Abstract
Harderian gland neoplasms were identified in 18 aged, adult Beechey ground squirrels (Spermophilus beecheyi) from the records of 167 wild-caught captive animals that were necropsied. All but one animal had tumors that were classified as carcinomas, with infiltrative growth and frequent metastases. This is the first detailed report of Harderian gland neoplasia in wild Sciuridae, although this neoplasm has been described in other rodent species. Clinically, affected ground squirrels typically were inappetent and presented with weight loss and exophthalmos. The biologic behavior of Harderian gland neoplasia is variable among rodent species; in Beechey ground squirrels there was a high incidence of malignant behavior. Eleven of 17 tumor-bearing animals for which the gender was known were male, and 6 were female. Nine of 16 for which data were available were uninfected, and 7 had evidence of current or prior infection with ground squirrel hepatitis virus. Tumor development occurred in older animals; all but 2 were 5.5 years of age or older. The presence of metastasis was not related to gender or chronic ground squirrel hepatitis virus infection.
Introduction
The Harderian gland is a bilobed, tubuloalveolar accessory lacrimal gland, found in many vertebrate groups such as amphibians, reptiles, birds, and mammals, including various species of laboratory rodents. The gland is located within the medial part of the orbit, and it secretes onto the surface of the nictitating membrane. 1 The mammalian Harderian gland secretion is primarily composed of lipids; however, in rodents, the gland also produces porphyrins and inoleamines. 1 The function of the gland is poorly understood; while lubrication of the nictitans and cornea is considered to represent the primary purpose of the gland, other proposed roles of the gland and its secretions in mammals include photoprotective effects of the porphyrins, 1 social signaling through pheromones, 2, 3 thermoregulation through contribution to pelage lipids, 1, 4 and coordination with the pineal-retinal axis through synthesis of serotonin and melatonin. 1, 5
Neoplasms of the Harderian gland have been described in several rodent species, including the mouse, 6 rat, 7, 8 hamster, 9 and vole; 10 and a single case report in the ground squirrel. 11 Neoplasms may arise spontaneously or as a result of exposure to a variety of chemical carcinogens. 12, 13 Since the gland is found in the retrobulbar space, the growth of these tumors often produces protrusion of the associated globe, termed exophthalmos, in affected animals.
The Beechey ground squirrel (Spermophilus beecheyi) has been studied as a model of human hepatitis B virus (HBV) infection because of the occurrence of a natural infection with ground squirrel hepatitis virus (GSHV), a member of the hepadnavirus family along with HBV and woodchuck hepatitis virus. 14, 15 Like chronic HBV infection, which carries a significant risk of hepatocellular carcinoma in humans, 16 GSHV infection is also associated with liver tumor development in chronically infected ground squirrels. 17 Since chronic GSHV infection was a risk factor for tumor formation in one gland, the liver, and other hepatitis viruses such as woodchuck hepatitis virus can replicate in extrahepatic tissues such as the pancreas and lymphoid organs, 18 we evaluated the association between chronic infection and tumor development in the Harderian gland as well. Although GSHV replication appears to be primarily limited to hepatocytes, the presence of virus in the Harderian gland has not been evaluated. 19
Previously, there has been 1 report, limited to the gross appearance of spontaneous Harderian gland neoplasia, in 4 ground squirrels from this captive colony, and these animals are included in this report. 11 This paper describes the incidence, biologic behavior, and histomorphology of Harderian gland tumors in a colony of wild-caught, naturally GSHV-infected ground squirrels maintained in captivity.
Materials and Methods
Necropsy reports and histologic tissue sections of from postmortem examinations of adult ground squirrels found to have Harderian gland neoplasms were retrospectively reviewed. The ground squirrels were part of a colony of animals trapped in the San Francisco Peninsula area of California in an ongoing GSHV study at Stanford University. Necropsies were performed on animals that were moribund, found dead, or those euthanatized as part of various study protocols. None of the animals bred successfully in captivity, so the colony was composed only of trapped animals. Animals were generally estimated to be 1 or 2 years of age at the time they were captured, based on patterns of tooth wear, unless they were juvenile. 15 Because these animals only breed once a year the ages could be estimated with reasonable accuracy. The average natural life expectancy of the animals in the colony was not known because of periodic euthanasia for experimental studies and a lack of sufficient records.
