Abstract
Type D enterotoxemia, caused by Clostridium perfringens epsilon toxin (ETX), is one of the most economically important clostridial diseases of sheep. Acute type D enterotoxemia is characterized by well-documented lesions in the nervous, cardiocirculatory, and pulmonary systems. However, discrepancies and confusion exist as to whether renal lesions are part of the spectrum of lesions of this condition, which is controversial considering that for many decades it has been colloquially referred to as “pulpy kidney disease.” Here, the authors assess renal changes in an experimental model of acute type D enterotoxemia in sheep and evaluate the possible role of ETX in their genesis. Four groups of 6 sheep each were intraduodenally inoculated with either a wild-type virulent C. perfringens type D strain, an etx knockout mutant unable to produce ETX, the etx mutant strain complemented with the wild-type etx gene that regains the ETX toxin production, or sterile culture medium (control group). All sheep were autopsied less than 24 hours after inoculation; none of them developed gross lesions in the kidneys. Ten predefined histologic renal changes were scored in each sheep. The proportion of sheep with microscopic changes and their severity scores did not differ significantly between groups. Mild intratubular medullary hemorrhage was observed in only 2 of the 12 sheep inoculated with the wild-type or etx-complemented bacterial strains, but not in the 12 sheep of the other 2 groups. The authors conclude that no specific gross or histologic renal lesions are observed in sheep with experimental acute type D enterotoxemia.
Type D enterotoxemia caused by Clostridium perfringens type D ETX is the most prevalent and economically important clostridial disease of sheep. By fulfilling molecular Koch’s postulates, 4 it has been demonstrated that ETX, which is encoded by the etx gene present in large conjugative plasmids, 12 is essential for the virulence of this pathogen in naturally susceptible hosts, including sheep and goats, and in rodent models. 7 These studies showed that when the etx gene of a wild-type virulent C. perfringens type D strain was knocked out, the resulting mutant strain was unable to produce ETX and, therefore, was avirulent. When the wild-type etx gene was reintroduced into the knockout strain, the resulting mutant regained the ability to produce ETX and with that its virulence. 7
After its production and release by C. perfringens in the intestinal lumen in the form of a prototoxin, ETX is activated by trypsin or other intestinal or bacterial proteases, which is followed by absorption and systemic distribution through the circulatory system.19,20 ETX binds to a specific receptor in endothelial cells and oligodendrocytes (and perhaps other cells), which results in toxin heptamerization with formation of a transmembrane pore leading to cytotoxicity. 14 Increased vascular permeability with plasma extravasation leading to edema is the immediate result of this damage to the vascular endothelium. Typical lesions of acute and subacute type D enterotoxemia in sheep include hydropericardium, hydrothorax, ascites, cerebral, pulmonary, and myocardial edema, and occasional necrotizing lesions in the brain and myocardium.5,6,8 –10,17 The necrotizing lesions in the brain and myocardium are likely the result of hypoperfusion secondary to increased vascular permeability, a direct action of ETX on neural and myocardial cells, or a combination of both. In some instances, the extent of endothelial cell damage determines the varying degrees of hemorrhage, which can occasionally be observed in several organs. 17
Other lesions have been inconsistently reported or poorly documented in sheep with type D enterotoxemia. Early recognition of soft kidneys in sheep with experimental enterotoxemia has led to the broadly used designation of “pulpy kidney disease,” 1 although this has long been recognized as a postmortem phenomenon. 18 It seems odd that this disease is named after a postmortem change, despite the lack of convincing evidence of specific gross, histologic, or ultrastructural lesions in the renal parenchyma of sheep detectable at the time of death.9,18 The aim of this study was to describe and score renal changes in sheep experimentally inoculated with a wild-type C. perfringens type D strain, 2 of its genetically modified derivatives, and sterile nontoxic culture medium, and to compare these changes between groups to assess the possible role of ETX-producing strains in their genesis.
Materials and Methods
Animal Experimental Inoculations
For this study, we used necropsy records, gross photographs, and formalin-fixed kidney samples of an experimental reproduction of acute type D enterotoxemia in sheep, carried out in 2011 at the School of Veterinary Medicine, University of California, Davis (Institutional Animal Care and Use Committee permit #16383). The experimental design, bacterial strains, clinical findings, and neurologic and cardiopulmonary lesions have been published elsewhere.7,8,10,12
Briefly, 24 sheep randomly assigned to 4 groups of 6 sheep were inoculated intraduodenally with 1 of 3 isogenic C. perfringens strains or with sterile nontoxic culture medium. Group 1 (sheep 1–6) was inoculated with C. perfringens type D wild-type strain CN1020. Group 2 (sheep 7–12) was inoculated with an isogenic C. perfringens strain (JIR4981) that is an etx knockout mutant and is thus unable to produce ETX. Group 3 (sheep 19–24) was inoculated with the isogenic C. perfringens strain that was complemented in trans with the etx gene (JIR12604), so that it regained the ETX toxigenic ability. Group 4 (sheep 13–18) was inoculated with sterile nontoxic culture medium (control group). Eight of the 12 (66.7%) sheep inoculated with ETX toxigenic strains (5/6 of group 1 [sheep 1–2 and 4–6] and 3/6 of group 3 [sheep 19–21]) either died of acute type D enterotoxemia or were euthanized due to severe clinical disease. None of the 12 sheep in groups 2 or 4, inoculated with the etx knockout strain and sterile nontoxic culture medium, respectively, developed significant clinical disease or died spontaneously. The end of the experiment was set at 24 hours postinoculation when all surviving sheep were euthanized. Results of the survival times, mortality, and overall clinical and pathologic findings, 7 as well as detailed descriptions of the neurologic, 8 and cardiopulmonary lesions 10 in these same sheep have been published elsewhere.
