Abstract
We report the development and field validation of 2 ELISAs for the detection of Ancylostoma caninum or Toxocara canis coproantigens in the feces of dogs with experimental and natural infections, and evidence of cross-reactivity with respective feline counterparts. A. caninum–specific coproantigens were detected in feces of experimentally infected dogs starting at 9 d post-infection (dpi), whereas eggs were not seen until 23 dpi. T. canis–specific coproantigens were detected in 3 of 5 experimentally infected dogs by 31 dpi, and 4 of the 5 animals by 38 dpi. T. canis eggs were seen in feces of 4 of the 5 animals by 38 dpi. One dog had delayed coproantigen detection and low egg output. Additionally, 817 canine and 183 feline fecal samples from naturally infected animals tested by flotation were subjected to coproantigen ELISA testing. Of these 1,000 canine and feline samples, 13 and 23 samples, respectively, were positive for “hookworm” or “roundworm” eggs; 19 and 26 samples were ELISA positive, respectively. The T. canis ELISA detected T. cati coproantigen in cat fecal samples. Discrepant ELISA and flotation results were obtained for 16 hookworm- and 13 roundworm-positive samples. Re-examination of the egg-positive, ELISA-negative samples indicated several instances of possible misidentification or coprophagy, whereas detection of antigen in samples without egg observations is likely a reflection of true infection status with egg shedding below detection levels. There is good indication, based on accumulated field data, that these antigen tests also detect other hookworm and ascarid species.
Introduction
The goal of diagnostic parasitology is to assess whether a patient is infected with a parasite of interest. A major strength of using a method whereby a fecal sample is examined in a manner that allows visual identification of microscopic stages in the sample (e.g., direct smear or the application of a flotation or sedimentation concentration procedure) is that such a method allows simultaneous detection of multiple different parasites and the discovery of parasites that may not have been expected. However, in testing of any type, the task is different when the goal of the analysis is to verify that any given parasite is present in a host, perhaps at a low population density or as a stage that remains cryptic to routine fecal examination methods. Thus, if there are low parasite burdens, intermittent shedding of eggs, single sex infections, or sexually immature forms and, hence, prepatent infections present, routine fecal examination becomes less sensitive or may even be incapable of detecting parasites. In these instances, secondary methods have been developed to aid in the detection of a number of parasitic infections, and have included detection of antigen in urine and sera for trematode infections,8,22 fecal antigen detection for Giardia, 5 collection and culture methods as for Tritrichomonas foetus, 9 and DNA detection with PCR for protists and helminths, such as Cryptosporidium and Strongyloides.2,16 It can be anticipated that additional methods, especially in those cases where a confirmatory test is needed, will continue to be developed to support the microscopic examination of feces.
In the case of Ancylostoma and Toxocara infections, the 3 scenarios in which an antigen test might supply additional information about the presence of intestinal infection would be 1) when immature prepatent stages are present in the intestine in a new infection or from reactivation of somatic larvae, 2) in cases with very low worm numbers or non-patent, single-sex infections, and 3) when spurious eggs are present in the feces. The prepatent period of Ancylostoma caninum is typically 2–4 wk,3,10 and while the infections are prepatent, feeding hookworms can cause severe blood loss. 11 The prepatent period of the common feline hookworm, A. tubaeforme, in the cat is typically a little longer than that of A. caninum, with eggs not appearing until 18–28 d after infection. 14 In the case of the common roundworm, Toxocara canis, the prepatent period can be 28–60 d,7,25 and thus, development can span 2 treatments with monthly anthelmintic products. The prepatent period of T. cati is similar to that of T. canis (i.e., 38–56 d). 4 When cats are infected with T. cati by ingesting larvae in infected mice, the larvae mature faster, but the post–mouse-ingestion prepatent period has not been well characterized. 21 The reactivation of larvae from somatic tissues is known to occur in the case of some hookworms. A. caninum is well recognized to have larvae sequestered within the body of a dog that periodically repopulate the intestine, and once there, have a prepatent period of <2 wk. 20 In the case of A. tubaeforme in the cat, larval sequestration does not appear to occur. 15 It is not known if the T. canis larvae in the tissues of a dog repopulate the intestine as do hookworms; however, it does appear that larval T. cati might be sequestered, perhaps in the stomach wall of cats, and periodically repopulate the intestinal tract.19,21
Monthly anthelmintic preventives vary in efficacy against different life stages of the nematodes for which they are labeled, but they are all approved for treating adult hookworm and roundworm infections. Thus, incoming or reactivated somatic larvae will be developing before being killed by the next month’s application of product. Depending upon the timing of the fecal examination relative to the last and next monthly product application, these infections would be detected by ELISA before they became patent based on fecal egg shedding. Furthermore, infections with a few worms are likely to be common in well-cared-for pets. In these cases, the greater fecundity of a female ascarid (“roundworm”) versus a single hookworm increases the microscopic diagnosis of a roundworm infection over that of a hookworm infection whenever low numbers of worms are present. Single-sex infections with these 2 nematodes are fairly uncommon; there is usually a near 1:1 sex ratio.18,23,24 However, in light infections, such as would be expected of a pet on preventive medication, or adult animals with low exposure levels, one would expect cases where only a few adult male or female worms might be present. It is likely that these infections may be detected by ELISA when eggs in a sample might not be observed. Relative to spurious findings in fecal samples, dogs, given their gustatory habits, are often shedding roundworm eggs from previously ingested cat feces, and occasionally the eggs of other ascaridoid nematodes (e.g., Ascaris suum eggs from pig feces; Douglas, JR, Baker NF. Some host–parasite relationships of canine helminths. Ann Biol Colloq Ore St Coll; 1966; Corvallis, OR). 13 Cats routinely shed the eggs of various parasites of mice. 12
We describe herein 2 new ELISAs similar to one previously developed for the canine whipworm, Trichuris vulpis. 6 Both of the new tests also use a recombinant antigen. One of these 2 assays was developed for the detection of a fecal antigen of T. canis and one for an antigen of A. caninum. The tests have been validated against routine fecal flotation results performed on samples from both naturally and experimentally infected dogs. Furthermore, when used on feline fecal samples, the T. canis ELISA proved useful in the detection of T. cati, and there are good indications that other related ascarid and hookworm parasites of cats and dogs are also detectable. These tests should prove a useful adjunct to routine fecal examination and should add to our basic knowledge of the biology of these worms, as these tests detect excreted and/or secreted antigen from the nematode itself rather than eggs in the feces of the infected host.
