Abstract
Strongyloides are small rhabditid nematodes primarily associated with enteric disease in a variety of animal species, including reptiles. Strongyloides spp life stages were associated with a disease outbreak in a large breeding colony of snakes. Multiple Pantherophis and Lampropeltis colubrids exhibited respiratory distress, anorexia, stomatitis, facial deformation, and waning body condition that resulted in death or necessitated euthanasia. Postmortem examinations of 13 snakes revealed epithelial hyperplasia and inflammation of the alimentary and respiratory tracts associated with varying numbers of adult and larval nematodes and embryonated or larvated ova. In a subset of snakes, aberrant nematode migration was also observed in the eye, genitourinary system, coelom, and vasculature. Histomorphology and gross examination of parasitic adult female nematodes from host tissues were consistent with a Strongyloides spp. Sedimented fecal material from 101/160 (63%) snakes housed in the affected facility was positive for nematodes and/or larvated ova. Polymerase chain reaction amplification and sequencing of portions of the 18S and 28S ribosomal ribonucleic acid (RNA) genes and the internal transcribed spacer region of adult female parasites and positive fecal samples supported the diagnosis of strongyloidiasis. Strongyloides spp possess a unique life cycle capable of alternating between parasitic (homogonic) and free-living (heterogonic) stages, resulting in the production of directly infective larvae. Commonly utilized husbandry practices in reptile collections can amplify the numbers of infective larvae generated in the captive environment, increasing the risk for rhabditid hyperinfections. This report documents morbidity, mortality, and non-enteric disease manifestations due to Strongyloides hyperinfections in a captive colubrid snake colony.
Encompassing more than 1800 species, the Colubridae is the largest and most diverse snake family. 13 The phenotypic diversity and often innocuous nature of colubrid snakes make them attractive pets with reptile enthusiasts. Due to their increasing popularity as household companions, there is a need for intensive captive reproduction to match consumer demand. 28 As with other animal production sectors, intensive production of reptiles can be associated with challenges, including disease outbreak and control.25,31,32 Parasitic disease is important in captive reptile collections due to the frequency with which infections may go undetected and the effects that heavy parasitism can have on animal health.5,43
Strongyloides spp nematodes, colloquially known as threadworms, are members of the order Rhabditida. Strongyloides spp have a direct life cycle divided into heterogonic and homogonic stages. The heterogonic life cycle involves free-living adults that reproduce sexually in the environment. Eggs laid by free-living females develop into third-stage larvae capable of direct infection of hosts through percutaneous or oral routes.29,38 Only a single free-living adult generation exists; all progeny develop into infective third-stage larvae. 36 Insects (notably cockroaches and flies) may serve as reservoirs. 40 Larvae enter the circulation and develop in the pulmonary system, followed by migration to the intestine to complete their maturation. 38 The prepatent period is short; S. ophidiae infection is detectable after 7 days. 16 The homogonic life stage involves parthenogenetic reproduction by parasitic adult female nematodes within the host, generating both male and female ova. 36 Parthenogenetic females deposit eggs in the mucosa of the small intestine.29,33 Ova and first-stage larvae pass in feces where males develop into free-living adults, and females either develop into free-living adults or infective third-stage larvae.
Strongyloides spp cause disease in a wide range of animal species, including reptiles, amphibians, birds, companion animals, nonhuman primates, and livestock.14,22,29,36 Common species of veterinary importance include S. stercoralis in canids; S. felis, S. planiceps, and S. tumefaciens in felids; S. papillosus in ruminants; S. ransomi in pigs; and S. westeri in equids. 29 Although improvements in animal husbandry and increased availability of prophylactic anthelmintic treatments have relegated Strongyloides spp to a pathogen of minor importance for much of veterinary medicine, these parasites remain clinically important in reptiles and amphibians.29,36,38,42 Of the 60 described species of Strongyloides, 8 have been documented in reptiles. 16 In snakes, 5 species of Strongyloides have been documented, including S. gulae, S. serpentis, S. ophidiae, S. natricis, and S. mirzai.14,19,27,34,41
Most Strongyloides infections are not marked by signs of infection and self-limiting, but serious disease and death can occur in the young or immunocompromised.29,36 Clinical signs of infection in animals include diarrhea and bronchopneumonia associated with mature and developing parasite life stages, respectively. 29 In piglets, migration of larvae in the lungs has been associated with alveolar hemorrhage, interstitial inflammation, and altered respiration. 33 Enteric infection can result in villous atrophy, enterocyte hyperplasia, and inflammation of the lamina propria with ulceration or squamous metaplasia of the mucosal epithelium. 33 In reptiles, clinical signs associated with Strongyloides infections are respiratory and/or enteric in nature. Open-mouthed breathing, respiratory distress, and pneumonia may be seen due to migrating Strongyloides larvae. 38 These signs may be indistinguishable from those caused by Rhabdias spp, another rhabditid that inhabits the respiratory system. Enteric strongyloidiasis causes anorexia, weight loss, lethargy, and diarrhea.9,10,38,41 Fluid and protein loss and electrolyte abnormalities secondary to enteric disease are thought to contribute to snake mortality. 9 Secondary bacterial infections have also been described and, in some cases, have been the proximate cause of death in infected snakes.5,10,34,41
Parasites are important causes of morbidity and mortality in both wild and captive reptiles.11,20,23 Gastrointestinal parasitism is common in captive reptiles. In a parasitological survey of snakes from German households and zoos, Strongyloides spp and Rhabdias spp were commonly identified and second only in prevalence to another enteric nematode, Kalicephalus spp. 5 Although Strongyloides spp are well-recognized intestinal pathogens of reptiles, published cases of infection in snakes are sporadic and descriptions of pathologic changes associated with these parasites are lacking. Even fewer details exist regarding non-enteric manifestations of strongyloidiasis or parasite histomorphology. This report characterizes the lesions caused by Strongyloides spp parasites in a group of captive colubrid snakes that initially presented for respiratory disease.
Material and Methods
Ethics Statement
All postmortem examinations were performed as part of a diagnostic investigation and, as such, ethical review and approval was not required for this study. Euthanasia, when necessary due to quality of life concerns, was performed using American Veterinary Medical Association–approved chemical means by the Zoological Medicine Department at the College of Veterinary Medicine at the University of Florida. 1 Fecal sample collection through cloacal flushing was approved by Institutional Animal Care and Use Committee at the University of Florida (Protocol no. 201910698).
Study Population
The investigated population of snakes was part of a large commercial colony of mixed species maintained for the purpose of breeding and supply to the pet trade. Snakes were housed in multiple buildings on the premises and primarily organized by family (Pythonidae, Boidae, and Colubridae). For the colubrid portion of the colony, the majority of the breeding stock was of captive bred and born origin; however, previous management practices permitted periodic introduction of wild-caught reptiles into the captive collection. Snakes were housed individually in a rack-style system, with vented plastic tubs maintained in climate-controlled buildings, with up to 300 reptiles per building. Cypress mulch substrate was used to line the tubs and was stored outdoors on pallets under covered awnings. Feeding was performed regularly with live rodents that, if not consumed within a predetermined time period, would be transferred to feed a snake in a neighboring tub. Reproductive practices consisted of comingling one male snake with one female snake and, in a single breeding season, it was common for individual males to be rotated among multiple females.
