Abstract
Panulirus argus virus 1 (PaV1) is the first and only naturally occurring pathogenic virus described in the Caribbean spiny lobster, Panulirus argus. PaV1 infection in decapod species that commonly co-occur with P. argus, including the spotted spiny lobster Panulirus guttatus, has not been previously described. In 2016, 14 Caribbean and 5 spotted spiny lobsters were collected near Summerland Key, Florida, to supplement the resident population of the Audubon Aquarium of the Americas in New Orleans, Louisiana. After 5 months in quarantine, Caribbean and spotted spiny lobsters began to exhibit clinical signs of lethargy and dying in the molt. Initial histologic evaluation revealed intranuclear inclusion bodies in circulating hemocytes in the spongy connective tissue of the epidermis, suggesting a viral infection. Samples of hepatopancreas and hemolymph from deceased Caribbean and spotted spiny lobsters tested negative for white spot syndrome virus and positive for PaV1 using real-time quantitative polymerase chain reaction (qPCR). Intranuclear, eosinophilic to amphophilic, Cowdry type A inclusion bodies observed primarily within fixed phagocytes and circulating hemocytes in the hepatopancreas of freshly euthanized Caribbean spiny lobsters were consistent with PaV1 infection. Transmission electron microscopy revealed that hemocytes associated with hepatopancreatic tubules contained viral inclusions with location, size, and morphology consistent with previously described PaV1 infection. These findings highlight the significance of using molecular diagnostics in conjunction with histopathology and electron microscopy in the investigation and diagnosis of PaV1 in spiny lobsters. Further study is required to investigate the relationship of PaV1-associated mortality events and microscopic lesions in the spotted spiny lobster.
Keywords
Panulirus argus virus 1 (PaV1) is the first and only described naturally occurring pathogenic virus of juvenile Caribbean spiny lobsters, Panulirus argus.4,11–13,18,19 First discovered over 2 decades ago in the Florida Keys, 19 the virus has since spread throughout the Caribbean, affecting geographical regions including St. Croix (US Virgin Islands), 5 the Yucatan Peninsula (Mexico), 10 Belize, 9 Cuba, 7 as well as Puerto Rico, the Dominican Republic, the Bahamas, Honduras, and Panama. 14 It has been suggested that PaV1-infected planktonic postlarvae of the Caribbean spiny lobster may play a role in the environmental dispersal of the virus.14,15 Local prevalence of PaV1 can reach over 70% in some areas, not including subclinical detection of viral infection. 4
The Caribbean spiny lobster is a major economic resource for commercial and recreational fisheries in the South Atlantic and Gulf of Mexico. According to the National Oceanic and Atmospheric Administration Fisheries’ commercial fishing landings database, landings of Caribbean spiny lobsters in the South Atlantic and Gulf of Mexico reached a total of 4.7 million pounds with a value of $42.2 million in 2021. 16 Since its discovery, PaV1 has been established as one of the most significant pathogens contributing to a decline in spiny lobster fisheries.18,19
Currently, there are no published reports of PaV1 naturally infecting other species of co-occurring decapods such as the spotted spiny lobster (Panulirus guttatus). A previous study utilizing wild-caught, experimentally infected spotted spiny lobsters, stone crabs (Menippe mercenaria), and channel clinging crabs (Mithrax spinosissimus) was unable to demonstrate disease after inoculation with PaV1-infected hemolymph. 5 The absence of PaV1 infection was based solely on a lack of clinical signs and histologic lesions in examined tissues including the hepatopancreas, heart, gills, hindgut, and connective and hematopoietic tissues. Animals were not molecularly screened prior to inclusion in the study or tested postmortem to identify the presence of asymptomatic carriers.
This report describes a mortality event affecting a group of 19 wild-caught Caribbean spiny lobsters (P. argus) and spotted spiny lobsters (P. guttatus) exhibiting clinical signs suggestive of an infectious disease while undergoing quarantine at the Audubon Aquarium of the Americas in New Orleans, Louisiana. Clinical and gross examination, histopathology, transmission electron microscopy (TEM), and molecular diagnostics were used in conjunction with each other to identify the pathogen and investigate the nature of this event.
