Abstract
The morphology of 58 apocrine tumors from 31 domestic Richardson’s ground squirrels (Urocitellus richardsonii) was examined, comparing tumor subtypes with clinical information and histological findings. The squirrels included 23 males (74%) and 8 females (26%). Of the 58 tumors, 36 (62%) were located from the cervical skin to the dorsal skin, 17 (29%) were from the perioral region to the cheek, and the location was unrecorded for the remaining 5 squirrels (9%). Histologically, 49 tumors (84%) were apocrine adenocarcinomas, 6 were apocrine adenomas (10%), and 3 were apocrine cysts (5%). The 49 apocrine adenocarcinomas were subclassified as 16 (33%) cystic papillary type, 16 (33%) tubulopapillary type, 12 (24%) solid type, and 5 (10%) micropapillary invasive type. Fisher’s exact test revealed correlations between tumor subtypes and tumor diameters, nuclear atypia, tumor margins, vascular invasion, and periodic acid-Schiff (PAS) positivity. This study showed a predominance of apocrine adenocarcinomas with various histological subtypes. In addition, subclassification of these tumors was useful for predicting the biological behavior of apocrine gland tumors in Richardson’s ground squirrels.
Richardson’s ground squirrel (Urocitellus richardsonii) is a rodent species belonging to the family Sciuridae, order Rodentia, and class Mammalia. Wild Richardson’s ground squirrels are native to the prairies of southern Canada and the northern United States. Richardson’s ground squirrels are occasionally kept in captivity but are uncommon as companion animals. 9 Reports of neoplastic diseases in this species are limited. Hepatocellular carcinomas caused by infection with ground squirrel hepatitis virus have been reported in wild cases.11,16 In addition, cheek salivary gland adenocarcinoma and mast cell tumors have been reported in captive squirrels.5,17
Ground squirrels are known to have integumentary scent glands located from the perioral region to the cheek (“oral scent glands”), from the cervical skin to dorsal skin (“dorsal glands”), and in the anal region.2,8 Oral and dorsal scent glands are composed of predominantly apocrine epithelial cells. Marking behaviors such as rubbing the perioral and dorsal areas against the ground or rocks (“twist-marking”) and rubbing the corners of the mouth against each other (“greeting”) play important roles in the social development of Richardson’s ground squirrels. 8 In 2018, an apocrine ductal carcinoma originating from the oral scent gland was reported for the first time in this species. 2 However, pathological information about tumors originating from these glands remains limited. The goal of this study was to assess the morphological features of these tumors.
Material and Methods
Study Population
The samples examined were 58 tumors from 31 Richardson’s ground squirrels submitted to the Laboratory of Veterinary Pathology at Nihon University (9 cases including 1 necropsy case) and IDEXX Laboratories (49 cases), including 57 surgical pathology specimens removed as therapeutic interventions by clinical veterinarians from 30 live squirrels and 1 necropsy case. In 10 squirrels, 2 or more masses were removed. The tumors removed were primary in 28 squirrels, and recurrent in the remaining 3. Signalment including sex, age, and tumor location was recorded, and maximum diameters of removed tumors, which were fixed in formalin, were measured by a ruler after bisection. The maximum diameters of tumors were categorized as <1, 1–2, and >2 cm.
Histopathology
The excised tumors were fixed in 10% neutral buffered formalin. After trimming and routine processing, representative tissues were embedded in paraffin, sectioned at a thickness of 5 μm, and stained with hematoxylin and eosin and periodic acid-Schiff (PAS) stains. All cases were histologically evaluated and classified as apocrine cysts, apocrine adenomas, or apocrine adenocarcinomas according to the criteria set forth in Surgical Pathology of Tumors of Domestic Animals Volume 1. 4 Apocrine adenocarcinomas were further subclassified into histological subtypes according to the classification of mammary tumors reported by Goldschmidt et al in 2011. 3 In addition, nuclear atypia, mitoses within a 2.37-mm2 area (equivalent to ten 40× fields), presence or absence of tumor margins, vascular invasion, and PAS positivity were recorded. Nuclear atypia was categorized as mild, moderate, or prominent based on the degree of anisokaryosis and pleomorphism. Tumor margins were composed of 2–4 lateral and the ventral margins, depending on tumor size. When the neoplastic cells infiltrated into at least 1 margin, the tumor margins were recorded as “not free.” For PAS staining, a positive result for >50% of neoplastic cells was recorded as “positive.”