Postmortem Examination and Histopathology
Disease investigation was prompted in the late fall of 2018 when numerous snakes within the collection were observed with nonspecific clinical signs of respiratory disease, including dyspnea and open-mouthed breathing. Over 7 months, 13 adult snakes were submitted for postmortem examination, including 8 corn snakes (Pantherophis guttatus), 3 California king snakes (Lampropeltis getula californiae), and 2 Pueblan milk snakes (L. triangulum campbelli; Table 1). Individuals died or were euthanized prior to submission following observation of one or more of the following signs: dyspnea, tachypnea, stomatitis, facial deformation with oral malocclusion, anorexia, and/or waning body condition. Fresh tissue samples of oral mucosa, esophagus, trachea, lung, liver, kidney, spleen, and heart were archived at −80° C. Representative samples of all visceral organs were preserved in 10% neutral-buffered formalin for 24 hours prior to processing. In addition, formalin-fixed heads were decalcified in ethylenediaminetetraacetic acid solution for approximately 120 hours prior to obtaining serial sections through the nares, eyes, and calvaria for histologic processing. The mandibles were disarticulated from the quadrate bones. A single longitudinal section spanning the glottis, glossal sheath, tongue, and gular skin was sampled for histopathology. Paraffin-embedded tissues were routinely processed, sectioned at thicknesses of 5 µm, and stained with hematoxylin and eosin.
Microscopic distribution of Strongyloides-associated lesions in necropsied colubrid snakes.
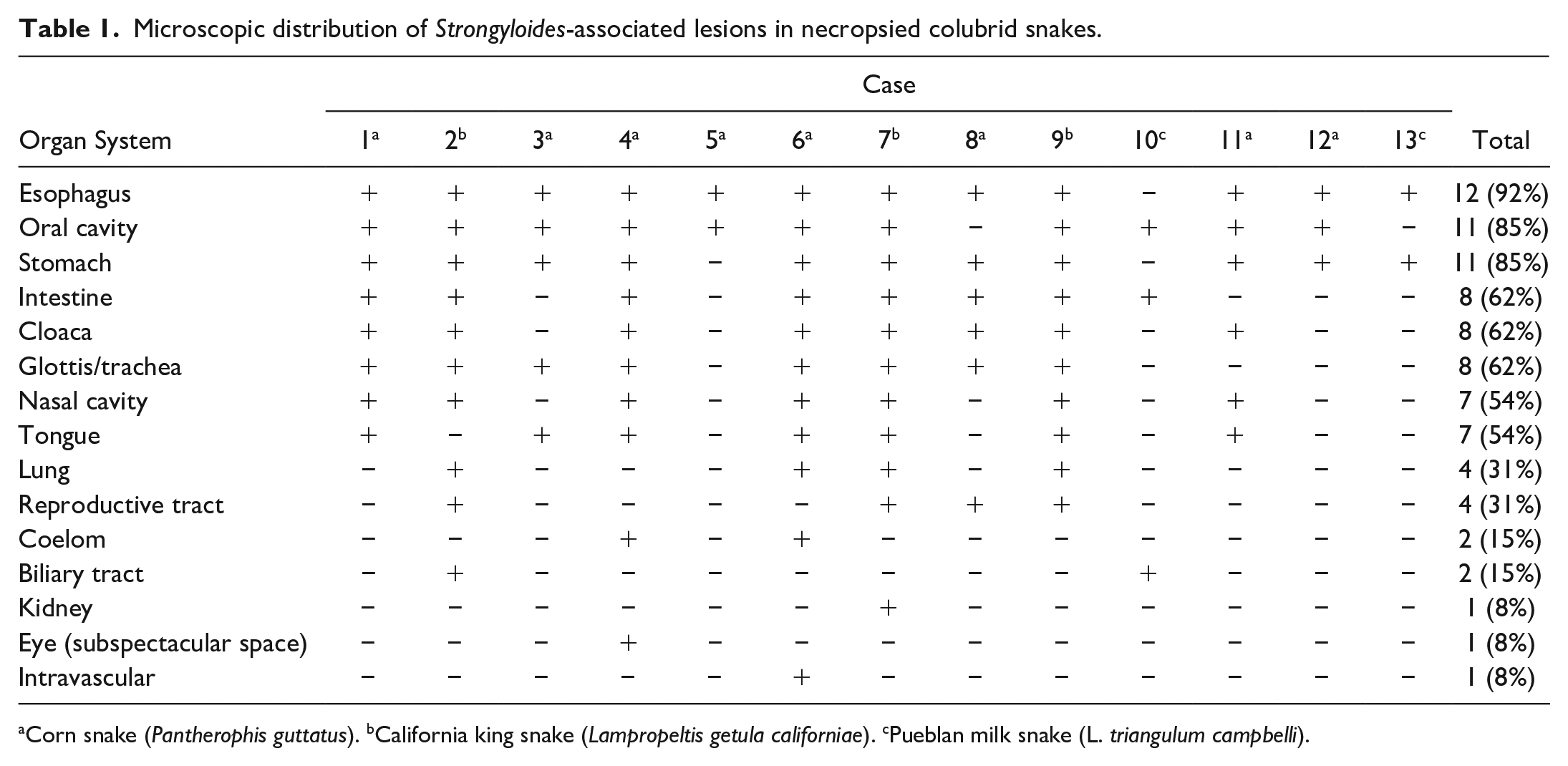
Corn snake (Pantherophis guttatus). bCalifornia king snake (Lampropeltis getula californiae). cPueblan milk snake (L. triangulum campbelli).