Gross Examination
All 24 sheep were subjected to a complete necropsy within 2 hours of death; the carcasses had no to minimal autolysis. Special attention was given to the gross examination of the kidneys, which were inspected for perirenal and parenchymal edema; capsular and superficial cortical hemorrhages or discoloration; consistency of the parenchyma; discoloration of the parenchyma of the cortex and medulla on cut section; and appearance of the renal papillae, calyxes, and pelvis. At necropsy, samples from multiple organs, including the kidneys, were collected, fixed in 10% neutral buffered formalin, processed routinely, and stained with hematoxylin and eosin (HE) for histology. 7 The samples of kidney always included cortex, medulla, and pelvic epithelium.
Histopathologic Scoring
Histologic examination of all renal sections was performed in a blinded fashion by one of the authors of this study (F.G.). The kidney sections were screened for 10 predefined microscopic changes detailed in Table 1. Each histologic change was scored individually using a 4-tier scheme with scores from 0 to 3, in which score 0 indicated absence of the change, and scores 1–3 increasing degrees of severity and/or area affected by the changes. The criteria used for scoring microscopic changes that had scores ≥1 are shown in Supplemental Table S1.
Ten predefined histologic changes assessed in the sections of kidney of all sheep inoculated with Clostridium perfringens isogenic strains or sterile culture medium.

When inflammatory cell infiltrates were observed, the types of inflammatory cells (lymphocytes, histiocytes, plasma cells, neutrophils, and/or eosinophils) were identified morphologically.
Statistical Analyses
Two-tailed Fisher’s exact test was used to assess differences in the proportions of sheep with or without microscopic changes in the kidneys. These proportions were compared between all 4 groups, as well as grouping the 12 sheep from groups 1 + 3 (exposed to ETX toxigenic strains) versus those from groups 2 + 4 (not exposed to ETX). The Kruskal-Wallis test was performed to assess differences in the histopathologic scores between all 4 groups, and between the 12 sheep of groups 1 + 3 and the 12 sheep of groups 2 + 4. Results were expressed as median (minimum-maximum) score. The significance level was set as α = 0.05. The analyses were conducted using R v3.6.2 (R Core Team) and RStudio v1.2.5033 (RStudio Team).
Results
Gross Lesions
None of the 24 sheep showed gross renal lesions. In particular, no edema, reduced consistency, hemorrhage, discoloration, or loss of gross architecture was observed in the kidneys of any of the animals of the 4 groups (Fig. 1).
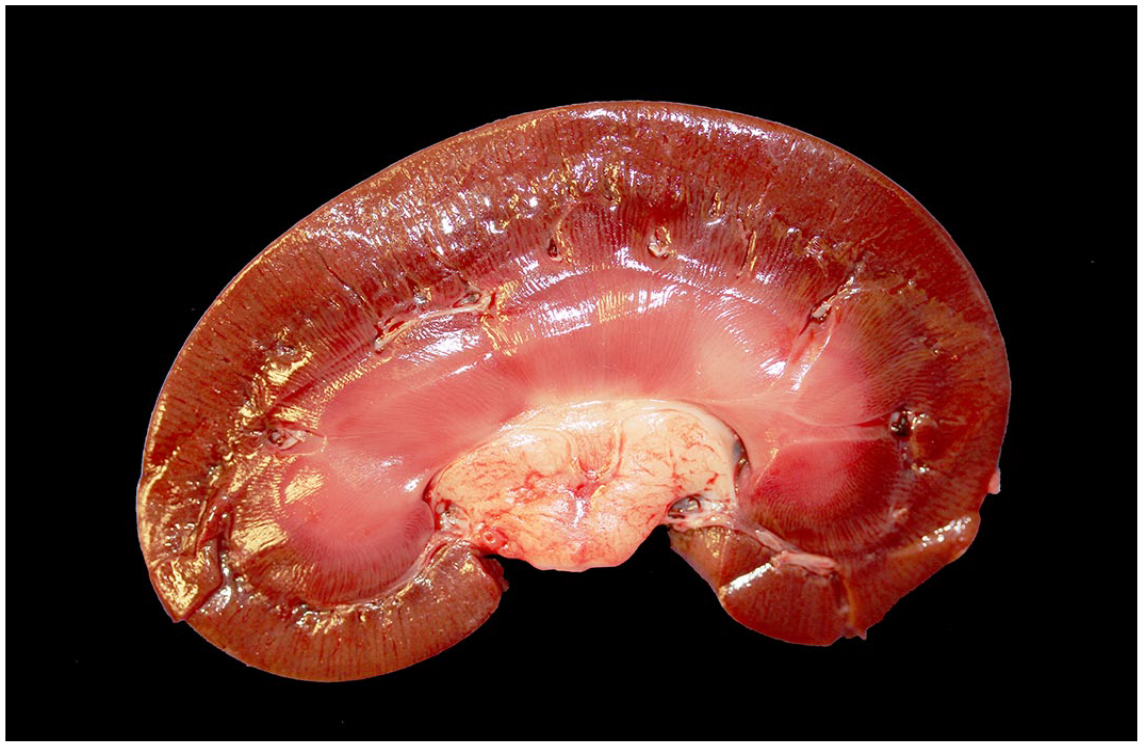
Clostridium perfringens type D enterotoxemia, kidney, sheep. No gross lesions are seen in a longitudinal section of the kidney of a sheep that was euthanized approximately 6 hours 45 minutes after inoculation with the etx-complemented strain of C. perfringens type D because it manifested severe acute enterotoxemia (sheep 19, group 3).
Histopathology
The proportions of sheep with microscopic changes by group are shown in Table 2. All 24 sheep (100%) had at least 1 of the assessed microscopic renal changes with scores ≤2; no scores of 3 were observed for any change in any of the animals. When the assessed microscopic changes were present, they generally affected sheep of all 4 groups in proportions that did not differ significantly between groups (Table 2). Similarly, the proportion of sheep with any of the changes in groups 1 + 3 versus groups 2 + 4 did not differ significantly (P > .371).
