Abstract
Keratoma is an aberrant keratin mass thought to originate from epidermal horn-producing cells interposed between the stratum medium of the hoof wall and the underlying third phalanx. The cause is unknown, although the presence of keratomas is frequently associated with chronic irritation, focal infection, or trauma. A total of 167 donkeys with keratomas were presented in this study. The diagnosis of a keratoma was based on clinical signs, radiography, and histopathologic examination. Surgical excision was attempted on all donkeys with lameness unless euthanasia was advised. Histopathologic examination, including Giemsa, periodic acid Schiff, and Young’s silver special histochemical stains, was performed and showed the presence of fungal hyphae and spirochete bacteria within the degenerate keratin. Polymerase chain reaction (PCR) for treponeme bacteria was performed on 10 keratoma lesions and 9 healthy pieces of hoof (controls). All healthy donkey tissues were negative for the 3 recognized digital dermatitis (DD) treponeme phylogroups, whereas 3 of 10 (30%) donkey keratoma samples were positive for one of the DD treponeme phylogroups. Routine fungal culture and PCR for fungi were performed on 8 keratoma lesions and 8 healthy pieces of hoof (controls). Keratinopathogenic fungi were detected in 1 of 8 (12.5%) keratomas, while only non-keratinopathogenic, environmental fungi were detected in 8 control healthy hoof samples. This is the first time the DD treponemes phylogroup and keratinopathogenic fungi have been detected in keratomas. Further studies are required to assess the significance of this finding.
Keratoma is an aberrant keratin mass thought to originate from epidermal horn-producing cells interposed between the stratum medium of the hoof wall and the underlying third phalanx.11,20,23,41,48 Keratomas have been described as horn cysts or benign horn tumors; however, there is no evidence that they are neoplastic in nature.20,48 Typically, keratomas are either spherical or cylindrical and are commonly located at the toe region, the quarter of the hoof, and less frequently at the sole or heel, with one case report describing a keratoma located at the frog.3,11,20,31,32,36,48 Single or multiple keratomas affecting one or more hooves in the same horse have been described.9,19,42,46,48 The cause is unknown, but the presence of keratomas is frequently associated with chronic focal irritation, focal infection such as hoof abscesses, or focal trauma.40,48 A thorough clinical history, in combination with clinical examination, may raise suspicion of a keratoma. Diagnostic imaging including radiographs, ultrasound of the sole (for solar keratomas), computed tomography, and low-field magnetic resonance imaging can be valuable in reaching a provisional clinical diagnosis, but histopathologic examination is required for a definitive diagnosis.17,28,29,48 Keratomas have not been studied extensively histologically. 48 Differential diagnoses should include other rare conditions such as hoof neoplasms (eg, melanoma, squamous cell carcinoma), proliferative pododermatitis (canker), or intraosseous epidermoid cysts of the third phalanx.22,37,48 The treatment of choice is complete or partial surgical excision which may lead to complete recovery.6,8 Keratomas have been recognized as a condition in donkeys. 47 The purpose of this study was to describe the histopathologic changes in the hoof and third phalanx of donkeys’ feet affected with keratomas.
Treponema spp. are spiral-shaped bacteria of the phylum Spirochaetes. Specific phylogroups of treponemes that include Treponema medium, Treponema pedis, and Treponema phagedenis–like species are particularly associated with the pathogenesis of bovine digital dermatitis (BDD), but have recently been associated with other hoof diseases of domestic and wild animals such as contagious ovine digital dermatitis of sheep, hoof diseases of American elk, and proliferative pododermatitis (canker) of horses.2,10,12,14,21,34,35,38,44,45,50 To date, Treponema spp. have not been identified in keratoma lesions. The significance of detecting BDD-associated Treponema spp. from keratomas is discussed in this study.
In humans, invasion of keratinopathogenic molds and keratinophilic dermatophytes is regarded as the most important factor in disease of the nail plate, which is called onychomycosis. 24 There is a paucity of information in the literature regarding the significance of keratinopathogenic molds in equine hoof disorders. The isolation of keratinopathogenic molds from keratomas and the significance of this finding are discussed in this study.