The animal in the necropsy case was a 3-year and 2-month-old female with a mass in each of the cheek and in the dorsal region. Surgical removal of the masses was performed, but the animal died postoperatively due to hemothorax. Autopsy was performed by a clinical veterinarian, and representative organs were submitted to the Laboratory of Veterinary Pathology at Nihon University for histopathological examination. Organs were fixed in 10% neutral buffered formalin. After the trimming of representative tissues, histologic sections were prepared, as described above.
Statistical Analyses
Associations between histological subtypes of adenocarcinoma and sex, tumor diameter, tumor location, nuclear atypia, presence or absence of tumor margins, vascular invasion, and PAS positivity were statistically tested using Fisher’s exact test. Values of P < .05 were defined as significant.
Results
Characteristics of Signalment and Gross Findings
The 31 Richardson’s ground squirrels comprised 23 (74%) males and 8 (26%) females. The 28 squirrels with primary tumors ranged in age from 1 year 3 months to 5 years 2 months (median, 3 years 7 months). The age was unknown for 2 squirrels. The median tumor diameter was 10 mm (range, 1–30 mm), and the tumor size was unrecorded in 10 cases. Of the 28 squirrels, 16 (57%) had solitary tumors and 12 (43%) had multiple tumors. Of the 58 tumors included in this study, 36 (62%) were located from the cervical skin to the dorsal skin and 17 (29%) were from the perioral region to the cheek. The location was unrecorded for 5 tumors (9%; Fig. 1).
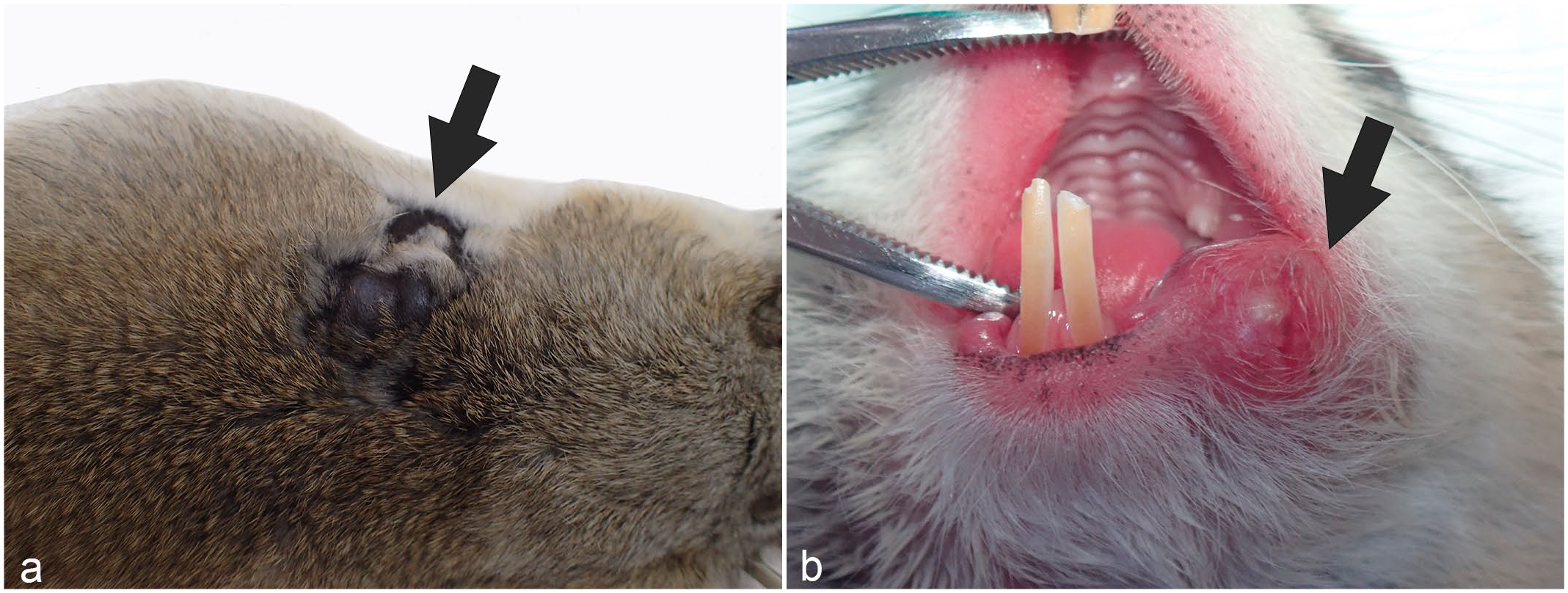
Gross appearance of apocrine tumors in Richardson’s ground squirrels. (a) Dorsal skin. A multinodular black mass is present on the back (arrow). (b) Skin at the margins of the mouth. A red mass is present at the margins of the mouth (arrow).
