Abstract
Madagascar hissing cockroaches (MHC, Gromphadorhina portentosa) are members of the Blaberidae (giant cockroaches) family of the Insecta class. They are native to the African island of Madagascar where they live within leaf litter on the rainforest floor. Due to their large size, relative tameness, and general easy keeping, they have become popular in classrooms, zoological collections, museums, research laboratories, and as private exotic pets; however, descriptions of diseases of MHC in the literature are rare. The objective of this study is to describe and characterize postmortem histological findings in 18 captive MHC from a single zoological collection. In this retrospective study, 18 (4 females and 14 males) adult MHC necropsies were submitted to Northwest ZooPath between 2016 and 2020 for evaluation. The main organs with histological lesions were chitinous gut (foregut and/or hindgut; n = 17), tracheae (n = 15), fat body (n = 14), ventriculus (midgut) (n = 13), body wall (n = 12), Malpighian tubules (n = 12), and hemolymphatic sinuses (n = 12). All animals had inflammatory lesions affecting various organs. Inflammatory lesions typically consisted of aggregates of hemocytes with variable amounts of melanization and/or encapsulation. Bacterial, fungal, and parasitic infections were common and variably associated with hemocytic inflammation. Many of these organisms may represent symbiotic organisms of the MHC that cause opportunistic infections. This study contributes to the current knowledge of pathological findings and disease response of MHC and reviews diseases reported in multiple cockroach species.
Cockroaches are members of the Blattodea order of the Insecta class.13,14 The Blattodea order contains at least 7 families and approximately 4000 species of cockroach with worldwide distribution. Only 1% of these species are considered pests and colonize human dwellings.13,14 Madagascar hissing cockroaches (MHC, Gromphadorhina portentosa) are members of the Blaberidae (giant cockroaches) family. 13 Adults are 5 to 10 cm in length, wingless, and have 2 tubercles on the dorsal surface of the prothorax. 36 The tubercles are more prominent in males. 36 They are native to the African island of Madagascar where they live within leaf litter on the rain forest floor. 36 They are nocturnal and omnivorous, typically feeding on ripened fruits that fall to the rain forest floor. 36 Both sexes have modified second abdominal spiracles through which air can be forced, resulting in the unique hissing sound that gives them their name. 36 The hissing sound is produced by both sexes when they are threatened or feel disturbed and is produced by males during mating/courtship and intermale fighting for territory. 36 Interestingly, they are considered an ovoviviparous species as fertilized eggs are incubated within the female’s ootheca for approximately 60 days before the nymphs hatch and emerge from the dam’s body resembling the birthing of live young. 36 The nymphs undergo gradual metamorphosis over approximately 5 months to become sexually mature adults. 36 Due to their large size, relative tameness, and general easy keeping, MHC have become popular in classrooms, zoological collections, museums, research laboratories, and as private exotic pets.9,10,17,34,36,61
In veterinary and human medicine, cockroaches are often considered a pest species due to their role in disease transmission, as a source of allergy development, or as intermediate or paratenic hosts to parasites of humans and various animal species.2,8,12,49,54,61 Descriptions of natural disease of MHC are limited to only a few reports. The objective of this study is to describe and characterize postmortem histological findings in 18 captive MHC from a single zoological collection.
Materials and Methods
Husbandry Details
Throughout the timeframe of this study, cockroaches were housed together in an exhibit enclosure at Central Florida Zoo & Botanical Gardens. The exhibit measured 35.6 × 17.8 × 38 cm. It was made of plastic with a frosted back wall, black on the side walls, and clear on the front wall to allow guests to visualize the MHC. The top panel of the exhibit was a metal screen. The substrate for the enclosure was 2.5 cm deep cypress mulch, which was spot cleaned daily and replaced as needed. There were 4 pieces of cork bark in the exhibit that allowed the roaches to hide and utilize the vertical space. A 43-watt incandescent bulb illuminated the exhibit and provided warmth. There was a 10-hour light and 14-hour dark/ambient room light cycle. The exhibit temperature was 25.5°C (78°F) and humidity was 50%. The exhibit was lightly misted each morning to maintain humidity. There was a naturalistic looking plastic water bowl filled with small rocks to prevent accidental drowning. Diet was offered in a naturalistic plastic food dish and consisted of wheat bran, fish flakes, and various vegetables. The produce was replaced daily if not completely consumed.
Pathology Review
A retrospective study was performed by reviewing archival data from a group of captive MHC submitted from Central Florida Zoo & Botanical Gardens to Northwest ZooPath between 2016 and 2020. Whole bodies were preserved in 10% neutral-buffered formalin. After fixation, the viscera were removed, routinely processed for histology, and stained with hematoxylin and eosin. Sections of the body wall were decalcified and dechitinized prior to processing using published protocols. 3 Cases were reviewed microscopically by 2 of the authors (C.F.M. and E.E.B.L.). To assist in microscopic evaluation, normal histology of insects was reviewed and a list of organs for examination was compiled (Table 1).28,29,51 Inflammatory lesions were routinely stained with histochemical stains (Giemsa, Grocott’s methenamine silver [GMS], Gram, and/or Ziehl-Neelsen acid-fast stains) to assess for infectious agents.
Organs for evaluation in Madagascar hissing cockroaches (Gromphadorhina portentosa).
Results
Archived necropsy tissues from 18 (4 females and 14 males) adult MHC were examined. All MHC were found dead and submitted for necropsy per Association of Zoos and Aquariums (AZA) requirements. When present in the histological section, the following organs most commonly had lesions: chitinous gut (including foregut and/or hindgut; 17/18; 94.4%), tracheae (15/18; 83.3%), fat body (14/18; 77.8%), ventriculus (midgut) (13/18; 72.2%), body wall (12/18; 66.7%), Malpighian tubules (12/18; 66.7%), hemolymphatic sinuses (12/18; 66.7%), glands (3/12; 25.0%), skeletal muscle (3/18; 16.7%), heart (1/8), and gonads (1/18, 5.6%). No lesions were observed in the brain, ventral nerve cord, peripheral nerves, or eyes.
