Abstract
Diseases affecting the hair follicle are common in domestic animals, but despite the importance of an intact skin barrier and a fully functional hair coat, knowledge about the detailed morphological features and the diversity of these complex mini-organs are often limited, although mandatory to evaluate skin biopsies with a history of alopecia. The factors that regulate the innate hair follicle formation and the postnatal hair cycle are still not completely understood in rodents, only rudimentarily known in humans, and are poorly understood in our companion animals. This review aims to summarize the current knowledge about hair follicle and hair shaft anatomy, the arrangement of hair follicles, hair follicle morphogenesis in the embryo, and the lifelong regeneration during the postnatal hair cycle in domestic animals. The role of follicular stem cells and the need for a multitude of interacting signaling events during hair follicle morphogenesis and regeneration is unquestioned. Because of the lack of state of the art methods that can be applied in rodents but are not feasible in companion animals, most of the information in this review is based on rodent studies. However, the few data from domestic animals that are available will be discussed, and it can be assumed that at least the principal molecular mechanisms are similar in rodents and other species.
In mammals, hair is an important characteristic of the skin and has a wide range of functions, such as thermal regulation; physical and immunological protection against external insults, including among others, protection against ultraviolet light and microbes; dispersion of sebum and pheromones; sensory perception; social interactions; and camouflage. 94 The body of most terrestrial mammals is covered by a large number of hair shafts (HSs), also referred to as hair fibers. The HSs are of varying length, thickness, and density, and these characteristics may vary depending on the body site. All hair fibers together form the hair coat, also referred to as fur, wool (in sheep), or just hair. The fur is produced by millions of mini-organs called hair follicles (HFs) that invaginate from the epidermis into the subcutis. A characteristic feature of HFs is that they self-renew during the hair cycle (HC) throughout the entire life of an individual to continuously produce new HSs. HF development in the embryo and the maintenance of the HC during postnatal life is a highly conserved process and tightly regulated. It involves follicular stem cells (SCs); numerous molecules derived from epithelial, mesenchymal, and neuroectodermal cells; and the extracellular matrix of the follicular and dermal environment. In addition, it is influenced by systemic factors like hormones, age, or genetics as well as environmental factors such as the time of the year.13,88 The interplay of follicular SCs and the signaling molecules triggered by the factors mentioned above results in the huge hair coat diversity of terrestrial mammals. Despite the importance of an intact skin barrier and functional HFs, our knowledge about the factors that regulate HF morphogenesis in the embryo and the postnatal HC, as well as hair diversity, are still not completely understood in rodents; only rudimentarily known in humans, Cashmere goats, and sheep; and knowledge is poor in the other mammals.
Follicular Anatomy
In terrestrial mammals, HFs can be distinguished according to their type and pattern of distribution. The current concept in the veterinary literature is that in most domestic animals, central primary HFs and lateral primary HFs produce heavy and light guard hairs (outercoat hairs). The central primary follicles emerge first during ontogenesis, whereas the lateral primary HFs develop later. The guard hairs determine the coat color in dogs and cats. Both primary HF types are associated all over the body with sebaceous glands and apocrine tubular glands, and both glands secrete into the infundibulum of the HF. Furthermore, an arrector pili muscle inserts at each primary HF. The secondary HFs produce the undercoat or wool hair and are neither associated with apocrine glands nor with an arrector pili muscle. It is assumed, but has not been shown in dogs and cats, that secondary HFs develop later during morphogenesis. For mice and pigs, it has been shown that at least 2 guard hair types and several other hair types (awl, auchene, and zigzag) exist and that their percentage distribution varies in different body location.17,42 In mice, it has been shown that the HFs that produce the different hair types are induced at different time points during HF morphogenesis, and 3 waves of HF induction have been described.17,42 The terms primary and secondary HFs are not used outside the veterinary literature. Given the huge hair coat diversity in dogs and cats, it is possible that more than just 2 guard hair types (referred to as derived from primary HFs) and 1 wool hair type exist (referred to as secondary). All HFs are surrounded by an extensive network of capillaries and nerve fibers. Whereas the bulb of the primary HFs reaches into the subcutis in some mammals, the secondary HFs are shorter, and their deep end is located in the mid to deep dermis. 59
Extending vertically from the epidermis to the base of the HF, the anagen HF can be divided longitudinally into 2 major compartments. The permanent upper segment and the lower inferior segment. The permanent part is further divided into the infundibulum and the isthmus. The inferior segment undergoes cyclical changes during HF regeneration. It regresses during catagen, is absent during telogen, and is reconstituted during the next anagen phase.18,107 The inferior segment can be further subdivided into the suprabulbar region and the HF bulb. Thus, a total of 4 longitudinally aligned anatomic regions can be distinguished in the late anagen HF (Figs. 1, 2).
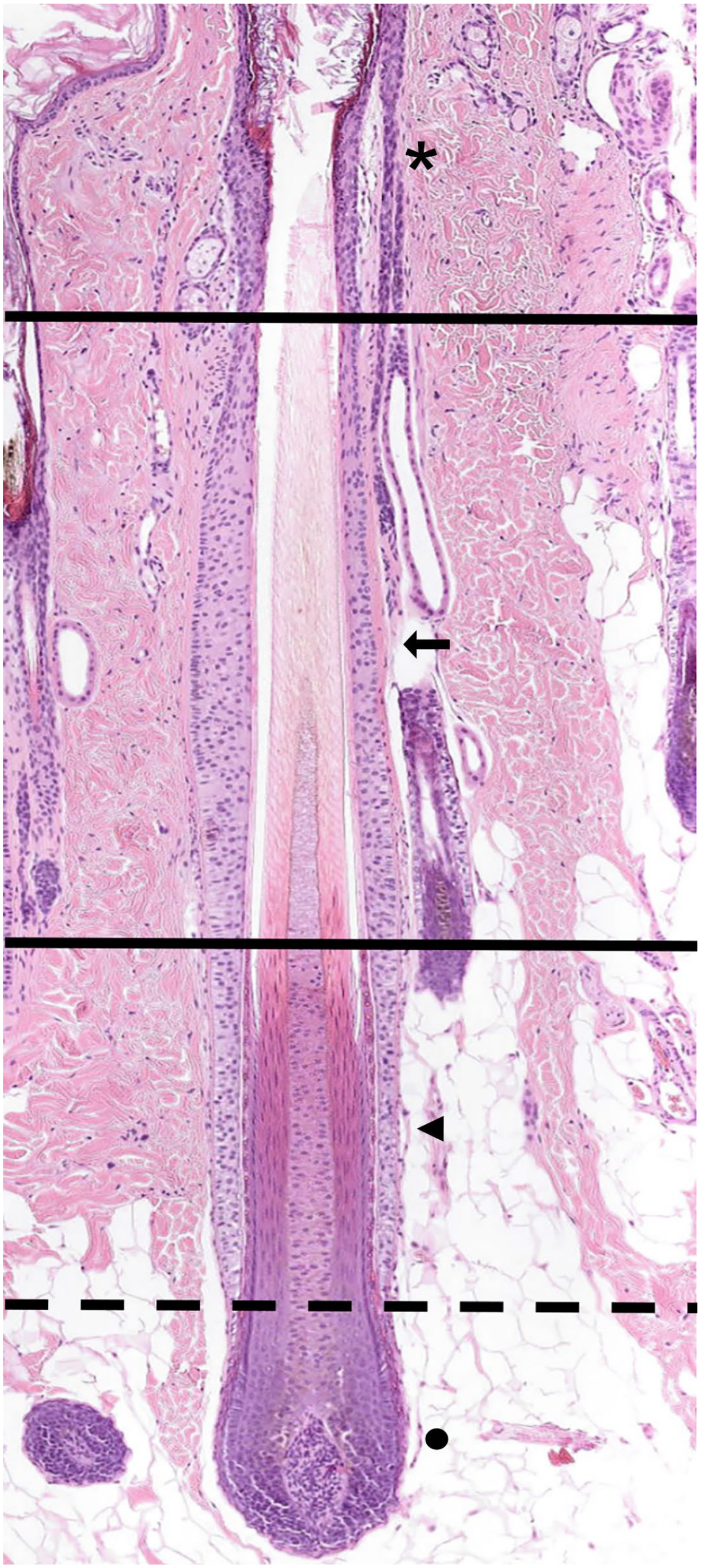
Late anagen hair follicle (HF). The anagen HF can be divided longitudinally into 2 major compartments. The permanent upper segment and the inferior segment. The permanent part is further divided into the infundibulum (black asterisk) and the isthmus (black arrow). The infundibulum extends from the orifice of the HF to a small area at which a ring of trichilemmal cornification is seen (approximately the opening of the sebaceous gland duct into the HF, upper black horizontal line). The isthmus extends from the lower end of the infundibulum to the interface between the completely cornified inner root sheath and the first noncornified cell of the Huxley layer (lower black horizontal line). The inferior segment is divided into the suprabulbar (black triangle) and bulbar regions (black dot), separated by a horizontal dashed black line. The suprabulbar region can be recognized by the presence of a not yet fully cornified inner root sheath. The bulbar region is composed of matrix cells intermingled with melanocytes that enclose the dermal papilla. Hematoxylin and eosin.
