Abstract
Citrobacter freundii, an opportunistic bacterial pathogen belonging to the family Enterobacteriaceae, has been sporadically reported in sea turtles. Here, the authors describe 3 unusual lesions associated with C. freundii infection in 3 loggerhead sea turtles stranded on the coast of Gran Canaria Island, Spain. It is possible that these 3 distinct lesions played a major role in the death of these turtles. The first turtle had caseous cholecystitis, a lesion not previously described in sea turtles. The second turtle had large intestinal diverticulitis, a rare condition in loggerheads. The third turtle had bilateral caseous salt gland adenitis. Histologically, numerous gram-negative bacilli were observed at the deepest edge of inflammation in all cases. Pure cultures of C. freundii were obtained from these 3 lesions. Molecular detection of C. freundii DNA in formalin-fixed paraffin-embedded samples from the lesions of the 3 turtles confirmed the microbiological isolation. These cases, in addition to expanding the limited body of knowledge on bacterial infections in sea turtles, highlight the potential pathogenic role of C. freundii in loggerhead turtles.
The loggerhead turtle (Caretta caretta) is the most common sea turtle species in the Canary Islands, whose subpopulation is included in the International Union for Conservation of Nature Red List of Threatened Species as “endangered.” 5 According to a 17-year retrospective study in which the causes of stranding of 1860 loggerhead turtles admitted to the Tafira Wildlife Rehabilitation Center (TWRC) (Gran Canaria Island, Spain) were analyzed, the most frequent causes of stranding were entanglement in fishing gear and/or plastics (51%), unknown/undetermined causes (20%), ingestion of hooks (12%), infectious diseases (5.5%), and trauma (boat strike) (5.2%). 16 The survival/release rate observed for turtles stranded due to entanglement was high (92.4%). 16 However, the survival/release rate for turtles stranded due to infectious diseases decreased to 68.6%. 16 Authors suggested that financial and time constraints in many rehabilitation centers often make it difficult to establish an accurate diagnosis, particularly for infectious diseases. 16
Citrobacter freundii, an opportunistic bacterial pathogen belonging to the family Enterobacteriaceae, has been sporadically reported in sea turtles with septicemic cutaneous ulcerative disease, 11 septic arthritis, 10 ulcerative and fibrinous esophagitis caused by fishing hooks,13,17 pulmonary lesions, 6 and steatitis associated with cold-stunning. 22 It has also been isolated from the cloaca of both healthy1,3 and stranded sea turtles. 8 In addition, recent studies on the global concern of antimicrobial resistance have been focused on sea turtles as potential carriers of multiresistant bacterial strains, particularly Enterobacteriales.1,3 We describe the gross and histopathological characteristics of unusual and fatal lesions associated with C. freundii infection in 3 loggerhead sea turtles stranded on the coast of Gran Canaria Island, Spain, between 2016 and 2020. These cases also highlight the potential pathogenic role of C. freundii in loggerhead turtles.
The first turtle (subadult; female; straight carapace length = 52.5 cm; weight = 21 kg) was found floating on the shore, lethargic, weak, and nonresponsive to external stimuli, and died 5 hours later at the TWRC. The second turtle (subadult; male; straight carapace length = 62 cm; weight = 34.6 kg) was found stranded in a rocky area and was very weak and covered with numerous algae and epibionts. Once placed in one of the TWRC pools, the turtle presented a buoyancy disorder, floating mainly along its caudal body. Radiographic (SRL Collimator Ralco, Italy) and computed tomography (Toshiba Astelion 16, Japan) imaging failed to establish a diagnosis, and the turtle was treated with enrofloxacin (5 mg/kg intramuscularly, every 48 hours for 1 month) and lactated Ringer’s solution (20 mL/kg intracoelomically, every 48 hours for 2 months). The turtle gradually lost weight and died 4 months later. The third turtle (juvenile; male; straight carapace length = 40 cm; weight = 9.5 kg) was found stranded, presenting with dehydration and cachexia. Intravenous fluid therapy with lactated Ringer’s solution (10 mL/kg, every 12 hours) was attempted, but it died 1 day later.
