pseudopelade,” 15 and less-well-understood conditions such as feline exfoliative dermatitis with or without underlying thymoma. Coexistance of cytotoxicity of follicular epithelia and epidermis, for example in cutaneous lupus erythematosus, is considered a cytotoxic pattern rather than a mural folliculitis. Clinical lesions vary and include alopecia, crusting (superficial folliculitis), papules or nodules (deep folliculitis, furunculosis), scaling (canine sebaceous adenitis), and draining tracts (German shepherd pyoderma).
Intraepidermal Vesicular and Pustular Dermatitis
The intraepidermal vesicular/pustular dermatitis pattern (Fig. 3a) is characterized by the presence of fluid-filled and/or inflammatory cell-containing spaces within the epidermis. The pattern is subdivided by the mechanism of vesicopustule formation. The most common forms are (1) spongiosis (intercellular edema), characterized by keratinocyte separation by extracellular fluid as in pyoderma and (2) acantholysis, characterized by the loss of cohesion induced by damage to desmosomes as seen in pemphigus complex. Vesicles may also form as a result of frictional pressure and following rupture of swollen keratinocytes, known as “ballooning degeneration,” as seen with viral infections (eg, herpes cold sores in people and parapox in goats). Secondary differentiating features include (1) pustule or vesicle size and number, (2) lesion profile (raised versus intraepidermal), (3) location within the epidermis or mucous membrane epithelium, and (4) the type of associated inflammatory cells. Owing to the thin epidermis in haired areas in animals, particularly companion animal species, the vesicles and pustules seldom persist, except on footpads and mucosal surfaces with a thicker epidermis. Clinical correlates are typically crusting, collarettes, and erosion. Examples include canine bullous impetigo, superficial pyoderma, pemphigus foliaceus, pemphigus vulgaris, and spongiotic vesicles in hypersensitivity reactions.
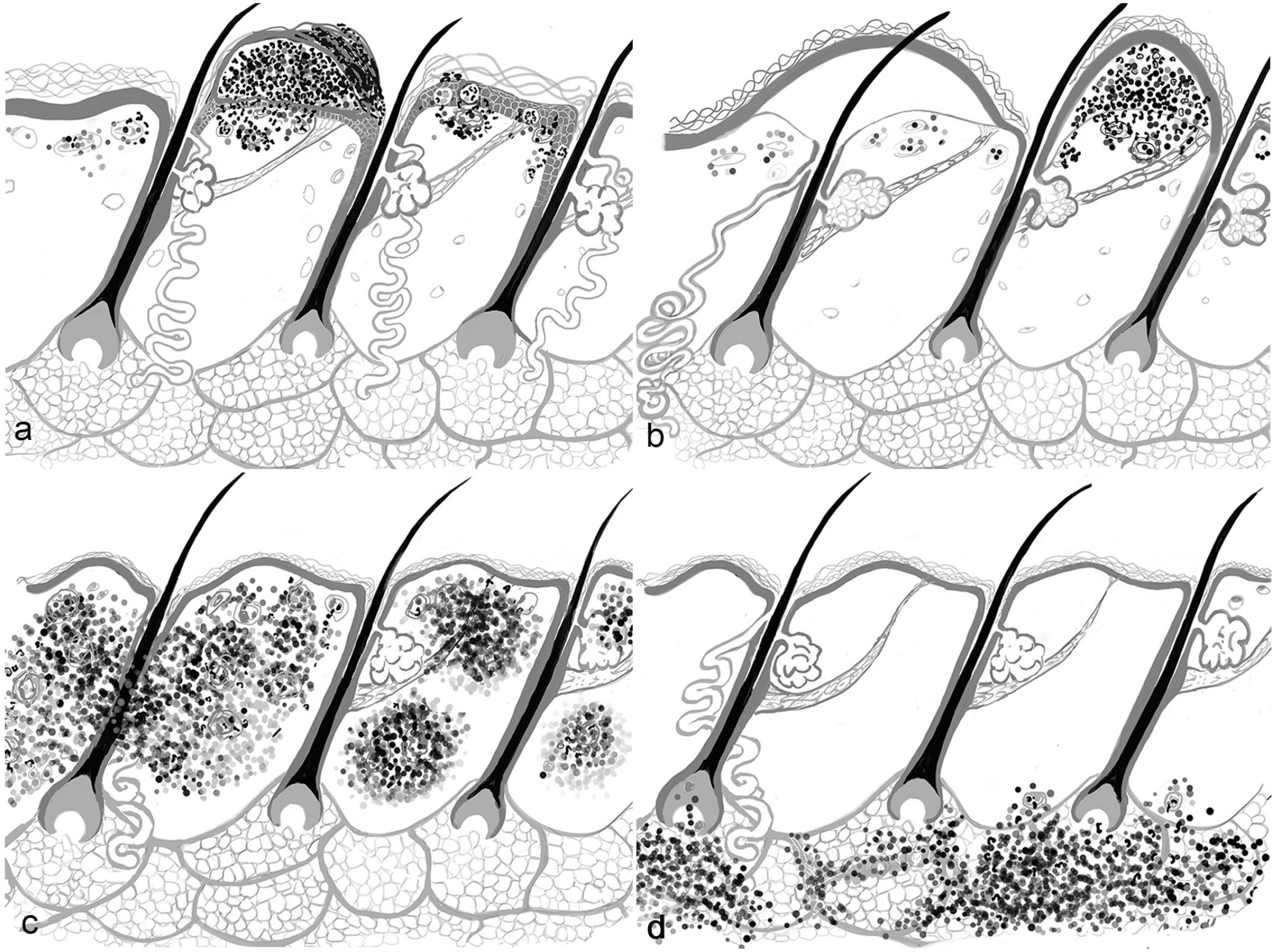
Schematics visualizing the major patterns of inflammatory diseases in domestic animals (drawings by: JA Yager): (a) intraepidermal vesicular/pustular dermatitis, (b) subepidermal vesicular/pustular dermatitis, (c) nodular to diffuse dermatitis, and (d) panniculitis. Note for simplicity, the hair follicles are represented as simple type not compound as occurring in many domestic animals (eg. dogs and cats).
Subepidermal Vesicular and Pustular Dermatitis
