Abstract
Lipopolysaccharide (LPS) has dose-dependent biphasic functions (cell protective versus cell toxic). To clarify the different effects of LPS on liver homeostasis or liver diseases, comparisons were made between low and high doses of LPS, in terms of the mutual relation of hepatic macrophages, autophagy, and damage-associated molecular patterns (DAMPs) in male F344/DuCrlCrlj rats. Rats injected with low dose (0.1 mg/kg) or high dose (2.0 mg/kg) of LPS were examined at 6, 10, and 24 hours following single injections. Histologically, focal hepatocellular necrosis was occasionally present in high-dose animals, whereas there were no significant changes in low-dose animals. In low-dose animals, Kupffer cells reacting to CD163 and CD204 were hypertrophic and regarded as M2 macrophages, which promote resolution of inflammation and tissue repair, whereas in high-dose animals, infiltration of M1 macrophages expressing CD68 and major histocompatibility complex class II, which enhance cell injury, was seen. Hepatocytes with high-mobility-group box-1 (HMGB1) (one of DAMPs)-positive cytoplasmic granules appeared more frequently in high-dose animals than in low-dose animals, indicating the translocation of nuclear HMGB1 into the cytoplasm. However, although light-chain 3 beta–positive autophagosomes in hepatocytes increased in both doses, abnormally vacuolated autophagosomes were only seen in injured hepatocytes in the high-dose group, indicating possible extracellular release of HMGB1, which might result in cell injury and inflammation. These findings suggested that low-dose LPS induced a favorable mutual relationship among hepatic macrophages, autophagy, and DAMPs leading to cytoprotection of hepatocytes, whereas failures of the relationship in high-dose LPS caused hepatocyte injury.
Lipopolysaccharide (LPS) is a constituent of gram-negative bacterial cell walls. Exposure to high doses of LPS (close to the lethal dose), which may be relevant to bacterial infection such as colibacillosis, leads to liver injury characterized by midzonal hepatocellular necrosis with inflammation, 38 whereas exposure to a low dose of LPS may result in a noninjurious response without inflammation. 10 Interestingly, it has been reported that pretreatment with a nontoxic amount of LPS increases resistance to tissue damage caused by ischemia/reperfusion 25 or an hepatotoxic substance. 22 In our previous study, it was reported that the pretreatment with a low dose of LPS (0.1 mg/kg body weight) at 24 hours before thioacetamide (TAA; a hepatotoxicant) injection significantly reduced the liver lesions, compared with those in rats injected by TAA alone. 23 It is likely that macrophages and autophagy in hepatocytes might be attributable, at least in part, to the reduction of hepatic lesions induced by TAA. 23 LPS may have biphasic functions (cell toxic versus cell protective), depending on the low or high dose. However, the mechanisms of the biphasic functions remain to be investigated.
LPS mainly stimulates hepatic macrophages, especially Kupffer cells via Toll-like receptors (TLRs), and induces production of inflammatory mediators.21,33 In particular, activation of TLR in Kupffer cells by LPS is known as a key mechanism for liver injury, such as hepatocellular necrosis, which is induced through the production of inflammatory cytokines.27,39 Macrophages appearing in pathological settings are divided as classically activated macrophages (M1) and alternatively activated macrophages (M2).35,36 CD68- and MHC class II–expressing macrophages are considered as M1, which enhance cell injury by producing proinflammatory cytokines.20,35 CD68 expression implies the extent of phagocytic activity, 8 and the MHC class II molecule is involved in antigen-presentation. 41 M2 macrophages are identified by CD163 and CD204 expression 36 and promote resolution of inflammation and tissue repair.20,35 M2 macrophages are reported to have beneficial properties against inflammatory disorders. 34 The CD163 antigen is a glycoprotein, which belongs to the scavenger receptor cysteine-rich group B family and functions as the scavenger receptor for hemoglobin-haptoglobin complexes. 24 CD204 is known as a macrophage scavenger receptor, and its expression is related to the metabolism of oxidized low-density lipoprotein. 31 Macrophages appear to play biphasic roles in the development of pathological lesions.
LPS also plays important roles in the activation of autophagy and release of damage-associated molecular patterns (DAMPs). Autophagy is an intracellular self-degradation process that delivers cytoplasmic constituents such as damaged organelles and invading microbes to lysosomes. Such cytoplasmic debris are enclosed by an isolation membrane to form autophagosomes. Autophagosome is characterized pathologically by the formation of double-membrane vesicles, demonstrable with LC3B (microtubule-associated protein 1 light-chain 3 beta) immunohistochemistry. 19 The autophagosome subsequently fuses with lysosomes (formation of autolysosomes), and the internal unnecessary materials are degraded. 18 Autophagy in hepatocytes is activated via a TLR-dependent activation pathway.5,17
DAMPs are released from injured or necrotic cells and act as ligands that can activate pattern-recognition receptors, such as TLRs, resulting in the activation of immune and inflammatory responses. 12 TLRs are mainly expressed by macrophages and hepatocytes in the liver.12,15 One of the best-known DAMPs is high-mobility-group box-1 (HMGB1), a nonhistone nuclear protein. Under stress conditions, such as cell injury, HMGB1 translocates from the nucleus to the cytoplasm and is subsequently released extracellularly.
Autophagy can regulate the release of HMGB1 in the affected cells.9,29 Under certain conditions, it is reported that HMGB1-containing LC3-positive cytoplasmic vesicles (likely autophagosomes) induced HMGB1 aggregation and inhibit HMGB1 extracellular release. 16 In addition, HMGB1 is also known as an inducer of autophagy.9,29
To our knowledge, there are no studies describing the mutual interaction among macrophages, autophagy, and DAMPs in rat livers under the influence of LPS. Importantly, in mammals, the liver is exposed continuously to gut-derived LPS via the portal vein; 6 therefore, exposure to a small amount of LPS may affect liver homeostasis. In order to clarify the interaction among macrophages, autophagy, and DAMPs in the biphasic functions (hepatocellular injury versus protection) of LPS, which may lead to liver homeostasis or diseases, the data were compared between rats treated with low-dose LPS (experiment 1) and those treated with high-dose LPS (experiment 2).
Materials and Methods
Experiments
Experiment 1: Low-dose LPS study
Sixteen, 5-week-old, male F344/DuCrlCrlj rats (85–105 g body weight) were used.
Twelve rats were injected intraperitoneally with an LPS dose of 0.1 mg/kg (Escherichia coli 055: B5; Sigma-Aldrich, St. Louis, MO USA), while the remaining 4 control rats were injected with an equal amount of sterile saline. Four rats were examined at 6, 10, and 24 hours after injection. Control rats were sacrificed at 6 hours after injection.
Experiment 2: High-dose LPS study
Twelve, 5-week-old, male F344/DuCrlCrlj rats (70–90 g body weight) were used. Eight rats were injected intraperitoneally with an LPS dose of 2.0 mg/kg, and the remaining 4 control rats were injected with sterile saline. Four rats were examined at 6 and 10 hours after injection, whereas control rats were sacrificed at 6 hours after injection.
Animals
Rats used in the aforementioned experiments 1 and 2 were obtained from Jackson Laboratory Japan (Kanagawa, Japan). They were maintained in a room at 21 ± 3°C with a 12-hour light-dark cycle, fed a standard diet for rats (DC-8; CLEA Japan, Tokyo, Japan), and supplied with tap water ad libitum. After 1-week acclimatization, they were used in experiments 1 and 2. The low and high doses of LPS were determined according to previous studies22,23 and preliminary studies done in our laboratory. At necropsy, rats were euthanized by exsanguination under deep isoflurane anesthesia, and the blood was collected from the abdominal aorta. The separated sera were subjected to a biochemical assay for aspartate transaminase (AST), alanine transaminase (ALT), alkaline phosphatase (ALP), and total bilirubin (T-Bil) by SRL Inc (Tokyo, Japan). The animal experiments were conducted under the institutional guidelines approved by the ethical committee of our university for the care and use of experimental animals.
Histopathology and Immunohistochemistry
Liver tissues from the left lateral lobe were collected and fixed in 10% neutral-buffered formalin and a periodate-lysine-paraformaldehyde solution processed by the PLP-AMeX (acetone, methyl benzoate, and xylene) method. 28 Formalin-fixed tissues were dehydrated, embedded in paraffin, and sectioned at 3–4 µm in thickness. The deparaffinized sections were stained with hematoxylin and eosin for histopathological examination.
Tissue sections with PLP-AMeX were deparaffinized and used for immunohistochemical analyses with primary antibodies such as CD163 (hemoglobin scavenger receptor), CD204 (macrophage scavenger receptor), CD68 (macrophage-specific member of the lysosomal-associated membrane protein family), MHC class II (major histocompatibility complex class II), LC3B (autophagosome marker), and HMGB1 (Table 1). After pretreatment, tissue sections were stained by the Histostainer (Nichirei Biosciences, Tokyo, Japan). Sections were incubated with 5% skimmed milk for 15 minutes, followed by 1-hour incubation with primary antibodies. After treatment with 3% H2O2 for 15 minutes, a horseradish peroxidase–conjugated secondary antibody (Histofine Simple Stain MAX PO; Nichirei Biosciences) was applied for 1 hour. Then, they were incubated with 3,3′-diaminobenzidine (Nichirei Biosciences) for 10 minutes. Sections were counterstained with hematoxylin for 1 minute. The specificity of antibodies used in this study has been confirmed using negative controls in our laboratory;22,23,32 tissue sections were treated with mouse or rabbit nonimmunized immunoglobulin G (IgG) instead of the primary antibody.
The list of antibodies used for immunohistochemistry.
