Abstract
An outbreak of morbidity and mortality in an African dwarf frog (Hymenochirus curtipes) colony was reported following arrival at an animal research facility. Animals were found dead on arrival or became moribund shortly thereafter, and additional animals showed clinical signs of lethargy, weight loss, and anorexia over the following 3 weeks. Externally, some affected animals presented with multifocal areas of hyperemia in the inguinal and axillary areas and on the limbs, and mottled tan discoloration along the ventral abdomen. Histologically, lesions were consistent with generalized septicemia, characterized by granulomatous meningitis, otitis media, peritonitis (coelomitis), myocarditis and pericarditis, nephritis, pneumonia, and arthritis. Gram staining identified gram-negative rod-shaped bacteria free within tissues and within macrophages. Culture results of coelomic swabs identified moderate to numerous Elizabethkingia miricola. Testing of water from tanks housing affected animals showed elevated levels of nitrites and ammonia, and the presence of Citrobacter, Aeromonas, Pseudomonas, and Staphylococcus spp. cultured from several tank biofilters. E miricola is a newly recognized and rapidly emerging opportunistic pathogen in anurans and has been reported as a cause of septicemia in humans. This report documents the first occurrence of E. miricola septicemia in African dwarf frogs and illustrates the importance of this potential pathogen in the laboratory setting for amphibian research colonies, as well as those individuals directly working with them.
Keywords
Elizabethkingia spp. are gram negative, aerobic, nonmotile, nonspore forming, nonfermenting, oxidase-positive bacilli that are ubiquitous in natural environments such as water (fresh and salt water), soil, and animals such as fish, frogs, and insects.1–3,5–8,14,17,18 In recent years, case reports of Elizabethkingia spp. within amphibian populations, specifically anurans, have been on the rise, with disease outbreaks occurring in different frog species around the world in both wild and captive populations. 25 Moreover, Elizabethkingia spp. are also an occasional cause of serious, life-threatening infections in humans and have been isolated from hospital environments including water supplies and equipment.4,5,12,14,18,27 While opportunistic and rare, the incidence of Elizabethkingia infection in humans has significantly increased over the past decade, resulting in a public health concern with high case-fatality rates, intrinsic antimicrobial resistance, and reports of zoonotic potential. 4
The current case series documents a large-scale outbreak of Elizabethkingia miricola in a colony of African dwarf frogs (Hymenochirus curtipes). This report is of particular importance because the relevance of this pathogen is largely unrecognized; it has not been reported previously to cause significant disease in the laboratory setting and is an important cause of acute morbidity and mortality in anurans. In addition, given its multidrug resistance profile and zoonotic potential, it is of particular concern for those individuals working with anurans in the laboratory setting. Here we document the clinical, gross and histologic, and microbiologic features of a large-scale outbreak caused by this rare opportunist and discuss the relevance of this disease in humans and anurans and the importance of recognizing this organism in the laboratory setting to minimize disease outbreaks, prevent loss of data, and protect laboratory personnel.
Materials and Methods
Animals and Husbandry
Animals (n = 102) were initially purchased from a single commercial vendor (vendor A) and transported to the University of Michigan animal facilities overnight in temperature-controlled tanks. Subsequent acquisition of animals was provided by 2 separate commercial vendors (vendor B, n = 164; vendor C, n = 44). Animals were of juvenile age. Animals were on an Institutional Animal Care and Use Committee (IACUC)–approved protocol to study chytridiomycosis biology. Upon arrival at the University, animals were placed in quarantine and heat-treated to clear any potential fungal infection. Heat treatment began 1 week after arrival and continued for 3 weeks. Using an aquarium heater, heat treatment was performed by slowly increasing the quarantine aquaria by 1°C per day to an end goal of 30°C and maintained at this temperature for 7 days. After 7 days, the water temperature was slowly returned to housing temperature by 1°C per day. Animals were housed in 9 to 10 gallon tanks containing up to 20 individuals. Each aquarium contained various enrichments such as polyvinyl chloride (PVC) pipes, gravel, and plastic plants and was enclosed with a lid. Water temperatures were set and maintained the same as the room temperatures, between 19°C and 21°C. In the animal room, fluorescent lighting was fixed to a 13-hour-on, 11-hour-off cycle. Daily health checks were performed for moribund or unhealthy animals. Diet for the animals included pelleted foods (Fluval Bug Bites Tropical Micro Granules, Rolf C. Hagen Inc., Mansfield, Massachusetts), live brine shrimp (Aquafauna Bio-Marine, Inc., Hawthorne, California), and bloodworms (San Francisco Bay Brand, Inc., Newark, California) every 2 to 3 days. Water testing was performed (API® Freshwater Test Kits, API FishCare, Chalfont, Pennsylvania) weekly for levels of nitrates, nitrites, and ammonia, and daily for pH levels.