Materials and Methods
Animal Collection and Clinical History
In September 2016, 14 Caribbean spiny lobsters and 5 spotted spiny lobsters were collected near Summerland Key, Florida, to supplement the resident population at the Audubon Aquarium of the Americas in New Orleans, Louisiana. The small adult lobsters, measuring approximately 20 to 30 cm in total length, were transported and held in separate quarantine tanks at Audubon’s Aquatic Center, housed at the Freeport McMoran Audubon Species Survival Center. After 5 months in quarantine, Caribbean and spotted spiny lobsters began exhibiting clinical signs of lethargy and dying in the molt. Initially, 3 sections of formalin-fixed carapace, 2 fresh vials of hemolymph, and 2 fresh sections of hepatopancreas from the affected Caribbean spiny lobsters were submitted to the Louisiana Animal Disease Diagnostic Lab (LADDL) at Louisiana State University School of Veterinary Medicine for diagnostic evaluation.
Histopathology and Ancillary Testing
Formalin-fixed carapace was processed routinely, embedded in paraffin, and sectioned and stained using hematoxylin and eosin according to standard laboratory procedures for histologic evaluation. Homogenized samples of fresh frozen hepatopancreas were submitted to the molecular diagnostic laboratory at LADDL to screen for white spot syndrome virus (WSSV) using a modified quantitative TaqMan polymerase chain reaction (qPCR) previously described by Durand and Lightner.2,8 Fresh hemolymph collected from the middle joint of the walking legs was streaked across plates of blood agar for bacterial culture. Based on initial findings, additional samples of hemolymph from 6 Caribbean spiny and 3 spotted spiny lobsters were collected in vials of lithium heparin and 90% ethanol and submitted to the University of Florida for PaV1 qPCR following previously established methods.6,13
Due to ongoing clinical signs of disease, 4 remaining moribund Caribbean spiny lobsters were examined and subsequently euthanized in 10% magnesium chloride in 100% ethyl alcohol. Tissues including the carapace, gill, hepatopancreas, midgut, and hindgut were fixed in Davidson’s solution and in 2.5% glutaraldehyde in 0.1M cacodylate buffer supplemented with 5% sucrose. Davidson-fixed tissues were processed routinely, embedded in paraffin, and sectioned and stained using hematoxylin and eosin according to standard laboratory procedures for histologic evaluation.
TEM
Glutaraldehyde-fixed tissues were processed for TEM. Briefly, samples were postfixed in 1% osmium tetroxide, dehydrated in ascending ethanol series and propylene-oxide, and embedded in Epon-Araldite. 21 Thin 70-nm sections were stained with uranyl acetate and Reynold’s lead citrate and examined in a JEOL JEM 1011 transmission electron microscope equipped with HAMAMATSU ORCA-HR digital camera (Tokyo, Japan).
Results
Histopathology and Ancillary Testing
During initial histologic evaluation of the carapace samples from the Caribbean spiny lobster, numerous intranuclear viral inclusion bodies were observed in circulating hemocytes within the spongy connective tissue underlying the cuticle. Nuclei were frequently enlarged and contained amphophilic to basophilic viral inclusion bodies, which were often surrounded by a clear halo with margination of chromatin along the nuclear membrane (Cowdry type A inclusion bodies). Some inclusions were more eosinophilic, lacking a distinct halo with a distorted, smudged appearance to the nuclei. The fresh samples of hepatopancreas were negative for WSSV using qPCR. Four out of 6 Caribbean spiny lobster and 1 out of 3 spotted spiny lobster hemolymph samples tested positive for PaV1 using qPCR, with interpretation ranging from light to heavy infection (Table 1). Bacterial culture identified Vibrio sp. in 1 of 2 Caribbean spiny lobster hemolymph samples submitted to LADDL.
Results of Panulirus argus virus 1 (PaV1) real-time quantitative polymerase chain reaction (qPCR) in Caribbean spiny lobsters (Panulirus argus, n = 6) and spotted spiny lobsters (P. guttatus, n = 3).
