Abstract
Odontomas are variably differentiated, hamartoma-like proliferations of odontogenic epithelium, pulp ectomesenchyme (odontoblasts), and dental matrix. Frogs are polyphyodont and homodont. Their teeth also differ from mammals in that they are restricted to the upper jaw in adults and lack a periodontal ligament and cementum, attaching directly to the underlying bone. Odontomas were identified in an African clawed frog (Xenopus laevis), a false tomato frog (Dyscophus guineti), and a tomato frog of unknown species (Dyscophus sp.). All of the examined odontomas were composed of numerous tooth-like structures comprising an arc of dentinal matrix lined on the convex surface by ameloblasts and on the concave surface by odontoblasts. Masson’s trichrome and immunohistochemistry with pan-cytokeratin supported these findings. The pathogenesis of these lesions may be displacement of the dental lamina, which has been shown in research studies to lead to de novo proliferation of dental elements in frogs.
Odontomas are multilineage, well-differentiated tumors or tumor-like malformations (hamartomas) that are uncommon in domestic species. They have been historically classified as either complex or compound based on the degree of organization of the dental tissues. With that said, most odontomas in domestic species have both compound and complex features. Complex odontomas are disorganized and do not form recognizable tooth-like structures, while compound odontomas are more organized and form numerous small tooth-like structures called denticles or prototeeth. 9 Compound odontomas are most commonly seen in young dogs but also are reported in cattle, horses, cats, primates, a white-tailed deer (Odocoileus virginianus), a Prevost’s squirrel (Callosciurus prevostii), a vole (Cricetidae), an African elephant (Loxodonta africana), and many species of fish. 3,6,9,10,12 Odontomas are especially common in angelfish and have been previously called “lip fibromas” due to the large amount collagenous tissue associated with the lesion. 6 The pathogenesis of odontomas is unknown, and although locally destructive and often challenging to surgically remove in domestic animals, they are associated with a favorable outcome after complete surgical excision along with aggressive curettage. 9
Adult frogs have teeth on the upper jaw but lack teeth on the lower jaw; true teeth are absent in tadpoles. 2,11 Their teeth are generally similar to those of other vertebrates with a few differences, including a lack of a periodontal ligament and cementum. Teeth of all anurans (frogs and toads) are homodont (ie, all teeth have a uniform morphology); most anurans are bicuspid, except for pipids (primitive, tongueless frogs), which are monocuspid. Teeth are continuously produced in anurans (polyphyodont). The mineralized matrix of adult teeth in amphibians has 2 distinct zones: a basal pedicel and a distal crown. These zones are separated by a well-demarcated transverse zone called the dividing zone. The dividing zone is weak, poorly mineralized, and composed of a ring of dentin and cementum-like matrix, and it allows the crown to break off of the tooth without damaging the underlying bone, to which the teeth directly attach (acrodont dentition). The dividing zone and pedicel are composed of atubular dentin. The pedicel attaches to the subjacent bone by bony attachment tissue (sometimes called “attachment bone”) secreted by osteoblasts. All amphibians have a pulp cavity surrounded by a dentinal cone. The pulp cavity is similar to that in mammals—it is composed of loose connective tissue and blood vessels, and it is lined by a layer of odontoblasts that produce dentin. Pipids (family Pipidae), including African clawed frogs (Xenopus laevis), differ from other anuran taxa in that the teeth lack a pedicellate dividing zone. In adult pipids, only a small portion of the tooth (approximately 50 microns) projects above the epithelium and into the oral cavity. Tomato frogs (Dyscophus sp.) have similar tooth morphology to all other anurans. 2
Although there are multiple, thorough reviews of amphibian neoplasia, 4,13 as well as a recent review of amphibian disease, 11 there are no reports of odontogenic lesions in frogs, to our knowledge. This report describes histologic findings of odontomas in 3 frogs.