Subepidermal vesicular/pustular dermatitis (Fig.3b) is characterized by a separation of the epidermis from the underlying dermis due to an impaired integrity of the basement membrane zone (BMZ). Owing to the complexity of this structure, differentiation between diseases leading to dermo-epidermal separation requires adjunct testing (eg, western blot, immunohistochemistry, immunofluorescence, molecular techniques, and electron microscopy). Patient age, clinical distribution of the lesions, and breed susceptibility can be helpful. Pathomechanisms include (1) congenital defect in one of the BMZ components, leading to a cleft and (2) autoantibodies binding to one of the BMZ components, leading to accumulation of fluid (vesicles or bullae) with or without inflammatory cells. Clinically, these lesions are bullous in people but are usually ulcerative in companion animal species, unless the lesions affect the footpads and mucosal tissues. The proteins targeted also vary in different species. The archetypal human disorder bullous pemphigoid is rare in dogs and cats but is seen more often in horses.24,26 Mucous membrane pemphigoid is the most frequent of this rare group of diseases in the dog. 27
Nodular to Diffuse Dermatitis
The pattern of nodular to diffuse dermatitis (Fig.3c) is characterized by aggregates of inflammatory cells in the dermis that may extend into the panniculus and subcutis. The nodules often expand and coalesce, resulting in a diffuse dermatitis. The first rule out for this pattern is an infectious disease, typically caused by higher bacteria, fungi, oomycetes, or protozoa. However, idiopathic, allergic conditions and rare inflammatory lymphomas also manifest as a nodular pattern.13,22 Lesions are subdivided based on (1) the nature of the inflammatory population and (2) the presence/absence of an etiologic agent including infectious pathogens, such as opportunistic fungi or higher bacteria, and endogenous or exogenous foreign material. Sterile nodular skin diseases are common and include lympho-histiocytic aggregates, as seen with reactive histiocytosis, and sterile pyogranuloma/granuloma syndrome. The former is considered an immune-dysregulatory process and the cause of the latter is unknown. Nodular dermatitis is often, but not always, associated with clinical papules or nodules that may ulcerate, crust, or develop draining tracts. Infectious examples include sporotrichosis and mycobacteriosis. Sterile conditions include feline or equine eosinophilic granuloma, calcinosis cutis, and reactive histiocytosis.