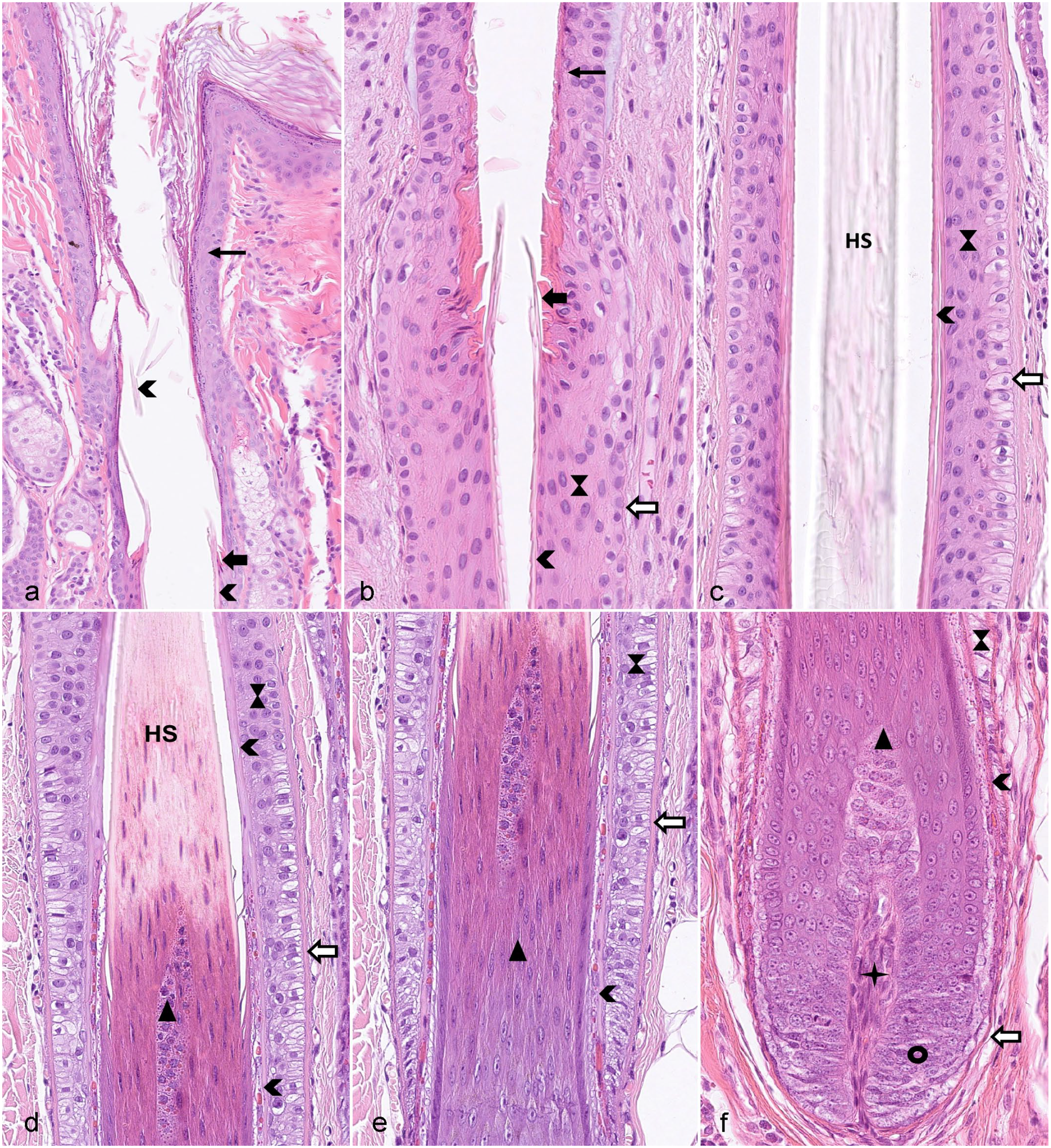
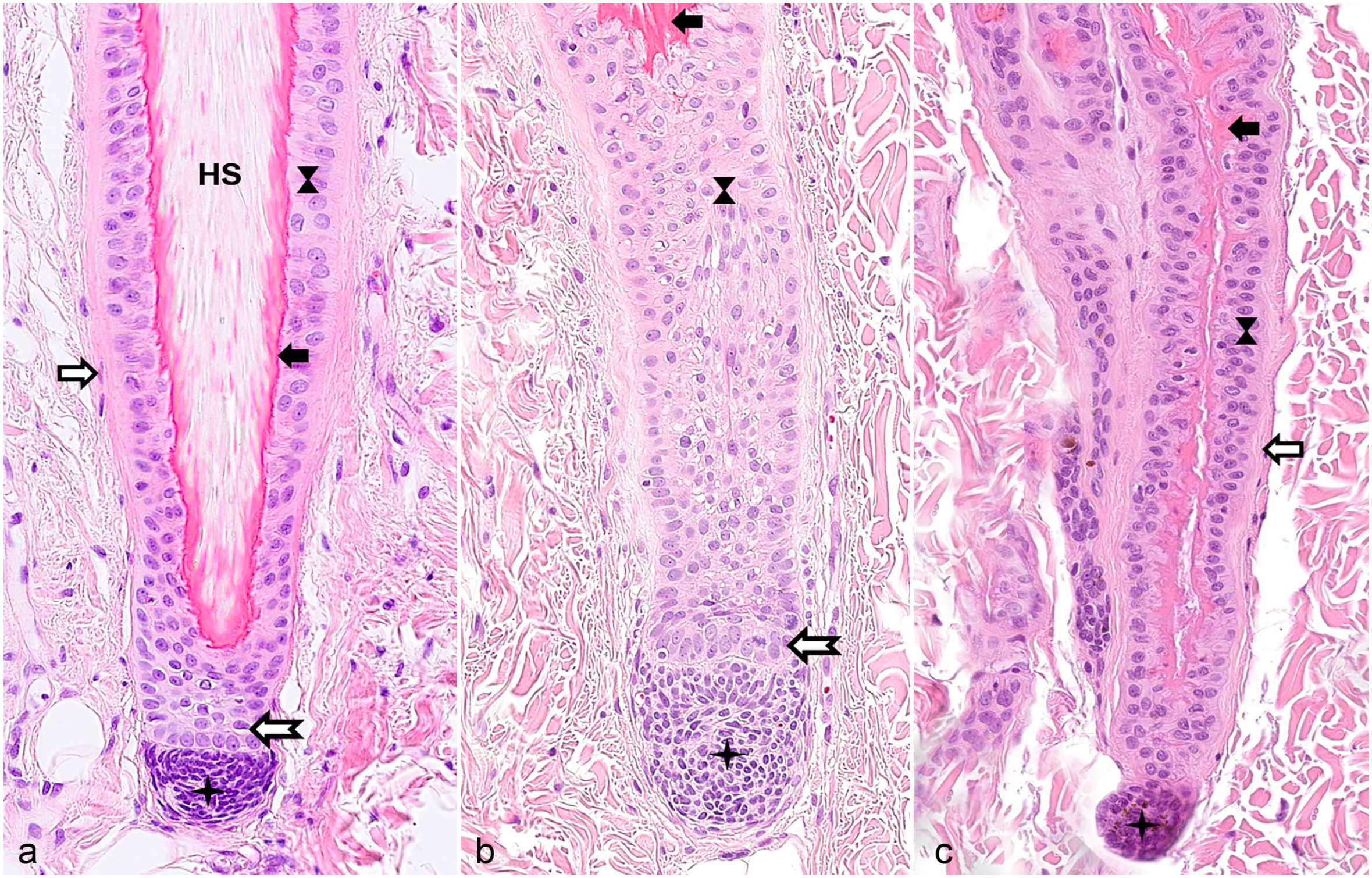
Different segments of a late anagen hair follicle (HF). Symbols are used in a consistent way throughout the figure plate. (a) Infundibulum filled with cornified material derived from the infundibular wall, as well as proteins from the sebaceous glands, the sweat glands, and lower portions of the HF and lined by the infundibular wall (thin black arrow), characterized by the presence of keratohyalin granules. The thick black arrow indicates the ring of trichilemmal keratin that marks the border between infundibulum and isthmus. The arrowhead marks the cornified inner root sheath in the isthmus and fragmented parts of the inner root sheath in the infundibulum. (b) Transition area between infundibulum and isthmus. The hourglass marks the outer root sheath and the white arrow surrounded by black indicates the fibrous sheath. (c) Isthmus. The inner root sheath is fully cornified and is surrounded by the outer root sheath and the fibrous sheath. The companion layer located between inner and outer root sheath is not visible. The hair shaft is indicated by HS. (d) Transition zone between isthmus and the suprabulbar region of the inferior segment. Note that the inner root sheath indicated by the arrowhead is fully cornified in the isthmus whereas only the Henle layer is cornified (visible as thin pink line) in the inferior segment. The Huxley layer of the inner root sheath is not yet cornified and can be identified by the presence of bright red trichohyalin granules. These are also present in the trichocytes (black triangle) forming the HS medulla. The inferior segment ends and the isthmus starts at the level where the inner root sheath and the HS is fully cornified (Adamson fringe). (e) Suprabulbar region. At the proximal end of this segment, neither the Huxley nor the Henle layer of the inner root sheath is cornified, whereas the cornified Henle layer is visible as a thin pink line in the more distal part. The outer root sheath ends at the border to the HF bulb. (f) HF bulb and lower segment of the suprabulbar region. The black circle indicates the matrix cells and the star indicates the dermal papilla. Hematoxylin and eosin.