All animals were maintained and treated in accordance with the contemporary Animal Care and Use Committee guidelines of Stanford University.
Tissue sections had been routinely prepared and stained with hematoxylin and eosin (HE).
Diagnostic criteria for classification of Harderian gland neoplasms were based on established criteria for laboratory rodents. 20
The presence of GSHV surface antigen (GSHsAg) or antibodies (anti-GSHsAg) in serum was evaluated using cross-reacting solid-phase commercial radioimmunoassay kits for HBV surface antigens or related antibodies (Ausria II, Abbott Labs, Chicago IL). 17
Because data were not available on animal numbers, gender, and virus status from the entire colony over the time that tumors were identified, it was not possible to statistically assess the incidence data for the Harderian glands in relation the gender and GSHV-infection status, but the frequency of metastasis in the different groups was evaluated using a 2-tailed Fischer's exact test. 21
Results
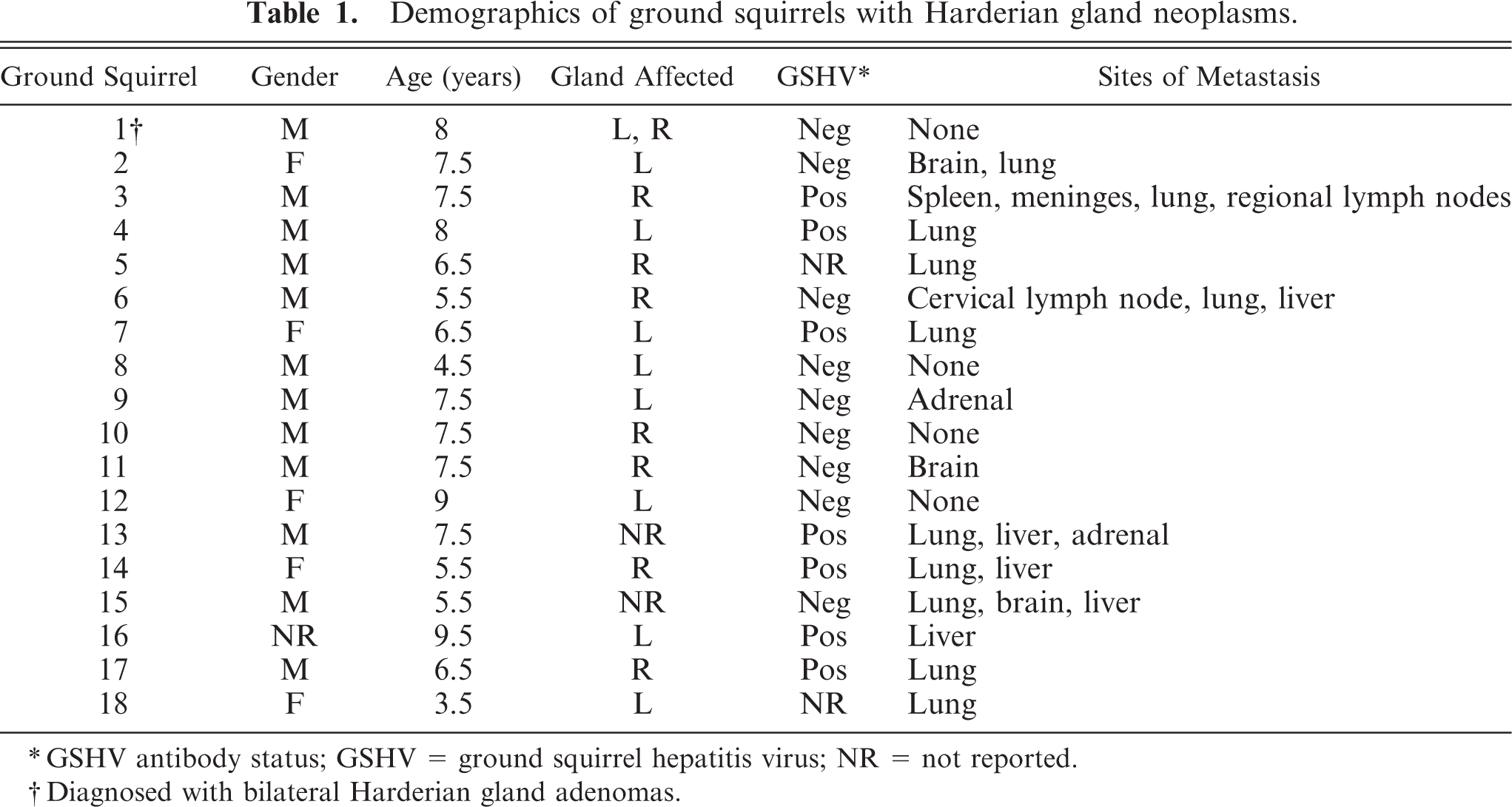
Harderian gland neoplasms were detected in 18 out of 167 (10.8%) necropsies performed over a period from 1988 to 1997 (Table 1). Clinically, affected animals frequently presented inappetent and lethargic, often with weight loss. Occasional cases were found dead without premonitory clinical signs. Four necropsy reports specifically report protruding