Abstract
A male Malayan tiger cub developed well-circumscribed, erythematous, alopecic lesions on the face, torso, and paws when 1-wk-old. Biopsies of a torso lesion and a right front paw lesion at 1-mo-old confirmed cutaneous mast cell tumors (MCTs). MCTs on the paws grew into pendulous masses up to 6.5 cm in diameter by 3-mo-old, but those on the face and torso regressed. Fine-needle aspiration of the spleen at 3-mo-old revealed marked mast cell infiltration. The spleen and the right paw cutaneous MCT were removed; the paw MCT recurred within 7 d. A 12-bp tandem duplication, suggesting a somatic mutation, was identified in exon 8 of c-KIT in DNA extracted from the cutaneous MCT on the right paw and from one over the torso, but not from the spleen. Remaining MCTs on the paws regressed slowly following splenectomy and had completely regressed by 1-y-old. At 7-y-old, there was no recurrence of any mast cell disease. Mast cell disease in this tiger cub is similar to a report in a domestic kitten and to pediatric mastocytosis in humans, which commonly begins in infancy, improves by adolescence, and is associated with somatic c-kit mutations. To our knowledge, mastocytosis has not been reported previously in a juvenile exotic felid.
A male Malayan tiger (Panthera tigris jacksoni) cub developed ulcerated, round, raised, well-circumscribed, erythematous, and alopecic lesions on the face, torso, and paws (Figs. 1, 3) when 1–2-wk-old. Its 2 siblings were unaffected. At 4-wk-old, histologic examination of biopsies confirmed the lesions as cutaneous mast cell tumors (MCTs). At 5-wk-old, the cub was growing and was behaviorally normal. However, it was smaller than its siblings (Suppl. Fig. 1). No new tumors were noted at this time. At 6-wk-old, the largest MCT was observed on the right hind paw and was treated at that time, and again at 8-wk-old, with an intralesional triamcinolone injection (1.8 mg dose each time) with no effect. MCTs on the paws continued to enlarge and became pendulous at 9-wk-old. At 12-wk-old, the MCTs on the paws were still enlarging but the cub was gaining weight and was clinically normal. MCTs on the face and torso were up to 2 cm in diameter. In contrast, MCTs on the palmar and plantar surfaces of the paws were up to 6.5 cm in diameter and ulcerated, often spanning 2 digits and compressing the paw pads.
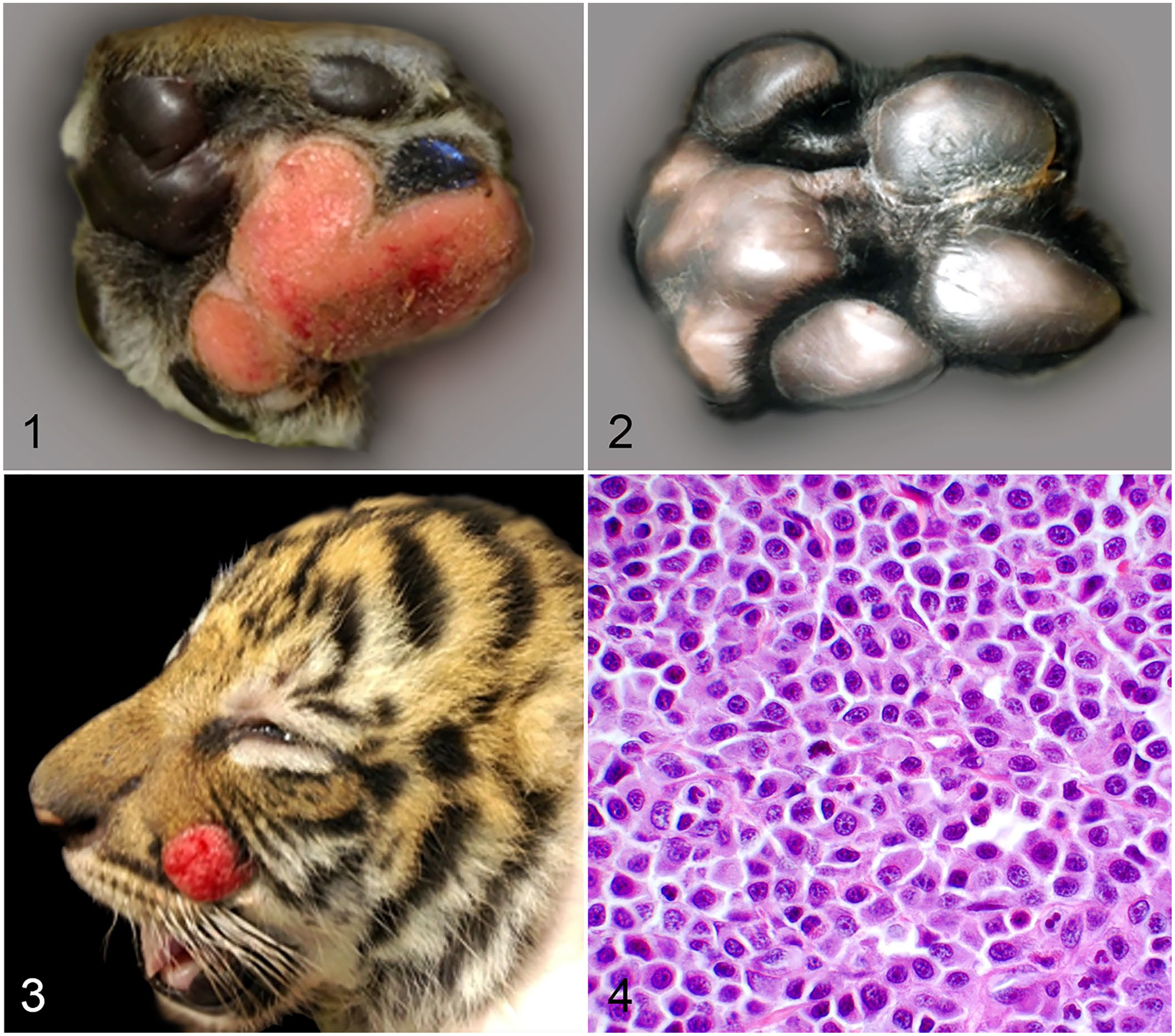
Cutaneous mast cell tumors in a Malayan tiger cub.