Cloacal Flushing for Fecal Collection
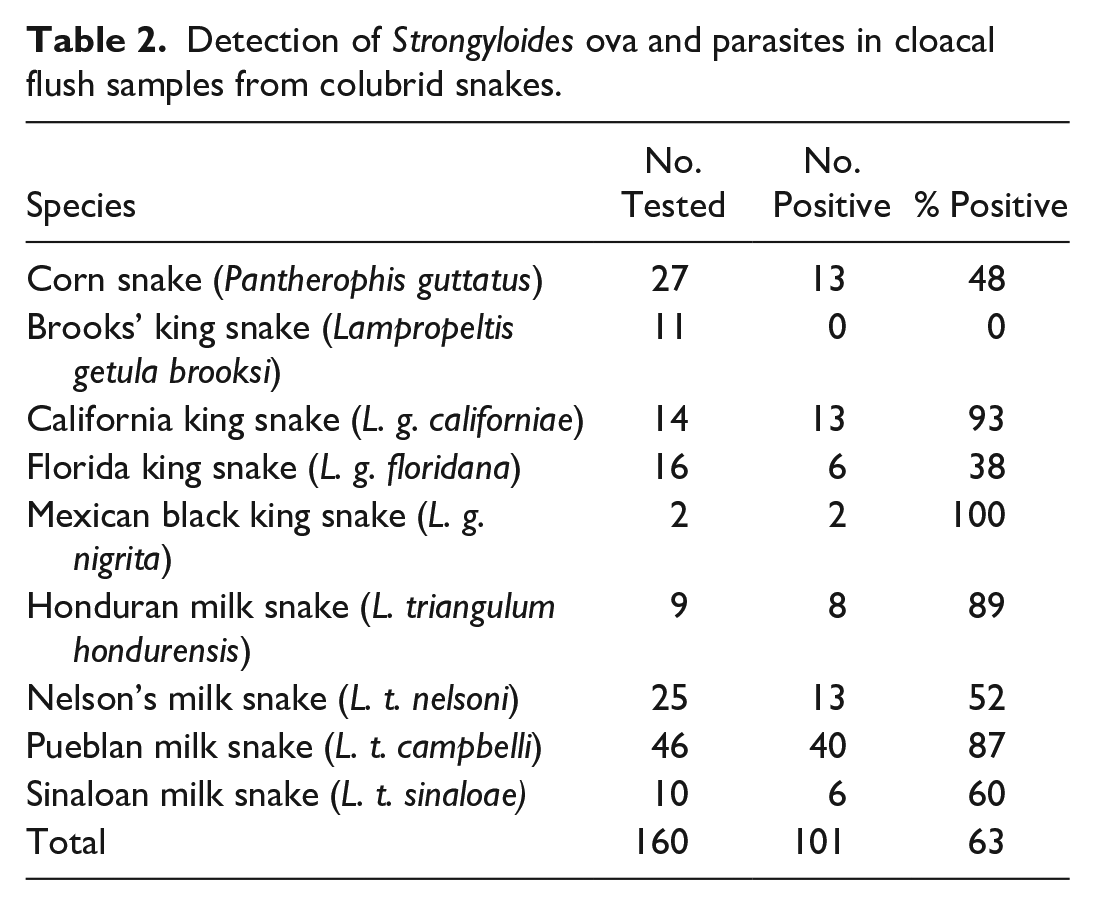
In June 2019, cloacal flushes were obtained from 160 colubrids housed in the same building as the index cases, representing 9 species: corn snake (P. guttatus; N = 27), Brooks’ king snake (L. g. brooksi; N = 11), California king snake (L. g. californiae; N = 14), Florida king snake (L. g. floridana; N = 16), Mexican black king snake (L. g. nigrita; N = 2), Honduran milk snake (L. t. hondurensis; N = 9), Nelson’s milk snake (L. t. nelsoni; N = 25), Pueblan milk snake (L. t. campbelli; N = 46), and Sinaloan milk snake (L. t. sinaloae; N = 10; Table 2). Cloacal flushes were collected using a sterile 16-French catheter flushed with 5 mL sterile phosphate buffered saline. Substrate samples from dirty cages (N = 6) and clean, unused bags (N = 4) were also collected in conjunction with the cloacal washes. Samples were stored in 15 mL conical tubes and preserved at 4° C in 70% ethanol.
Detection of Strongyloides ova and parasites in cloacal flush samples from colubrid snakes.
Parasitological Examination
Samples of fresh or frozen oral mucosa and lung from 2 deceased snakes (cases 2 and 4, Table 1) were examined for the presence of parasites. Cloacal flush fecal samples and substrate samples were evaluated by simple sedimentation methods as previously reported with modifications. 44 Modifications included the use of a household liquid dish detergent solution (2 drops of dish detergent in 500 mL water), which was added to the feces following ethanol decanting. The resulting fecal-detergent solution was mixed with a sterile pipette and centrifuged at 1500 rpm for 10 minutes. Pelleted sediments were analyzed for the presence of parasites under a compound microscope. Dirty and clean substrate samples were also evaluated for free-living parasite life stages by sedimentation as described above. Recovered adult and larval nematodes were preserved in 70% ethanol, cleared with lactophenol, and examined by compound and dissecting microscopy.
Parasite Molecular Characterization
Nucleic acids were extracted from affected tissues (oral mucosa or esophagus) of 2 snakes (cases 2 and 4, respectively; Table 1) or fecal sediment from 2 snakes, using a DNeasy kit (Qiagen, Valencia, California) per manufacturer’s recommendations. Portions of ribosomal ribonucleic acid (RNA) genes and internal transcribed spacers, including the ITS1/5.8S/ITS2 region (~700 base pairs [bp]), 4 the 18S gene (~900 bp), 3 and the 28S gene (~300 bp), 30 were amplified by conventional polymerase chain reaction (PCR) using previously described primers and conditions. The PCR products were resolved on a 1.5% agarose gel, and bands of the expected size were excised and purified using a QIAquick gel extraction kit (Qiagen, Valencia, California). Products were commercially sequenced bidirectionally (Genewiz, South Plainfield, New Jersey); contigs were assembled and primer sequences were trimmed using commercial software (Geneious 11.1, Biomatters, Inc, Auckland, New Zealand). The determined sequences were compared with known nematode sequences using the NCBI Basic Local Alignment Search Tool. 12
Results
Gross Postmortem Examination
Examined snakes were consistently in thin body condition with scant coelomic adipose stores and dark brown, atrophic livers. The most commonly observed lesion was irregular thickening and hyperemia of the oral mucosa, with distortion of the glottis and accumulation of copious mucus in the oral cavity (Fig. 1a). In 3/13 (23%) snakes, friable, tan oral masses measuring up to 2 × 1 × 1 cm surrounded the tongue and displaced the glottis. Oral swelling was often associated with dentary (mandibular) deformation and malocclusion. Gastrointestinal lesions were frequently distributed along the proximal esophagus, stomach, and small intestine. The esophageal mucosa was thickened and covered by tan, granular material. Affected stomachs contained increased amounts of intraluminal mucus. There was variable small intestinal distension and mural thickening, with accumulation of mucoid to pasty material. Gross lesions of the large intestine and cloaca were less commonly observed but appeared similar to the small intestine when present.