Materials and methods
Experimental A. caninum and T. canis infections
The experimental infection animal care and protocols were reviewed and approved by the vendor’s Institutional Animal Care and Use Committee in accordance with the Guide for the Care and Use of Laboratory Animals (https://goo.gl/g8EQ8V). Ten uninfected dogs acquired from a Class A U.S. Department of Agriculture animal vendor were treated on day -14 with an anthelmintic (Interceptor, Novartis Animal Health, Greensboro, NC) per the dosages prescribed on the package insert (0.5–1.0 mg milbemycin oxime per kg body weight). Fecal samples were collected prior to infection on days -14, -11, and -1 to verify absence of helminth infections. Five dogs were allocated into either the A. caninum infection group, or the T. canis infection group. Dogs were given orally either 150–300 infective A. caninum larvae or 150–300 larvated T. canis eggs and monitored for 2 h, at 20-min intervals, to ascertain that no vomiting had occurred. Fecal samples were collected from all 10 dogs at 0, 2, 9, 17, 23, 31, 38, 41, 44, 48, 52, 62, 69, 76, 84, and 87 d post-infection (dpi). At 89 dpi, 3 of the 5 dogs from each infection group (A. caninum: dogs 1, 2, and 4; T. canis: dogs 8–10) were again treated with an anthelmintic (Novartis Animal Health) at the prescribed dosages, and had fecal samples collected on days 93, 97, 100, 105, 107, and 111. Egg counts were performed on all samples using 1.25 specific gravity (SG) Sheather sugar solution centrifugal flotation, as described below. Two dogs from each infection group (A. caninum: 3 and 5, and T. canis: 6 and 7) were autopsied on day 89, and adult nematodes were confirmed, but not enumerated. Dogs 1, 2, 4, and 8–10 were returned to the colony upon completion of the study.
Egg recovery and quantitation procedure for experimental nematode infections
One gram of feces from each experimentally infected dog was homogenized with ~10 mL of water and poured through a strainer; the homogenate was pressed until no more fluid passed through. The strained filtrate was poured into a 15-mL centrifuge tube, and the tube was spun at 174 RCF for ~10 min. Following centrifugation, the supernatant was discarded, and the fecal pellet resuspended in ~12 mL of 1.25 SG Sheather sugar solution (2,267 g of sugar, 29.5 mL of melted phenol, 1,451 mL of distilled water) before centrifugation, as before. Following centrifugation, the tube was carefully filled with the sugar solution until a small positive meniscus was formed. A coverslip was placed on the meniscus and allowed to stand for at least 10 min after which the coverslip was removed and placed on a microscope slide. Examination for A. caninum or T. canis eggs was made at 100× magnification, and the number of eggs counted beneath the coverslip was considered the number of eggs per gram of feces.
Egg recovery from canine and feline field fecal samples
Egg flotation data were collected from 1,000 field fecal samples (817 dog and 183 cat) submitted to IDEXX Reference Laboratories located in North Grafton, MA, Dallas, TX, or Sacramento, CA. Samples from CA, CO, CT, HI, MA, ME, NH, NV, NY, OR, RI, and TX were represented in the collection. The method used to process these samples was as follows: ~2 g of feces from each fecal sample were mixed with 10–15 mL of 1.24 SG ZnSO4 and homogenized. The homogenate was strained and the liquid collected. The strained filtrate was poured into a 15-mL centrifuge tube, filled with additional ZnSO4 to within 2 mm of the rim, and spun at 500 RCF for 5 min. Following centrifugation, the tube was carefully filled with more ZnSO4 solution until a small positive meniscus was formed. A coverslip was placed on the meniscus, allowed to stand for 10 min, and then removed to a microscope slide. Examination for parasite eggs was made at 100× magnification, and the number of eggs counted beneath the coverslip was reported as rare (1–2 eggs), few (3–10 eggs), moderate (11–30 eggs), or many (>30 eggs). Sample identity was blinded for ELISA testing. A second fecal flotation, as described above, was conducted on these field samples when a discordant ELISA result was obtained. If eggs were recovered, minimal and maximal egg diameter were recorded.
Fecal slurry preparation
Approximately 0.05 g of fecal material were suspended in 0.5 mL of sample buffer (Tris buffer, pH 7.2, supplemented with Tween-20, 5% mouse serum [Equitech, Kerrville, TX], 10% rabbit serum [Pel-Freez, Rogers, AR], and 40% fetal bovine serum [Life Technologies, Grand Island, NY]). Fecal material and sample buffer were mixed well by stirring with an applicator stick. Fecal slurries were prepared immediately before testing and were not stored.
Fecal extract preparation
One gram of fecal material was suspended in 4 mL of sample buffer (Tris buffer, pH 7.2, supplemented with Tween-20, 5% mouse serum [Equitech], 10% rabbit serum [Pel-Freez], and 40% fetal bovine serum [Life Technologies]). Sample homogenization was accomplished by addition of four or five 3-mm silica beads (product 11-312A, Fisher Scientific, Pittsburg, PA) followed by 2 min on a vortex. The fecal slurry was clarified by centrifugation at 17,000 RCF for 30 min. The fecal extract supernatant was collected by pipette and stored at 4°C for up to 1 wk or frozen at −20°C.
A. caninum and T. canis ELISA
Polyclonal antibodies (pAb) and monoclonal antibodies (mAb) were developed against recombinantly expressed A. caninum Asp-5 protein. 26 Capture antibodies were polyclonal and detection antibodies were monoclonal IgG kappa subtypes. Monoclonal antibodies were developed against recombinantly expressed T. canis protease inhibitor homolog.1,17 Both capture and detection T. canis mAbs were IgG kappa subtypes.
Capture pAb (hookworm) or mAb (roundworm) coating of well plates was performed by first diluting the antibody in 10 mM phosphate-buffered saline (PBS, pH 7.3) buffer to a concentration of 3 μg/mL. A 96-well microtiter ELISA plate (Immulon 1B, product 6310, Thermo Electron, Milford, MA) was then coated with 100 μL of diluted antibody per well, covered, and allowed to incubate overnight at 4°C. Coated plates were washed 3 times with 1× PBS plus 0.05% Tween-20 (PBST) on an automated plate washer (ELx405, BioTek Instruments, Winooski, VT). Tris buffer (Trizma, pH 8.0, product T2694-IL, Sigma-Aldrich, St. Louis, MO) plus 1% bovine serum albumin (product 68020, Proliant, Ames, IA) was prepared, and 300 µL was added to each well and incubated for 3 h at room temperature as a blocking step. A 2.5% sucrose solution in 0.1 M Tris buffer (pH 7.0) was prepared, and 300 µL were added to each well and incubated for 3 h at room temperature. Sucrose over-coated plates were tapped dry and stored at 4°C in sealed bags with 4 g of desiccant. Over-coated plates are stable for up to 1 y. Fecal extract samples (100 µL) were added to plate wells. The plate was covered and allowed to incubate for 1 h at room temperature followed by a wash step with PBST. This step was repeated 4 additional times for a total of 5 washes. Detection mAb antibody (succinimidyl-4-(N-maleimidomethyl)cyclohexane-1-carboxylate [SMCC; product P122322, Thermo Fisher Scientific, Waltham, MA]) horseradish peroxidase (product 10815462103, Roche Molecular BioChemicals, St. Louis, MO) was prepared at 3 μg/mL in sample buffer and 100 μL were added to each well and incubated for 30 min at room temperature followed by 5 PBST wash steps. A 3,3′,5,5′-tetramethylbenzidine (TMB; product TM102-91000-128, SeraCare Life Science, Milford, MA) solution containing 0.2 g/L TMB and 0.01% hydrogen peroxide was prepared, and 50 μL of substrate was added to each well and incubated for 10 min at room temperature. Color development was stopped by addition of 50 μL of 0.1% sodium dodecyl sulfate solution to each well. Plates were read at a wavelength of 650 nm within 10 min of addition of stop solution. An ELISA cutoff of 0.100 was established by adding 6 standard deviation values to the mean value of several hundred negative samples run in the assay (data not shown). ELISA optical density (OD) signal measured over a titration of the cognate recombinant protein, tested as a sample for each assay, correlated with protein concentration.
