Abstract
In this report a 9-year-old female German Shepherd dog with a membranoproliferative glomerulonephritis (MPGN) type III associated with concomitant infection of Dirofilaria immitis and Leishmania infantum is presented. Light microscopic evaluation of kidney revealed a diffuse hypercellularity and thickening of glomerular basement membrane. Heavy and coarse granular complement C3 deposition and a weaker positive reaction to immunoglobulin G were present along peripheral glomerular basement membrane and in the mesangium in the immunofluorescent study. Transmission electron microscopy revealed deposits in the mesangium, subendothelium, and subepithelium. These lesions are compatible with membranoproliferative glomerulonephritis type III in humans.
Membranoproliferative glomerulonephritis (MPGN) is a histologic lesion characterized by intraglomerular hypercellularity (mesangial cells, endothelial cells, and inflammatory cells), matrix expansion, and thickening to the walls of the peripheral capillary loops. Immunocomplex deposits in the capillary walls and within mesangium are seen on electron microscopy. Membranoproliferative glomerulonephritis is usually associated with an immune complex-mediated pathogenesis and may be idiopathic (primary) or secondary to viral, bacterial, and parasitic infections. According to the World Health Organization classification, MPGN is subdivided, based on ultrastructural alterations in the glomerular basement membrane (GBM), as type I (subendothelial deposits), type II (intramembranous dense deposits), and type III. 6,7 In MPGN type III, immune deposits are found in the subendothelial and mesangial region, similarly to type I; in addition, numerous subepithelial deposits are present. 1,6,7,13,14 This work describes the features of MPGN type III in a dog associated with simultaneous infection with Dirofilaria immitis and Leishmania infantum. To the authors’ knowledge, this is the first description of MPGN type III in dogs.
A 9-year-old female German Shepherd dog was referred with a 4-week history of poliuria/polydipsia, anorexia, vomiting, and diarrhea with melena. A hematologic evaluation 3 days earlier had revealed blood urea nitrogen increase and elevated serum creatinine and antibodies to Leishmania spp. (IFAT 10 ) and D. immitis antigens (SNAP a ). On clinical examination, the animal was in lateral recumbency with a severely dull and depressed mental state (uremic status). Its body condition score was poor, and it exhibited generalized muscle atrophy, tachypnea (60/min), and tachycardia (170/min). Mucous membranes were dry and a pale-yellowish color while all lymph nodes were subjectively normal to decreased in volume. After blood and urine collection and fine needle aspiration of lymph nodes, the dog was hospitalized. Fluid therapy (lactated Ringers) was infused, and antiemetic and gastro protective therapy (metoclopramide, ranitidine) was initiated. Hematologic data showed elevated serum creatinine (13.7 mg/dl; reference range, 0.5–1.5 mg/dl); a marked increase of blood urea nitrogen (132 mmol/1; reference range, 3.6–10,1 mmol/1); hyperphosphatemia (15.3 mg/dl; reference range, 2.6–6.2 mg/dl); and a marked normochromic, normocytic nonregenerative anemia (red blood cell count 2.710 106/mmc3; reference range, 5.7–8.8 106/mmc3; hemoglobin 6.2 g/dl; reference range, 12.9–18.4 g/dl; hematocrit 17.5 %; reference range, 37.1–57.0 %) compatible with chronic renal failure. Moderate hyperproteinemia (8.1 g/dl; reference range, 5.4–7.5 g/dl), characterized by hyper-globulinemia (β and γ globulin) and moderate hypoalbu-minemia (2.3 g/dl; reference range, 2.8–3.5 g/dl) with low albumin-globulin ratio (0.4; reference range, 0.6–1.1), and detection of Leishmania spp. amastigotes in the cytologic lymph node smears confirmed the diagnosis of leishmaniasis. The urine sample was isosthenuric with a specific gravity of 1,014 (reference range, 1,015–1,045), a moderate proteinuria of 298 mg/dl, and a high protein/creatinine ratio of 4.5 (reference number, <0.6). Electrophoretic examination of the proteinuria (sodium dodecyl sulfate polyacrylamide gel electrophoresis) showed a mixed protein content suggestive of lesions within the glomerular and tubulointerstitial compartments. Due to the progression of clinical signs and the very poor prognosis, the dog was euthanized, and a postmortem examination was performed.
