Abstract
With the aim of evaluating the presence of Fusarium spp. in sea turtles with and without lesions and assessing the risk factors favoring colonization and/or infection, 74 loggerhead sea turtles (Caretta caretta) admitted to rescue and rehabilitation clinics in Italy were analyzed. The study compared 31 individuals with no apparent macroscopic lesions and 43 individuals with macroscopic lesions. Shell and skin samples were analyzed using Calcofluor white with 10% potassium hydroxide, standard histopathological examination, and fungal cultures. Fusarium spp. were isolated more frequently from animals with superficial lesions (39%) than from those with no macroscopic lesions (16%). Isolates from animals with superficial lesions were Fusarium solani species complex (FSSC) lineages haplotypes 9, 12, and 27 (unnamed lineages), FSSC-2 (Fusarium keratoplasticum), Fusarium oxysporum (27%), and Fusarium brachygibbosum (3%). In contrast, only F. solani haplotypes 9 and 12 were isolated from animals with no macroscopic lesions. The presence of lesions was identified as a risk factor for the occurrence of Fusarium spp. Of the 74 animals, only 7 (9.5%) scored positive on microscopic examination with Calcofluor, and histological examination of those 7 animals revealed necrosis, inflammatory cells, and fungal hyphae in the carapace and skin. The results of this study suggest that fusariosis should be included in the differential diagnosis of shell and skin lesions in sea turtles. Direct examination using Calcofluor and potassium hydroxide was not useful to diagnose the infection. Histopathological examination and fungal culture should be performed to ensure correct treatment and infection control.
Fusarium species are filamentous fungi distributed worldwide in soil, plant parts, water, and other organic substrates. 29,32 The genus includes plant pathogens and species causing human and animal infections. 9,32 A broad range of clinical manifestations has been described in humans, ranging from superficial infections (such as keratitis and onychomycosis) to disseminated infections in immunocompromised patients. 32
Fusarium organisms have been described as causing infection in marine animals (delphinids, 13,49,50 pinnipeds, 13,27 fish, 7,12,18,37,39,48 and with increasing frequency in turtles* and alligators 14 ). In addition, colonization of eggs with Fusarium is considered among the main causes of globally declining turtle populations (Chelonia mydas, Caretta caretta, Eretmochelys imbricata, Lepidochelys olivacea, Dermochelys coriacea, and Natator depressus). 43,44
The clinical manifestations caused by Fusarium spp. in aquatic animals vary from superficial invasion of the skin to pulmonary or systemic infections affecting the lungs, liver, heart, and cartilages. 3,23,39,48 Superficial infections occur both in marine animals living freely in the sea, such as dolphins and pinnipeds, and in various species of sea turtles, while pulmonary or invasive infections have been described in Kemp’s ridley turtles (Lepidochelys kempi), baby loggerhead turtles (Caretta caretta), and occasionally in other marine animals such as baby sharks. 13,29,36,42
Fusarium solani (Mart.) Saccardo (1881) has been recognized as the main etiological agent of fusariosis in turtles, and recent reports have shown that this pathogen is associated with mass mortalities in natural and relocated nests of the sea turtle species, C. caretta. 3,39,42,43 The detected fungus is actually a monophyletic “species complex” (ie, F. solani species complex [FSSC]), which includes over 60 phylogenetic species. 33,34,43,45,54 This complex is widely distributed and comprises soil-borne saprotrophs that are among the most frequently isolated fungal species from soil and plant debris. The phylogenetic relationships of the F. solani species responsible for important human and plant diseases have been widely studied. 33,46 However, the F. solani isolates from sea turtles have not been similarly investigated, being limited to only a few isolates belonging to Fusarium falciforme (Multilocus sequence typing [MLST] haplotype 3+4-ww), Fusarium keratoplasticum (haplotype 2 u), or the still unnamed haplotypes 9 (a) and 12 (e). 33,35 In addition, although the occurrence of this fungus in loggerhead turtle nests is common, its role in causing skin or systemic lesions in adult sea turtles is seldom reported. 3,42,43 From an epidemiological point of view, the origin of this opportunistic infection is considered related to the presence of F. solani in rescue center tanks and to immunosuppression due to the traumatic lesions suffered, the surgical treatment applied, and other stressful conditions associated with transportation or rehabilitation of these marine turtles. 3,13
The frequent isolation of these fungal species from skin samples of sea turtles brought to our rescue centers during routine examination (unpublished data) stimulated interest in performing a larger epidemiological study by enrolling adult animals from 2 rescue centers in southern Italy with the aims of (1) evaluating the presence of Fusarium spp. in adult/subadult sea turtles with and without apparent macroscopic shell and skin lesions, (2) assessing the risk factors favoring colonization, (3) establishing the role of this fungal species in causing superficial lesions, and (4) studying the phylogenetic relationships of cultured Fusarium strains with those previously isolated from marine animals.