Preparation of monoclonal and polyclonal resin
Monoclonal antibody conjugates for A. caninum and pAb conjugates for T. canis were prepared for target-specific antibodies and for non–target-specific antibodies following manufacturer’s instructions with a few modifications (AminoLink plus coupling resin, product 20501, Thermo Fisher Scientific). Briefly, 2 mg of each antibody at 1 mg/mL in PBS was reduced with 50 mM sodium cyanoborohydride (AminoLink reductant, product 44892, Thermo Fisher Scientific) solution. The reduced antibody was mixed with 2 mL of resin overnight at 4°C. Coupled resin was blocked with 1 M Tris buffer (pH 8.0), and washed with 1 M NaCl. The conjugated resin was resuspended in an equal volume of 10 mM PBS (pH 7.3). Conjugated resin was stored at 4°C.
Immunoprecipitation assay
An immunoprecipitation assay was developed to confirm that positive ELISA signal was in response to either A. caninum or T. canis coproantigen rather than nonspecific signal. This is particularly important to determine for its validation with field samples. The antibody-coated beads were prepared by pelleting 200 µL of resin and removing the supernatant. Either A. caninum target or T. canis target and unrelated antibody resin was prepared for each fecal sample. The antibody-coated beads were mixed with 260 µL of fecal extract and incubated at room temperature for 30 min with tumbling. The antibody-bead complex was removed by centrifugation at 17,000 RCF for 3 min. Pretreatment of each sample with immunoprecipitation reagents resulted in 2 samples post-treatment: A, immunoprecipitated with unrelated antibody and B, immunoprecipitated with A. caninum or T. canis target–specific antibody. Treated samples A and B (100 µL each) were tested in either the A. caninum or T. canis ELISA as described above. The immunoprecipitation assay was applied to samples collected from 2 experimentally infected dogs from day 0 (pre-infection), days 2 and 48 (prepatent), and day 84 (patent) for detection of adult worm infections of both A. caninum (dog 5) and T. canis (dog 9). All ELISA-positive field fecal samples were also tested in the immunoprecipitation assay.
Data analysis
The following formula was used to calculate the percent inhibition for the immunoprecipitation assay: percent inhibition = (1 – B/A)100%, where A indicates the optical density at 650 nm (OD650) value for the unrelated antibody pretreatment and B indicates the OD650 value for the target-specific antibody pretreatment. Percent inhibition ≥50% was considered an indication of specific A. caninum or T. canis coproantigen in the sample.
Assessment of cross-detection potential among ELISA coproantigen assays on experimentally infected dogs
Feces from A. caninum, T. canis, and Trichuris vulpis experimentally infected dogs, used for the development of respective coproantigen ELISAs, were cross-tested in each ELISA platform to assess cross-reactivity among these 3 nematode parasites (e.g., A. caninum antigen from the feces of experimentally infected dogs was tested on the A. caninum, T. canis, and T. vulpis ELISA; likewise for T. canis and T. vulpis antigens; data not shown.)
Review of coproantigen ELISA performance on a large pool of canine and feline field fecal samples
Egg flotation and corresponding coproantigen ELISA results were collected from an additional 9,083 field fecal samples submitted to IDEXX Reference Laboratories. Samples from CA, CO, CT, HI, MA, ME, NH, NV, NY, OR, RI, and TX were represented in the collection. These fecal samples were comprised of 7,352 canine, 1,719 feline, and 12 samples for which no “dog” or “cat” species designation was given. These samples had been examined previously by both fecal flotation and coproantigen ELISAs, as detailed above. The goal of this analysis was to explore the capability of the coproantigen tests to detect other related hookworm and ascarid parasites in canine and feline samples.
Results
A. caninum eggs were observed on fecal flotation in all 5 experimentally infected dogs starting on day 23 (Fig. 1A). Eggs per gram varied within and between individual animals, but no infected animal stopped shedding eggs over the time course of fecal collection prior to treatment or autopsy (on day 89). A. caninum–specific coproantigens were detected as elevated ELISA signals (>0.1 OD) starting on day 9 for all 5 infected dogs. ELISA signal was strong (>2.0 OD) by day 23 for all 5 animals and remained elevated throughout the study (Fig. 1B). Three A. caninum–infected dogs were followed after treatment (day 89). Samples collected 4 d after treatment had markedly decreased egg counts (<100 and no eggs were detected in a sample from dog 1). Eight days post-treatment (day 97), all 3 dogs had dropped to “egg-negative” status and maintained this status through the end of the study. Coproantigen levels for these dogs fell below the 0.1 OD cutoff level for 2 of the 3 dogs 8 d post-treatment and for all 3 dogs 11 d post-treatment. However, levels were observed to rise once more 16 d post-treatment for all 3 animals.
