Abstract
The skin covers the external surface of animals, and it is constantly exposed to and inhabited by different microorganisms, including bacteria. Alterations in the skin barrier allow commensal and/or pathogenic bacteria to proliferate and penetrate deep into the lower layers of the skin. Being the first barrier to the external environment, the skin is prone to injuries, allowing the penetration of microorganisms that may lead to severe deep infections. Companion animals, especially dogs, are prone to bacterial infections, often secondary to allergic dermatitis. When environmental conditions are unfavorable, horses, cattle, sheep, and goats can develop superficial infections, such as those caused by Dermatophilus congolensis. Deep inflammation is commonly caused by Mycobacterium spp., which results in granulomatous to pyogranulomatous dermatitis and panniculitis. Likewise, bacteria such as Nocardia spp. and Actinomyces spp. can cause deep pyogranulomatous inflammation. Bacteria that lead to deep necrotizing lesions (eg, necrotizing fasciitis/flesh-eating bacteria) can be severe and even result in death. This review includes an overview of the most common cutaneous bacterial infections of domestic animals, highlighting the main features and histologic morphology of the bacteria, cutaneous structures involved, and the type of inflammatory infiltrates.
The skin is the largest organ in the body and provides an external barrier to the organism. It is also constantly exposed to and inhabited by different microorganisms, including bacteria. Alterations in the skin barrier allow commensal and/or pathogenic bacteria to proliferate and penetrate deep into the lower layers of the skin.
Different superficial and deep bacterial infections will lead to different lesion patterns involving the skin of animals (Fig. 1a–d). For instance, the overgrowth of pathogenic bacteria colonizing the skin surface is more likely to result in superficial pustules. However, with skin barrier disruption, bacteria can penetrate the skin and result in deep suppurative to pyogranulomatous inflammation. Certain bacteria, such as Mycolicibacterium spp. (former Mycobacterium spp.), are attracted by lipid-rich environments 53 and will often cause panniculitis. It is crucial for pathologists to identify the distribution and pattern of lesions involving the skin to narrow down possible differential diagnoses of cutaneous infections. This review focuses on the most common cutaneous bacterial diseases of domestic animals, highlighting the characteristic clinical and histologic lesions. It is divided into five sections, discussing the normal cutaneous microbiota inhabiting the skin of domestic animals and the pathogens associated with cutaneous infections based on the affected portion of the skin, such as epidermis, hair follicles, dermis and panniculus, and blood vessels.
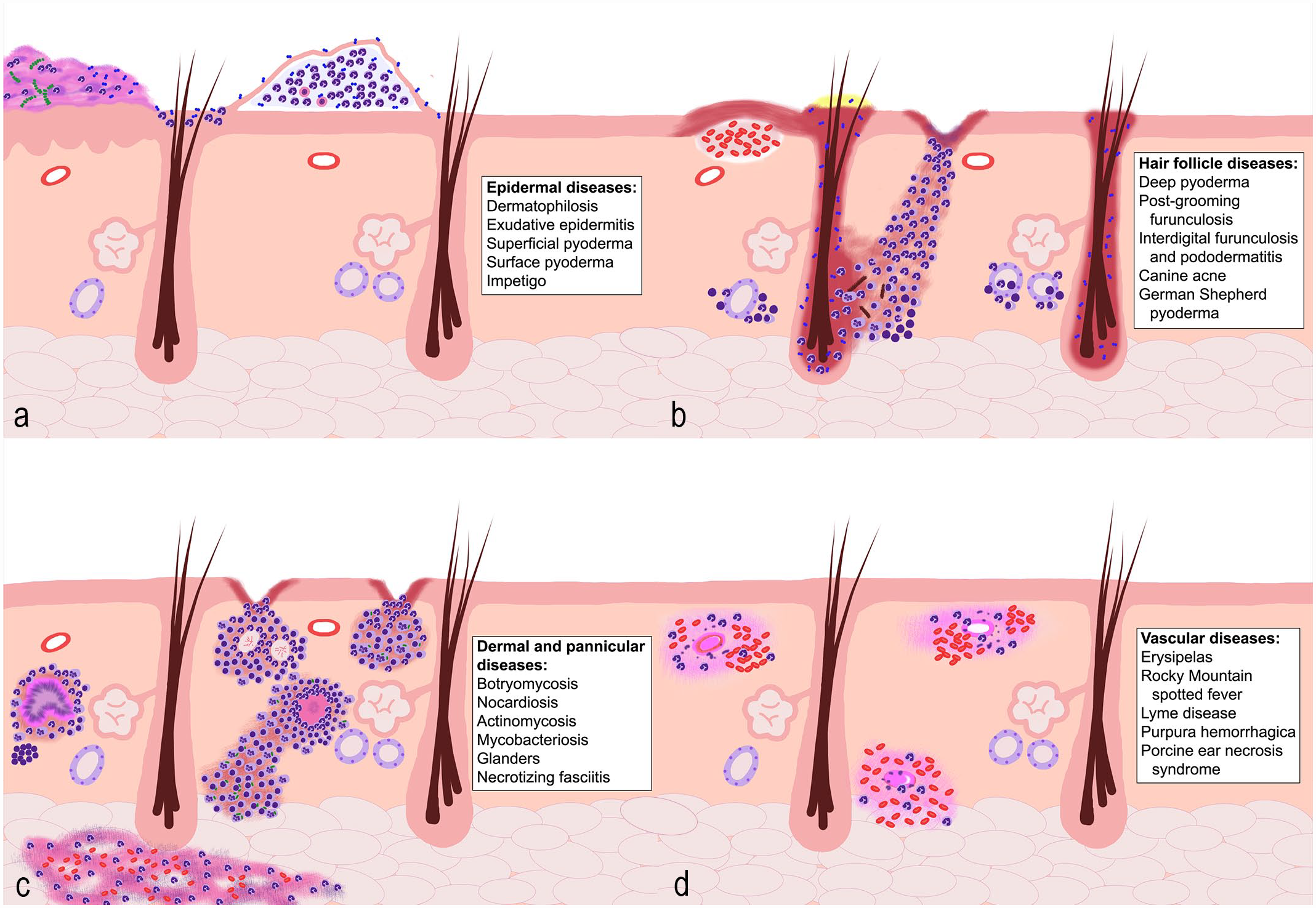
Schematic representation of the main patterns of bacterial diseases affecting the skin and the differential diagnoses associated with each pattern. (a) Bacterial diseases that affect the epidermis. These are often characterized by pustules that may extend to the follicular ostia filled with degenerate neutrophils and occasional acantholytic cells. Chronic lesions can also result in crusts with cellular debris, epidermal hyperplasia, and hyperkeratosis. Bacteria may be present within the pustules, crusts, or in the hair follicle opening. (b) Bacterial diseases that affect the hair follicles. This type of infection causes folliculitis with rupture of the hair follicles, leading to furunculosis and the formation of draining tracts with ulceration of the overlying epidermis. In some conditions, such as postgrooming furunculosis and German Shepherd pyoderma, hemorrhagic bullae can be present. In chronic cases, apocrine adenitis can also be observed. (c) Bacterial diseases affecting the dermis and panniculus. This group of diseases results in nodular inflammation involving the dermis and panniculus. It can lead to draining tract formation, as well as ulceration of the overlying epidermis. This type of inflammatory response will vary from suppurative to granulomatous or pyogranulomatous. The presence of organisms within the lesions varies, and special stains are often required. (d) Bacterial diseases affecting the blood vessels. This group results in vasculitis characterized by fibrinoid necrosis, thrombosis, and hemorrhage. Both superficial and deep blood vessels may be affected. The causative agent is usually not seen in the histological sections.
The Microbiome Inhabiting the Skin Surface of Domestic Animals
It is well established that the skin harbors a variety of microorganisms, including viruses, bacteria, archaea, and fungi, as well as mites.17,36 Compared to humans, animals have a higher diversity of their microbiota. The microbiota is primarily influenced by the host taxonomic order and geographical location, 81 with the canine microbiota being mainly influenced by individual variability and body site. 79 The most common bacterial phyla found on dog skin are Proteobacteria, Actinobacteria, Firmicutes, Bacteroidetes, and Fusobacteria. 80 Cats are inhabited by similar bacterial phyla, although Bacteroidetes is one of the most prevalent phyla on their skin. 65 This phylum predominates in their oral cavity, and it is suggested that the cats’ grooming behavior is responsible for spreading the bacteria to other body sites. 65 The most common bacterial genera on equine skin include Psychrobacter, Macrococcus, Pseudomonas, Acinetobacter, Planomicrobium, Arthrobacter, Carnobacterium, Desemzia, and Corynebacterium, and the microbiota composition in this species is mainly influenced by body site. 70 In cattle, microbiome studies have focused on the udder, which is predominantly colonized by the families Corynebacteriaceae and Staphylococcaceae, 76 and the feet. 101
The skin is a nutrient-poor environment, which by itself regulates the composition of its bacterial community. Among the bacteria that can successfully survive and colonize the skin, some have developed additional strategies to thrive over their competitors. Commensal species, including Cutibacterium acnes (formerly Propionibacterium acnes) and Staphylococcus epidermidis, can metabolize lipids, 18 leading to the production of antimicrobial metabolites and decreased cutaneous pH, which creates a hostile environment for pathogenic organisms. 36 Another survival strategy is the microbial production of bacteriocins and antimicrobial peptides (AMPs). Several coagulase-negative staphylococci, such as S. epidermidis, S. hominis, and S. felis have been shown to produce AMPs that inhibit the growth of Staphylococcus aureus and S. pseudintermedius.19,20,43,60,68 S epidermidis can also induce the production of AMPs by the host (also known as host defense peptides), which can synergistically promote and modulate the immune response, increasing the effectiveness of the innate immune response.19,20,31,60,82,90
Cutaneous dysbiosis is defined as an imbalance of skin bacterial populations. 31 Two models of dysbiosis have been proposed to explain why this phenomenon occurs: (1) dysbiosis driven by microbial communities, where a shift in microbial composition alters the immune response, leading to damage to the barrier homeostasis and subsequent penetration or overgrowth of harmful bacteria; (2) dysbiosis driven by host biology/pathology that alters the barrier and/or immune system and changes the composition and distribution of the microbial population, which results in bacterial overgrowth or penetration and leads to impaired or inappropriate immune responses. 31 Regardless of the mechanism, dysbiotic conditions alter the cutaneous bacterial populations, which can lead to disease.