Histologic Features
Of the 58 tumors, 49 (84%) tumors were apocrine adenocarcinomas, 6 were apocrine adenomas (10%), and 3 were apocrine cysts (5%; Fig. 2).
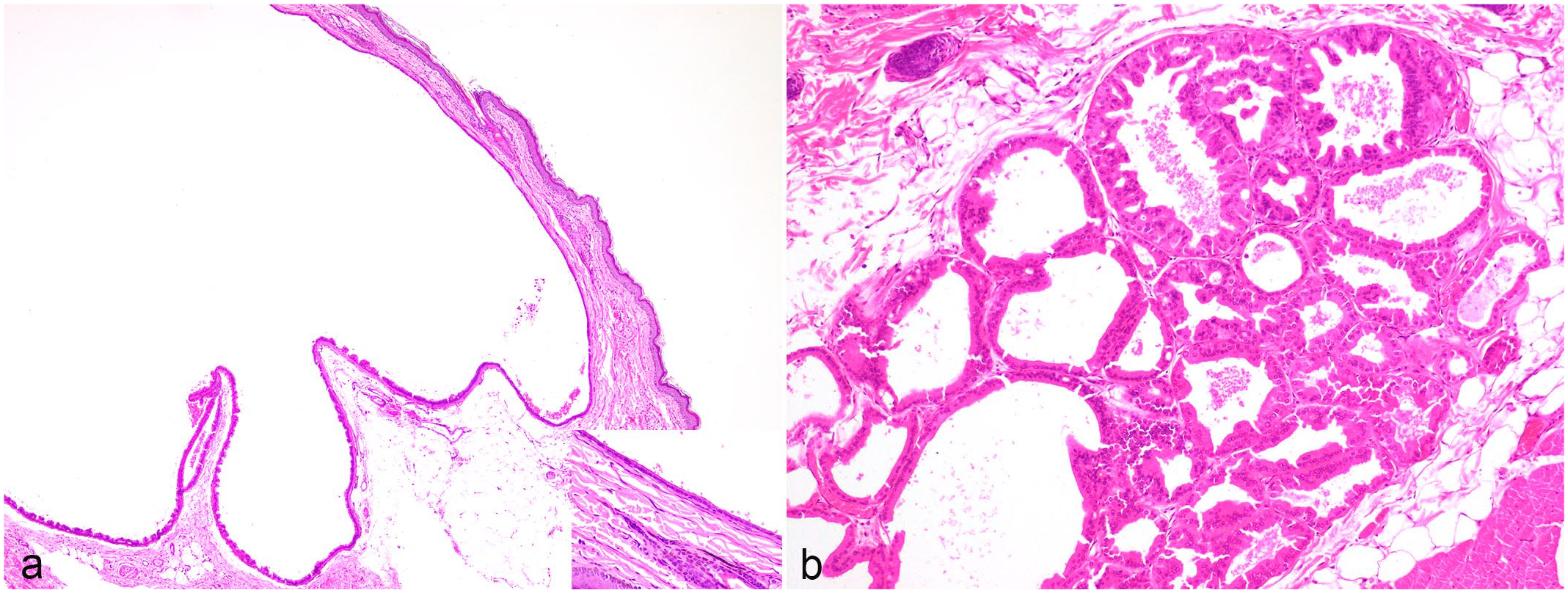
Histopathological features of apocrine tumors in Richardson’s ground squirrels. Hematoxylin and eosin. (a) Apocrine cyst, dorsal skin. The cyst is lined by a single layer of epithelial cells. (b) Apocrine adenoma, dorsal skin. The cells forming tubules are slightly pleomorphic. No mitotic figures were seen.