Materials & Methods
History and Clinical Presentation
All 167 donkeys were from a population of equids living at The Donkey Sanctuary in the South West of England, UK. A premortem diagnosis of keratoma was based on either clinical appearance alone or a combination of clinical appearance, radiographic findings, and histopathology results. A characteristic clinical appearance consisted of a soft-tissue mass of white-cream-colored keratin on the axial surface of the hoof wall, with varying consistency from soft to hard. The abnormal tissue led to either a bulge in the hoof wall or axial deviation of the white line dependent on the extent of the lesion within the hoof capsule. Lameness was observed in 133 of 167 (80%) cases, and in 21 of 167 (12.5%) cases, the keratomas were associated with infection. Fifty-seven of 167 (34%) donkeys with keratomas were euthanized due to welfare issues associated with uncontrolled lameness despite treatment.
Radiographic Examination
Dorso 60° proximal to palmarodistal oblique views were taken when radiographic evaluation was performed. A positive radiographic finding was considered to be a radiolucent defect in the solar margin of the distal phalanx with a smooth contour and minimal sclerosis, consistent with a space-occupying lesion in the hoof capsule (Fig. 1b).6,41 This is in comparison with an unaffected third phalanx radiograph where no radiolucent defects were observed (Fig. 1a).
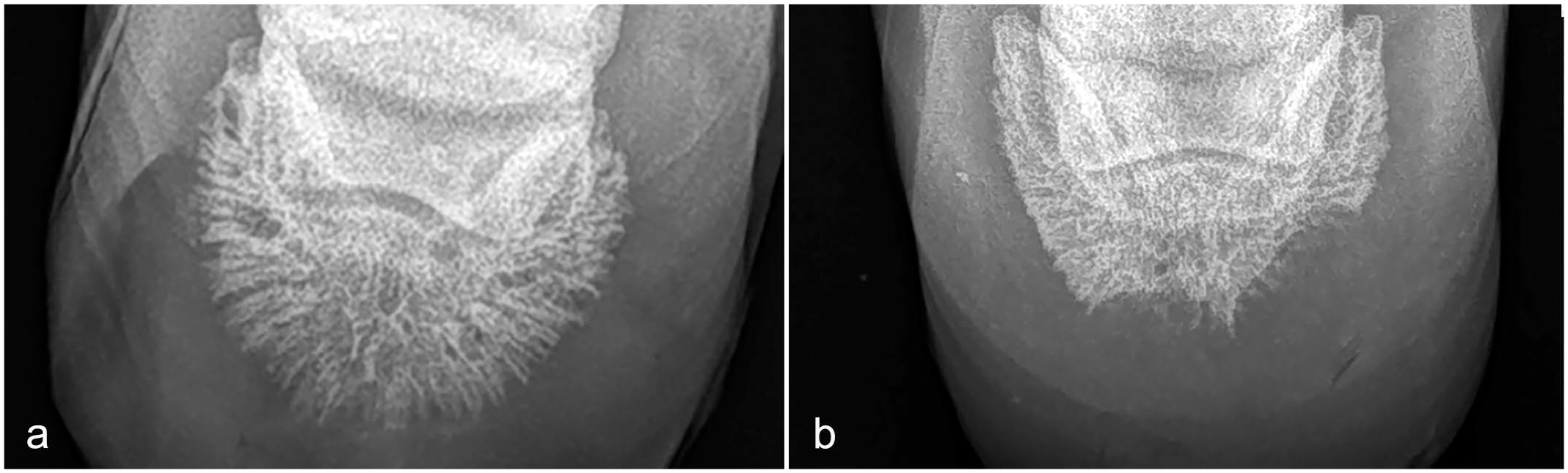
(a) Donkey hoof, normal radiograph. Dorso 60° proximal to palmarodistal oblique view. (b) Donkey hoof, keratoma, radiograph. Dorso 60° proximal to palmarodistal oblique view. Two radiolucent defects in the solar margin of the distal phalanx with a smooth contour and minimal sclerosis, consistent with a space-occupying lesion in the hoof capsule.