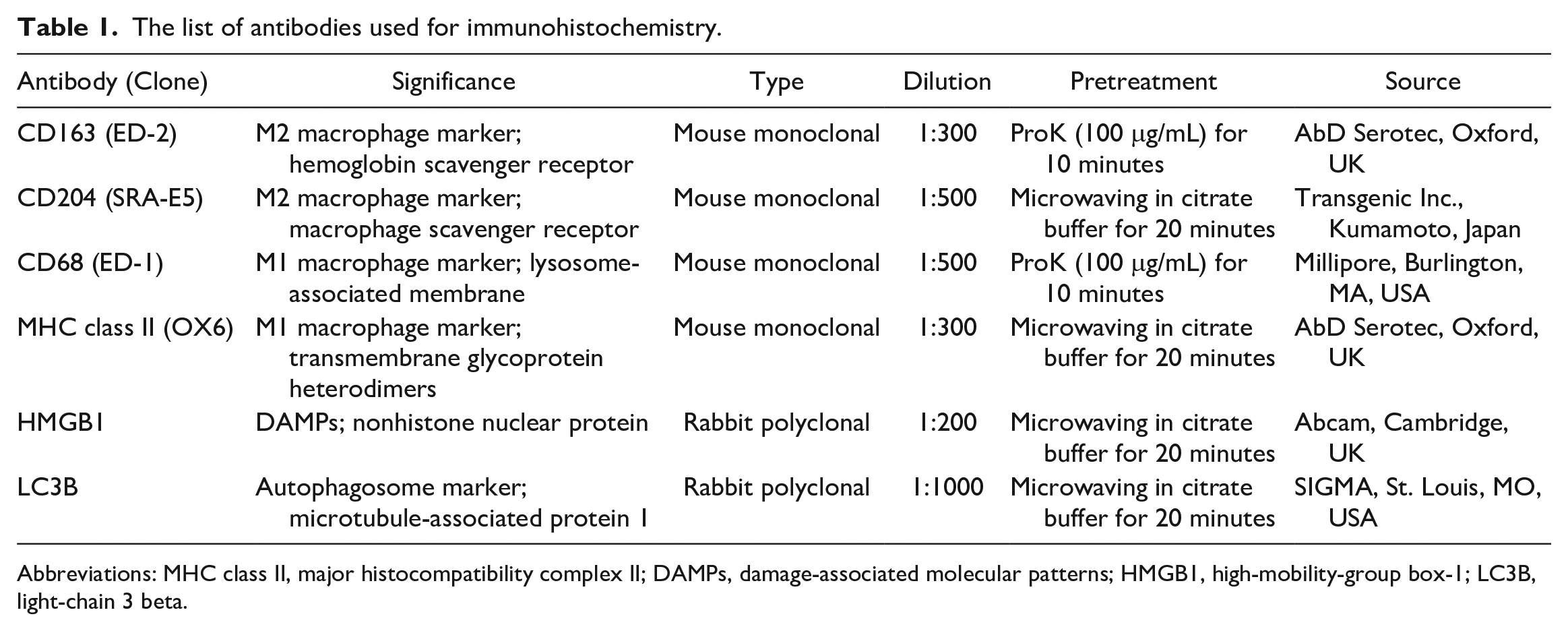
Abbreviations: MHC class II, major histocompatibility complex II; DAMPs, damage-associated molecular patterns; HMGB1, high-mobility-group box-1; LC3B, light-chain 3 beta.
Double Immunofluorescence
Fresh-frozen liver sections measuring 10 µm in thickness from rats injected with a low dose of LPS were used for double immunofluorescence of LC3B in combination with CD163 or CD68. After fixation in a mixed acetone:methanol (1:1) solution for 10 minutes at 4°C, the sections were incubated with 10% normal goat serum for 30 minutes followed by reaction with the primary antibody overnight at 4°C. After rinsing with phosphate-buffered saline, the sections were incubated for 45 minutes with the secondary antibody: goat antirabbit IgG conjugated with Alexa 568 (Invitrogen, Thermo Fisher Scientific, Waltham, MA USA) for LC3B and goat antimouse IgG conjugated with Alexa 488 (Invitrogen) for CD163 or CD68. The sections were coverslipped with Fluoro-KEEPER Antifade Reagent, Non-Hardening Type with DAPI (Nacalai Tesque, Kyoto, Japan), and analyzed by using a virtual slide scanner (VS-120; Olympus, Tokyo, Japan).
Cell Counts
The numbers of CD68-, CD163-, CD204-, and MHC class II–positive cells in the centrilobular-to-midzonal area were counted using WinRoof software (Mitani Corp. Tokyo, Japan) and were expressed as the number of positive cells per unit area (cells/mm2). The cell size of macrophages reacting to each antibody was measured in randomly selected 20 positive cells using the WinRoof software (Mitani Corp.). The number of single- or double-labeled cells with LC3B/CD68 and LC3B/CD163 immunofluorescence was counted with OlyVIA software (Olympus). The number of cytoplasmic LC3B-positive granules per hepatocyte was counted in 20 hepatocytes in randomly selected 4 high-power fields at perivenular areas of the liver.
Real-Time Reverse Transcriptase-Polymerase Chain Reaction
Liver samples from the left medial lobe were immediately immersed in RNAlater (Qiagen GmbH, hilden, Germany) overnight at 4°C and stored at −80°C until use. Total RNA was extracted from liver tissues by using an SV total RNA isolation system (Promega, Madison, WI USA) according to the manufacturer’s instructions. The extracted total RNA was reverse-transcribed with Super script VILO cDNA Synthesis Kit (Invitrogen). Real-time PCR was performed using TaqMan gene expression assays (Life Technologies, Thermo Fisher Scientific, Waltham, MA USA) in a PikoReal Real-Time 96 PCR System (Thermo Scientific, Waltham, MA USA). The TaqMan probes used in this study target Tnfα (tumor necrosis factor-α; proinflammatory factor), Mcp1 (monocyte chemoattractant protein-1; proinflammatory factor), Il1β (interleukin-1β; proinflammatory factor), Tgfβ1 (transforming growth factor-β1; anti-inflammatory factor), Tlr2, and Tlr4 (both, pattern recognition receptor) (Table 2). The level of messenger RNA (mRNA) expression was normalized against that of 18s rRNA as a reference gene. The data were analyzed using the comparative Ct method (ΔΔCtmethod).
The list of TaqMan probes used for real-time RT-PCR.
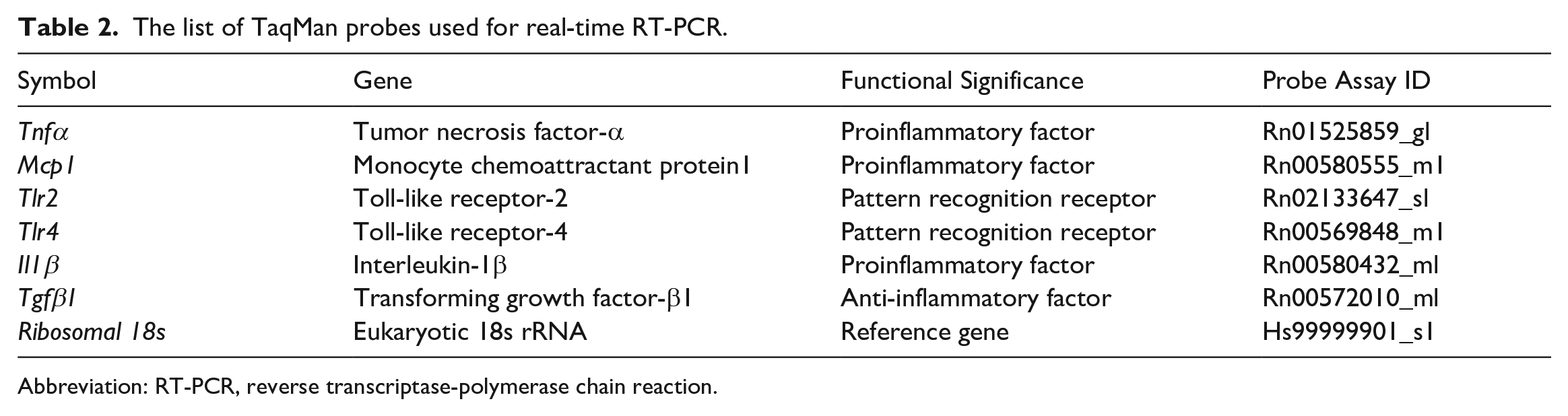
Abbreviation: RT-PCR, reverse transcriptase-polymerase chain reaction.
Western Blot
Liver samples from the left medial lobe were homogenized in a RIPA buffer (20 mM Tris-HCl pH 7.5, 150 mM NaCl, 1 mM ethylenediaminetetraacetic acid, 1 mM ethylene glycol tetraacetic acid, 1% Nonidet-P40, 0.1% deoxycholate, 0.1% sodium dodecyl sulfate, 1 mM NaF, 0.1 mM Na3VO4, 1 mM phenylmethylsulfonyl fluoride, and proteinase inhibitor cocktail; Nacalai Tesque). After centrifugation at 13,000 × g for 10 minutes, the supernatant was mixed with an equal volume of 2× SDS sample buffer (125 mM Tris-HCl, pH 6.8, 4% SDS, 30% glycerol, and 10% 2-mercaptoethanol) and then boiled at 95°C for 5 minutes. The protein concentration was determined by an absorption spectrometer using Bio-Rad Protein Assay (Bio-Rad Laboratories, Hercules, CA USA). Samples were separated on 5%–20% gradient polyacrylamide gels (ATTO, Tokyo, Japan) and transferred to polyvinylidene difluoride membranes (Bio-Rad Laboratories). Membranes were incubated overnight at 4°C with primary antibodies LC3B, p62 (autophagy degradation index), and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) (internal control) (Table 3), followed by incubation with peroxidase-conjugated secondary antibodies (Histofine Simple Stain MAX PO; Nichirei Biosciences) for 30 minutes. Signals were visualized with ECL prime (GE Healthcare) and quantified with a luminescent image analyzer (LAS-4000; GE Healthcare, Chicago, Illinois USA).
The list of antibodies used for Western blot.

Abbreviations: LC3B, light-chain 3 beta; GAPDH, glyceraldehyde 3-phosphate dehydrogenas.
Statistical Evaluation
Data obtained were expressed as mean ± standard deviation. Statistical analysis was performed using Dunnett’s test. Significance was considered at P < .05.