eyes, termed exophthalmos, with white to tan nodular retrobulbar soft tissue masses evident at gross necropsy (Fig. 1). Because of exophthalmos, enucleations had been performed 1 to 3 months prior to euthanasia in 2 animals. Affected animals were classified as adult to geriatric, ranging from 3.5 to 9.5 years of age (mean = 6.9 years of age). Both males (n = 12) and females (n = 5) developed tumors (gender was not specified in 1 case). Right and left Harderian glands were involved with approximately equal frequency. Of these cases, 7 of 16 (44%) ground squirrels had evidence of current or prior infection based on presence of antibody to GSHV or the presence of GSHV viral proteins in the blood, and 9 of 16 (56%) were negative. GSHV antibody status was not available for 2 animals. It was not possible to determine a gender-related or virus status–related tumor incidence because the data on overall colony gender and virus status were not available.
Demographics of ground squirrels with Harderian gland neoplasms.
GSHV antibody status; GSHV = ground squirrel hepatitis virus; NR = not reported.
Diagnosed with bilateral Harderian gland adenomas.
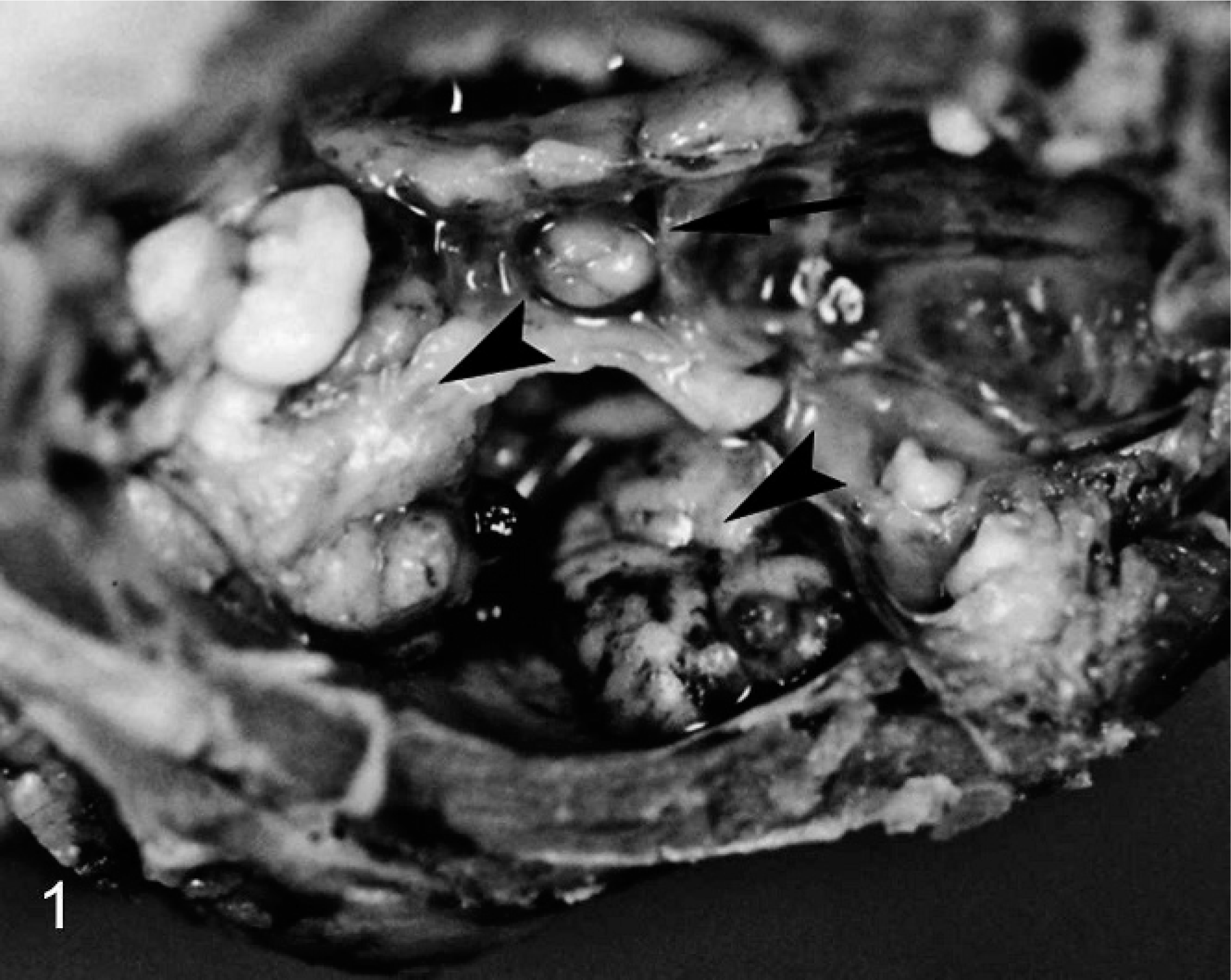
Adult Beechey ground squirrel with calvarium removed. A multilobulated, pale soft tissue mass is present in the retrobulbar space and protruding into the braincase (arrowheads). The pituitary is indicated for orientation (arrow).