Bacterial, fungal, and parasitic infections were equally common (13/18; 72.2%). Co-infections with bacteria, fungi, and parasites occurred in 7/18 (38.9%) cases, bacteria and fungi in 4/18 (22.2%) cases, fungi and parasites in 2/18 (11.1%) cases, bacteria and parasites in 2/18 (11.1%) cases, parasites only in 2/18 (11.1%), and no infectious agents were observed in 1/18 (5.6%) case. Gram staining was performed in 8 cases where bacteria were observed associated with lesions, but Gram staining characteristics of bacteria associated with lesions were determined in only 3/18 (16.7%) cases due to loss of bacteria in sectioning. Bacteria populations in those 3 cases included one case with a mixed population of gram-negative rods and cocci and gram-positive rods within a hindgut inflammatory lesion, one case with a monotypic population of gram-positive diplococci within the coelomic cavity, and one case with a population of gram-positive cocci arranged in chains within the chitinous gut. No acid-fast bacteria were observed. GMS staining highlighted fungal yeasts and, to a lesser extent, pseudohyphal and hyphal forms associated with and without inflammatory lesions in the body wall, tracheae, and gut lesions.
Chitinous Gut
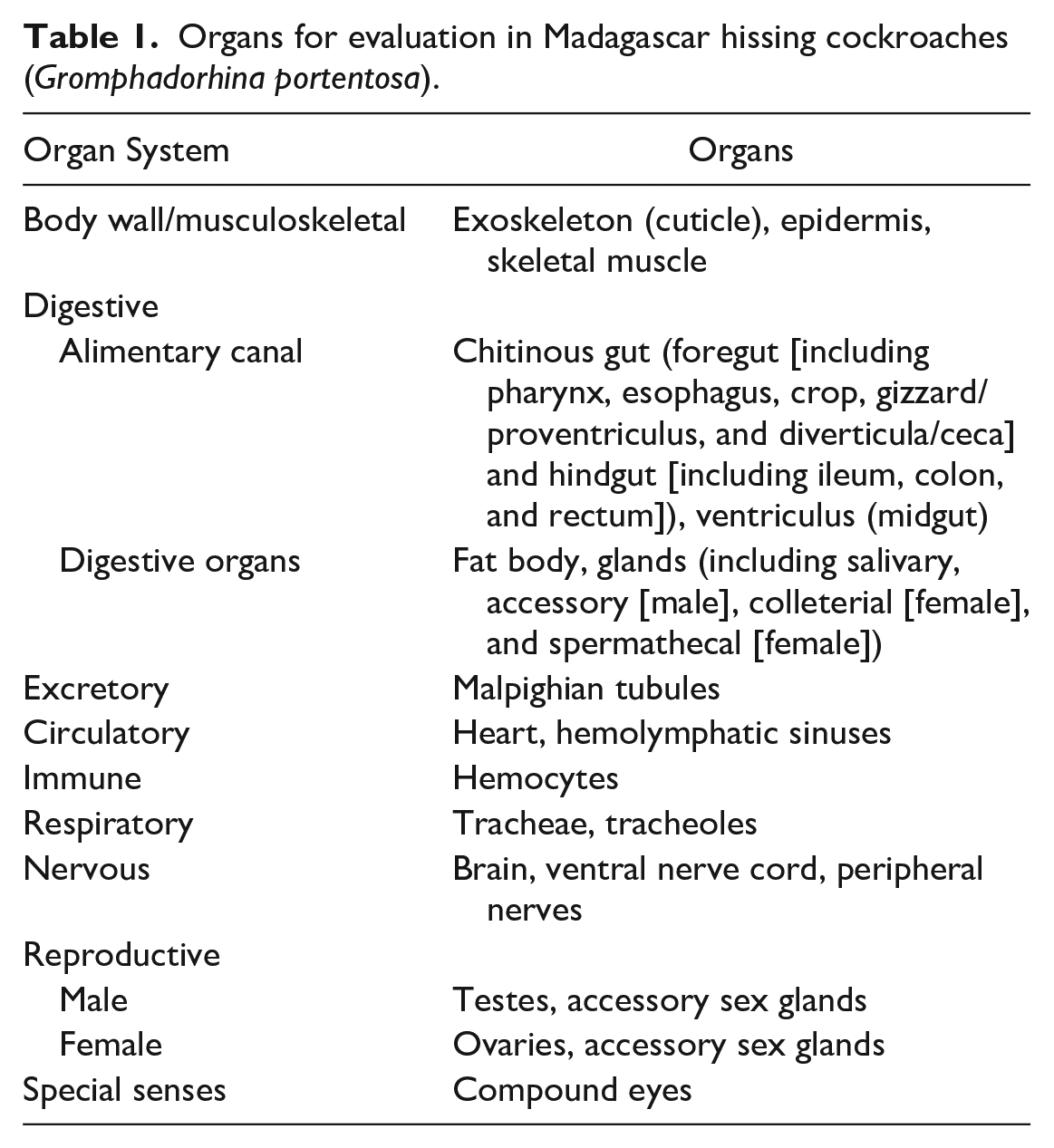
Inflammatory lesions within the chitinous gut (n = 17) were often associated with mucosal damage varying from erosion to ulceration of the lining epithelium. Three cases (3/17; 17.6%) had erosion only and were associated with superficial melanin deposition. Six cases (6/17; 35.3%) had ulceration associated with hemocytes and melanin deposition. In 4 of the ulcerated cases, the inflammation extended transmurally into and through the muscular wall (Fig. 1a). In one case, hemocytic inflammation was observed within the wall without associated mucosal damage. Bacterial overgrowth (11/17; 64.7%) or fungal overgrowth (1/17; 5.9%) was associated with the mucosal and inflammatory lesions. In one case, the bacteria were characterized with Gram staining and showed a mixed population of gram-positive and gram-negative rods and cocci. Bacteria and GMS-positive 3- to 5-µm (in diameter) fungal yeasts were limited to the immediately adjacent lumen and to the superficial aspect of the inflammatory lesions.