Necropsy and Histopathology
Moribund animals were removed from the colony and humanely euthanized using MS-222 immersion. Following external examination, each animal was immersion-fixed in 10% neutral-buffered formalin for 24 to 48 hours. Following fixation, each specimen was decalcified in Immunocal (StatLab, Columbia, Maryland) for 4 to 6 hours. After sufficient decalcification, the limbs were removed, and the body of each animal was bisected longitudinally, placed in cassettes, and processed for histology by the In Vivo Animal Core facility in the Unit for Laboratory Animal Medicine at the University of Michigan by routine methods. Tissues were processed to paraffin in an automated tissue processor (TissueTek VIP, Sakura Finetek USA, Torrance, California), embedded, sectioned at 4 μm thickness on a rotary microtome, and mounted on glass slides. Sections were stained with hematoxylin and eosin by routine histological methods on an automated histostainer (Leica ST5010 XL, Leica Biosystems, Buffalo Grove, Illinois) and coverslipped with Micromount (Leica; #3801731) using a Leica CV5030 automatic coverslipper. Gram staining was performed using alcoholic crystal violet-oxalate stain followed by aqueous Lugol’s iodine solution, decolorized in acetone, and stained with Twort stain (Newcomer Supply, Middleton, Wisconsin).
Microbiology and Water Quality Assessments
Coelomic swabs were obtained from all clinically affected (n = 45) and unaffected (n = 5) animals, and environmental swabs were obtained from tanks that housed affected animals. Swabs were submitted for general bacterial culture to the Michigan State University Veterinary Diagnostic Laboratory. Samples were plated on blood agar, MacConkey agar, and colistin-nalidixic agar plates (Hardy Diagnostics, Santa Maria, California), incubated overnight at 35°C to 37°C in a 5% CO2 incubator, and observed for growth of bacterial colonies. The bacterial colonies were identified using matrix-assisted laser desorption ionization-time of flight mass spectrometry (Bruker Microflex LT; Bruker Daltonics, Bremen). A skin swab was also performed on each animal, and was submitted for Xenopus infectious disease (Batrachochytrium dendrobatidis, Mycobacterium chelonae, Mycobacterium marinum, Pseudocapillaroides xenopi, Ranavirus spp., Cryptosporidium spp., Pseudomonas aeruginosa, Salmonella spp.) PCR panel (Idexx BioAnalytics, Columbia, Missouri). Water quality testing was performed daily to detect any abnormalities in pH (pH >7.5 or <6.5), and weekly for elevations in ammonia (>0.5 mg/L), nitrite (>0.5 mg/L), and nitrate (>50 mg/L).
Results
Of the 102 animals initially received from vendor A, approximately 10% of the arrived were deceased. Over the following 24 hours, 11 frogs became moribund and either died or were euthanized. Over the next 3 weeks, the receiving laboratory reported approximately 1 to 5 deaths per day. By week 3, roughly 50% of the colony remained and were ultimately culled. As deaths subsided in frogs obtained from vendor A, elevated morbidity and mortality began occurring in frogs obtained from vendor B. Out of 164 animals from vendor B, 15 became moribund and were euthanized for assessment. Animals from vendor C did not develop clinical signs and no deaths were reported. Clinically affected frogs were presented with clinical signs of bloating, reddish discoloration of the skin, lethargy, weight loss, anorexia, and floating at the surface of the water. Ultimately, 45 frogs were humanely euthanized for pathology examination due to clinical morbidity over the span of 5 weeks, along with additional clinically unaffected animals from vendors B (n = 2) and C (n = 3) to serve as controls. At the time of gross examination, a majority of affected animals were in fair to poor body condition, with noticeable weight loss. Eleven clinically affected animals had gross lesions characterized by locally extensive tan to orange discoloration along the ventral abdominal skin surface (Fig. 1a), 3 animals had reddish discoloration along the forelimbs (Fig. 1b), and 2 animals presented with dark red watery abdominal fluid.

Elizabethkingia miricola infection, African dwarf frogs. Clinically affected animals were thin, in poor body condition, some with (a) locally extensive tan to orange discoloration along the ventral abdominal skin surface (arrow) and/or (b) multifocal cutaneous hyperemia along the forelimbs (arrowhead).