Harderian gland tumors were all of epithelial origin. Seventeen of 18 cases were classified as adenocarcinomas. Adenocarcinomas were typically unencapsulated and poorly demarcated, forming large retrobulbar masses that infiltrated into the adjacent retrobulbar and periocular tissues (Fig. 2). Tumors were densely cellular, composed of large solid lobules and nests, anastomosing tubules and papillary structures, and irregular acini, supported by fine collagenous stroma, with multiple foci of necrosis and mineralization (Fig. 3). Neoplastic cells were cuboidal to columnar, with finely granular, amphophilic cytoplasm, and populations of large, polygonal cells with abundant, finely vacuolated cytoplasm. Nuclei were round to oval, with finely stippled chromatin, and lacked prominent nucleoli. Tumor cells exhibited moderate anisocytosis and anisokaryosis, and mitoses were generally 0 to 2 per 400× field. Variable neutrophilic to lymphoplasmacytic inflammatory infiltrates extended between neoplastic lobules. Metastases were detected in 14 (77%) of the cases; metastatic sites included lungs, liver, kidneys, adrenal glands, and brain. Metastatic foci were composed of neoplastic cells morphologically similar to the primary retrobulbar mass. The incidence of metastasis of the Harderian gland tumors was not associated with gender or with GSHV-infection status.