Lesions in the chitinous gut and tracheae of captive Madagascar hissing cockroaches (Gromphadorhina portentosa). (a) Chitinous gut. The wall is expanded and effaced by a nodular, encapsulated aggregate of hemocytes surrounding a region of melanization with central bacteria (asterisk). Hematoxylin and eosin (HE). (b) Chitinous gut. Within the lumen, there is a cross section of a nematode with lateral alae and an intestinal tract lined by columnar epithelium near a region of mucosal melanization. HE. (c) Chitinous gut. Within the lumen, there is a longitudinal section of an arthropod with a scalloped, chitinized body wall, an intestinal tract lined by cuboidal to columnar epithelial cells, a suspected tracheae, and a uterus containing embryonated eggs. Inset: A 90–100 × 60 µm, ovoid to reniform, embryonated egg with a thick brown to gold capsule and unipolar operculum. HE. (d) Hemocytic tracheitis, tracheae. The tracheal wall is locally extensively and transmurally expanded by sheets of hemocytes associated with mucosal melanization and luminal pigmented fungi. Inset: Higher magnification of pigmented fungal hyphae with septae. HE. (e) Fungal colonization, trachea. The lumen of the tracheae contains numerous fungi, which are not associated with inflammation. Grocott’s methenamine silver.
While typically not associated with mucosal damage, parasites were observed in multiple cases. In 5 cases (5/18; 27.8%), there were adult nematodes and arthropods in cross and longitudinal sections, and in 11 cases (11/18; 61.1%), nematode ova were observed with and without adults. The nematodes had lateral alae, platymyarian musculature, and an intestinal tract and measured 500 to 600 µm in width (Fig. 1b). In one case, a nematode cross section was intimately associated with a region of mucosal damage and hemocytic inflammation. The arthropods had a scalloped, chitinized body wall with an intestinal tract lined by cuboidal to columnar epithelial cells, suspected tracheae, fat body-like material, and a uterus containing 90–100 × 60 µm, ovoid to reniform, embryonated eggs with a thick eosinophilic capsule (Fig. 1c). One population of nematode ova were 70–80 × 40 µm, ovoid, embryonated ova with a thick brown to gold capsule. The other population was 90–100 × 60 µm, ovoid to reniform, embryonated ova with a thick brown to gold capsule and an occasional operculum (only unipolar was observed) (Fig. 1c, inset). In one case, a nematode ovum was embedded within a region of hemocytic inflammation.
When a more specific anatomical location could be determined for the inflammatory lesions (n = 5/17; 29.4%), lesions were identified in the ventriculus (1/17; 5.9%), foregut diverticula/ ceca (2/17; 11.8%), and hindgut (2/17; 11.8%).
Tracheae
Inflammatory lesions within the trachea (n = 15) were often characterized by hemocytic inflammation (13/15; 86.7%; one without melanin), melanin deposition (14/15; 93.3%; 2 without hemocytes), and/or ulceration (2/15; 13.3%) (Fig. 1d). Fungi (8/15; 53.3%) or bacteria (2/15; 13.3%) were associated with the inflammatory lesions. The fungi consisted of 3 to 5 µm yeasts that were occasionally associated with pseudohyphal and hyphal forms (Fig. 1e). In one case, the bacteria were characterized as gram-positive diplococci. In 4 cases (4/15; 26.7%), similar fungi were seen within the tracheal lumen without associated inflammation. In 3 cases (3/15; 20.0%), the inflammatory lesions were located near the spiracle opening. In 4 cases (4/15; 26.7%), the tracheal lumen contained flocculent eosinophilic fluid.
Fat Body
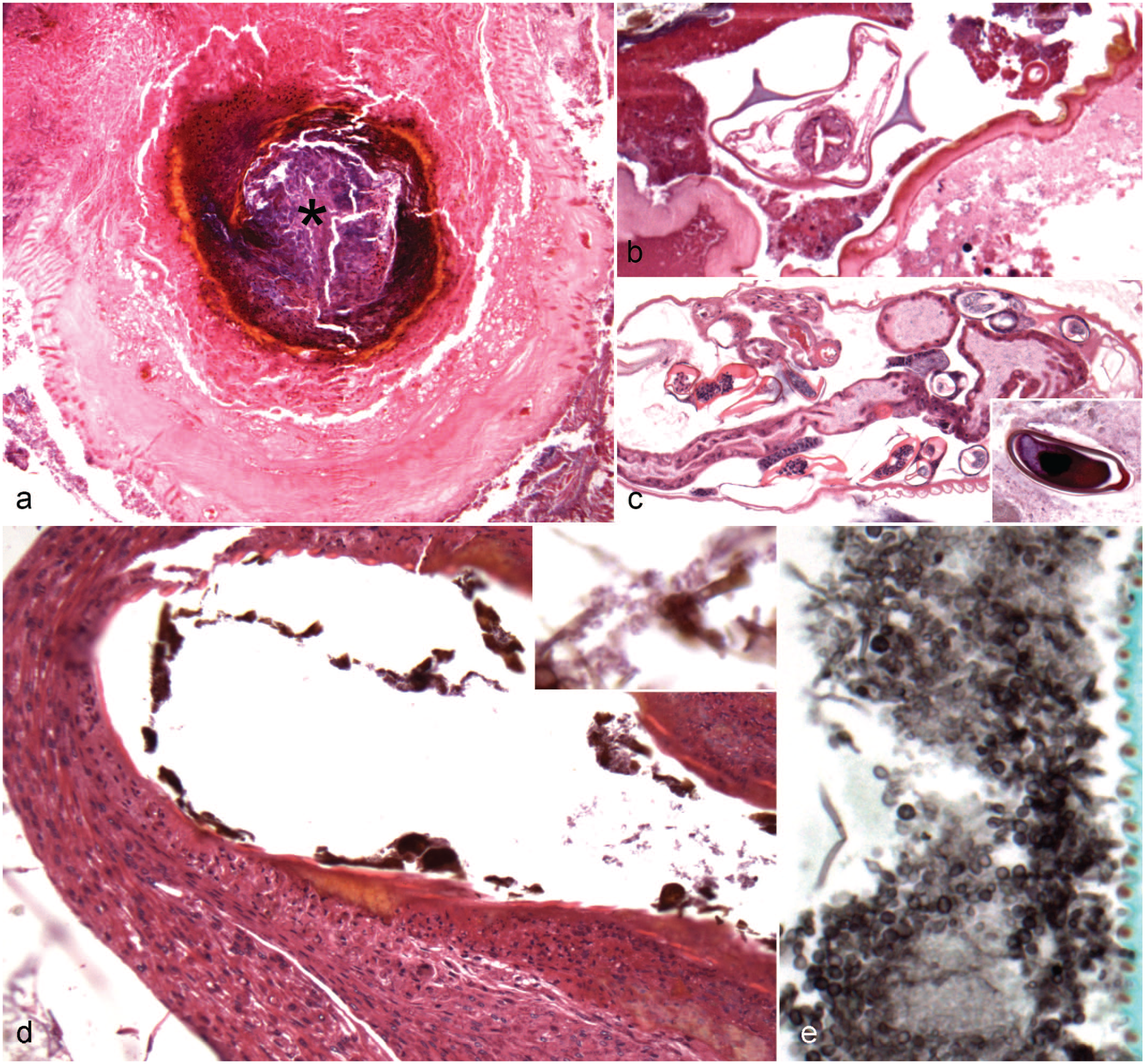