The Northwest ZooPath (Monroe, WA) archives were searched for the terms odontogenic, ameloblastoma, odontoma, and epulis. Three odontomas were identified in amphibians. Slides had been prepared prior to this study using standard histologic techniques, specifically fixation in 10% neutral buffered formalin, sectioning at 5 microns, and staining with hematoxylin and eosin (HE); decalcification was not performed. Additional case information, including clinical history, radiographs, and gross photographs, was requested from clinicians. Control slides and formalin-fixed, paraffin-embedded blocks of the maxilla of X. laevis and Dyscophus sp. (which were prepared using aforementioned methods with the addition of decalcification in 5% trichloroacetic acid for 24–48 hours) were obtained from the Smithsonian National Zoological Park archives.
Immunohistochemistry (IHC) for AE1/AE3 and Masson’s trichrome stain (Artisan Masson’s Trichrome Stain Kit; Dako, Santa Clara, CA) were performed (Walter Reed National Military Medical Center Laboratory) on 4-µm sections of paraffin-embedded tissues from all control tissues and cases. Primary antibody applied to each section was anti-pancytokeratin (AE1/AE3) (760-2135, predilute, mouse monoclonal; Ventana Medical Systems, Oro Valley, AZ). Sections were counterstained in Mayer’s hematoxylin. Sections of control tissues from Smithsonian National Zoo, treated in the same manner, were used as positive controls for Masson’s trichrome and AE1/AE3, while phosphate-buffered saline replaced the primary antibody for IHC-negative controls. The AE1/AE3 antibody was validated in mammalian, not amphibian, species. An amphibian-validated pancytokeratin antibody was not available.
Case No. 1 was a captive-bred African clawed frog (X. laevis) of unknown age and sex from California that was euthanized due to an oral mass extending from the gingiva. Mandible, kidney, liver, adrenal gland, spleen, intestines, testicle, lung, and heart were examined histologically, and the only substantial finding was the oral mass in the gingiva. Case No. 2 was a captive, 5-year-old tomato frog (Dyscophus sp.) of unknown sex from Florida that had a biopsy performed of a mass described as being on the upper lip and extending into the oral cavity; follow-up was not available. Case No. 3 was a captive, false tomato frog (D. guineti) of unknown age and sex from Texas that had a biopsy performed of an oral mass that was noted during treatment for hypovitaminosis A; follow-up was not available.
All oral masses had histologically similar features. The oral mucosa was subtended by an unencapsulated, densely cellular proliferation composed of numerous small tooth-like formations (denticles) comprising 3 elements: arcs of dentinal matrix, palisading odontogenic epithelium (ameloblasts), and proliferative mesenchymal cells (odontoblasts) (Figs.1,2). Dentin was a brightly eosinophilic, slightly fibrillar matrix and frequently arranged in arcs. The concave surface of the dentinal arcs was basally palisaded by proliferative spindloid mesenchymal cells that formed loosely arranged aggregates with long, thin, spinous, cytoplasmic processes and small, condensed, oval nuclei (pulp ectomesenchyme, recapitulating odontoblasts). The convex surface of the dentinal arcs was generally lined by a palisade of columnar epithelial cells with frequent central nuclei and basilar clearing (odontogenic epithelium, recapitulating ameloblasts). Denticles ranged from approximately 60 to 200 microns in greatest dimension. Inflammation, edema, and ulceration were minimal.