Results
Experiment 1: Low-Dose LPS Study
Gross, histopathological, and clinical pathological analyses
In the livers of control rats and LPS-injected rats, there were no significant changes in gross morphology or histological architectures in hematoxylin and eosin–stained sections. In addition, there were no significant differences in serum AST, ALT, T-Bil, or ALP (Supplemental Fig. S1) levels between control and LPS-injected rats. Collectively, rats injected with a low dose of LPS did not show any abnormalities not only in clinical pathology but also in pathological findings.
Analysis of hepatic macrophages
Macrophages reacting to CD163 (Fig. 1a, e), CD204 (Fig. 1b, f), CD68 (Fig. 1c, g), and MHC class II (Fig. 1d, h) were present in the livers of control rats and rats injected with a low dose of LPS. There were no significant differences in the number of macrophages reacting to CD163, CD204, CD68, or MHC class II between the liver of control and LPS-injected rats (Supplemental Fig. S2). However, the cell size of macrophages reacting to each antibody was different (Fig. 2). Macrophages reacting to CD163 at 6 and 10 hours and CD204 at 10 and 24 hours in LPS-injected rats had increased cytoplasm compared with those in control rats. In contrast, CD68- and MHC class II–positive macrophages did not reveal any morphological change in rats injected with a low dose of LPS. Collectively, although there were no changes in the numbers of macrophages reacting to CD68, CD163, CD204, or MHC class II, macrophages reacting to CD163 and CD204 were larger in cell size.
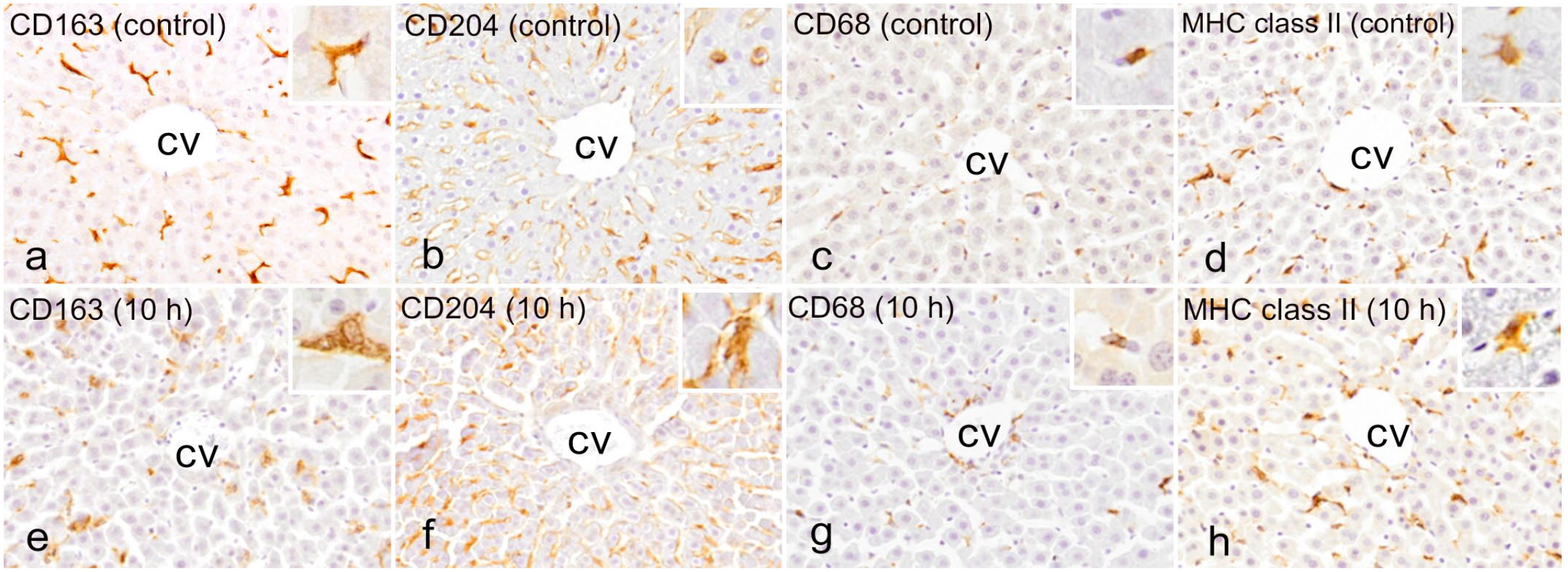
Liver, control rats (a–d) and rats injected with a low dose of LPS at 10 hours (e–h). Immunohistochemistry for macrophage markers. (a) Immunohistochemistry for CD163 in a control rat. CD163-positive cells (mainly Kupffer cells) are seen along the sinusoid in the centrilobular-to-midzonal area of the liver. (b) CD204-positive cells are diffusely distributed in centrilobular-to-midzonal areas of the liver in a control rat. (c) CD68-positive cells are sporadically present in centrilobular-to-midzonal areas of the liver in a control rat. (d) MHC class II–positive cells are occasionally present in centrilobular-to-midzonal areas of the liver in a control rat. (e) CD163-positive cells and (f) CD204-positive cells in rats injected with a low dose of LPS are hypertrophic in contrast to controls; however, there are no significant changes in shape of (g) CD68-positive cells and (h) MHC class II–positive cells. (insets: higher magnification of positive cells). Abbreviations: LPS, lipopolysaccharide; CV, central vein; MHC class II, major histocompatibility complex II.
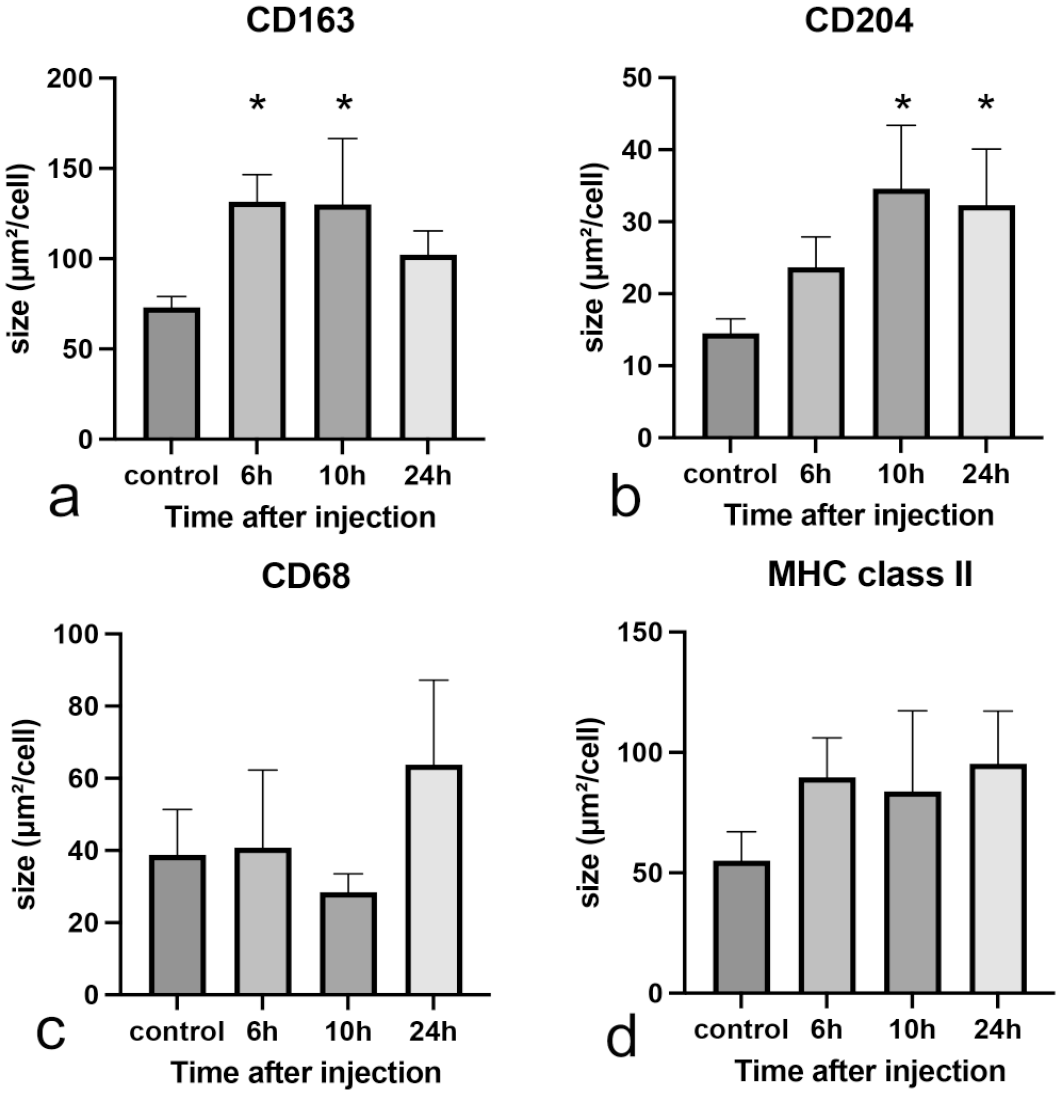
The size of cells positive for macrophage markers in rats at different times after the injection of a low dose of LPS. The cell sizes of (a) CD163-positive cells, (b) C204-positive cells, (c) CD68-positive cells, and (d) MHC class II–positive cells. Abbreviations: LPS, lipopolysaccharide; MHC class II, major histocompatibility complex II. Dunnett’s test; *P < .05, significantly different from control rats.
mRNAs Expression of Tnfα at 6 hours (P = .0205, Supplemental Fig. S3a), Il1β at 6 hours (P = .0147, Supplemental Fig. S3b), and Tgfβ1 at 6 (P = .0079) and 24 hours (P = .0202, Supplemental Fig. S3d) significantly increased in LPS-injected rats. Mcp1 did not show significant change (Supplemental Fig. S3c).