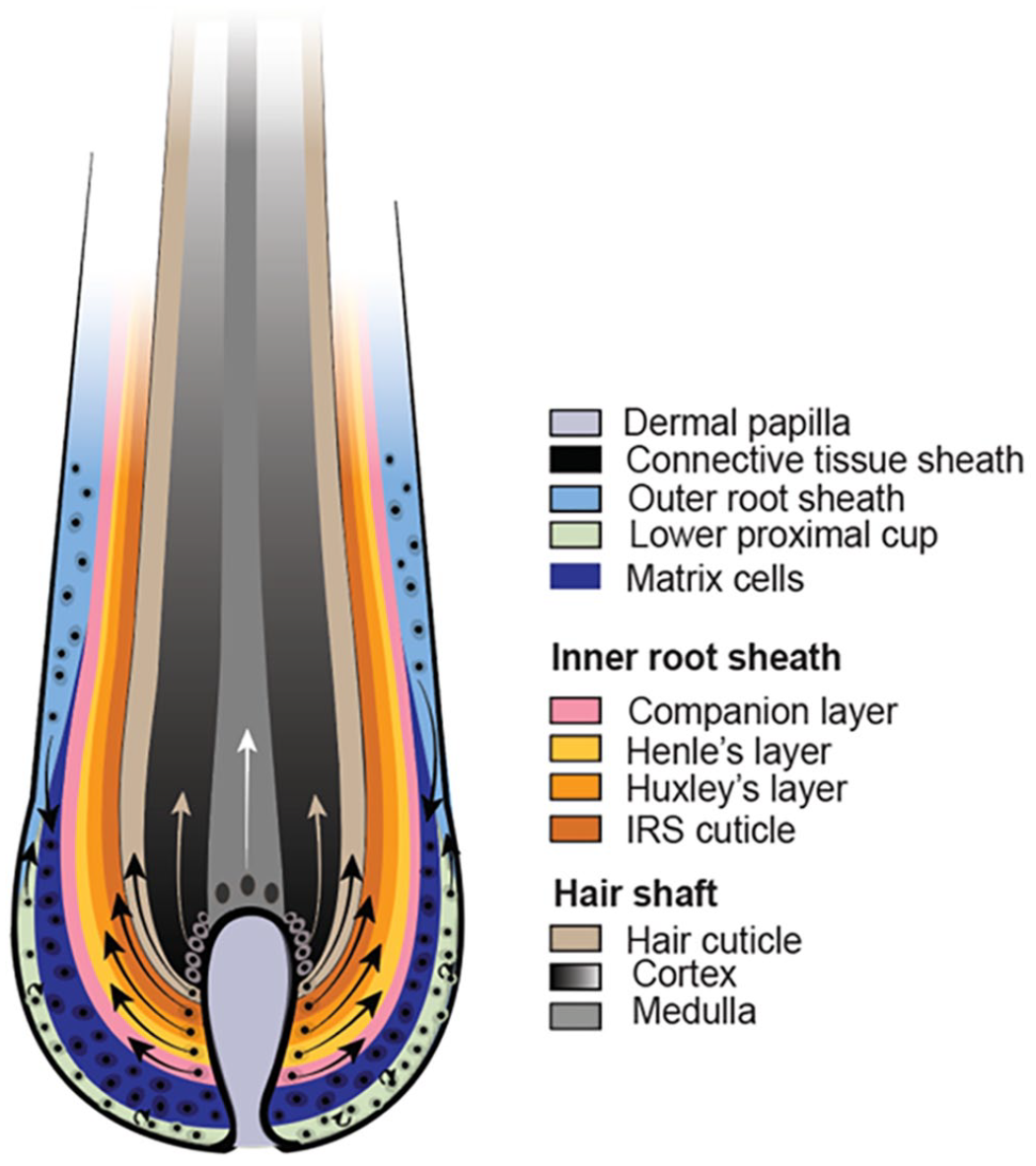
The infundibulum extends from the ostium (opening to the skin surface) of the HF to a small area at which a ring of trichilemmal cornification is seen (approximately at the opening of the sebaceous gland duct into the HF). The isthmus extends from the proximal end of the infundibulum to the interface between the completely cornified inner root sheath (IRS) and the first noncornified cell of the Huxley layer (approximately at the insertion of the arrector pili muscle). The suprabulbar region can be most easily recognized by the presence of a not yet fully cornified IRS characterized by trichohyalin granules in the cytoplasm of the cells of the Huxley and, in the more proximal part, also the Henle layer. The bulbar region is composed of highly proliferative matrix cells, also called transit amplifying cells (TACs) or matrix progenitors, intermingled with melanocytes. The matrix cells enclose the dermal papilla (DP). The DP is separated from the matrix cells of the bulb by a basement membrane. This basement membrane and a mesoderm-derived connective tissue layer (fibrous sheath), composed of collagen and stromal cells, surround the entire HF (Figs. 2, 3). 1
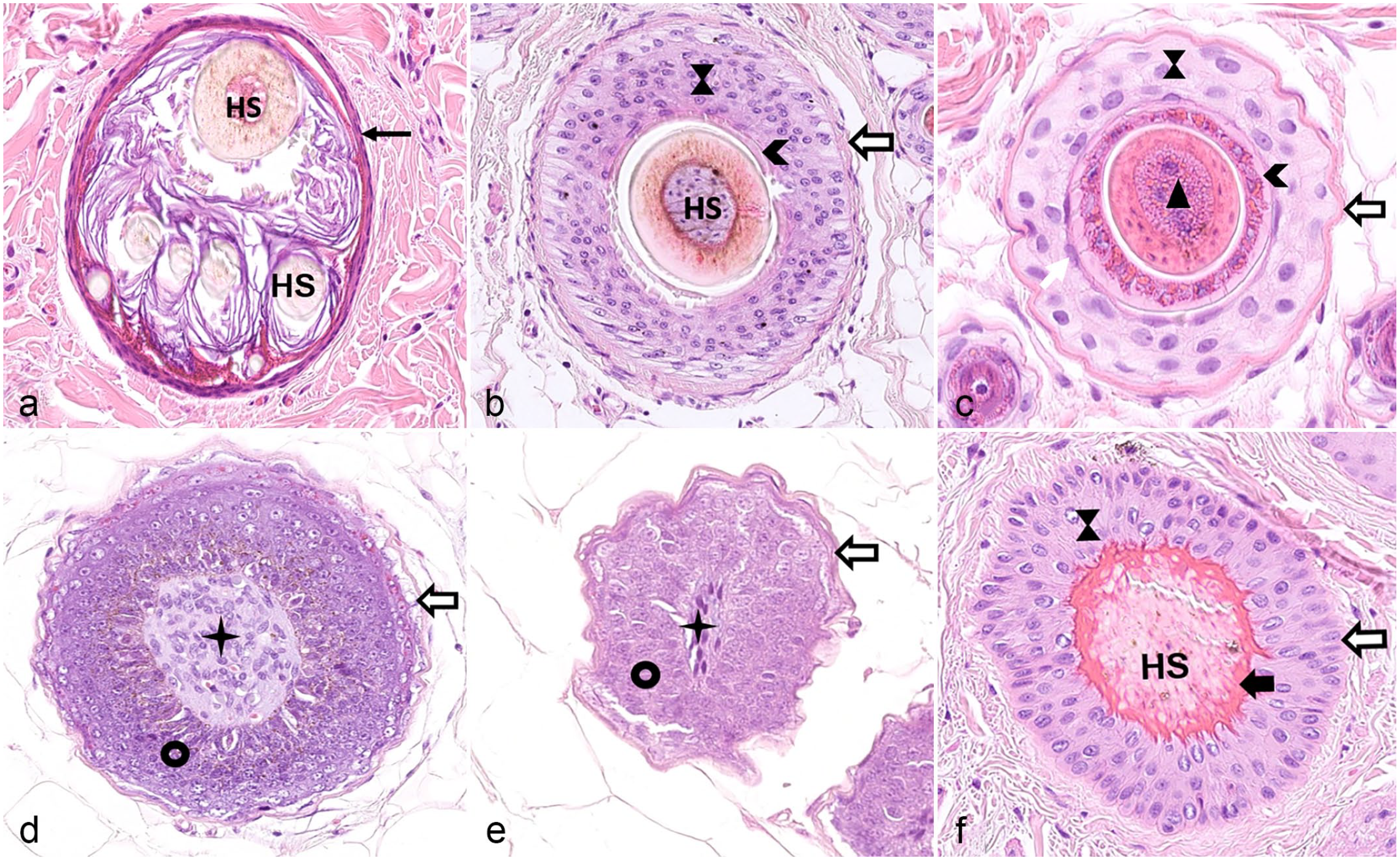
Horizontal sections through the different segments of the hair follicle (HF). (a) Cross section through an infundibulum. The infundibular wall is indicated by the black arrow. The lumen is filled with several hair shafts (HS) and infundibular keratin. The hair cycle stage cannot be determined looking at the infundibulum. (b) Cross section through the isthmus of an anagen HF. The HS is composed of the innermost medulla, the cortex, and the thin cuticle. It is surrounded by a fully cornified inner root sheath (arrowhead), the companion layer (not visible), the outer root sheath (hourglass), and the fibrous sheath (white arrow surrounded by black). (c) Cross section through the suprabulbar region of an anagen follicle. Note that the Henle layer of the inner root sheath is fully cornified whereas the Huxley layer is not yet cornified and is, like the not yet cornified trichocytes of the HS medulla, characterized by the presence of trichohyalin granules. The white arrow indicates the cells of the companion layer. (d) Cross section through the upper part of the hair bulb. The dermal papilla (star) is surrounded by matrix cells (black circle) which are in turn surrounded by the fibrous sheath (white arrow surrounded by black). (e) Cross section through the lower portion of the hair bulb. (f) Cross section through the isthmus of a telogen HF. The club hair is anchored by trichilemmal keratin to the outer root sheath, which is in turn surrounded by a fibrous sheath. Hematoxylin and eosin. HS, hair shaft.
In a cross-sectional view, the infundibulum is lined by stratified squamous epithelium with a stratum granulosum characterized by the presence of keratohyalin granules. The keratinocytes of the infundibular wall terminally differentiate by undergoing cornification. The cornified keratinocytes are seen as squames in the infundibular lumen. Eventually also a HS is seen in the infundibulum. It looks alike in all HC stages.
The isthmus and the suprabulbar region of the anagen HF are organized in concentric layers. Note that in mice, the isthmus is defined as the area between the opening of the sebaceous gland duct and the bulge, which is the region where the arrector pili muscle inserts. The bulge is the SC bearing region in mice.
The concentric layers of the isthmus and the suprabulbar region of a late anagen HF are composed of an outer root sheath (ORS), a companion layer (CL), and an IRS, which surrounds the HS. 96 The ORS can be subdivided from the outside (abaxial) to the inside (axial) in a basal and a suprabasal layer. The CL, consists of barely visible cells and separates the ORS from the IRS. It is yet unclear if it represents an independent layer or is part of the IRS.61,96 The IRS differs morphologically between the suprabulbar region and the isthmus. In the lower part of the suprabulbar region, it is composed of, from the outside to the inside, the Henle layer, the Huxley layer, and the IRS cuticle. Trichohyalin granules are present in the cytoplasm of the cells from the Henle and Huxley layers. The IRS cells differentiate gradually upward, and the IRS is fully cornified when it reaches the isthmus. The cells of the Henle layer are cornified already after approximately the end of the lower third of the suprabulbar region and the Henle layer is from then on visible in hematoxylin and eosin (H&E)–stained sections as a pale eosinophilic line. The cells of the Huxley layer are fully cornified only when they reach the border of the isthmus. In the isthmus, the Henle and Huxley layers cannot be distinguished anymore because both are fully cornified and appear homogeneously pale eosinophilic. The IRS cuticle at the interface to the HS is barely visible in histological sections. The HS is constituted of 3 layers: an outer cuticle, the cortex, and the innermost medulla, which is not present in all HSs.51,94,101,107,111 The trichocytes of the HS in the suprabulbar region are still nucleated, and trichohyalin granules are present in the cytoplasm of the medullary cells.