The apocrine adenomas formed well-demarcated, moderately cellular, expansile neoplastic masses composed of cuboidal cells arranged in glands and tubules separated by fine fibrovascular connective tissue. No mitotic figures were seen. For the apocrine cysts, the dermis and subcutis was expanded by a cyst, which was lined by a single layer of epithelial cells. The epithelium sometimes became flattened and atrophic.
The 49 apocrine adenocarcinomas were subclassified as 16 (33%) cystic papillary type, 16 (33%) tubulopapillary type, 12 (24%) solid type (24%), and 5 (10%) micropapillary invasive type. No cases of tubular, cribriform, comedocarcinoma, anaplastic, complex, or mixed types were identified.
Cystic papillary apocrine adenocarcinomas formed enlarged cysts lined by neoplastic cells with papillary protrusion into the lumen. Cuboidal cells with distinct cell borders lined the inside of the cysts and were often multilayered. The papillae were supported by fibrovascular connective tissue stroma and the lumens often contained eosinophilic granules admixed degenerate neutrophils and macrophages. Neoplastic cells sometimes had PAS-positive cytoplasmic granules (Fig. 3). The median number of mitoses per 2.37 mm2 was 2 (range, 0–29). One case had a concomitant apocrine cyst.
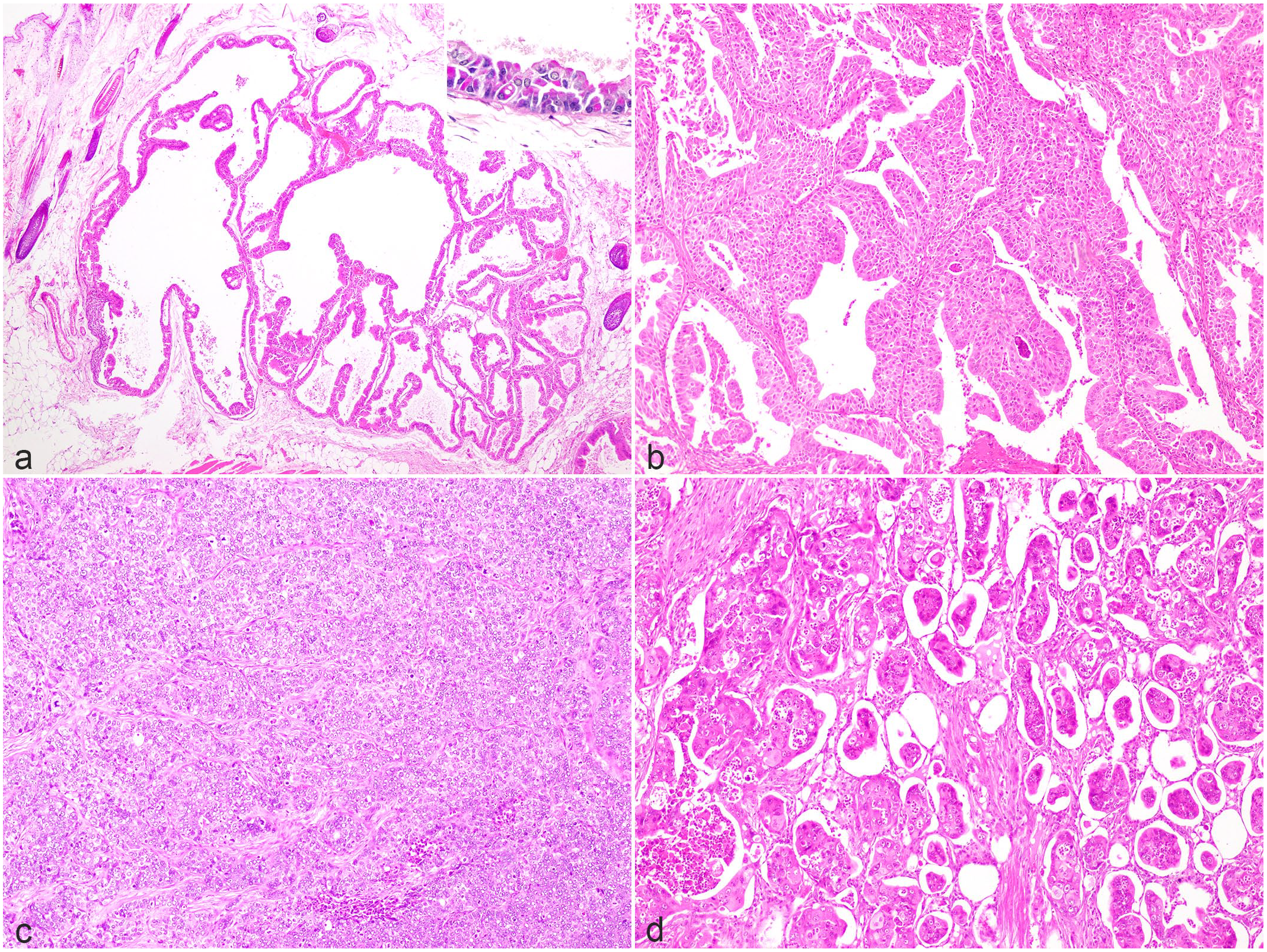
Histopathological features of apocrine adenocarcinomas on dorsal skin in Richardson’s ground squirrels. Hematoxylin and eosin. (a) Cystic papillary type. Neoplastic cells extend into the center of the cyst. Inset: Neoplastic cells contain periodic acid-Schiff (PAS)-positive material in the cytoplasm. PAS stain. (b) Tubulopapillary type. Multiple layers of neoplastic cells form papillary projections lining tubular lumina. (c) Solid type. Irregular lobules of neoplastic cells are surrounded by fine fibrous stroma. (d) Micropapillary type. Neoplastic cells are present within variably sized lumina.
Tubulopapillary apocrine adenocarcinomas were diagnosed as often as cystic papillary apocrine adenocarcinomas. In tubulopapillary apocrine adenocarcinomas, neoplastic cells formed 1–2 layers around lumens and extended into the lumen-like papillae. The lumens were formed by tubular arrays of cuboidal cells with distinct cell borders and some were multilayered. The stroma was composed of fibrous connective tissues. The median number of mitoses per 2.37 mm2 was 9.5 (range, 0–37; Fig. 3).