In 11 cases (11/18; 61.1%), the fat body was deemed adequate to abundant when lobules had abundant lipid and smooth, concave membranes (Fig. 2a). In the cases deemed abundant, the fat body filled the free space within the coelomic cavity. In 4 cases (4/18; 22.2%), the fat body was deemed to be atrophic or small with reduced lipid and comparatively small lobules with wrinkling of the outer membrane. In these cases, the fat body was sparse with frequent empty space within the coelomic cavity. In 3 cases (3/18; 16.7%), the fat body ranged from adequate to atrophic/small.
Histological findings in the fat body and ventriculus (midgut) of captive Madagascar hissing cockroaches (Gromphadorhina portentosa). Hematoxylin and eosin. (a) Fat body. This fat body is deemed adequate as it is large with abundant lipid components and smooth, concave membranes and fills the free space within the coelomic cavity. (b) Fat body. This adequate fat body has abundant lipid and bright eosinophilic droplets. (c) Fat body. This adequate fat body has frequent refractile, nonstaining crystalline material within its membranes. Inset: Refractile crystalline material (viewed with polarized light). (d) Ventriculus. The mucosa is focally ulcerated associated with transmural accumulation of hemocytes. (e) Gregarine-like organism, ventriculus. The lumen contains a gregarine gamont-like organism approximately 800 × 1000 µm with a distinct protomerite (arrow) along with suspected deutomerite (asterisk), and an occasional suspected nucleus (arrowhead) in section.
In 2 cases (2/18; 11.1%), there were numerous eosinophilic and lipid droplets throughout the fat body (Fig. 2b). Both cases were female and had adequate fat bodies. In 6 cases (6/18; 33.3%), there were abundant, nonrefractile, nonstaining crystalline-like material throughout the fat body. These cases included both males and females. In 5 of the cases (5/18; 27.8%) and one case (1/18; 5.6%) without the nonstaining crystalline-like material, there was accumulation of lesser amounts of light brown, crystalline-like material that was refractile under polarized light (Fig. 2c). All these cases, except one, were males. Five of the cases with nonrefractile, crystalline-like material (5/6) had adequate fat bodies and one had adequate to atrophic/small fat bodies.
In 8 cases (8/18; 44.4%), there was hemocytic inflammation within the fat body, which extended from inflammatory lesions within other organs or within the coelomic cavity. In 2 of these cases (2/8), the inflammation was associated with degeneration and focal accumulation of bright eosinophilic droplets. Six of these cases (6/8) had adequate fat bodies and 2 had atrophic/small fat bodies.
Ventriculus (Midgut)
Inflammatory lesions within the ventriculus (n = 13) were often associated with mucosal damage characterized by disruption of the peritrophic membrane and/or loss of the lining epithelium (Fig. 2d). Six cases had ulceration, which was associated with hemocytes in 5 cases and/or melanin deposition in 4 cases. In 2 of the ulcerated cases, the inflammation extended transmurally. In one case, the mucosal lesion was located at the junction with the chitinous gut. Bacterial overgrowth (5/13; 38.5%) or fungal overgrowth (2/13; 15.4%) was associated with the mucosal and inflammatory lesions. In one case, Gram staining revealed that the bacteria were a monotypic population of gram-positive diplococci. Bacteria and fungi were limited to the immediately adjacent lumen and to the superficial aspect of the inflammatory lesions. While not associated with lesions, protozoa were observed in multiple cases. In 6 cases (6/13; 46.2%), there were presumed gregarine (apicomplexan alveolates that infect invertebrates) within the lumen (Fig. 2e). The presumed gregarine gamont ranged in size from 250 µm to 2000 µm, variably showed a distinct protomerite along with the suspected deutomerite, and contained a nucleus in the section. In 2 cases (2/13; 15.4%), nematode ova were observed without adults. These ova had similar features as those described in the chitinous gut.