Material and Methods
Study Population and Sampling
From December 2010 to June 2011, 74 loggerhead sea turtles were referred to the Department of Veterinary Medicine in Bari by the WWF Sea Turtle Rescue Center based in Molfetta (Apulia, Italy) and by the WWF Sea Turtle Rescue Center in Rauccio (Apulia, Italy) for routine diagnostics after rescue (eg, fishing net entrapment, ingestion of hooks and lines, cold stunning). Each Sea Turtle Rescue Center was equipped with 6 tanks measuring 1.5 × 1.4 × 0.75 m (about 1500 l of sea water at a salinity of 35–37 per thousand). The tanks were connected to a mechanical-biological filter in a closed system with weekly water changes. The turtles were kept in individual tanks until they were set free at sea (about 2–30 days). Samples were collected from all animals that had entered the rescue center during the study period. These had typically been present in the center for 2 to 30 days or were sampled soon after being rescued from the sea. Two study groups were defined: 31 turtles with no apparent macroscopic shell or skin lesions and 43 turtles with macroscopic shell or skin lesions. The latter group had localized lesions on the carapace (n = 4), on the carapace and flipper (n = 21), on the carapace and plastron (n = 1), or generalized skin/shell lesions (n = 17). Lesions on the carapace or the exposed skin appeared as small white to light brown spots (Fig. 1).
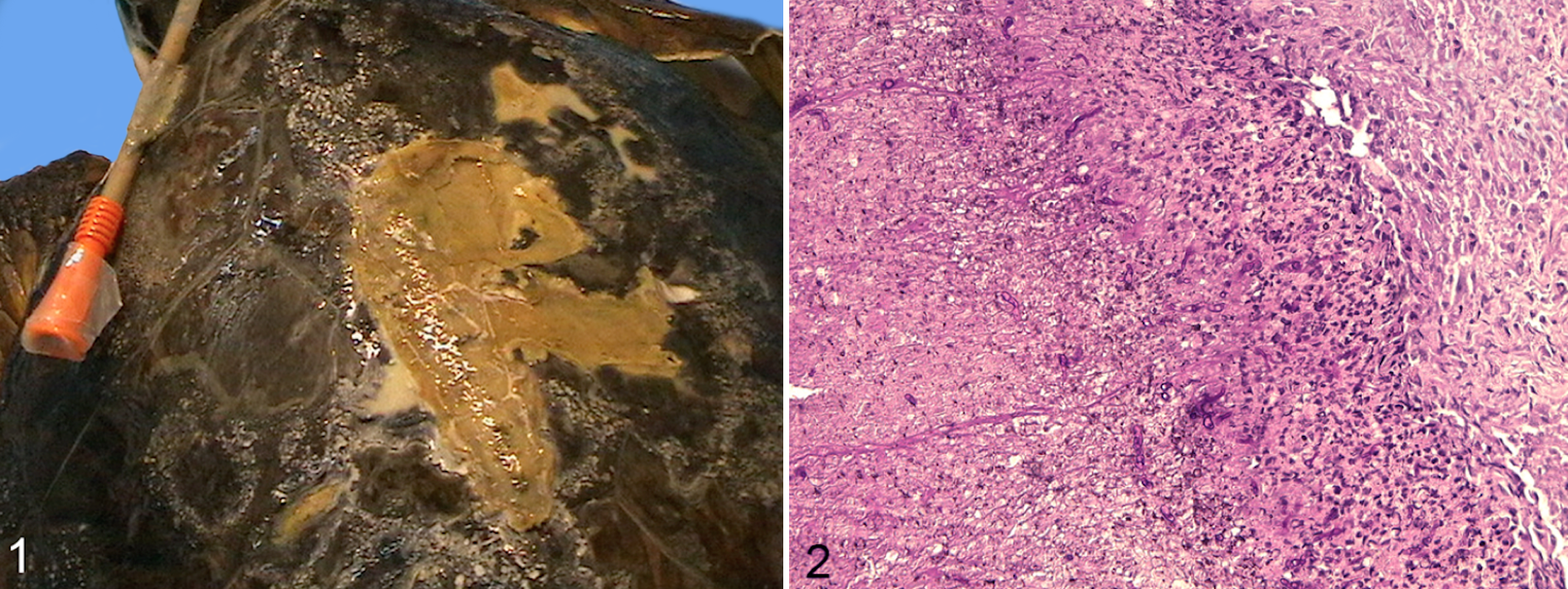
The sex of the turtles, manner of capture, and length of stay at the center were recorded into a medical history file. The turtles were classified by age into juvenile, subadult, and adult accordingly to carapace length. 12,20 To evaluate seasonal trends in Fusarium infections, the sampling period was divided into spring (from 21 March to 21 June), summer (from 21 June to 21 September), autumn (from 21 September to 21 December), and winter (from 21 December to 21 March).