Solid apocrine adenocarcinomas were formed by sheets of polygonal cells without lumens, divided by fibrous connective tissues. The median number of mitoses per 2.37 mm2 was 22.5 (range, 4–52; Fig. 3).
Micropapillary invasive apocrine adenocarcinomas formed neoplastic masses composed of numerous small lumens. The neoplastic cells were polygonal with distinct borders and had prominent nuclear atypia. Vascular invasion was identified in all cases of this type. The median number of mitoses per 2.37 mm2 was 24 (range, 10–28; Fig. 3).
Recurrence
Of the total 31 surgical pathology cases, local recurrences were observed in 8 (26%) cases. Of those 8 cases, 2 cases recurred twice. The interval from the first mass removal to recurrence was known in 5 cases, with a median of 6 months (range, 4 months to 2 years 7 months). Of the 12 apocrine carcinomas that occurred in recurrent cases, 3 (25%) cases were of the cystic papillary type, 3 (25%) cases were of the tubulopapillary type, 4 cases (33%) were of the solid type, and 2 cases (17%) were of the micropapillary invasive type.
Necropsy Case
Tumors of the cheek were composed of lobules of cuboidal to polygonal cells, supported by fine fibrovascular stroma. The neoplastic cells had distinct cell borders and moderate eosinophilic cytoplasm. Nuclei were round with coarsely stippled chromatin and distinct nucleoli. Anisocytosis and anisokaryosis were prominent. In total, 28 mitoses per 2.37 mm2 were seen. Histological findings of the cheek masses were consistent with solid-type apocrine adenocarcinoma. The dorsal tumor formed papillae that extended into an enlarged cyst. Neoplastic cells were cuboidal cells with distinct cell borders and mild anisocytosis and anisokaryosis. A total of 3 mitoses were seen in an area of 2.37 mm2 (equivalent to ten 40× fields). Histological findings for this dorsal tumor were consistent with cystic papillary–type apocrine adenocarcinoma. Nodular lesions comprising neoplastic cells similar to those seen in the cheek were identified in the lungs. Lymphatic vessels surrounding the cheek mass showed evidence of infiltrating tumor cells, which were presumed to have metastasized via lymphatics to the lungs (Fig. 4). Other significant lesions included myocardial hypertrophy, centrilobular hepatocellular degeneration, lymphoplasmacytic interstitial nephritis, hemorrhagic cystitis, and cystic endometrial hyperplasia.