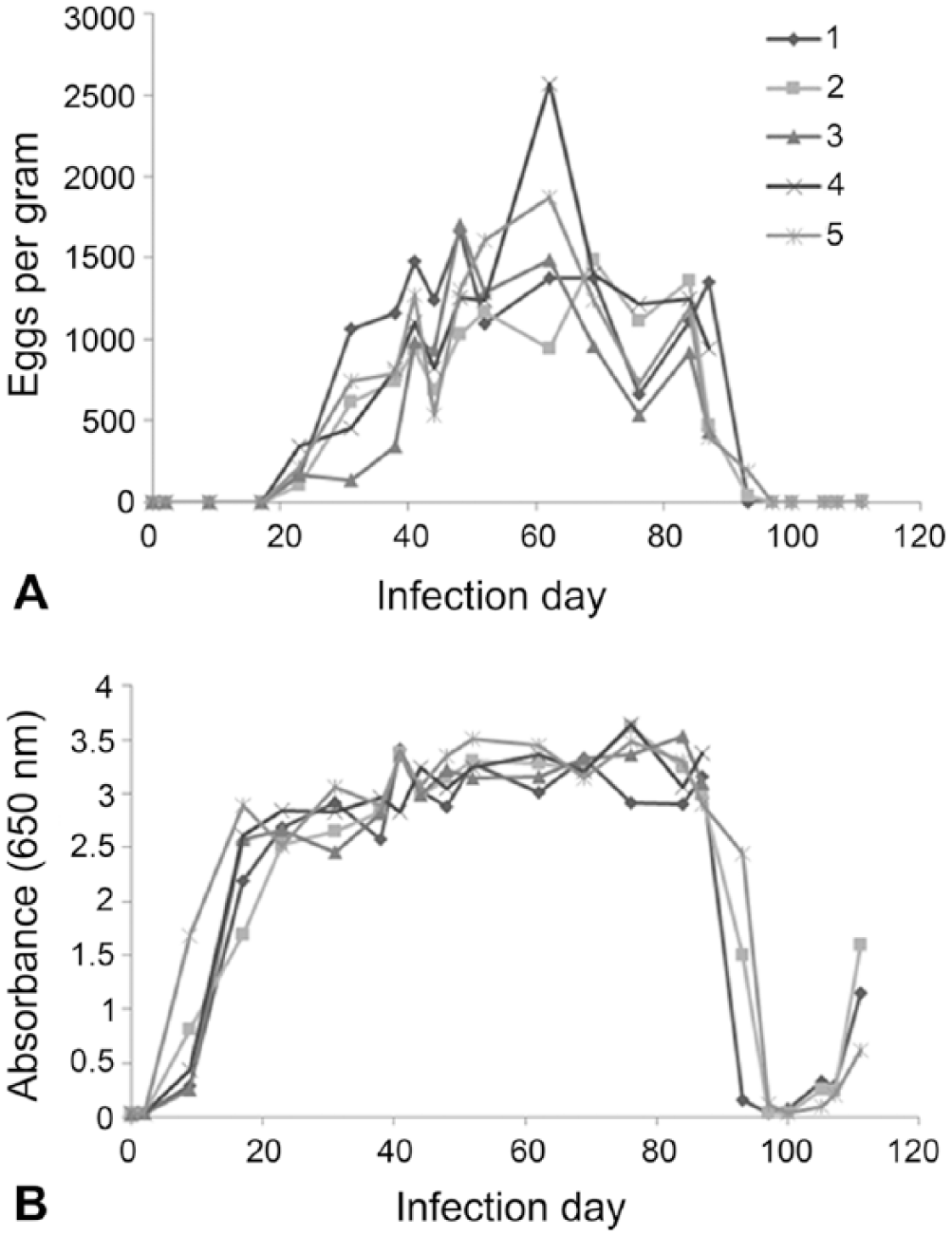
Time course of fecal egg shedding and coproantigen detection in 5 Ancylostoma caninum experimentally infected dogs. A. Eggs per gram determined by fecal flotation. B. A. caninum coproantigen level detection by ELISA. Neither A. caninum eggs nor coproantigen signal were detected by flotation and ELISA in a set of uninfected dogs over the same time course (data not shown).
In order to assess if the ELISA reactivity was specific to A. caninum–secreted antigens, an immunoprecipitation assay was conducted on selected days of the infection time course for dog 4. Four times in this hookworm’s life cycle were assayed: day 0, uninfected; day 2, infected but without adult worms; and days 48 and 84, patent infection. The elevated hookworm signals observed at days 48 and 84 were significantly reduced (>90%) by the target-specific treatment, indicating that the signal is not the result of nonspecific binding of antibody reagents. Furthermore, no elevated signals were obtained over the time course of the study on either the T. canis or T. vulpis coproantigen ELISAs for this group of dogs, further validating the specificity of the assay against other common families of parasitic nematodes.
T. canis eggs were observed on fecal flotation in 4 of the 5 experimentally infected dogs starting on day 38. Confirmation of egg shedding in dog 8 was delayed until day 41, and eggs per gram were lower than for the other dogs throughout the time course of the study (Fig. 2A). T. canis–specific coproantigens were detected as elevated ELISA signals (>0.1 OD) starting on day 31 for 4 of the 5 dogs. T. canis coproantigen was not detected in the feces of dog 8 until day 41, the same day that eggs were first evidenced for this animal. T. canis ELISA signal was very strong (>3.0 OD) by day 38 for all animals except dog 8, which reached similar ELISA signal levels by day 69 (Fig. 2B).
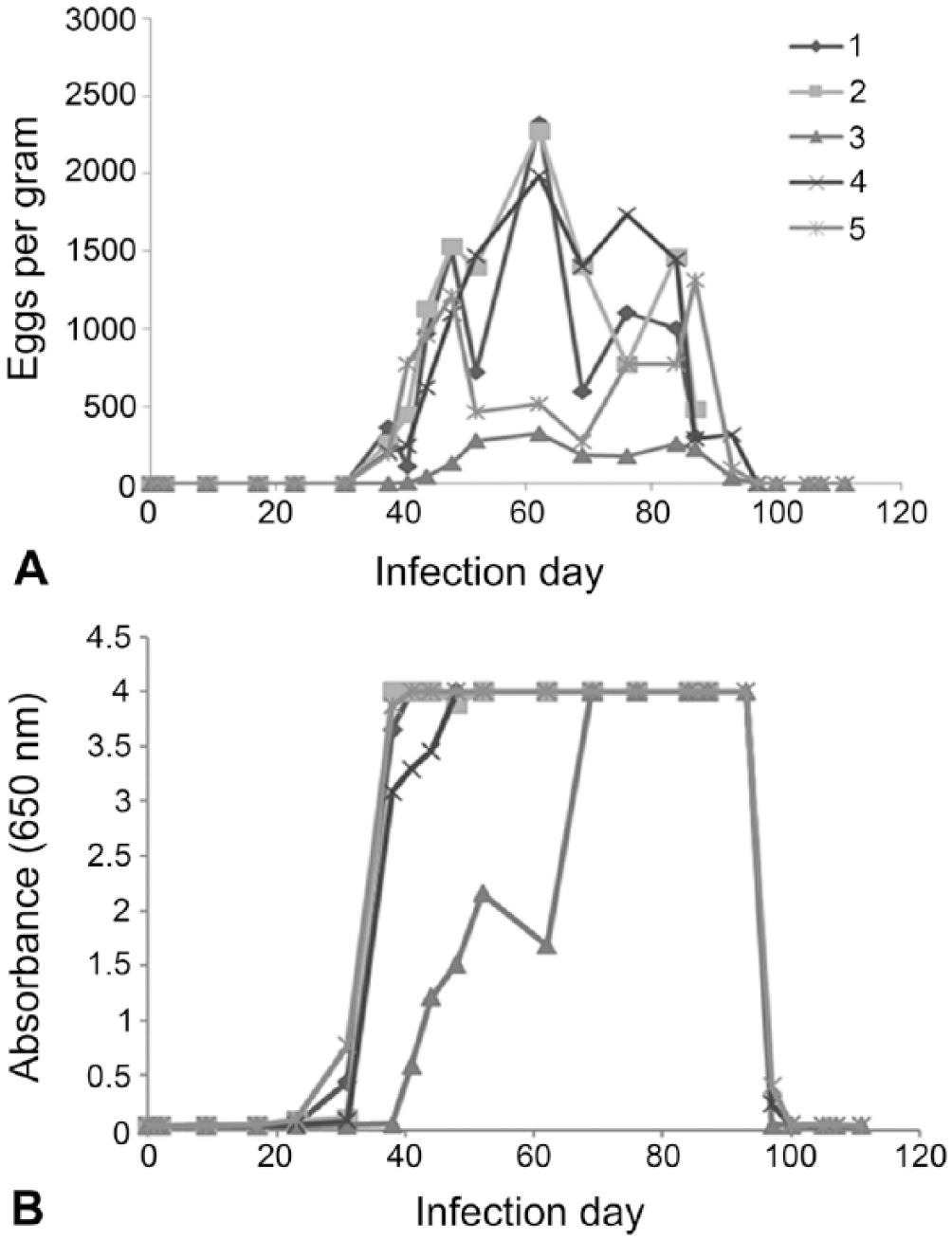
Time course of fecal egg shedding and coproantigen detection in Toxocara canis infected dogs. A. Eggs per gram determined by fecal flotation. B. T. canis coproantigen level detection by ELISA. Neither T. canis eggs nor coproantigen signal were detected by flotation and ELISA in a set of uninfected dogs over the same time course (data not shown).