Shell and skin samples were collected from each turtle (with or without lesions) from 3 different anatomical sites (ie, carapace, flipper, and plastron). Animals with no apparent macroscopic lesions were sampled using sterile lancet scribing over an area of 25 cm2 per site, whereas animals with lesions were sampled using a sterile lancet collecting material from the advancing edges of the lesions. To investigate the source of Fusarium spp., for each rescue center, 3 samples of water from the tanks (50 ml each), one of sand from the filters, and one of fresh water (50 ml) entering the tanks were collected during the study period.
Microscopic Examination and Scanning Electron Microscopy
Shell and skin samples from all enrolled animals were examined using Calcofluor white (Calcofluor White Droppers; Becton Dickinson, Sparks, USA) with 10% potassium hydroxide (KOH) for rapid microscopic fluorescent detection of fungal elements, according to the manufacturer’s instructions.
To diagnose fungal infection or colonization, 17 a histological examination was performed on shell and skin samples collected from all 43 animals with visible shell and skin lesions and from 4 or 5 sea turtles without apparent macroscopic lesions that scored positive on fungal culture. Scanning electron microscopy (SEM) was performed on 8 shell samples collected from the carapace of 4 healthy sea turtles and 4 animals with visible lesions. For histology, the samples were fixed in 10% formalin, embedded in paraffin, sectioned at 5 microns, stained with hematoxylin and eosin (HE) or periodic acid–Schiff (PAS), and examined by light microscopy (Leica DM 4000; Leica Microsystems GmbH, Nussloch, Germany).
For SEM analysis, the samples were mounted on stubs, sputter-coated with gold-palladium, and examined using a LEO S420 SEM (Leo Electron Microscopy Ltd., Cambridge, UK). 8,10
Fungal Culture and Fusarium Species Identification
All samples were first cultured in Sabouraud agar with chloramphenicol (0.5%) (Liofilchem Diagnostici, Roseto degli Abruzzi, Italy). Potato dextrose agar was used to subculture fungal strains that were not easily identified by microscopy, and only Fusarium-like organisms were further processed for identification at species level. Water collected from the tanks, sand solution, shell, and skin samples were inoculated onto Sabouraud agar and incubated at 25°C for 15 days. Water samples were centrifuged at 1000 rpm for 10 minutes and 0.1 ml of the precipitate was cultured, while sand samples were diluted in sterile saline solution (1:10), homogenized, and 0.1 mL was cultured.
Colonies grown on the medium were subcultured onto potato dextrose agar and identified to genus level based on their morphology and by microscopic examination of their hyphae and conidia. 9 All Fusarium-like isolates were subcultured onto synthetic nutrient-poor agar (SNA) and were morphologically identified at species level. 22,30,31
Multilocus Sequence Analysis of Fusarium-Like Organisms
Morphological identification to genus and species (complex) level was then confirmed by multilocus sequence analysis based on internal transcribed spacer (ITS), translation elongation 1-α (EF1-α), and RNA polymerase II second largest subunit (RPB2). 51,52 Fungal isolates were grown for 48 hours in shaker cultures (125 rpm) at 25°C in 100 ml of Wickerham medium (4% D-glucose, 0.5% peptone, 0.3% yeast extract, 0.3% malt extract). The resulting mycelium was vacuum-filtered on Whatman No. 4 filter paper, washed with distilled water, frozen, and lyophilized. About 10 mg of lyophilized mycelium was collected and used to prepare genomic DNA (DNA Purification System for Food Kit; Wizard Magnetic, Promega, Madison, WI, USA). Extracted DNA was resuspended in 100 μl of sterile water and stored at –20°C.
The general barcode ITS region was amplified using the oligonucleotide primers ITS5 and ITS4. 52 Genomic DNA (30 ng) was added to the polymerase chain reaction (PCR) mix (25 μl) containing 0.75 μl of each primer 10 μM, 2.5 μl of buffer 10×, 0.5 μl of each dNTP 10 μM, and 0.13 μl of Ampli TAQ 5 U/μl (Hot Master TAQ Polymerase 5 PRIME, Quanta Biosciences, Gaithersburg, USA). The PCR was performed in a thermal cycler (2700; Applied Biosystems). Specifically, the PCR was carried out at 94°C for 10 minutes (for polymerase activation and denaturation), followed by 30 cycles at 94°C for 30 seconds (denaturation), 55°C for 30 seconds (annealing), and 72°C for 30 seconds (extension), followed by 10 minutes at 72°C (extension). Amplicons were resolved in 1.5% w/v agarose (Ambion) gels, stained with ethidium bromide (10 mg/ml), and then photographed using the Gel Documentation System (Gel Doc 2000; BioRad). Amplicons were purified using the ExoSAP-IT Kit (Amicon, Millipore, Bedford, MA) and then sequenced directly using the TAQ DyeDeoxyTerminator Cycle Sequencing Kit (v.2; Applied Biosystems) in an automated sequencer (ABI-PRISM 377; Applied Biosystems, CA, USA). Sequences were determined from both strands (using the same primers individually as for the PCR) and the electropherograms visually analyzed, and where necessary, the sequences were corrected. For the additional multilocus gene amplification, the sequencing of part of the EF1-α gene and RPB2 gene was done as described. 8 In addition, using multilocus-based maximum likelihood phylogenetic analysis, Fusarium isolates were identified by pairwise sequence alignment query against the Fusarium MLST database (http://www.westerdijkinstitute.nl/fusarium) and the GenBank databases (http://www.ncbi.nlm.nih.gov).