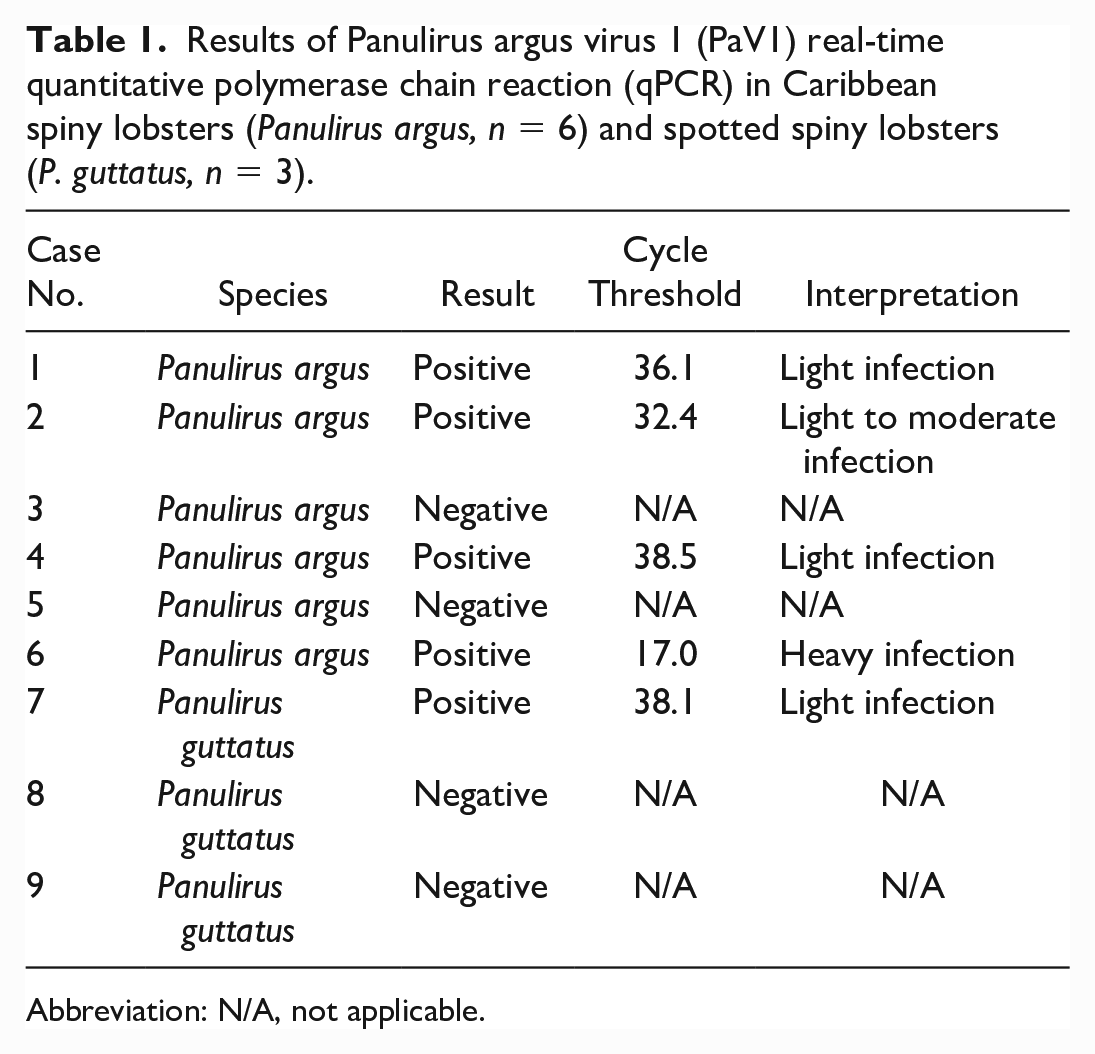
Abbreviation: N/A, not applicable.