Necropsies and histological processing were performed following previously described protocols. 17 Special stains performed on selected samples included Brown-Hopps (modified) stain for gram-positive and gram-negative bacteria, Ziehl-Neelsen stain for acid-fast organisms, and Grocott’s methenamine silver nitrate stain for fungi.
Tissue samples from the main gross lesions (discarding the caseous material and selecting the leading edge of inflammation) and spleen of each turtle were cultured on brain-heart infusion (BHI) medium (Thermo Fisher Scientific, Waltham, MA, USA) and incubated at 25°C and 37°C for 48 hours. Reseeding was done on chocolate agar, blood agar, and MacConkey agar (Thermo Fisher Scientific) and was incubated at 25°C and 37°C for 24 hours. Identification and antimicrobial susceptibility testing were performed using the Vitek-2 Automated System (BioMérieux, France) with Vitek-2 GN ID Card and Vitek-2 AST-GN98 Card, respectively. A polymerase chain reaction assay to detect C. freundii DNA targeting a specific region of the cfa gene encoding a cyclopropane fatty acids synthase 9 was performed on formalin-fixed paraffin-embedded samples from the 3 turtles using procedures previously described. 12
At necropsy, the first turtle had generalized subcutaneous edema. The most significant gross lesions were hepatic atrophy and severe distention (30 cm × 35 cm) of the gallbladder (Fig. 1a), which was distended and filled with tightly packed lamellar deposits of green, caseous material (Fig. 1b). The wall of the gallbladder was thickened. No parasites were detected. Histologically, heterophilic cholecystitis with loss of the gallbladder epithelium, infiltration of the gallbladder mucosa by numerous heterophils with bile pigment deposits, presence of numerous gram-negative bacilli at the deepest edge of inflammation, and edema of the propria-submucosa were observed (Fig. 1c). Thin hepatic cords with intracytoplasmic accumulation of bile pigment and canalicular bile stasis were observed in the hepatic sections. No histological lesions were detected in other major organs.
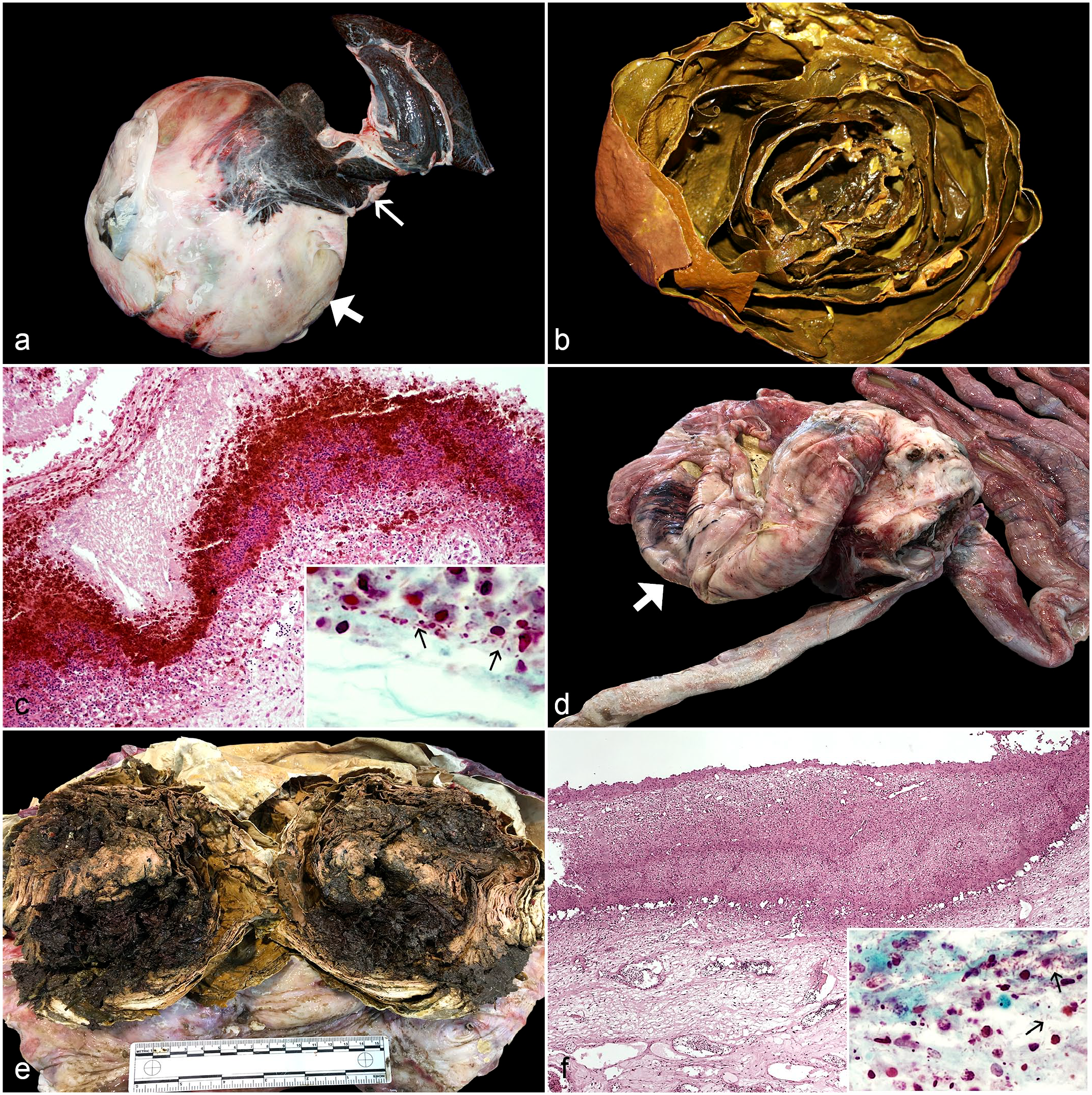