Three T. canis–infected dogs were followed after treatment (day 89). Decreased egg counts were observed 4 d post-treatment (day 93), and all 3 dogs had no detectable eggs in fecal samples 11 d (day 100) post-treatment or through the end of the study. The coproantigen levels in these 3 animals also decreased post-treatment, and all 3 were ELISA-negative (<0.1 OD cutoff level) by day 100. Antigen levels were not observed to rise thereafter.
As for the hookworm assay, ELISA specificity to the T. canis–secreted antigens was evaluated in the immunoprecipitation assay. Four times in this roundworm’s life cycle were assayed in dog 9: day 0, uninfected; day 2, infected but without adult worms; day 48, prepatent adult infection; and day 84, patent infection. The elevated signals observed at days 48 and 84 were significantly reduced (>90%) by the target-specific treatment, indicating that the signal is not the result of nonspecific binding of antibody reagents. Once more, validating the specificity of the assay against less closely related nematodes showed that no elevated signals were obtained over the time course of the study on either the A. caninum or T. vulpis coproantigen ELISAs for this group of dogs.
The 2 new coproantigen assays were performed on a collection of 1,000 fecal samples from naturally infected dogs (n = 817) and cats (n = 183). Retained samples with an amount ≥2 g were selected from field samples submitted for fecal flotation testing at IDEXX Reference Laboratories. Flotation result was not known prior to ELISA testing. In these samples, 13 canine and 0 feline samples were reported to contain “hookworm” eggs, and “roundworm” eggs were observed in 17 canine and 6 feline samples. Four canine samples were egg-positive for both hookworm and roundworm eggs. Other parasites observed included: Cystoisospora, Giardia, and Taenia in some hookworm-positive egg samples, and T. vulpis, Taenia, and Eimeria in some roundworm-positive egg samples. When examined with the Ancylostoma ELISA, 19 samples were antigen positive. All 19 were canine samples. Antigen detection and egg observation agreed on 8 samples, and there were 5 antigen-negative, egg-positive samples. In the 13 samples that had hookworm eggs observed, ELISA signal ranged from below cutoff to 2.9 OD (Fig. 3). There were 11 antigen-positive, egg-negative samples (Fig. 4). Overall, when eggs were observed, the percent agreement between the ELISA A. caninum coproantigen test and fecal flotation test was 61.5%, whereas the percent agreement between this ELISA and the flotation when no eggs were observed was 98.9% (Table 1). In the case of the Toxocara ELISA, there were 26 antigen-positive samples: 17 canine and 9 feline samples. Positive antigen detection and egg observation matched in 18 samples (12 canine and all 6 feline samples). There were, however, also 5 antigen-negative, yet egg-positive samples (all canine samples). For the 23 egg-positive samples, ELISA signals ranged from below cutoff to 4.0 OD (Fig. 3). There were 8 antigen-positive, egg-negative samples (5 canine and 3 feline samples; Fig. 4). Similarly to what was observed for the A. caninum coproantigen ELISA, the overall agreement between increased OD level and egg observation after flotation was 78.3% for the T. canis coproantigen ELISA, whereas the percent agreement between ELISA OD level to samples without egg observations was 99.2% (Table 2). Of the 4 samples that were hookworm and roundworm egg–positive, 3 samples tested positive on both ELISAs; the fourth sample was ELISA-negative on both tests.
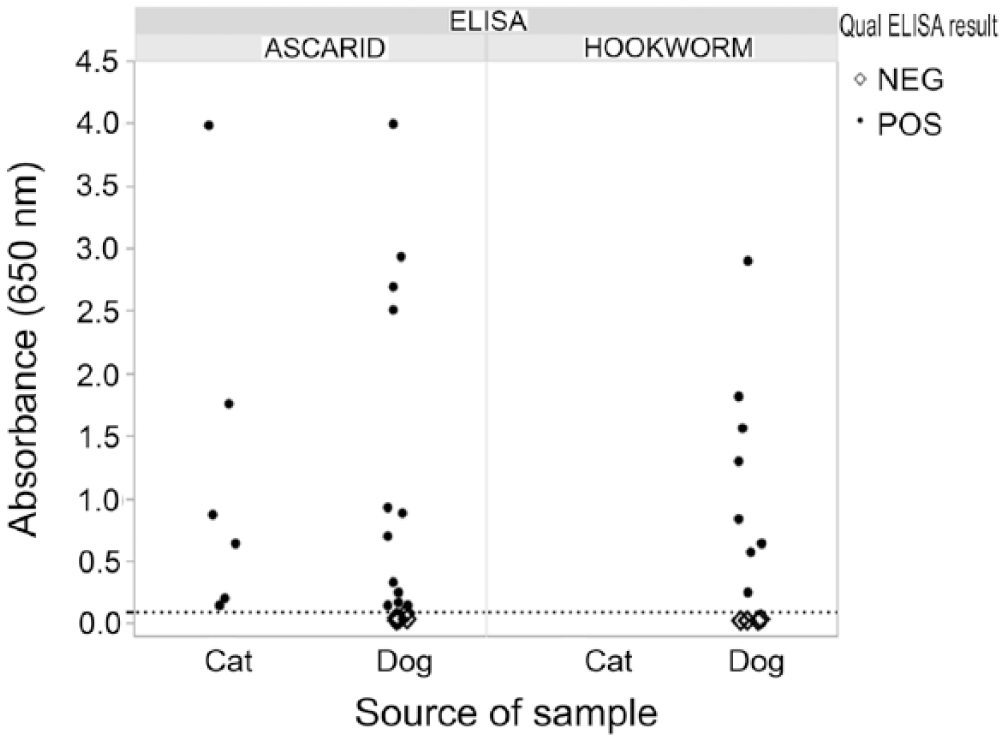
Ancylostoma and Toxocara enzyme-linked immunosorbent assay (ELISA) results for egg-positive field samples. Dotted line is the ELISA optical density cutoff of 0.100.