Bacterial Diseases Affecting the Epidermis
Pyoderma can be classified into three categories based on the depth of the inflammation/infection: surface pyoderma, superficial pyoderma/folliculitis, and deep pyoderma and furunculosis. 10 Surface pyoderma is the least understood category of pyoderma and comprises pyotraumatic dermatitis and fold dermatitis (intertrigo, Supplemental Fig. S1a). In surface pyoderma, bacteria are confined to the skin surface with a dominant inflammatory response. 49 Some authors also include bacterial overgrowth syndrome in this category, 49 although this may not be considered a true pyoderma, as it lacks neutrophilic inflammation. 33
Superficial pyoderma primarily involves the epidermis and can extend into the upper portion of the hair follicles. In deep pyoderma, rupture of hair follicles leads to the expansion of the inflammation toward the adjacent dermis and deep into the panniculus. 49 Because of their distinct histological patterns, these two categories will be discussed in detail in this and the next sections.
Superficial Pyoderma
Superficial pyoderma, also clinically known as bacterial folliculitis, is one of the most common skin diseases in dogs. 49 It involves the penetration of bacteria through the epidermis and invasion into the follicular ostia, leading to folliculitis. The primary pathogen associated with this condition is Staphylococcus spp., primarily S. pseudintermedius, followed by S. intermedius, S. delphini, and S. schleiferi (S. coagulans—coagulase positive, and S. schleiferi—coagulase negative), 10 although other bacterial species can also be associated with this condition.38,56 Staphylococci are normal commensals in the skin of healthy dogs, 79 and it is therefore suggested that underlying factors related to the host may be the primary cause of bacterial pyoderma. In fact, healthy canine skin harbors a significantly diverse bacterial community, while dogs with pyoderma lesions have a loss of diversity and a higher relative abundance of Staphylococcus spp. 66 Likewise, allergic dogs have increased populations of Staphylococcaceae in their skin compared to healthy dogs, especially in the groin and nostrils.12,79 The main conditions associated with pyoderma are hypersensitivities, atopic syndromes, ectoparasites, endocrinopathies, dermatophytosis, and cornification abnormalities.10,49,87 A recent study has shown that 68% of the dogs diagnosed with recurrent pyoderma had one of the aforementioned underlying conditions with allergies being the most common (59%). 87 Other reasons why dogs are considerably more predisposed to pyoderma are related to specific characteristics of the canine stratum corneum, which is thinner and more compact than in other species, lacks a lipid follicular plug, has decreased intercellular lipids, and has a higher pH. 33 These characteristics may favor the survival and adherence of staphylococci on canine skin.
Dogs present with papules, pustules, and epidermal collarettes with peripheral erythema and exfoliation, particularly involving the abdomen, groins, and axillae (Fig. 2a, Supplemental Fig. S1b–d).33,49 Alopecia is variable. 38 Cytological evaluation at the margins of the epidermal collarettes is recommended, where bacteria and neutrophils are far more easily detectable, compared to the center of the collarette. 5 Histologically, pustules with neutrophils and a variable number of eosinophils are in the superficial epidermis, and infundibulum or ostium of superficial hair follicles (Fig. 2b), with or without few acantholytic keratinocytes. Bacteria are not always detected, and crusts are common with older pustules. Inflammation can also follow the isthmus and encircle adnexal glands. In some cases, the inflammatory infiltrate can pierce the hair follicle infundibulum, leading to superficial perforating folliculitis. This phenomenon is distinct from furunculosis, as it remains superficial and does not efface the entire follicle. 38
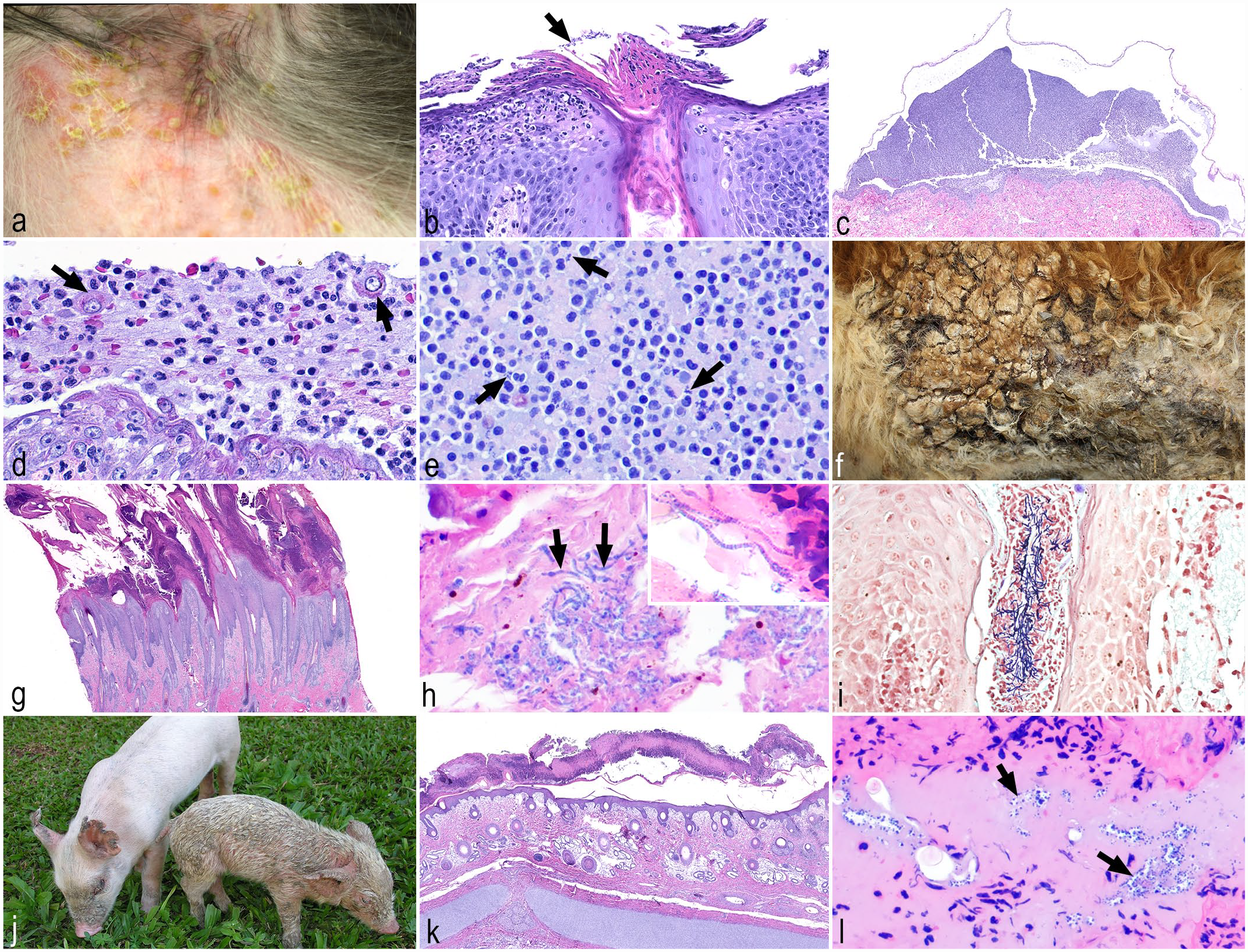
Bacterial diseases affecting the epidermis. (a) Superficial pyoderma, dog. Pustules and epidermal collarettes surrounded by erythema in the axillary region. (b) Superficial pyoderma, dog. Neutrophils infiltrate and aggregate within the epidermis forming superficial pustules with superficial bacterial colonies (arrow), that may extend into the follicular ostia. Hematoxylin and eosin (HE). (c) Impetigo, dog. Intact subcorneal pustule filled with neutrophils. Note shallow erosion of the underlying epidermis. Higher magnification of the cells in the subcorneal pustule is seen in (d) and (e). HE. (d) Impetigo, dog. Few acantholytic cells (arrows) are within pustules, detaching from the underlying epidermis. HE. (e) Impetigo, dog. Coccoid bacteria (arrows) are within neutrophilic inflammation. HE. (f) Dermatophilosis, calf. The skin is covered with dry and domed thickly adherent crusts. (g) Dermatophilosis, horse. Marked epidermal hyperplasia covered with a thick crust with inflammatory cells and necrotic debris. HE. (h) Dermatophilosis, horse. Parallel rows of coccoid bodies with a typical “railroad track” appearance within the crust (arrows, inset). HE. (i) Dermatophilosis, horse. Gram-positive bacteria within the lumen of a hair follicle. Gram stain. (j) Exudative epidermitis or greasy pig skin disease, piglet. Thick crusts cover the face and ears of the piglet on the left, and a greasy and brown exudate with multifocal crusts covers the entire body of the piglet on the right. (k) Exudative epidermitis or greasy pig skin disease, piglet. Nonintact neutrophilic pustules forming extensive and thick serocellular crusts overlay the hyperplastic epidermis. Moderate edema is in the superficial dermis. HE. (l) Exudative epidermitis or greasy pig skin disease, piglet. Aggregates of gram-positive coccoid bacteria (arrows) are within the serocellular crust, which contains numerous degenerative neutrophils. Gram stain.