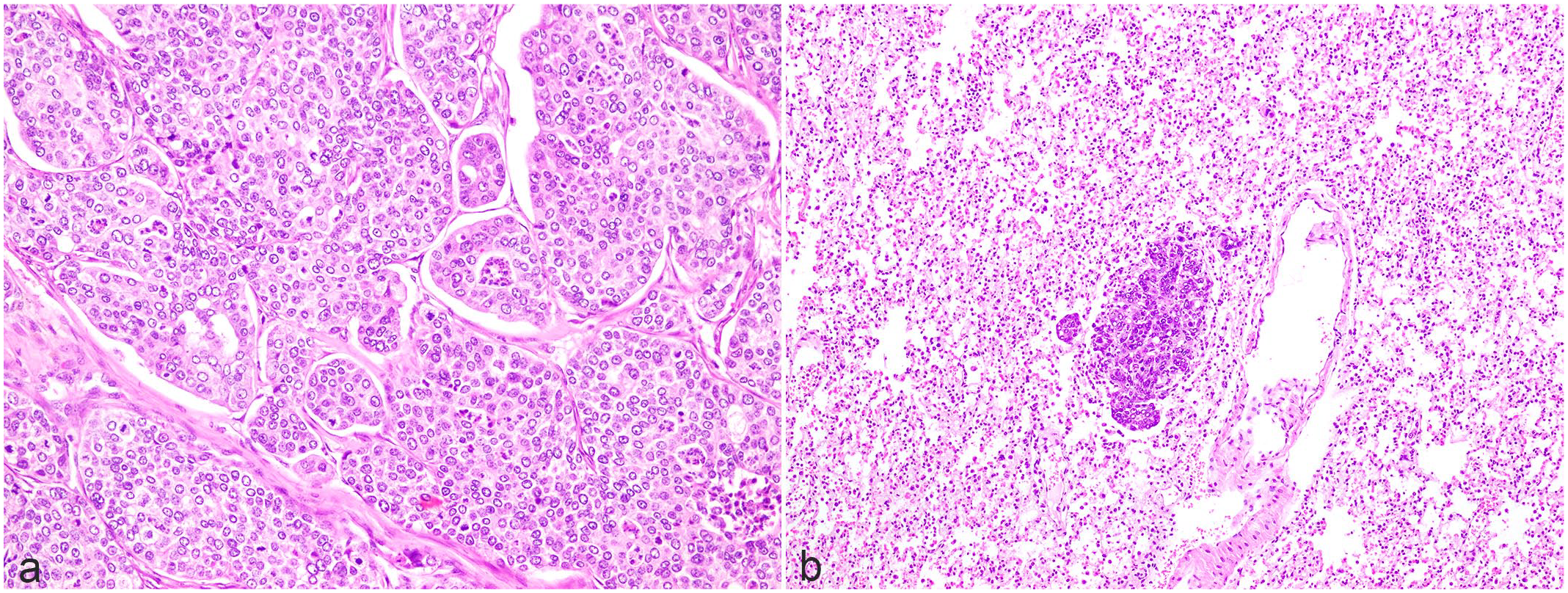
Histopathology of apocrine adenocarcinoma in a Richardson’s ground squirrel, necropsy case. Hematoxylin and eosin. (a) Mass from the cheek skin. Neoplastic cells in lobular arrangement. (b) Lung metastasis.
Statistical Analyses
Morphological features showing correlations with histological subtypes are summarized in Table 1. Statistical analyses showed clear correlations between tumor subtypes and tumor diameter (Fisher’s exact test; P = .01964, 41 cases per group), nuclear atypia (Fisher’s exact test; P = .003644, 49 cases per group), tumor margins (Fisher’s exact test; P = .02974, 49 cases per group), vascular invasion (Fisher’s exact test; P = .007077, 49 cases per group), and PAS positivity (Fisher’s exact test; P = .01602, 49 cases per group). Cystic papillary–type adenocarcinomas tended to be the smallest and micropapillary invasive–type adenocarcinomas tended to be the largest. Nuclear atypia was mild in cystic papillary–type adenocarcinoma and prominent in solid-type and micropapillary invasive–type adenocarcinoma. Furthermore, neoplastic cells in cystic papillary–type tumors showed more PAS-positive cytoplasm than those in other types. Solid-type and micropapillary invasive–type tumors showed a greater frequency of neoplastic cell infiltration into the resection margins. With regard to intravascular invasion, cystic papillary–type tumors showed a low tendency to invade vessels compared with other types, while micropapillary invasive types showed a high tendency to invade.