Number and percentage of sheep with microscopic renal changes in groups inoculated with Clostridium perfringens type D wild-type strain (group 1), etx knockout strain (group 2), etx-complemented strain (group 3), or sterile culture medium (group 4, control).
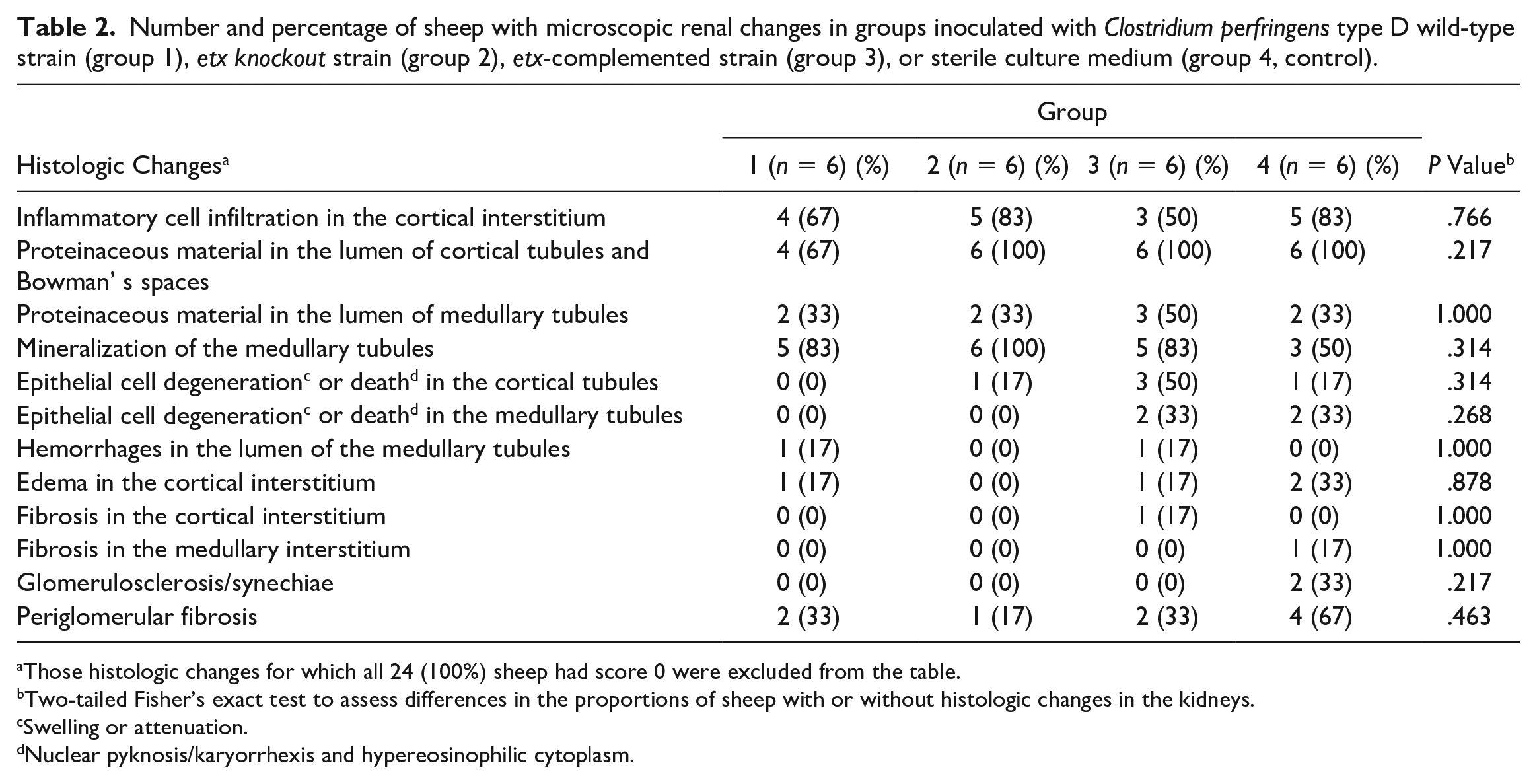
Those histologic changes for which all 24 (100%) sheep had score 0 were excluded from the table.
Two-tailed Fisher’s exact test to assess differences in the proportions of sheep with or without histologic changes in the kidneys.
Swelling or attenuation.
Nuclear pyknosis/karyorrhexis and hypereosinophilic cytoplasm.
The most frequent change was the presence of proteinaceous material, in the form of droplets or homogeneous material, in the lumen of cortical tubules and Bowman’s spaces (22 sheep from all groups) (Fig. 2), followed by focal or multifocal mineralization of the medullary tubules (19 sheep from all groups), inflammatory cell infiltration in the cortical interstitium (score 1 in 17 animals from all groups), the presence of proteinaceous material in the lumen of medullary tubules (score 1 in 9 sheep from all groups), and periglomerular fibrosis (score 1 in 9 sheep from all groups). The maximum number of changes in a single sheep was 6 and was observed in 3 sheep (1 of group 1, 1 of group 3, and 1 of group 4).