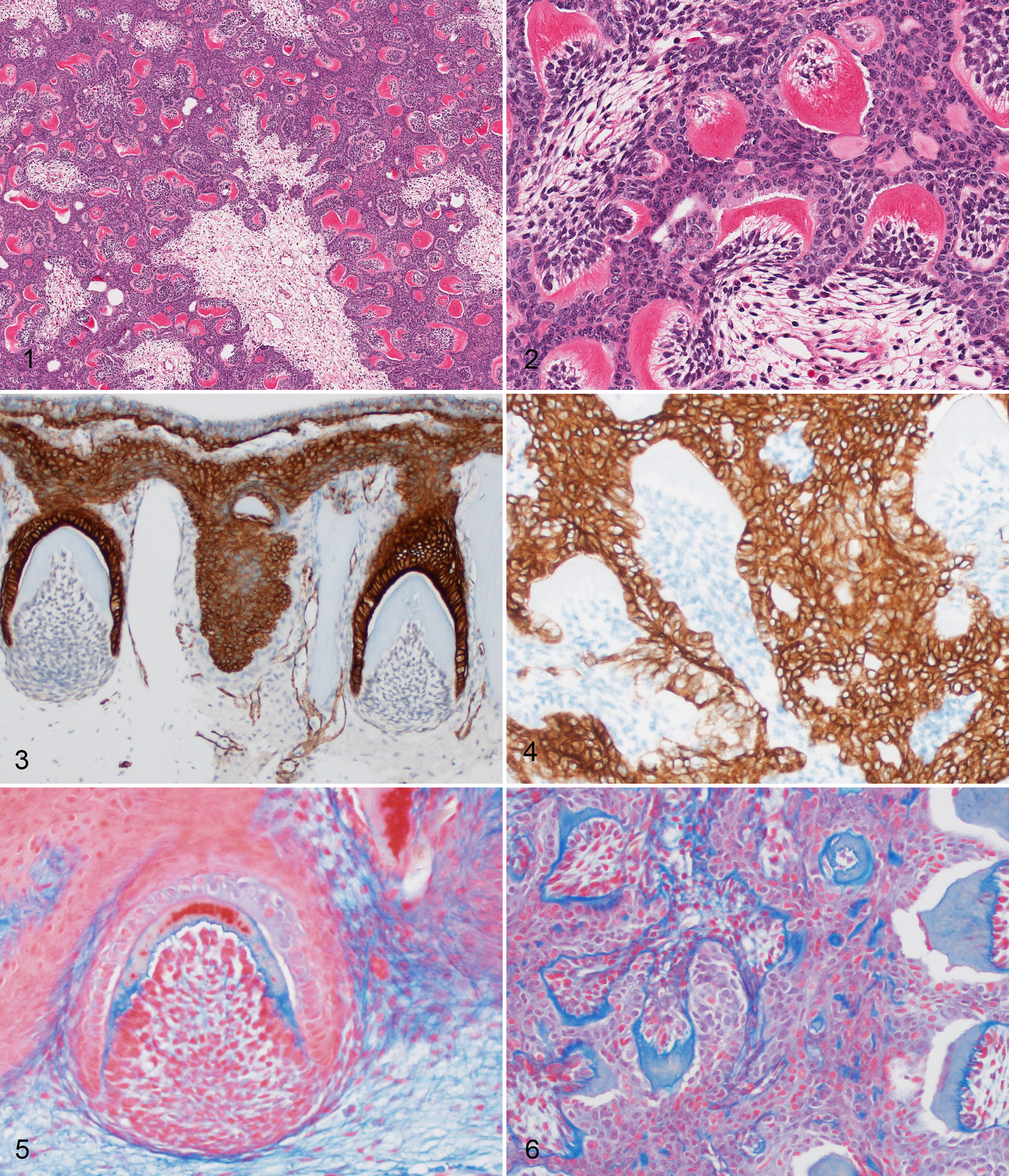
In oral tissues from the control animals, IHC with AE1/AE3 revealed strong, diffuse, cytoplasmic immunoreactivity of gingival epithelium that was contiguous with subjacent odontogenic epithelium (Fig.3). In the odontomas, proliferative cells along the convex surface of the dentinal arcs had strong, diffuse, cytoplasmic immunoreactivity to AE1/AE3. Proliferative cells on the concave surface of the dentinal arcs formed loose streams that were cytokeratin negative (pulp ectomesenchyme/odontoblasts) (Fig.4). In the control animals, in Masson’s trichrome-stained sections, the dentin was bright blue directly adjacent to odontoblasts (predentin) and pale to bright red adjacent to the ameloblasts (Fig.5). With Masson’s trichrome, the dentin in the odontoma lesions stained diffusely blue, consistent with predentin (Fig.6).