At necropsy, samples from all organs were fixed in 10% formalin and paraffin embedded for routine histology. Sections of kidneys were stained with periodic acid-Schiff hematoxylin and Masson's trichrome and phosphotungstic acid hematoxylin. Acid fuchsin orange G (AFOG) stain was used to demonstrate immune deposits and to value the interstitial fibrosis. An immunofluorescent study was performed on frozen sections of renal tissue, using fluorescein-conjugated polyclonal antibodies against the following: dog immunoglobulin G (IgG) (H+L), dog IgA, dog IgM, and dog complement component 3 (C3). b Immunohistochemistry to detect Leishmania antigen was performed on liver, spleen, and kidney sections using a rabbit polyclonal antibody. c The liver was pale and diffusely congested. The kidneys appeared of normal shape and size, and the spleen was increased in volume. Twenty-four filarial worms were found in the right atrium, and 3 adult filariae were found in the lungs.
Total genomic DNA was extracted from frozen kidney tissue using a commercial kit d performed under the conditions suggested by the manufacturer. DNA extract was amplified according to Ferroglio and colleagues 5 and Mar and colleagues 12 for detection of L. infantum and D. immitis DNA.
Histologically, microfilariae were found throughout the vessels of different organs: liver, lungs, spleen, and kidneys. The most significant lesion observed in the liver was a chronic granulomatous inflammatory reaction, characterized by intralobular hepatic granuloma formation with variable numbers of macrophages. Several amastigotes, immunolabeled with anti-Leishmania antigen, were detected in Kupffer cells of the liver, spleen, and kidneys (Fig. 1). Light microscopy examination of the kidney revealed diffuse enlargement of glomerular tufts and glomerular hypercellularity, attributed primarily to mesangial cell proliferation. Mesangial matrix expansion and sclerosis were particularly severe in some glomeruli, and multiple areas of mesangiolysis were present associated with microaneurisms. The glomerular tufts, in some instances, showed a prominent lobular pattern (Fig. 2). Examination of the tissue stained with periodic acid-Schiff revealed thickening of the glomerular capillary walls and occasional duplication of GBM. By AFOG stains at higher magnification, massive deposits of bright red material were visible in the capillary walls and in the mesangium (Fig. 3). Interstitial inflammatory infiltrates were diffusely distributed in the cortex and were predominantly composed of lymphocytes, macrophages, and plasma cells. Focal tubular atrophy was associated with areas of fibrosis demonstrated by staining with Masson's trichrome and AFOG. Immunofluorescence showed heavy, coarse granular complement C3 deposition along peripheral GBM and in the mesangium and a weaker positive reaction to IgG (Fig. 4). Electron microscopic evaluation of renal tissue revealed a marked endocapillary proliferation (proliferating endothelial and mesangial cells) occluding the capillary lumen. Numerous large subepithelial and occasional mesangial deposits were present. Interposition of mesangial cells between endothelial cells and GBM was also noted. The epithelial foot processes were widely effaced (Fig. 5). Polymerase chain reaction products run on 2% agarose evidenced the band of 302 bp and 145 bp, which respectively confirm the presence of D. immitis and L. infantum DNA in kidney tissues.