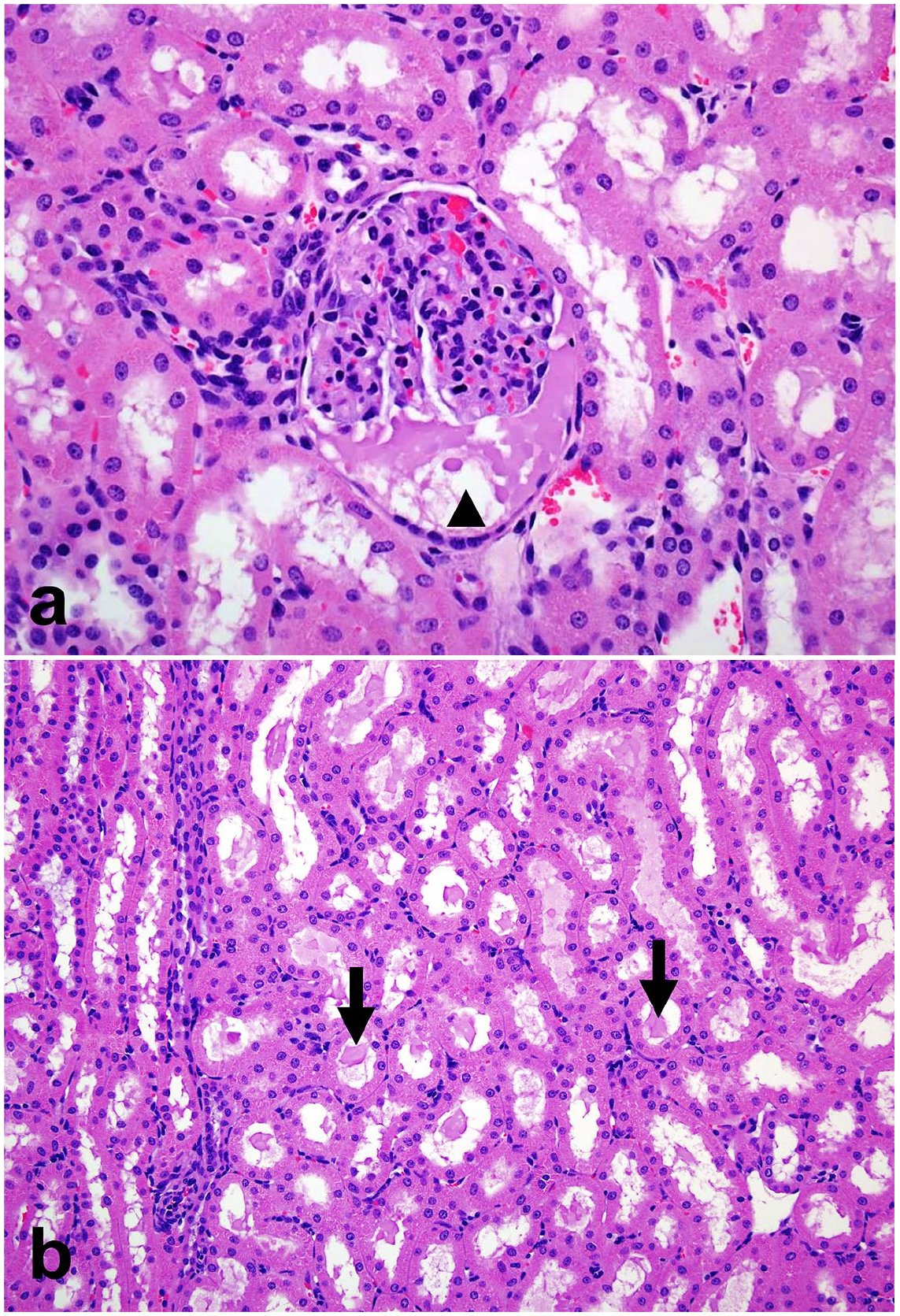
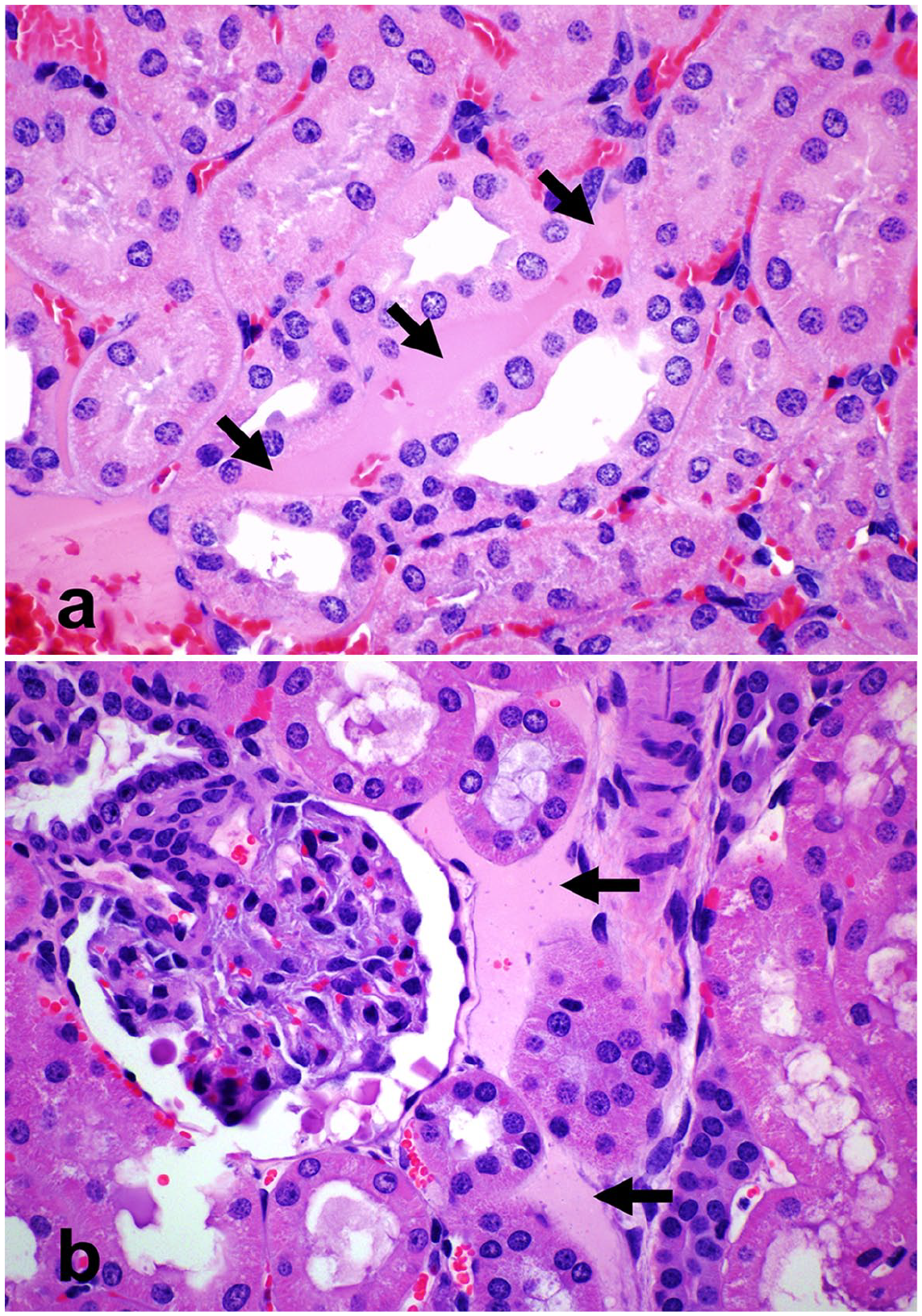
Proteinaceous material in the (a) Bowman’s spaces and (b) lumen of cortical tubules, kidney, sheep. The Bowman’s space (
Score 2 changes, the maximum score detected, included the presence of proteinaceous material in the lumen of cortical tubules and/or Bowman’s space in 5 sheep (2 of group 2, 1 of group 4, and 2 of group 3), mineralization of medullary tubules in 1 sheep of group 2, and epithelial cell death (Fig. 3) in medullary tubules of 4 sheep (2 of group 3 and 2 of group 4). Score 1 edema in the cortical interstitium (Fig. 4) was observed in 4 sheep (1 of group 1, 1 of group 3, and 2 of group 4). Inflammatory cell infiltration was only observed in the cortical interstitium and affected 17 sheep of all groups (4 of group 1, 5 of group 2, 3 of group 3, and 5 of group 4). In all 17 of these sheep, the inflammatory infiltrate was composed of lymphocytes, histiocytes, and plasma cells.
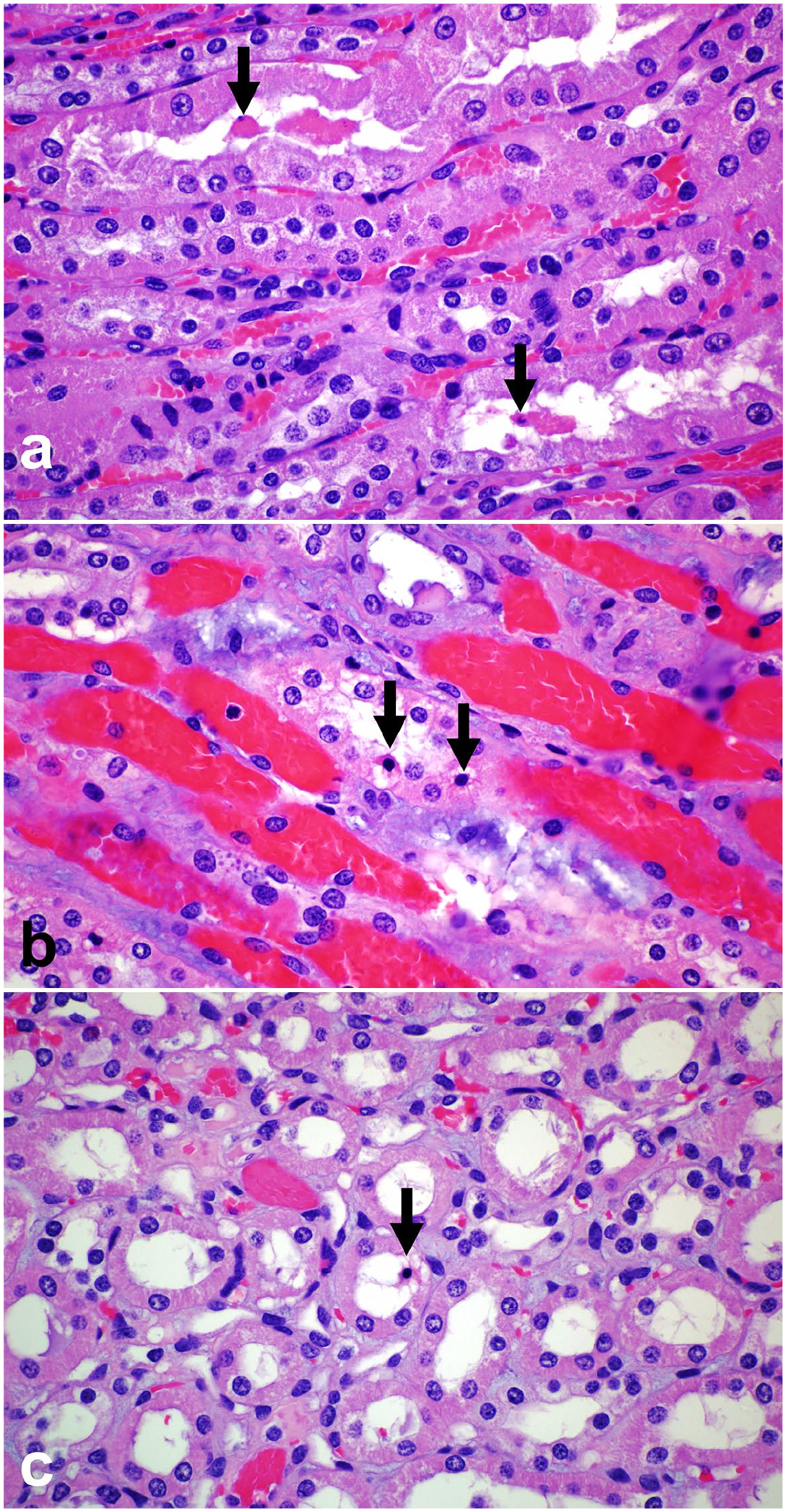
Epithelial cell death in the medullary tubules, kidney, sheep. Individual medullary tubules (
Edema in the cortical interstitium, kidney, sheep. The cortical interstitium is expanded by homogeneous eosinophilic proteinaceous material that separates the tubules (arrows), consistent with mild (score 1) interstitial edema, in sheep inoculated with the wild-type C. perfringens strain—group 1—(
The only change that was present, although infrequently, in sheep inoculated with ETX toxigenic strains (groups 1 and 3), but not in those inoculated with the etx knockout strain or sterile nontoxic culture medium (groups 2 and 4), was mild (score 1) hemorrhage in the lumen of a few (<5%) medullary tubules (Fig. 5). This change affected 2/12 sheep of groups 1 and 3 (1 from each group) and none of the 12 sheep of groups 2 and 4; these proportions did not differ significantly (P = .478). In addition, fibrosis in the cortical interstitium (score 1) was observed in only 1 of the 24 sheep, belonging to group 3. None of the 24 sheep showed any of the other predefined microscopic changes (Table 3). The intergroup comparison of the severity scores for microscopic changes between groups 1 and 4 (Table 3), and between groups 1 + 3 and 2 + 4 (Supplemental Table S2) did not reveal significant differences either.
