Abstract
Granulomatous mural folliculitis (GMF) is an uncommon reaction pattern occasionally observed in nonadapted ruminant hosts infected with malignant catarrhal fever viruses. This report characterizes GMF and concurrent cutaneous lesions in 16 goats with crusting dermatitis using histochemistry including hematoxylin and eosin, periodic acid–Schiff, and Grocott’s methenamine silver, and immunohistochemistry for CD3, CD20, ionized calcium binding adaptor molecule 1, and cytokeratin AE1/3. Infiltrates in all 16 GMF cases consisted of macrophages and fewer T lymphocytes, and variably included eosinophils, multinucleated histiocytic giant cells, and/or neutrophils. Formalin-fixed paraffin-embedded skin and fresh skin samples from caprine GMF cases were tested using pan-herpesvirus nested conventional polymerase chain reaction (PCR) and partial sequencing, ovine herpesvirus-2 (OvHV-2) real-time PCR, and OvHV-2 colorimetric in situ hybridization (ISH). Five of 16 goats with GMF (31%) were PCR positive for malignant catarrhal fever viruses, including caprine herpesvirus 3 in 1 goat and OvHV-2 in 4 goats. Three goats also had positive intranuclear OvHV-2 hybridization signal in follicular keratinocytes, among other cell types, localized to areas of GMF. Herpesviruses were not detected in the formalin-fixed paraffin-embedded skin of 9 goats without GMF. This case series describes relatively frequent detections of malignant catarrhal fever viruses in the skin of goats with GMF, including the first report of caprine herpesvirus 3, and localizes OvHV-2 infected follicular keratinocytes within areas of GMF.
Keywords
Malignant catarrhal fever (MCF) is an often fatal, economically important syndrome of domestic and wild even-toed ungulates caused by a group of closely related gammaherpesviruses, collectively referred to as MCF viruses (MCFVs).4,5,14,18,30 Naturally occurring MCF has been reported in more than 30 even-toed ungulate species, mainly Bovidae and Cervidae, and less often Giraffidae or Suidae; in addition, experimental infection and disease have been reproduced in laboratory animal species, including rabbits and hamsters.19,30 MCF is typically a sporadic disease, but significant epidemics occur. 12
Gammaherpesviruses have a tropism for lymphocytes, typically establishing latency therein and inducing lymphocytic proliferation, and can cause a wide spectrum of clinical manifestations ranging from inflammation to neoplasia.4,25 The gammaherpesvirus subfamily comprises seven genera: Bossavirus, Lymphocryptovirus, Macavirus, Manticavirus, Patagivirus, Percavirus, and Rhadinovirus. 17 Macaviruses, which include both MCFVs and other lymphotropic herpesviruses, share a conserved region in the DNA polymerase gene and express the 15A common antigen.4,5,21,30,32
MCFVs can be further categorized into the Alcelaphinae/Hippotraginae and Caprinae subgroups on the basis of their reservoir species. 5 Historical understanding of MCF is largely based on the Alcelaphinae/Hippotraginae subgroup—specifically alcelaphine herpesvirus 1—since these viruses can be propagated in cell culture, unlike viruses in the Caprinae subgroup, such as ovine herpesvirus 2 (OvHV-2).5,19 Currently, the International Committee of Taxonomy of Viruses (ICTV) formally recognizes 5 MCFVs: alcelaphine herpesvirus 1, alcelaphine herpesvirus 2, hippotragine herpesvirus 1, OvHV-2, and caprine herpesvirus 2 (CpHV-2); however, many more MCFVs have been identified and putatively named based on their reservoir host, when known.5,17,30
At least seven different MCFVs cause naturally occurring infection and disease. Alcelaphine herpesvirus 1, the first identified MCFV, is endemic in wildebeest (Connochaetes sp.) and causes wildebeest-associated MCF in domestic cattle and even-toed ungulates in zoological collections.8,19,21,30 OvHV-2 is endemic in domestic sheep and causes sheep associated MCF in domestic cattle, Bali cattle (Bos javanicus domesticus), bison (Bison bison), water buffalos (Bulbalus bulbalis), moose (Alces alces), various deer species, a variety of zoo even-toed ungulate species, and swine.8,19,44,49 Occasionally, sheep develop clinical disease. 34 CpHV-2 is endemic and asymptomatic in domestic and wild goats and causes disease in various deer species, moose, water buffalos, bison, pronghorn (Antilocapra americana), and pigs.8,11,19,30,44,49 The provisionally named malignant catarrhal fever virus of white-tailed deer (MCFV-WTD) subclinically infects goats and has been detected within nasal swabs, suggesting the possibility of viral shedding, 26 and causes disease in WTD and red brocket deer (Mazama americana).8,19,30,33 Renaming this virus to caprine herpesvirus 3 (CpHV-3) has been suggested, 18 and will be used in this report hereafter. Alcelaphine herpesvirus 2-like virus, which may either be a strain of alcelaphine herpesvirus 2 or a closely related novel virus, caused disease in a Barbary red deer (Cervus elaphus barbarus).8,16 MCFV-Ibex subclinically infects Nubian ibex (Capra nubiana) and causes disease in bongo antelope (Tragelaphus euryceros).8,12 OvHV-2-like virus (putatively named ovine herpesvirus-3) causes disease in bighorn sheep (Ovis canadensis). 5
The number of known MCFVs is continually expanding, as recently exemplified by a Californian zoological surveillance study, which tested samples from 96 healthy and diseased ruminant species and subspecies and identified 44 different gammaherpesvirus DNA polymerase genotypes, 34 being new putative gammaherpesviruses; of these, three were novel macaviruses detected in ruminants with clinical MCF. 32
Most if not all ruminant species are thought to carry their own unique adapted macavirus, typically causing asymptomatic persistent infection.5,12,44 In contrast, poorly adapted dead-end hosts usually do not shed infectious virus, and outcomes of infection range from subclinical to severe and fatal.4,5,19 Infection occurs through inhalation, ingestion of virus-laden secretions, or possibly through the ingestion of contaminated feed or water. 19
Domestic goats are the reservoir host for CpHV-2 and can also be naturally infected with OvHV-2 and CpHV-3.7,10,30 It has been suggested that the majority of goats are infected with CpHV-2, with patterns of transmission similar to that of OvHV-2 in sheep flocks.4,21,22,30 MCF-like disease in goats caused by CpHV-2 has not been reported previously. 14 Goats are infected with OvHV-2 following transmission from sheep, and although productive caprine infection is reported, subsequent transmission of OvHV-2 from infected goats to naïve goats or other species has not been reported; thus, their capacity to act as a reservoir host is uncertain.4,10,11,21,30 OvHV-2 caprine infection is most often subclinical, although there have been occasional reports of infection, determined using polymerase chain reaction (PCR) or in situ hybridization (ISH), with vasculocentric and/or cutaneous lesions consistent with MCF.10,14,30,45,47 CpHV-3 has been detected in clinically normal domestic goats in Europe and North America, and it is suspected that goats may be the reservoir host.18,26,30
MCF was first recognized in cattle and can manifest clinically with pyrexia; depression; catarrhal nasal discharge; lymphadenopathy; and/or acute gastrointestinal, central nervous system (CNS), or ocular signs, often resulting in high mortality rates.5,7,8,10–12,24,52 Histologically it typically causes: (1) lymphoproliferation, (2) lymphohistiocytic arteritis with or without fibrinoid necrosis in small to medium caliber arteries, and (3) ulcerative cutaneous and mucosal lesions.5,7,8,10–12,24,52 This triad of histologic lesions, or at minimum, arteritis alone, is sometimes suggested in the MCF literature to be a defining diagnostic feature of clinical MCF across all species. 52 However, this dogma fails to adequately capture the full spectrum of MCF as a broad syndrome, with significant variability in the clinical course, ranging from peracute to chronic, that can cause an array of variously distributed lesions.15,16,21,24 For example, MCFV-Ibex infection in bongo antelope is characterized by necrotizing cholangiohepatitis and neutrophilic and necrotizing myocarditis, and the absence of any grossly apparent erosive oronasal lesions or enteritis. 12
Granulomatous mural folliculitis (GMF) is perhaps the most unusual and striking pathological divergence from the typical acute MCF lesion-triad. GMF was first reported in association with chronic MCF in 2002, in a case series describing infections in 2 Sika deer with CpHV-2. 4 Subsequent to this study, histiocytic mural infiltrates have been reported in an additional Sika deer, as well as a white-tailed deer, two goats, a bighorn sheep, and a cow (Supplemental Table S1).4,10,11,24,28,44,45 These histiocytic mural infiltrates were variably mixed with multinucleated histiocytic giant cells (MNGCs), eosinophils, lymphocytes, and/or plasma cells, and were associated with either CpHV-2 or OVHV-2 infection.4,10,11,24,28,44,45 GMF has otherwise been observed with dermatophytosis and presumed idiosyncratic drug reactions associated with L-thyroxine, cefadroxil, and amitraz in dogs, and methimazole in a cat.3,39,41,42 In the last example, there was complete resolution of skin lesions 15-days following cessation of methimazole administration. 3 Idiopathic GMF has also been recently described in a canine case report, in which stabilization of skin lesions was achieved with combination immunosuppressive therapy. 31 Equine linear alopecia is another idiopathic entity with GMF, clinically characterized by circular areas of alopecia arranged in a linear, vertically-oriented configuration on the neck, shoulders, and/or lateral thorax. 40
Domestic goats have many different skin diseases with overlapping gross lesions; however, the known differential diagnoses for caprine GMF are limited to OvHV-2 infection and dermatophytosis. 50 The latter, caused by Trichophyton verrucosum, Trichophyton mentagrophytes, and Microsporum canis, occurs occasionally in goats, usually affecting the head, ears, and/or neck, and typically resolves after 5-weeks in immunocompetent animals. 45 Dermatophytosis can be differentiated from OvHV-2 induced GMF using special histochemical stains—such as Grocott’s methenamine silver or fungal culture.