Surgical Procedure
Surgical excision was attempted in all donkeys with lameness associated with keratoma unless confounding factors led the clinician to advise euthanasia instead of treatment. Usually, keratomas that were subject to surgical excision showed no infiltration into the laminae and were commonly able to be “peeled” away.
Euthanasia
Euthanasia was performed on ethical grounds if there was lameness associated with keratoma and additional confounding factors including concurrent health concerns, lesions in multiple hooves, or extensive lesions that would lead to severe distortion and failure of the hoof capsule following resection.
Post Mortem Examination
Fifty-seven euthanized donkeys were subject to full postmortem examination by a board-certified veterinary pathologist at The Donkey Sanctuary. During the postmortem examination, all 4 hooves from each donkey were sectioned in sagittal and multiple transverse planes (Fig. 2a, b).
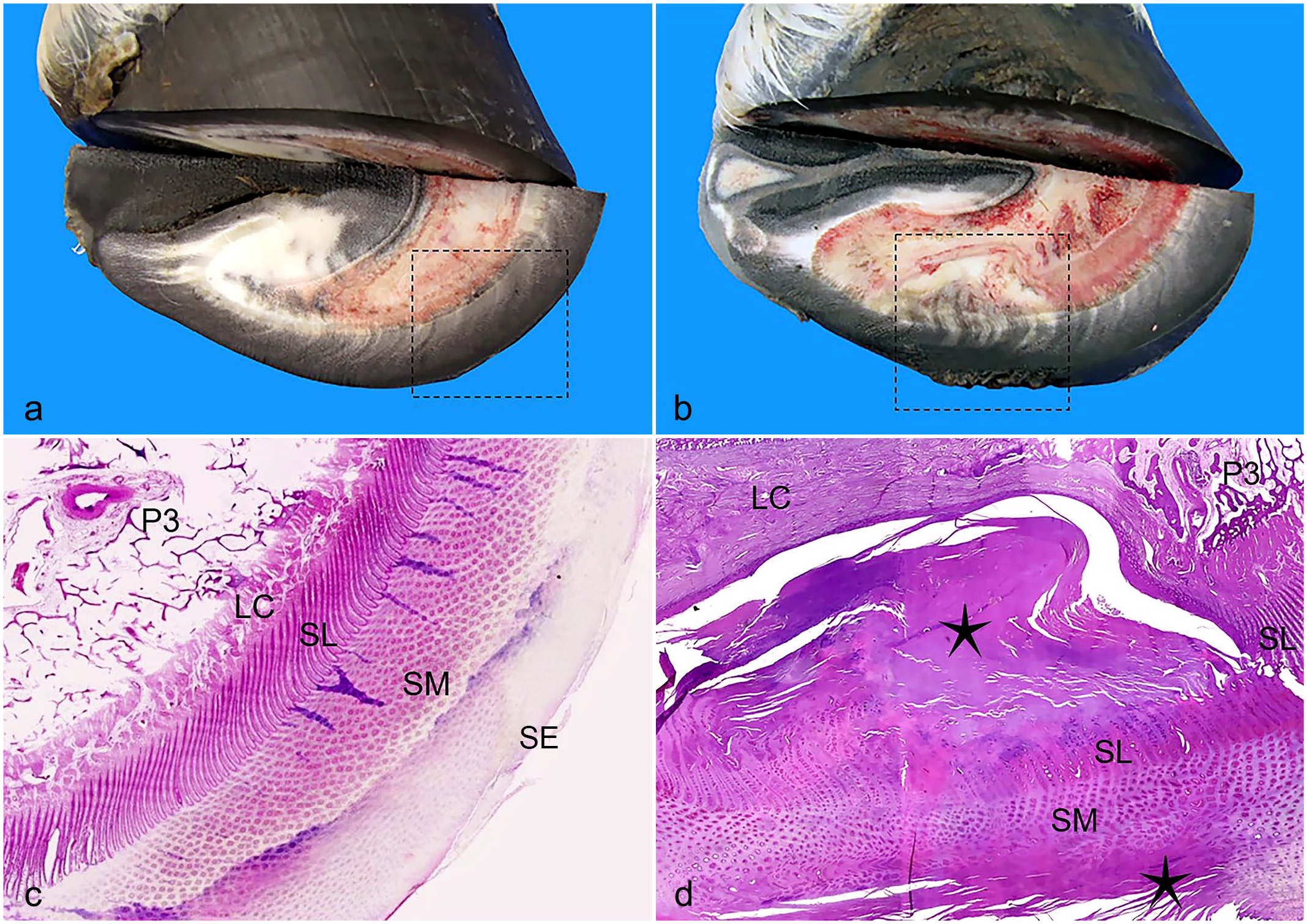
(a) Donkey hoof, normal, transverse section. (b) Donkey hoof, keratoma, transverse section. (c) Donkey hoof, normal. Subgross, area within dashed square in (a). P3, third phalanx; LC, laminar corium; SL, stratum lamellatum; SM, stratum medium; SE, stratum externum. Hematoxylin and eosin (HE) stain. (d) Donkey hoof, keratoma. Subgross, area within dashed square in (b) showing an irregular mass composed of abundant laminar degenerate keratin (stars) compressing and effacing the SL and SM. The third phalanx (P3) is compressed and subject to bone remodeling. There is sclerosis of the LC. HE.