Intergroup comparison of scores of histologic changes in groups inoculated with Clostridium perfringens type D wild-type strain (group 1), etx knockout strain (group 2), etx-complemented strain (group 3), or sterile culture medium (group 4, control).
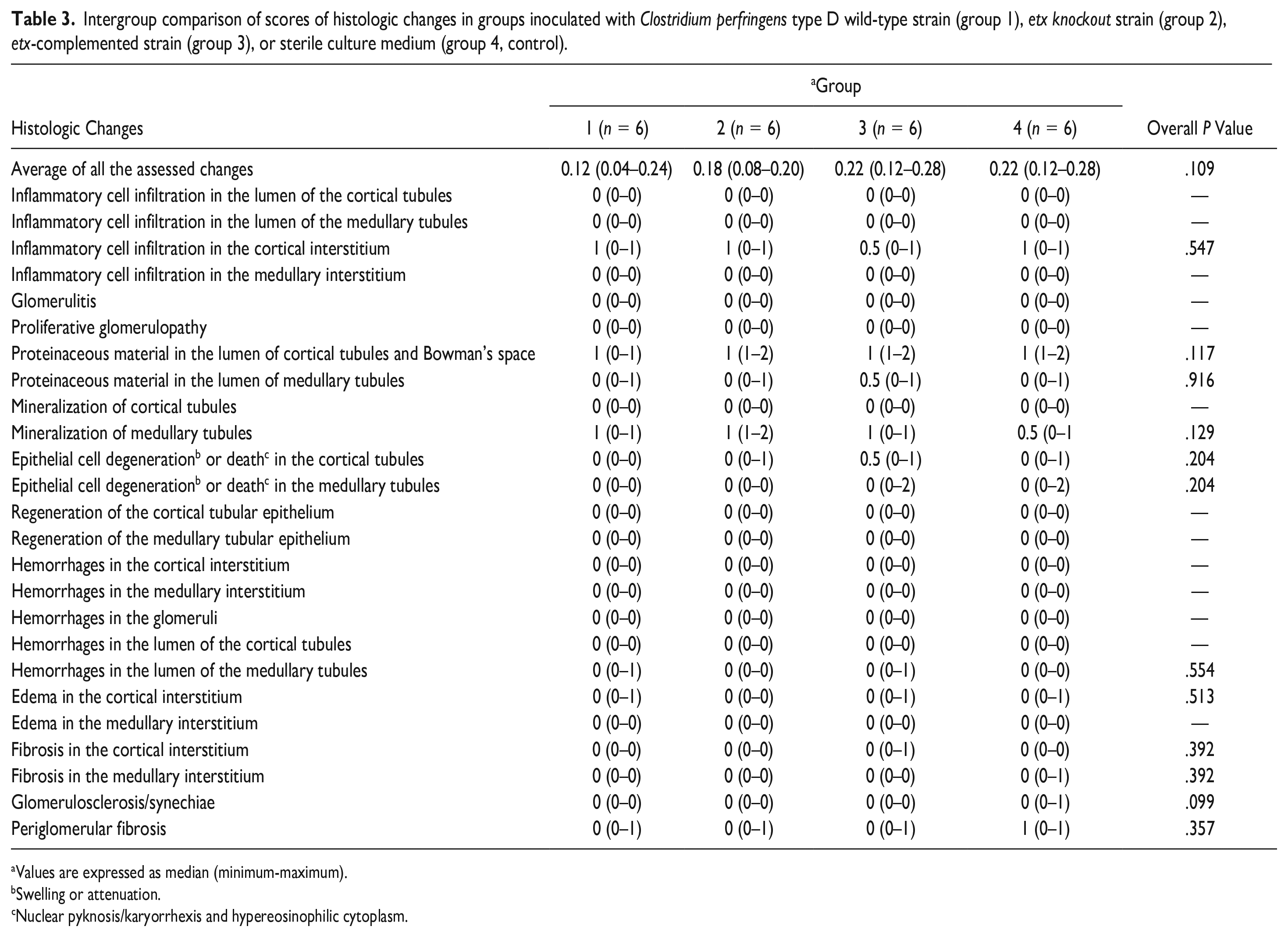
Values are expressed as median (minimum-maximum).
Swelling or attenuation.
Nuclear pyknosis/karyorrhexis and hypereosinophilic cytoplasm.
