Abstract
An adult castrated male Doberman Pinscher was presented with a 6-month history of well-demarcated alopecic patches with reticulated hyperpigmentation and fine peripheral scaling on the axillae, thorax, abdomen, inguinal region, and thighs. The dog later developed hyperthermia, lethargy, apparent joint pain, peripheral lymphadenomegaly, vomiting, and diarrhea. Relevant laboratory tests results included anemia, thrombocytopenia, proteinuria, and an elevated antinuclear antibodies serum titer. Histologically, skin biopsy specimens had a lymphocyte-rich interface dermatitis and interface mural folliculitis ending in follicular destruction. Altogether, these signs were consistent with a unique alopecic variant of chronic cutaneous lupus erythematosus, eventually associated with the development of systemic lupus erythematosus. This rare form of chronic cutaneous lupus needs to be added to the expanding list of lymphocyte-mediated autoimmune alopecias in dogs.
In humans, the classification of cutaneous manifestations of lupus erythematosus (LE) is based on 2 factors. 8 First, LE-associated dermatoses are separated into those with microscopic lesions specific for LE (ie, a lymphocyte-rich interface dermatitis with basal keratinocyte damage) and those without such lesions (ie, LE-nonspecific skin diseases). 8 Second, LE-specific skin disease, also known as cutaneous lupus erythematosus (CLE) sensu stricto, is further divided into acute, subacute, and chronic CLE based on the duration of the lesions (ACLE, SCLE, and CCLE, respectively). 6,8 Within this nosology, discoid LE (DLE) represents a unique clinical variant of CCLE. In contrast, lupus-nonspecific skin lesions (eg, vasculitis or subepidermal vesicular lesions) are seen not only in LE but also as part of other diseases. 8 Importantly, systemic LE (SLE) can exist with or without skin lesions, which can be either LE specific (ie, CLE) or not. On the other hand, CLE can be seen with or without SLE. 6,8 Less than 20% of human patients with DLE are reported to develop SLE. 6 Whereas inflammatory lesions, sometimes leading to permanent alopecia, are often seen on the scalp of humans with DLE, other patients can have alopecic patches without clinically visible inflammation; such lesions resemble those of the autoimmune alopecias pseudopelade or alopecia areata. 10
The purpose of this report is to describe a unique alopecic form of CCLE in an adult dog that subsequently manifested signs of SLE.
A 2.5-year-old castrated male Doberman Pinscher was presented to the North Carolina State University Veterinary Teaching Hospital for a 6-month history of scaly skin lesions. These lesions reportedly began as erythematous, exudative, and scaly patches on the axillae and groin, and, with time and following several courses of oral antibiotics and antiseptic shampoos, became nonerythematous as they slowly spread centrifugally. Anorexia was subsequently reported by the owner. At presentation to the dermatologist, skin lesions consisted of well-demarcated bilaterally symmetrical patches of spontaneous hair loss on the axillae, cranio-ventro-lateral thorax, lower flanks, and caudomedial aspects of the thighs and abdomen (Figs. 1 –3). In alopecic areas, only small tufts of hair remained (Fig. 3). The transition between haired and alopecic patches was abrupt with a rim of fine scaling at the lesional edge (Fig. 3). Expanding epidermal collarettes with large scales and hyperpigmented centers, a hallmark of exfoliative superficial pyodermas, were not seen. In contrast, an unusual pattern of reticulated hyperpigmentation was visible throughout all hairless areas. Erythema, edema, or scarring were not found in any of the alopecic patches. In addition to skin lesions, several peripheral lymph nodes were enlarged, and mild hyperthermia (39.2°C) was noted. Furthermore, the owner reported the rapid development of lethargy, apparent joint pain, and dyspnea in the following week.