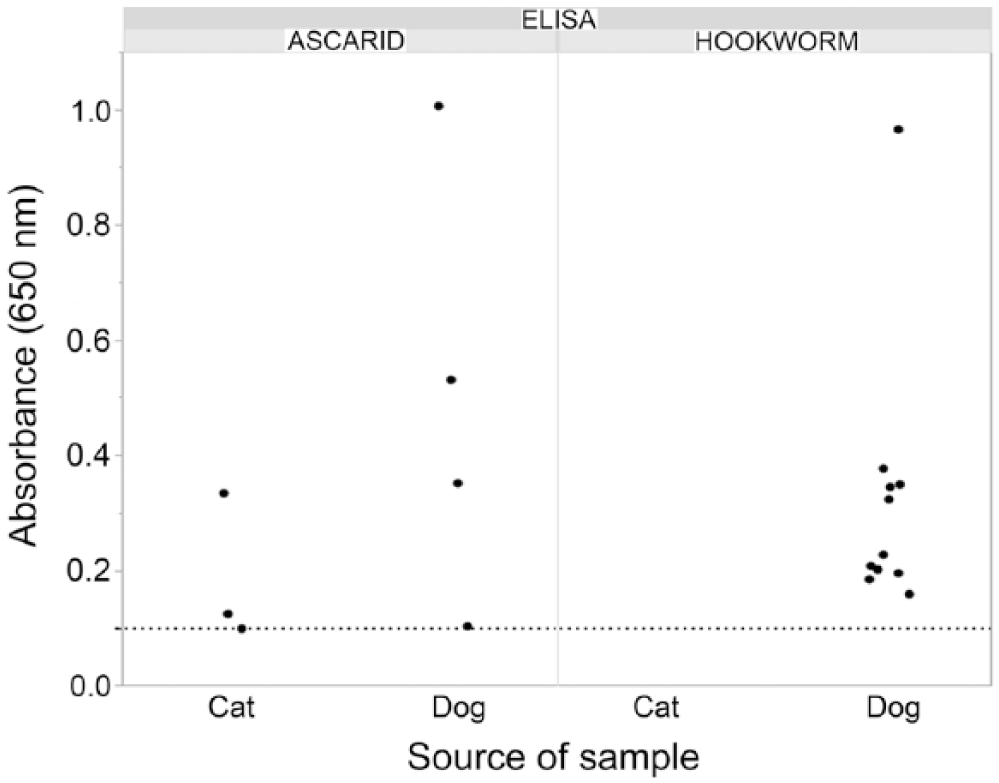
Ancylostoma and Toxocara ELISA results for egg-negative, ELISA-positive field samples. Dotted line is the ELISA optical density cutoff of 0.100.
Ancylostoma ELISA compared to the egg flotation gold standard.
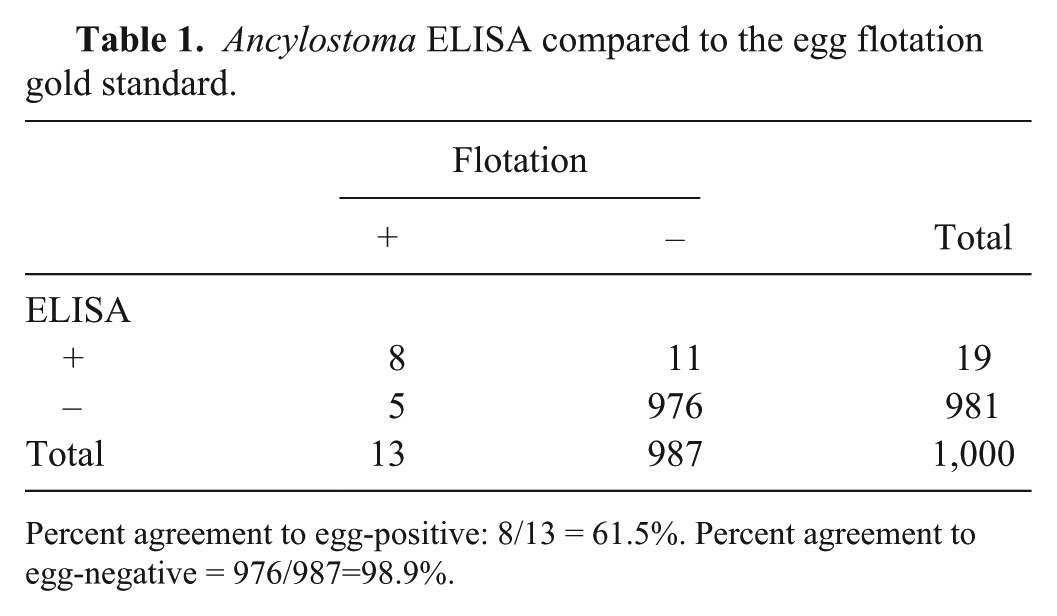
Percent agreement to egg-positive: 8/13 = 61.5%. Percent agreement to egg-negative = 976/987=98.9%.
Toxocara ELISA compared to the egg flotation gold standard.
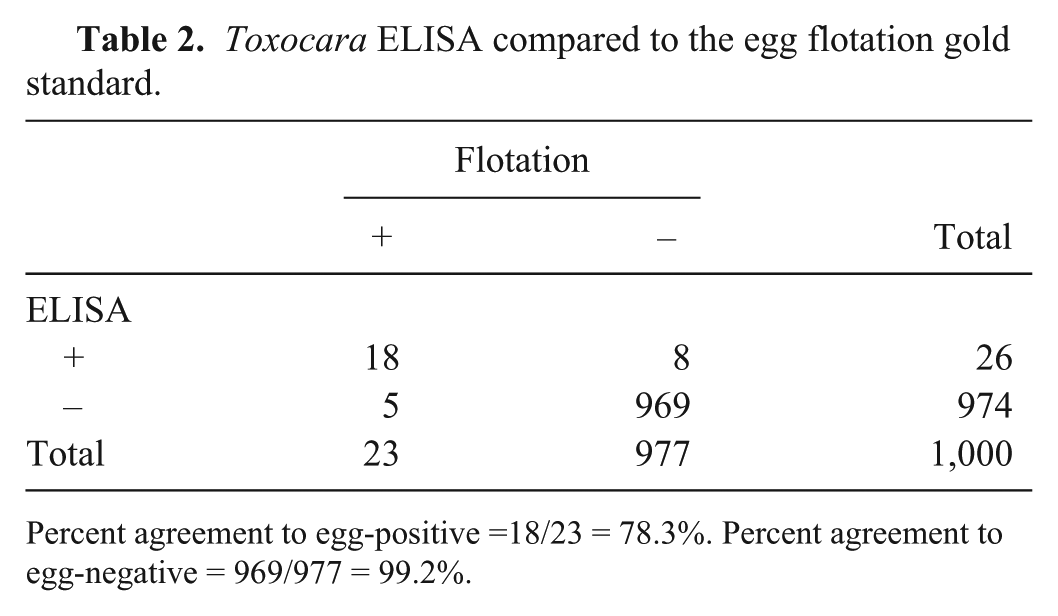
Percent agreement to egg-positive =18/23 = 78.3%. Percent agreement to egg-negative = 969/977 = 99.2%.