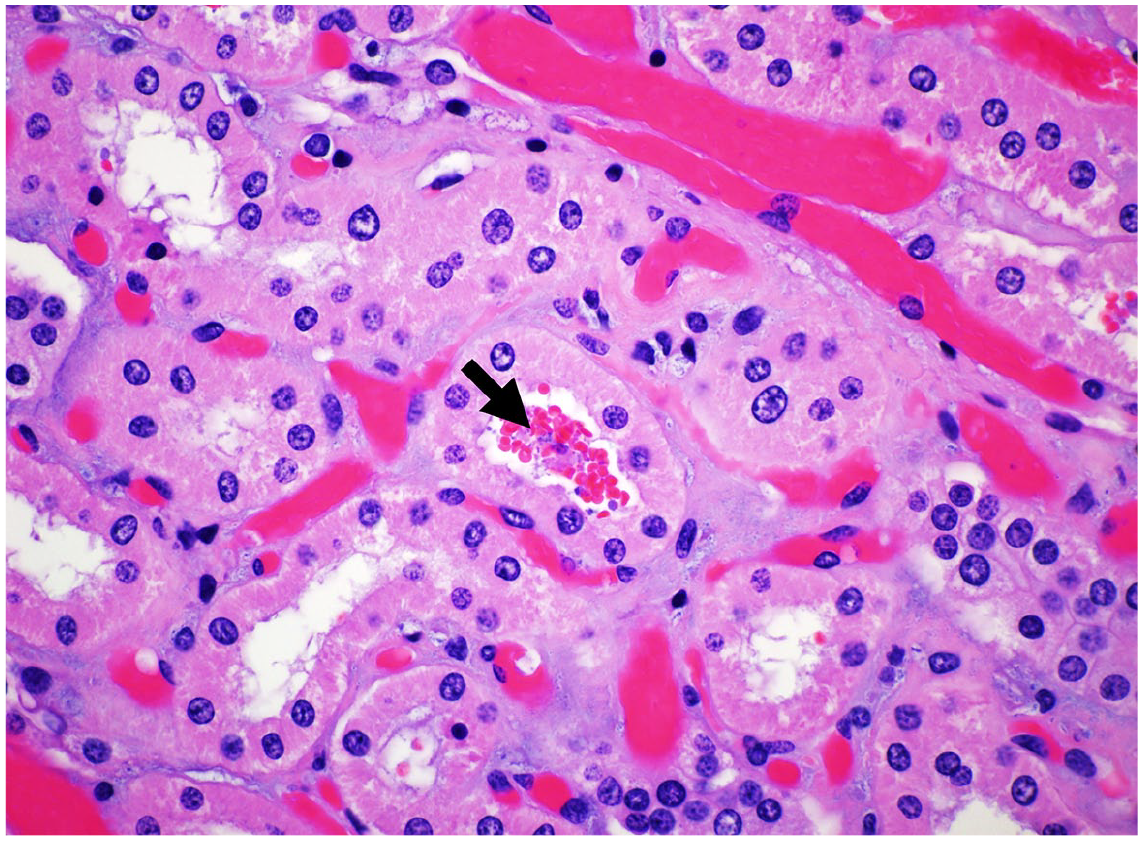
Kidney, sheep. A medullary tubule in a sheep inoculated with the etx-complemented C. perfringens strain—group 3—(sheep 20) contains intraluminal erythrocytes (arrow, score 1 hemorrhage). Hematoxylin and eosin.
Discussion
In this study, we demonstrated that sheep experimentally exposed to ETX toxigenic strains of C. perfringens type D, many of which succumbed to acute type D enterotoxemia, did not show specific gross or histologic lesions in the kidneys that would aid in the diagnosis of this disease. Despite the historical designation of “pulpy kidney disease,” the lack of gross renal lesions in all sheep in this study, as well as observations in previous studies,2,9,11,17 indicate that sheep that die of acute type D enterotoxemia have unaltered renal consistency if necropsied before significant autolysis takes place. Softening of the kidneys is most likely a change occurring postmortem. While it has been postulated that autolysis occurs more rapidly in the kidneys of sheep that die of type D enterotoxemia, 9 this has not been proved, and kidney softening can also occur after death in sheep regardless of the cause of death. Therefore, the name “pulpy kidney disease” can be misleading and, in our view, should be avoided.
To the best of our knowledge, there is only 1 study that assessed the speed of postmortem kidney softening in sheep with enterotoxemia. 9 The study suggests that renal softening may occur more rapidly in sheep that succumb to type D enterotoxemia than in euthanized controls held under identical postmortem conditions. However, the results should be interpreted with caution as they represent qualitative observations of a single study conducted with 2 groups of only 2 sheep each and, therefore, lack statistical validation.
Other gross renal alterations that have been described, although very infrequently, in sheep with experimental acute/peracute type D enterotoxemia, such as subcapsular petechiae or reddening of the papillae, 17 were not observed in the present study. The most frequent histologic change in our study was the presence of proteinaceous material in the lumen of cortical tubules and Bowman’s spaces, which was observed in 22/24 (91.7%) sheep. While these changes would fit with the proposed mechanism of action of ETX, if this toxin resulted in increased permeability of the glomerular capillaries with protein leakage to the glomerular space, their occurrence in many control sheep and animals inoculated with the etx knockout C. perfringens strain indicates that they are not associated with the action of ETX. Marked dilation of the Bowman’s space with proteinaceous fluid accumulation and atrophy of the glomerular tuft, referred to as “glomerular cystic atrophy,” has been described as a nonspecific finding in older animals and may occur because of impaired tubular fluid flow due to chronic damage such as tubulointerstitial scarring. 3 Conversely, in the sheep of this experiment, the Bowman’s spaces that contained proteinaceous material were not markedly dilated, there was no evidence of atrophy of the glomerular tufts in the affected glomeruli, and only 9 of the 22 sheep (40.9%) with this histologic change had evidence of chronic cortical damage, as indicated by the presence of mild cortical interstitial fibrosis, periglomerular fibrosis, and/or glomerulosclerosis in glomeruli not affected by protein accumulation. While it is possible that the accumulation of proteinaceous material in the Bowman’s space and proximal tubules in our sheep could be pathologic, physiologic, or artifactual, our results indicate that these changes have no diagnostic value for type D enterotoxemia. Elucidating the mechanism/s by which they occurred is beyond the scope of this article and would not be of clinical or diagnostic relevance.