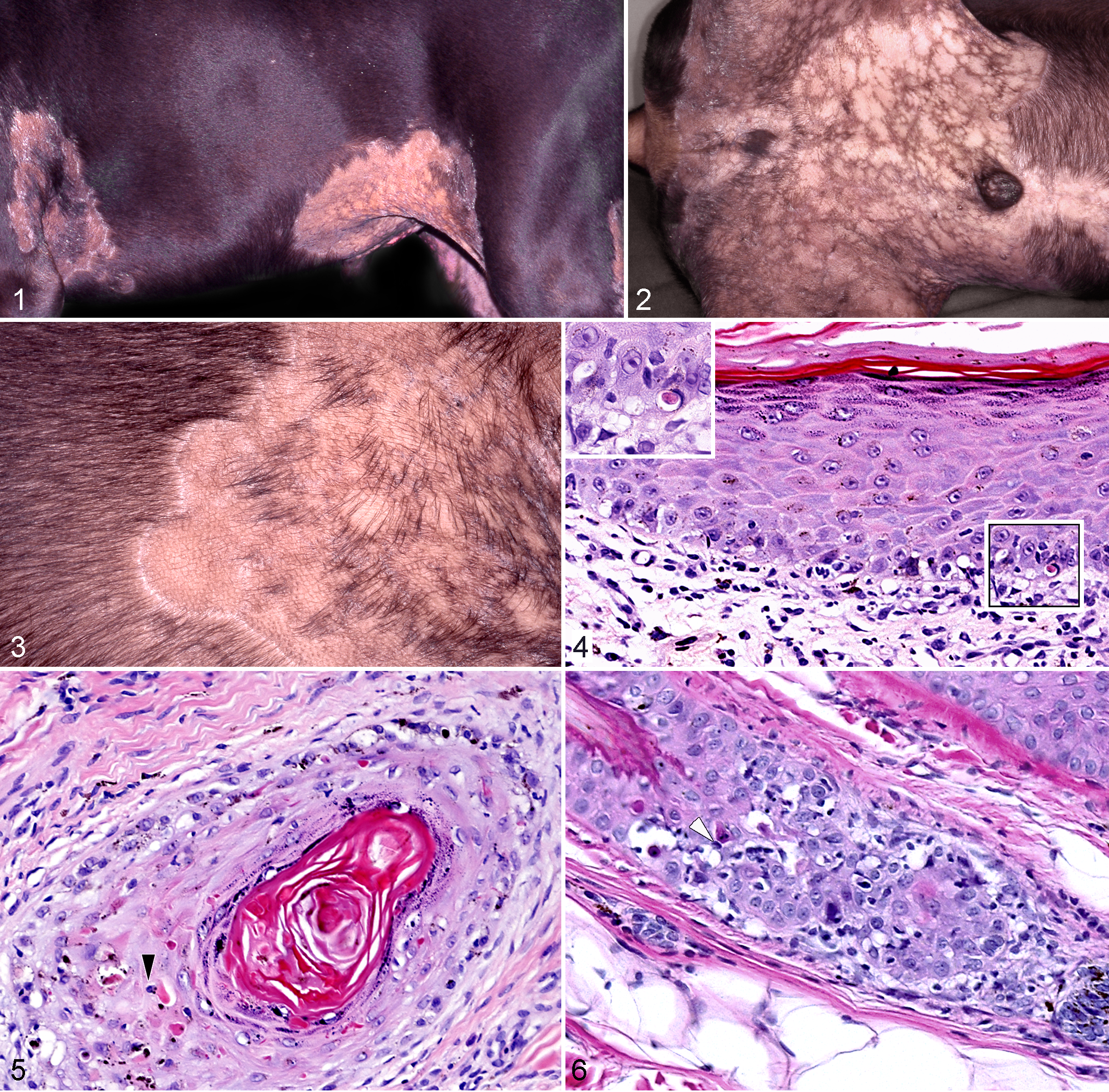
A dermatophyte culture yielded no growth after 2 weeks. Skin biopsy samples, collected from alopecic areas, had a lymphocyte-rich interface dermatitis (Fig. 4) and mural folliculitis (Figs. 5, 6). Many lymphocytes and fewer plasma cells formed a band-like infiltrate just below a moderately hyperplastic epidermis where lymphocytic exocytosis into the deep epidermis was associated with basal cell vacuolation, apoptosis, and focal disappearance of the stratum basale (Fig. 5). Pigment dispersal to superficial dermal melanophages occurred in areas of poor epidermal pigmentation, which were interspersed with smaller areas of prominent epidermal pigmentation. Mild to moderate thickening of the epidermal basement membrane zone was observed multifocally. The epidermal lymphocytic interface reaction commonly extended along follicular infundibula (Fig. 5). In addition, lymphocytes infiltrated the infundibular wall and external root sheath of 40% to 80% of hair follicles in association with multifocal keratinocyte vacuolation, apoptosis, and lymphocytic satellitosis (Fig. 6). Collapse of the external root sheath and follicular atrophy with scant remaining epithelium developed in more severely and chronically affected follicles. Hair bulbs and sebaceous glands did not seem affected; many follicles were in the telogen phase. Taken together, the microscopic changes were consistent with a unique alopecic variant of CCLE with prominent lymphocyte-mediated hair follicle destruction.