Impetigo
Impetigo is a superficial pustular dermatitis, most commonly caused by coagulase-positive staphylococci, such as S. pseudintermedius in dogs or S. aureus in other species. 98 Dogs, cats, cattle, goats, and sheep are the most commonly affected species. Lesions usually occur at the time of puberty and are nonpruritic, nonpainful, and frequently heal spontaneously. 56 Predisposing factors are a moist environment, skin abrasions, parasitism, stress, or poor nutrition.56,98 Patients present with small, erythematous papules and pustules affecting the interfollicular epidermis. 56 Lesions occur most often in the axillary and inguinal regions in dogs. In kittens, areas that are in contact with the mouth of the queen during transportation are most affected. 56 Cows, does, and ewes have lesions mainly on the base of the teats and intermammary sulcus of the udder, ventral abdomen, perineum, vulva, medial thighs, and ventral tail.56,98 Pustules are fragile and frequently rupture, producing a honey-colored crust over a shallow erosion (Supplemental Fig. S1e).38,56
Bullous impetigo is a more severe form of the disease that can also affect adult dogs. Pustules are more extensive and span several hair follicle ostia. Patients with impetigo often have underlying conditions, such as iatrogenic or naturally occurring hyperglucocorticoidism, diabetes mellitus, or hypothyroidism.38,56,98 Bacteria other than staphylococci may play a role, such as E. coli or Pseudomonas. 56
The main histologic changes are subcorneal pustules filled with neutrophils that are not associated with hair follicles (Fig. 2c).38,56,98 Bacterial toxins cleave intercellular adhesion molecules (desmoglein 1) leading to keratinocyte detachment and formation of acantholytic keratinocytes, which needs to be differentiated from pemphigus foliaceus (Fig. 2d).38,56,98 In impetigo, fewer acantholytic keratinocytes are seen and cocci bacteria are within intact pustules (Fig. 2e), which can be highlighted with a Gram stain. Specimens with nonintact pustules can be a challenge, as secondary bacterial infections can occur in patients with pemphigus foliaceus. 38
Dermatophilosis
Dermatophilosis, also known as streptotrichosis, rain rot, or rain scald, is caused by Dermatophilus congolensis, a gram-positive facultative anaerobic bacterium. The disease is common in tropical and subtropical climates. Cattle are most often affected. In temperate climates, goats and sheep are commonly affected. Occasional infection occurs in horses and camels.56,84,98 Dermatophilosis can cause major economic losses as the disease can become generalized and animals become seriously ill and udder or teat lesions can hinder the suckling of calves.56,84 The dorsum and distal extremities are most commonly affected, and consist of papules, pustules, and domed, adherent crusts (Fig. 2f).56,98 There are two main predisposing factors: wet conditions/prolonged wetting (hence called also “rain rot” or “rain scald”) and trauma to the skin (eg, due to ticks, insects, or UV damage).56,84,98 Softening of stratum corneum and dissolving of lipid film after prolonged rain or trauma to the skin makes it easier for zoospores/coccoid bodies to overcome the skin barrier. Zoospores then produce branching filaments that infiltrate into the epidermis and outer root sheath of the hair follicles. After penetration, keratinocytes begin to cornify and neutrophils accumulate and inhibit further invasion of the organisms. Then the cycle repeats, resulting in layered, thick crusts.56,98 D congolensis may be more difficult to grow in culture than other bacteria and can be zoonotic.84,98
The histologic hallmark of dermatophilosis is a thick crust with alternating layers of orthokeratosis and parakeratosis, and inflammatory cells and necrotic debris (Fig. 2g). The typical “railroad track” bacteria consist of parallel rows of coccoid bodies seen within these crusts (Fig. 2h). Gram stain is often helpful to highlight typical gram-positive bacteria within crusts (Fig. 2i). There may be intraepidermal neutrophilic micropustules or subcorneal pustules, and suppurative, luminal folliculitis. Inflammation in the dermis is usually mild.56,98
Exudative Epidermitis
Exudative epidermitis (EE), or greasy pig disease, is a well-recognized bacterial proteolytic acantholytic dermatosis of piglets and weaners.28,67 The main cause of EE is the bacterial strains of Staphylococcus hyicus that express exfoliative toxins known as ExhA, ExhB, ExhC, ExhD, SHETA, and SHETB. 30 Methicillin-resistant strains of S aureus (MRSA), 94 S sciuri, 16 and S chromogenes 1 have also been associated with EE in pigs. Although S. hyicus is considered a commensal bacterium on the conjunctiva, snout, ear, and vagina of pigs, it enters the epidermis through abrasions. 27 Desmoglein-1 is the target protein of the exfoliative toxins, which lyse the extracellular domain of this cadherin family glycoprotein. 30 The first lesions of EE are red and moist and start around the eyes, mouth, and ears; in the axilla and groin; and in areas of trauma. Lesions coalesce and quickly form a thick, odoriferous, greasy, brown exudate throughout the entire skin, herein called greasy pig disease (Fig. 2j). 28 Dehydration and anorexia may lead to death. In recently weaned pigs, lesions are confined to the head, and in adults, on the back and flanks. Histologically, the earliest lesions are subcorneal vesicles and neutrophilic pustules with a few acantholytic keratinocytes that may involve hair follicles. As the lesions progress, there is superficial serocellular crusting with large numbers of neutrophils, exfoliated stratum corneum (Fig. 2k), and numerous colonies of gram-positive coccoid bacteria (Fig. 2l).28,67 In chronic cases, the crusts are composed of marked orthokeratosis and parakeratosis; the underlying epidermis is acanthotic with hyperplastic rete pegs; and the dermis is infiltrated with histiocytes, lymphocytes, and plasma cells.28,67
Bacterial Diseases Affecting Hair Follicles and Adjacent Dermis
Deep Folliculitis, Furunculosis, and Pyoderma
Deep pyodermas often develop secondary to traumatic furunculosis, actinic comedones, and even demodicosis; these can be a sequela of superficial bacterial folliculitis. 38 Some forms of deep pyoderma are clinically or histologically distinct, and therefore categorized separately, such as the German Shepherd pyoderma, postgrooming furunculosis, canine acne, and others. 38 A detailed discussion of each of these entities is beyond the scope of this article. However, they share some common pathological features that will be covered below.