Body Wall
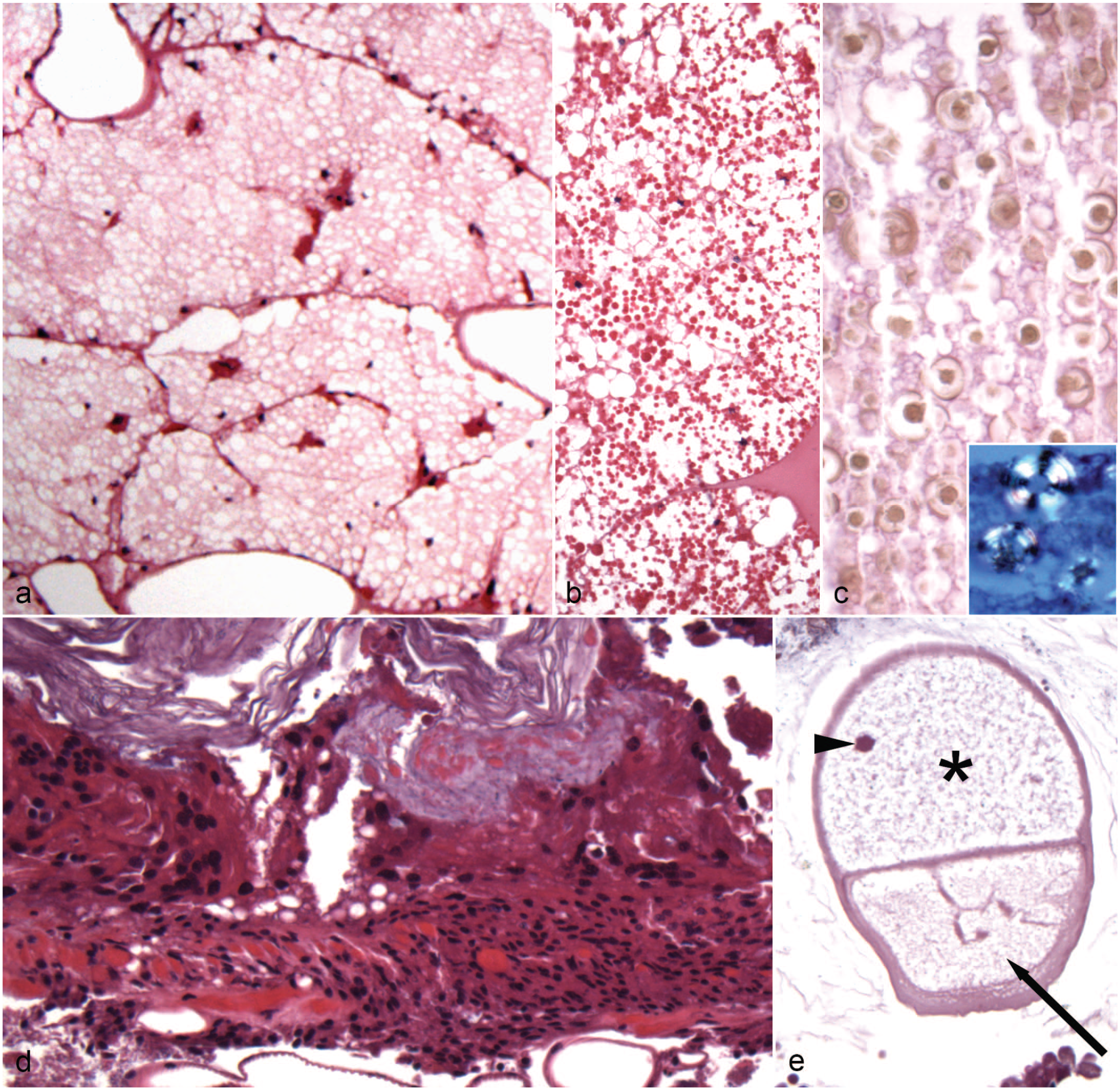
In body wall lesions (n = 12), there was often cuticle damage associated with melanin deposition and/or inflammation (Fig. 3a). Cuticle damage was characterized by basophilia that was interpreted as cuticle degeneration (5/12, 41.7%; Fig. 3b), erosion (4/12; 33.3%) associated with melanization, and ulceration with full thickness loss of cuticle and epidermis (6/12; 50.0%) associated with hemocytic inflammation and melanization. Fungal yeasts were associated with 2 erosions and 5 ulcers (Fig. 3a). In 3 cases (3/12; 25.0%), similar fungal organisms were seen on the chitinous lining away from inflammatory lesions (including in mouth parts and anus). The histological appearance of these fungi was similar to those observed in the tracheae. In one case, there were presumed bacteria associated with an erosive lesion, and a portion of a presumed arthropod mite, which was not associated with body wall lesions, was observed in another case. The section of presumed arthropod mite was 300–400 × 200–250 µm, had a thin chitinous wall, and contained indistinct, largely degenerate internal structures. When the anatomical location could be determined, lesions were present on the head (n = 7; 5 near antennae, 3 near the mouth, and/or 1 near the eye—these represent 7 total with some overlap), thorax (n = 4; one near spiracle), and abdomen (n = 3; one near appendage articulation and one near anus).
Histological findings in the body wall and Malpighian tubules of captive Madagascar hissing cockroaches (Madagascar hissing cockroaches; Gromphadorhina portentosa). (a) Degenerate cuticle, body wall. The cuticle is locally extensively basophilic and melanized suggestive of degeneration. Hematoxylin and eosin (HE). (b) Cuticle ulceration with melanization, body wall. The cuticle is focally ulcerated associated with melanization and infiltration of hemocytes within the dermis. HE. Inset: Similar cuticle lesion with intralesional fungi. Grocott’s methenamine silver (GMS). (c, d) Suspected fungi, Malpighian tubules. (c) The lumen contains multiple 10–20 × 5 µm, ovoid- to crescent-shaped organisms with a single basophilic nucleus arranged individually and in small clusters. HE. (d) These organisms stain with GMS staining, which highlights rare intracytoplasmic organisms within the lining epithelial cells in lumen along with multiple organisms within the lumen. Inset: Higher magnification of organisms. GMS.