Digestive lesions associated with Citrobacter freundii infection in loggerhead sea turtles (Caretta caretta). (a) Caseous cholecystitis characterized by a distention of the gallbladder (thick arrow). Note the hepatic atrophy (thin arrow). (b) The gallbladder contains greenish solid caseous material arranged in concentric layers. (c) Histopathological features include loss of the gallbladder epithelium, infiltration of the gallbladder mucosa by numerous heterophils with bile pigment deposits, and edema of the propria-submucosa (Hematoxylin and eosin, HE). Inset: the presence of numerous gram-negative bacilli (arrows) at the deepest edge of inflammation (Brown-Hopps [modified], BH). (d) Intestinal diverticulum (arrow) emerging from the mesenteric aspect of the large intestine. (e) The intestinal diverticulum contains abundant yellow, firm, lamellated, caseous exudate. (f) Histopathological features include loss of the intestinal epithelium and infiltration of the mucosa by heterophils and multinucleated giant cells; no muscular layers are present (HE). Inset: numerous gram-negative bacilli (arrows) in the deeper layers of the mucosa (BH).
The most relevant gross lesion observed in the second turtle was the presence of a 26-cm-diameter intestinal diverticulum that protruded from the mesenteric aspect of the large intestine (Fig. 1d). The intestinal diverticulum weighed 3.1 kg, was distended by yellow, firm, lamellated, caseous exudate (Fig. 1e), and the wall was attenuated to 1.5 to 2.0 mm. Several adhesions were also observed between the serosa of the diverticulum and the serosa of the stomach and small intestine. No perforations of the intestinal wall were observed. The liver was friable. No gross lesions were visible in other organs. No parasites were detected. Histologically, heterophilic intestinal diverticulitis was diagnosed. The caseous material within the center of the diverticulum was composed of layers of degenerate heterophils and numerous gram-negative bacilli. The mucosa was ulcerated and completely replaced by heterophils and multinucleated giant cells; numerous gram-negative bacilli were also observed in the deeper layers of the mucosa (Fig. 1f). No muscular layers were observed. Severe dilation of the lymphatic vessels of the serosa of adjacent intestinal sections was also observed. A granuloma characterized by a necrotic center surrounded by macrophages and multinucleated giant cells was observed in a hepatic section; no gram-negative or gram-positive bacteria, acid-fast organisms, or fungi were detected in this granuloma. No histological lesions were detected in other major organs.