There were no apparent gross lesions on the 4 remaining, moribund Caribbean spiny lobsters prior to euthanasia. Histologic evaluation of the Caribbean spiny lobster tissues revealed significant pathological changes primarily affecting the hepatopancreas. In comparison to adjacent hepatopancreatic tubules, the hemal sinuses were variably dilated and filled with moderate to abundant circulating hemocytes and proliferative spongy connective tissue (Fig. 1a). Fixed phagocytes were variably enlarged and exhibited disruption of their typical pseudorosette-like arrangement surrounding a central arteriole. Circulating hemocytes, proliferative spongy connective tissue cells, and fixed phagocytes frequently contained hypertrophied nuclei containing large eosinophilic to amphophilic intranuclear inclusion bodies (Fig. 1b), similar to those identified in the spongy connective tissue underlying the cuticle. In the most severely affected lobsters, tubules throughout the hepatopancreas had sparse lipid vacuolation, consistent with atrophy. In addition to the hepatopancreas, intranuclear inclusion bodies were observed within circulating hemocytes in the midgut (Fig. 1c), epidermis (Fig. 1d), and gill lamellae (Fig. 1e).
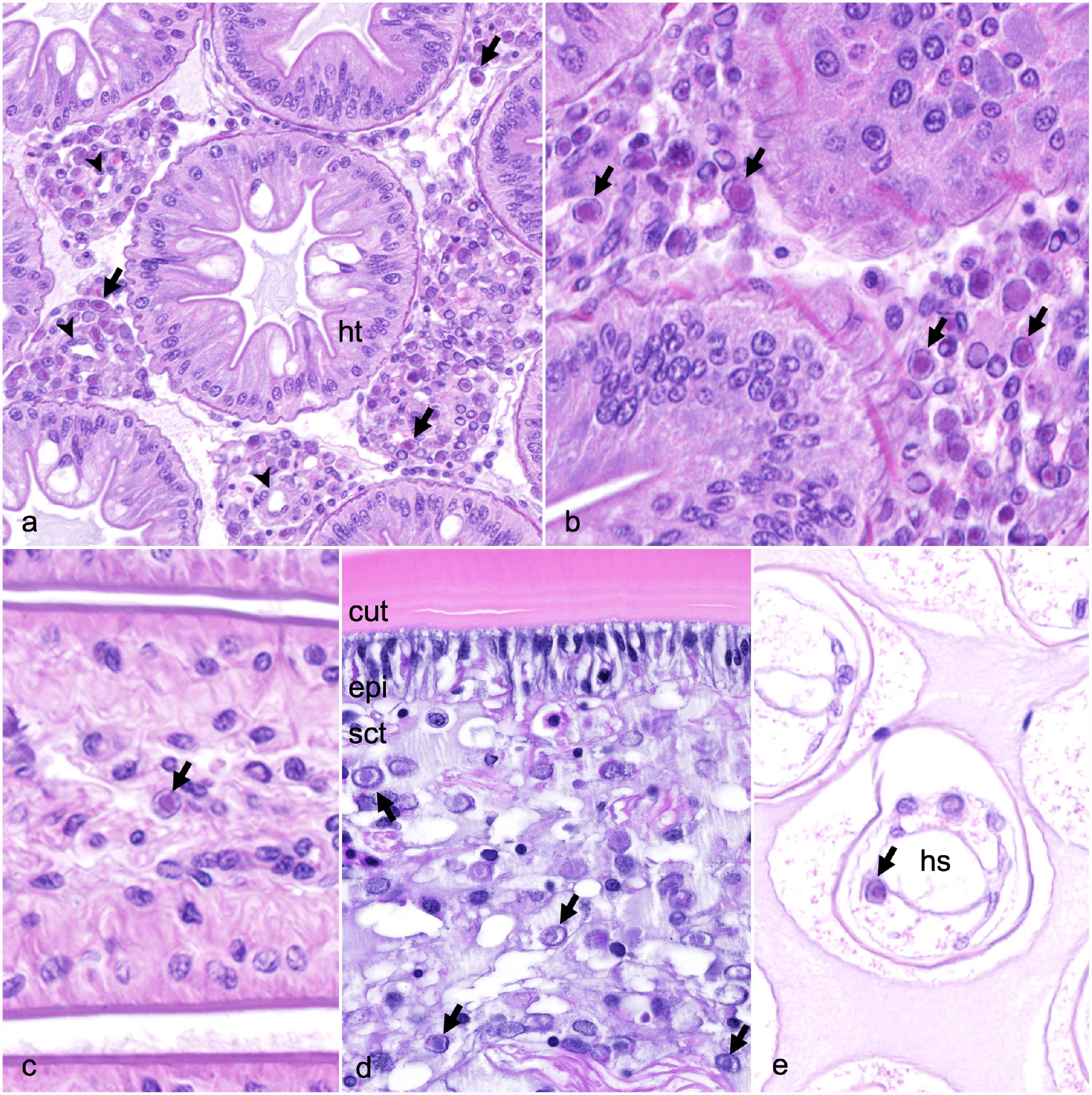
Panulirus argus virus 1 infection, Caribbean spiny lobsters. Hematoxylin and eosin (HE). (a) Hepatopancreas. Atrophied hepatopancreatic tubules (ht) are separated by variably dilated hemal sinuses containing a central arteriole (arrowheads) surrounded by a pseudorosette of proliferative fixed phagocytes, spongy connective tissue, and circulating hemocytes containing numerous intranuclear inclusion bodies (arrows). (b) Hepatopancreas. The nuclei of fixed phagocytes and circulating hemocytes are frequently enlarged and contain large eosinophilic inclusions (arrows), often surrounded by a clear halo with margination of the nuclear chromatin (Cowdry type A inclusion bodies). (c) Midgut. Eosinophilic