Further examination of the discordant results for A. caninum revealed that 11 of the 987 samples that lacked a “hookworm” egg observation, but had elevated ELISA signals, had OD levels ranging from 0.160 to 0.968. Also, of the 977 samples that lacked a “roundworm” egg observation, the 8 samples (3 feline and 5 canine) that tested positive with the ELISA had OD levels ranging from the cutoff level of 0.100 to >1.0 (Fig. 4). All egg-negative, ELISA-positive samples were confirmed in the immunoprecipitation assay, supporting detection of nematode-specific antigens.
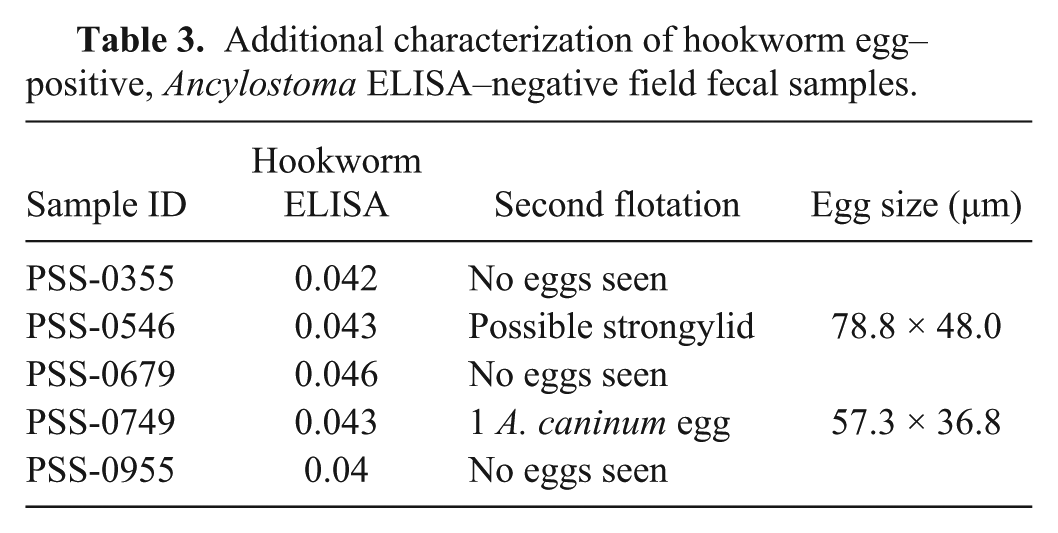
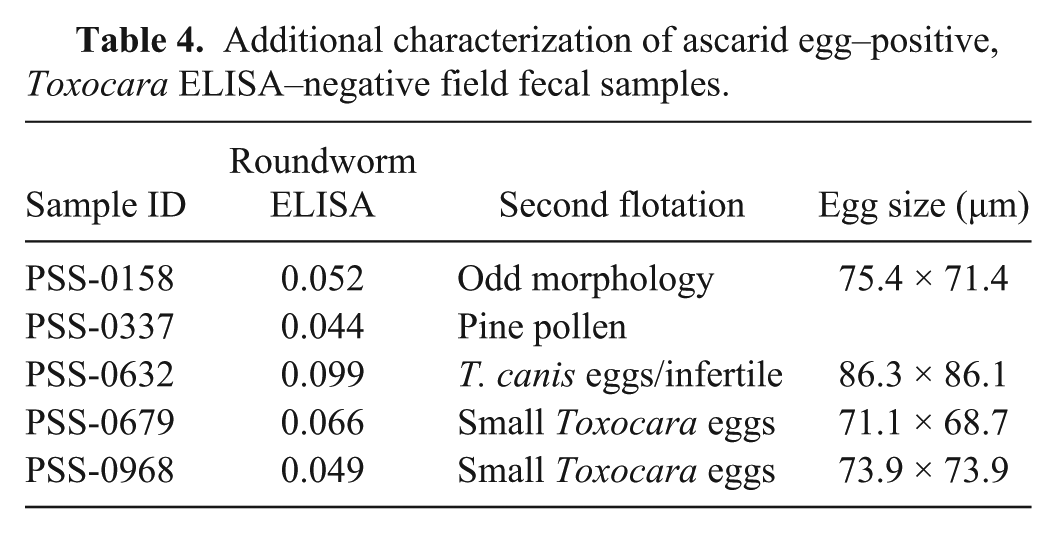
Egg-positive field samples that were negative by the respective coproantigen ELISA were additionally further characterized by a second fecal flotation. No eggs were observed in 3 of the 5 “hookworm” samples with such discrepancies, 1 sample had 3 large strongylid eggs, and a fifth sample had a single A. caninum egg observed upon a second flotation (Table 3). Of the 5 canine “roundworm” egg–positive fecal samples that were T. canis coproantigen ELISA-negative, 1 sample appeared to contain distorted Toxocara-like eggs, a second sample contained pollen grains most likely originally misidentified as eggs, 2 other samples had small Toxocara eggs consistent with those of T. cati, and the fifth sample was confirmed to contain T. canis eggs (Table 4).
Additional characterization of hookworm egg–positive, Ancylostoma ELISA–negative field fecal samples.
Additional characterization of ascarid egg–positive, Toxocara ELISA–negative field fecal samples.
A. tubaeforme was expected to cross-react on the A. caninum coproantigen ELISA, yet none of the 183 feline fecal samples originally examined in the field-collected pool of 1,000 bore evidence of hookworm infection. Thus, flotation and respective coproantigen ELISA results were compiled for 9,083 fecal samples from naturally infected animals previously submitted to and processed at the IDEXX Reference Laboratories. Of these, 149 samples (15 cat and 134 dog) had observations of “hookworm” eggs recorded (0.9% and 1.8% prevalence based on fecal egg shedding for cats and dogs, respectively). Results for coproantigen detection using the A. caninum ELISA on these 9,083 samples indicated agreement with the egg detection results for hookworms in 12 of 15 cat and 107 of 134 dog samples (i.e., overall agreement in 119 of 149 samples, based on egg detection). Of the 9,083 samples examined, discrepant results were obtained for 160 samples, where 30 samples (3 cat and 27 dog) had eggs but no antigen detected, and 130 had antigen but no hookworm eggs detected (11 cat, 110 dog, and 9 where no host designation was provided).