Renal involvement has been reported in dogs infected with D. immitis and in canine leishmaniasis. To the authors’ knowledge, this is the first report describing renal lesions associated with simultaneous infection with D. immitis and L. infantum. The histopathologic, immunofluorescent, and electron microscopic findings are consistent with a membranoproliferative glomerulonephritis type III in humans. This specific type of MPGN has never been described in a dog, because immunofluorescence and electron microscopy are not routinely used diagnostic techniques in veterinary pathology. The pathogenesis of glomerulonephritis associated to D. immitis has been widely discussed. 2 The most severe glomerular and interstitial lesions are usually observed in dogs with high microfilarial counts and long infection periods, due to prolonged release of antigenic material into the blood stream, by inducing in situ formation or trapping of preformed complexes. 18,19 In this dog the presence of heartworms in the heart and the relevance of a high count of microfilaremia confirm one possible origin of this type of glomerulonephritis. 17 Acid-fuchsin-orange-G-positive material deposited in the glomeruli, especially in the GMB, may be related to the presence of circulating complexes, as reported in experimental D. immitis infection, or to direct damage by microfilariae to the glomerular capillary endothelium. 8,15 Likewise, infection with L. infantum may have played a role in the development of the glomerulonephritis in this dog. 16 The glomerular immune deposits in people with MPGN are typically positive of C3 and often positive for IgG, as was found in this case, further supporting the immunopatho-genesis of this lesion. 3,9,11 In this case, it was not possible to define whether the renal lesion was a result of one parasite or the another. Due to the features of the glomerulonephritis observed in this dog, immunohisto-chemical results, and polymerase chain reaction analysis on renal tissue, it is possible that both infections were involved in the pathogenesis of this MPGN type III. Previous works describe lesions from both parasites in the kidney resulting from deposition of immunocomplexes. 2,16 The interstitial inflammation, associated with crescentic formations observed in some glomeruli, are indicative of the severity of the disease. Severe mesangial proliferation and significant mesangial sclerosis are likely expressions of progression of these lesions.
Membranoproliferative glomerulonephritis is considered to be the most frequent type of glomerulonephritis in dogs. 4 According to the human classification, the light microscopy features do not define the possible type of MPGN. The use of electron microscopy permits the identification of the distribution of the deposits inside the mesangium and especially in GBM. In our work, the main ultrastructural findings were mesangial, subendothelial, and subepithelial deposits; these features resemble MPGN type III, according to the World Health Organization. Further investigations to focus the mechanism leading to different sites of deposition of immunocomplexes will be helpful for a better characterization of renal disease in dogs and outcome for target therapies.

Liver; dog. Microfilaria in sinusoidal vessel and strongly immunolabeled amastigotes within Kupffer cells. Streptavidinbiotin-peroxidase complex method, 400−.

Kidney; dog. Thickened capillary walls (arrows) and an area of nodular glomerulosclerosis (arrowhead) in a glomerulus associated with interstitial nephritis (asterisk). Periodic acid–Schiff, 200−.

Kidney; dog. Deposits red colored in the capillary wall (arrow) and in the mesangium (arrowhead) with protein cast in renal tubules. Acid fuchsin orange G, 200−.
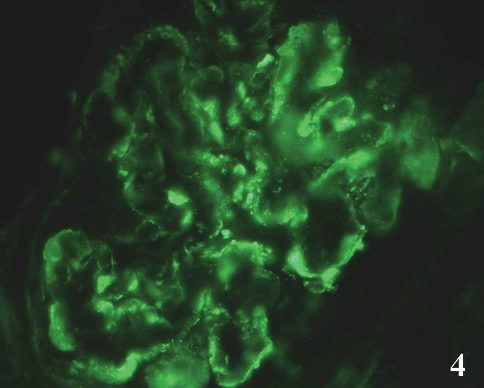
Kidney; dog. Diffuse capillary walls and mesangial granular staining of complement C3. Immunofluorescence, 400−.

Kidney; dog. Segment of capillary wall (arrow) with numerous subepithelial (asterisks) and intramembranous deposits (arrowhead) and spikes of glomerular basement membrane–like material. Transmission electron microscopy, 8,000−.
Footnotes
a.
Idexx Laboratories, Westbrook, ME.
b.
Bethyl Laboratories Inc., Montgomery, AL.
c.
Agrolabo, Scarmagno, Italy.
d.
GenomeElute, Sigma-Aldrich, St. Louis, MO.