Malpighian Tubules
Inflammatory lesions within the Malpighian tubules (n = 12) were often characterized by hemocytic inflammation (8/12; 66.7%), melanin deposition (7/12; 58.3%; 2 without hemocytes), and/or mineralization (1/12; 8.3%; associated with inflammation). In 6 cases (6/12; 50.0%), the inflammation extended into or was also present within the coelomic cavity. Suspected fungi were associated with the inflammatory lesions in 7/12 cases (58.3%). These presumed fungi were 10–20 × 5 µm, and ovoid to crescent shaped with a single basophilic nucleus arranged individually and in small clusters (Fig. 3c). These presumed fungi stained with GMS and Giemsa (Fig. 3d). Rarely, presumed fungi were seen within the cytoplasm of the epithelial cells lining Malpighian tubules. In 2 cases, similar presumed fungi were seen within the Malpighian tubule lumen not associated with inflammation, and in the other cases, these presumed fungi were seen within the Malpighian tubule lumen away from areas of inflammation.
Hemolymphatic Sinuses
The primary lesion within the hemolymphatic sinuses (n = 12) was hemocytosis (11/12; 91.7%), which was concentrated near areas of inflammation within other organs (9/12; 75.0%). In 2 of the cases, hemocytes were associated with melanin deposition. In 2 cases, the hemolymph was hypereosinophilic.
Other Organs
In the glands, inflammatory lesions (n = 3) were characterized by hemocytic inflammation and melanization. In one case, the lesion was near the mouth. In the skeletal muscle, lesions included myofiber atrophy (1/18; 5.6%), melanin deposition (1/18; 5.6%), and hemocytic inflammation (1/18; 5.6%), which extended from the adjacent body wall inflammatory lesion. In one case (1/8), approximately 90% of the heart was replaced by abundant luminal and transmural hemocytic inflammation with multifocal melanin deposition. In one case (1/14; 7.1%), approximately 70% of the testes were replaced by hemocytic inflammation associated with melanin deposition, aggregates of bacteria, and free sperm.
Discussion
This report describes histological lesions in a group of captive MHC from a single zoological collection. Hemocytic inflammation was the most common finding. The primary immune cell of insects is the hemocyte.18,23 The typical insect inflammatory response involves aggregation of hemocytes with melanin deposition, encapsulation, and nodulation.18,23 In this report, infectious organisms were often observed associated with inflammation. The clinical significance of these organisms is complicated by the fact that many present within the inflammatory lesions were also seen away from areas of inflammation. So, many of these are suspected to represent opportunistic infections by normal symbionts.
The gut microbiota of German cockroaches (Blattella germanica) is diverse with various microorganisms, including bacteria, viruses, fungi, protozoa, and archaea.31,54 The MHC mycoflora includes species of Rhizopus, Penicillium, Mucor, Trichoderma, and Alternaria. 58 In this report, many of the chitinous and ventriculus lesions were associated with abundant luminal bacteria that superficially extended into the areas of inflammation associated with mucosal damage. To a lesser extent, there were also fungi associated with mucosal damage. In most cases, the bacteria and fungi were not seen to extend into the deeper tissues and thus were considered to mostly represent secondary infection from commensals.
As these MHC were fixed whole, and no culture samples were obtained prior to fixation, the identity of these bacteria is unknown. Gram staining, when performed to characterize a bacterial-associated lesion that may or may not have been associated with a gut lesion, typically showed a mixed population of bacteria within the gut. In 2 cases, the bacteria appeared more monotypic (one with gram-positive diplococci within the coelomic inflammation and one with gram-positive cocci in chains within the chitinous gut inflammatory lesion) rather than heterogeneous, which is more concerning for a primary pathogen.
There is one report of a bacterial infection associated with histological lesions in cockroaches. A previous study described a Rickettsiella sp. associated with heavy body swelling and reproductive stagnation in the Oriental cockroach (Blatta orientalis). 41 Microscopically, intracytoplasmic aggregates of bacteria termed “bacterial vacuoles” were observed mainly within the fat body, but also in the gut epithelium, Malpighian tubules, blood cells, and ovarioles. 41 The morphology of these Rickettsiella sp. differed from those of the symbiotic bacteria of the genus Blattabacterium that typically occur within the mycetocytes (one of the cells of the fat body that harbors bacterial endosymbionts of the previous listed genus) of this species. 41 It was speculated that vertical propagation and fecal spread may occur in these infections. 41 Guzman et al 21 isolated a novel species (Entomobacter blattae) from the gut of a MHC. E. blattae is a gram-negative rod, classified as a member of Acetobacteraceae, and was not reported to be associated with disease or lesions. 21
Similar fungi were observed in the body wall and tracheal lesions as those seen in the gut. These fungi did not extend beyond the superficial aspect of the inflammatory lesions. As the morphology of these fungi was similar to those observed in the gut lesions, it is speculated that these may represent a similar fungal species. Also, similar fungi were present without inflammation within the tracheae lumen and along the body wall, so these may represent opportunistic infections by normal commensals.
Multiple fungal infections in various cockroach species have been described. A novel fungal species (Metarhizium argentinense) was described causing an undescribed disease in wild cockroach (Blaberidae: Epilamprinae) that was pathogenic to German cockroach nymphs in a laboratory setting. 20 A naturally occurring fatal fungal pathogen (Metarhizium blattodeae) was described affecting a sylvatic (forest-dwelling) cockroach in Brazil. 35 M. blattodeae was pathogenic to American cockroach (Periplaneta americana) nymphs under laboratory conditions. 35 Other described fungal infections in cockroaches include Hymenostilbe ventricosa that was found to affect nymphs attached to the underside of leaves in Thailand, 25 Ophiocordyceps blattarioides that was described to affect wild adult cockroaches from Columbia, 46 Ophiocordyceps blattae that was found on cockroaches in Sri Lankan gardens, 40 and Herpomyces shelfordellae that was found to affect Turkestan cockroaches (Shelfordella lateralis) from Hungary, Poland, and the USA. 22 Unfortunately, most of these reports did not investigate pathological findings in the affected insects, so it is unclear whether these infections are similar to those observed in this group of MHC.