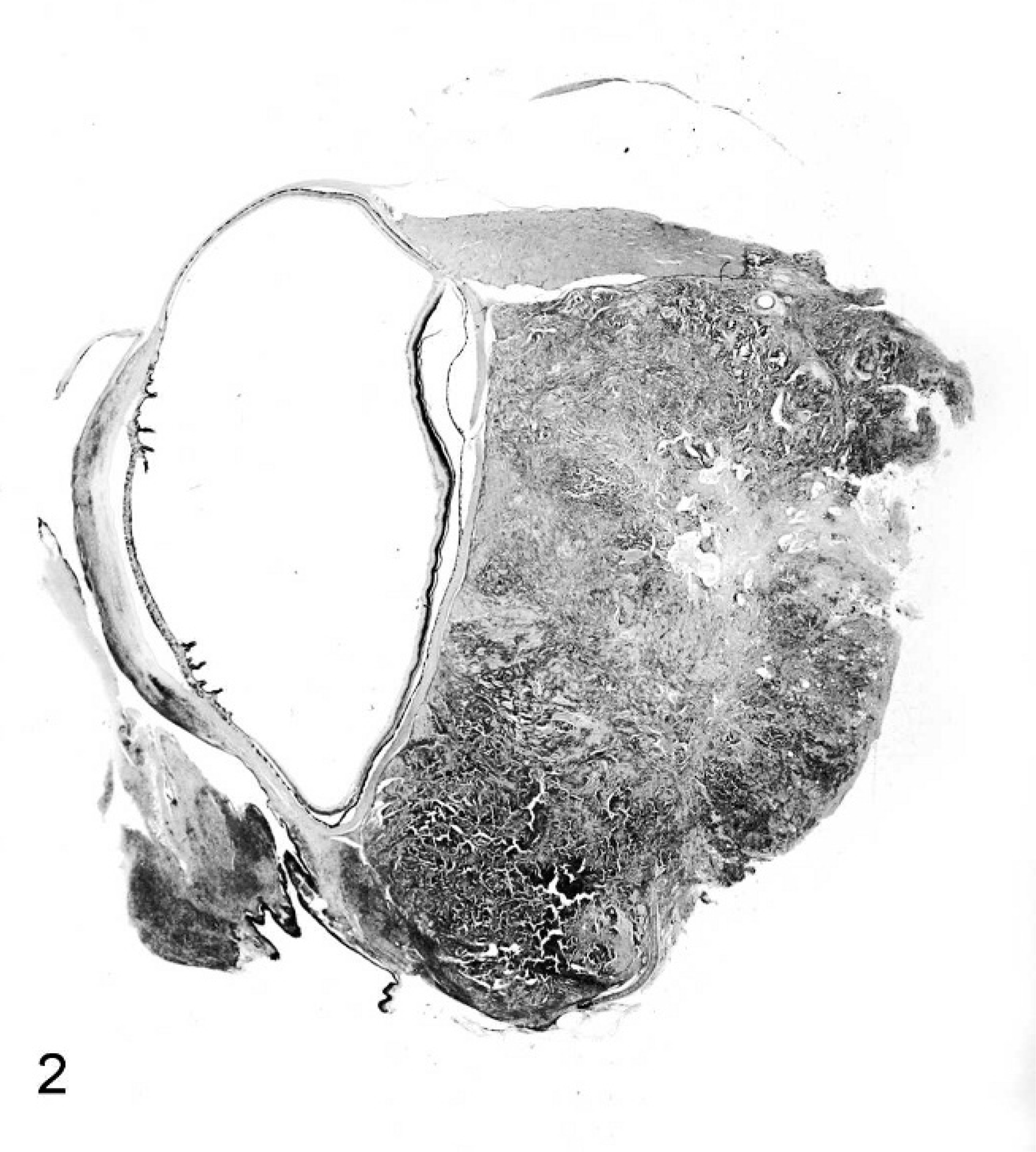
Subgross image of a multilobated Harderian gland adenocarcinoma forming a large retrobulbar mass in a Beechey ground squirrel. Central regions of necrosis are present. The cornea and adjacent conjunctival tissues are infiltrated by mixed inflammatory cells. HE stain.