This retrospective case series provides the first evaluation of a large, unrelated group of domestic goats with GMF with systematic histologic assessment, interpreted in conjunction with pan-herpesvirus nested conventional PCR coupled with partial confirmatory sequencing, OvHV-2 real-time PCR, and OvHV-2 ISH.
Materials and Methods
Sixteen goats with a diagnosis of GMF were selected from the Cornell University, College of Veterinary Medicine Animal Health Diagnostic Center (AHDC) case archive, from submissions dating between 2013 and 2022. Submission forms were used to compile information about the property of origin, herd history, patient signalment, clinical signs, gross skin lesion appearance, and treatments. Submitting clinicians were contacted for follow-up information. All skin samples were routinely processed and stained with hematoxylin and eosin, periodic acid-Schiff, and Grocott’s methenamine silver.
Two of 16 goats were full necropsy cases (goats 2 and 5) and had a representative range of tissues that were histologically assessed. Examined tissues in goat 2 included lymph node, lung, skeletal muscle, heart, liver, kidney, and small intestine. Examined tissues in goat 5 included brain, spinal cord, sciatic nerve, eye, thyroid glands, adrenal glands, lung, skeletal muscle, tongue, heart, spleen, liver, kidney, mesenteric lymph node, pancreas, stomach, small intestine, and large intestine. Goat 5 was the subject of a previous case report. 36
Pan-herpesvirus nested conventional PCR followed with partial sequencing and OvHV-2 specific probe-hydrolysis real-time PCR (Taqman) were performed by the Virology Laboratory at the AHDC following established protocols and procedures. Samples tested consisted of formalin-fixed paraffin-embedded (FFPE) skin tissue scrolls for 15 of the goats with GMF, a fresh skin sample for the remaining goat with GMF (goat 4), and FFPE skin tissue scrolls from nine randomly selected goats with one or more dermatological condition other than GMF. These nine non-GMF cases were also taken from the AHDC case archive and included zinc-responsive dermatosis, ectoparasitism, selenium deficiency, and/or bacterial skin infection. Viral DNA was extracted from FFPE samples using the RecoverAll Total Nucleic Acid Isolation Kit for FFPE (Invitrogen), and from fresh tissue using the Viral RNA Mini Kit (Qiagen) following the manufacturer’s instructions.
Pan-herpesvirus conventional PCR was performed on the extracted nucleic acid using the SuperScript III One Step RT-PCR System (Invitrogen). Primers were previously described in VanDevanter, 48 with the exception of the DFASA primer, which was modified (CODEHOP) according to Rose. 36 Secondary PCR products were run on a 1.5% agarose gel. Amplicons from the secondary PCR were purified using the QIAquick PCR Purification Kit (Qiagen) and submitted for Sanger sequencing at Cornell Bioresource Center.
Real-time PCR was performed on the extracted nucleic acid using the Path-ID Multiplex One-Step RT-PCR Kit (Applied Biosystems) following manufacturer’s instructions. OvHV-2 specific primers and probe for the assay were designed at the AHDC based on sequence obtained from amplicons generated using primers previously described. 23 Thermal cycling conditions were 10 minutes at 48°C, 10 minutes at 95°C, and then 40 cycles of 15 seconds at 95°C followed by 1 minute at 60°C.
ISH was performed using 5-µm-thick sections of FFPE skin from the goats with GMF using a nucleic acid probe targeting open reading frame (ORF) 25 and 50 of OvHV-2 (ACD, catalog #501098), following previously published methods. 33 All slides were tested in tandem with negative control probe DapB (ACD, catalog #312038) and a positive control probe for caprine housekeeping gene Ch-ISG15-sense (ACD, catalog # 566121). In addition, an OvHV-2 real-time PCR positive goat with GMF was used as an internal positive control, with the OvHV-2-orf25-orf50 probe.
Immunohistochemistry (IHC) was also performed on sections of FFPE skin from a subset of 7/16 goats with GMF (goats 1, 2, 3, 5, 7, 14, and 15), using antibodies against: CD3, a T lymphocyte marker (mouse monoclonal anti-CD3 antibody, Leica, Newcastle Upon Tyne, United Kingdom, catalog #PA0553); CD20, a B lymphocyte marker (rabbit polyclonal anti-CD20 antibody, ThermoFisher/Invitrogen, Massachusetts, United States, catalog #PA5-16701); ionized calcium binding adaptor molecule 1 (Iba1), a histiocytic cell marker (rabbit polyclonal anti-Iba1, Wako, Osaka, Japan, catalog #019-19741); and CKAE1/3 (syn. pancytokeratin), an epithelial cell marker (mouse monoclonal anti-AE1/AE3 antibody, Dako, California, United States, catalog #M3515). Positive control tissues included sections of FFPE caprine lymph node and spleen for CD3, CD20, and Iba1, and FFPE caprine skin for CKAE1/3. Briefly, 5 μm tissue sections were deparaffinized with Bond Dewax Solution (Leica, catalog #AR9222). Pretreatment with heat-induced antigen retrieval was performed using citrate-based buffer pH 6 for CD20 (30 minutes) (Bond Epitope Retrieval Solution 1, Leica, catalog #AR9961), and using Tris/EDTA pH 9 for Iba1 (10 minutes), and CD3 and CKAE1/3 (both 20 minutes) (Bond Epitope Retrieval Solution 2, Leica, catalog #AR9640). Endogenous peroxidase activity was blocked with a 3% peroxide solution for 5 minutes (Leica, catalog #DS9800). CD3 (ready-to-use), CD20 (diluted at 1:400), and Iba1 (diluted at 1:3000) antibodies were applied for 15 minutes; CKAE1/3 antibody (diluted at 1:200) was applied for 60 minutes. Next, for CD3, the PowerVision polymeric horseradish peroxidase (PV-HRP) anti-mouse (Leica, catalog #PV6114) IgG reagent was applied for 10 minutes; for both CD20 and Iba1, PV-HRP-anti-rabbit (Leica, catalog #PV6119) IgG reagent was applied for 10 minutes; and for CKAE1/3, PV poly-alkaline phosphatase anti-mouse (Leica, catalog #PV6110) IgG reagent was applied for 30 minutes. Subsequently, tissues were incubated with Bond Polymer Refine Detection for 10 minutes (Leica, catalog #DS9800). Tissues were developed with 3,3-diaminobenzidine (DAB) chromogen (Leica, catalog #DS9800) for 10 minutes. The slides were counterstained with hematoxylin (Leica, catalog #DS9390) for 5 minutes, dehydrated, cleared, and mounted.