Histologic findings associated with subtypes of apocrine adenocarcinomas in Richardson’s ground squirrels.
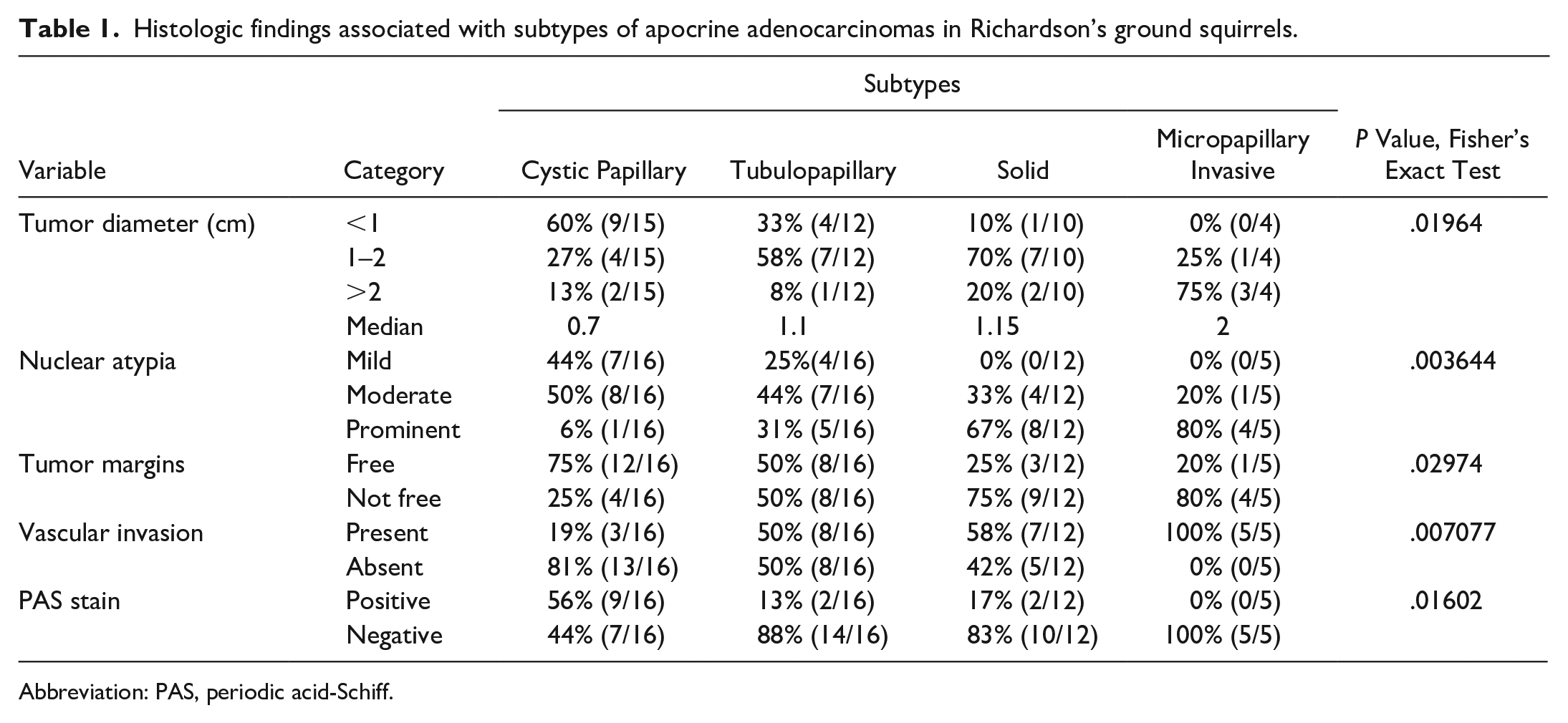
Abbreviation: PAS, periodic acid-Schiff.