Analysis of autophagy
Autophagy in CD68- and CD163-positive hepatic macrophages (Supplemental Fig. S4) was evaluated using double immunofluorescence with an autophagosome marker LC3B. In LPS-injected rats at 6, 10, and 24 hours, there were no significant changes in the proportion of LC3B/CD68 or LC3B/CD163-double-expressing macrophages, compared with that of controls. These findings indicated that the formation of autophagosomes might not be changed in hepatic macrophages (reacting to CD68 and CD163) in rats injected with a low dose of LPS.
To evaluate the autophagy change in hepatocytes, immunohistochemistry was performed for LC3B in the liver of control rats and rats injected with a low dose of LPS (Fig. 3). In control rats, LC3B-positive cytoplasmic granules in hepatocytes were detected as the basal level (Fig. 3a); the granules were small in number and fine. LC3B-positive cytoplasmic granules per hepatocyte markedly increased in LPS-injected rats at 6, 10 (Fig. 3b), and 24 hours (Supplemental Fig. S5); the cytoplasmic granules were more clearly visible than those in the control.
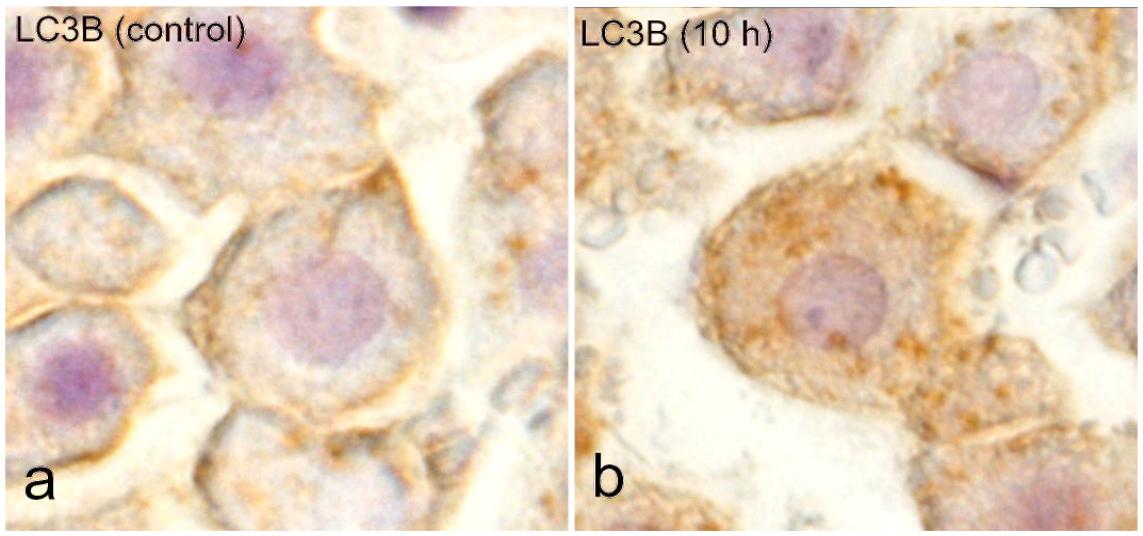
Liver, control rats and rats injected with a low dose of LPS at 10 hours. Immunohistochemistry for LC3B. (a) Fine LC3B-positive cytoplasmic granules in hepatocytes are sporadically detected at a basal level in control liver. (b) Increased numbers of LC3B-positive cytoplasmic fine granules in hepatocytes of the LPS-injected rat at 10 hours in comparison to control rats. Abbreviations: LPS, lipopolysaccharide; LC3B, light-chain 3 beta.
Tlr2 mRNA expression significantly increased in LPS-injected rats at 6 hours (P = .0068) (Fig. 4a); on the other hand, mRNA expression of Tlr4 did not change in LPS-injected rats (Fig. 4b). Western blot analysis revealed the increased expression in autophagosome markers (LC3B; LC3B-I at 24 hours [P = .0128] or LC3B-II at 10 [P = .0465] and 24 hours [P = .0485]) in LPS-injected rats (Fig. 4c, d). The expression of p62 (Fig. 4e), a protein degraded by the autophagosome, significantly decreased (P = .0485) in LPS-injected rats at 24 hours.
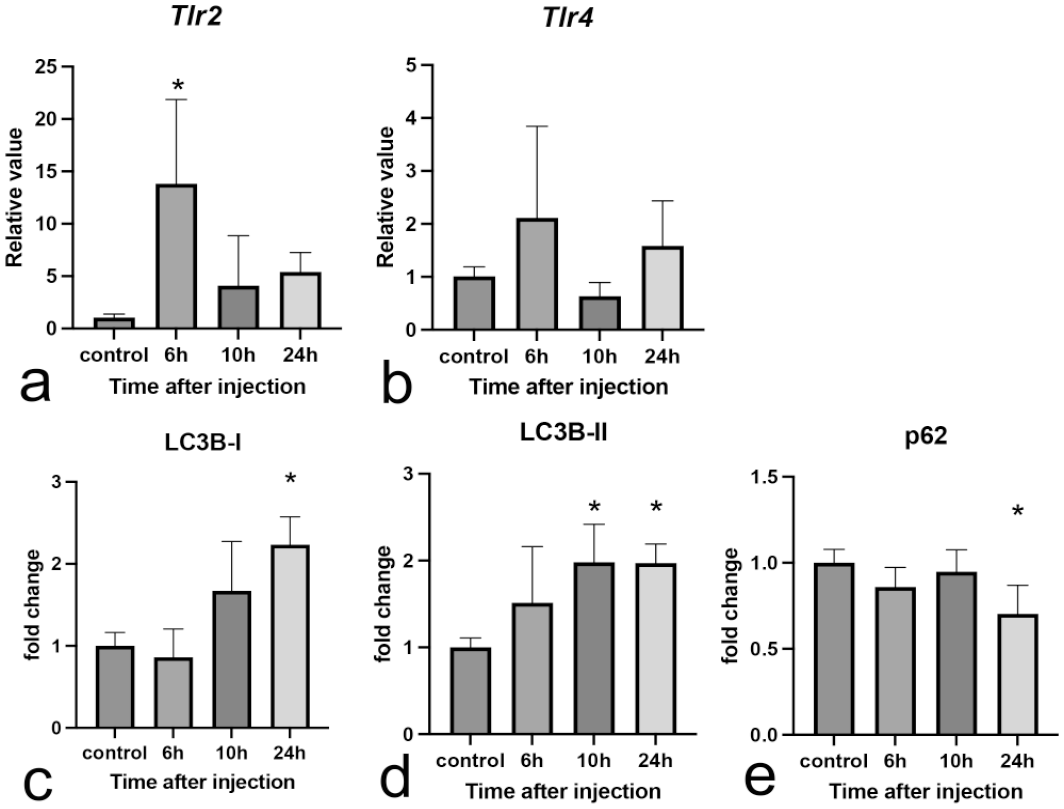
Expression of autophagy-related genes and proteins. (a, b) mRNA Expressions of autophagy-related receptors in livers of control rats and rats injected with a low dose of LPS. (a) mRNA Expression of Tlr2. (b) mRNA Expression of Tlr4. Expression levels are normalized to the 18s rRNA level. Dunnett’s test; *P < .05, significantly different from control rats. (c-e) Western blot analysis of livers of control rats and rats injected with a low dose of LPS. Fold change of (c) LC3B-I, (d) LC3B-II, and (e) p62. Intensity quantification using Multi Gauge software, Version 1.0, Fujifilm (Tokyo, Japan); GAPDH is used as loading control. Abbreviations: mRNA, messenger ribonucleic acid; LPS, lipopolysaccharide; LC3B, light-chain 3 beta; GAPDH, glyceraldehyde 3-phosphate dehydrogenase. Dunnett’s test; *P < .05, significantly different from control rats.
Analysis of DAMPs
Nuclei of hepatocytes in control rats were exclusively positive for HMGB1 (Fig. 5a). In LPS-injected rats at 6 and 24 hours, nuclear positivity was at the same level as seen in controls (Fig. 5b, d). On the other hand, there were some hepatocytes with cytoplasmic HMGB1 and loss of nuclear HMGB1 in LPS-injected rats at 10 hours (Fig. 5c, arrows). These findings indicated that the translocation of HMGB1 into the cytoplasm occurred temporarily at 10 hours after LPS injection.