Results
Pan-herpesvirus nested conventional PCR with partial sequencing and OvHV-2 real-time PCR results for the 16 goats with GMF are summarized in Table 1. In addition, Table 1 also includes a summary of the major histopathologic findings. Five of the 16 goats with GMF had FFPE or fresh skin samples that were pan-herpesvirus PCR positive (31%), including goats 1 to 4 with OvHV-2, and goat 5 with CpHV-3. None of the 9 goats without GMF were MCFV PCR positive.
Pan-herpesvirus conventional nested PCR, partial sequencing, and OvHV-2 real-time PCR results for goats with GMF.
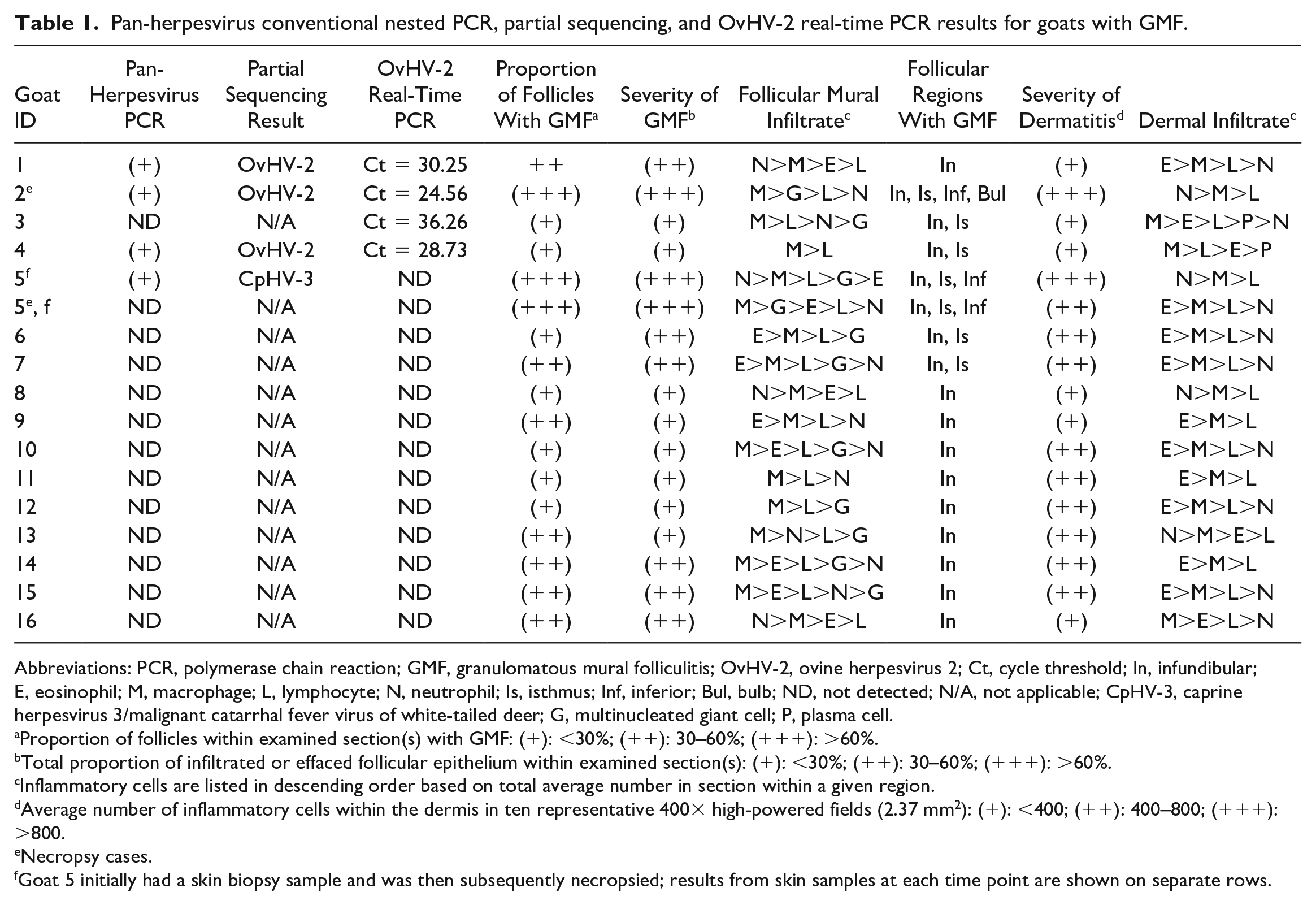
Abbreviations: PCR, polymerase chain reaction; GMF, granulomatous mural folliculitis; OvHV-2, ovine herpesvirus 2; Ct, cycle threshold; In, infundibular; E, eosinophil; M, macrophage; L, lymphocyte; N, neutrophil; Is, isthmus; Inf, inferior; Bul, bulb; ND, not detected; N/A, not applicable; CpHV-3, caprine herpesvirus 3/malignant catarrhal fever virus of white-tailed deer; G, multinucleated giant cell; P, plasma cell.aProportion of follicles within examined section(s) with GMF: (+): <30%; (++): 30–60%; (+++): >60%.
Total proportion of infiltrated or effaced follicular epithelium within examined section(s): (+): <30%; (++): 30–60%; (+++): >60%.
Inflammatory cells are listed in descending order based on total average number in section within a given region.
Average number of inflammatory cells within the dermis in ten representative 400× high-powered fields (2.37 mm2): (+): <400; (++): 400–800; (+++): >800.
Necropsy cases.
Goat 5 initially had a skin biopsy sample and was then subsequently necropsied; results from skin samples at each time point are shown on separate rows.
Summarized for each of the 16 goats with GMF is the signalment, property information, gross lesions, diagnostic tests, treatments, clinical duration and progression, and outcomes, in Supplemental Table S2. Property information included location, number of conspecifics, and presence of other even-toed ungulates. Recorded information pertaining to gross lesions included skin lesion characteristics and distribution, and the presence of noncutaneous lesions. All 16 goats with GMF were from the Northeast or Midwest of the United States. Ages ranged from 5-months to 12-years. There was no sex or breed predilection, nor was there a seasonal distribution.
All 16 goats had similar gross lesion characteristics (Fig. 1a–d). The most common were crust (13/16, 81%), alopecia (7/16, 44%), scale (6/16, 37%), lichenification (6/16, 37%), fissures (4/16, 25%), erosions (4/16, 25%), and/or discharge (2/16, 12.5%). The goats were variably pruritic (six with, six without, four unspecified). Lesions were most often distributed on the facial region (14/16, 87%), which included the following: face, pinnae, periocular area, muzzle, lips, and chin. Of these areas, the pinnae (8/16, 50%) and periocular area (8/16, 50%) were most common. Other frequently affected regions included the legs (9/16, 56%), tail (5/16, 31%), dorsal trunk (4/16, 25%), and lateral trunk (4/16, 25%).