Statistical Analysis
Data were analyzed using the χ2 test using the SPSS statistical package and a binomial test using BioEstat 5.0. 2 They were considered statistically significant when P ≤ .05.
Results
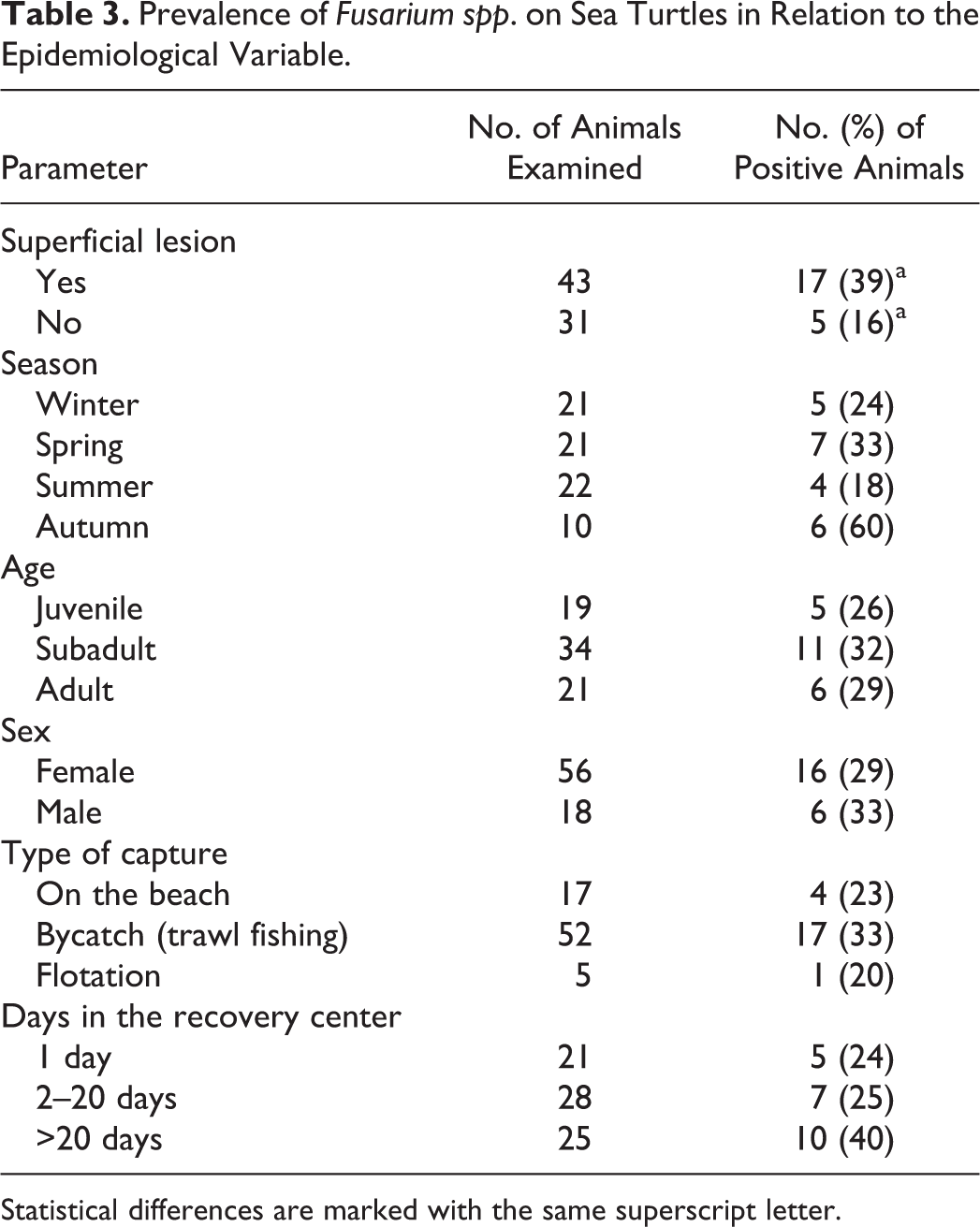
Out of 74 animals, 65 (88%) were positive on fungal culture. A total of 7 fungal genera were isolated from at least 1 anatomical site of the enrolled animals (Table 1). Penicillium spp. (43%) predominated, followed by Fusarium-like organisms (30%), which were the most frequently cultured fungal species in both study groups (Table 1). The occurrence of Penicillium spp. was higher in animals with no macroscopic lesions (52%) than from those with macroscopic lesions (37%), whereas Fusarium spp. occurred more frequently in animals with macroscopic lesions (39%) than those with no macroscopic lesions (16%). In animals with localized lesions, Fusarium spp. were most frequently isolated from the site of the lesions (Table 2).