Histologically, lesions within affected animals were variable in severity and distribution, and largely consisted of multifocal granulomatous inflammation affecting multiple organ systems, consistent with septicemia. Of the total number of clinically affected animals (n = 45), 15 had histologic lesions in one or more body systems, including the meninges of the brain and/or spinal cord, middle ear, coelomic cavity, lung, myocardium and/or pericardium, kidney, and joints (Fig. 2). Gram staining of affected animals demonstrated gram-negative rods within areas of inflammation (Fig. 3a), as well as within the cytoplasm of macrophages (Fig. 3b). Clinically unaffected animals did not have detectable gross or histologic lesions.
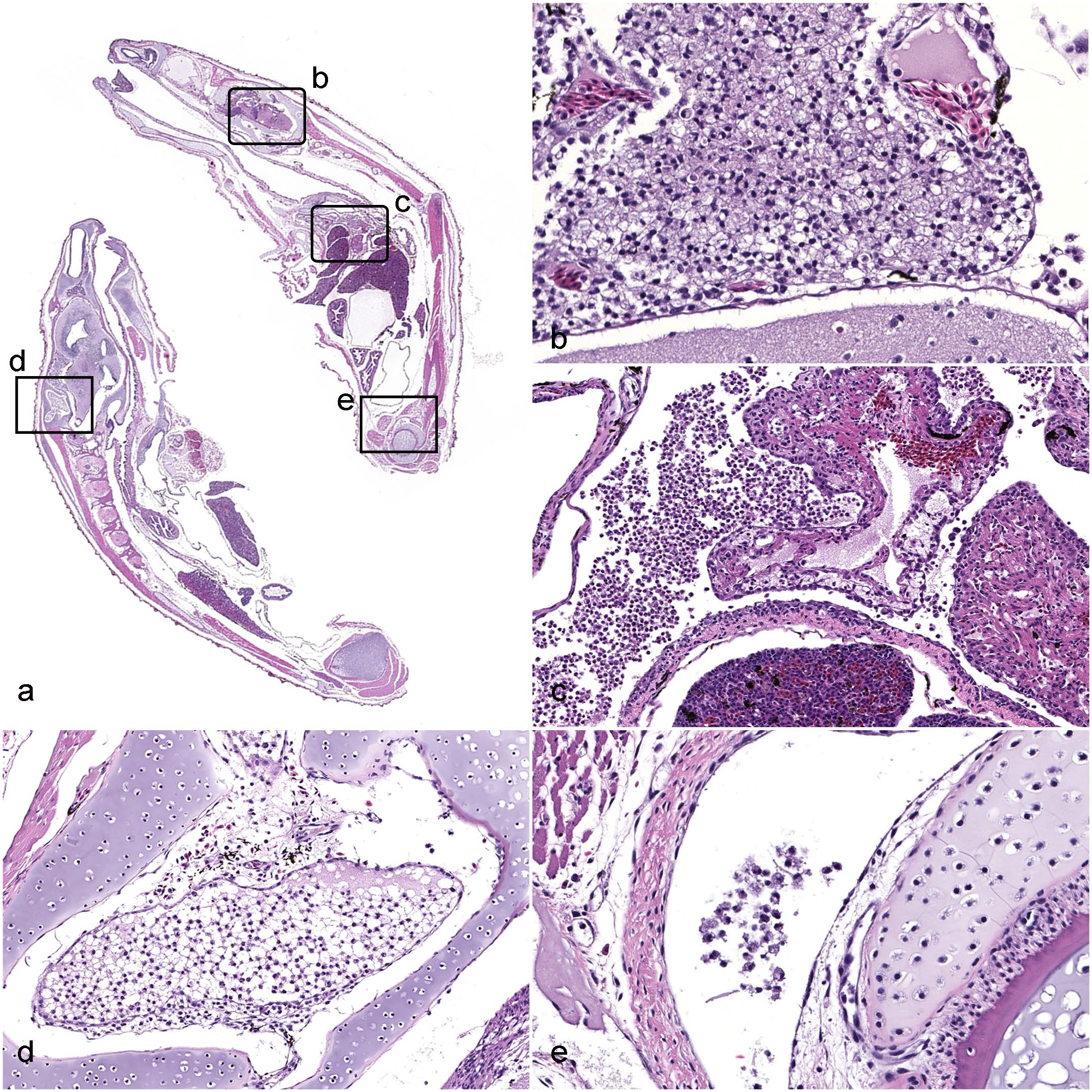
Elizabethkingia miricola infection, African dwarf frogs. Hematoxylin and eosin. (a) Subgross image of longitudinal section of the head and body of an affected frog showing involvement of multiple organ systems. Boxes highlight areas represented in the figures denoted by the lowercase letters. (b) Granulomatous meningitis, cerebrum. Granulomatous inflammation expanding the meninges is characterized by numerous large macrophages with abundant foamy cytoplasm. (c) Granulomatous coelomitis, pericarditis, and myocarditis. Macrophages are present within the coelomic cavity and infiltrate the pericardium and myocardium of the heart. (d) Granulomatous otitis media, middle ear. Foamy macrophages are present within the tympanic bulla of the middle ear. (e) Granulomatous arthritis, coxofemoral joint. Infiltrating macrophages are present within the joint space.