At 13-wk-old, fine-needle aspiration of the spleen was performed, and mastocytosis was identified. Splenectomy was performed, and the MCT on the right hind paw was removed surgically. The spleen was enlarged and firm with frequent pinpoint white foci; it weighed 59.5 g (0.65% of body weight) and was 15 cm in length. The MCT on the right front paw recurred 7 d postoperatively. At 19-wk-old, aspirates of the liver, kidney, bone marrow, and popliteal lymph nodes and buffy coat smears were evaluated and revealed no evidence of systemic mast cell disease. Between 5 and 8 mo of age, it could no longer be determined if the MCTs on the paws were still enlarging. The cub was growing quickly and was behaviorally normal. The MCTs were noted to be regressing at 8–12-mo-old, and by 12-mo-old only scar tissue was noted at the previous tumor site (Fig. 2). When 7-y-old, the tiger did not have any recurrence of mast cell disease, and mast cells have not been found in buffy coat smears.
Skin biopsies of cutaneous MCTs obtained at 1- and 3-mo-old were similar and had diffuse dermal infiltrates of mast cells 12–18-µm diameter with moderate-to-abundant eosinophilic cytoplasm that contained faintly basophilic granules (Fig. 4). Nuclei were round, oval, or irregularly shaped, and contained coarsely stippled chromatin and 1 or 2 small nucleoli in most cells. Occasional binucleate mast cells and mature eosinophils were present. The epidermis was hyperplastic or ulcerated. Small numbers of mast cells were identified in regional lymph node aspirates. Mast cell infiltrates in the spleen were multifocal and identical to those in cutaneous tumors. Mast cells were present in splenic vessels and within the subcapsular and medullary sinuses of splenic lymph nodes removed with the spleen. Mastocytemia was not observed.
Immunohistochemical labeling to detect Ki67 and KIT were performed on MCTs obtained by biopsy at 1-mo-old. The right front paw MCT had 11 mitoses in 2.37 mm2, and a Ki67 labeling index of 35.4 (positive nuclei per 1 cm2 grid reticle, average of 5 grids counted). A torso tumor had 14 mitoses in 2.37 mm2, and a Ki67 labeling index of 40.6 (positive nuclei per 1 cm2 grid reticle, average of 5 grids counted). These Ki67 grid-based indices reflect Ki67 indices of ~5.1% and 6.8% of neoplastic cells, respectively. MCTs had weak perimembranous-to-cytoplasmic immunolabeling for KIT. Occasional perinuclear clumping was noted in the tumor from the right front paw.