Mild microscopic cortical interstitial edema (Fig. 4a) was observed in 2 sheep inoculated with the ETX toxigenic C. perfringens strains. Although this change would also fit with the proposed mechanism of action of ETX (ie, increase in vascular permeability), its presence in 2 control sheep (Fig. 4b) suggests that this was not an ETX-related effect in this study. As for all other changes, the percentage of sheep with this change and the severity scores did not differ significantly between groups (Tables 2 and 3).
We did not find evidence of tubular epithelial cell degeneration as a standalone change. Single-cell death was observed in the epithelium of medullary tubules in 2 sheep inoculated with the etx-complemented C. perfringens strain (group 3) as well as in 2 control sheep (group 2), suggesting that this histologic change is not specific to ETX action. In view of these results, we speculate that to some extent individual cell death may reflect the physiologic senescence and replacement of the tubular epithelium that takes place either independently of any significant tubular damage or because of nonspecific subclinical/sublethal insults. Native or recombinant ETX labeled either with radioactive iodine or green fluorescent protein accumulate in the kidneys of mice,13,15,16 and we cannot completely rule out that ETX may exert some degree of cytotoxicity on ovine renal epithelial cells in vivo. However, our findings indicate that there was no evidence of severe renal injury in sheep with experimental type D enterotoxemia.
The only histologic change found in 2/12 sheep exposed to ETX toxigenic strains, but not in any of the 12 sheep not exposed to this toxin, was mild (score 1) acute intratubular hemorrhage affecting <5% of the medullary tubules. Both sheep had died of acute type D enterotoxemia 7 hours 40 minutes (sheep 4) and 10 hours 10 minutes (sheep 20) after inoculation. Hemorrhaging is part of the spectrum of systemic lesions of this disease, and both animals also had moderate (sheep 4) or severe (sheep 20) hemorrhages in other anatomic locations such as the endocardium and myocardium. 10 Mild hemorrhaging into the medullary tubules could have been caused by ETX as part of the broader pathological picture in these 2 sheep. However, we cannot completely rule out that such a small number of erythrocytes might have been artifactually pushed/displaced within the tubular lumen during fixation, tissue trimming, or histologic processing/sectioning. Multifocal hemorrhage in the cortical renal interstitium, which has been very rarely observed histologically in sheep with experimental acute/peracute type D enterotoxemia, 17 was not observed in any of the sheep of the present study.
The inflammatory (lymphoplasmacytic interstitial nephritis) and degenerative (interstitial and periglomerular fibrosis, glomerulosclerosis, and medullary tubular mineralization) histologic lesions were interpreted as chronic, pre-existing subclinical changes considered to be incidental. These lesions were present before the experimental inoculations in all affected sheep of all 4 groups.
In a previous study, acute type D enterotoxemia was induced in 24 lambs. 9 The only gross alteration described in the kidneys at the time of death of affected lambs was a variable degree of congestion. The anatomic location within the kidneys, its severity, and the number of affected lambs that developed this nonspecific change were not provided. Histologic examination of samples of kidney collected at the time of death, fixed in formalin, processed, and stained with HE revealed no detectable differences in the light microscopic appearance in intoxicated and control lambs, and there was no evidence of tubular necrosis or interstitial hemorrhages. In addition, in the same study, 9 frozen sections of formalin-fixed kidney were processed by the azodye method to demonstrate alkaline phosphatase activity. This method did not reveal detectable histochemical differences in the activity of this enzyme in the brush border of the proximal tubular epithelium of control and intoxicated animals. Another study 17 compared the histologic changes in the kidneys of 3 lambs with experimentally induced type D enterotoxemia that developed clinical disease for 2–4 hours with those of 3 unaffected control lambs. All 6 lambs were autopsied 6 hours after euthanasia. Autolytic changes consisting of nuclear pyknosis, karyorrhexis, and karyolysis in the proximal and distal tubular epithelium were identical in animals from both groups.
Altogether, our study agrees with previous studies9,17 in that experimental acute type D enterotoxemia is not associated with gross or histologically detectable renal lesions that would be specifically attributable to ETX; in other words, similar changes can be observed in control sheep. A limitation of our study and the previous studies9,17 is that type D enterotoxemia was reproduced experimentally, which may not necessarily reflect the naturally occurring disease.
We conclude that, if the postmortem examination is conducted shortly after death to avoid significant autolysis, sheep experimentally inoculated with ETX-producing C. perfringens do not develop specific renal lesions, as renal changes in these sheep do not differ from those of sheep inoculated with the nontoxigenic C. perfringens or sterile culture medium. Based on the lack of evidence of antemortem loss of consistency of the renal parenchyma in sheep with this condition, we suggest that the common name “pulpy kidney disease” can be misleading and should be avoided. Further research is needed to elucidate whether autolysis leading to postmortem loss of renal consistency and/or histologic changes in the kidney occur more rapidly or to a greater extent in sheep with type D enterotoxemia.
Supplemental Material
sj-docx-1-vet-10.1177_03009858231171669 – Supplemental material for Experimental acute Clostridium perfringens type D enterotoxemia in sheep is not characterized by specific renal lesions
Supplemental material, sj-docx-1-vet-10.1177_03009858231171669 for Experimental acute Clostridium perfringens type D enterotoxemia in sheep is not characterized by specific renal lesions by Federico Giannitti, Jorge P. García, Vicki Adams, Joaquín I. Armendano, Juliann Beingesser, Julian I. Rood and Francisco A. Uzal in Veterinary Pathology
Footnotes
Acknowledgements
Federico Giannitti received a mobility grant (MOV_CA_2018_1_150021) from the Uruguayan “Agencia Nacional de Investigación e Innovación” (ANII) and is a member of ANII’s “Sistema Nacional de Investigadores” (SNI).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grant R01 AI056177 from the National Institute of Allergy and Infectious Diseases (NIAID). Research at Monash University was also supported by funding provided by the Australian Research Council to the Australian Research Council Centre of Excellence in Structural and Functional Microbial Genomics (grant no. CE0562063).
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