Histopathologic Preparation
Lesions from the affected hooves of all donkeys with keratomas including transverse sections of the affected hooves in 4 donkeys (Fig. 2b), as well as transverse sections of normal/control donkey hooves (Fig. 2a), were collected either at postmortem examination or during surgical excision of keratomas, fixed in 10% neutral-buffered formalin, embedded in paraffin wax, and stained with hematoxylin and eosin for histopathological examination. In addition, examinations with Giemsa, periodic acid Schiff (PAS), and Young’s silver special histochemical stains were performed.
PCR Detection of Infectious Lameness-Associated Bacteria and Fungal Culture
For the isolation of genomic DNA (gDNA), tissues from healthy donkey and foot lesions were thawed, DNA was extracted using a DNeasy kit (Qiagen, Manchester, IK) as detailed in manufacturer’s instructions, and it was stored at −20°C. The donkey lesion gDNA samples were investigated using nested PCR assays, both genus specific for Treponema and species specific for each of the 3 aforementioned recognized BDD treponeme phylogroups using FIREPol polymerase (Solis, Estonia) as previously described. 18 Assays used reaction conditions and primers as originally detailed and included an initial universal bacterial 16S rRNA gene step, followed by the nested genus-/species-specific assays producing 300–500 bp products. gDNA Extractions of the 3 culturable treponemes and double distilled water were used as positive and negative control material, respectively.12,33 A Dichelobacter nodosus–specific PCR assay which amplified a 586-bp region of the D. nodosus 16S rRNA gene was also used to assess the samples, as previously described. 44 For Fusobacterium necrophorum detection, a species-specific PCR assay was also used which targets the lktA gene, as previously described.4,44 All PCR assays were analyzed in triplicate. All resulting PCR products were subjected to separation by 1% (w/v) agarose (Bio-Rad, Hemel Hempstead, UK) electrophoresis at 110 V, 400 mA, for 40 minutes and visualized by 0.5 mg/ml ethidium bromide staining and subjected to ultraviolet (UV) illumination and image-recording using a standard gel documenting system.
Healthy controls and lesion samples were routinely cultured on Sabouraud dextrose broth (2% [wt/vol] glucose, 1% [wt/vol] peptone) supplemented with chloramphenicol (1 mg l−1), subcultured onto Sabouraud dextrose agar slants, and kept at 4°C for fungal culture.