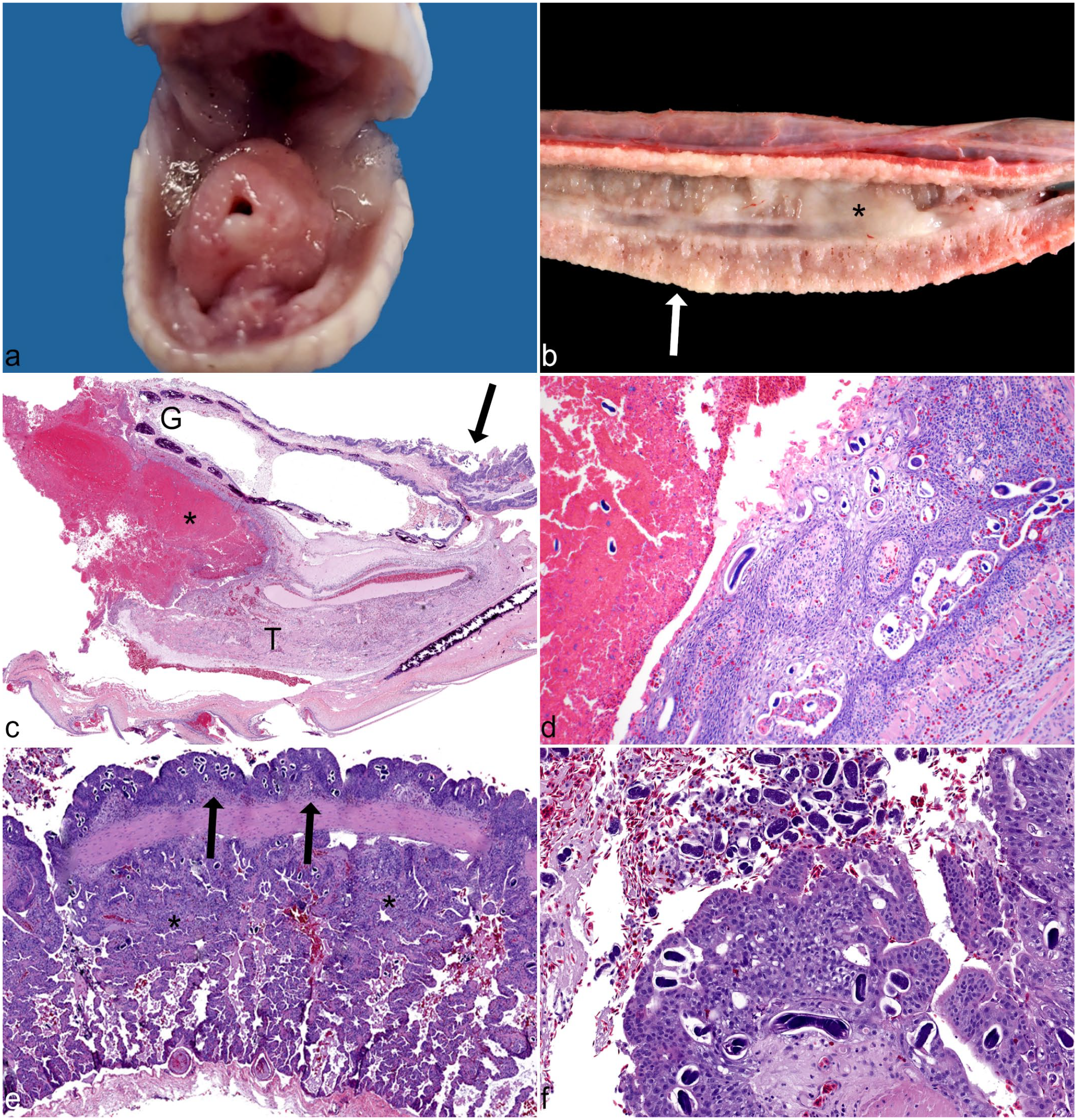
Strongyloidiasis, oral cavity and lung, California king snake (L. g. californiae). (a) Oral cavity. Diffuse mucosal proliferation and hyperemia of the mucosa with distortion and elevation of the glottis and mucus accumulation. (b) Lung. Stromal thickening and pallor of the pulmonary parenchyma (arrow) with abundant thick mucus occupying the lumen of the vorbronchus (*). (c) Oral cavity. Longitudinal section through the mandible showing displacement of the glottis (G) and tongue (T) by a large coagulum of granulocytic debris (*) with diffuse mucosal epithelial hyperplasia (arrow). Hematoxylin and eosin (HE). (d) Oral cavity. Higher magnification of granulocytic coagulum and junction with hyperplastic glossal sheath and many nematode life stages embedded in granulocytic debris and adjacent to the mucosa. HE. (e) Lung. Diffuse respiratory epithelial hyperplasia with compressed faveolar spaces (*), edema, hemorrhage, and many embedded nematodes (arrows). HE. (f) Lung. Higher magnification of nematodes (adults and ova) embedded in hyperplastic epithelium and free in the vorbronchus, with hemorrhage and edema. HE.
Respiratory disease was the predominant finding in 4/13 (31%) snakes, with increased pulmonary pallor and widespread stromal thickening (Fig. 1b). Faveolar spaces were indistinct, with abundant catarrhal exudate occluding the vorbronchus. In 2/13 (15%) snakes, pinpoint yellow foci were noted in the pulmonary parenchyma and pleura. Nematode larvae were observed in preliminary cytologic squash preparations of pulmonary nodules from 1 snake (case 7, Table 1). Unilateral, cloudy, red discoloration of the spectacle and buphthalmia was observed in a single snake (case 4).
Histopathology
Oral masses were formed by large coagula of degenerate granulocytes and necrotic debris, partially encompassed by mucosal epithelial hyperplasia of the glossal sheath with displacement of the tongue and glottis (Fig. 1c). Many nematode life stages, including adult females, larvae, and ova (see section “Parasite Histomorphology”), were observed within the granulocytic debris and adjacent hyperplastic mucosa (Fig. 1d). Nematode life stages were associated with granulocytic and lymphoplasmacytic inflammation of the tongue, oral mucosa, proximal trachea, and esophagus. Of the 11 snakes with oral cavity lesions, 7 exhibited varying degrees of necrotizing glossitis with hypereosinophilic and vacuolated glossal myofibers, sarcoplasmic fragmentation, internalization of nuclei, and nuclear rowing suggestive of early myofiber regeneration.
In the lungs, there was epithelial hyperplasia of the terminal septa, resulting in a papillated appearance of the epithelium bordering the central lumen. Faveoli were diffusely lined by proliferating type II pneumocytes with narrowing and compression of faveolar spaces (Fig. 1e). The pulmonary interstitium was often expanded by granulocytes and lymphocytes. Numerous nematode life stages were embedded in the pulmonary epithelium and free in the lumen of the vorbronchus and faveoli, with associated hemorrhage, edema, and inflammation (Fig. 1f). No parasites were observed in the examined sections of 3/13 (23%) snakes although microscopic changes consistent with proliferative pneumonia were present.
Pathology of the alimentary tract and cloaca consisted of lymphocytic and granulocytic inflammation and mucosal epithelial hyperplasia associated with luminal and intramucosal nematode life stages. In the stomach, nematodes were associated with glandular loss, lymphocytic to granulocytic gastritis, and segmental mucosal epithelial hyperplasia, attenuation, and erosion. Occasionally, the esophagus and stomach contained abundant mucus with entrapped necrotic debris, bacteria, degenerate granulocytes, and nematode life stages.