At necropsy, the third turtle had mild cachexia. The most important gross lesion was bilateral caseous salt gland adenitis, with yellow caseous necrotic debris occupying up to a third of the total volume of each gland (Fig. 2a). Histologically, abundant eosinophilic material composed of heterophils and cell debris was observed occupying the lumen of the main ducts and some secondary ducts of the glandular lobes; numerous gram-negative bacilli were observed in some of these secondary ducts (Fig. 2b). Incidentally, an Anisakis sp larva was observed embedded in the hepatic capsule. No histological lesions were detected in other major organs.
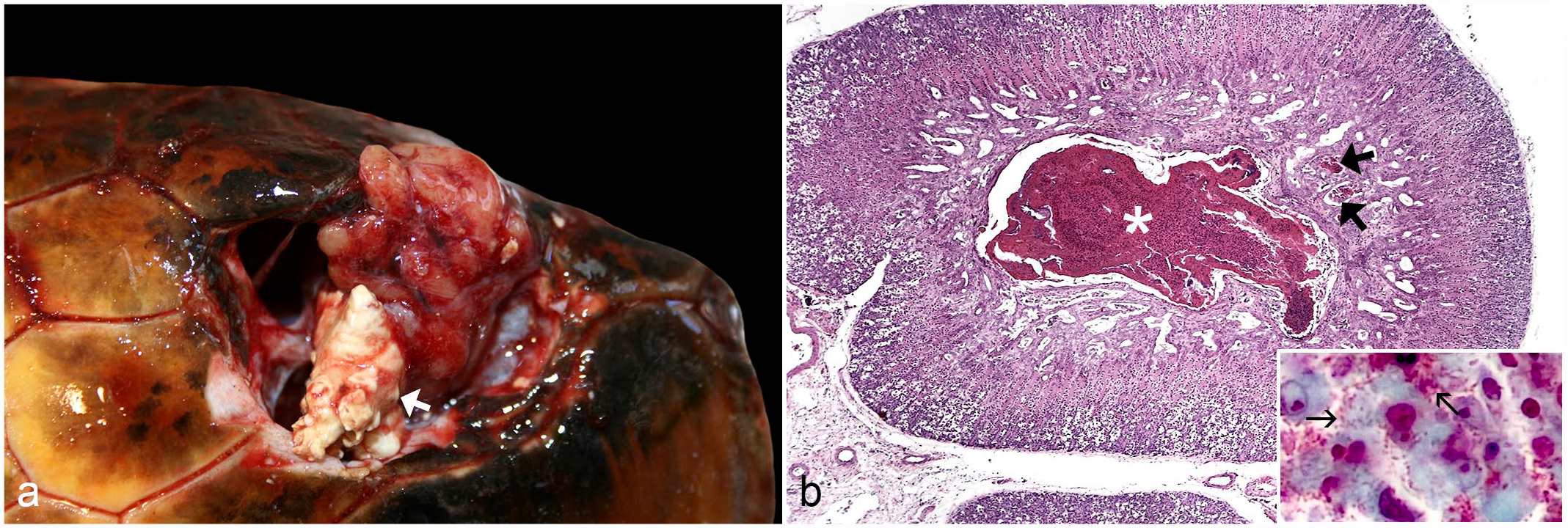
Caseous salt gland adenitis associated with Citrobacter freundii infection in loggerhead sea turtle (Caretta caretta). (a) Note the yellow caseous necrotic debris (arrow) occupying the ventral third of the right salt gland. (b) An abundant eosinophilic exudate composed of heterophils and cell debris is observed occupying the lumen of the main duct (asterisk) and some secondary ducts (thick arrows) of this glandular lobe (Hematoxylin and eosin). Inset: numerous gram-negative bacilli (thin arrows) in a secondary duct with cell debris (Brown-Hopps).
Pure cultures of C. freundii were obtained from the gallbladder (turtle 1), intestinal diverticulum and spleen (turtle 2), and salt glands (turtle 3). Molecular detection of C. freundii DNA in formalin-fixed paraffin-embedded samples from the lesions of the 3 turtles confirmed the microbiological isolation. Results of antimicrobial susceptibility tests are summarized in Supplemental Table S1.