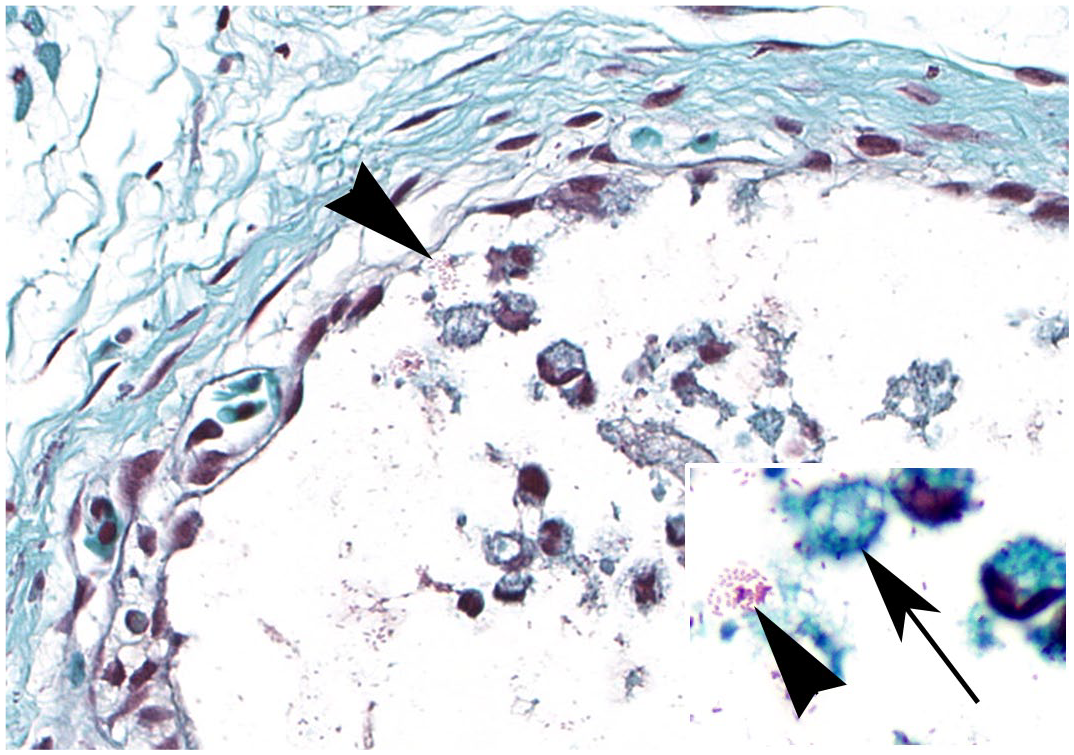
Elizabethkingia miricola infection, coxofemoral joint, African dwarf frogs. Scattered gram-negative bacilli are present (arrowheads). Inset: higher magnification shows the bacilli both within the cytoplasm of foamy macrophages (arrow) and free within areas of inflammation (arrowhead). Gram stain.
Coelomic culture isolated moderate to numerous E miricola in 29 of the 45 affected frogs from vendors A and B. Coelomic swabs from vendor C were negative for E miricola and other pathogens (Table 1). Susceptibility results for frogs infected with E miricola from vendors A and B showed that isolates were resistant to all cephalosporins (cephalexin, ceftazidime, cefpodoxime, cefazolin, and cefovecin), aminoglycosides (amikacin and gentamycin), ampicillin, and tetracycline (Table 2). Isolates were susceptible to trimethoprim sulfamethoxazole, piperacillin/tazobactam, doxycycline, and fluoroquinolones (marbofloxacin, orbifloxacin, and pradofloxacin). Moderate to numerous numbers of Citrobacter, Aeromonas, Pseudomonas, and Staphylococcus spp. were cultured from all 3 tank biofilters, but tank biofilter cultures were negative for E miricola. PCR testing of skin swabs was negative for chytrid fungus and other pathogens tested on the Idexx polymerase chain reaction (PCR) panel. Water quality testing showed elevated levels of ammonia (0.5–1.5 mg/L) and nitrite (0.5–2 mg/L) in 5 tanks (of 9 total) over a period of 7 days in which clinically affected frogs were housed.
Summary of lesions and E. miricola cultures in affected African dwarf frogs and sampled aquaria.