In one snake (case 6), nematode larvae were present in blood vessels, cardiac atria, and coelomic cavity, with associated regional coelomitis. This snake also had histologic lesions supportive of septicemia, including disseminated fibrin thrombi, intravascular bacteria, and embolic hepatitis and pneumonia. Aberrant Strongyloides migration was identified in 9/13 (69%) snakes with nematode life stages in one or more of the following tissues: nares, vomeronasal organ, periodontium, semicircular canals, oviduct, deferent duct, and biliary tract. Necrosis and bacteria often accompanied the presence of nematodes in aberrant locations, consistent with secondary bacterial infection. One snake with gross evidence of ocular disease (case 4) had granulocytic inflammation, edema, and neovascularization of the spectacular stroma, with accumulation of granulocytes and nematode larvae in the subspectacular space, periorbital connective tissue, and orbital gland.
Parasite Histomorphology
As expected, given the homogonic life cycle of Strongyloides spp, only adult female nematodes were observed in host tissues. Parasitic females were 33 to 40 µm in diameter, with an intestinal tract lined by simple epithelium and coiled paired uteri containing uninucleate ova (Fig. 2a). The paired reproductive tract is a distinctive histologic characteristic of Strongyloides spp that permitted differentiation of these parasites from other rhabditid nematodes. In transverse sections, adult females had a 2-µm wide, eosinophilic, striated cuticle with triradiate esophagus, platymyarian-meromyarian musculature, pseudocoelomic cavity, and flat vacuolated lateral cords (Fig. 2b). In longitudinal sections, the adult esophagus was filariform and lined by a thin, slightly refractile cuticle. Deposited ova ranged from 20 to 30 µm in diameter and were embryonated or contained a single folded or coiled larva (Fig. 2c, d).
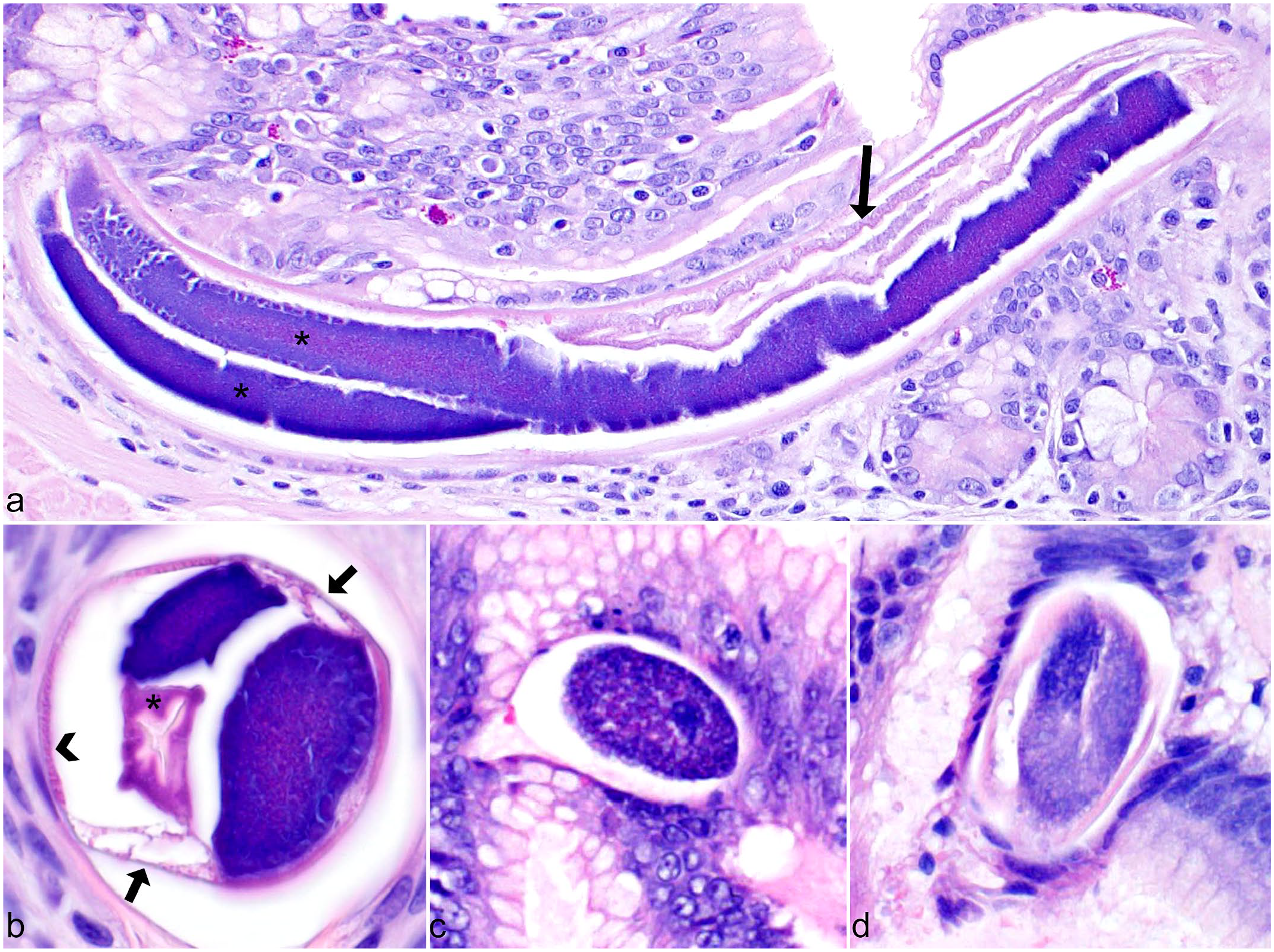
Parasite histomorphology, Strongyloides spp. Hematoxylin and eosin. (a) Parthenogenetic adult female in longitudinal section showing partially coiled paired uteri (*) containing ova and intestinal tract lined by simple epithelium (arrow). (b) Adult nematode in transverse section with paired uteri, vacuolated lateral cords (arrows), triradiate esophagus (*), and platymyarian musculature (arrowhead). (c) Embryonated ovum embedded in mucosa. (d) Embedded ovum containing folded larva.
Parasitological Examination
Multiple parasite life stages were recovered from examined tissues, cloacal flushes, and substrate samples. Seven parasitic adult female nematodes were recovered from the lung of one infected snake (case 2; Fig. 3a). Measurements, including length, width, esophageal length, mouth to vulva length, and tail length, were determined for each nematode (Supplemental Table S1). Ranges and means were determined (Supplemental Table S1; Table 3) and measurements were compared with other known snake Strongyloides spp. Of cloacal flush fecal samples, 101/160 (63%) were positive for larvated ova, intact adults, and/or fragmented adults, including segments of uterus containing embryonated ova (Table 2). Ova were 64.1 (+/− 1.5) µm long and 32.5 (+/− 0.9) µm wide (Fig. 3b). Adult nematode morphology in fecal sediments was identical to that of nematodes recovered from tissues. Examination of substrate samples yielded infective third-stage larvae, with characteristic forked tails from used substrate only (Fig. 3c); no parasites were present in clean substrate samples.