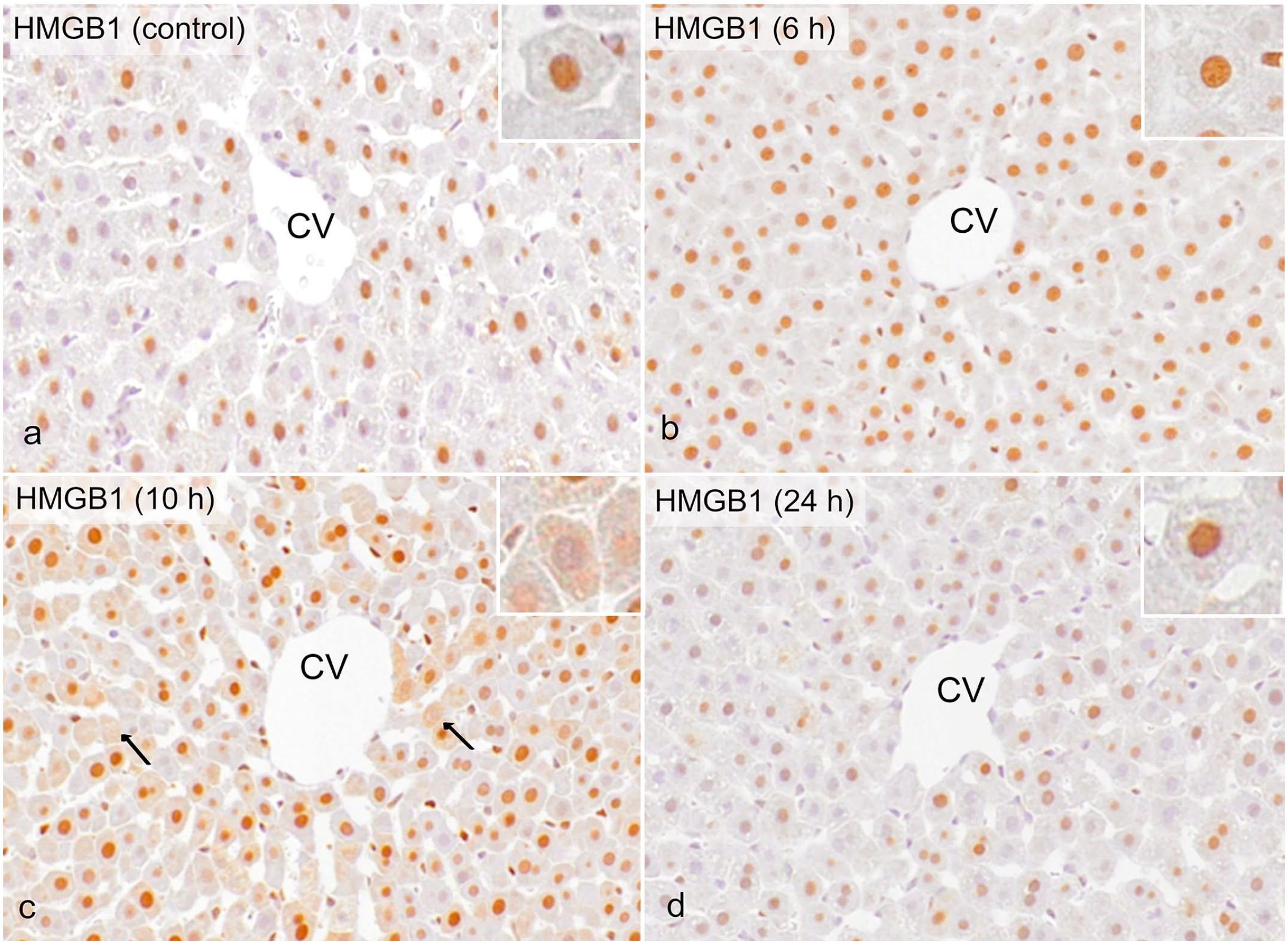
Liver, control rats and rats injected with a low dose of LPS. Immunohistochemistry for HMGB1. (a) In control rats, hepatocellular nuclei are positive, and cytoplasm are negative for HMGB1. (b) At 6 hours after LPS challenge, nuclear and cytoplasmic positivity is at the same level as in controls. (c) Hepatocytes with cytoplasmic HMGB1 and loss of nuclear HMGB1 (arrows) are scattered in the centrilobular area at 10 hours. (d) At 24 hours after LPS challenge, nuclear and cytoplasmic HMGB1 expression become the same level as in controls. (insets: higher magnification of hepatocytes). Abbreviations: LPS, lipopolysaccharide; CV, central vein; HMGB1, high-mobility-group box-1.
Experiment 2: High-Dose LPS Study
Gross, histopathological, and clinical pathological analyses
Grossly, no lesions were seen in the livers of control rats and LPS-injected rats at 6 hours. In contrast, whitish spots were diffusely distributed throughout the left lateral lobe of the liver in LPS-injected rats at 10 hours (Fig. 6a). Histologically, although there were no lesions in the livers of control rats and LPS-injected rats at 6 hours, focal coagulative hepatocellular necrosis was seen occasionally in the midzonal areas of the liver in LPS-injected rats at 10 hours (Fig. 6b). Histologically, there were no significant changes in other tissues of rats injected with low- and high-dose LPS in hematoxylin and eosin-stained sections. Serum AST, ALT, and T-Bil levels significantly increased in LPS-injected rats at 10 hours (Supplemental Fig. S6).
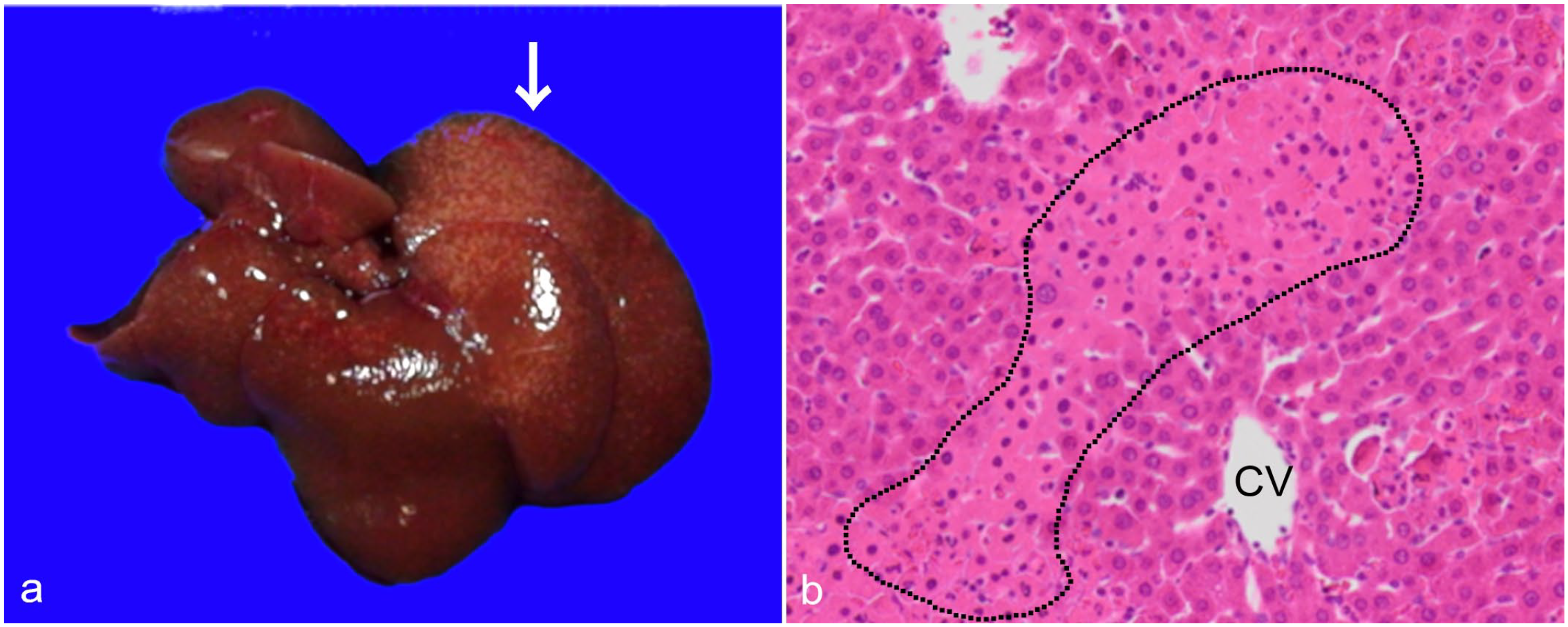
Liver injury induced by a high dose (2.0 mg/kg body weight) of LPS, liver, rat. (a) Gross appearance of the liver. There are whitish spots mainly in left lateral lobe of the liver (arrow) of the LPS-injected rat at 10 hours. (b) Histopathology of the liver. At 10 hours, focal hepatocellular coagulation necrosis (encircled by dotted line) is seen in the midzonal area. Abbreviations: LPS, lipopolysaccharide; CV, central vein.
Analysis of hepatic macrophages
The distribution of CD163- and CD204-positive macrophages was similar to that in rats injected with a low dose of LPS, whereas CD68- (Fig. 7c) and MHC class II–positive macrophages (Fig. 7d) in LPS-injected rats at 10 hours were seen sporadically with increased number compared with control rats (Fig. 7a, b), as mentioned below. The numbers and cell size of macrophages expressing CD163, CD204, CD68, and MHC class II are shown in Fig. 8 and Supplemental Fig. S7, respectively. Although CD163- and CD204-positive macrophages did not show any significant changes in the numbers (Fig. 8a, b), their cytoplasm became larger in LPS-injected rats at 10 hours (Supplemental Figs. S7a, b). Similar to low-dose LPS-treated rats, Kupffer cells reacting to CD163 and CD204 might become activated by LPS stimulus. In contrast, the numbers of CD68- and MHC class II–positive macrophages significantly increased (Fig. 8c, d) without showing morphological changes, in LPS-injected rats (Supplemental Figs. S7c, d).