intranuclear inclusion body within a circulating hemocyte (arrow). (d) Carapace. Endocuticle (cut) lined by columnar epithelium (epi) with underlying spongy connective tissue (sct). Circulating hemocytes throughout the spongy connective tissue contain intranuclear basophilic to amphophilic inclusion bodies (arrows). (e) Gill. Cross section through the lateral portion of the gill lamellae. A circulating hemocyte within the hemal sinus (hs) contains an intranuclear inclusion body (arrow).
TEM
TEM of the Caribbean spiny lobster hepatopancreas supported the results of histologic examination. Hemocytes infiltrating connective tissue in the vicinity of hepatopancreatic tubules displayed conspicuous signs of infection (Fig. 2a). Phagocytes were moderately to markedly distended with vacuoles containing viral particles, virogenic stroma, membrane bodies associated with viral transport, and debris derived from the cells destroyed by the virus. Hemocyte nuclei, in which proliferation and assemblage of viral particles occurred, were enlarged, contained numerous viral inclusions, and exhibited electron lucent nucleoplasm with remnants of chromatin at the periphery (Fig. 2a, b). Assemblage of virions within the nuclei included several stages of capsid morphogenesis and enveloping (Fig. 2c–e). In immature virions, the electron lucent core was surrounded by a multilayered membrane-like sheath transformed into crystalline capsid structure in mature virions. Immature viral particles were often associated with characteristic tubular structures (Fig. 2a, e). Mature virions displayed an icosahedral nucleocapsid surrounded by a bilayered envelope. An electron-dense core (presumably a nucleoid) was seen in some virions (Fig. 2e, insert). On sections, enveloped virions measured 183 nm ± 14.7 (SD, n = 12) ranging from 156 to 209 nm. The diameter of nucleocapsids without enveloping membranes was 150.7 ± 8.7 nm (SD, n = 12) ranging from 140 to 162 nm.