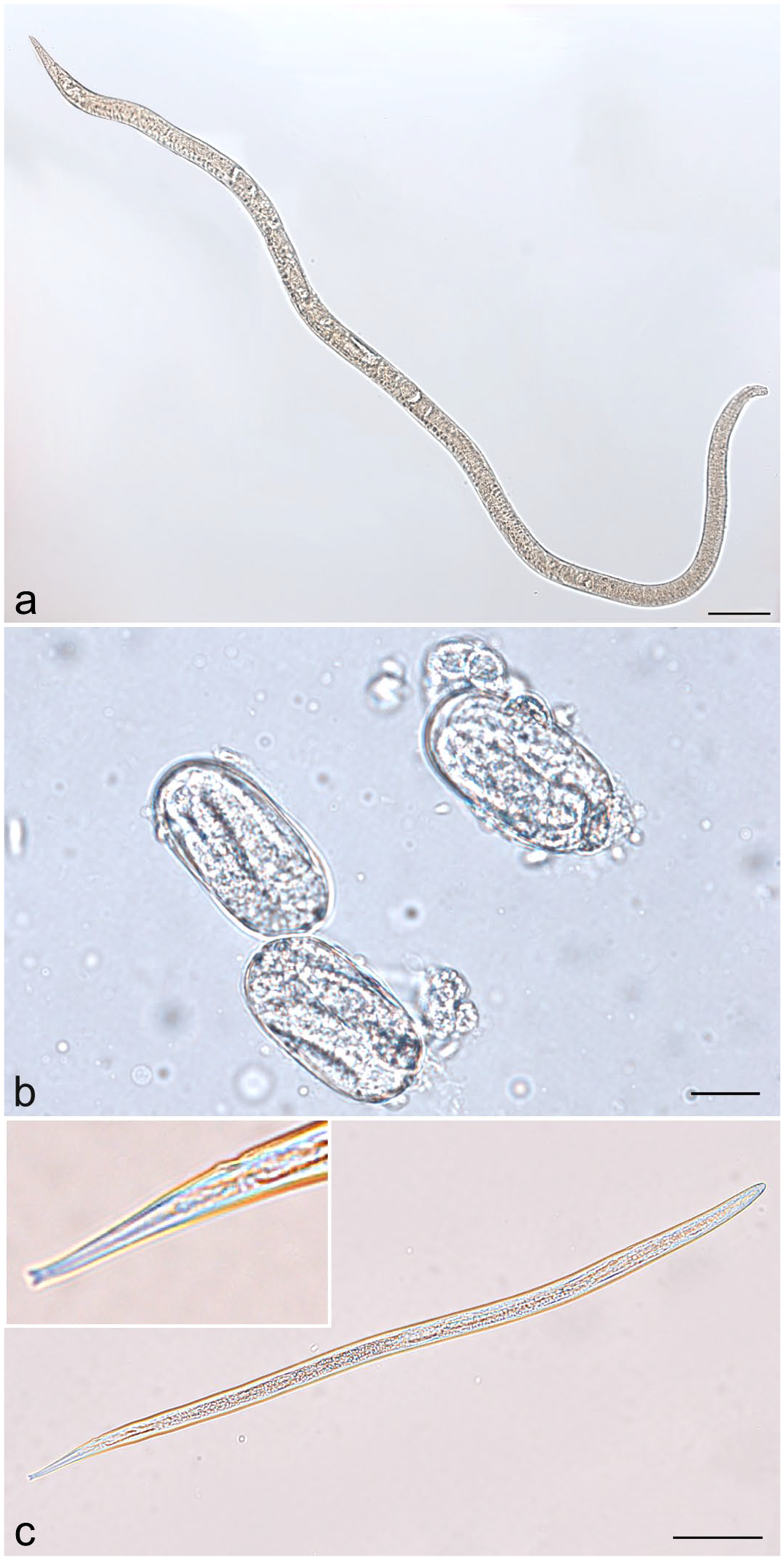
Morphology of Strongyloides life stages identified using light microscopy. (a) Parasitic adult female nematode from the lung of a California king snake (L. g. californiae). Scale bar = 100 µm. (b) Larvated ova from cloacal flush of a California king snake. Scale bar = 2 µm. (c) Infective third-stage larva with characteristic forked tail (inset) from used cypress mulch substrate. Scale bar = 50 µm.
Body measurements of parasitic Strongyloides females recovered from the lung of an affected snake and ova measurements from fecal samples compared with analogous measurements reported for other snake Strongyloides spp.
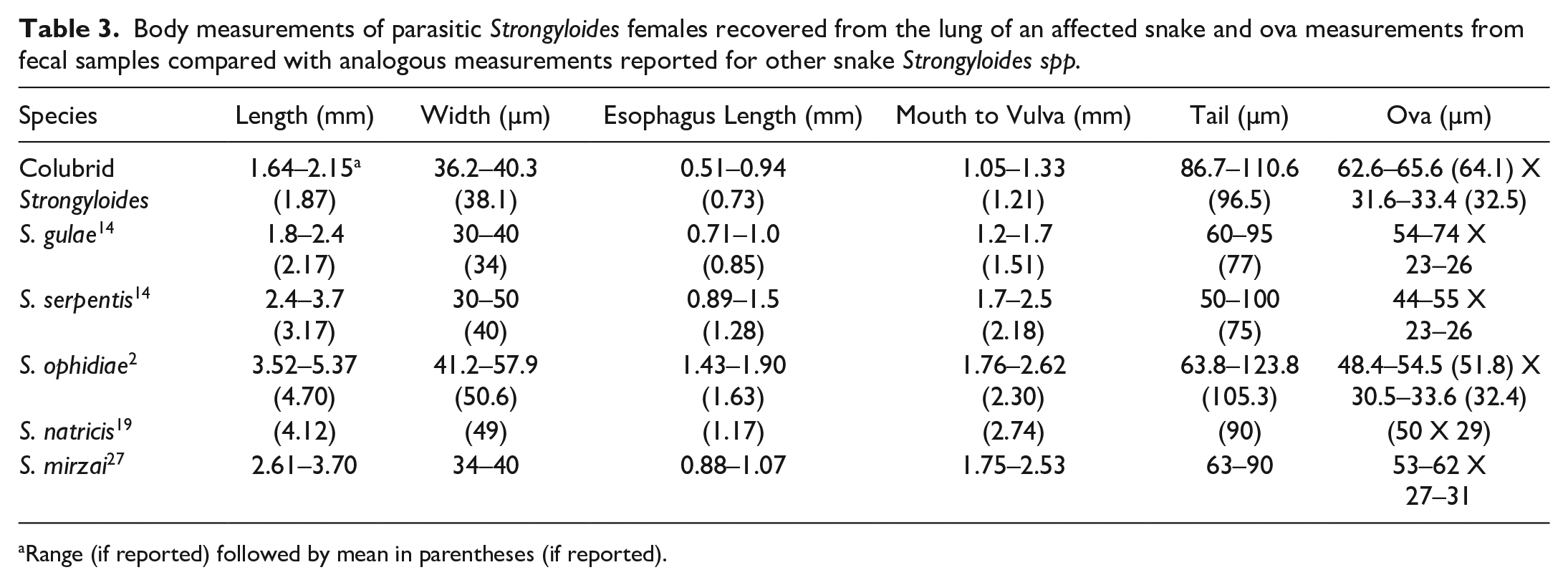
Range (if reported) followed by mean in parentheses (if reported).