Isolation of Different Fungi From Sea Turtles With and Without Macroscopic Lesions of the Skin or Shell.
Two samples were positive to 2 fungal species (Rodothorula spp. + Penicillium spp., Fusarium spp. + Penicillium spp., Alternaria spp. + Penicillium spp., or Aspergillus spp. + Penicillium spp.).
Isolation of Fusarium spp. From the Carapace, Flipper, or Plastron of Sea Turtles With and Without Macroscopic Lesions.

The data show the number of animals with each combination of lesions and fungal isolation.
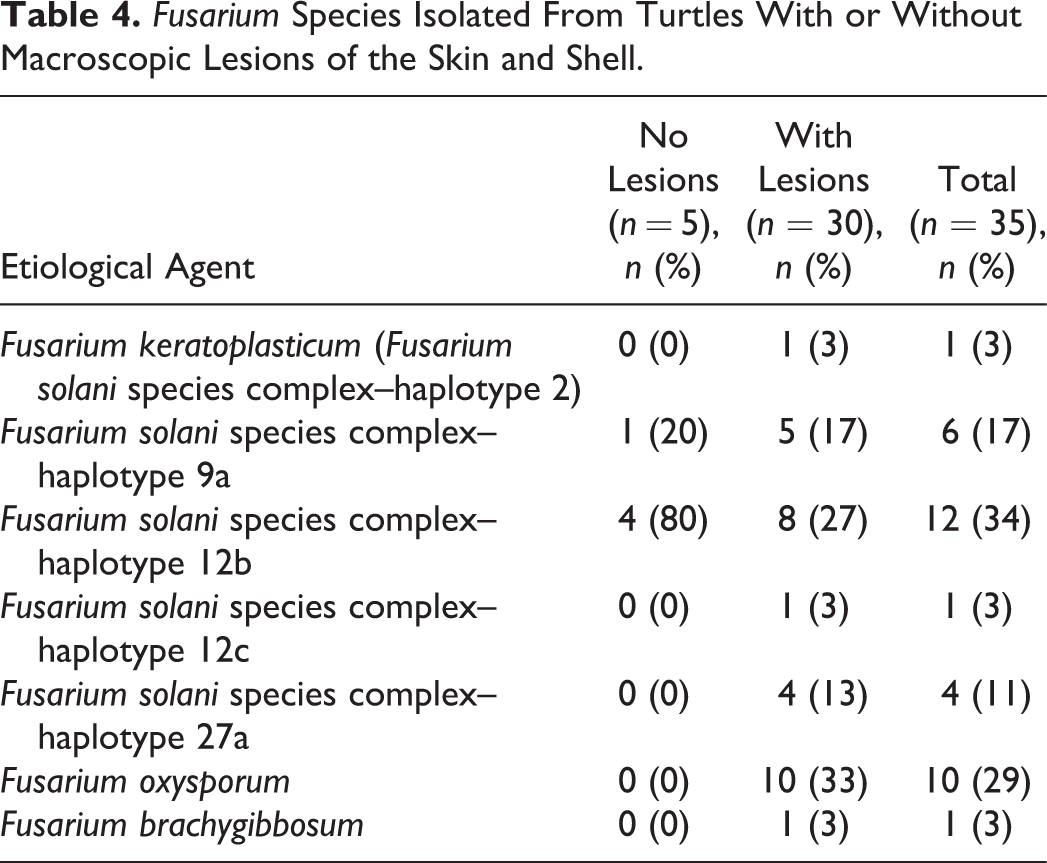
Risk factors for the occurrence of Fusarium spp. from sea turtles are listed in Table 3. The presence of superficial lesions (binomial test: Z = 2.1734, P = .0149; Table 3) proved to be a predisposing factor for Fusarium spp. colonization or infection (P ≤ .05; Table 3). In addition, turtles captured during the autumn season in trawl nets were more predisposed to Fusarium spp. colonization or infection than those captured during other seasons and with other capture methods. Finally, turtles that stayed in the recovery center longer than 20 days were more often colonized/infected by Fusarium spp. (Table 3).
Prevalence of Fusarium spp. on Sea Turtles in Relation to the Epidemiological Variable.
Statistical differences are marked with the same superscript letter.