This report describes the histologic features of odontomas in 3 frogs. Each case had an oral mass extending from the gingiva and jaw. In mammals, odontomas present as firm, slow-growing, expansile masses within the mandible or maxilla of younger animals that can lead to failure of tooth eruption and/or displacement of erupted teeth. 9 In the cases described here, odontomas were limited to the maxilla as frogs lack teeth on the lower jaw. Other clinical signs reported with odontomas include facial swelling, ocular signs, and eruption of tooth-like structures within the oral cavity; these clinical signs were not noted in the animals in this report, which had a limited clinical history and either only a single biopsy with lack of clinical follow-up or euthanasia due to the oral mass. Odontomas feature variably organized, proliferative dental elements (odontogenic epithelium/ameloblasts), pulp ectomesenchyme (odontoblasts), and dental matrix (dentin with or without enamel). These elements were identified via HE-stained sections and supported by IHC and Masson’s trichrome stain with comparison of the odontomas to control tissues. Ameloblasts were identified using cytokeratin IHC and palisaded along the convex surface of the dentinal matrix in both control animals and odontomas. Ectomesenchymal-derived odontoblasts were identified by negative immunoreactivity to cytokeratin and were present along the concave surface of the dentinal matrix in both control tissues and odontomas. In the odontomas, the dental matrix stained blue with trichrome, consistent with early dentin. The orderly association between ameloblasts, interposed dentinal matrix, and odontoblasts recapitulated normal tooth formation as seen in the controls; this is diagnostic for “compound” odontoma.
To our knowledge, the odontomas in this report are the only odontogenic lesion reported in amphibians. It is possible that the lack of reported odontogenic neoplasia in amphibians may be due to the absence of periodontal ligament and cementoblasts, which are a source for odontogenic proliferative lesions. 7 It is also likely that oral masses in amphibians are less commonly biopsied than in domestic mammals.
The cause for odontomas in these frogs is unknown. One possible cause is trauma to the dental lamina, which is an invagination of the gingival epithelium that leads to tooth bud formation. The dental lamina is present throughout the life of amphibians due to polyphyodont dentition. There are multiple studies in amphibians in which dental lamina is transplanted by either autograft or allograft transplantation. In these studies, transplantation of the dental lamina leads to formation and even replication of tooth buds and/or formed teeth, even when transplanted outside of the mouth. 5,8 When dental lamina is transplanted along with sections of the jaw, tooth development continues in an orderly fashion with a regular relationship between the teeth and underlying bone. When dental lamina is transplanted without associated bone, tooth development and replication may continue, but dentinal relationships may change, resulting in alteration of tooth morphology to a more embryological appearance (eg, bicuspid to monocuspid). 1 The signaling that occurs between amphibian teeth and bone is still poorly understood. Normal tooth development (size and shape) can reportedly continue with experimental disruption or loss of underlying bone. 7
Trauma or disruption to the dental lamina was not reported in any of these animals, and the pathogenesis of odontomas in these cases is speculative. In dogs, most odontomas occur in young animals, and it is speculated that they may be a developmental hamartoma. 9
Footnotes
Acknowledgements
We thank institutions and individuals for contributing case material to this study, including individuals at Fort Worth Zoo, Jacksonville Zoo, University of California–Berkeley, and Smithsonian National Zoo. In addition, we thank Christie Buie and Cathy Minogue of Northwest ZooPath for data retrieval and Leroy Brown of Histology Consultation Services for slide preparation. Thank you to Beth Westerin-Romig for clinical consultation on this entity for these cases. Thank you to the histology staff at the Joint Pathology Center and Walter Reed National Military Medical Center for preparation of slides, IHC, and special stains, especially Warren McNeil, Stacey Tamer, Steven Mcnair, Kenenya Gathers, Ann Brown, and Andrea Cherilus.
Disclaimer
The views expressed in this manuscript are those of the authors and do not reflect the official policy of the Department of Army/Navy/Air Force, Department of Defense, or US government.