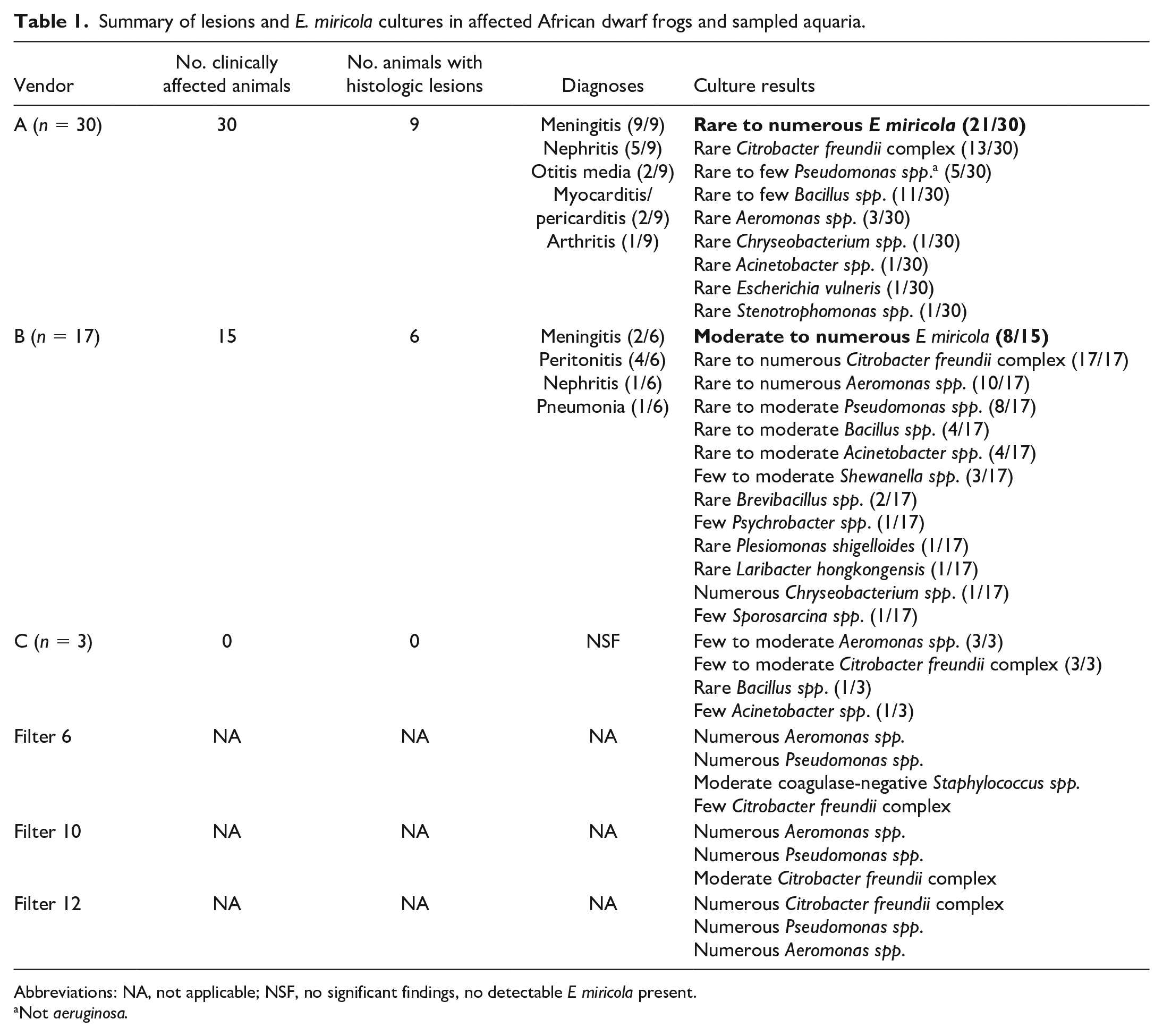
Abbreviations: NA, not applicable; NSF, no significant findings, no detectable E miricola present.
Not aeruginosa.
Antimicrobial susceptibility interpretations of E miricola in affected African dwarf frogs.