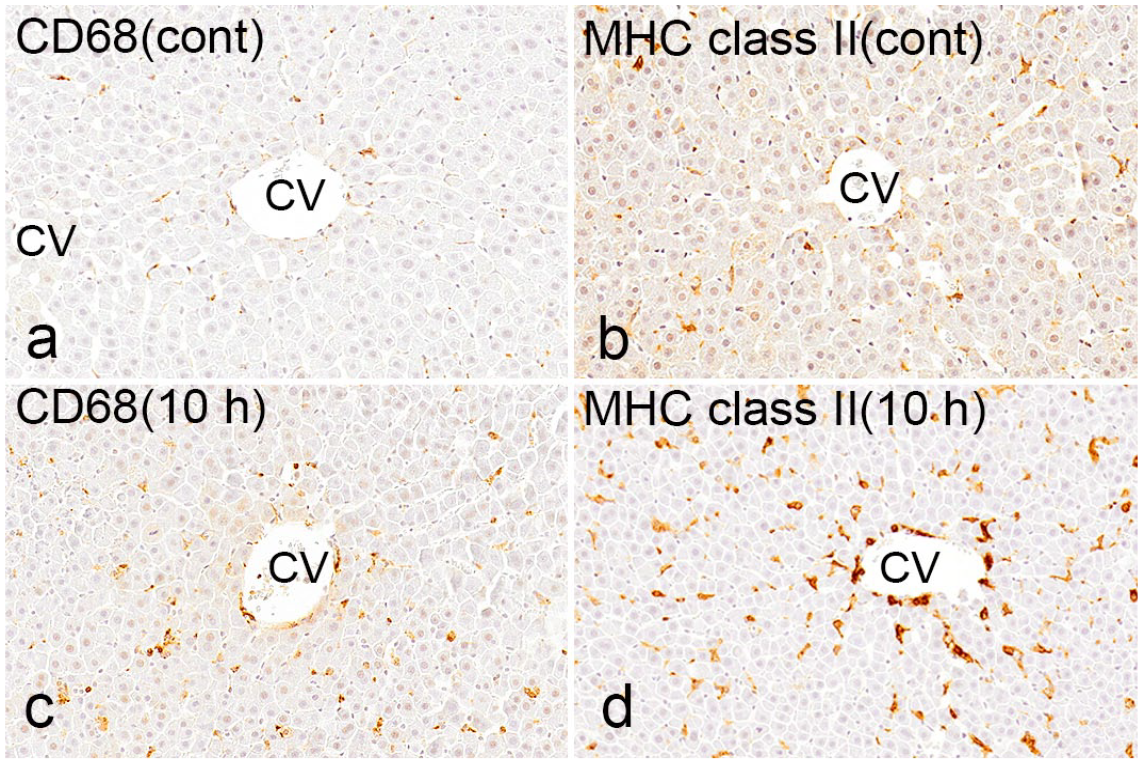
Liver, control rats (a, b) and rats injected with a high dose of LPS (c, d) at 10 hours. Immunohistochemistry for CD68 and MHC class II. Immunohistochemistry for both (a) CD68 and (b) MHC class II in a control rat. (c) Immunohistochemistry for CD68 in an LPS-injected rat at 10 hours. The positive cells increased compared with those in controls and are seen diffusely around the central vein. (d) Immunohistochemistry for MHC class II in an LPS-injected rat at 10 hours. MHC class II–positive macrophages are increased in number. Abbreviations: LPS, lipopolysaccharide; MHC class II, major histocompatibility complex II; CV, central vein.
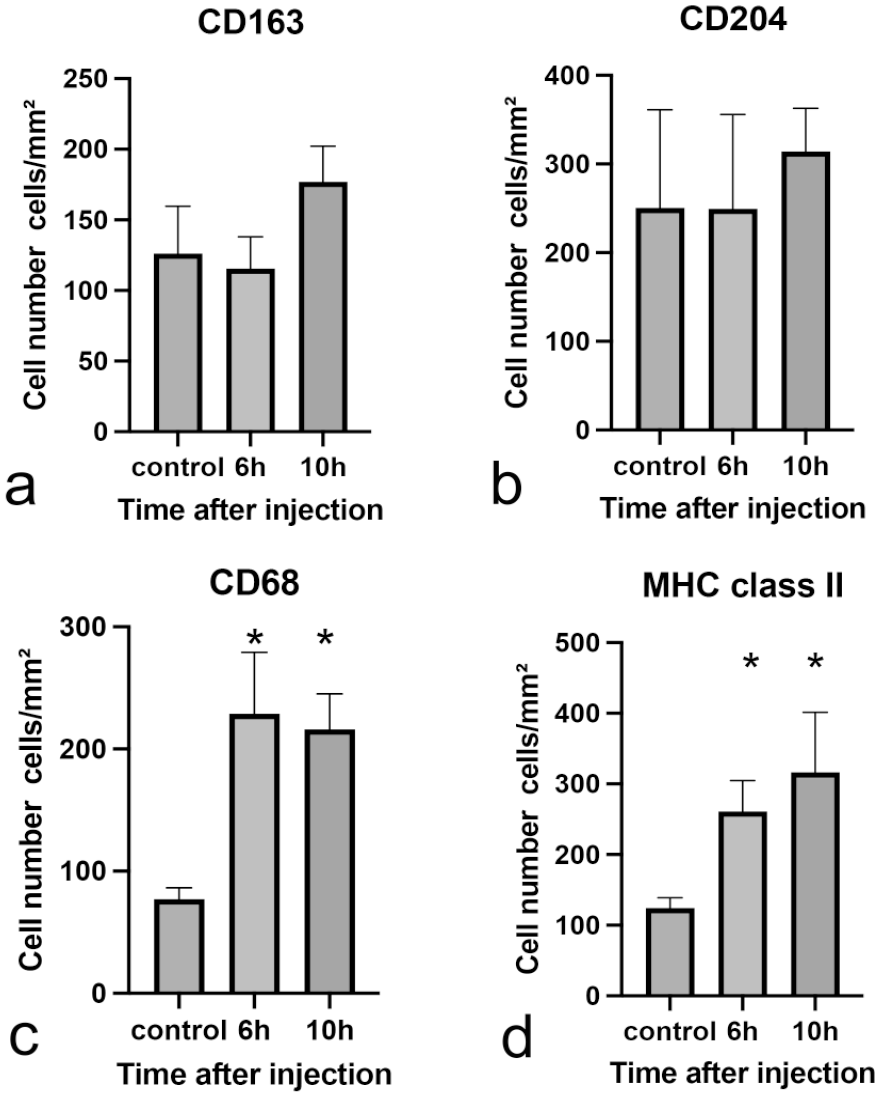
The number of cells positive for macrophage markers in rats at different times after the injection of high-dose LPS. The number of (a) CD163-positive cells, (b) CD204-positive cells, (c) CD68-positive cells, and (d) MHC class II–positive cells. Abbreviations: LPS, lipopolysaccharide; MHC class II, major histocompatibility complex II. Dunnett’s test; *P < .05, significantly different from control rats.
mRNA Expression of Tnfα (6 hours: P = .0003, 10 hours: P = .0011), Il1β (6 hours: P = .0056, 10 hours: P = .0005), and Mcp1 (6 hours: P = .014, 10 hours: P = .0004) significantly increased in LPS-injected rats at 6 and 10 hours (Fig. 9a–c). mRNA Expression of Tgfβ1 also significantly increased in LPS-injected rats at 10 hours (P = .0038, Fig. 9d).
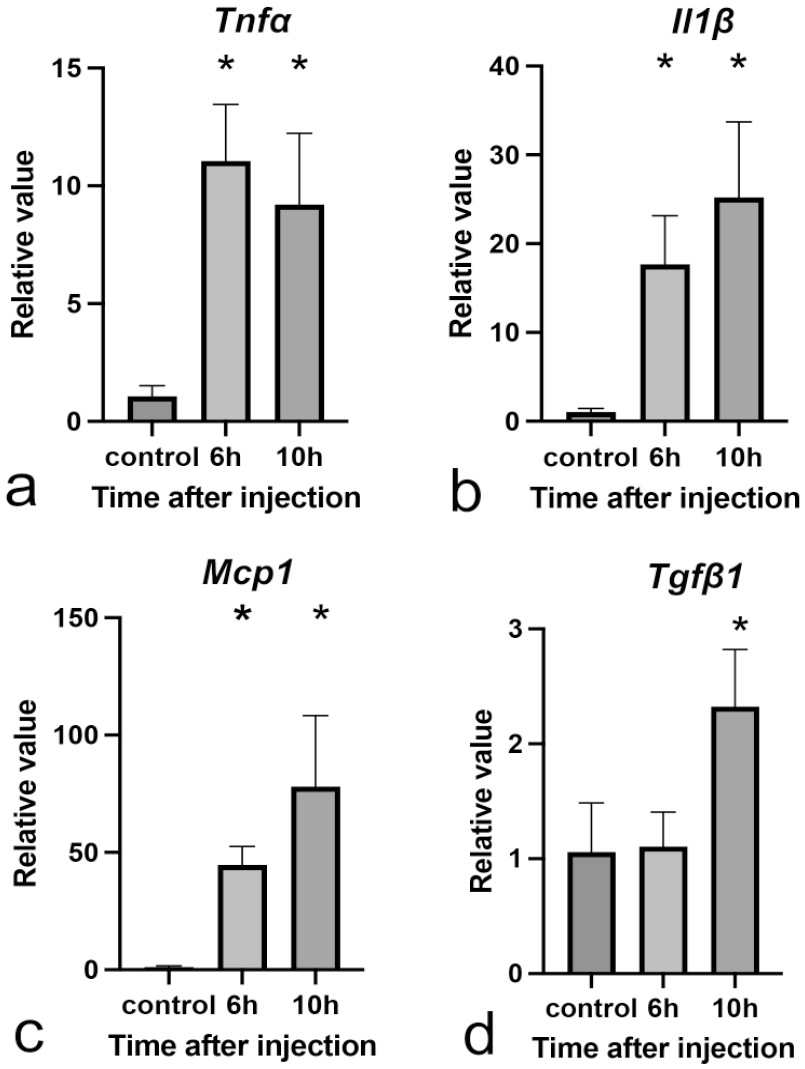
mRNA Expressions of macrophage-related cytokines in livers of control and LPS-injected rats. (a) Tnfα, (b) Il1β, (c) Mcp1, and (d) Tgfβ1. Expression levels are normalized to the 18s rRNA level. Abbreviations: LPS, lipopolysaccharide; mRNA, messenger ribonucleic acid; rRNA, ribosomal ribonucleic acid. Dunnett’s test; *P < .05, significantly different from control rats.
Analysis of autophagy
LC3B-positive cytoplasmic granules per hepatocyte increased in rats injected with a high dose of LPS at 6 and 10 hours. Interestingly, hepatocytes with vacuolated or irregular structures reacting to LC3B in the cytoplasm were occasionally seen in LPS-injected rats at 10 hours (Fig. 10).