Discussion
Although the head, inguinal, and axillary areas are the most frequent sites of apocrine adenocarcinoma in dogs and cats, this study did not identify tumor occurrence in these areas in Richardson’s ground squirrels. 10 This difference was attributed to differences in anatomical positions of the scent glands. Richardson’s ground squirrels have peculiar scent glands located from the perioral region to the cheek, from the cervical skin to the dorsal skin, and in the anal area. 8 The dorsal scent gland field extends from the scapular region to the cranial and caudal areas, reaching the periauricular area in the cranial direction.7,8 The oral scent gland is located at the oral angle and extends about 2.5–3 mm posteriorly. 8 In both scent glands, the glandular ducts lined by apocrine epithelia terminate in a collecting sinus and a short duct originating from this sinus opens onto the free surface of the skin. 8 A previously reported apocrine ductal adenocarcinoma had been suggested to arise from these scent glands. 2 However, no masses arising from anal areas were seen in this study and the locations of the tumors were disproportionately observed in dorsal and perioral skin. This was presumably due to anatomical differences as the oral and dorsal scent glands consist mainly of apocrine epithelium cells, and scent glands of the anal area consist of both apocrine and sebaceous epithelial cells.7,14 In addition, functional differences may exist in relation to the fact that oral and dorsal scent glands are frequently used by the animal for marking, for individual and group recognition purposes, whereas scent glands in the anal area are used for vigilance and marking behavior in males.7,15 Cases were more frequent among males than among females. Because androgen-dependent tumors originating from ventral scent glands are common in male gerbils—another rodent species—the possibility must be taken into account that sex hormones are related to tumor development in ground squirrels. 1 In fact, in Columbian ground squirrels (Urocitellus columbianus), which belong to the same family as Richardson’s ground squirrels, males have been reported to mark much more frequently using the oral and dorsal scent glands than females, suggesting a relationship between sex and the frequency of tumor development. 7 As a future endeavor, investigation into the expression of sex hormone receptors in tumors may be worthwhile.
Because malignant neoplasms were more common diagnoses in this study than apocrine adenoma or apocrine cyst, strong suspicion of the possibility of malignancy is important for skin masses that form from the perioral region to the cheek or from the cervical skin to the dorsal skin. Apocrine adenocarcinoma is a malignant neoplasm that originates from the apocrine secretory epithelium and is known to have various morphologies, including tubular, cystic, and solid. In addition to a desmoplastic reaction, lymphovascular invasion and metastasis may occur. 4
Cystic papillary apocrine adenocarcinomas were comparatively more likely to be PAS positive, and nuclear atypia was also more likely to be mild. Cystic papillary apocrine adenocarcinomas were therefore considered to form malignant neoplasms with apocrine secretory capacities. This type has been considered to display the best prognosis with less invasive behavior at tumor margins. In solid-type and micropapillary invasive–type apocrine adenocarcinomas, more cases showed prominent nuclear atypia and frequent mitoses compared with cystic papillary and tubulopapillary types. In addition, vascular invasion was identified in more cases. With regard to the canine mammary carcinomas referenced, retrospective studies of the prognostic value of the 2011 classification reported a correlation between the 2011 classification and histologically observed lymphovascular invasion.6,12,13 This study showed a difference between tumor subtypes and the presence of lymphovascular invasion. In addition, the solid and micropapillary invasive types had incomplete excisions. Therefore, these data suggest that solid and micropapillary invasive apocrine adenocarcinomas should be followed up with caution, as they appear to have more aggressive behavior.
Given the limited duration of follow-up in this study, whether tumor types and atypia were associated with recurrence or metastasis was unclear. However, because many cases showed vascular invasion and the necropsy case displayed metastasis to the lungs, such an association might be likely depending on the tumor morphology. Tumor subtypes were presumably unrelated to recurrence because various tumor subtypes showed local recurrence in this study. In conclusion, this study examined apocrine tumors originating from the peculiar scent glands of Richardson’s ground squirrels, revealing sex differences, predominant sites, and morphological features. In addition, we also demonstrated various histological subtypes and the correlations between histological subtype and features suggestive of aggressive tumor behavior.
Footnotes
Acknowledgements
We would like to thank IDEXX Laboratories for providing tissue samples. We would also like to thank Dr Iori Koizumi from Koizumi Nest Animal Hospital and Toshiro Ito from Iris Animal Hospital for providing the macrographs.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