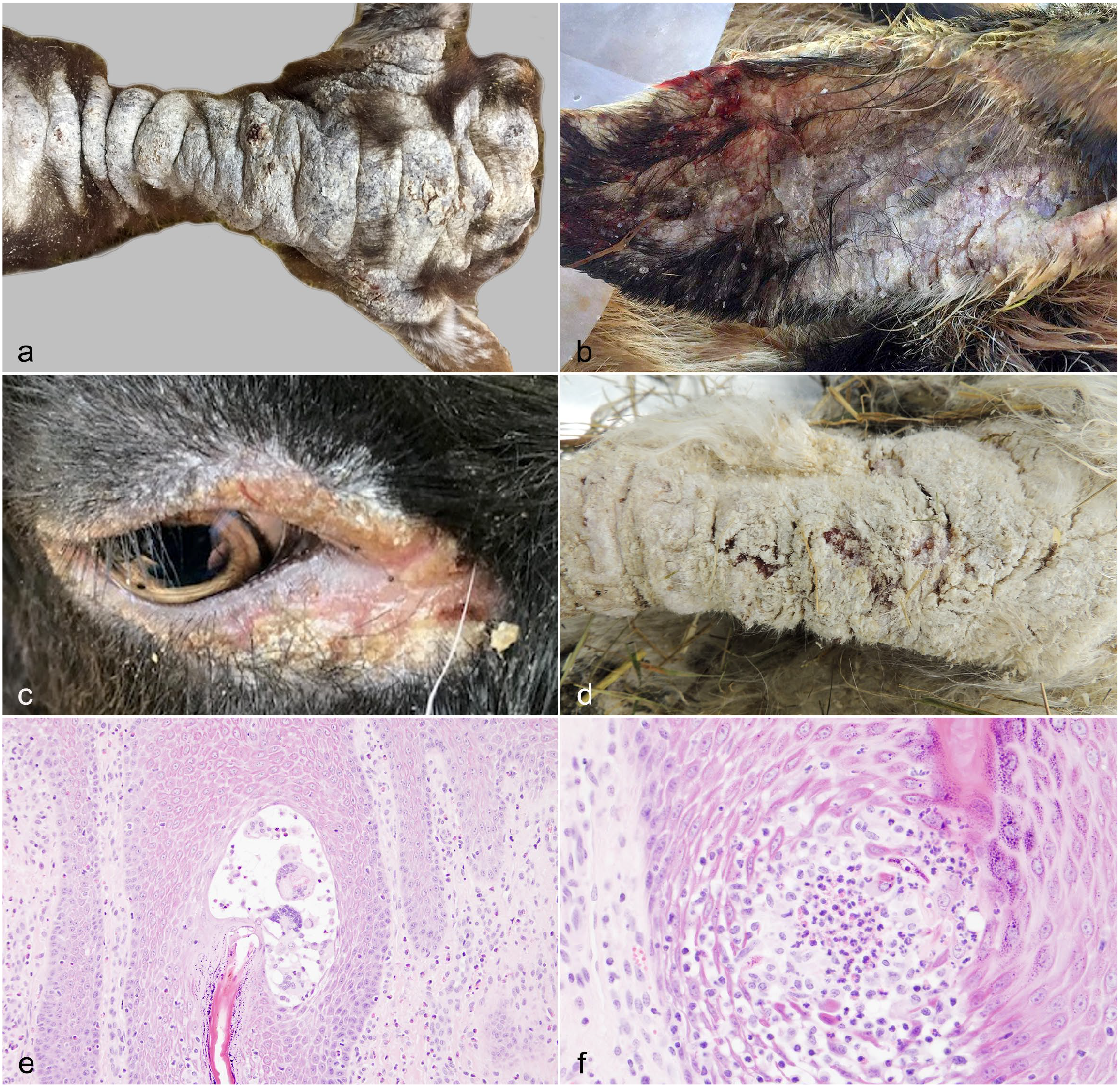
Granulomatous mural folliculitis, goats. (a) Goat 1, ovine herpesvirus 2 real-time polymerase chain reaction (PCR) positive. Severe generalized alopecia, scale, and lichenification with multifocal crusts. (b) Goat 2, ovine herpesvirus 2 real-time PCR positive. Alopecia, scale, crust, and fissuring on the medial pinna. (c) Goat 3, ovine herpesvirus 2 real-time PCR positive. Periocular alopecia with erythema and scale. (d) Goat 5, pan-herpesvirus conventional nested PCR positive, partially sequenced as caprine herpesvirus 3. Alopecia, crust, and deep fissuring on the limb. (e) Goat 15. A spongiotic mural pustule with several multinucleated giant cells in the infundibular region. Hematoxylin and eosin (HE). (f) Goat 15. Transmural eosinophilic and histiocytic infiltration and spongiosis in the infundibular region. HE.
Localized skin disease that became progressively more generalized was noted in 3/16 goats (19%), first occurring on the dorsal trunk (goat 1); in the periocular area (goat 7); or the pinnae, dorsum, and legs (goat 16). In goats 5 and 8, pyrexia and widely distributed skin lesions progressed to generalized debilitation necessitating euthanasia. Goat 2 was unique in first presenting with pyrexia and respiratory signs, followed by widespread skin lesions and then systemic illness prompting euthanasia. The same clinical signs and outcome occurred in three other unsampled kid herd mates of goat 2.
Of the 16 goats with GMF, 11 were reported to live with conspecifics (69%), of which 4/11 had herd mates with skin lesions (36%). When specified, even-toed ungulate species on the property shared with the goats included sheep (goats 1 & 2), llamas and alpacas (goat 5), and pigs (goat 9); none of which were reported to have skin lesions.
Exclusion of other differential diagnoses for skin disease by the referring veterinarians prior to biopsy sampling, as reported on submission forms, was limited. Testing performed included skin scraping without detection of mites (3/16, 19%), negative parapoxvirus PCR tests (3/16, 19%), and zinc blood levels (2/16, 13%), which were low in both tested animals, but with no response to subsequent zinc therapy.
In 14/16 goats with GMF there were no reported abnormal noncutaneous physical exam or gross findings (88%). The exceptions were goats 2 and 12, both of which had weight loss. Goat 2, which was one of two goats that were necropsied, also had verminous pneumonia and severe, multifocal, subacute myonecrosis with dystrophic mineralization and histiocytic and plasmacytic infiltrates of uncertain etiology.
The clinical duration varied from 2 weeks (goat 2—euthanized) to greater than 4 years (goat 15, partial resolution of skin lesions, which remained in a stable condition). In total, 5/16 goats with GMF were euthanized (31%), 3/16 had partial resolution of cutaneous lesions (19%), 2/16 had complete resolution of cutaneous lesions (13%), and outcomes were unavailable for 6/16 goats (38%). Of the 5 goats euthanized, 3 were MCFV PCR positive (goats 1, 2, and 5). Outcomes were unavailable for the remaining 2 MCFV PCR positive goats. CpHV-3 PCR positive goat 5 was initially managed with an immunosuppressive dose of corticosteroids (dexamethasone, intramuscular). This led to improvement after a week with crusts falling off and revealing normal skin beneath. However, after 1 month the dexamethasone dose was reduced and there was onset of hindlimb paresis and recrudescence of skin lesions, necessitating euthanasia. 37 Of the three goats reported to have partial resolution of skin lesions, specific information regarding how the condition had improved was only available for goat 15. This goat was said to have a reduced severity of lesions, with only mild scaling, crusting, and alopecia multifocally across its body. Injectable corticosteroids were administered to all three goats with partial resolution of cutaneous lesions, among other concurrent therapies. Complete resolution of cutaneous lesions occurred in 2/16 goats, including goat 9 following administration of topical permethrin and injectable methylprednisolone and goat 11 following topical application of zinc oxide.
Histologic findings in affected follicles of goats with GMF included spongiosis (16/16, 100%) (Fig. 1e, f), areas of transmural effacement by the granulomatous infiltrate (12/16, 75%) (Fig. 2a, b), and keratin debris within areas of GMF (13/16, 81.3%). This keratin debris was sometimes present within MNGCs (Fig. 2a inset). Suppurative luminal folliculitis was present in 8/16 goats (50%) and was considered suggestive of secondary bacterial folliculitis. There was no consistent association between this finding and a clinical history of pruritus. Sebaceous and epitrichial sweat glands were unaffected in all goats.