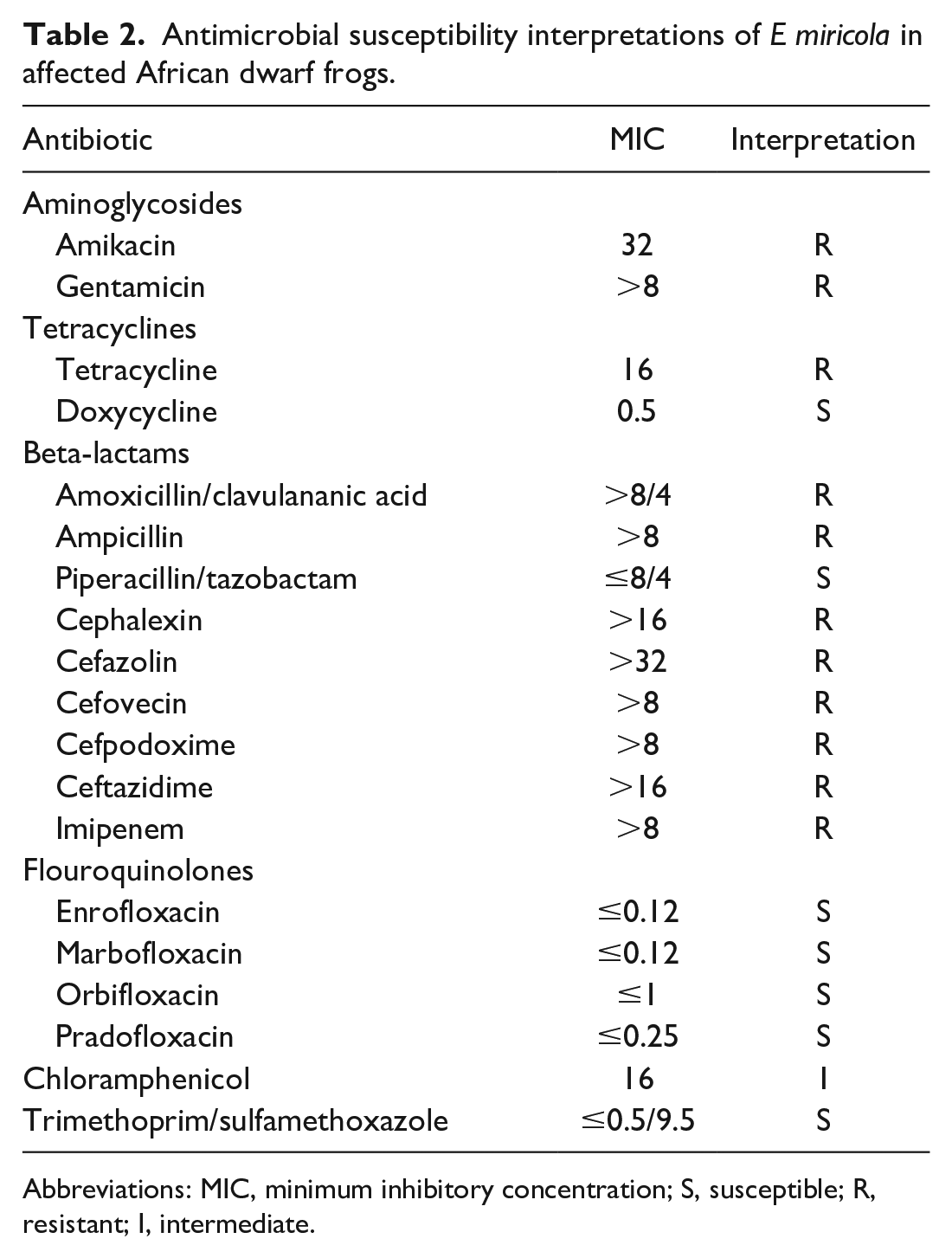
Abbreviations: MIC, minimum inhibitory concentration; S, susceptible; R, resistant; I, intermediate.
Discussion
The current case series is the first to report E miricola as a cause of fatal septicemia in African dwarf frogs in the laboratory setting. E miricola has been recently reported as a rapidly emerging opportunistic bacterial pathogen affecting a wide range of anuran species, most typically wild populations. 25 In addition, recent literature reports that this pathogen is highly resistant to antimicrobials and is a potential human pathogen.4,5 The current study emphasizes the importance of recognizing this bacterium as a pathogen in laboratory anurans to prevent disease outbreaks that may result in significant loss of animals and data, and expose laboratory personnel to a potential zoonotic pathogen.