A total of 35 Fusarium strains were isolated from the 22 animals that tested positive on fungal culture. Based on multigene phylogenetic analysis as well as BLAST searches (data not shown), the Fusarium spp. isolates were identified as belonging to the FSSC (24/35, 68.6% of the isolates), to Fusarium oxysporum (10/35, 29%), or to Fusarium brachygibbosum (1/35, 3%). The Fusarium isolates from all animals that had no macroscopic lesions (5/5 100%) and some of those with macroscopic lesions (20/30; 67%) matched most closely to Fusarium keratoplasticum (FSSC-2) or to haplotypes 9(a), 12(b and c), and 27(a) (unnamed phylogenetic species; Table 4). Additional isolates from animals with macroscopic lesions included F. oxysporum (29%), a single isolate of F. brachygibbosum (3%), and some FSSC lineages (ie, haplotypes matching closely to 12c and 27a) (Table 4). Isolates matching F. solani species complex–haplotype 12b were also isolated and identified from the sand in the filters.
Fusarium Species Isolated From Turtles With or Without Macroscopic Lesions of the Skin and Shell.
Of 74 animals, only 7 (9%) scored positive for fungal elements on microscopic examination with Calcofluor (ie, 4 animals with macroscopic lesions and 3 animals with no macroscopic lesions). Histological examination identified fungi in 12 of 43 samples from animals with macroscopic lesions and 4 of 31 samples from animals with no macroscopic lesions. All samples that were positive on histological examination were also positive on fungal culture for Fusarium spp.
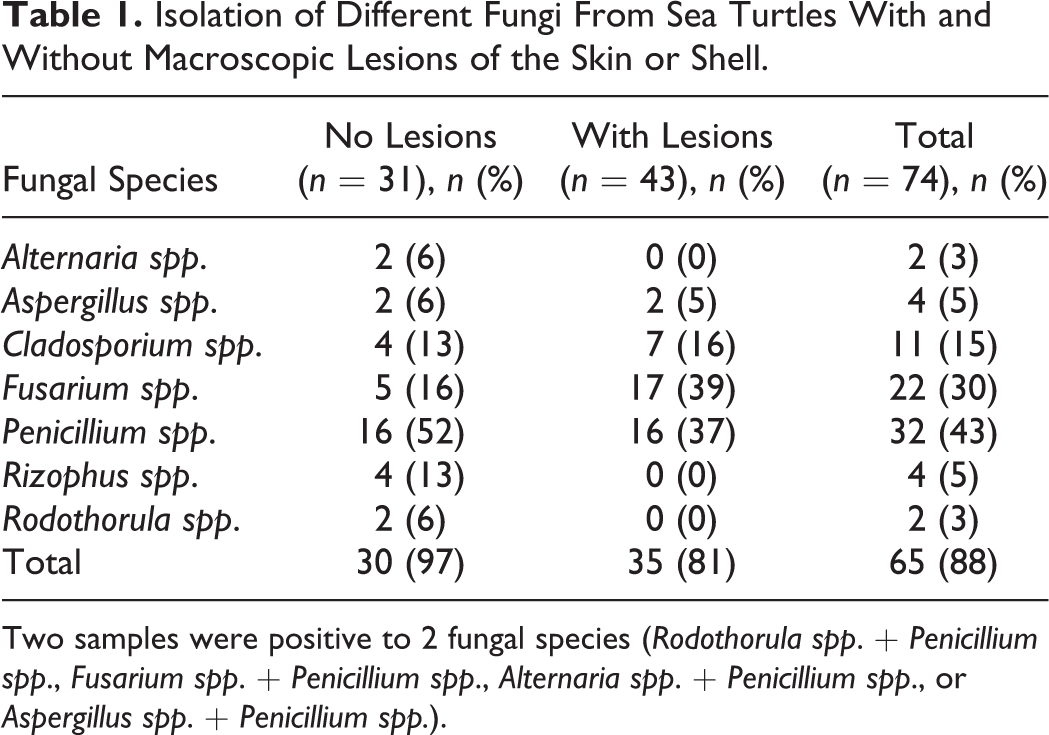
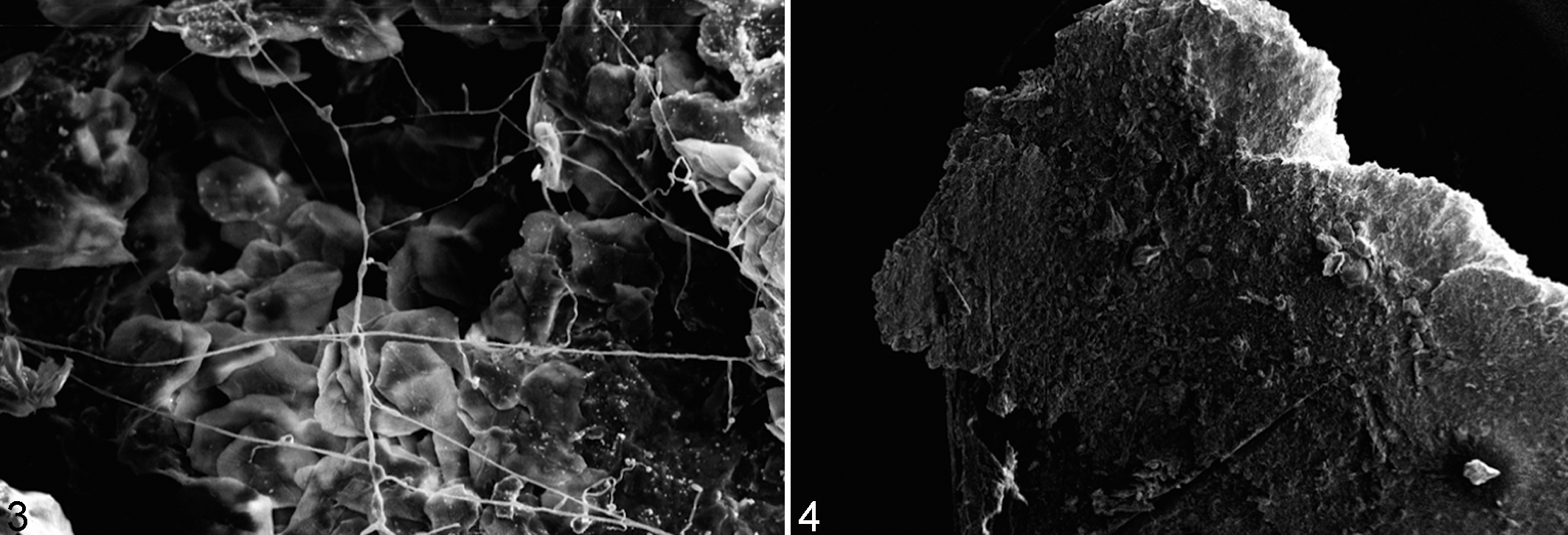
Histological examination revealed necrosis and the presence of inflammatory cells (heterophils, monocytes, and lymphocytes) at the level of the basement membrane and in the dermis (Fig. 2). SEM confirmed the presence of fungal hyphae on and in the internal structures of the carapace in all samples from animals with lesions (Fig. 3). Small round and oval ∼50-μm-diameter granules associated with fungal hyphae were observed in 3 samples from carapaces of animals with lesions and 1 sample from an animal without. All samples in which fungal elements were examined ultrastructurally were also positive on fungal culture for Fusarium spp. In 3 animals without apparent macroscopic lesions, no fungal structures were observed by SEM (Fig. 4), and these samples were also negative for Fusarium spp. on fungal culture. All water samples collected from the tanks and fresh water entering the tank were negative for Fusarium-like organisms.