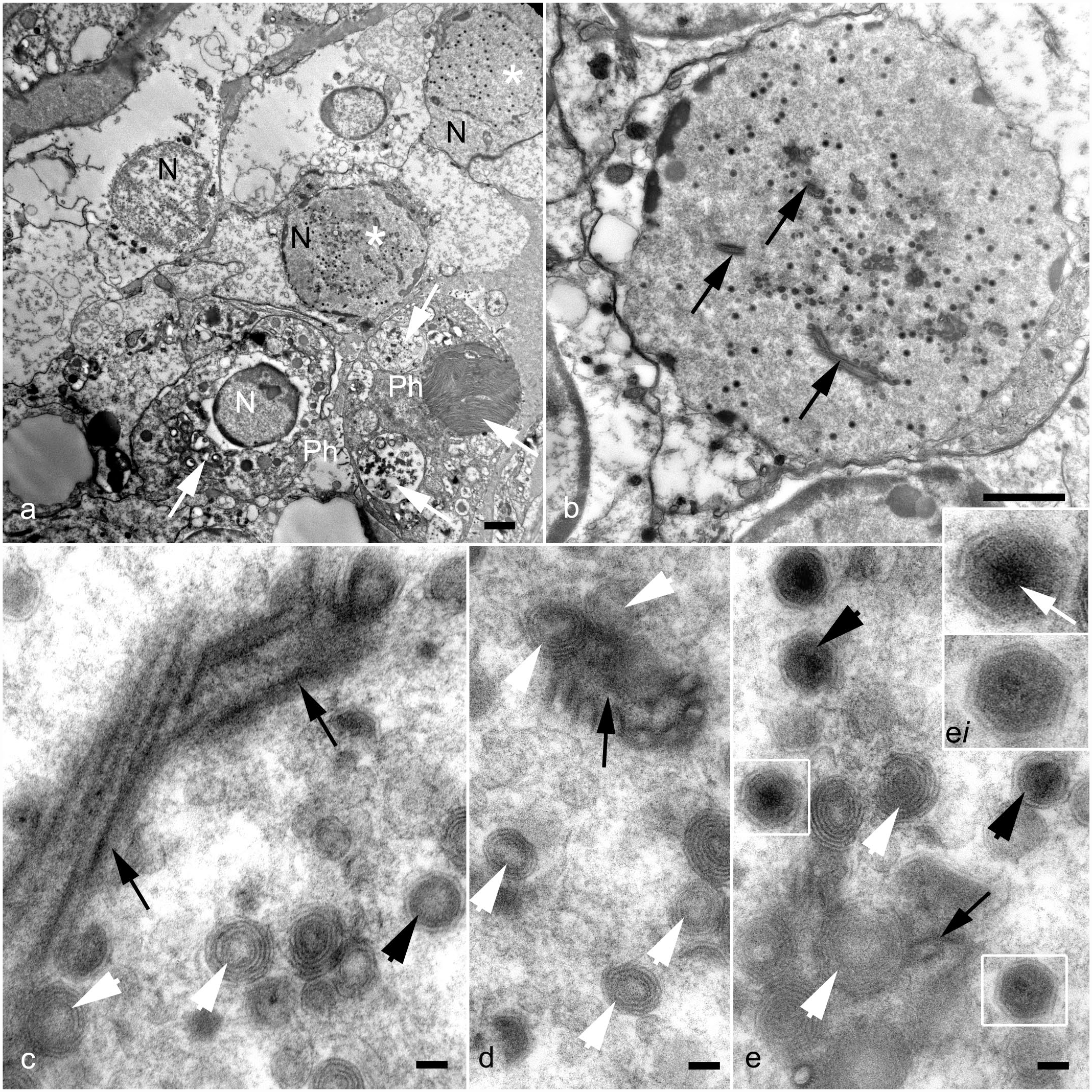
Panulirus argus virus 1 infection, Caribbean spiny lobsters. Transmission electron microscopy. (a) Hemocytes in vicinity of hepatopancreatic tubules display conspicuous signs of infection: phagocytes (Ph) are full of vacuoles containing cell debris derived from the cells destroyed by the virus (white arrows). Hemocyte nuclei (N), in which virus proliferation occurs (white asterisks), are enlarged, and contain numerous viral inclusions. (b) Infected nuclei exhibit electron lucent nucleoplasm with remnants of chromatin at the periphery, and numerous virions at different stages of morphogenesis. Characteristic Panulirus argus virus 1-associated tubular structures are indicated by black arrows. (c–e) Assemblage of virions in the nucleoplasm. Immature virions (white arrowheads) display an electron lucent core surrounded by a multilayered membrane-like sheath. Immature virions are often seen adjacent to the rod-shaped tubular structures (black arrows) suggesting the role of these structures in virus morphogenesis. (d) A cross section of the rod-shaped tubular structure is apparent. (e) Mature virions (black arrowheads; c, e) are characterized by an icosahedral nucleocapsid of crystalline structure, a bilayered envelope, and an electron-dense core, presumably a nucleoid (white arrow, inset). Scale bars: Fig. 2a, b, 2 µm. Fig. 2c–e, 100 nm.