The bulb is composed of highly proliferative matrix cells and at the lowest end, close to the DP, the cells of the lower proximal cup. The bulbar cells are spatially organized, but this organization cannot be appreciated histologically. Only the pigmented cells that form the HS can be distinguished from the unpigmented cells on H&E-stained sections. The unpigmented cells form the IRS, the CL, lower parts of the ORS, or retain stemness (cells of the lower proximal cup). Furthermore, the DP, which is composed of fibroblasts, capillaries, and extracellular matrix, is seen in the bulb (Figs. 2, 3).
During catagen, most cells of the inferior portion of the HF undergo regression and the club hair forms. The club hair is characterized by a brush-like hair base and becomes gradually anchored to the ORS that undergoes trichilemmal cornification. The trichilemmal cornification of the ORS starts at the proximal end of the remaining part of the follicle, and the IRS simultaneously degrades at the distal end of the isthmus. The DP diminishes in size, changes its shape, and remains connected to the regressing epithelial cells. The fibrous sheath, which was surrounding the inferior portion of the HF remains as connective tissue trailing strand during catagen.
In telogen, the entire inferior portion of the HF has regressed, and the IRS is not present anymore. The club hair is firmly anchored along the entire isthmus to the surrounding ORS by trichilemmal keratin. The DP is situated underneath the secondary hair germ (SHG), which forms the base of the telogen HF and is one of the SC compartments. The DP is not surrounded by epithelial cells anymore (Figs. 4, 5).
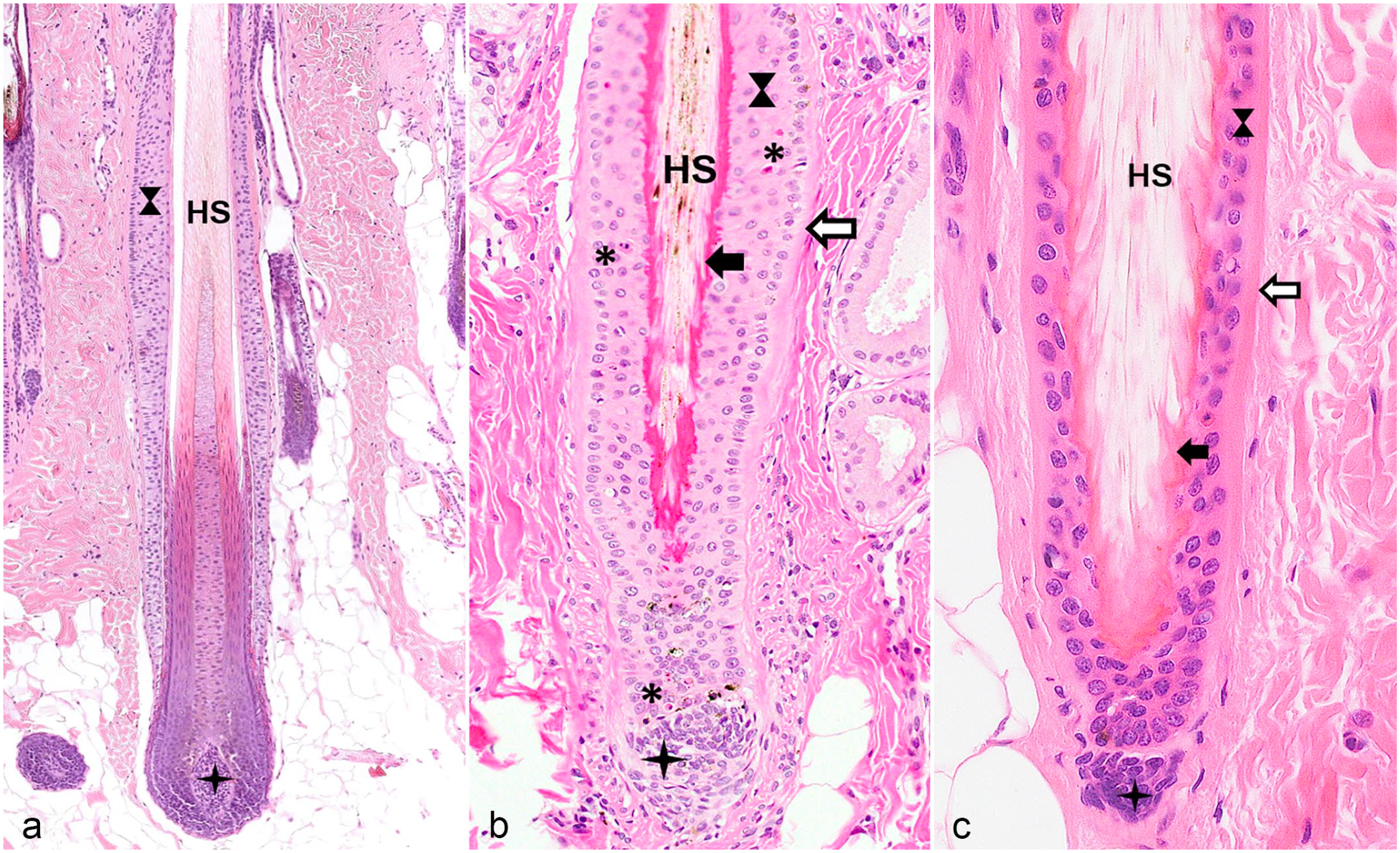
Hair follicles (HFs) in different hair cycle stages. The same symbols are used for the different anatomic regions as in Figs. 2 and 3. (a) Late anagen HF with a fully developed inferior segment and the isthmus. (b) Catagen HF. The inferior segment regresses by cessation of the mitotic activity and by apoptosis (indicated by asterisk) and the club hair surrounded by trichilemmal keratin forms starting at the proximal end of the isthmus. The dermal papilla (star) changes its shape and remains attached to the regressing epithelial strand. The trailing connective tissue sheath, which is located below the dermal papilla, is not visible in this image. (c) Telogen HF characterized by a fully developed club hair and a dermal papilla (star) that is located close to the outer root sheath surrounding the club hair. The inner root sheath of the isthmus and the inferior segment is not present in the telogen HF. The small cell layer between the outer root sheath and the dermal papilla represents the secondary hair germ. Hematoxylin and eosin. HS, hair shaft.
Hair follicles (HFs) in different hair cycle stages. The same symbols are used for the different anatomic regions as in Figs. 2 and 3. (a) Early anagen stage. The cells of the secondary hair germ (white arrow with an indented end and surrounded in black) start to proliferate downward to form the inferior segment. (b) Early anagen at a later stage. Note the elongated epithelial strand of the outer root sheath and the mitotic figure indicated by an arrow. (c) Kenogen HF. Trichilemmal keratin is anchored to the outer root sheath, but a HS is missing in the follicular lumen. Hematoxylin and eosin. HS, hair shaft.
A detailed morphological description of the distinct HC phases for mice, humans, and dogs can be found in the literature.66,67,71
The HS
As mentioned above, the HS is composed of 3 layers: the cuticle, the cortex, and the medulla. The overlapping, thin scales of the cuticle are oriented toward the tip of the hair and form a protective layer around the cortex. The orientation of the cuticle cells of the HS is opposite to the cuticle cells of the IRS, facilitating the retention of the HS within the HF. The cortex constitutes the bulk of the fiber mass, is composed of trichocytes, and is accountable for the tensile strength of the HS. 83 The innermost medulla consists of loosely packed, horizontally oriented cells interspersed with vesicles and trichohyalin granules up to the level of the Adamson fringe, which marks the border between the not yet fully cornified IRS and HS, and the fully cornified IRS and HS.31,51,91 The major protein component of the HS are different keratins.16,49 Acidic type I and basic type II keratins twine around each other to form dimers in the shape of a helix. The helix is maintained by disulfide bonds. Several of these helices pair up as tetramers to form protofilaments, which in turn form intermediate filaments (IFs, microfibrils). These IFs are the basic structure of the trichocyte.81,111 The IFs are embedded in a matrix of keratin-associated proteins (KAPs) and form macrofibrils. The assembly of numerous macrofibrils embedded in KAPs form the spindle-shaped trichocytes of the cortex. Keratins and KAPs account for 85% of the HS proteins. The strength of a hair fiber is provided by a network of intermolecular and intramolecular cross-links. These cross-links are composed of the disulfide bonds that connect the keratins of the IFs, the KAPs, and the adhesion molecules that form intracellular bridges between the trichocytes. The latter are desmogleins and desmocollins that mediate the mechanical stability of the keratin-KAP network.16,30,91
Arrangement of HFs
HFs can be distinguished by their arrangement as simple and compound HFs (Fig. 6). Most omnivores and herbivores (eg, horses, cattle, pigs, rats, mice) have simple HFs. Simple HFs are distributed individually throughout the dermis and each HS emerges through one orifice (Fig. 6b). Simple HFs can be either primary or secondary, and the percentage of secondary follicles is approximately 60% to 70% in cattle and horses. In Cashmere goats, secondary HFs produce the wool and account for the vast majority of HFs. 114 Compound HFs are seen in carnivores (eg, dogs, cats). In compound follicles, several HFs are grouped together, and the HSs produced by these HFs share one infundibulum and emerge through one orifice (Fig. 6a, 7). In most animals with compound follicles, each compound group is composed of a central large primary HF surrounded by variable numbers of smaller lateral primary HFs and secondary HFs (Fig. 6a, 7). In dogs, depending on the breed, a follicular compound is composed of 10 to 30 HFs, 62 and up to 85% of the HFs are secondary and produce the undercoat (Fig. 7). Several groups (in dogs and cats mostly 3) are arranged closely together, forming a follicular unit (complex) (Fig. 6c). However, there are some dogs for which the central large primary hair has its own orifice, at least in some body locations. This is seen also in cats (Fig. 6c). In this case, the follicular units are called mixed. Humans, sheep, and goats have, depending on the body location and the breed (in goat and sheep), 62 simple or compound follicles. On the human scalp, it is common that 2 to 3 HSs share one infundibulum and protrude from the same orifice, whereas simple HFs predominate on the body. Sheep have, depending on the body location, simple follicles or compound follicles composed of 3 primary HFs and 15 to 16 (up to 60 in merino sheep) secondary HFs. 62 In goats, compounds are composed of 3 primary and 3 to 6 (8–22 in Cashmere goats) secondary HFs. A detailed insight into comparative HF morphology between different species is, for example, provided in prior publications.58,59,62
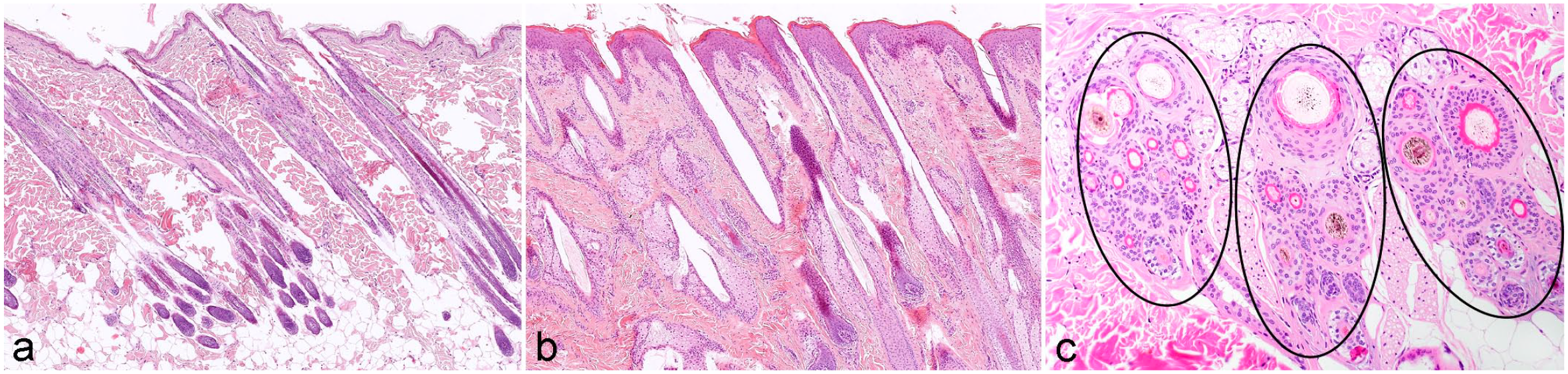
Hair follicles (HFs) of different species. (a) Feline skin with compound HFs. The HFs are grouped together and the hair shafts (HSs) produced by these HFs share one infundibulum and emerge through one orifice to the outside. (b) Equine skin with simple HFs. Follicles are distributed individually throughout the dermis and each HS emerges through one orifice. (c) Canine follicular complex (unit). Compound HFs form a group (encircled) composed of a central large primary HF surrounded by variable numbers of smaller lateral primary and secondary HFs. Primary HFs are pigmented, whereas secondary HFs are unpigmented. The number of HFs per group varies depending on the dog breed. Within 1 HF group, HFs of different cycle stages are present. The telogen follicles can be identified by the bright red trichilemmal keratin that surrounds the HS and the anagen follicles present with a pale eosinophilic inner root sheath. Several groups (in dogs and cats mostly 3) are arranged closely together forming a follicular complex. Hematoxylin and eosin.