DNA extraction, preparation of the PCR mixture, and post-PCR analysis were carried out in separate rooms using equipment designated for each area to minimize the possibility of specimen contamination. 16
The fungal strains were inoculated in 1.5 ml Eppendorf tubes containing 0.5 ml of Sabouraud dextrose broth supplemented with chloramphenicol and incubated overnight in an orbital shaker at 150 rpm and 30°C. Thereafter, fungal cultures were adjusted photometrically (absorbance at 530 nm; McFarland 0.5 standard) to a concentration of 1 × 106 to 5 × 106 cells/ml. In the case of filamentous fungi, the conidia were separated from the rest of the mycelium by filtration through sterile glass wool. 26 The fungal suspensions with predetermined concentrations were centrifuged at 5000 × g, and then the pellet was frozen at −20°C for 1 hour and incubated at 65°C for 1 hour in 0.5 ml of extraction buffer (50 mM Tris-HCl, 50 mM Ethylenediamine tetraacetic acid, 3% sodium dodecyl sulfate, 1% 2-mercaptoethanol). The lysate was extracted with phenol-chloroform-isoamyl alcohol (25:24:1, vol/vol/vol). Then, 65 μl of 3 M sodium acetate and 75 μl of 1 M NaCl were added to 350 μl of the supernatant, and the resulting volume was incubated at 4°C for 30 minutes. DNA was recovered by isopropanol precipitation and washed with 70% (vol/vol) ethanol. The concentration was measured by monitoring the UV absorbance at 260 nm (Gene Quant System; Pharmacia, LKB Biochrom). 16
The extracted DNA was amplified using a RoboCycler 96 temperature cycles (Stratagene, La Jolla, Calif). The primers used are specified below. PCR amplification was carried out in 2 steps. 16 The universal primers used for fungal amplification were ITS1 (5′ TCC GTA GGT GAA CCT GCG G 3′), which hybridizes at the end of the 18S rRNA gene, and ITS4 (5′ TCC TCC GCT TAT TGA TAT GC 3′), which hybridizes at the beginning of the 28S rDNA (Life Technologies, Barcelona, Spain). 49
For the second amplification, the primers used were ITS86 (5′ GTG AAT CAT CGA ATC TTT GAA C 3′), which hybridizes with the 5.8S rDNA region, and ITS4 (Life Technologies, Barcelona, Spain). 27 PCR products were sequenced on both strands with the amplifying primers and identification done using www.boldsystems.org. The method used was Sanger sequencing.
Results
Radiographic Findings
Dorso 60° proximal to palmarodistal oblique views of the affected hooves were taken in 140 of 167 (83%) donkeys. Lesions strongly suspected of a keratoma, namely radiolucent defect with smooth contour and minimal sclerosis affecting the solar margin and/or the wings of the third phalanx, were noted in 115 of 140 (82%) donkeys (Fig. 1b).
Macroscopic Findings
All 4 hooves of the affected donkeys subjected to postmortem examination were examined, and a sagittal section as well as multiple transverse sections of the hooves were made to better reveal the extent of the lesions (Fig. 2a, b). Focally, the hoof wall of one or more feet of the affected donkeys was expanded and replaced by a spherical, cylindrical, or irregularly shaped, pale white or gray to dark gray, and varying in consistency from soft and friable to hard and solid mass that effaced and/or compressed the stratum lamellatum and the laminar corium. Often, compression bone resorption of the third phalanx was present. The mass either affected the whole hoof wall thickness or just the inner part. Similar masses in the solar part of the hoof were less often observed.
Histopathologic Findings
Samples of keratomas from all 167 donkeys were examined histologically. The histopathologic findings in the affected hooves were compared to the normal histologic anatomy of nonaffected hooves (Figs. 2c and 3a, c). Focally, the normal architecture of the stratum medium and often stratum externum of the hoof was effaced and replaced by degenerate laminar keratin, often admixed with nucleated keratinocytes (orthokeratotic and parakeratotic hyperkeratosis) (Fig. 2d). This aberrant keratin mass, which was rarely admixed with moderate to high numbers of neutrophils (suggesting secondary bacterial involvement), compressed the underlying structures, namely the stratum lamellatum, the laminar corium, and the third phalanx (Fig. 2d). Focally, there was either loss of differential staining (necrosis) (Fig. 3d) or atrophy, stunting, and fusion of the primary and secondary epidermal and dermal lamellae (Fig. 3b). Focally, there was regular epidermal hyperplasia (Fig. 3b). Multifocally, the primary dermal lamellae were infiltrated by low numbers of lymphocytes and plasma cells. Focally, the third phalanx trabecular bone was lined by osteoblasts and fewer osteoclasts (remodeling). There was focal bone lysis of the third phalanx (Fig. 2b, d). There was focal sclerosis affecting the laminar corium (Fig. 2d).