Discussion
Two critically important viral diseases of crustaceans, white spot syndrome virus (WSSV) and Panulirus argus virus 1 (PaV1), were highly suspected and considered as potential differential diagnoses based on the clinical signs and histologic findings in this captive group of Caribbean spiny lobsters (Panulirus argus) and spotted spiny lobsters (P. guttatus). WSSV is a well-established, devastating viral disease of several species of penaeid shrimp, and was more recently identified as a significant cause of morbidity and mortality in Louisiana red swamp crayfish (Procambarus clarkii).2,23 While WSSV has been experimentally induced in 6 species of Panulirus lobsters, it has not been documented in the wild. 17 Based on the histologic appearance of the basophilic intranuclear inclusion bodies resembling WSSV in shrimp and crayfish, WSSV was considered a primary differential diagnosis and ruled out following negative qPCR results.
PaV1 is a devastating viral disease affecting wild Caribbean spiny lobsters. The virus is an unclassified, nonenveloped, icosahedral DNA virus with a nucleocapsid of approximately 182 nm that shares characteristics with Herpesviridae and Iridoviridae.18,19 PaV1 causes the highest morbidity and mortality in early benthic juveniles, while adults tend to serve as asymptomatic carriers of the virus.4,18 Histologic lesions are typically observed within 10 to 15 days of infection; the most significant changes are characterized by infection of fixed phagocytes and circulating hemocytes, including hyalinocytes and semigranulocytes, resulting in enlarged nuclei containing Cowdry-like eosinophilic inclusion bodies.4,11 Consistent with published reports, the most significant histologic findings in these lobsters were localized within the hepatopancreas. In late stages of infection, the hemolymph develops a characteristic milky, white appearance and fails to clot.18,19
In most decapod crustaceans, the hepatopancreas is a large, compact, paired glandular organ occupying the cephalothorax and surrounding the midgut.1,20 The hepatopancreas of crustaceans, along with various aquatic species, is normally filled with lipid reserves appearing as vacuolation of hepatocytes, which is a useful marker of nutritional and metabolic status. Lack of adequate vacuolation in severely affected lobsters is highly suggestive of metabolic wasting, which has been associated with mortality due to PaV1 infection 13 ; however, it is uncertain whether increased stress due to capture and captivity, inadequate nutrition, or any combination of these factors may have also contributed in this case.
Molecular diagnostics including PCR and fluorescent in situ hybridization (FISH) aid in confirmation of PaV1.12,13,18 In our case, the positive qPCR for PaV1 in the Caribbean spiny and spotted spiny lobsters, together with the histopathology and TEM findings in the Caribbean spiny lobsters, indicates PaV1 as the cause of disease and mortality in the subset of captive Caribbean spiny lobsters. While the diagnostic sensitivity for the TaqMan real-time qPCR is reported at 100%, it has a diagnostic specificity of 84%, which raises some concern for potential false positive results. 6 This further emphasizes the need for additional diagnostic methods, such as histopathology and/or TEM, to confirm the diagnosis of PaV1 in the group of captive spotted spiny lobsters.