Genomic DNA was extracted from paraffin-embedded sections of the right front paw cutaneous MCT sampled at 1- and 3-mo-old, the torso MCT sampled at 1-mo-old, and the spleen removed at 3-mo-old using a published protocol. 20 A PCR assay for the analysis of exon 8 of the c-KIT gene was performed as described previously. 6 A 12-bp tandem duplication mutation in exon 8 of c-KIT, as described previously in a cat, 8 was identified and confirmed by sequencing in all cutaneous MCTs, but not within the spleen, indicating that the mutation was somatic and not genomic. It is unknown why the mast cells in the spleen did not have this mutation. However, no analysis for mutations within other exons of c-KIT was performed. It is also possible that the splenic mast cells were not neoplastic and simply represented an immune reaction. Finding a mutation in the cutaneous MCTs confirms neoplastic transformation at these sites and may indicate a more aggressive phenotype.
MCTs are reported rarely in exotic felids as cutaneous or visceral tumors in geriatric animals.2,4,5,16 To our knowledge, cutaneous MCTs have not been reported in a juvenile exotic felid. The MCTs in this tiger cub are morphologically and behaviorally similar to those reported in domestic kittens, most commonly Siamese, with the exception of the unusually large tumors in the paws and splenic involvement in the tiger cub. In domestic kittens, cutaneous mast tumors are hypothesized to be reactive lesions that regress spontaneously.3,18 Because of the large paw tumors in this tiger cub and concern for splenic metastasis, steroid treatment and surgical excision of one tumor were attempted (with no success), splenectomy was performed, and proliferation indices, KIT immunohistochemistry, and c-KIT exon 8 sequencing were performed. Generally, a mitotic count >5 per ten 400× fields, tumor multiplicity, a high Ki67 labeling index (>25% of neoplastic cells), and cytoplasmic KIT expression are poor prognostic indicators in feline MCTs.11–14 Mitotic count, multiple tumors, and mutation status all suggested a poor prognosis in this tiger cub, but tumors regressed. Based on a new histologic grading scheme for feline cutaneous MCTs, these tumors would have been considered low grade, given that they did not meet the criteria for a high-grade designation. Although the tiger cub MCTs had high mitotic counts and tumor diameters >1.5 cm, they did not have significant nuclear atypia. 13 In a published case series of 20 cutaneous MCTs in domestic cats, 12 of which had a c-KIT mutation, no association was found between c-KIT mutation and histologic grade or mitotic count in the domestic cats. 17 That study also included cutaneous MCTs from 4 cheetahs, but exact grading and mitotic counts were not reported for those tumors. 17
Splenectomy may induce tumor regression and prolong survival in domestic cats with cutaneous and visceral MCTs, 7 and may have been helpful in our case. However, because facial and torso tumors were already regressing prior to splenectomy, the role of splenectomy in tumor regression in this tiger cub is uncertain. It is also uncertain why the paw tumors persisted longer and were substantially larger than those on the face and trunk. Paw tumors were traumatized by ambulation and the cubs playing with them, but trauma is not reported to exacerbate growth of cutaneous MCTs.
The somatic c-KIT exon 8 tandem duplication mutation identified in the proliferating cutaneous mast cells was similar to a mutation described in a cat with systemic mastocytosis and mastocytemia. 8 Exons 8 and 9 encode the fifth immunoglobulin-like domain (IgD) of the extracellular domain of KIT in cats, and are the most common sites for gain of function mutations in feline MCTs; these mutations may render the tumors sensitive to treatment with tyrosine kinase inhibitors. 9 Similar somatic c-KIT mutations, and the clinical presentation observed in our case, have been described in pediatric mastocytosis in humans. 1 Data in humans suggests that the presence of additional mutations in multiple other genes correlates better with progression to malignant disease.10,15,19 The findings in our case similarly suggest that high proliferation indices and multiple cutaneous tumors in a kitten with mast cell disease do not necessarily indicate a poor prognosis.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387221074709 – Supplemental material for Cutaneous and splenic mastocytosis in a juvenile Malayan tiger
Supplemental material, sj-pdf-1-vdi-10.1177_10406387221074709 for Cutaneous and splenic mastocytosis in a juvenile Malayan tiger by Rebecca C. Smedley, Nancy L. Stedman and Matti Kiupel in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank the veterinary and animal care staff at Busch Gardens Tampa, FL and Dr. Helga Bleyaert of BluePearl Pet Hospital, Tampa, FL.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