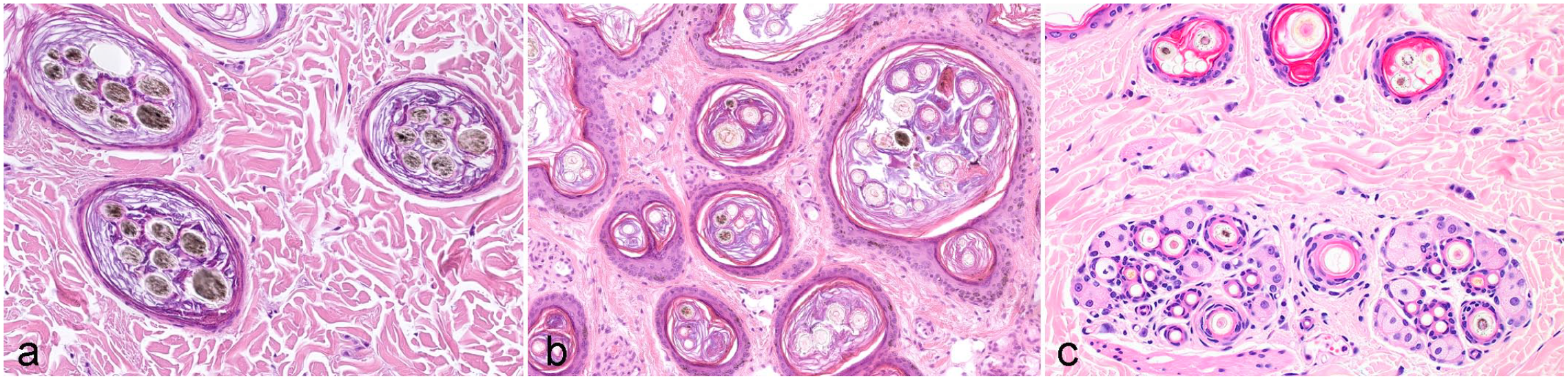
Horizontal sections through the infundibula of different canine breeds. (a) Poodle. Each infundibulum contains about the same number of pigmented (primary) hair shafts (HSs). (b) Bernese Mountain Dog. The number of HSs per infundibulum varies greatly, and each infundibulum contains primary and secondary HSs. (c) Mixed follicular unit of a cat. The large central primary hair is derived from a simple hair follicle (HF) that has its own infundibulum. This simple HF is surrounded by 2 groups composed of compound HFs. Hematoxylin and eosin.
HF Morphogenesis
HF formation in the embryo requires communication between ectodermal, mesodermal, and neuroectodermal components and is regulated by a well-orchestrated interaction of several molecules affecting the wingless (WNT), sonic hedgehog (SHH), bone morphogenic protein (BMP), Notch, and the ectodysplasin receptor (EDAR) signaling pathways.17,54,94 HF morphogenesis is divided into 3 main developmental steps: induction, organogenesis, and cytodifferentiation. Each of these can be further subdivided into morphologically distinct stages (Fig. 8). The embryo surface emerges as a single layer of epithelial cells, which gives rise to the epidermis. The dermis is formed from the underlying mesoderm composed of mesenchymal cells (stage 0). During a specific embryonic stage (E14.5 in mouse embryos and week 12 in human and Cashmere goat embryos for primary HFs), HF morphogenesis starts at predetermined sites.17,19,24 During this induction phase (stage 1), activation of canonical WNT/β-catenin signaling leads to multifocal thickening of the surface epithelium, and HF placodes are formed. Interestingly, it has been shown recently, combining long-term three-dimensional (3D) live imaging and single-cell transcriptomics, that the position of the cells within the placode already determines the terminal fate of the daughter cells within the fully developed HF. 65 Organogenesis (stages 2 and 3) starts when SHH signaling from the placode to the underlying mesenchymal cells induces condensation of mesenchymal cells underneath the placode. This leads to the down-growth of epithelial cells and the formation of the hair germ, also named bud. The hair germ proliferates and invaginates deeper into the dermis forming the hair peg (stage 4).19,94 When the down-growing epithelium has reached its final length (stage 5, bulbous peg stage), cytodifferentiation starts. During cytodifferentiation (stages 5 to 8), the edges of the down-growing follicular epithelium gradually engulf the mesenchymal condensate (peg stage), which from thereon is called the DP. Signals from the DP to the adjacent epithelial cells induce their differentiation into the IRS, which will provide a molding channel for the developing HS. When the IRS has been formed, the epithelial cells that surround the DP (also known as matrix cells) start to differentiate into the distinct lineages of the HS, which elongates and eventually protrudes through the epidermis. The development of an infundibular lumen and the orifice within the cell column that was down-growing allows the developing HS to emerge to the surface. Lumen formation is regulated separately and is not yet entirely understood. In a recent publication, it was shown that a column of keratin (KRT)79+ cells is found in the center of the hair germ of the developing HF. These cells protrude from the HF, change their shape, and integrate into the interfollicular epidermis during stages 6 to 8 of HF morphogenesis. Simultaneously, cells of the adjacent surface epithelium fold inward at the future site of the orifice. Lumen formation seems to be associated with apoptosis around the time when the cells of the column change their shape. In the mature HF, keratin (KRT)79+ cells line the inner surface of the infundibulum.60,103 These general developmental stages occur during the morphogenesis of all hair types, but it has been shown in mice and Cashmere goats that the induction of different HF types (eg, primary and secondary HFs) occurs at different embryonic stages (eg, mouse awl and auchene hairs at E16.5 and zig zag hair around P0) and that their development is tuned by the interplay of different molecules.17,94 The disturbance of the finely tuned HF morphogenesis at any level may result in congenital alopecia. 59
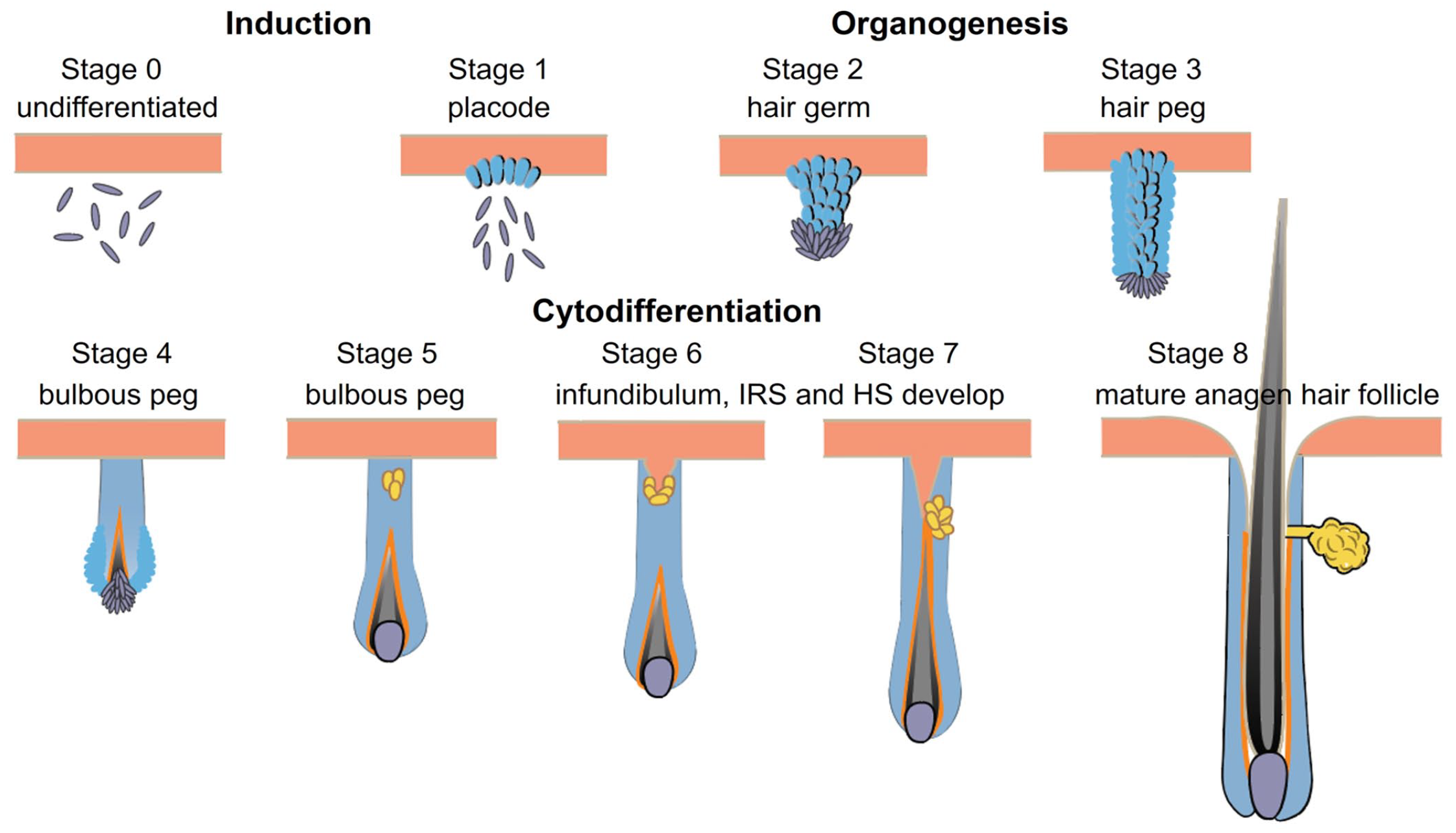
HF morphogenesis is divided into 3 main developmental steps: induction, organogenesis, and cytodifferentiation. Each is further subdivided into morphologically distinct stages. During the induction phase (stage 1), multifocal thickening of the surface epithelium results in the formation of HF placodes. Organogenesis (stages 2 and 3) starts when condensation of mesenchymal cells underneath the placode occurs. This leads to the down-growth of epithelial cells and the formation of the hair germ, also named bud. The hair germ proliferates and invaginates deeper into the dermis (stage 4, peg stage). When down-growing epithelial cells have reached their final length (bulbous peg stage, stage 5), cytodifferentiation starts. During cytodifferentiation (stages 5–8), the edges of the down-growing follicular epithelium gradually engulf the mesenchymal condensate (bulbous peg stage) which, from thereon, is called the dermal papilla. Signals from the dermal papilla to the adjacent epithelial cells induce their differentiation into the IRS, which provides a molding channel for the developing HS, which elongates and eventually protrudes through the epidermis. The development of an infundibular lumen and the orifice occurs during stages 6 to 8 of HF morphogenesis within the cell column that was down-growing. HF, hair follicle; IRS, inner root sheath; HS, hair shaft.
It is noteworthy that dogs (and most likely also the other animals that have compound follicles) are born with simple HFs. Once morphogenesis of these follicles is complete, dogs gain their adult coat by the formation of follicular compounds, which are produced in puppies older than approximately 12 weeks. 95
In dogs, the coat type is an essential characteristic of the breed. It is, in most breeds, determined by the combination of distinct variants of only 3 genes, namely
The HC
Hair growth is maintained by the lifelong cyclical regeneration of the HF. This is needed to renew the protective covering of the body and to enable adaption of coat quality (eg, hair length, diameter, and density) and color to environmental changes and sexual maturation. After completion of HF morphogenesis, the HF is in anagen (Figs. 1, 4a, 9). Consequently, the first HC stage is catagen, during which the inferior portion of the HF regresses (Figs. 4b, 9). In telogen, the HF is in its “default state,” a phase of relative proliferative quiescence. In telogen, the fully keratinized HS is maintained as club hair, which is an energy-efficient state to keep the body protected by a fur (Figs. 4c, 9).26,94,105 At the end of telogen, a new anagen stage is induced (Figs. 5a, b, 9).
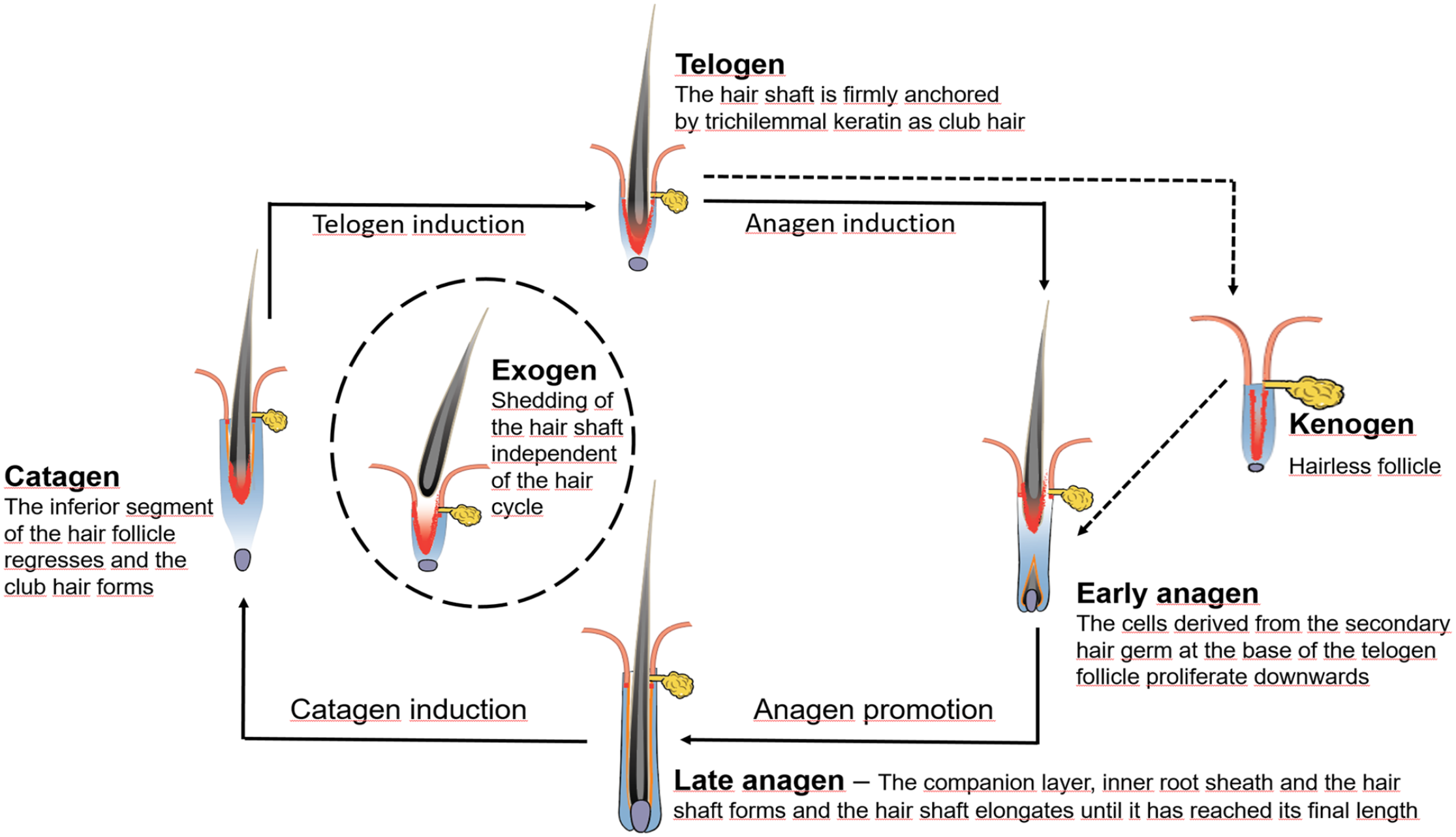