Unfortunately, the group of deceased spotted spiny lobsters in this report were too severely autolyzed to confirm a histologic diagnosis of PaV1 in conjunction with the molecular diagnosis. However, clinical signs in association with the molecular detection of virus and subsequent mortality suggest that PaV1 infection played some role in the death of these lobsters. It is uncertain if stress related to the capture, handling, and subsequent captivity of these lobsters contributed to potential immunosuppression and expression of disease. Further study would be indicated to help correlate the clinical and molecular diagnostic findings with any microscopic evidence of viral infection.
The viral morphology of PaV1 and similar crustacean viruses has been progressively compared and linked to the morphology of iridoviruses, which are members of a diverse group of nucleocytoplasmic large DNA viruses. 22 One distinguishing characteristic of PaV1 in contrast to other iridoviruses and nucleocytoplasmic large DNA viruses is the completion of viral morphogenesis within the infected host cell nucleus as opposed to the cytoplasm. Based on this unique characteristic and recent phylogenomic analysis, a new classification under a distinct family, “Mininucleoviridae,” has been suggested for PaV1 and similar crustacean viruses. This new classification has yet to be formally acknowledged by the International Committee on Taxonomy of Viruses; at present, PaV1 remains an unclassified DNA virus.
The ultrastructural analysis performed in this study demonstrated viral infection of hepatopancreatic hemocytes, intranuclear assemblage of virions, the presence of characteristic intranuclear tubular (“rod-shaped”4,19) structures and their association with viral morphogenesis, and the shape and approximate size of virions. These ultrastructural characteristics were consistent with the original description of PaV1, 19 along with recent literature highlighting shared characteristics with Iridoviridae and of nucleocytoplasmic large DNA viruses. 22 To the authors’ knowledge, these are the only 2 existing reports detailing the virus’ fine morphology.
In the wild, healthy lobsters exhibit an interesting behavior in which they tend to avoid diseased lobsters, which may aid in reduced disease transmission, but lead to increased shelter competition and increased predation of diseased animals.3,4,18 In a captive environment, healthy and diseased lobsters may have decreased opportunity for avoidance; in this case, it is presumed that shared close contact between groups of Caribbean and spotted spiny lobsters likely contributed to the introduction of the virus into the latter group. It was only by chance that these lobsters were held in quarantine for an unusually extended period, leading the authors to question the adequate length of quarantine required to prevent spread of disease to other aquarium-housed animals. Furthermore, it may be beneficial to house individual animals separately while under quarantine, when feasible.
In conclusion, the combination of clinical signs, molecular diagnostic testing, histopathology, and TEM resulted in a confirmed diagnosis of PaV1 in these captive Caribbean spiny lobsters and a presumptive diagnosis of PaV1 in the subset of spotted spiny lobsters. Interestingly, the lobsters involved in this case were considered small adults, which typically exhibit less prevalence of disease and serve as asymptomatic carriers of PaV1.4,18 Vibrio sp. identified in the hemolymph of 1 Caribbean spiny lobster was considered a secondary, incidental finding, and not likely a major contributor of disease in this case. The overall results stress the significance of evaluating clinical signs and microscopic lesions in conjunction with ancillary diagnostics, including molecular biology, virology, and bacteriology techniques. While pathologists may readily consider this diagnostic approach in domestic species, it is equally important to utilize these methods while investigating diseases in less commonly encountered species, including invertebrates. This report also illustrates the significance of adopting adequate quarantine procedures to aid in the early detection of disease, thus preventing unnecessary exposure of healthy animals.
Footnotes
Acknowledgements
The authors acknowledge the Audubon Aquarium of the Americas in New Orleans, Louisiana, for the submission of this case; members of the Histology and Molecular sections at the Louisiana Animal Disease Diagnostic Laboratory for their assistance; and the Behringer Lab at the University of Florida for providing the PaV1 molecular diagnostic testing. The authors also acknowledge Dr Mariano Carossino (Louisiana Animal Disease Diagnostic Laboratory and Department of Pathobiological Sciences, School of Veterinary Medicine, Louisiana State University) for his support in reviewing the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially supported by the Louisiana Animal Disease Diagnostic Laboratory, Louisiana State University School of Veterinary Medicine (PG009095).