Staphylococcus spp. are the major pathogens associated with folliculitis, furunculosis, and deep pyoderma. However, other types of bacteria, such as Streptococcus spp., 23 Proteus spp., Pseudomonas spp., E. coli, 38 Bacillus spp., Corynebacterium spp., and Pasteurella multocida, 56 have also been isolated. Mixed bacterial infections are common. 42 S. pseudintermedius and S. schleiferi are the two most prevalent staphylococcaceae isolated from dogs. Both can develop methicillin resistance, which is a major issue in the treatment of this condition. 42 The zoonotic potential of multidrug-resistant bacteria is another serious concern, and although uncommon and transient, 29 owners of dogs with methicillin-resistant S. pseudintermedius and veterinarians may become carriers of multidrug-resistant staphylococcal organisms.25,73,96
In general, these infections are initially centered within hair follicles, leading to a crusted papule and fragile transient pustules. Affected hair follicles lose the hair shafts, causing patchy progressive alopecia. As lesions become more severe, ulceration and suppurative or serosanguinous exudate are seen, with the formation of crusts (Fig. 3a). When furunculosis occurs, the skin becomes nodular and dark red, with draining tracts, ulcers, severe edema (Fig. 3b), and occasional hemorrhagic bullae. In severe cases, the animal may present with reactive lymphadenomegaly, fever, and anorexia. 56
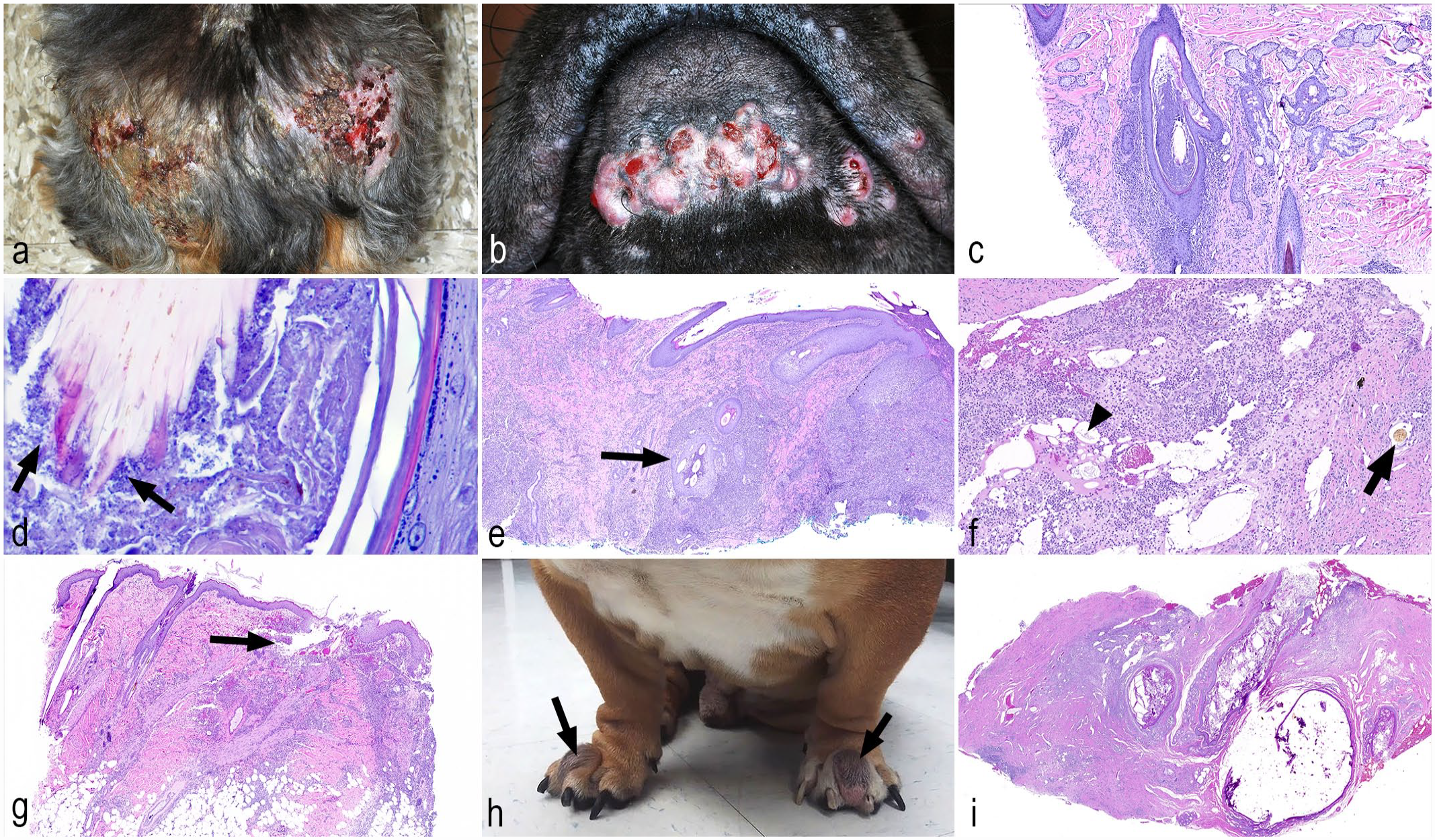
Bacterial diseases affecting hair follicles and adjacent epidermis. (a) Deep pyoderma, dog. Crusts, ulcers, and alopecia at the dorsocaudal region. (b) Deep folliculitis, furunculosis, and pyoderma, dog. Multifocal to coalescing raised alopecic papules/nodules with multifocal exudation of serosanguineous material in the chin and lips. (c) Deep folliculitis, dog. The follicular lumen is dilated with abundant keratin, and there is perifollicular hypercellularity to due to numerous inflammatory cells. Adjacent sebaceous glands are irregularly hyperplastic. Hematoxylin and eosin (HE). (d) Deep folliculitis, dog. Closer view of (c) showing numerous coccoid bacteria (arrows) within the follicular lumen surrounding a hair shaft. HE. (e) Deep folliculitis, furunculosis, and pyoderma, dog. Subgross image demonstrating inflammation centered around and within a hair follicle (arrow) and extending into the deep dermis. The epidermis is hyperplastic and covered with crusts on the right margin. HE. (f) Deep folliculitis, furunculosis, and pyoderma, dog. Inflammation surrounds free keratin (arrowhead) and a single free hair shaft (arrow), with abundant fibrosis on the right lower corner. HE. (g) Post-grooming furunculosis, dog. Ruptured hair follicle with an “exploded” appearance within the infundibular region (arrow), surrounded by inflammation and hemorrhage. HE. (h) Interdigital furunculosis and pododermatitis, dog. In both feet, the interdigital region is swollen and erythematous, forming raised nodules (arrows). (i) Interdigital furunculosis and pododermatitis, dog. Subgross image showing markedly dilated follicles with abundant keratin and surrounded by inflammatory infiltrates. HE.
The histological changes vary greatly, depending on the duration of the infection and the presence of furunculosis. In the initial stages, neutrophilic folliculitis predominates (Fig. 3c, d), which may result in hair follicle distention and rupture (Fig. 3e), leading to a foreign body-type reaction to the free keratin and hair shafts in the adjacent dermis (Fig. 3f). At this stage, a pyogranulomatous reaction is more commonly appreciated.38,56 In the interfollicular dermis, aggregates of neutrophils and hemorrhage can form hemorrhagic bullae. Eosinophils may be present if lesions are accompanied by ectoparasites or allergic conditions. Hemorrhage and fibrosis are variable. Just like in superficial pyodermas, bacteria are not always seen even with the use of special stains, 56 as a high colony-forming unit count is needed for bacteria to be seen histologically.39,100
In dogs3,13 and cats, 99 cases of severe folliculitis, furunculosis, and deep pyoderma have been recently attributed to Burkholderia cepacia (formerly known as Pseudomonas cepacia) associated with contaminated devices, shampoos and grooming products, disinfectants, ear-cleaning solutions, and contaminated surgical cold sterile solution.3,13 Erythematous to hemorrhagic papules, plaques, nodules, alopecia, crusts, and ulcers with draining tracts are primarily in the dorsum, trunk, and upper limbs, and less commonly in the face and inguinal region. Histologically, there is severe pyogranulomatous dermatitis with folliculitis and furunculosis in the dermis and panniculus. Giemsa and Gram stains highlight gram-negative rods inside macrophages and free within the tissue.3,13 The disease can evolve into systemic infection, leading to death by sepsis. In these cases, the bacteria and fibrin thrombi are seen in the liver, lung, spleen, lymph nodes, brain, kidneys, and other organs.13,99
Postgrooming Furunculosis
Postgrooming furunculosis is a severe type of deep folliculitis, furunculosis, and pyoderma associated with vigorous grooming with contaminated bathing products and instruments. 38 The most common associated bacterium is Pseudomonas aeruginosa,37,75,92 but S. pseudintermedius (including methicillin-resistant strains), S. epidermidis, Enterobacter cloacae, S. hominis, Klebsiella oxytoca, B. cepacia, Proteus sp., E. coli, and Serratia marcescens have also been reported.13,14,37 These bacteria have been isolated from shampoos, electric clipper blades, clipper handles, and grooming leashes.34,74
The lesions have an acute onset of 2 days after grooming or water immersion, but they may take up to 7 days to develop. Hemorrhagic crusts, papules, pustules, pinpoint ulcers or draining tracts, and nodules or hemorrhagic furuncles commonly affect the dorsum of the neck and trunk in the majority of patients, 14 as they are application sites of contaminated grooming products. 37 Lesions can also occur in the inguinal region, limbs, and abdomen. 14 Alopecia is also noted as the severity increases. 38 Major clinical signs include pain, fever, and inappetence. 14 Rarely, the disease can evolve into sepsis and death, especially if it involves multidrug-resistant bacteria. 75
Histologically, the hair follicles are ruptured, as if they had “exploded” (Fig. 3g), 38 with severe suppurative furunculosis associated with fragmented hair shafts and keratin, usually within the superficial dermis. Other findings include hemorrhage, edema, and congestion.14,38 Bacteria are not always visible, and Gram stain may be negative. 14 Given the severity of this disease, the lesions tend to be biopsied in its acute stage; therefore, chronic granulomatous furunculosis and scarring are unusual. 38
Interdigital Furunculosis and Pododermatitis
Pododermatitis with interdigital furunculosis, also known as pedal furunculosis, interdigital bullae, and interdigital pyogranuloma, is a deep bacterial infection almost always associated with an underlying factor. 42 It is considered a subgroup of deep pyoderma 38 that affects the interdigital area of dogs, especially short-coated breeds. 42 Initially, the condition starts with single or multiple nodules of interdigital furunculosis with occasional hemorrhage and ulceration, forming draining tracts (Fig. 3h). The underlying causes of furunculosis can be trauma, excessive licking, demodicosis, atopic dermatitis, or allergic diseases that cause pruritus, or conditions that alter weight-bearing.2,61 Most common bacterial species involved include S. pseudintermedius, Streptococcus spp., E. coli, Klebsiella spp., and Pseudomonas spp.42,61 Some lesions resolve spontaneously, while others wax and wane, or can persist indefinitely. 42
Clinically, the interdigital nodules contain purulent or caseous material with numerous hair fragments when expressed.24,42 Other areas of the body are usually not affected. 24 Histologically, the dermis contains multifocal, nodular to diffuse pyogranulomatous dermatitis with furunculosis (Fig. 3i). With chronicity, fibrosis is also appreciated. Special stains are usually negative for infectious agents, unless a secondary bacterial infection develops, and culture and antimicrobial sensitivity tests are recommended in these cases. 42
Bacterial Diseases Primarily Involving the Dermis and Panniculus
Botryomycosis
Botryomycosis is a chronic deep dermatitis that occurs in several species, including dogs, cats, pigs, cattle, horses, laboratory animals, and humans. The disease is commonly known as bacterial pseudomycetoma. The most common agent is S. aureus; however, other bacteria such as Streptococcus spp., Pseudomonas spp., E. coli, Proteus spp., and Pasteurella spp. have been isolated. 71 The infection most often occurs as a result of trauma, such as lacerations, surgery, or punctures, with immunosuppressed individuals being more susceptible.