The genus Elizabethkingia was originally discovered in 1959 by Elizabeth O. King, an American microbiologist at the Centers for Disease Control and Prevention studying meningitis and septicemia in infants; the bacterium was initially named Flavobacterium meningosepticum.5,18 In 1994, the bacterium was moved to a new genus and was named as Chryseobacterium meningosepticum. In 2003, a new member of the genus was discovered from condensation water from the Russian space station Mir collected in 1997 and named as Chryseobacterium miricola. Finally in 2005, these species were reclassified into a new genus, Elizabethkingia, based on 16S rRNA gene sequencing.3,16,18 The third pathogenic species, E anopheles, was isolated from the midgut of the Anopheles gambiae mosquito in Gambia, Africa in 2011;6,14,18,22 however, no causal link has been established between the mosquito as a vector and transmission of E anophelis to humans. 6
There are currently 7 species in the Elizabethkingia genus: E meningoseptica, miricola, anophelis, bruuniana, ursingii, argenteiflava, and occulta,2,18,22 the first 3 of which (meningoseptica, miricola, and anophelis) constitute the 3 medically important and pathogenic species.8,22,26 E meningoseptica infection is most often associated with pneumonia, septicemia, and meningitis in immunocompromised people and neonates. E anophelis has been associated with similar infections including pneumonia, meningitis, and septicemia, with high mortality rates, 8 and was associated with the largest outbreak of Elizabethkingia infection, occurring in the Midwest in 2015–2016, in which 65 individuals in Wisconsin, Illinois, and Michigan were affected, with 20 reported deaths.6,18 E miricola sporadically causes pneumonia, sepsis, and other infections in people, and has been increasing in incidence worldwide.4,18
These 3 pathogenic species cannot be differentiated based on phenotypic differences and have been prone to misdiagnosis as contaminants or misidentification due to lack of sensitive systems in clinical settings to detect species differences.11,18,22 While E meningoseptica has been historically the most common isolate in human infections, problems with identification in clinical settings may have previously underestimated the incidence of infections by other isolates. In fact, E miricola has been frequently misidentified in the past as E meningoseptica and was later correctly identified through 16S rRNA sequencing.12,18,22
Reports of life-threatening Elizabethkingia infections in humans with high case-fatality rates have increased dramatically in recent years across the globe.1,4 Generally associated with some degree of immunocompromise or comorbidity, Elizabethkingia is an opportunistic infection, typically affecting older individuals, and most cases involve preexisting disease such as cancer, diabetes mellitus, renal insufficiency, liver cirrhosis, or cystic fibrosis, or long-term corticosteroid or chemotherapy treatment or chronic indwelling catheter placement.5,7,8,19,22 In addition, it has been increasingly shown that hospitalization in critical care units and mechanical ventilation are risk factors for acquiring Elizabethkingia spp. infections, as the bacteria has been isolated from washbasins and hospital reservoirs, and may be transmitted from health care workers to patients as a source of nosocomial infection.1,2,4,5,12,13 The first case of E miricola identified in humans was in a patient with mantle cell lymphoma who was undergoing stem cell transplantation and on mechanical ventilation.1,3,7,8,13,14,21 Other infections which have been reported include urinary tract infection,1,11 bacteremia and sepsis,9,21,23 pneumonia,13,17,21,26 pulmonary abscesses, 9 intracranial infection, 8 septic arthritis, 3 and oral infections. 26
Elizabethkingia spp., including E miricola, are multidrug resistant. Within this genus, approximately 40 resistance genes have been found, with almost half involved in resistance to beta-lactams. 26 Whole genome sequencing of E miricola revealed 27 putative resistance genes and 38 predicted virulence-associated genes, with 2 novel metallo-β-lactamase genes (blaBloB-16 and blaBog-19) in an isolate from an amphibian. 15 In addition to beta-lactams, Elizabethkingia have been found to be resistant to colistin and aminoglycosides, but susceptible to fluroquinolones, piperacillin-tazobactam, and trimethoprim sulfamethoxazole. 5 Similar multidrug resistance profiles were observed in the current case series. Due to the inherent resistant nature of this pathogen to many antimicrobials, treatment options for Elizabethkingia infections are limited and difficult. In addition, Hu et al 15 showed through comparative genomic analysis that human and anuran E miricola strains share similar virulence-associated gene profiles. In their study, all tested amphibian isolates clustered with one human E miricola isolate in the phylogenetic analysis. Although zoonotic spread of E miricola from anurans to humans has not been definitively recognized in the clinical setting, these data suggest that E miricola may be a potential zoonotic pathogen. This challenge highlights the importance of not only considering E miricola as potential zoonotic bacteria and reservoir of multidrug resistant genes, 15 but also the need for accurate testing diagnostic platforms and species identification in optimizing the best treatment against these bacteria.