There are very few studies on the pathogenicity of C. freundii in sea turtles.6,10,11,17,22 Some studies suggest that C. freundii may form part of the intestinal microbiota of sea turtles, although there is no unanimity because other studies have not detected it.18,19
Abnormal conditions of the hepatobiliary system are rare in sea turtles and are usually associated with trematode infection. 2 Chronic bacterial infection has also been suggested for sporadic cases in which the inflammatory exudate can obstruct the gallbladder and extrahepatic bile ducts. 2 However, there are no descriptions of such severity or of biliary infections due to C. freundii in sea turtles. 2
Diverticulitis affecting the digestive system is rare in loggerheads. Two cases of esophageal diverticulitis were reported in loggerheads, 13 one of them associated with Aerococcus viridans infection. 21 However, large intestinal diverticulitis is a common necropsy finding in leatherback turtles.15,20 These lesions are generally small, 2 to 9 cm in diameter, and not implicated as a cause of stranding or death, although authors suggested the possibility of perforation, obstruction, and septicemia.15,20 Morganella morganii and Proteus spp have been isolated from these lesions.15,20
Partial blockage of the gastrointestinal tract by the pressure exerted by the diverticulum (as shown by the severe dilation of the lymphatic vessels of the serosa of adjacent intestinal sections) could be responsible for the turtle’s poor body condition and buoyancy disorder. Abnormal buoyancy in sea turtles is not always due to lung disease but also due to the presence of gas and/or foreign bodies within the gastrointestinal tract. 6 Acquired digestive diverticula are described as either pulsion or traction forms. 21 The most common pulsion diverticula result from increased intraluminal pressure or herniation of mucosa through a disruption in the muscular layer of the intestinal wall, and they do not contain layers of the muscle wall. Traction diverticula are less common and occur secondary to peri-intestinal inflammation in which all layers of the intestinal wall are pulled away from the lumen to form an evagination in the intestinal wall. 21 The lack of identification of all layers of the intestinal wall in the diverticulum of this loggerhead suggests it may have been a pulsion diverticulum.
Members of the family Enterobacteriaceae may play an important role as opportunistic agents, becoming pathogenic in immunocompromised animals. 10 Although the digestive location of the lesions observed in the first 2 turtles could have favored colonization by C. freundii, the pathogenic role of C. freundii cannot be ruled out, especially in the first turtle, in which no additional underlying causes were identified.
Salt gland adenitis as the only cause of stranding of loggerheads, as seen in the third case, was reported for the first time in 2011. 14 The most frequently isolated bacteria were Aeromonas hydrophila, Staphylococcus sp, and Vibrio alginolyticus. 14 Although Citrobacter sp was also isolated from one case, no species identification was attempted. 14 Authors reported that plasma concentrations of sodium and chloride from 2 affected loggerheads were higher than normal values (by 45.7% and 69.2%, respectively), suggesting that severe lesions in the salt glands can cause failure to maintain homeostasis and subsequent stranding and/or death. 14 Several studies to characterize the ocular bacterial flora in free-living and captive sea turtles have failed to identify Citrobacter spp.4,7 The absence of lesions in other organs in this turtle rules out possible septicemia, thus being a primary infection of the salt glands. Based on our studies, although the incidence of these bilateral lesions represents less than 1% of strandings, their notable impact on homeostasis requires the attention of the veterinary services of the rehabilitation centers. In addition, C. freundii must be included in the etiological differential diagnosis for this disease.
Further studies are necessary to provide more information about the clinical significance of C. freundii in sea turtles and to analyze their ability to acquire multiple resistance genes to various antibiotics.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231183983 – Supplemental material for Citrobacter freundii-associated lesions in stranded loggerhead sea turtles (Caretta caretta)
Supplemental material, sj-pdf-1-vet-10.1177_03009858231183983 for Citrobacter freundii-associated lesions in stranded loggerhead sea turtles (Caretta caretta) by Alicia Inurria, Alejandro Suárez-Pérez, Pascual Calabuig and Jorge Orós in Veterinary Pathology
Footnotes
Acknowledgements
We thank all the staff of the Tafira Wildlife Rehabilitation Center (Cabildo de Gran Canaria), and A. Santana and P. Briganty (Department of Morphology, ULPGC) for technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