The subcutis is most often affected; however, multiple organs can be involved. 57 Multiple firm, nonpruritic subcutaneous nodules often have draining tracts and ulcers. 85 The center of the nodules contains purulent material and white granules. 44 In horses, widespread infection follows the lymphatic vessels. 85 Resolution of localized lesions often requires complete surgical excision, as antimicrobials alone are not effective in clearing these infections. 57 Histologically, these nodules are characterized by pyogranulomatous inflammation with numerous multinucleated cells (Fig. 4a). Clusters of gram-positive coccoid bacteria, often observed within the center of these pyogranulomas, are surrounded by deeply eosinophilic club-like material forming the Splendore–Hoeppli reaction (Fig. 4b), which corresponds to the white grains observed macroscopically. 44
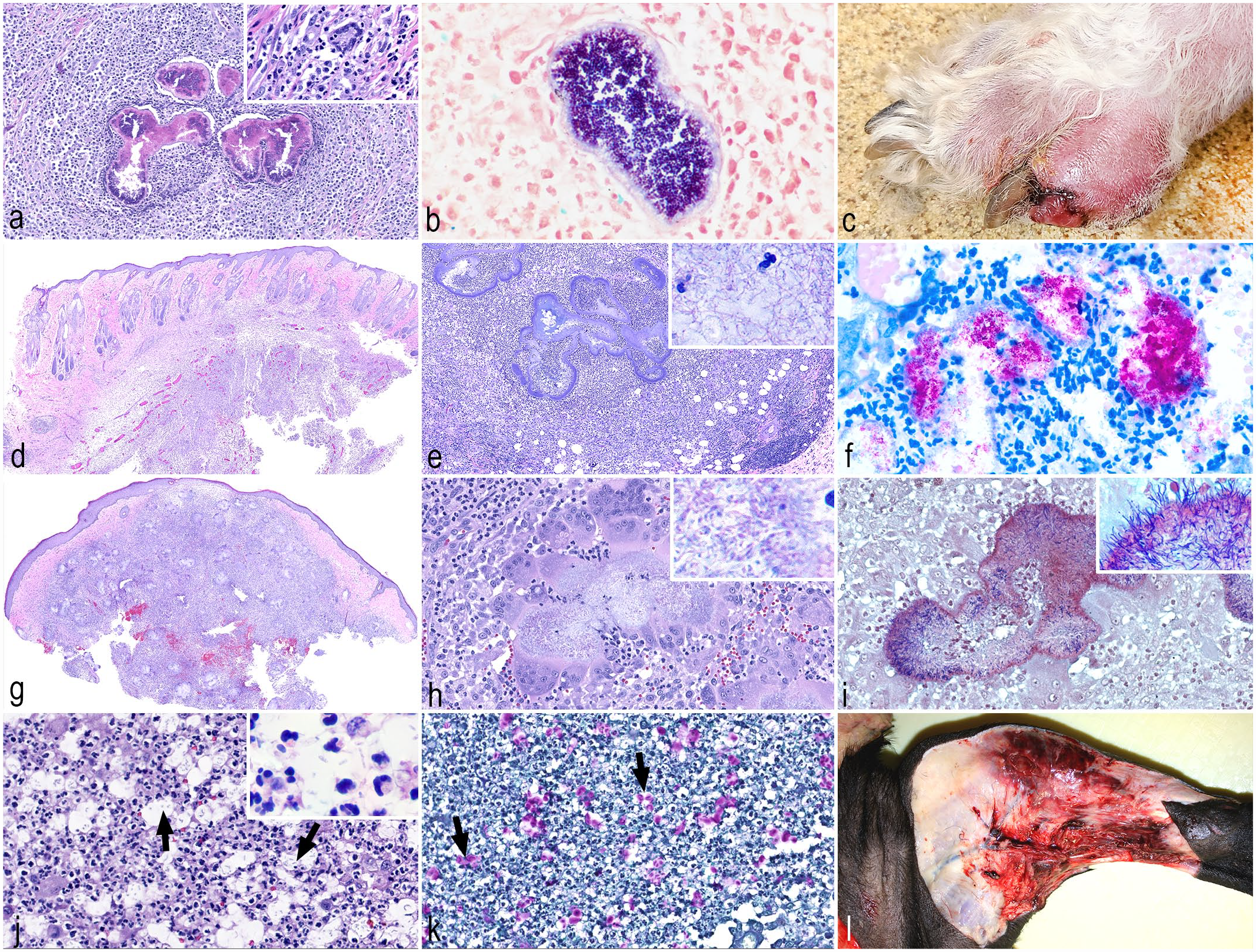
Bacterial diseases primarily involving the dermis and deep panniculus. (a) Botryomycosis, dog. Pyogranulomatous inflammation (inset) with central clusters of coccoid bacteria surrounded by deeply eosinophilic club-like material (Splendore–Hoeppli reaction). Hematoxylin and eosin (HE). (b) Botryomycosis, dog. Numerous gram-positive cocci surrounded by Splendore-Hoeppli reaction. Gram stain. (c) Nocardiosis, dog. Nodule affecting the hind limb digit with ulceration and hemorrhage. (d) Nocardiosis, cat. Subgross image of a locally extensive nodular area of inflammation extending from the mid dermis into the deep dermis and panniculus. HE. (e) Nocardiosis, cat. The organisms aggregate within the center of the inflammatory reaction as serpiginous colonies of beaded and filamentous bacteria (inset). (f) Nocardiosis, cat. Nocardia spp. are acid-fast bacteria. Fite-Faraco. (g) Actinomycosis, dog. Confluent inflammation extending from the superficial dermis to the deep dermis and panniculus with central hemorrhage. HE. (h) Actinomycosis, dog. Colonies of filamentous bacteria (inset) surrounded by Splendore-Hoeppli material and numerous multinucleated giant cells and neutrophils. HE. (i) Actinomycosis, dog. Gram-positive filamentous bacteria (inset) surrounded by the Splendore–Hoeppli material. Gram stain. (j) Pseudomonas luteola, cat. Neutrophils and scattered epithelioid macrophages admixed with 1- to 3-μm-diameter oval organisms with a clear capsule (arrows, inset). HE. (k) Pseudomonas luteola, cat. Elongated bacteria have an approximately 3-μm periodic acid-Schiff (PAS)-positive capsule (arrows). (l) Necrotizing fasciitis, dog. Medial aspect of the leg with overlying skin removed showing necrotic and hemorrhagic subcutaneous tissue.
Nocardiosis
In companion animals, Nocardia spp. can cause chronic skin infections, in addition to frequently infecting the lungs and pleura, whereas in cows it is more commonly responsible for mastitis. These organisms are ubiquitous in the environment, being found in organic matter in soil, water, and plants that can be inoculated into the skin via puncture wounds. Nocardia spp. are facultative intracellular aerobic filamentous bacteria. The most common species infecting the skin are from the Nocardia asteroides complex, which results in cutaneous nodules that may become ulcerated and form draining tracts (Fig. 4c). The infections can be refractory to conventional therapy, and cutaneous infections may be complicated by the invasion of adjacent tissues, leading to pulmonary infections and even disseminated disease, especially in immunosuppressed animals. 78 Histologically, nodular to diffuse pyogranulomatous inflammation in the dermis and subcutis (Fig. 4d) aggregates around tangled colonies of beaded and filamentous bacteria (Fig. 4e). 44 It is uncommon for these organisms to form tissue grains or to be surrounded by the Splendore–Hoeppli reaction. Histologically, Nocardia spp. are gram positive and partially acid-fast positive (Fig. 4f), allowing it to be distinguished from Actinomyces spp., which are negative with acid-fast stains. A modified acid-fast stain like Fite-Faraco, instead of Ziehl-Nielsen, may be more beneficial to confirm Nocardia infection, as it is less susceptible to decolorization during processing.38,44 An issue that is often faced by pathologists is that the bacterial colonies in tissues may be sparse and multiple sections may be needed to be evaluated for organisms to be found.
Actinomycosis
Actinomyces spp. can be normally found in the oral cavity, and cutaneous infections are usually a result of bite wounds or penetrating injury, occurring most often in hunting dogs or animals that have access to outdoor environments. Small animals will often present with cutaneous or subcutaneous nodules, as well as deep cellulitis that may become ulcerated and fistulate. 42 The intralesional colonies can occasionally be seen clinically as yellow-tan sand-like granules.
Actinomyces spp. cause large and often confluent areas of pyogranulomatous inflammation predominantly composed of large numbers of neutrophils surrounded by epithelioid macrophages, similar to those observed in Nocardia infections (Fig. 4g, h). Both organisms are gram positive (Fig. 4i); however, Actinomyces is acid-fast negative. The Splendore–Hoeppli reaction is more commonly observed in Actinomyces, but infrequent in Nocardia infection.38,44 A similar pattern may also be seen in botryomycosis; however, the latter will have intralesional cocci bacteria instead of filamentous bacteria. Multiple lesions are recommended to be biopsied, as these organisms can be difficult to find in histological specimens, and multiple deeper sections may be needed for these aggregates of filamentous bacteria to be found. 38 Rarely, colonies of Actinomyces spp. can form narrow branched hyphae-like filaments in tissues, 59 a characteristic that is also seen in culture, with the formation of spore, spore chain, sporangia, and mycelium. 48
Pseudomonas Luteola
Pseudomonas luteola is a rare gram negative pathogen described in cats 58 and ferrets.6,55,83 It is usually associated with septicemia and pyogranulomatous inflammation affecting the panniculus, pleura, mediastinum, salivary glands, and lungs.6,55,83 A case report in a cat 58 described pyogranulomatous dermatitis and panniculitis with numerous 1- to 3-μm-diameter oval organisms (Fig. 4j) with a 3-μm periodic acid-Schiff (PAS)-positive capsule (Fig. 4k). The organism also stains weakly with Grocott’s methenamine silver. Mayer’s mucicarmine is negative. Because of the unusual morphology of the microorganisms, P. luteola should be a differential diagnosis for other infections, such as leishmaniasis, histoplasmosis, and even cryptococcosis.