Parasite Molecular Characterization
Samples of affected oral mucosa from California king snake case 2 (L. g. californiae), affected esophagus from corn snake case 4 (P. guttatus), and a fecal sediment from a Nelson’s milk snake (L. t. nelsoni) were all positive by PCR for nematode 18S, 28S, and ITS1/5.8S/ITS2 targets. An 828-bp portion of the 18S gene was amplified from each sample (deposited as Genbank accession OQ107214). The 18S sequence from each sample was 100% identical to each other and was most similar to Strongyloides mirzai (98.4% identity; Genbank AB453311) collected from a Japanese pit viper, Protobothrops flavoviridis. 6 The only other snake Strongyloides spp for which there was reference sequence, S. ophidiae (EU287935) collected from a false coral snake (Oxyrhopus guibei) in Brazil, was 97.5% identical over a smaller portion (286 bp) of the amplified target. 2 A 293-bp portion of the 28S gene was also amplified from each sample (deposited as Genbank accession OQ107213). As for the 18S sequences, the sequences from all samples were identical to each other. The 28S sequence was most similar to S. papillosus (94.9%; AB923882), a parasite of cattle and small ruminants. Finally, a 559-bp portion of the ITS1/5.8S/ITS2 region was amplified from each sample (deposited as Genbank accession OQ107246); the sequence from this region was most similar to Strongyloides callosciureus (81.1%; AB272229) described in free-ranging Formosan squirrels (Callosciurus erythraeus thaiwanensis). 26 No reference sequence was available for the 28S and ITS1/5.8S/ITS2 regions of S. mirzai. No reference sequence for any genetic target was available for the other 3 reported snake Strongyloides spp, S. gulae, S. serpentis, and S. natricis.
Discussion
In addition to enteric disease typically associated with Strongyloides spp infection, we present a cohort of colubrid snakes with parasite-associated pathology of the upper alimentary and respiratory tracts. Esophageal involvement was documented most often (12/13 snakes), followed by the oral cavity and/or stomach (11/13 snakes). Disease investigation was initially prompted by snakes with clinical signs of respiratory disease, and proliferative pneumonia secondary to Strongyloides spp infection was attributed as the proximate cause of death in 4 snakes. Aberrant Strongyloides spp migration to the eye (1/13 snakes) and reproductive tract (4/13 snakes) were considered unique presentations that, to our knowledge, have not been previously described. Preliminary molecular characterization of recovered parasites was suggestive of a novel Strongyloides species although further comparison with existing Strongyloides species is needed.
Of the 5 Strongyloides species previously reported in snakes (S. gulae, S. serpentis, S. ophidiae, S. natricis, and S. mirzai), only S. ophidiae and S. mirzai have publicly available sequences. Comparison of the 18S sequence of the Strongyloides spp found in these snakes with S. mirzai (AB453311; 98.4% nucleotide identity over 810 bp) and S. ophidiae (EU287935; 97.5% nucleotide identity over 286 bp) shows genetic variation consistent with this parasite likely representing a distinct species. For example, S. stercoralis (AB923888) exhibits 99.4% to 99.6% identity to isolates of S. procyonis (AB205054 and AB272234) and 97.6% identity to S. cebus (AB272236) and S. ratti (AB923889). Unfortunately, no sequence was available for comparison with S. gulae, S. natricis, and S. serpentis; as such, it was necessary to compare observed and reported parasite morphologies. Although there was considerable overlap of parasite length, width, and esophagus length for the Strongyloides spp found in these snakes and those reported for S. gulae, the mouth to vulva length of the parasites in these snakes was shorter and the tail lengths were longer. 14 For ova, although the lengths of eggs of both S. gulae and the eggs collected from cloacal washes of our snakes were overlapping, the reported width of S. gulae ova is considerably narrower than we observed in eggs from the snakes in this collection.
There are substantial geographic differences between the documented occurrences of S. mirzai (India), S. natricis (Spain), and S. ophidiae (Brazil), and the cases of strongyloidiasis reported here, although international transport by the reptile trade has been documented for other reptile parasites.17,35 Both S. gulae and S. serpentis were reported in free-ranging snakes from Louisiana, United States.2,14,16,19,27 The captive snakes from this collection resided in central Florida, United States and, while range maps for either S. gulae or S. serpentis have not been determined, it is possible those parasites naturally extend into Florida.
It remains unclear whether the parasite reported herein represents S. gulae or a novel Strongyloides species. Although vouchered museum reference specimens of S. gulae and S. serpentis do exist, they have been maintained in formalin, excluding the potential of genetic characterization of the historical samples. The apparent lack of Strongyloides reference specimens stored in a way to preserve parasite nucleic acid is problematic. Standards for parasite specimen vouchering to meet the needs of morphologic and molecular parasitology are warranted.
Specific instances of ophidian strongyloidiasis are infrequently reported. Infections with unspeciated Strongyloides spp have been documented in a variety of species in captivity, including the speckled king snake (L. g. holbrooki), yellow rat snake (Elaphe obsoleta quadrivittata), gray rat snake (E. o. obsoleta), and reticulated python (Malayopython reticulatus). Each case was variably associated with episodic coughing, anorexia, diarrhea, and weight loss.9,10 Infection with S. mirzai was identified in a single report of a green tree python (Morelia viridis) that died following a bout of anorexia, lethargy, and weight loss. 41 Necropsy revealed proliferative diphtheritic enteritis of the proximal duodenum associated with numerous adult nematodes embedded within the diphtheritic membrane. The nematodes were identified as S. mirzai based on morphological characteristics only, and the authors noted that observed parasite morphometrics were inconsistent with previously published reports of S. mirzai in snakes.27,41 In snakes naturally and experimentally infected with S. ophidiae, the most consistent postmortem finding was catarrhal enteritis of the duodenal intestinal segment. 16 In all of the snakes examined in this report, severe epithelial hyperplasia of the oral cavity, respiratory system, and alimentary tract with granulocytic and lymphocytic inflammation was a consistent response to the parasites. The wide distribution of parasites observed in colubrids in this study is different than previous reports of snake strongyloidiasis, particularly as it relates to respiratory involvement. As such, rhabditid nematodes other than Rhabdias spp should also be considered in cases of verminous respiratory disease in snakes.