The hair cycle (HC). The telogen hair follicle (HF) is in a phase of relative proliferative quiescence and the fully keratinized club hair is anchored by trichilemmal keratin to the outer root sheath. At the end of telogen, a new anagen phase is induced. During early anagen, epithelial cells derived from the stem cells located in the secondary hair germ, and later also from the bulge, grow down toward the subcutis to form the inferior portion of the HF. During late anagen, the different layers of the hair shaft and the inner root sheath form. They are derived from transit amplifying cells of the HF bulb. During catagen, the inferior portion of the HF regresses by apoptosis and cessation of mitotic activity. The shedding of the club hair (exogen) is actively controlled and can occur any time during the next HC phase or even later. If anagen induction is insufficient, the hair fiber is lost and a kenogen follicle develops. It is characterized by an empty HF, which has previously lost the club hair fiber and remains quiescent until appropriate signals initiate a new anagen phase. Figure modified from Figure 17.6 in
The shedding of the club hair (exogen) represents another distinct and actively controlled phase of the HC and prevents the premature release of the club hair fiber before a new HS has formed, and thus ensures the protection of the body by a full hair coat (Fig. 9). In mice, it is known that the overfur hair is shed in the subsequent anagen phase, while the underfur hair is shed either in the following anagen or the next telogen phase.63,94,98 In dogs, exogen is mainly observed in the subsequent anagen phase, but detailed data on the shedding of primary and secondary HSs are lacking. 68 The mechanisms involved in club fiber shedding—by some authors also referred to as teloptosis—is not yet completely understood, although proteolysis of intercellular adhesion molecules between the club hair and the surrounding epithelium of the ORS is supposedly involved.34,87 Kenogen, in some veterinary textbooks also described as hairless telogen, is another distinct HC stage. It is characterized by an empty HF, which has previously lost the club hair fiber and remains quiescent until appropriate signals initiate a new anagen phase (Fig. 5c).86,87 Kenogen HFs are physiologically present, to a small percentage, in humans and contribute up to 20% to a canine follicular unit. However, a prolonged kenogen phase may result in follicular atrophy, and a higher percentage of kenogen follicles is characteristic for noninflammatory HC disorders resulting in alopecia (Fig. 10).68,87
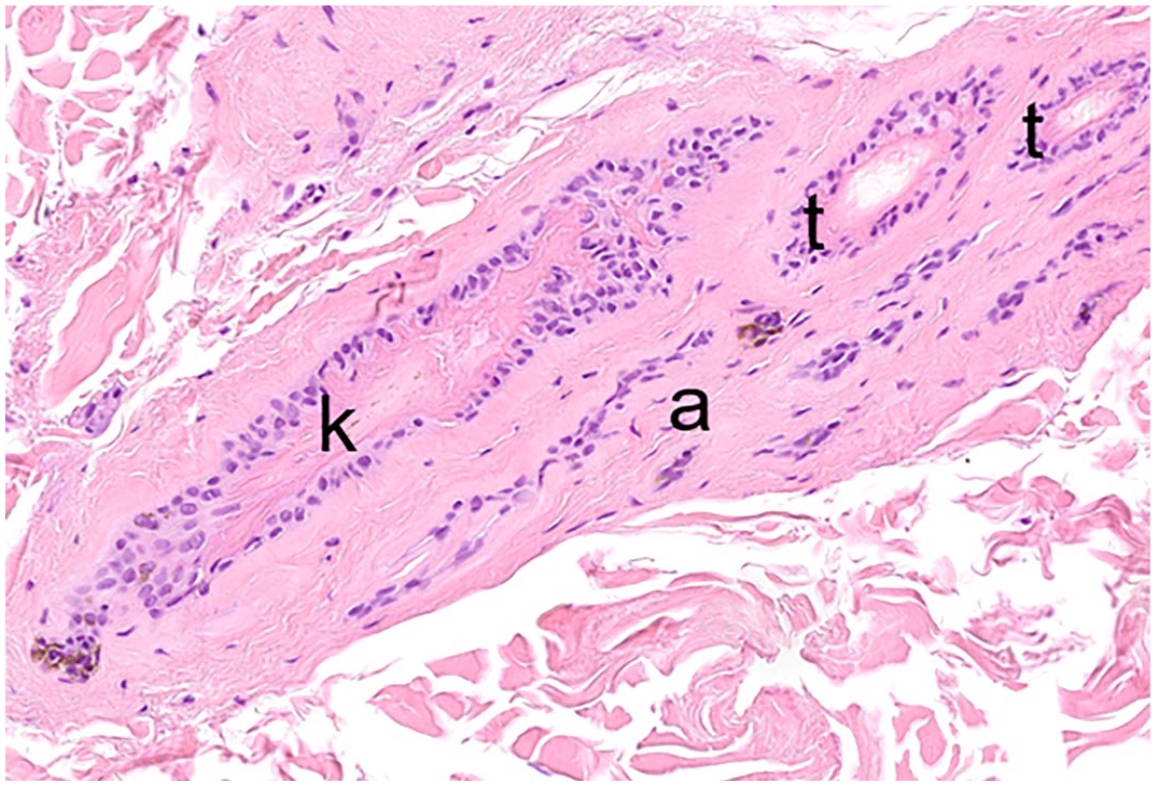
Hair cycle disturbance in a dog. The follicular group is composed of telogen (t), kenogen (k), and atrophic hair follicles (a). Hematoxylin and eosin.
The length of the HC phases determines the length of the HS and its replacement rate during shedding. 108 In contrast to mice, where the first HC is synchronized and thus easy to study,8,77,94 in humans and domestic animals, the postnatal HC is never synchronized and follows mosaic, nonsynchronous patterns. It is noteworthy that even HFs within 1 follicular unit cycle independently. 74 Although not well studied, it is obvious that the duration of the HC phases varies between species, breeds, body locations, age, and sex. In humans, scalp HFs stay in anagen for 2 to 8 years, the duration of catagen is 3 to 4 weeks, and telogen lasts for approximately 9 months. In contrast, human eyebrows are in anagen for 4 to 7 months, catagen lasts 3 to 4 weeks, and telogen lasts about 9 months. 74 In sheep, especially in Merino sheep, about 99% of follicles are continuously growing for up to 8 years. In contrast, in the ancestral Wiltshire sheep, a season-dependent HC, including hair shedding, can be observed. 90 In dog breeds, such as poodles and other dogs that need regular clipping, 98% of follicles are in anagen. Yet in most dog breeds, an average of about 30% of follicles are in the telogen phase.67,68
The HC is associated with extensive remodeling of the entire skin. This includes the expansion of blood and lymph vessels, the remodeling of nerve fibers, maturation and expansion of adipocytes, production of extracellular matrix by fibroblasts, and changes in the number and type of immune cells.26,44
Hair Follicle SCs
The cyclic regeneration of the HF throughout an animal’s lifetime relies on follicular SCs. Follicular SCs are slow cycling and can self-renew and maintain pluripotency throughout their life. Thus, they are also important for wound repair. In vitro, it has been shown that they grow clonally and can differentiate into neuronal cells, glial cells, melanocytes, and smooth muscle cells.6,70,85,93
Within the HF, different SC populations reside in distinct microenvironments, which are essential for their maintenance and function. These microenvironments are called the SC niches. An SC niche is defined by its cellular components, cytokines, signaling molecules, and extracellular matrix proteins.
The current view, based on studies in mice, is that there are 2 main regions harboring follicular SCs in the telogen HF. Each region is compartmentalized into several niches. One SC region is the bulge, the other SC region is the SHG.28,112 In young mice, the bulge is apparent as a protrusion of the ORS.94,99 This protrusion is not seen in domestic animals nor in humans, but the term bulge has been used in several publications and refers to the isthmus region.46,47 In dogs, different SC populations have been identified along the entire isthmus. 25 Like in the dog, the SC bearing regions in the human HF are not yet well defined.72,73
The SCs of the bulge maintain the quiescent SCs of the long-term SC pool.28,29 The SHG is located at the very proximal end of the telogen HF, directly above the DP, which provides key signals for SC activation.26,28,39 The SCs located in the SHG are more prone to proliferation and respond quickly to various stimuli to initiate new anagen phases. It has been shown that the transcriptional profile of bulge SCs is homogeneous, whereas the SCs of the SHG are heterogeneous, and their spatial arrangement to the DP determines their lineage fate in the subsequent anagen phase.45,110,112
The different SC populations and subpopulations are characterized by the expression of distinct surface proteins and transcription factors, and these markers may vary among species. In mice, SC markers have been extensively studied, and CD34 and KRT15 have been identified as bulge SC markers. These SCs also express the transcription factors SRY-related high mobility group-box (SOX) 9, transcription factor (TCF)3, TCF4, LIM/homeobox protein (LHX)2, the leucine rich repeat containing G protein-coupled receptor (LGR)5, and nuclear factor of activated T-cells cytoplasmic (NFAT)1. In addition, bulge SCs have been labeled with the zinc finger protein GLI1, S100A4, S100A6, KRT19, and homeodomain-only protein (HOPX). Cells of the SHG express the SC markers LGR5, SOX9, KRT15, and GLI1.1,27,28,84,89,93,100,104 Noteworthy, SCs in mice are not only located in the bulge and in the SHG, but also in the isthmus region above the bulge. These cells do not express the SC markers CD34 and KRT15, but express MTS24, LGR6, and leucine rich repeats and immunoglobulin-like domains 1 (LRIG1).40,69,97 In mice downstream analysis, employing lineage-tracing experiments and single-cell RNA-seq of the distinct SC and progenitor populations has allowed for the determination of the final differentiated cell type arising from the various progenitors. 112