Necrotizing Fasciitis
Necrotizing fasciitis, also known as flesh-eating disease, is a rapidly progressive and potentially life-threatening condition characterized by deep necrotizing panniculitis and fasciitis. 93 It is usually caused by bacterial inoculation to the deep tissues, such as cat scratches, 4 vaccinations,35,93 or after traumatic injuries, 11 but the source of the infection might not always be apparent. 97 In dogs, it is most commonly associated with Streptococcus canis, S. pseudintermedius, E. coli, S. zooepidemicus, and P. multocida.4,35,93,97 In cats, this condition has been associated with S. canis, Prevotella bivia, S. pneumoniae, and Acinetobacter baumannii.11,22,91,93 Fournier’s gangrene is a type of necrotizing fasciitis that affects the genital, perianal, and perineal regions. This entity has been described in a cat, caused by Enterococcus faecium, S. epidermidis, and E. coli. 8
Affected animals are usually lethargic with fever and pain, and can present ecchymosis and swelling of the affected areas (Fig. 4l). The lesion can progress to the formation of hemorrhagic bullae and skin necrosis. Histologically, severe necrosis and suppurative inflammation involve the dermis, panniculus, fascia,11,93,97 and muscles, 91 occasionally reaching the bone, causing osteomyelitis 93 and involving joints. 22 Blood vessels are usually necrotic, causing thrombosis and ischemia, with subsequent dermal necrosis.4,93 The condition can rapidly progress to septicemia, affecting multiple internal organs. 91
Mycobacterial and Mycolicibacterial Infections
Based on the extensive phylogenomic analysis, the genus Mycobacterium has been recently emended and four new genera were created to accommodate former members. Therefore, Mycobacterium should only be used for microorganisms of the “Tuberculosis-Simiae” complex, which includes all the major human pathogens. Microorganisms that belong to the clade “Fortuitum-Vaccae” now belong to the genus Mycolicibacterium; those of the “Terrae” clade are now of the Mycolicibacter genus; the “Triviale” microorganisms belong to the Mycolicibacillus genus; and the “Abscessus-Chelonae” clade is now part of the Mycobacteroides genus. 40
In the next sections, we will discuss different mycobacterial and mycolicibacterial infections in different animal species, using the current nomenclature for each microorganism.
Fast-Growing Mycolicibacterial/Mycobacteroidal Infections
Fast-growing mycolicibacterial/mycobacteroidal infections are opportunistic infections caused by species of the genera Mycolicibacterium or Mycobacteroides which grow rapidly in culture (often in less than 7 days). These bacteria are included within the Runyon Group IV (Mycolicibacterium smegmatis complex, M. fortuitum, M. flavescens, M. thermoresistible, and Mycobacteroides Chelonae-Abscessus complex). 53 These organisms can be found in soil and water throughout the world, and infections are often the result of traumatic penetration of these microorganisms into broken skin. 38 These organisms have tropism for lipid, and lesions are most often centered in the subcutaneous tissue. 53 In cats, these infections are often seen in the ventral abdominal region, involving the inguinal fat pads. 38
Clinically, the infection may lead to cutaneous and subcutaneous abscesses, which may fistulate and form draining tracts (Fig. 5a, Supplemental Fig. S1f). Histologically, multifocal to coalescing pyogranulomatous panniculitis often extends to and involves the dermis and panniculus (Fig. 5b). The inflammation is composed of aggregates of neutrophils surrounded by large numbers of epithelioid macrophages mixed with fewer lymphocytes and plasma cells. Neutrophils and macrophages often surround clear, empty lipid vacuoles (lipid from broken adipocytes), and the visualization of this feature justifies the use of special stains. Thin and elongated entangled bacilli to short filamentous bacteria may be lodged within the lipid vacuoles (Fig. 5c). Fite-Faraco acid-fast stain is preferred to identify these organisms, and these may also stain with Grocott’s methenamine silver. 38 The main challenge in the histological diagnosis of this infection is that the organisms may be difficult to find histologically, even with the use of special stains. The lesions can be very similar and must be differentiated from cases of sterile pyogranulomatous dermatitis and panniculitis. Thus, direct smears and culture must be performed prior to ruling out this infection and considering lesions of this nature to be sterile.
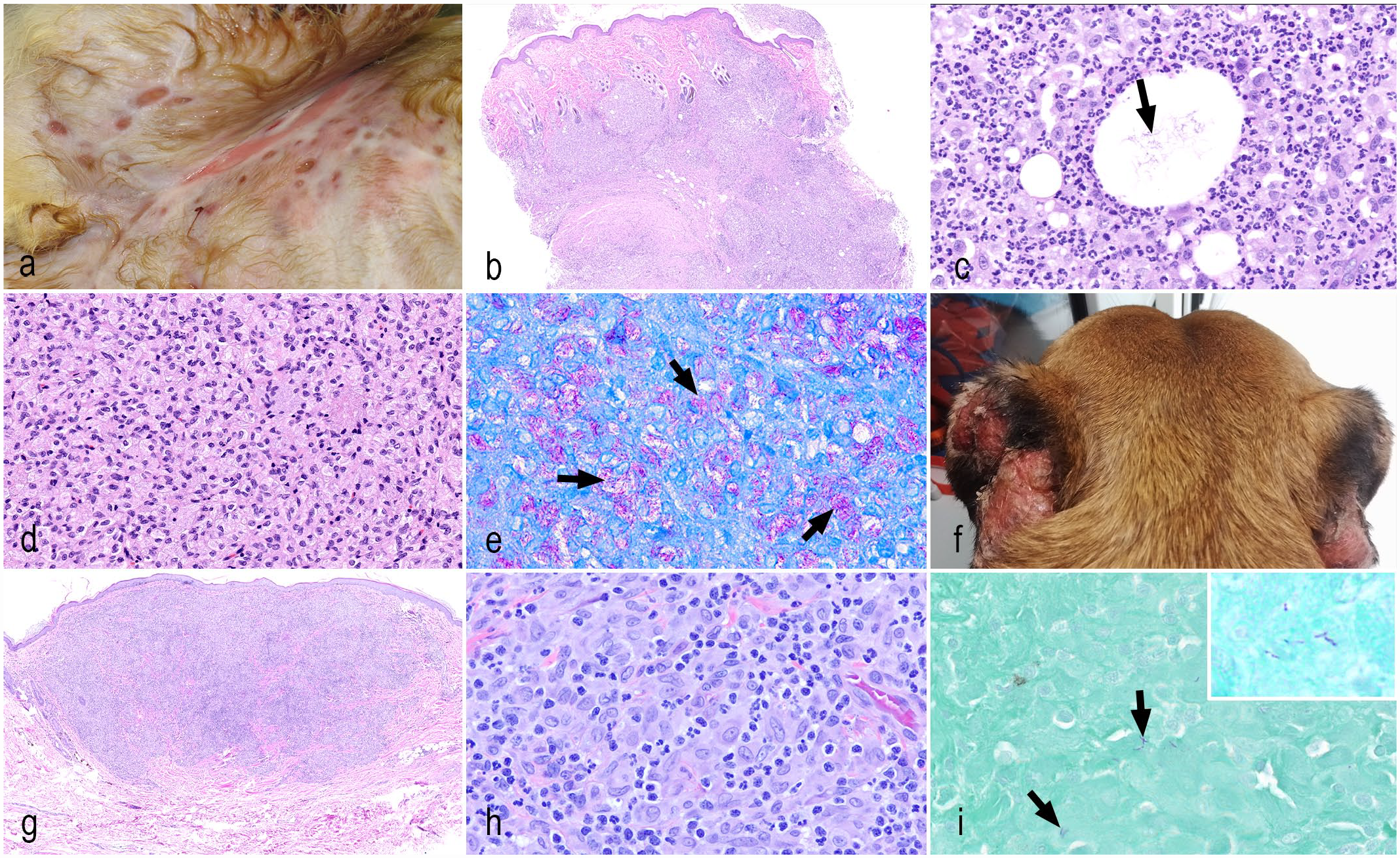
Bacterial diseases primarily involving the dermis and deep panniculus, mycobacterial infections. (a
Slow-Growing Mycobacterial Infections
Slow-growing mycobacterial infections result in cutaneous nodules that may be caused by the tuberculous agents, including Mycobacterium tuberculosis and M. microti or nontuberculous agents, including Mycobacterium avium, M. genavense, M. xenopi, and M. ulcerans. These organisms are fastidious, taking 4–6 weeks to grow and often failing to grow in culture. 52
M tuberculosis, which poses a significant public health risk, rarely causes lesions in the skin of cattle, dogs, cats, and horses. Of these animal species, only cats are more likely to have cutaneous lesions, whereas the other species likely develop pulmonary or alimentary infections. Cutaneous infections are characterized by plaques, nodules, or abscesses that form draining tracts and may involve the head and limbs. It is common for these patients to have systemic disease with fever and anorexia. Histologically, these lesions form pyogranulomas with central necrosis and mineralization surrounded by multinucleated cells and epithelioid macrophages. Scattered organisms are observed within the center of these granulomas. 56
Although less concerning than the tuberculous Mycobacterium spp., many of the nontuberculous slow-growing Mycobacterium spp. may also pose public health risks, especially in immunocompromised individuals. 32 Cats are one of the main species affected by these infections, with lesions mimicking those seen in feline leprosy syndrome. Differentiation between these infections often requires molecular assays, as these organisms often fail to grow in culture. In dogs, miniature Schnauzers and Basset hounds are particularly predisposed to infections with the M. avium complex. Miniature Schnauzers have been identified to have an inherited defect in CARD9, a molecule that plays a role in innate immunity, which makes them more susceptible to M avium infection. 32 The nontuberculous slow-growing Mycobacterium spp. Are primarily distributed within the dermis, and patients present with single or multiple dermal masses. The nodules may fistulate, forming a draining tract, and lymph nodes may be involved. 38 In immunosuppressed individuals, these lesions may become disseminated. Histologically, granulomatous to pyogranulomatous inflammation is characterized by sheets of epithelioid macrophages with few scattered multinucleated giant cells and neutrophils (Fig. 5d). Large numbers of bacteria are often seen in the cytoplasm of epithelioid macrophages with acid-fast stains, especially for lesions caused by the M avium complex (Fig. 5e).