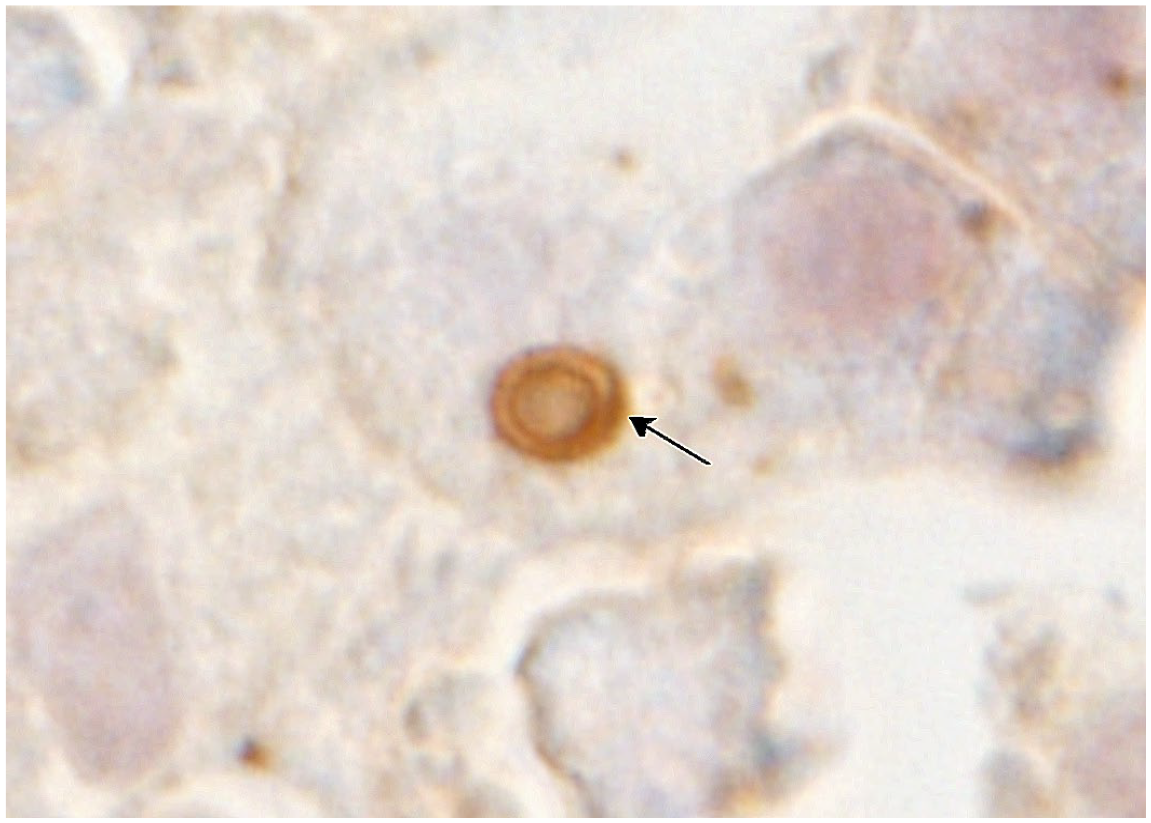
Liver, rats injected with a high dose of LPS at 10 hours. Immunohistochemistry for LC3B. A hepatocyte has a large, vacuolated structure expressing LC3B in the cytoplasm (arrow), indicating abnormality of autophagosome. Abbreviations: LPS, lipopolysaccharide; LC3B, light-chain 3 beta.
Tlr2 mRNA expression significantly increased in LPS-injected rats at 6 (P = .0011) and 10 hours (P = .0006, Fig. 11a); on the other hand, mRNA expression of Tlr4 significantly decreased in LPS-injected rats at 6 hours (P = .0264) and became similar to the level of controls at 10 hours (Fig. 11b).
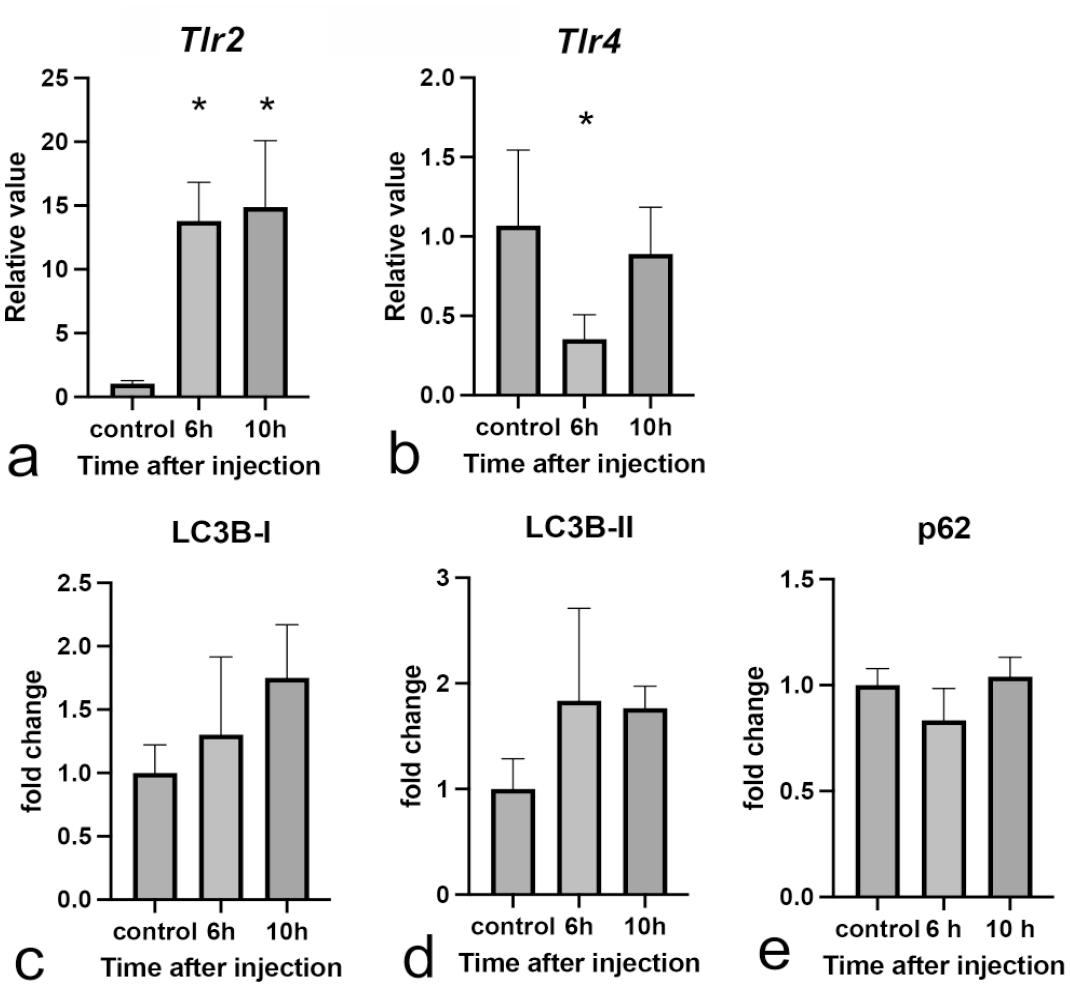
Expression of autophagy-related genes and proteins. (a, b) mRNA Expressions of autophagy-related receptors in livers of control rats and rats injected a high dose of LPS. mRNA Expressions of (a) Tlr2 and (b) Tlr4. These expression levels are normalized to the 18s rRNA level. Dunnett’s test; *P < .05, significantly different from control rats. (c–e) Western blot analysis of livers from control rats and rats injected with a high dose of LPS: (c) LC3B-I, (d) LC3B-II, (e) p62. The intensity of these bands is quantified using the Multi Gauge software, Version 1.0, Fujifilm; GAPDH is used as loading control. Abbreviations: LPS, lipopolysaccharide; mRNA, messenger ribonucleic acid; rRNA, ribosomal ribonucleic acid; LC3B, light-chain 3 beta; GAPDH, glyceraldehyde 3-phosphate dehydrogenase. Dunnett’s test; *P < .05, significantly different from control rats.
In rats injected with a high dose of LPS, the expression of LC3B-I showed a tendency to increase (Fig. 11c), but LC3B-II did not show significant changes (Fig. 11d). The expression of p62 did not show any significant changes, as well (Fig. 11e).
Analysis of DAMPs
At 6 hours in rats injected with a high dose of LPS, nuclear positivity was the same as seen in controls. On the other hand, in LPS-injected livers at 10 hours, there were many hepatocytes with cytoplasmic HMGB1-positive granules, particularly in and around necrotic hepatocytes; in addition, HMGB1-negative nuclei were seen around the necrotic areas (Fig. 12). These findings indicated that the translocation of nuclear HMGB1 into the cytoplasm already occurred at 10 hours after LPS injection. Interestingly, there were some hepatocytes that had HMGB1-negative cytoplasm and nuclei around necrotic foci, indicating that HMGB1 might have been released extracellularly.