Of the 9,083 canine and feline fecal samples from IDEXX Reference Laboratories, 260 samples had “roundworm” eggs detected (107 cat and 153 dog; 6.2 % and 2.1% prevalence based on fecal egg shedding for cats and dogs, respectively). The T. canis coproantigen ELISA showed good agreement with egg detection, with 91 of 107 cat and 128 of 153 dog samples showing agreement between the flotation and coproantigen tests. However, 152 samples showed discrepancies between these 2 tests: 41 samples (16 feline, 25 canine) were roundworm egg–positive yet had no elevated antigen level evidenced on the ELISA, and 101 samples were ELISA-positive, but no eggs were observed (54 cat, 45 dog, and 2 samples where no host designation was indicated).
Discussion
In the case of the experimental infections with the common canine hookworm and roundworm, A. caninum and T. canis, the 2 newly developed coproantigen ELISAs had 100% concordant positive results with those of microscopic fecal examinations when infections were patent based on egg detection in fecal samples. The concordance was 100% beginning 23 dpi in the case of A. caninum and 38 or 41 dpi in the case of T. canis. However, the coproantigen ELISA was also capable of detecting infections in the prepatent period, as was also observed and reported previously in the coproantigen detection of the canine whipworm, T. vulpis. 6 In fact, the A. caninum ELISA detected infections 2 wk before the patency period. Thus, it appears this ELISA is capable of detecting immature adult worms in the intestine (detection at 9 dpi). In the case of T. canis, the ELISA detected the presence of young adult worms in 4 dogs 1 wk prior to patency (at 31 dpi), but only at the initiation of egg-patency in the remaining dog (at 41 dpi). After anthelmintic treatment, the ELISA results tightly mimicked the egg count results from fecal examinations. For A. caninum, eggs were no longer seen in fecal samples 8 d post-treatment (and through the remainder of the study), and the coproantigen ELISA results dropped to negative status 11 d post-treatment for these dogs. For T. canis, by 11 d post-treatment, eggs were no longer seen in feces and the ELISA coproantigen results were also negative.
After treatment of the hookworm-infected dogs, the coproantigen levels rose past the cutoff value, becoming positive again 16 d after treatment; microscopic fecal examination still evidenced no eggs. This increase in antigen after treatment had also been observed in the case of T. vulpis where there was a rise in fecal antigen beginning a short time after treatment. 6 If the anthelmintic used is assumed to have been efficacious because antigen detection indicates the presence of immature-to-mature adult worms, a post-treatment rise in antigen level should be interpreted as an indication that worms (perhaps sequestered in tissue at the time of treatment) have repopulated the intestinal lumen of the treated animal.
Experimental infections induced in the development of the 3 coproantigen tests (for A. caninum and T. canis, described here, and the T. vulpis ELISA reported previously 6 ) allowed for the controlled testing of cross-reactivity among these 3 common parasites of dogs from 3 phylogenetically distinct nematode groups. No cross-reactivity was observed when feces from animals infected with 1 of the 3 parasites was tested on the 2 antigen tests designed to detect the other parasites (e.g., feces from dogs infected with T. canis did not produce positive ELISA readings on the A. caninum or T. vulpis ELISAs). Although no such experimental infections were done in cats, there is good supporting evidence, based on the use of the A. caninum and T. canis coproantigen ELISAs on field samples (i.e., naturally infected animals), that they are able to detect their feline counterparts (i.e., A. tubaeforme and T. cati). Such results are not surprising, as the feline parasite homologs of the proteins from the canine parasites used in the development of these tests show high levels of identity when their alignments are compared in GenBank (T. canis and T. cati protease inhibitor homologs showed 96.5% identity; A. caninum and A. ceylanicum Asp-5 homologs share 88% identity [no A. tubaeforme homolog sequence was available for direct comparison at the time of manuscript preparation]).
Use of these antigen tests as a “reflex” test to the traditional ova and parasite flotation assay (i.e., coproantigen ELISA being performed on samples where eggs from the parasite of interest are not seen on fecal flotation) would likely increase the detection of infections in pets. In the 1,000 fecal samples from naturally infected animals, 13 A. caninum egg–positive samples, reflecting 13 infected individuals, would be increased to 24 A. caninum infections if egg or coproantigen detection was used to ascertain infection status. For Toxocara in this sample population, 23 positive samples based on eggs would be increased to 24 based on egg or coproantigen detection.
Both the A. caninum and T. canis ELISAs detect more infected individuals than egg flotation alone (A. caninum, 19 antigen-positive vs. 13 egg-positive; Toxocara, 26 antigen-positive vs. 23 egg-positive). The smaller increase observed with respect to Toxocara may reflect the greater ascarid fecundity. The detection of A. caninum antigen without eggs being present in feces might therefore be expected to be the more common finding, and is supported by the data above. This could be due to a combination of factors, including the more rapid development of these worms into young adults, and their known ability to repopulate the intestine after larval emergence from somatic tissues and growth to adulthood without a need of another infective dose of larvae. 20
If egg-positive samples are further interrogated by the antigen ELISAs, it is apparent that there is a subset of egg-positive samples that are antigen-negative. This may be helpful in reducing the misidentification of spurious parasites in dogs by identifying samples that are egg-positive that do not contain related antigens. There were 5 hookworm egg–positive canine samples that were ELISA-negative. Upon reexamination, the discrepancy could not be explained in 3 of the 5 samples. However, in 1 of the 2 remaining samples, the hookworm eggs were apparently rare and were identified on a recheck of the same sample. And in the other remaining sample, the recovered strongylid egg was partially embryonated, indicating the egg was likely a contaminant. Reexamination of the 5 Toxocara egg–positive, ELISA-negative samples similarly indicated the possibility of misidentification or coprophagy as a reason for the disparate egg or antigen results. Indeed, 2 dog samples had Toxocara eggs morphologically identified as T. cati eggs. It is also interesting to note that of the 17 dog and 6 cat Toxocara egg–positive samples, all 5 discrepant samples were of canine origin, perhaps linking coprophagic behavior to increased spurious egg detection. It seems likely that as more and more samples are examined, the reference labs may ultimately be able to examine the resulting data to determine if these tests are actually assisting the veterinary industry by identifying false-positive samples resulting from predation and/or scavenging, coprophagic, and grooming behaviors, or from environmental contamination.
In the case of both hookworms and roundworms, client-owned cats and dogs remain at risk for infection at any time from the skin penetration of hookworm larvae, the ingestion of infective Toxocara eggs or Ancylostoma larvae, or the ingestion of paratenic hosts (prey animals) containing larvae of either or both genera of parasites. Thus, even if pets are on a year-round heartworm preventive program (e.g., monthly administration of anthelmintic treatments) with added activity against hookworms and roundworms, these worms might have a small window of opportunity to develop to adulthood and patency. Thus, a routine fecal examination might disclose a few eggs in the feces of even a well-cared-for pet, and if there are eggs, and if the ELISA is positive, the pet is probably infected. The veterinarian will need to consider the species of worm(s) involved and the anthelmintic product being used, if any, and then she or he will have to make the decision as to whether to treat the pet with another product at the time of diagnosis or accept that the infection will likely be cleared with the next monthly application of the broad-spectrum heartworm-preventive product.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
DA Elsemore, J Geng, J Cote, and R Hanna were funded by IDEXX.