Canine Leproid Granulomas
This condition is mainly seen in short-coated breed dogs, especially Boxers. 51 Patients present with nodules on the ear margins (Fig. 5f), and the feet occasionally affected. It is speculated that these infections may be transmitted by insect bites due to the predilection for sparsely haired areas. Documented outbreaks among related and/or closely housed individuals also suggest the transmissibility of this condition. 89 Patients can recover with complete surgical excision of lesions, and it may be self-limiting in some immunocompetent individuals. 51 The causative agent has yet to be identified, but it appears to be a member of the Mycobacterium genus15,51 that fails to grow in culture media. Studies using similar methods of 16S rRNA gene sequencing have determined a high degree of identity (99–100%) among samples from similarly described cases in Australia, Brazil, Ireland, New Zealand, and the USA.26,89 Ongoing genomic investigations recently performed by our group show this is a novel Mycobacterium species, most likely within the M genovense group.
Cutaneous infection caused by this organism leads to the formation of confluent pyogranulomatous inflammation (Fig. 5f) with sheets of epithelioid macrophages admixed with neutrophils, lymphocytes, and plasma cells (Fig. 5h). Sparse, 2–5 µm acid-fast positive bacilli can be seen within the cytoplasm of epithelioid macrophages (Fig. 5i). The organisms are more likely to stain with Fite-Faraco acid-fast stain.
Feline Leprosy
Feline leprosy is often seen in cats that have access to outdoor environments, and this condition can be divided into three different presentations, which are caused by different pathogens. 62 One of the most common presentations is caused by Mycobacterium lepraemurium, which is seen in many countries, especially in coastal regions. 64 M. lepraemurium can be found in rats, and the development of lesions is believed to be a result of potential encounters with rodents. It is not a zoonotic agent, and no direct transmission between cats has been reported. Few to numerous nodules within the skin and subcutaneous tissues, which may become ulcerated, are seen mainly in cats younger than 3 years of age. They are primarily on the head and limbs, with fewer nodules involving the body and tail, and, only infrequently, the disease can be generalized. The infection has an indolent course, and is associated with a good prognosis even in untreated patients with persistent disease, but without the involvement of internal organs.
The second presentation of feline leprosy has been attributed to a novel Mycobacterium within the M. simiae complex, Candidatus “M. tarwinense,” with lesions seen in the head, especially involving periocular tissues. 63 It is believed that cats acquire this infection during fights with other cats, or from fights or hunting prey species. This condition has an indolent course with a favorable prognosis in most cases. This novel pathogen is almost entirely confined to the Victoria region in Australia.
The third presentation is associated with Candidatus “Mycobacterium lepraefelis” infection, which results in a much more severe presentation of feline leprosy, where the organisms may become disseminated. 62 This organism has been identified in Australia and New Zealand, and one case was identified in British Columbia, Canada. This disease affects middle-aged to older, preferably male, cats with outdoor access. Patients present with widespread cutaneous lesions, which can be followed by systemic involvement. Due to the progressive and systemic involvement, this infection may lead to death or result in euthanasia.
Histologically, these Mycobacterium spp. may cause lepromatous or tuberculoid leprosy in cats. In lepromatous leprosy, sheets of epithelioid macrophages admixed with neutrophils, plasma cells, and lymphocytes are seen within the dermis and panniculus, with large numbers of organisms observed within the cytoplasm of macrophages. In tuberculoid leprosy, dermal to subcutaneous granulomas with central caseous necrosis are observed, and low numbers of organisms are seen within necrotic areas, similar to lesions caused by tuberculosis. Lepromatous leprosy was the most common presentation in cases caused by Candidatus “M. lepraefelis.”
Farcy
This is a chronic skin disease in cattle that causes lymphangitis and dermatitis that is primarily distributed within Sub-Saharan Africa. The causative agents include Mycolicibacterium senegalensis and Mycolicibacterium farcinogenes. The infection can be zoonotic, although only rare cases have been reported in humans. However, the main public health concerns related to farcy are because lesions can mimic those seen in cattle with tuberculosis. It is believed that farcy enters the skin via abrasions and wounds. Tick transmission may also play a role due to the geographic distribution of this disease, as well as the fact that breeds resistant to ticks are also resistant to farcy. The bacteria initially penetrate the subcutaneous tissues resulting in cellulitis, invading lymphatics and involving regional lymph nodes. Clinically, linear nodules with draining tracts and leakage of yellow purulent material are the most characteristic lesion seen in the chronic phase of this disease. Histologically, classic granulomas with central necrosis and mineralization surrounded by macrophages and multinucleated giant cells are observed. Only rare to few acid-fast organisms may be seen histologically, although oftentimes these may be missed due to the paucity of bacilli within histological sections. 41
Glanders
Glanders is a zoonotic infection seen in horses, mules, and donkeys caused by Burkholderia mallei. This agent is highly contagious and can be fatal in humans. It is included as one of the category B bioterrorism agents by the Centers for Disease Control and Prevention. The disease has been eradicated in many countries, although scattered outbreaks are still seen in some regions around the world. Asymptomatic patients and those during early infection periods can serve as the source of infection in other animals. The organisms can invade the skin, mucous membranes, and gastrointestinal tract. Infected animals may also develop systemic signs including fever, anorexia, weight loss, and cough. Patients can present with a cutaneous, nasal, and/or pulmonary form, with a combination of cutaneous and pulmonary forms being the most common clinical presentation in disease outbreaks. The cutaneous lesions of glanders are similar to those seen in farcy in cattle, and “farcy” is also a term used to describe glanders in affected animals. Nodular cutaneous ulcers in a “crate-like” pattern that appears as chains with drainage of serous and purulent exudate are the characteristic lesions. The linear aspect of the lesions is the result of severe lymphangitis. 46
Bacterial Diseases Causing Vasculitis
Erysipelas
Erysipelothrix rhusiopathiae is a gram-positive bacillus, 95 known to cause several syndromes in pigs, such as septicemia, arthritis, vegetative endocarditis, cutaneous vasculitis, and abortion. 69 This pathogen is zoonotic and is reported in a variety of mammals, birds, reptiles, and invertebrates. 95 Wild boars are thought to be a reservoir for the bacteria, posing a threat to domestic pigs and other species. 88
With acute septicemia, the pigs develop cyanotic or erythematous extremities and small papules or large, raised, firm plaques with characteristic diamond or rhomboid shapes (Fig. 6a). As the disease progresses, the lesions become dry and darker with a necrotic center, and the skin can be easily peeled from the underlying tissues (Fig. 6b). In severe cases, the ears, tail, and foot can slough due to necrosis. 69