While the fungi within gut, tracheae, and along the body wall were morphologically similar, the organisms positive for GMS and Giemsa staining within the Malpighian tubules had a different morphology and may represent a separate organism unique to the Malpighian tubules. Multiple unicellular eukaryotes, including apicomplexans, ciliates, amoeba, trypanosomes, and microsporidians, are described to infect insects with some of these eukaryotes serving symbiotic relationships with their hosts. 30 In cockroaches, there are 2 similar symbionts of the Malpighian tubules that are the primary differentials for the organisms observed in this case. A symbiont protozoan was described within the lumen and attached to the microvilli of the tubule cell of the Malpighian tubules of the American cockroach. 55 It was speculated that these symbionts may play an important role in the host insect’s metabolism similar to ruminal protozoa and ruminants. 55 Nephridiophaga sp. are believed to be a distinct clade of fungi that colonize the lumen and microvilli border of tubule cells, and are rarely found within the epithelial cells of the Malpighian tubules of insects, including cockroach species.42,52 The life cycle of this Nephridiophaga sp. includes merogony with multinucleate plasmodia and sporogony leading to small, uninucleate spores.42,52 The sporogonial plasmodia form 5-µm-wide, 10-µm-long, oval, flattened spores with one nucleus.42,52 Giemsa staining can be used to highlight the spores. 52 Nephridiophaga sp. have not been reported to be associated with lesions, so they are thought to be symbiotic commensals.42,52 Based on the size, staining affinity (GMS and Giemsa positive), the rare intra-epithelial localization, and the anatomic location, Nephridiophaga sp. is considered the primary differential for the organisms in the Malpighian tubules in this study. A microsporidian-like organism cannot be ruled out given the strong GMS staining. The previously described protozoal symbiont remains a differential but is considered less likely given the GMS and Giemsa staining, which would be atypical for protozoa. Molecular analysis would be required to further classify this organism. It remains uncertain whether these organisms represent primary pathogens, which would be the first report of it in cockroaches, or whether this represents opportunistic invasion or commensal overgrowth secondary to the inflammatory process of unknown cause.
In this report, multiple internal nematodes and internal and rarely external arthropods were identified. On the body wall of one MHC, there was a cross section of a suspected arthropod that may represent the cockroach mite (Androlaelaps (Gromphadorolalelaps) schaeferi). The internal structures were largely degenerate, so it was difficult to correlate specific features. A. schaeferi is a known symbiont of the MHC.38,58,63 A. schaeferi is an approximately 1-mm-long, brown mite, which is most commonly found on the underparts, near the mouth, at the base of the legs, and around the spiracles of MHC. 38 A. schaeferi is host-specific, so much so that upon the death of the MHC, A. schaeferi dies with it rather than move to a different MHC.47,62,63 It has been reported that A. schaeferi predates on developmental stages of other species of cockroach parasitic mites to help limit the mite burden, feed on saliva and organic debris that serves as a substrate for fungal growth resulting in a reduced fungal load on the exoskeleton, and has been associated with more efficient gut transit and clearance in MHC colonized by A. schaeferi compared with non-A. schaeferi-infested MHC.38,47,48,58 –60 A previous study reported that A. schaeferi-infested MHC had their life spans extended by approximately 9 months compared with A. schaeferi-free MHC. 58 However, there are rare reports where A. schaeferi has been associated with grossly evident yellow foci (necrosis) with lifting of the arthrodial membrane near mite aggregates. Inflammatory cell infiltrates were observed within the leg muscles in A. schaeferi-infested colonies, but were absent in A. schaeferi-free colonies. 38 Further investigation is needed to better understand the pathogenic and symbiotic relationships of A. schaeferi and MHC.
The lumen of the ventriculus of multiple MHC contained presumed gregarine gamonts. This is supported by their morphological description in multiple reports, size, and presence only within the ventriculus.1,4,5,11,50,64 Lethal disease manifesting as swollen abdomens and watery feces has been associated with large numbers of gregarines and watery fluid within the intestinal tracts of an MHC colony. 1 The gregarines were not proven to be the cause of the disease, but it was stated that the large number of parasites within the gut along with the clinical disease was evidence that they were the likely cause of enteric disease in these insects. 1 Multiple reports have described morphologically similar parasites (Leidyana (Blabericola) migrator [Apicomplexa: Eugregarinida]), but in contrast to the previously described case, no lesions were associated with L. migrator in these studies.4,6,11,50 Microscopically, the alimentary tract was colonized by gametocysts (260–1066 µm in diameter [depending on species]) that release 8 × 4 µm dolioform oocysts. 11 Trophozoites typically attach to the host ventricular and enteric cecal epithelium, while the gamonts are often free within the host ventricular peritrophic membrane and ventricular epithelium. 11 In the current case series, similar to most previous cases, gregarines were not associated with mucosal damage or obvious disease.
The lumen of the chitinous gut contained 2 adult organisms: a nematode and a suspected arthropod. The nematode had lateral alae and may represent an oxyurid, which have been described to inhabit the hindgut of MHC and other cockroach species.4,37,44 The brown shelled ova observed within the lumen may be those of this nematode. Rare cross sections of nematodes and brown shelled ova were observed immediately adjacent or embedded within regions of hemocytic inflammation. It is uncertain whether these nematodes are the cause of the inflammation or merely innocent bystanders. There was also another parasite ovum with an operculum (only unipolar was observed in these sections), which is not a feature of oxyurid ova; therefore, this is suspected to represent an ovum of a different parasite for which the adult was not observed. Given the operculum, nematode species within the order of Trichocephalida (including Trichuris spp.) are possible. Enteric nematodes have been described in MHC, but none have been described to be associated with lesions. These include oxyurid nematodes Leidynema portentosae and Leidynema appendiculota, which are found within the hindgut of MHC.4,44 It is unclear from these reports whether these represent distinct nematodes or the same nematode. Morphologically, L. portentosae was described to have unusually wide lateral alae, which show spine-like endings in females. 44 Therefore, more data are required to determine whether L. migrator, L. portentosae, and L. appendiculota are commensal symbionts of the alimentary tract or enteric pathogens of MHC.