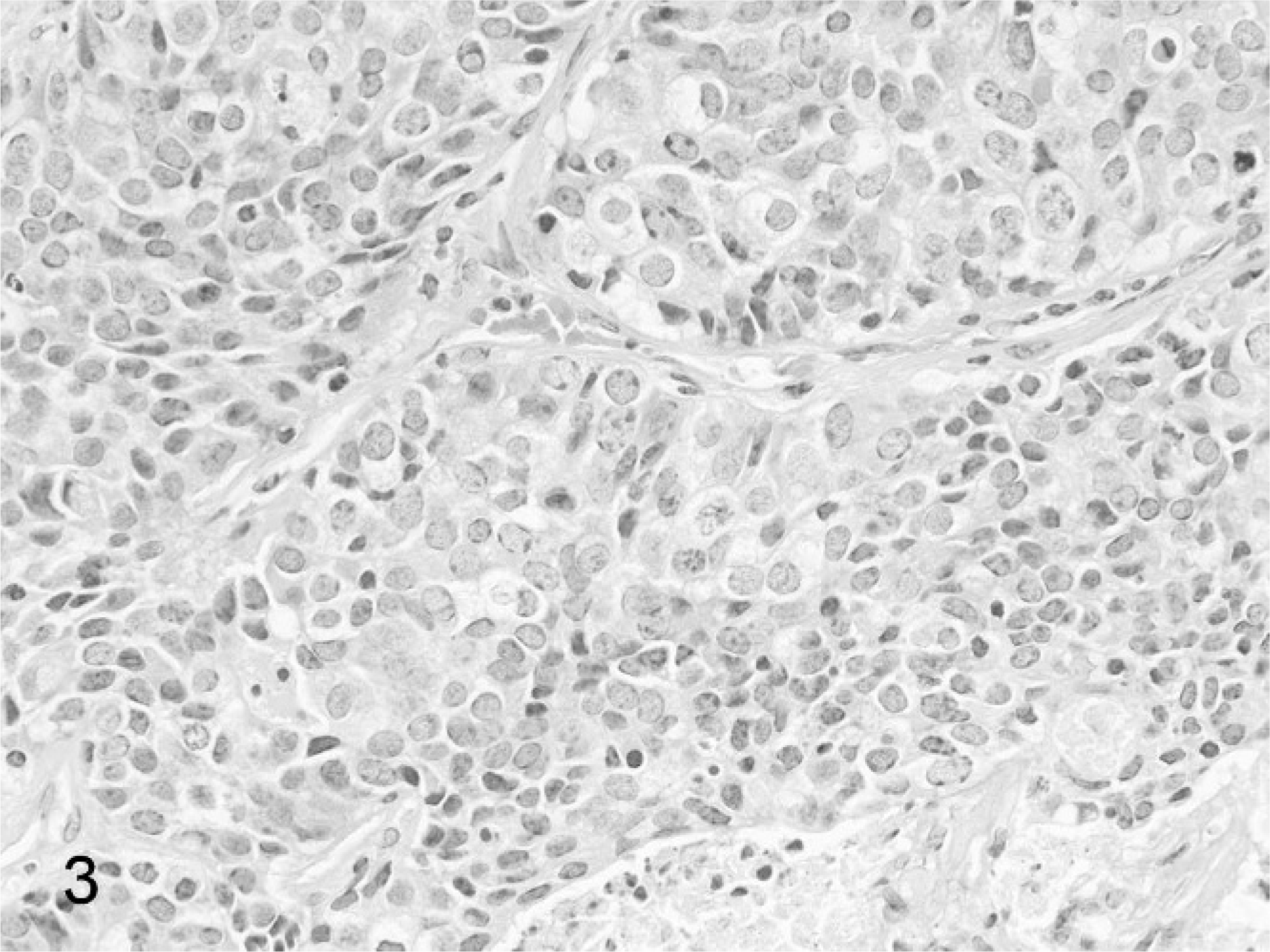
Harderian gland adenocarcinoma in a Beechey ground squirrel. Neoplastic cells are arranged in nests, papillary cords, and irregular acini, supported by fine collagenous stroma. An area of necrosis is present at the bottom of the image. HE stain.