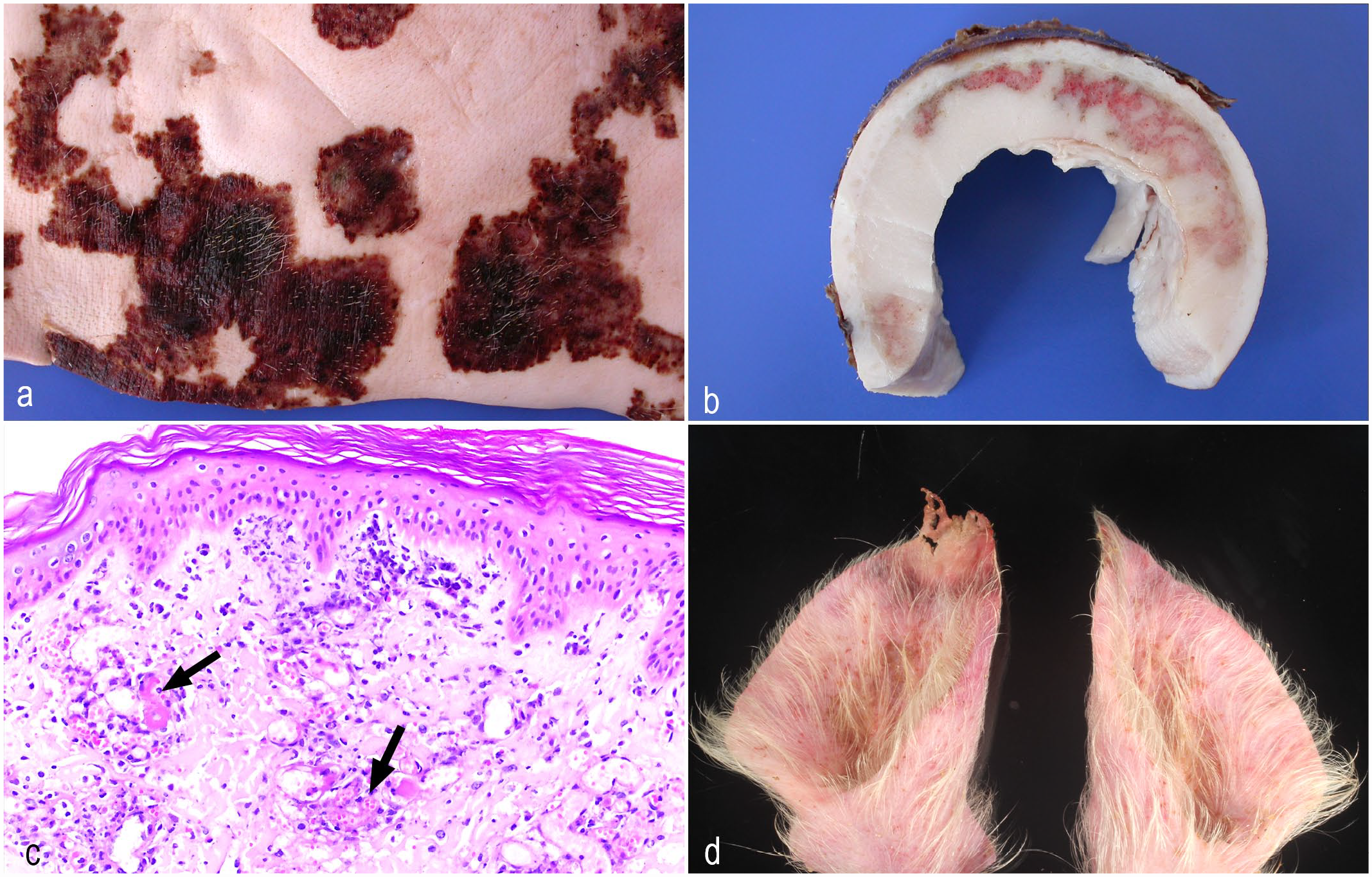
Bacterial diseases causing vasculitis. (a) Erysipelas, pig. Haired skin with multifocal to coalescing dry and brown plaques with a necrotic center. (b) Erysipelotrichosis, pig. Cut section of haired skin depicting necrotic surface with underlying areas of hemorrhage and necrosis in the dermis and panniculus. (c) Erysipelotrichosis, pig. Multifocal vascular thrombosis with scattered neutrophilic infiltrates within and around blood vessels with overlying acute necrotic epidermis. Courtesy of Dr. David Driemeier and Dr. Paula Pereira. Hematoxylin and eosin (HE). (d) Porcine ear necrosis syndrome. The ear tips are necrotic with a rim of congestion between the necrotic and viable tissue.
Histologically, there is marked dermal neutrophilic vasculitis with thrombosis (Fig. 6c), which gives the characteristic diamond appearance grossly, with cutaneous necrosis and suppurative hidradenitis. 56 The cutaneous form of erysipelas has also been reported in dogs with alopecia and erythematous macules. Histologically, they consisted of neutrophilic vasculitis and fibrinoid necrosis at all levels of the dermis, mixed with fibrin, hemorrhage, and edema. 54
Rocky Mountain Spotted Fever
Rocky Mountain spotted fever is caused by Rickettsia and is transmitted by ticks, mainly by Dermacentor variabilis or D. andersoni, Rhipicephalus sanguineus, and Amblyomma sculptum, and is, therefore, a seasonal disease.47,98 The disease is most prevalent in North, Central, and South America. Dogs are especially susceptible to infection and clinical disease, and dogs may even serve as sentinels for the disease in humans. 47 Fever is a common early clinical sign and develops three to seven days after the tick bite. Other clinical signs include lethargy, anorexia, skin rash, tremors, ocular lesions, thrombocytopenia, and leukocytosis. The integumentary, respiratory, central nervous, gastrointestinal, and urogenital systems can be affected.47,98 Clinically, erythema, petechiae, necrosis, ulceration, and edema are visible. Rickettsia spp. targets the host’s endothelial cells and causes direct endothelial damage, which results in endothelial proliferation, inflammation, and hemorrhage. Histologically, neutrophilic small vessel vasculitis with leukocytoclasia, fibrinoid necrosis, and fibrin thrombi are observed.56,98 Treatment of insufficient duration may lead to disease relapse. 47
Lyme Disease
Lyme disease is caused by Borrelia burgdorferi, a gram negative spirochete, transmitted by Ixodes spp. The disease is described in dogs, horses, and humans. In humans, an expanding ring-like erythema develops about 1–2 weeks after the tick bite. 56 Dogs can have a rash, urticaria, and moist dermatitis. 56 In horses, cutaneous lymphoid hyperplasia, formerly pseudolymphoma, has been reported. One reported horse displayed dermal papules ranging from 2–7 mm in size across the ventral aspect of the right masseter. 86 Histologically, densely cellular infiltrates consisting of large, lymphoblastic cells, intermixed with small, mature lymphocytes are observed. The infiltrate was separated from the epidermis by a Grenz zone. Immunohistochemistry showed a mixed CD3 and CD79a-positive cell population. 86 Cutaneous lymphoid hyperplasia may be a more general reaction of tick bites.
Purpura Hemorrhagica
Purpura hemorrhagica is common in horses and is most commonly associated with Streptococcus equi subsp. equi (strangles-associated) and Corynebacterium pseudotuberculosis infections, or in correlation with vaccinations or other infections, such as Rhodococcus equi, equine influenza virus, or equine herpesvirus type 1.77,98 The skin, respiratory tract, or gastrointestinal tract may be affected. Clinically, there is well-demarcated subcutaneous edema on all four limbs, hemorrhages in the mucous membranes, and purple macules in the ventrum, head, and distal limbs.77,98 Systemic signs include fever, depression, and anorexia. The histologic lesions are an immune-mediated vasculitis due to antigen-antibody-complex deposition in vessel walls (type III hypersensitivity reaction) with fibrinoid necrosis and neutrophilic, leukocytoclastic vasculitis. Lesions may progress to ulceration and sloughing of the skin. 98 Most horses survive the disease with treatment 77 ; however, there may be complications due to dermal necrosis, laryngeal edema, thrombophlebitis, glomerulonephritis, and colic. 7
Porcine Ear Necrosis Syndrome
Porcine ear-tip necrosis (ETN) results in progressive loss of the ear helix in the nursery and early-grower pigs. 21 Several synonyms have been used for this syndrome over the years, such as porcine ear necrosis, ear necrosis syndrome, ulcerative spirochetosis of the ear, or streptococcal auricular dermatitis. 50 ETN has a global distribution, being most common in countries with intensive pig farming, leading to welfare implications. 9 Risk factors such as high stocking rate, fully slatted flooring without straw, high ambient temperature, poor air quality, and high humidity have been associated with outbreaks of ETN. 72
Neither the etiology nor the pathogenesis of ETN has been fully elucidated. Three hypotheses have been proposed for the development of ETN: (a) necrosis of the epidermis due to exfoliative toxins of staphylococci, mainly S. aureus; (b) occlusion of capillaries by cold agglutinins or immune complexes produced during infection with specific pathogens, such as Mycoplasma suis; and (c) trauma, such as ear biting or chewing, with β-hemolytic streptococcal infection.21,50 Furthermore, Treponema pedis has been detected in the lesions of ETN, with proposed infection by biting and licking; however, experimental infection failed to reproduce the disease. 45
Initial lesions consist of local erythema, which is often associated with ear biting, followed by necrosis (Fig. 6d) and loss of part of the ear pinna if healing occurs. Open wounds are a port of entry for opportunistic pathogens. 21 Histologic lesions include crust formation on the epidermis with granulation tissue in the dermis and necrosis of the chondrocytes. 21 Vasculitis is not a consistent finding in ETN. 72
Conclusion
The definitive diagnosis of cutaneous bacterial infections still relies on clinical signs confirmed by bacterial culture and/or molecular techniques. However, the recognition of specific histological patterns helps narrow down the differential list to better allocate the resources for ancillary diagnostic tests. Therefore, it is crucial for pathologists to recognize the different patterns that bacterial diseases can cause, when special stains can or cannot be useful, and what are the main anatomical structures involved in each case, to provide clinicians a more comprehensive diagnostic report.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231176558 – Supplemental material for Common superficial and deep cutaneous bacterial infections in domestic animals: A review
Supplemental material, sj-pdf-1-vet-10.1177_03009858231176558 for Common superficial and deep cutaneous bacterial infections in domestic animals: A review by Mayane Faccin, Dominique J. Wiener, Raquel R. Rech, Domenico Santoro and Aline Rodrigues Hoffmann in Veterinary Pathology
Footnotes
Acknowledgements
The authors acknowledge Drs. Adam Patterson, David Driemeier, and Paula Pereira for gently providing pictures for this paper. They also would like to acknowledge the Gastrointestinal Laboratory staff at Texas A&M University for slide scanning.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