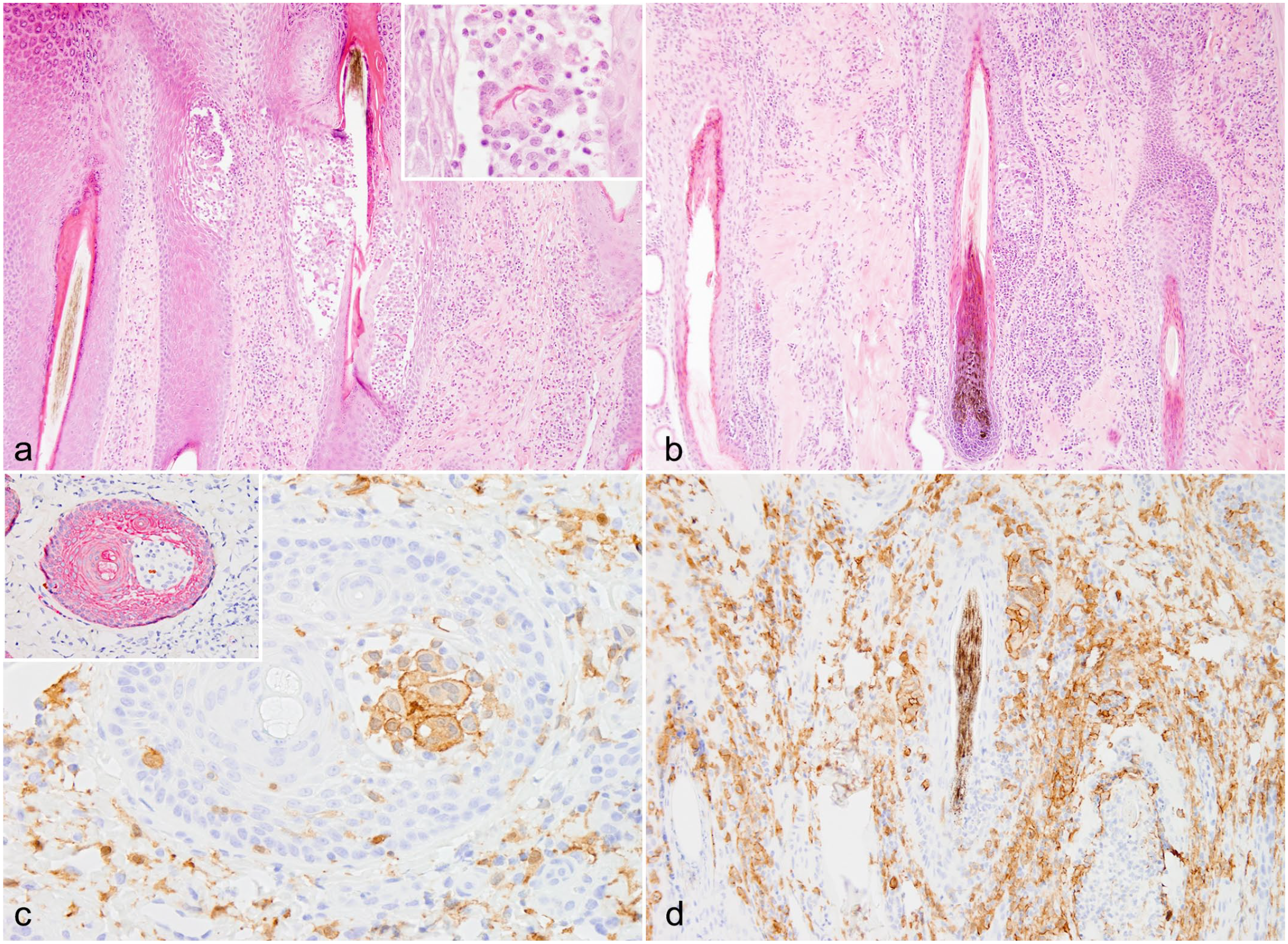
Granulomatous mural folliculitis, goats. (a) Goat 14. Transmural granulomatous mural folliculitis spanning from infundibular to isthmus regions. Hematoxylin and eosin (HE). Inset: multinucleated giant cell with intrahistiocytic keratin debris. HE. (b) Goat 2. Transmural effacement of the inferior region by macrophages, neutrophils, fewer lymphocytes and eosinophils, and occasional multinucleated giant cells with a similar infiltrate extending throughout the dermal interstitium. HE. (c) Goat 7. Intramural multinucleated giant cells have positive membranous and cytoplasmic ionized calcium binding adaptor molecule 1 immunolabeling (brown), consistent with histiocytic origin. Ionized calcium binding adaptor molecule 1 immunohistochemistry (IHC). Inset: Unlike the surrounding follicular keratinocytes, multinucleated giant cells do not have membranous or cytoplasmic pancytokeratin (CKAE1/3) immunolabeling (red), indicating these cells are not epithelial syncytia. CKAE1/3 IHC. (d) Goat 2. Mononuclear cells throughout follicular walls and the dermal interstitium have positive membranous and cytoplasmic ionized calcium binding adaptor molecule 1 immunolabeling, consistent with a granulomatous infiltrate. Ionized calcium binding adaptor molecule 1 IHC.
Consistent findings in the epidermis of all goats with GMF included hyperkeratosis and irregular epidermal hyperplasia. Hyperkeratosis was mixed para- and orthokeratotic in 11/16 (69%) goats. Other common epidermal changes included crusts in 15/16 (94%) goats, which were typically serocellular (14/16, 88%), and subcorneal and intraepidermal pustules in 10/16 (63%) goats. Pustules consistently featured neutrophils, variably mixed with macrophages (6/16, 38%), MNGCs (4/16, 25%), and/or eosinophils (4/16, 25%). All goats had dermatitis, spanning from the superficial to the deep dermis, and it was typically a perivascular and perifollicular pattern (15/16, 94%). Vasculitis was absent in all goats, including both that underwent full necropsy evaluations. Periodic acid-Schiff and Grocott’s methenamine silver stains did not reveal dermatophyte or other fungal structures in examined skin sections from any of the 16 goats.
Iba1 IHC was performed in 7/16 goats with GMF including four goats with either OvHV-2 or CpHV-3 infection and three goats without detectable MCFV infection. It consistently demonstrated moderate to large numbers of macrophages in affected follicles, the epidermis, and the dermis (Fig. 2c, d). The MNGCs were confirmed to be of histiocytic origin, and not follicular keratinocyte syncytia (Fig. 2c inset), using Iba1 and pancytokeratin IHC. CD3 IHC also showed the presence of T lymphocytes in moderate to large numbers in the dermis, and in low to moderate numbers in the epidermis and affected follicles, in 6/7 goats. Goat 14 was an exception, having large numbers of follicular intramural T lymphocytes. Small numbers of B lymphocytes were present in the sections and only formed occasional dermal aggregates in 3/7 goats. These B lymphocyte aggregates were scattered randomly in the mid and deep dermis.
Of the 16 goats with GMF, three of the four OvHV-2 real-time PCR positive goats were OvHV-2 ISH positive. These included goats 1 (Fig. 3a, b), 2 (Fig. 3c), and 4. Identification of cell types with positive hybridization signal was not always possible due to relatively poor cellular morphology. Those that could be confidently distinguished included epidermal keratinocytes, follicular keratinocytes, endothelial cells, and MNGCs. Cells within heavily disrupted follicles were often nonidentifiable and could have been keratinocytes, follicular dendritic cells, or infiltrating lymphocytes or macrophages. Cells within the dermis were also often nonidentifiable and may have been fibroblasts or inflammatory cells. When the cell type could be confidently determined by morphology, the positive signal was most common in follicular keratinocytes (Fig. 3a, b). Positive signal was less common in epidermal keratinocytes and endothelial cells lining thin-walled vessels (capillaries, venules, and/or lymphatics, present in the mid dermis, deep dermis, and subcutis). Positive signal was rare within follicular intramural MNGCs (Fig. 3c). Follicular keratinocytes and epidermal keratinocytes with positive signal were only observed within areas of degeneration and inflammation (Fig. 3a, b). Endothelial cells with positive signal were present in areas with and without adjacent dermal and subcutaneous inflammation.