Panniculitis
The panniculitis pattern (Fig.3d) refers to lesions affecting the panniculus exclusively because this narrows the diagnostic possibilities. However, processes beginning in the panniculus often extend into the subcutaneous fat and dermis. It is therefore important to differentiate panniculitis, as a pattern, from lesions characterized by dermatitis with extension into the panniculus and subcutaneous fat, a process that is addressed as panniculitis in the morphologic diagnosis. In people, panniculitis, is divided into septal and lobular categories, reflecting different groups of clinical diseases. Septal panniculitis is extremely rare in companion animals but has been seen in association with septal vasculitis. 35 Panniculitis is further subdivided on the basis of (1) inflammatory cell types and (2) the presence or absence of an etiologic agent. It is rare to see an infectious disease exclusively affecting the panniculus. An exception is feline dermatophytic mycetoma, which differs from “kerion,” a dermatophyte-associated furunculosis. 34 Rapidly growing mycobacteria also primarily affect the adipose tissue. 20 Idiopathic sterile nodular panniculitis is a relatively common but poorly defined condition or group of conditions only in dogs. It should not be confused with another group of equally poorly defined lesions called sterile granuloma/pyogranuloma syndrome, which is predominantly dermal. It is important to note that inflammatory lymphomas may mimic panniculitis in dogs. 23
Limitations and Caveats of Pattern Analysis
The Pattern of Skin Inflammation is not Equivalent to a Specific Disease
Some diseases and patterns have the same designation, which at times can be confusing. An example is “sebaceous adenitis,” a disease associated with the genodermatosis in certain breeds such as standard poodles. 29 This needs to be differentiated from inflammatory lesions, which may affect and destroy sebaceous glands secondarily.
It is also confusing when patterns and lesions have identical designations, such as panniculitis. By definition, the
It is not Uncommon for More Than one Pattern to be Present in a Single Biopsy
The “perivascular pattern” is almost always present but is frequently obscured or obliterated by the other more-diagnostic patterns, such as “nodular to diffuse dermatitis” or “luminal folliculitis.” For example, in direct arthropod-induced injury (insect bite or sting), “perivascular dermatitis” may coexist with “intraepidermal pustular dermatitis” and “folliculitis.”
One Disease May Manifest With Different Patterns Over its Clinical Course
A progression from one pattern to another has been referred to as the “lives of lesions.” 2 For example, subacute canine demodectic mange is characterized by mural lymphocytic folliculitis whereas nodular lesions due to furunculosis may occur in long-standing lesions, particularly with secondary bacterial infection.
One pattern may be a precursor to the development of lesions that represent another pattern. For example, in canine vesicular cutaneous lupus erythematosus, the “subepidermal vesicular” pattern, although often presenting as the most prominent pattern in the biopsy, is a secondary change. The dermo-epidermal separation is the consequence of damage to the basal cell layer of the epidermis, which typifies the “cytotoxic pattern.” Determining the relationship between the 2 is critical in reaching the correct diagnosis.
Not all Patterns Have the Same Diagnostic Utility
It is useful to think in terms of “diagnostic power” of different patterns. The “perivascular” pattern is the least useful, because it represents generic inflammation in the skin. However, the presence of additional features, such epidermal or dermal changes, may be powerful enough to achieve a definitive diagnosis. 32 For example, perivascular dermatitis with pallor of the spinous layer of acanthotic epidermis, marked diffuse parakeratosis, and accentuation of the basal layer supports the diagnosis of superficial necrolytic dermatitis.
“Nodular to diffuse dermatitis” is a moderately powerful pattern depending on whether there is an identifiable cause, such as microbes or foreign material. “Intraepidermal vesiculo-pustular” pattern generally indicates an immune-mediated process or autoimmune disease, once an infectious cause has been eliminated.