SC marker expression has also been studied in humans, dogs, pigs, and Cashmere goats. In humans, the marker expression is only partially similar to the expression described for mice. For example, CD34 is not expressed in human SCs. In contrast, human bulge cells are identified by the cell surface glycoprotein CD200 and the expression of KRT15 and KRT19.15,84 In dogs, CD34+ cells are located along the entire isthmus and the suprabulbar region of the anagen follicle, whereas in telogen, CD34+ cells are present only in the lower ORS. KRT15 is expressed in the outermost layer of the ORS along the isthmus during anagen and in the ORS of the upper isthmus region during telogen. SOX9 expression was solely found in the innermost cells of the ORS in anagen follicles. LGR5+ cells are present in the SHG of telogen HFs in dogs. Furthermore, PCR analysis revealed upregulation of SOX9, LHX2, TCF3, and NFATC1 in canine bulge cell-enriched follicular keratinocytes.25,47 Transcriptome analysis of microdissected HFs revealed the expression of both CD34 and CD200 as well as KRT15 and 19, SOX9, LGR5, LHX2, TCF3 and 4, NFATC1, and the integrins ITGA6 and B1. 109 In Cashmere goats, transcriptome analysis of secondary HFs revealed the expression of the same SC markers as in dogs. 32 Recently in pigs, LGR5 has been identified as a marker of the ORS sheath and bulge cells in all stages of the HC, and low expression levels have been identified in the IRS. 82
The SC dynamics in the HF have been only studied in mouse models using in vivo lineage tracing and single-cell labeling applying state-of-the-art imaging technologies and single-cell genomic approaches. These experiments cannot be performed in companion animals and in humans. Thus, it can be only assumed, but is not proven, that cells expressing the same markers as SCs in mice are also SCs in domestic animals, and that SC dynamics are similar in humans, dogs, pigs, and goats.67,74,107
SC quiescence, activation, proliferation, and their final fate is regulated by finely tuned and balanced signaling events driven by factors derived from the HF niche and their interaction with factors derived from the DP and several other cell populations, such as melanocytes, sebocytes, adipocytes, smooth muscle progenitors, and nerve fibers.9,20,36,93,106 These factors constantly compete with each other, providing multiple inputs to the SCs, and finally result in SC quiescence or activation. Among other factors, the balance between signals derived from the WNT and BMP pathways are crucial to determine the SC fate.12,13 Further pathways involved include SHH, NOTCH, TGFB, FGF, and PI3K/AKT signaling.52,93,105
Anagen Induction
During the late telogen phase (competent telogen), BMP signaling is gradually reduced and the SCs of the SHG become increasingly responsive to WNT signaling.112,78 When WNT signals results in the stabilization of β-catenin in the nucleus and BMP inhibitors reach certain levels, SHG SCs become activated and get ready to generate downward growing TACs.28,56,112
Early Anagen (Anagen Promotion)
During early anagen, the SCs activated during anagen induction and located in the proximal layer of the SHG undergo oriented divisions. This results in the production and downward growth of progenies of the IRS, the CL, and the HS (Fig. 5a, b).7,92,93 The follicular SCs located in the bulge become activated shortly thereafter and undergo self-renewal to maintain the SC pool. At the end of anagen promotion, the TACs derived from the SHG envelop the basement membrane that surrounds the DP and form the matrix cells of the anagen bulb. Simultaneously, cells derived from the upper layer (distal layer) of the SHG move upward and establish the basal layer of the upper ORS. These cells also contribute, together with SCs of the lower bulge, to the formation of the lower proximal cup, a single-cell layer that surrounds the matrix cells of the bulb. Some of the cells of the lower proximal cup retain stemness, whereas others give rise to the lower ORS (Fig. 11).37,93,96,110 As a result of the downward extension of the ORS, the DP loses its close contact to the bulge, and it has been discussed that this results in the return to quiescence of the bulge SCs. 93
Cell dynamics at the end of early anagen and during cytodifferentiation. The transit amplifying cells derived from the secondary hair germ envelop the dermal papilla and form the matrix cells of the anagen bulb. Simultaneously, cells derived from the upper layer of the secondary hair germ, together with cells of the lower bulge, form the lower proximal cup, a single-cell layer that surrounds the matrix cells of the bulb. Some of the cells of the lower proximal cup retain stemness, whereas others give rise to the lower outer root sheath. The matrix cells of the hair bulb give rise to transit amplifying cells, which localize around the dermal papilla. Depending on their location around the dermal papilla and driven by signals derived from the dermal papilla microniches, the cells differentiate upward to form the distinct layers of the hair shaft, the inner root sheath, and the companion layer. Figure modified from Table 17.1 in
Late Anagen (Cytodifferentiation)
During late anagen, the signaling crosstalk between the DP and the TACs that surround the DP is driven by 4 molecularly distinct subpopulations of the DP microniche. Depending on the localization of the TACs relative to these microniches and the specific signals emanating from them, the TACs undergo asymmetrical divisions to renew their cell pool and to differentiate upward into the distinct layers of the IRS, the CL, and the HS (Figs. 2, 4, 5, 11).110,112 Interestingly, although TACs are lineage primed based on their location to the DP, they are capable of changing their positions around the DP and can give rise to multiple differentiated lineages. 112 The matrix cells that have undergone cytodifferentiation are replenished not only by SCs located around the DP but also by SCs derived from the lower bulge that have moved downward along the basal ORS. 110
It is noteworthy that few remaining SCs are able to sustain normal HF growth, when most SCs have been inhibited. This proves that SCs, although spatially primed, are still able to generate the entire set of TACs and thus have the capacity to differentiate in diverse lineages. 110 Whereas anagen promotion is mainly driven by WNT signals, cytodifferentiation of the HF lineages is mainly driven by WNT and BMP signals.11,43,48,57,112,113
HS Formation
Cytodifferentiation into the different HS lineages coincides with increased cell adhesion between the trichocytes. Approximately in the middle of the bulb, just above the DP, the expression of several keratin proteins starts, and these assemble to form the different layers of the hair fiber. 31 In cortex cells, the keratin proteins assemble into intermediate filament bundles, which are called proto-macrofibrils. Simultaneously, the expression of KAPs starts. These KAPs intersperse the bundles of proto-macrofibrils. The cells become laterally oriented and align into a network of macrofibril bundles. Synchronously, melanin granules are released from melanocytes into the trichocytes to determine the color of the HS. At the most distal level of the bulb, a multitude of keratins and KAPs start to be expressed in the cortex trichocytes.
Also in the midbulb cuticle, cells start to elongate as the fiber moves upward. After the completion of extensive cell reshaping, cortex cells undergo cornification. This process is gradual and starts distal to the bulb. Cornification, which results in hardening of the hair fiber, occurs via disulfide cross-links of keratin heterodimers and tetramers within the IFs. The intercellular space between the macrofibril bundles becomes tightened by a so-called “cell membrane complex.” 81 The process of cell membrane complex formation is still not understood, but the interaction of extracellular matrix components, desmosomes, and gap junctions seems to be involved. The consolidation of the fiber is completed by water loss, which results in the shrinkage of the cortex.