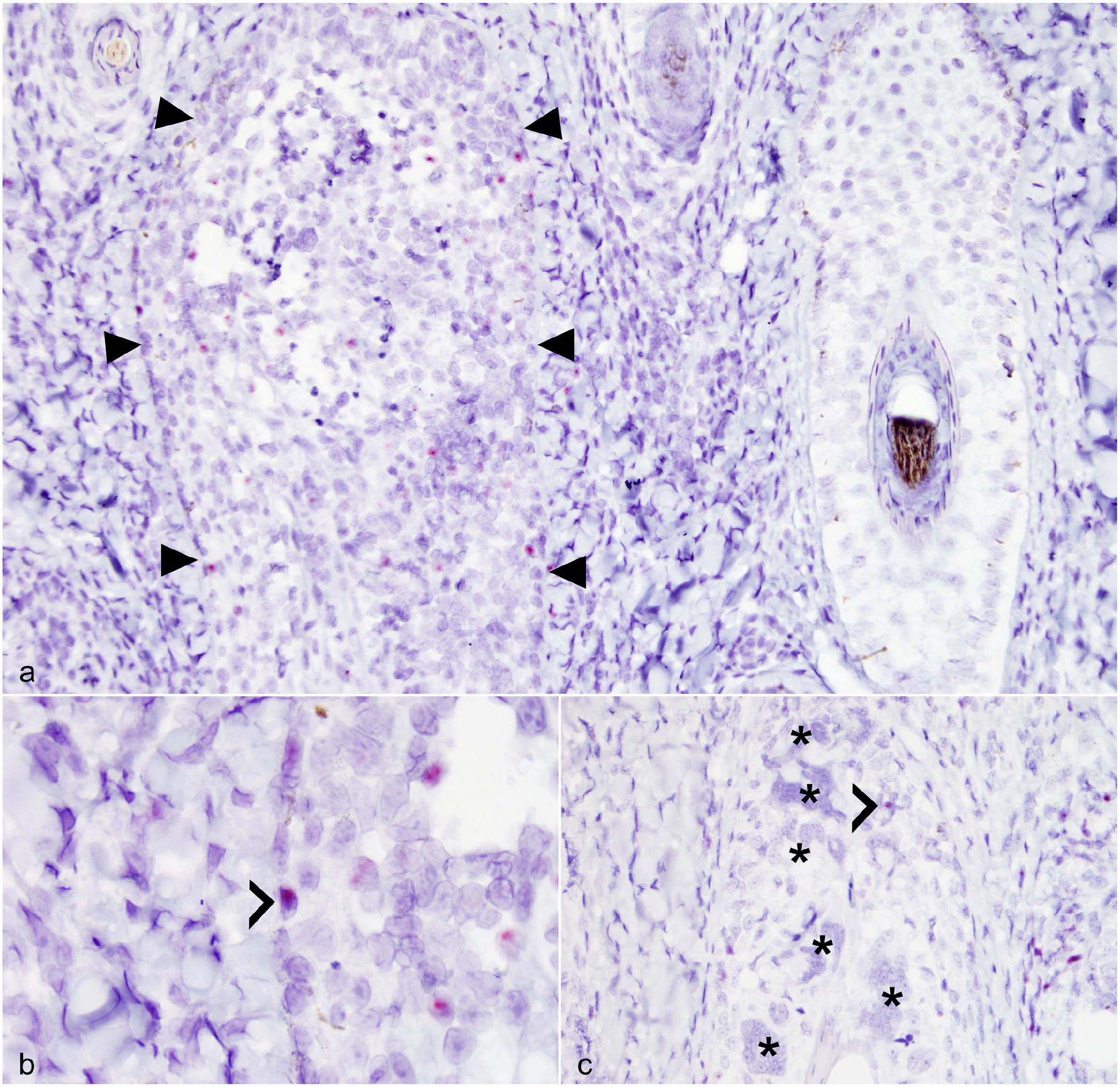
Granulomatous mural folliculitis, goats. (a) Goat 1. The follicle delineated by arrowheads has disrupted architecture with infiltrating mononuclear cells. In contrast to the adjacent normal follicle on the right, this infiltrated follicle has numerous intranuclear puncta of hybridization signal; several are within follicular keratinocytes, and others are within individualized mononuclear cells of uncertain histogenesis. Ovine herpesvirus 2 in situ hybridization. (b) Goat 1. Higher magnification of the inflamed follicle in (a) showing intranuclear ovine herpesvirus 2 hybridization signal within a basilar follicular keratinocyte (chevron). Ovine herpesvirus 2 in situ hybridization. (c) Goat 2. Granulomatous mural folliculitis with many multinucleated giant cells. Although the majority lack ovine herpesvirus 2 hybridization signal (asterisks), one multinucleated giant cell has an intranuclear puncta (chevron). Ovine herpesvirus 2 in situ hybridization.
Discussion
Exploration into the mechanisms underlying GMF in MCFV-infected ruminants is in its infancy. Contrastingly, the pathogeneses of the more typical, well characterized epidemiological forms of MCF, including sheep associated MCF and wildebeest-associated MCF, have been more extensively researched, but are also incompletely understood.14,28,30
Experimental infections of sheep with OvHV-2 have revealed initial lytic replication in alveolar epithelium,30,46 with subsequent switching of cell tropism to latently infect leukocytes.19,30 Infected leukocytes include CD4+ helper T cells and CD8+ T cells of undetermined type. 27 This transition from lytic to latent infection is thought to be achieved via expression of multiple immunomodulatory proteins, which likely influence innate, cell-mediated, and humoral responses. 27 It has been suggested that following this period of latency in lymphocytes, OvHV-2 reactivates, infects the nasal epithelium, and undergoes lytic replication resulting in viral shedding in nasal secretions.30,46
The same alteration in gene expression, from lytic to latent phases, is not observed in OvHV-2 experimentally infected rabbits and bison. 30 Instead, there is a continued lytic pattern of gene transcription (e.g., ORF25) in diseased tissues, indicating that lytic replication is involved in the pathogenesis of typical OvHV-2-induced acute MCF. 20 In this case series, OvHV-2 infection of follicular keratinocytes was localized to degenerate and inflamed follicles using OvHV-2 ISH, suggesting that virus-induced lysis may also be involved in the pathogenesis of OvHV-2 induced GMF. Given that herpesviruses are double stranded DNA viruses, conclusive demonstration of lytic replication in follicular keratinocytes would require DNAase pretreatment to ensure that the OvHV-2 ORF 25 and 50 ISH probe was hybridizing with RNA transcripts, rather than viral genomic DNA. 33 Alternative methods of identifying lytic infection include fluorescence immunohistochemistry using antibodies against OvHV-2 capsid and reverse transcriptase PCR for the detection of ORF-25 RNA transcripts.6,46
In this case series there was colocalization of OvHV-2 infected follicular keratinocytes and intramural leukocyte infiltration predominated by macrophages. This concurrence suggests that keratinocyte infection and subsequent recruitment of macrophages may also be involved in the pathogenesis of MCF-induced GMF. Follicular keratinocytes have a variety of specific functions in physical and immunological defenses, which have led to some authors categorizing them as a type of immune cell. 1 Keratinocytes have interferon (IFN)-gamma receptors and when bound by IFN-gamma, either derived from lymphocytes or keratinocytes, outcomes include upregulation of IFN-gamma production (i.e., a positive feedback loop), induction of proteins that prevent or interfere with viral replication, increased antigen presentation via expression of MHC II molecules, and production of chemokines that are specific for monocytes, lymphocytes, and natural killer cells.1,2,13
In contrast with the results in this case series, a 2022 case report of caprine GMF with OvHV-2 infection observed OvHV-2 hybridization signal in mononuclear inflammatory cells rather than follicular or epidermal keratinocytes, MNGCs, or endothelial cells. These infected mononuclear cells were found surrounding lesions of arteritis, transmigrating and surrounding follicles, and within eroded epidermis, possibly suggesting an immune-mediated pathogenesis. 45 Many previous studies involving various even-toed ungulate species with classic vasculocentric acute MCF have emphasized the importance of immune-mediated mechanisms of injury in the pathogenesis of MCF, rather than lytic viral infection. Precisely which leukocytes are involved, the proportion of intralesional leukocytes that are infected with virus, and to what degree other local cell types are infected within vascular lesions varies between studies.