The suspected arthropod had a chitinized body wall with suspected tracheae and fat body-like material. It is unusual for arthropods to colonize the gastrointestinal tract, so it is possible that as they were not associated with observed lesions, they may represent inadvertent ingestion of an external arthropod such as A. schaeferi. 63 There are also ubiquitous, free-living, saprophytic mites (including Tyrophagus putrescentiae) that have been reported to feed on the substrate within cockroach enclosures, but do not directly affect the insects, so it is possible that one of these mites could have been ingested from the environment. 38
Other parasites have been described to inhabit the gastrointestinal tract of cockroaches. Examination of fecal pellets of German cockroaches identified protozoan cysts/oocysts from Nyctotherus spp., Gregarina spp., Entamoeba spp., Cryptosporidium spp., coccidia, Lophomonas blattarum, and Balantidium coli. 54 Another group identified the presence of Entamoeba spp. by PCR in the intestines of 3 cockroach species: American cockroach, Argentinian forest cockroach (Blaptica dubia), and MHC. 26 In these reports, the presence of these parasites was detected, but no associated disease or lesions were mentioned.
In this report, there were no obvious viral lesions (eg, viral inclusions) observed. Viral infections are rarely reported in cockroaches. Hu et al 24 reported a disease affecting the smoky-brown cockroach (Periplaneta fuliginosa) caused by a densovirus termed “cockroach small spherical virus.” Densoviruses are invertebrate parvoviruses that are known to be a widespread pathogen of insects. 24 Disease manifestation or pathological findings were not described, and there were no reported ulcer-like lesions in the hindgut as had been previously described with “cockroach virus.”24,53 Radek and Fabel 43 described a cockroach entomopoxvirus causing chronic infection and reduced mobility in the German cockroach. Microscopically, 8 × 5 to 19 × 12 µm, ellipsoid virus inclusion bodies were observed intracellularly in tracheole cells, the hypodermis, fat body cells, and muscles. 43
The insect fat body is the primary site of lipid synthesis and storage, and carbohydrate and protein production. 27 Due to this, its health can be a good indicator of an insect’s body condition. It has been shown that during times of starvation, cockroaches mobilize glycogen and lipids from the trophocytes (adipocytes) of the fat body. 39 In this report, the fat body was deemed adequate to be abundant, small/ atrophic, or somewhere in between. Attempts to define body condition in insects can help provide insight to clinicians and caretakers as they set and adjust their husbandry and nutritional recommendations and choices.
In 2 cases (both females), there were numerous homogeneous, eosinophilic droplets throughout the fat body. Similar material has been described as suspected vitellogenin or female protein that is produced by the fat body and immediately released into the hemolymph during the normal female reproductive cycle leading up to ovulation.7,56,57 As this material was only present in 2 females and no male MHC in this case, this is the primary differential for this material, but other proteinaceous material cannot be ruled out.
Approximately a third of the cases contained crystalline-like material throughout the fat body. There were 2 morphologically distinct materials with one being non-refractile and nonstaining and the other being light brown and refractile under polarized light. These materials were observed in both sexes. The composition of these materials is uncertain, as is their clinical significance. Protein globules were described as refringent bodies in a previous report, but these were not further defined, so it is uncertain whether the refractile crystalline-like material observed in these MHC is similar. 56 It is thought that these changes may represent nutritional or metabolic effects on the fat body.
Many of the body wall lesions were located near articulations. These areas are often soft and permeable, making them more predisposed to trauma. Similarly, spiracle openings were affected in approximately one quarter of the cases with tracheal inflammation and/or melanization. As these are exposed to the external environment, they are more susceptible to damage and are a potential site for invasion by infectious organisms.
Hemocytosis was typically concentrated near inflammatory lesions within adjacent organs and therefore likely represented a secondary rather than a primary process. The hypereosinophilia of the hemolymph observed is thought to represent hemolymph concentration. Insects are reported to concentrate their hemolymph by decreasing their body water content as a response to various stressors.33,45
The only report of noninfectious or inflammatory disease described in MHC is a single case of a bilateral gynandromorph MHC with mixed female and male genitalia and a mosaic color pattern on the abdominal tergites (aberrant sexual mosaic). 19 There was no evidence of this condition in any of the MHC in this case series.
Due to their small size, gross examination of MHC is often difficult, and therefore limited. There are a few sources for gross anatomy of MHC or insect anatomy texts that include MHC, which can be helpful to better understand the anatomy of these insects.15,16,32 There have been multiple recent articles discussing anesthesia of MHC, radiology of invertebrates, and general advice for invertebrate medicine, which shows an increasing interest in invertebrate medicine by clinicians.9,17,34 Postmortem examinations, including histological examination by veterinary pathologists, is key to better understand diseases of invertebrates, including insects.
Conclusion
In this report, inflammatory lesions were common in MHC. Bacterial, fungal, and parasitic infections were common and were variably associated with hemocytic inflammation. Many of these may represent opportunistic infections by symbionts of the MHC. Ancillary testing would be beneficial to better characterize these infectious agents and to understand their significance to the disease processes of MHC. This study expands our knowledge of pathological findings and disease processes of MHC and provides a review of diseases reported in multiple cockroach species.
Footnotes
Acknowledgements
Thank you to Cathy Minogue and Christie Buie of Northwest ZooPath for collating materials. Thank you to Leroy Brown of Histology Consultation Services for specimen preparation. Thank you to Shelby Halley of New Hampshire Veterinary Diagnostic Laboratory for histochemical stain preparation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Disclaimer
The views expressed in this article are those of the authors and do not necessarily reflect the official policy of the Department of Defense or the U.S. Government.