Aberrant migration of Strongyloides spp in snakes has also been described. In a case report of a captive Burmese python (Python bivittatus), Strongyloides spp most closely resembling S. gulae identified in the kidneys and ureters were associated with interstitial nephritis and bilateral ureteral obstruction. 34 In our study, larval nematodes were seen in renal tubules in a single California king snake (L.g. californiae, case 7; Table 1) associated with tubulointerstitial nephritis. Interestingly, Strongyloides spp were seen in the reproductive tracts of 4 snakes, including 3 California king snakes (cases 2, 7, and 9) and a single corn snake (P. guttatus, case 8). Strongyloides life stages were noted in the oviducts of 2 females (cases 2 and 9). In 2 male California king snakes (cases 7 and 8), parasites were observed in the deferent ducts. To the best of the authors’ knowledge, this is the first report of parasitism of the reptile reproductive tract by Strongyloides spp. Given that all snakes with parasites in the reproductive tract and kidney also had parasites in the cloaca, an ascending infection is considered most likely. However, with the intensive reproductive practices used in this colony, it is possible that sexual transmission of the parasites may have contributed to the spread of this parasite in the collection. Finally, Serpentirhabdias dubielzigi, another nematode in the order Rhabditida, has been associated with subspectacular nematodiasis in ball pythons (Python regius).7,15 This presentation mirrors the ocular disease observed in a corn snake (case 4) in this colony, highlighting a similar pattern of aberrant parasite migration through subcutaneous tissues of the head and subspectacular space. Again, to the authors’ knowledge, this is the first report of a Strongyloides spp in the ocular tissues of a snake.
Clinical signs and lesions associated with Strongyloides and Rhabdias infections in reptiles and amphibians are largely dependent on parasite load, host age, stress, nutritional condition, stocking density, comorbidities, and the occurrence of opportunistic/secondary infections.5,18,21 Reptiles and amphibians may be housed on substrate suitable for nematode proliferation. In cases reported here, snakes were maintained on a cypress mulch substrate capable of supporting the homogonic life cycle, as evidenced by identification of an infective third-stage larva in used substrate. As substrate was primarily spot-cleaned and only sporadically changed in toto, the environment could serve as a continuous source of infective third-stage larvae and likely facilitated the hyperinfections noted in examined animals. This collection also historically would introduce animals directly from the wild to diversify genetics, which may have permitted introduction of a parasite from a free-ranging reptile into the captive collection. Moreover, routine husbandry practices likely facilitated the spread of infective larvae between individuals, notably at times of animal comingling during breeding and transfer of uneaten food items between containers. Snakes were housed in tubs in a rack-style system. Such tubs often limit snake movement, which can have negative impacts on animal health, both from stress and lack of opportunities to get away from feces.8,39 Finally, a few comorbidities, including biliary coccidiosis (N = 2), colonic adenocarcinoma (N = 2), and intestinal and biliary cryptosporidiosis (N = 2), were identified, which may have contributed to the severity of parasitic infection in some snakes.
As multiple snakes initially presented with respiratory signs, it was important to consider other primary or contributing respiratory pathogens, such as ophidian serpentovirus (nidovirus), paramyxovirus, orthoreovirus, bacterial or fungal pneumonia, or other pulmonary parasites in these reptiles. No viral inclusions, epithelial syncytia, nor epithelial necrosis were observed in any of the snakes, as may be expected with paramyxovirus or orthoreovirus infection. Although serpentovirus infections in colubrids have not been characterized microscopically, typical changes other than pneumonia seen in serpentovirus-infected pythons and boas, such as rhinitis, tracheitis, and stomatitis, were not present in the absence of parasites. Bacteria and fungi were not observed in pulmonary tissues. Other potential respiratory parasites of snakes include Rhabdias spp, renifers (trematodes), and pentastomes. While proliferative pneumonia without evidence of parasites was observed in 3 snakes (cases 8, 10, and 11), this could represent residual pathology from previous parasitic migration or remnant histologic changes following anthelmintic elimination of parasites. Two of these snakes were positive for Strongyloides spp through fecal examination and were euthanized within 1 week of the conclusion of an anthelmintic treatment trial owing to quality-of-life concerns. While histology could not confirm the suspicion of prior pulmonary strongyloidiasis, treatment was apparently unsuccessful in eliminating these parasites from other organ systems.
Diagnosis of rhabditid nematode infections is commonly achieved by detection of ova and/or first-stage larvae in feces or lung washes. Due to sporadic shedding of Strongyloides life stages, fecal examination of naturally infected snakes may only be intermittently positive. 16 Strongyloides and Rhabdias ova are also nearly identical and only infective third-stage larvae of the 2 genera can be differentiated due to a characteristic forked tail present in Strongyloides spp.37,38 In snakes, anthelmintic prophylaxis and treatment remain largely based on anecdotal evidence with limited safety or pharmacokinetic data.16,24 Fenbendazole and ivermectin are commonly considered for anthelminthic treatment of nematode infection in reptiles; usage of thiabendazole to treat strongyloidiasis in snakes has also been reported.9,10,18 Many reported anthelmintic dosages are based on empirical observations but, owing to the diversity of reptiles, it may not be possible to take into account unique sensitivities that result in adverse drug reactions or death. 24 Although not the focus of this article, a subset of 10 snakes representing all species in this study appeared to tolerate 0.2 to 0.3 mg/kg oral ivermectin given at 14-day intervals.
Treatment efficacy against Strongyloides infection in snakes is variable.9,16 In some studies, administration of anthelmintics eliminated the detection of ova and larvae from feces despite continuation of clinical signs and ultimate death of infected animals.10,16 Reports of complete resolution of disease with negative follow-up fecal examinations are exceedingly rare. 10 Although not monitored on a large or formal scale, an ivermectin treatment regimen instituted in the colony temporally correlated with improved clinical signs of respiratory disease. Despite the heavy nematode infections in the necropsied snakes, no snakes died during treatment without significant clinical comorbidities. Finally, environmental sanitation and hygiene are paramount to elimination of free-living populations to prevent reinfection or development of hyperinfection. 29 Following characterization of the disease syndrome, the producer worked with staff to more frequently do full substrate exchanges in the tubs, which also likely contributed to colony health improvement over time.
The findings of this study (1) document a unique presentation of widespread infection by a classically enteric nematode parasite, and (2) emphasize the importance of routine screening tests and proper animal husbandry in intensively managed animal populations for the detection and prevention of disease.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231186391 – Supplemental material for Proliferative strongyloidiasis in a colony of colubrid snakes
Supplemental material, sj-pdf-1-vet-10.1177_03009858231186391 for Proliferative strongyloidiasis in a colony of colubrid snakes by Erin A. Graham, Eric W. Los Kamp, Nina M. Thompson, Steven B. Tillis, April L. Childress, James F.X. Wellehan, Heather D.S. Walden and Robert J. Ossiboff in Veterinary Pathology
Footnotes
Acknowledgements
We thank the staff of the Histology Laboratory at University of Florida’s College of Veterinary Medicine for slide preparation and the collection owners for supporting the diagnostic investigation.
Supplemental material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: E.W.L.K. received funding for student research through the Boehringer Ingelheim Veterinary Scholars Program and the University of Florida College of Veterinary Medicine.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