Discussion
The results of this study of adult sea turtles admitted to WWF Sea Turtle Rescue Centers in the Mediterranean show that those with and without apparent macroscopic skin lesions differ in the fungal species colonizing their skin. Penicillium spp. and Fusarium spp. were the most frequently identified fungal species, most likely due to the environmental conditions in the centers favoring their growth. 25,32 The higher frequency of Fusarium fungi in animals with lesions suggests a role of this organism in the observed skin lesions. In humans, the ability of Fusarium spp. infection to cause disease is related to factors such as host immunity and contaminated water (eg, showering) as a possible source of infection. 32 In this study, the presence of Fusarium spp. in sand samples from the water tank filter and the amounts of airborne Fusarium conidia in the environment, which are usually high during spring and autumn, 47 might have encouraged colonization of the animals’ skin. In addition, the presence of lesions might have favored the germination of conidia, thus causing an infection and a deterioration of the health status. The finding that animals admitted to the center for more than 20 days were more frequently colonized also suggests an association between Fusarium-like organisms and the skin lesions that occurred, given the presence of F. solani in the tank at the rescue center, a recognized risk factor of infection in receptive adult turtles. 3 This finding suggests that the environmental conditions and management at the rescue center might favor Fusarium spp. growth and might be the source of colonization. This latter hypothesis is supported by the fact that sand from the filter was positive for Fusarium spp. On the other hand, it could mean that colonized animals may be the source of further infections at rescue centers. The absence of this mold in the fungal culture of the water samples might be due to the high dilution of spores in the water and/or to the presence of other molds, which might inhibit Fusarium spp. growth in fungal culture.