Additional changes on higher magnification, particularly acantholytic cells and waves of pustule formation, enable one to parse out pemphigus foliaceus from superficial spreading pyoderma. The “subepidermal vesiculo-pustular” pattern, when observed as the primary pattern, is classical for autoimmune subepidermal bullous dermatoses and some heritable diseases. However, confirmation of individual subepidermal blistering diseases requires ancillary testing, including immunohistochemistry, indirect immunofluorescence, western blotting or gene sequencing, and immune electron microcopy.3,25 The cytotoxic pattern, whether “interface” or “panepidermal,” is diagnostically powerful, because it generally implies an immune-mediated process effected by cytotoxic-T-cells as in erythema multiforme, some viral skin diseases, and some autoimmune diseases, such as cutaneous lupus erythematosus.11,33
Other Diagnostic Approaches
Tissue-Reaction Patterns
There is obviously no single correct approach to microscopic diagnosis of inflammatory skin lesions. In human dermatopathology, one standard textbook “Skin Pathology”31,28 uses a system of “tissue-reaction patterns” that emphasize groups of diseases with similar pathogeneses. Some are irrelevant to veterinary dermatology such as the “psoriasiform” tissue-reaction pattern.
Anatomic Approach
In some human textbooks, a purely “anatomical approach” is used. In “Fitzpatrick’s Dermatology,” the chapter on pathology of the skin divides the integument into “reactive” units: superficial (epidermis and superficial dermis), reticular (deep dermis), hair follicles and glands, and subcutaneous tissue. 18 However, assumptions that these are separable may be invalid. The very different microanatomy of the human skin precludes adopting this methodology in the authors’ opinion.
The main difference between pattern analysis and a purely anatomic approach lies in the initial question asked on scanning power examination of the section. In the pattern approach, the question is “what is (are) the major pattern(s) present?” In a purely anatomic approach, the question is “Which anatomic unit—the epidermis, dermis, adnexa, or fat- is mostly affected?” The latter presents a challenge for the following scenarios: (1) what to do with samples in which both dermal and epidermal lesions are equally present? and (2) what to do when the pathogenetically important lesion may not lie in the anatomical compartment with the most obvious histologic lesion? Seasoned pathologists fall back on their experiential knowledge, but for novices, the pattern approach has the advantage of immediately narrowing the list of differential diagnoses.
Combination of Patterns, Anatomic Location, and Cause
“Skin Diseases of the Dog and Cat” combines the use of patterns of inflammation with the anatomic location affected (epidermis, dermis, adnexa, or panniculus). In addition, as a third step, cause is listed as a category. For example, infectious versus sterile nodular to diffuse granulomatous and pyogranulomatous diseases of the dermis.14,13
Histopathology Remains the Link Between Clinics and Ancillary Techniques
The question could be asked: “why continue to promote methodologies based on the use of conventional microscopy in the age of image informatics and various ancillary techniques such as molecular analytics?”—In both human and veterinary dermatology, molecular methods have long been adjuncts to diagnosis, for example, in identification of genodermatoses, infectious agents, and in tumor diagnosis and management.9,12,19,21 The major thrust in human dermatology has been in oncology, with less emphasis on inflammatory skin diseases. 7 However, transcriptomic technologies are hastening the understanding of the pathogenesis of human inflammatory skin diseases, for example, systemic sclerosis (scleroderma) and psoriasis.30,31 While advanced molecular techniques are largely limited to research laboratories in veterinary dermatology, they are gradually being introduced to assist in diagnosis of inflammatory skin diseases in domestic animals.3,6,8
It is widely acknowledged in human and veterinary medicine that the light microscopic interpretation of hematoxylin-and-eosin-stained tissue sections will remain the mainstay of routine dermatopathology diagnosis.3,7 While including adjunct methods, such as clonality testing, immunohistochemistry, in situ hybridization, or polymerase chain reaction (PCR) is helpful, the results always need to be interpreted in the context of the histologic changes observed.3,6,8 There are many examples demonstrating that relying on adjunct methods alone can lead to incorrect diagnoses of disease processes.4,10 Thus interpretation of histologic features in context with clinical presentation remains a crucial step in veterinary dermatology, whilst keeping in mind that pattern analysis may prove useful in the development of algorithms for computer-assisted diagnosis in the future.
Footnotes
Acknowledgements
The authors would like to acknowledge the European Society for Veterinary Dermatology for providing them the opportunity to develop and refine the pattern analysis approach to dermatopathology diagnosis in veterinary medicine. This manuscript is based on material given by the authors at the ESVD Dermatopathology Workshops over the past 25 years. Although there are many other people to be thanked, they particularly acknowledge the ideas and the inspiration of the late Dr. Tony Stannard, who was a mentor to them all.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