E miricola is a highly contagious, emerging pathogen of anurans, affecting multiple species, and is very difficult to treat due to multidrug resistance. Within the past several decades, reports of Elizabethkingia infections within anuran populations across the world have been increasing, most of which are reported in natural settings, or in farmed or wild captive species. E miricola was first reported as the cause of a large outbreak of meningitis in cultured black-spotted frogs (Pelophylax nigromaculatus) in China in 2016, 14 and since has been reported as a cause of systemic disease in northern leopard frogs (Rana pipiens), Chapa bug-eyed frogs (Theloderma bicolor), Vietnamese warty toads (Bombina microdeladigitora), Sabana Surinam toads (Pipa parva), and Chinese spiny frogs (Quasipaa spinosa).17,25 F meningosepticum (now E meningoseptica) was isolated during a fatal outbreak in African clawed frogs (Xenopus laevis). 10 Infections with Elizabethkingia spp. in anurans are typified by septicemia, manifesting clinically as anorexia, depression, stupor, abdominal swelling, torticollis, and incoordination. Grossly, animals may show peripheral edema, ascites, ileus, petechial hemorrhage in the skin and musculature, cutaneous ulceration, ocular opacity, hepatomegaly, renomegaly, and splenomegaly.14,17,20,25 Similar to the animals in this study, microscopic lesions have been characterized by otitis interna, hepatitis, pneumonia, myocarditis, nephritis, and meningitis, as well as endophthalmitis, keratitis, and enteritis,17,25 associated with mixed to granulomatous inflammation and the presence of gram-negative rods within inflammatory lesions.10,17,20
While infection in humans is generally opportunistic and associated with some degree of immune compromise, this is less clear in anurans. In this study, E miricola was not isolated from healthy animals or the water environment. However, it is well known that environmental stressors can have a significant impact on the immune status of anurans. 24 As such, opportunistic infections are a common cause of morbidity and mortality in aquatic species in the laboratory setting, and a significant cause of confounding in animal studies. For a number of opportunistic pathogens, under normal conditions animals remain healthy, but when stressed by overcrowding, poor environmental conditions, or concurrent disease, animals succumb to rapid systemic disease due to a weakened immune system. 20 Numerous gram-negative opportunists make up normal flora of the skin of frogs, including Chryseobacterium, Aeromonas, Flavobacterium, Klebsiella, Citrobacter, and Proteus, among others, and may cause secondary infections depending on stress, environmental factors, diet, and overall immune health. F meningosepticum, now classified as E meningoseptica, is a reported opportunistic pathogen in anurans; while the opportunistic nature of E miricola is unclear, it is speculative that this organism plays a role as an opportunist.
In the current case series, it is considered that the source of infection likely originated from the water in the original transport containers, as initial deaths occurred during shipment and shortly after arrival. Additional mortality was likely a result of disease progression secondary to stress following their arrival to the receiving laboratory, including significant impacts from heat treatments and concurrent elevations of nitrogenous wastes in tank water. In addition, diseased animals arrived from 2 separate vendors. It is common practice for different vendors to obtain frogs from a common original source farm, which may mean that the origin of the contamination may have been at the source farm level. Animals from both vendors were housed in separate rooms, and there was no shared water source or equipment between the two populations. While the possibility of contamination between populations through handling by personnel cannot be ruled out, this is very unlikely as personal protective equipment (ie, gloves) is changed between rooms. Animals from vendor C obtained near the end of the initial outbreak did not develop clinical illness, suggesting that either the source water from vendor C was not contaminated or that the opportunistic infection ran its course and ultimately ended once animals were sufficiently adjusted to their new environment (Table 1). Vendor C animals were also larger in size and may have therefore been more resistant to opportunistic infection.
This is the first documented case of large-scale mortality due to infection by E miricola in a laboratory anuran species. One previous report documents F meningosepticum in African clawed frogs (X laevis), 10 a commonly utilized laboratory amphibian species, with similar clinical, gross, and histologic findings. However, similar to the situation in humans, difficulties in species identification of clinical isolates may historically have underrepresented the incidence of E miricola in favor of the diagnosis of E meningoseptica (formerly F meningosepticum). It is possible that the isolate from African clawed frogs (among many others previously identified as E meningoseptica) may have in fact been E miricola, and therefore the true overall incidence of this disease in anurans may be unrecognized.14,25
Conclusion
E miricola has been previously reported as a potential opportunistic pathogen in wild anuran populations and has not previously been observed in the laboratory setting. This report emphasizes the need to recognize E miricola as a potential differential diagnosis early in large-scale mortality events in laboratory anurans to minimize animal loss and prevent compromise of research data. In addition, it is important to better understand the true zoonotic potential of this organism, since it could should it pose a risk to laboratory personnel with comorbidities or impaired immune function. Laboratory personnel must be aware of this and other potential zoonoses involving anuran species and wear the proper personal protective equipment when handling animals, changing water, feeding, or otherwise interacting with such species, to prevent potential spread of disease both between aquaria and susceptible individuals.
Footnotes
Acknowledgements
The authors thank the members of the In Vivo Animal Core (IVAC) at the University of Michigan Medical School for their technical expertise, and the Timothy James Laboratory in the Department of Ecology and Evolutionary Biology at the University of Michigan for clinical support and assistance with sampling.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