In a 2019 OvHV-2 ISH study involving various even-toed ungulate species and rabbits with typical acute MCF lesions, hybridization signal was detected in lymphocytes within and around arterial walls, and no signal was evident in epithelium, endothelium, or smooth muscle, in various tissues. 33 The predominant leukocyte within vascular lesions was determined by IHC to be CD3+ lymphocytes. 33 Similar findings in past studies have been considered supportive of the theory that OvHV-2 induced proliferation and dysfunction of T lymphocytes, with subsequent reduced tolerance of medium-sized arterial self-antigens, is central to the pathogenesis of widespread vasculitis.28,43 In older studies, detection of a predominant CD8+ T cell population has been interpreted as being evidence of cytotoxic alpha-beta T cells; however, a 2010 study involving bison experimentally infected with OvHV-2 instead showed a majority of CD8+/perforin+ gamma delta T cells, which are cytotoxic lymphocytes of the innate immune system. 29 This is an interesting finding within the context of caprine GMF, as gamma delta T cells have been shown to play a role in granulomatous responses to certain infections in various animal species (e.g. with mycobacteriosis), and are particularly common in epithelial and mucosal barriers in goats.35,51
In contrast with the 2019 OvHV-2 ISH study, a more recent 2021 OvHV-2 ISH study in cattle, water buffalo, and bison with typical acute MCF lesions that utilized IHC and immunofluorescence found that macrophages predominated within vascular lesions, with far fewer T cells. 38 Both cell types were proliferating within affected vessel walls and were infected with OvHV-2. 38 Other cell types infected with OvHV-2 included endothelial cells, smooth muscle cells, and fibroblasts. 38 From these results, it was concluded that macrophages, in addition to T cells, are involved in causing vasculitis, both in recruiting other cells and in releasing pro-inflammatory mediators, possibly following recruitment and activation by infected endothelial cells. 38
In this case series, five of the 16 goats with GMF had FFPE or fresh skin samples that were pan-herpesvirus PCR positive (31%), including four goats with OvHV-2 (25%) and 1 goat with CpHV-3 (6%). None of the nine goats without GMF were MCFV PCR positive. Despite being considered endemic in domestic and wild goats, 19 CpHV-2 was not detected in goats with (16) or without (9) GMF in the present study. These results differ significantly from two previous large-scale surveys of MCFV infection in goats in North America in 2001 21 and in Europe in 2015, 26 which each found a different MCFV to be most prevalent in their study population. In the North American study, 104/124 goats (83.9%) were CpHV-2 PCR positive, 11/124 (8.9%) were OvHV-2 PCR positive, and 2/122 (1.6%) were CpHV-3 PCR positive.18,21 In the European study, the most prevalent MCVF was CpHV-3 (5/42, 12%), followed by CpHV-2 (4/42, 10%), and lastly OvHV-2 (3/42, 7%). 26 There are many possible factors influencing these differences, including different geographical locations, inclusion criteria for sampling, sample types, and testing methods.18,21,26
The absence of MCFV detection in 11/16 (69%) of the goats in this case series with GMF does not exclude the possibility of undetected virus. Testing for 15 of the goats was performed on FFPE skin, which had spent an uncertain amount of time in formalin prior to processing. Prolonged fixation is known to reduce test sensitivity, 49 hence the exclusion of tissues held for greater than six days in a 2019 OvHV-2 ISH study. 33 The detection of CpHV-3 in the skin biopsy sample of goat 5, but not the necropsy sample of skin collected 1-month later, suggests the possibility of either transient replication of the virus in the affected skin area, or fluctuations in levels of detectable virus within skin. In humans, GMF is a rare manifestation among several different cutaneous isotopic responses that have been observed following herpes varicella zoster virus infection. Some investigators have suggested persistence of varicella zoster virus DNA within the GMF lesion is not involved in the inflammation, and that GMF may instead be a form of delayed-type hypersensitivity reaction. 9
In this study, caprine GMF was observed in both male and female animals, with ages ranging from 5-months to 12-years, and in various breeds with no apparent predilections. Prior reports of GMF in goats included an 18-month-old crossbred doe in the United Kingdom and a 1-year-old Nigerian dwarf wether in the United States.10,45 GMF was observed in goats during all seasons, which is consistent with typical infection dynamics with sheep OvHV-2 transmission to cattle. 25 Cattle are infected year-round, with only a modest increase around lambing season. 25 This pattern of infection cannot be generalized to all MCFVs; for example, alcelaphine herpesvirus 1 is intensely shed by wildebeest during the first 90-days of life and, as such, cattle are usually infected during calving season. 25
Two of the goats in this case series with OvHV-2 infection had available information about other species of even-toed ungulate being kept on the same property and both had been kept with sheep. The same was true in previous case reports of caprine GMF with OvHV-2 infection,10,45 and similarly in a bovine GMF case report with OvHV-2 infection, in which there were sheep in the adjacent paddock. 27 Natural transmission of OvHV-2 from carrier sheep to naïve animals predominantly occurs through reactivation from latency and the shedding of virus in nasal secretions. 46 OvHV-2 is transmissible over large distances, of up to at least 5 km, based on natural outbreaks with transmission between sheep feed lots and distant bison operations. 20 The CpHV-3 infected goat was kept with llamas and alpacas, as well as horses and chickens. Camelid infection with CpHV-3 has not been previously reported, although such surveillance studies have not been performed, to our knowledge.
Of the 16 goats with GMF, 11 were reported to live with conspecifics, of which four had herd mates with macroscopically similar skin lesions, none of which were further investigated. Although MCF is usually sporadic and affects individual animals, outbreaks are also reported, most often in bison and deer, and sometimes in cattle. 38 In previous reports of caprine MCF, one OvHV-2 infected goat with GMF was housed with 3 other goats, none of which were affected. 45 In a case series of OvHV-2 infected goats with more typical acute MCF lesions, two of three goats on one property were affected, and one of three goats on another property were affected. 14 In a case report, also involving OvHV-2 infection in a goat with typical acute MCF lesions, the other 19 conspecifics were unaffected. 47 These results show that OvHV-2 induced caprine MCF, manifesting as either GMF or more typical acute MCF disease, can occur in either individual or multiple goats.22,30
The cutaneous lesions observed in this case series had similar macroscopic characteristics between all 16 goats, and were consistent with previous reports of caprine GMF,10,45 and GMF in other ruminant animal species.4,11,24,28,44 Although reports of GMF in MCF-infected cattle are rare, other cutaneous lesions are relatively common as a part of the systemic disease process in classic vasculocentric MCF, and are increasingly prominent with greater chronicity.4,24 Cutaneous lesions seen with typical chronic MCF in cattle feature many of the same gross characteristics described in this caprine GMF case series, such as crusting, alopecia, and erosion.7,28 However, histologically the cutaneous lesions are characterized by lymphohistiocytic perivascular or lichenoid dermatitis, dermal arteritis, and epidermal necrosis.7,28
Some or all of the gross lesion characteristics observed in this case series can also be seen with a variety of other caprine dermatological conditions, including lice and mite infestations (as reported in goat 2), most significantly, chorioptic mange and sarcoptic mange; staphylococcal dermatitis; dermatophilosis; dermatophytosis; orf (also known as contagious pustular dermatitis, which is caused by a parapoxvirus); zinc-responsive dermatosis; photosensitization or sunburn; pemphigus foliaceus; and adverse cutaneous drug reaction. 50 However, each of these conditions has histologically distinguishing features, which were not observed in the goats included in this case series, and other than dermatophytosis and adverse cutaneous drug reaction, do not cause GMF. Dermatophytosis was not observed using fungal histochemical stains and thus was considered unlikely. Adverse cutaneous drug reaction is also considered unlikely in this case series, given that none of the goats were reported to have developed skin lesions subsequent to recent drug administration.