As the growing fiber elongates, the tube-like keratinized channel of the IRS molds the HS. The scales of the IRS cuticle are intertwined with the scales of the HS cuticle, forming a tight connection, but allowing the IRS cells to glide upward with the growing HS and to gradually cornify. The IRS in turn is tightly attached to the CL via the “Flügelzellen.” These cells reach from the Huxley layer through the keratinized Henle layer, and attach to the CL via desmosomes. 50 When the hardened fiber detaches from the IRS at the distal end of the isthmus, the CL reabsorbs parts of the IRS and the remaining IRS particles are discharged via the hair canal.31,91 The molecular signals involved in HS differentiation comprise several pathways including Notch (with its upstream acting transcription factors MSX2 and FOXN1), WNT, BMP, and FGF.3,11,43,48
Catagen: HF Regression and Return to Relative Quiescence
After the HS has reached its predetermined length, the inferior portion of the HF regresses and the club hair is formed. The initiation of catagen is driven by the activation of BMP/TGFB/SMAD signaling with FGF5 being one of the main players.77,94 The regression of the inferior portion is caused by keratinocyte apoptosis and cessation of the mitotic activity of the matrix cells (Fig. 4b). As a result, the inferior portion of the HF is converted in an epithelial strand, which shortens progressively. The DP diminishes in size and changes its shape, but remains attached to this epithelial strand. Thus, it comes gradually closer and closer to the bulge region. Alpha-smooth muscle actin located in fibroblasts of the fibrous sheath may contribute to the retraction of the DP. 102 The fibrous sheath, which was surrounding the inferior portion, remains as trailing connective tissue sheath proximal to the retracted DP. The HS develops into the club hair by forming a brush-like hair base adjacent to the developing SHG. At the end of catagen, the club hair is firmly attached to the surrounding ORS by trichilemmal keratin. During the regression phase, SCs of the lower ORS that survived the regression migrate back to their niche and populate the bulge and the SHG. 38 SCs that were closest to the bulge in catagen become incorporated in the long-term SC pool within the bulge to fuel the next HC. Other ORS SCs that traveled further downward become SCs of the SHG, once back in telogen.
Telogen
Catagen is followed by the resting phase of the HF, named telogen. In telogen, the club hair is firmly anchored by trichilemmal keratin to the ORS (Fig. 4c). Telogen can be divided into 2 subphases, namely, refractory telogen and competent telogen. During “refractory” telogen, quiescence of bulge and SHG SCs is maintained by high levels of BMP signaling, while WNT and SHH signaling are suppressed.35,38,78 At the end of telogen, during “competent” telogen, the signals derived from the DP result in the inhibition of BMP signaling and the activation of WNT signaling, resulting in elevated transcriptional activity of the SCs located in the SHG and subsequent anagen initiation.
Systemic and Environmental Influences on the HC
The cyclical regeneration of the HF is governed by factors derived from the follicular microenvironment, the dermal macroenvironment, but also by systemic and environmental factors. These include factors released by the immune and nervous system and hormones. Other important factors that influence hair growth are nutrition, age, genetic background, the circadian rhythm, and seasonal changes.2,13,53,99 To date, no study showing the relation of the above-mentioned factors is available for dogs, but it can be assumed that HFs are regulated, at least in some aspects, similar to those in humans and mice. 2 Thus, some studies highlighting the effect of hormones, inflammation, and stress from mice and humans have been outlined in the review “General Approach to Canine Non-inflammatory Alopecia” in the same issue of this journal. Especially, the negative effect of mast cell degranulation on the HC via substance P has been shown in several mouse studies.4,5,55,75 In humans, it has been shown that examination stress results in shift of the immune response to TH1 and transiently hampers hair growth. 76 It would be beyond the scope of this review to cite all the reference that outlined the impact of the factors mentioned above. Thus, only some are highlighted below. The importance of the dermal macroenvironment, including the presence of adipocytes and preadipocytes, has been previously outlined. 13
An insight into the role of vitamins A, B, C, D, and E as well as the minerals iron, selenium, and zinc on hair growth has been previously reviewed. 2 An example of the effect of the circadian rhythm has been provided by a study that showed that the sensitivity of the HF to double stranded DNA breaks due to radiation therapy is dependent on the time of the day. The mitotic activity of the bulbar matrix cells is lower in the evening, and thus the HF is not equally sensitive to harming insults as in the morning. 79 Age has also been associated with the HF regeneration by affecting the follicular SCs. Details about the different factors contributing to this have been reviewed recently. 41 In humans, it is well known that genetic risk factors contribute to the development of androgenic alopecia.14,33
A well-known example for the adaptation of the hair coat to environmental factors is the seasonal changes of the hair coat. Molting is a prominent feature of wild mammals, and it is important to adapt the hair coat to different climatic conditions. In most mammals, the majority of HFs are in telogen throughout the year, and anagen peaks occur only in late autumn and spring. The anagen peak in autumn assures a long firmly anchored club hair and a thicker fur during the winter and guarantees a warm coat. After the warm coat is shed in spring, another anagen peak is needed to grow the shorter and thinner summer fur. The production of new HSs during anagen is very energy consuming, whereas the maintenance of a hair coat in telogen does not need much energy. Thus, seasonal hair growth and shedding is very energy-efficient. 26 Seasonal molting occurs to a minor degree in domestic animals, but never as prominent as in wild mammals. Personal observations (M.W.) looking at skin biopsies from dogs and horses are in agreement that the anagen peaks in autumn and spring and a higher percentage of telogen follicles is seen in summer and winter.4,5,13,41,55,75,76,79,80
Conclusion
The HF is a complex and fascinating mini-organ, and the detailed regulation of HF morphogenesis and regeneration during the HC is unclear in domestic animals. Despite this limitation, the understanding of its anatomy and physiology is rewarding if a pathologist has to describe and interpret skin biopsies of alopecic animals. A precise and detailed description of the follicular changes, interpreted in the context of the anamnestic data, the clinical history, and research results from rodents, is valuable to suggest a possible underlying pathogenesis.
Footnotes
Acknowledgements
I would like to thank Keith Linder for helpful discussions about the hair follicle during the writing process of the chapter about the integument for the 7th edition of
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