The high presence of Fusarium spp. from the carapace, in animals both with and without apparent macroscopic lesions, might indicate that the carapace is the preferential colonization site for Fusarium spp., perhaps related to the keratin content of turtle shell and the ability of Fusarium spp. to produce keratinolytic enzymes. 15,24,28 From this site, the fungus might colonize further tissues in receptive hosts causing disease. The presence of tissue breakdown or trauma in marine animal skin could be related to the mode of capture, or it could happen during stranding or predation or within the holding tanks as already described, 7,16 resulting in a higher occurrence of Fusarium spp. infections in animals with lesions than in those without macroscopic lesions. 19
In addition, the finding of Fusarium organisms in animals with and without macroscopic lesions might suggest a role of adult turtles in spreading this fungus in the environment as well in the sand where the turtles nest and lay their eggs. Members of the F. solani species complex, such as F. falciforme (haplotypes 3 + 4) and F. keratoplasticum (haplotype 2) as well as F. oxysporum, are associated with mass mortalities in natural and relocated nests of loggerhead turtles. 38,42,44 However, even though it has been clearly demonstrated that the presence of Fusarium in eggs is of sand-borne origin, since this fungal species has never been isolated from eggs taken directly from the ovipositor of nesting females (prior to contact with the sand) or from the cloacal mucus, 42,44 the role of adult turtles in spreading this fungus in the environment cannot be ruled out and requires future investigation.
The isolation of fungal cultures of Fusarium in animals both with and without apparent macroscopic lesions suggests that fungal culture alone is not useful to diagnose infection. However, the role of Fusarium spp. in causing skin lesions has been clearly investigated by using histology or SEM analyses, given that Calcofluor white with 10% potassium hydroxide was not found to be useful for diagnosing fungal infections in this study. Calcofluor white (CFW) is a nonspecific fluorochrome that binds to cellulose and chitin, which are components of the fungal cell wall, through intercalation into nascent chitin chains. It is a high-sensitivity stain for detecting fungal elements (eg, Fusarium-like organisms) in corneal scrapings or Pneumocystis jirovecii in bronchoalveolar lavage fluid, 1,5,11,26 but it was no useful for diagnosing fungal infection in sea turtles. Both histological examination and SEM analysis of selected samples clearly suggest that Fusarium spp. might have a role in causing superficial lesions in susceptible sea turtles. Even though SEM analysis is not a desirable technique for diagnosing fungal infection, due to its technical difficulties, it provides relevant information on fungal adhesion, growth, and colonization by revealing the presence of fungal hyphae on the surface and inside the carapace tissues of animals with lesions. 5,6 The constant presence of small granules in samples testing positive for Fusarium spp. in animals with and without lesions suggests that these structures might favor colonization and/or the initial stages of Fusarium infection. Future studies are needed to better address this hypothesis.
Based on multilocus sequencing, our isolates belonged to F. solani and F. oxysporum species complexes or to F. brachygibbosum. Some FSSC lineages (ie, the haplotypes matching to 12b/c and 27a) and F. oxysporum and F. brachygibbosum were identified for the first time as pathogens on turtles with skin lesions. Some of these FSSC lineages (ie, haplotypes 2 and 12) have previously been isolated from turtle, shark, and seahorse (Suppl. Table S1), 35,41 suggesting that marine-adapted lineages are involved in infections of marine, cold-blooded animals. The F. solani species complex was the most prevalent Fusarium species cluster in animals with and without lesions, but various Fusarium spp. were isolated from animals with lesions, thus suggesting that fungal culture is necessary to correctly diagnose fungal infection in turtles. Since the distribution of Fusarium species varies with geographic region 51 and different species have different drug susceptibility patterns, 32 accurate species assignment is important for epidemiological studies and to guide clinical management. Even though there is no correlation between FSSC species and antifungal susceptibility, F. solani and Fusarium verticillioides are usually resistant to azoles and exhibit higher amphotericin B minimal inhibitory concentration (MICs) than other Fusarium spp. 32,51 In contrast, F. oxysporum may be susceptible to voriconazole and posaconazole. 32,50
In conclusion, the results of this study suggest that fusariosis should be included in the differential diagnosis of shell and skin lesions in sea turtles and that proper species-level identification should be performed to enable proper treatment and infection control. Conversely, direct examination using Calcofluor and KOH was not useful, so a histopathological examination or SEM analysis as well as fungal culture should be performed to diagnose or confirm infection. 3 Furthermore, since infection was mainly found in animals admitted for more than 20 days to a rescue center, good management practices should be employed to prevent the risk of Fusarium infection in sea turtles.
Supplemental Material
Supplemental Material, Cafarchia_et_al_Supplemental_Table_S1 - Fusarium spp. in Loggerhead Sea Turtles (Caretta caretta): From Colonization to Infection
Supplemental Material, Cafarchia_et_al_Supplemental_Table_S1 for Fusarium spp. in Loggerhead Sea Turtles (Caretta caretta): From Colonization to Infection by Claudia Cafarchia, Romina Paradies, Luciana A. Figueredo, Roberta Iatta, Salvatore Desantis, Antonio Vito Francesco Di Bello, Nicola Zizzo and Anne D. van Diepeningen in Veterinary Pathology
Footnotes
Notes
Acknowledgements
We thank the WWF rescue center in Molfetta (BA, Italy) and the sea turtle rescue center in Rauccio (LE, Italy).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