The distribution of cutaneous lesions in this case series is consistent with the previous UK case report of caprine GMF, as well as descriptions of GMF in Sika deer, white-tailed deer, bighorn sheep, and cattle.4,10,11,24,28,44 Exceptionally, the US case report of caprine GMF instead described cutaneous lesions localized to the ventrum. 45
In this case series, goats most often presented with localized skin disease that became progressively more severe and generalized. Three goats additionally were pyrexic prior to or at the onset of skin lesions and one goat also had respiratory signs. Necropsy of this goat revealed verminous pneumonia and no lesions suggestive of systemic MCF; thus, the respiratory signs were most likely not caused by OvHV-2 infection in this goat. Treatment history was available in 13/16 goats and showed that of the five goats treated with injectable corticosteroid, the majority had a favorable outcome. These included all three goats with partial resolution of cutaneous lesions (all MCFV PCR negative); one of two goats with complete resolution of cutaneous lesions (both MCFV PCR negative); and 1 of the 5 euthanized goats (goat 5, which was CpHV-3 PCR positive), which had initial improvement in cutaneous lesions that worsened as the corticosteroid dose was decreased. Goat 11 had complete resolution of cutaneous lesions with lime sulfur dips and topical zinc oxide; however, these treatments were also used in other goats, without the same outcome. In previous case reports of caprine GMF, 1 goat in the United Kingdom was euthanized after 4-weeks, having become depressed and hyporexic, whereas the goat in the United States was still alive more than a year after diagnosis, with no change in skin condition; no treatments had been administered since diagnosis.10,45 All but one of the previously reported cases of GMF in other ruminants infected with MCFVs have resulted in euthanasia, with the exception of a cow in New Zealand that had complete resolution of skin lesions after 14-weeks.4,11,24,28,44 Therefore, MCFV-associated GMF appears to have a spectrum of possible outcomes: euthanasia after weeks or months is most common, with partial recovery and a stable condition being less frequent, and full recovery rare. Caprine infection with OvHV-2 can result in a variety of manifestations ranging from subclinical infection to localized GMF to typical acute MCF with neurological signs and vasculitis. The factors influencing these different disease manifestations are unknown.
Of the 16 goats included in this case series, only 2 underwent full necropsies and neither had systemic MCF-like lesions. Although neither of the previously published case reports of caprine GMF involved necropsy,10,45 other ruminant species with GMF have undergone full postmortem evaluations. A bighorn sheep infected with the provisionally named OvHV-3 had no evidence of systemic MCF-like disease. 44 By contrast, all cervine GMF cases have also had systemic lesions typical of MCF, including multifocal granulomatous and lymphoproliferative arteritis in two Sika deer infected with CpHV-2; hemorrhagic enteropathy with multisystemic granulomatous infiltrates and vasculitis in another Sika deer infected with CpHV-2; and lymphoplasmacytic meningoencephalomyelitis, myocarditis, and retinitis in a white-tailed deer infected with CpHV-2.4,11,24 Altogether, the findings in this case series and those in previously published reports of GMF with MCFV infection indicate that both localized cutaneous disease and systemic disease with cutaneous manifestations are possible.
In all goats in this case series, GMF was characterized by infiltrating macrophages and lymphocytes, and infundibular regions were consistently affected. The deepest two regions of the hair follicle only had mural infiltrates in goats 2 and 5, which were infected with OvHV-2 and CpHV-3, respectively. Both goats were the only animals with severe GMF or severe dermatitis (Table 1). Furthermore, of the four OvHV-2 real-time PCR positive goats, goat 2 had the lowest cycle threshold value, indicating the presence of a larger quantity of viral DNA compared with the other 3 goats which instead had mild GMF. These results may suggest an increased likelihood of MCFV detection in caprine GMF cases with deep and severe folliculitis and dermatitis. Dermal infiltrates invariably featured macrophages and fewer lymphocytes, often mixed with eosinophils and/or neutrophils. Altogether, these histologic findings are largely consistent with previous individual reports of GMF in MCFV-infected goats and other ruminant animals.4,10,11,24,28,44,45
Arteritis is one of the classic lesions observed with sheep associated MCF in cattle and is often cited as being a near-constant feature in all cases of MCF, regardless of the species of MCFV or host involved. This dogma is challenged by the absence of vasculitis in all 16 goats in this case series; however, only two goats underwent necropsy and neither included histologic evaluation of locations most typically prone to developing vasculitis, such as the carotid rete and the pampiniform plexus. 30 In the MCF-associated GMF literature, one goat had no evidence of cutaneous vasculitis, while the other had focal deep dermal arteritis.10,45 In other ruminant species, one bighorn sheep had no vasculitis, one cow had mild cutaneous vasculitis, one white-tailed deer had arteritis involving the rete mirable and subcutis, and three sika deer had arteritis in locations other than the skin.4,11,24,28,44 These findings suggest that vasculitis is variably present in MCFV-infected ruminants with GMF, and when present it may occur infrequently, most often in noncutaneous locations.
The significance of CpHV-3 infection in goat 5, which had severe GMF, is uncertain. While CpHV-3 DNA was detected within skin samples that featured GMF, there have been several reports of subclinical infection in goats. However, the histologic features are consistent with other published reports of MCFV-induced GMF, and all other known differential diagnoses, other than the originally published diagnosis of presumed sterile eosinophilic and GMF and furunculosis, have been excluded. 37 GMF has not been previously reported in an MCFV-infected reservoir host, as goats are suggested as being for CpHV-3. Whether or not this is the case, classic vasculocentric MCF has been occasionally reported in reservoir hosts, e.g., OvHV-2 in sheep.5,34
In conclusion, the colocalization of OvHV-2 within GMF lesions in this case series supports the involvement of MCFVs in the pathogenesis of GMF. Although the precise relationship between OvHV-2 and GMF is yet to be defined, infection of follicular keratinocytes suggests the possibility of direct virus-induced cellular lysis and/or keratinocyte activation and subsequent immune-mediated destruction. Immune-mediated disease may persist despite an eventual decline in detectable levels of OvHV-2 DNA. Alternatively, or concurrently, injury directly or indirectly caused by virus-infected lymphocytes and/or macrophages is also possible. Macrophages are the main inflammatory cells present within GMF lesions and intralesional MNGCs are histiocytic in origin, rather than being keratinocyte syncytia, as had been previously suggested. Future studies should focus on characterizing lysis vs. latency-associated OvHV-2 gene expression profiles within GMF lesions, and more definitively determining infected cell types. The role of CpHV-3 in the pathogenesis of caprine GMF is uncertain and further investigation is required.
Supplemental Material
sj-xlsx-1-vet-10.1177_03009858231189303 – Supplemental material for Granulomatous mural folliculitis in 16 domestic goats: Infection with malignant catarrhal fever viruses and colocalization with ovine herpesvirus-2 using in situ hybridization
Supplemental material, sj-xlsx-1-vet-10.1177_03009858231189303 for Granulomatous mural folliculitis in 16 domestic goats: Infection with malignant catarrhal fever viruses and colocalization with ovine herpesvirus-2 using in situ hybridization by Thomas Westermann, Elena Alina Demeter, Diego G. Diel, Randall W. Renshaw, Melissa A. Laverack, Rhea S. Gerdes and Jeanine Peters-Kennedy in Veterinary Pathology
Supplemental Material
sj-xlsx-2-vet-10.1177_03009858231189303 – Supplemental material for Granulomatous mural folliculitis in 16 domestic goats: Infection with malignant catarrhal fever viruses and colocalization with ovine herpesvirus-2 using in situ hybridization
Supplemental material, sj-xlsx-2-vet-10.1177_03009858231189303 for Granulomatous mural folliculitis in 16 domestic goats: Infection with malignant catarrhal fever viruses and colocalization with ovine herpesvirus-2 using in situ hybridization by Thomas Westermann, Elena Alina Demeter, Diego G. Diel, Randall W. Renshaw, Melissa A. Laverack, Rhea S. Gerdes and Jeanine Peters-Kennedy in Veterinary Pathology
Footnotes
Acknowledgements
We gratefully acknowledge Ally A Butler Cornell University DVM Candidate Class of 2024 for assistance with clinical follow-up data, Dr Mason Jager for assistance with OvHV-2 ISH, Dr Danny W Scott for his review of the manuscript, and the Cornell University College of Veterinary Medicine AHDC histology and virology laboratory staff for their technical expertise.
Supplemental material for this article is available online.
Author Contributions
TWW, DGD, MAL, and RSG performed the experiments; TWW, JPK, EAD, DGD, and RWR contributed to the experimental design; TWW, JPK, and AED performed histologic evaluations; and the manuscript was written by TWW with contribution from the other authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