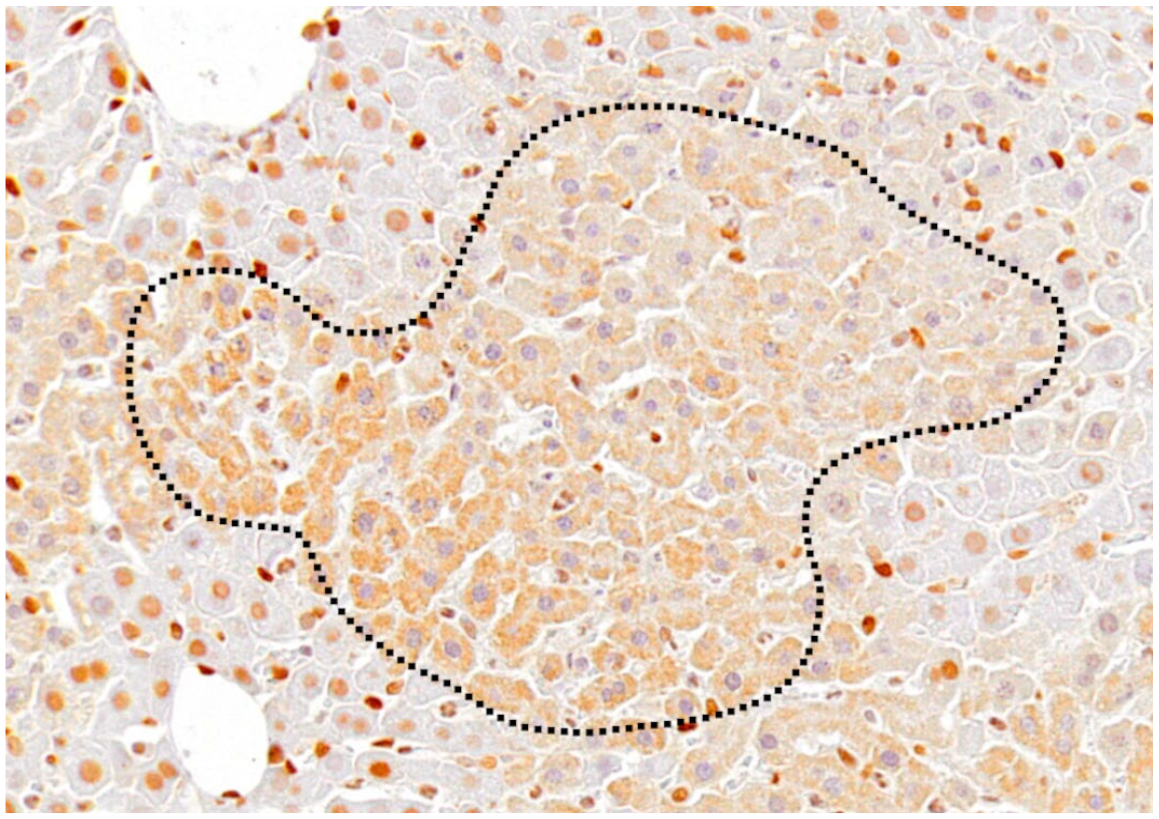
Liver, rats injected with a high dose of LPS at 10 hours. Immunohistochemistry for HMGB1. Coagulated hepatocytes that have HMGB1-positive cytoplasmic granules and negative nuclei are seen in and around a necrotic area in the liver; the area of coagulative necrosis of hepatocytes (encircled by dotted line) is the same as that seen in necrotic lesions encircled by dotted line in Fig. 6b in the hematoxylin and eosin–stained section. Abbreviations: LPS, lipopolysaccharide; HMGB1, high-mobility-group box-1.
Discussion
Low-dose LPS (0.1 mg/kg body weight) did not result in any abnormalities in the clinical findings or histological analysis, whereas high-dose LPS (2.0 mg/kg body weight) induced multifocal hepatocellular necrosis with increased liver enzymes. These results indicated that LPS may show dose-dependent liver injury.
Based on the concept of M1/M2 macrophage polarization, 36 increased M1 macrophages expressing CD68 and MHC class II seen in the high-dose group might be related to cell injury/necrosis in the LPS-injected rats. In fact, there were necrotic foci of hepatocytes in rats injected with a high dose of LPS at 10 hours. It was reported that M1 macrophages produce proinflammatory cytokines such as Tnfα, Il1β, and Mcp1.18,36 Increased mRNA expressions of Tnfα and Il1β were seen at 6 hours in the low-dose group and at 6 and 10 hours in those given high-dose LPS. It was considered that such persistent effects of proinflammatory cytokines could contribute to liver injury (such as coagulation necrosis) seen in the high-dose group. CD68 antigen expression is most commonly used for the detection of blood monocytes and exudate macrophages in pathological lesions.8,32,37 The MHC class II molecule has been reported to be expressed in dendritic cells and activated macrophages. 41 In this study, an increase in the number of macrophages expressing CD68 and MHC class II was only seen in rats given a high dose of LPS, indicating that these cells might be infiltrating cells in sinusoid from blood circulation.
On the other hand, low-dose LPS injection, which did not induce increased M1 macrophages, might stimulate the activity of CD163- and CD204-expressing M2 macrophages, which have beneficial properties against inflammatory disorders resulting in limitation of tissue injury, 34 although the effects of cytokines in low-dose LPS might be insufficient to induce cell injury.
It has been reported that CD163- and CD204-positive cells are regarded as Kupffer cells in normal rat livers.35,36 In the present study, macrophages reacting to CD163 and CD204 in both the low-dose and high-dose groups were hypertrophied compared with the control rats, indicating that LPS treatment activated Kupffer cells. Because the liver is continuously exposed to small amounts of gut-derived LPS, 6 Kupffer cells might be involved in liver homeostasis by constant performance of M2 functions.
Hepatocyte autophagy plays important roles in cytoprotection for liver injury by degrading damaged organelles. 2 The present study showed that treatment with low-dose LPS could stimulate the autophagy degradation cascade in hepatocytes, but not in macrophages. Autophagy in hepatocytes is activated via a TLR-dependent activation pathway;5,17,26 this is supported by increased mRNA expression of Tlr2 in both LPS-challenged groups. In contrast, Tlr4 expression decreased at 6 hours after injecting a high dose of LPS. Tlr4 mRNA expression decreases within a few hours of LPS treatment and returned to the original level; the finding of Tlr4 downregulation was previously reported in vitro. 26 However, TLRs activate not only autophagy in hepatocytes but also production of cytokines in macrophages and extracellular release of DAMPs.7,33 The significance of TLR expression in this study needs to be investigated further.
LC3B-positive cytoplasmic granules per hepatocyte increased in both the low- and high-dose groups. However, the expression of p62, a protein degraded by the autophagosome, 5 did not show any significant change in high-dose LPS rats, whereas p62 protein degraded and decreased in rats injected with a low dose of LPS. Furthermore, LC3B-II, the index of autophagosome formation, did not show significant changes. These results indicated that there might be abnormalities in the autophagosome-formation process. Interestingly, in hepatocytes of the high-dose group, LC3B-positive cytoplasmic granules were vacuolated or irregular in their morphology although those in the low-dose group were fine (apparently, normal structures). Autophagic proteolysis is considered to be inhibited in vacuolated autophagosomes. 14 In addition, it is reported that autophagy regulates HMGB1 release in some conditions.16,30 Abnormal autophagosomes seen in high-dose LPS rats in the present study also might have resulted in autophagy degradation disorder, leading to promotion of liver injury without processing DAMPs (in particular, HMGB1). In contrast, activated autophagy in hepatocytes in the low-dose group could be related to protection of hepatocytes against damage, which might be caused by LPS.
Hepatocytes that have cytoplasmic HMGB1-positive granules appeared more frequently in the high-dose group at 10 hours than those in the low-dose group. In addition, there were some hepatocytes that had HMGB1-negative cytoplasm and nuclei around and in necrotic foci in the high-dose group at 10 hours. These findings indicated that the translocation of nuclear HMGB1 into the cytoplasm occurred more frequently at 10 hours after high-dose LPS injection, and extracellular release of HMGB1 might have occurred.
It has been considered that LPS affects hepatocytes through proinflammatory cytokines produced by activated macrophages, mainly M1 macrophages.1,11 On the basis of the present results, a low dose of LPS induced the activation of CD163- and CD204-positive Kupffer cells, which may have high sensitivity to LPS. CD163- and CD204-positive cells may be functionally regarded as M2 macrophages, which can protect hepatocytes against tissue injury by producing anti-inflammatory cytokines. 3 Considering these findings, although Kupffer cells may produce proinflammatory cytokines under some conditions, they might rather work as M2 macrophages following a low-dose-LPS challenge; this should be investigated further.
Proinflammatory cytokines, which increased in rats injected with a low dose of LPS, may stimulate the translocation of HMGB1 (one of DAMPs) into cytoplasm in hepatocytes. It has been reported that cytoplasmic HMGB1 is necessary to enhance autophagy, which, in turn, degrades HMGB1 under certain situations.17,29,40 Based on these facts and the present results, the activation of autophagy in hepatocytes might play an important role in the degradation of HMGB1. Collectively, the low dose of LPS used in the present study was noninjurious, which might be related to the cytoprotective function mainly by activated M2 macrophages reacting to CD163 and CD204 (mainly Kupffer cells in the present study) and autophagy. The significant reduction of injured hepatocyte areas in TAA-injected rat liver after pretreatment with a low dose of LPS, in contrast to TAA-alone-injection rats, might be due to the activated M2 macrophages with cytoprotection. 23 It is interesting to investigate possible cytoprotective roles of low-dose LPS using other hepatotoxicants.
In contrast, the high dose of LPS used in the present study might induce infiltration of CD68-positive and MHC class II–positive M1 macrophages, leading to persistent secretion of proinflammatory cytokines, resulting in greater stimulus to hepatocyte injury. 4 It is reported that inflammatory stimuli can induce HMGB1 nuclear-cytoplasmic translocation in hepatocytes. 42 Proinflammatory cytokine levels following a high-dose-LPS challenge could induce the translocation of HMGB1 and then damage cytoplasmic organelles in hepatocytes. These cytoplasmic proteins might not be degraded by abnormal autophagy, 14 resulting in hepatocyte injury. In addition, HMGB1, which was not degraded by autophagy, might be extracellularly released and exacerbate inflammation. 13
In conclusion, dose-dependent effects of LPS on liver homeostasis or diseases were investigated in the present study. In addition, a low dose (0.1 mg/kg body weight) of LPS works as cytoprotection to hepatocytes, which may be performed by a favorable mutual relationship among hepatic macrophages, autophagy, and DAMPs; presumably, the mutual relationship may be related to homeostasis. On the other hand, a high dose (2.0 mg/kg body weight) of LPS induced hepatocyte injury (necrosis), which might have been due to failures in the mutual relationship among hepatic macrophages, autophagy, and DAMPs; possibly, such failures may be associated with hepatic diseases. These findings suggest that it is necessary to consider the effects of LPS on hepatocytes, which may be supplied from gut microbiota, when we evaluate the mechanisms of hepatic lesions.
Footnotes
Acknowledgements
The technical assistance of the members of our laboratory is gratefully acknowledged as well.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported partly by JSPS KAKENHI Grant Number 19H03130 (to J.Y.) and by the Platform Project for Supporting Drug Discovery and Life Science Research (Basis for Supporting Innovative Drug Discovery and Life Science Research [BINDS]) from AMED under the grant number JP20am0101123.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