One case was diagnosed with bilateral Harderian gland adenomas, characterized by nodular proliferations of glandular structures lined by well-differentiated, polyhedral to columnar cells with abundant foamy cytoplasm.
Discussion
Harderian gland neoplasms have been described in a number of laboratory rodent species, including the mouse, rat, hamster, and vole. 6– 10 The biologic behavior of these tumors varies by species. In mice, Harderian gland adenomas are common in the B6C3F1 strain, with a 4.7% incidence in control males and a 3.3% incidence in control females reported in NTP carcinogenicity studies; carcinomas were reported at a rate of 0.7% in both males and females in the same study. 22 The majority of the Harderian gland adenomas are classified as papillary cystadenomas. 6 Harderian gland carcinomas feature greater cellular pleomorphism, with local invasion and distant metastases. 20 In contrast, Harderian gland tumors are less common in rats than in mice, with a reported frequency of 0.1% in control F344 rats in NTP carcinogenesis studies. 23 In rats, most are classified as adenocarcinomas, often poorly differentiated, and while they exhibit infiltrative growth, metastasis is uncommon. 8
The incidence of Harderian gland tumors detected at necropsy in this population of Beechey ground squirrels was approximately 11% (18/167), higher than that seen in other laboratory rodents such as rats and mice.
Seventeen of 18 cases examined (94.4%) were classified as adenocarcinomas and were most commonly solid to tubular in arrangement. Biologically they demonstrated an aggressive pattern of behavior with local invasion, with widespread metastases demonstrated in 14 of 18 cases (77%), which indicates that this tumor is characteristically more aggressive in ground squirrels than in other species. One case was diagnosed with bilateral Harderian gland adenomas.
The tumors appear to be age-related, as these tumors occurred in animals at or beyond the end of their life expectancy in the wild. The life expectancy of this species in the wild is not well understood but estimated to be approximately 2 to 4 years. 24 Most of our animals developed tumors at ages considerably older than that. It is not clear what the natural incidence of this tumor is in wild populations as, few of these animals are afforded complete necropsies, and affected animals would be more likely to suffer from predation as a result of impaired vision or other effects of the neoplasm. Since the animals did not breed successfully in captivity, the genetic makeup of the captive population was not different than the wild populations from which they were derived, thus a genetic basis for the tumor incidence in the captive population is not likely. Because of the lack of information on the composition of the animal colony, appropriate statistical analysis was not possible to evaluate the relationship between tumor development and gender or virus infection status, but there was no dramatic preponderance of tumors in either gender, and tumors occurred with nearly equal frequency in uninfected or animals with previous or current infections. Other factors associated with captivity, such as altered photoperiod, disturbed hibernation periods, and access to commercial food, rather than a natural diet, as well as other changes associated with captivity, may have contributed to the tumor development.
In summary, we present data on the presence of Harderian gland tumors in a novel rodent group, the squirrel family, Sciuridae. Harderian neoplasms in these animals appear to be more frequent than in other species and have an aggressive phenotype, with a majority of tumors developing metastasis.
Footnotes
Acknowledgement
We acknowledge Carie Kimbrough, Safety Assessment, GlaxoSmithKline, Research Triangle Park, North Carolina, for helpful statistical analysis.