Because of the microscopic lesions suggestive of CLE in this dog, further investigations were performed to rule out concurrent SLE. Submandibular lymph node aspiration demonstrated numerous lymphoblasts. A complete blood count revealed poorly regenerative anemia, severe thrombocytopenia, neutrophilia, and atypical reactive lymphocytosis. Serum chemistry results included moderate hypoalbuminemia, mild hypoglycemia, and mild elevations of liver enzymes. Abnormal urinalysis results included marked proteinuria with amorphous crystals; the urine protein/creatinine ratio was moderately elevated. Antinuclear antibody serology was positive at 1:160 with a speckled pattern. Direct Coombs test was negative at 4°C and 37°C. Serology for Ehrlichia canis antibodies was negative. With direct immunofluorescence on formalin-fixed, paraffin-embedded sections, 5 patchy, variably thick deposits of IgG were seen at the dermo-epidermal junction (positive “lupus band”).
Altogether, the fever, lethargy, anemia, thrombocytopenia, proteinuria, LE-specific skin lesions, apparent joint pain, elevated antinuclear antibody titer, and a positive “lupus band” test led to the diagnosis of alopecic CCLE with subsequent development of SLE. 1 Oral prednisone was prescribed at low immunosuppressive dosages. Within 2 weeks, profuse diarrhea, projectile vomiting, and weight loss developed. The prednisone dose was halved, but these signs persisted. The owner requested euthanasia, but necropsy was not performed.
At the time of this writing, only 2 large case series have reported the clinical manifestations of canine SLE, but the associated skin lesions were not described in detail. 2,7 In a retrospective study of 116 dogs with SLE published prior to 1982, skin lesions were present in 38 dogs (33%). 7 The authors added 26 additional dogs with SLE and skin lesions, the latter including pruritic seborrheic skin disease, mucocutaneous ulcerations, ulcerative or hyperkeratotic footpads, or subcutaneous nodules. 7 Although not mentioned in the text, alopecia was reported in the legends of the first two figures. In a series of 75 dogs with SLE, mucocutaneous lesions were reported in 60% of dogs, 2 and were described only as “photosensitization, alopecia with or without dermatitis, erythema, ulceration and occasional hyperkeratosis.” 2
The skin lesions of our patient are unique in that they appeared early in the course of the disease, before clinical signs of SLE were noted. The duration of the dermatosis and the presence of a lymphocyte-rich interface dermatitis and mural folliculitis warrant their inclusion, using the current human classification, under the umbrella of CCLE. 6,8 Classic DLE, the most common form of human CCLE, usually manifests as well-demarcated red-purple macules or papules that develop into coin-shaped “discoid” plaques with peripheral hyperpigmentation. 6 As the lesions progress, there is central depression, scarring, depigmentation, and telangiectasia. 6 The CCLE lesions of our patient do not resemble those of classic human DLE. They are compatible, however, with the less common and poorly characterized “clinically non-inflammatory alopecic patches” of CCLE that resemble those of advanced pseudopelade or alopecia areata. 10 In this dog, as in humans, the main clinical differentials for this type of hair loss would be these two autoimmune alopecias that are both due to the cytotoxic T-lymphocytes attack of differing hair follicle segments. 3,4,9 However, the reticulated hyperpigmentation, sometimes seen in human patients with CCLE, is a clinical sign that would distinguis CCLE from the other autoimmune alopecias. In addition, in canine and human pseudopelade and alopecia areata, the interfollicular epidermis normally remains unaffected microscopically. In conclusion, the distinctive chronic alopecic skin lesions with reticulated hyperpigmentation in this dog preceded and subsequently coexisted with SLE. Histologically, the cutaneous lesions of lymphocyte-rich interface dermatitis and mural folliculitis supported the diagnosis of CCLE. Alopecic CCLE should be considered as another autoimmune alopecia with interface mural folliculitis in dogs.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