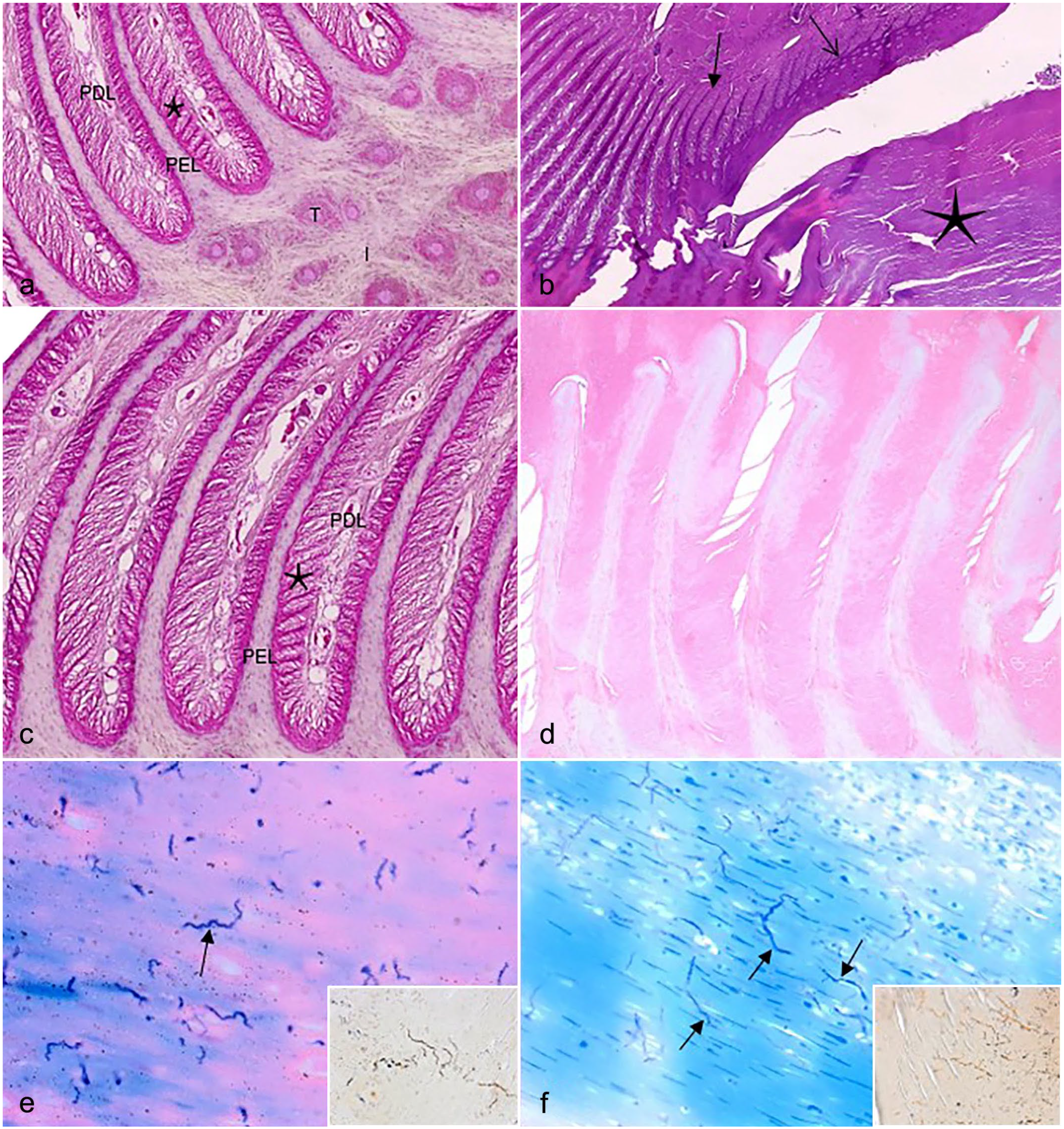
(a) Donkey hoof. Normal histomorphology of stratum lamellatum and stratum medium. PEL, primary epidermal lamellae; PDL, primary dermal lamellae; Star, secondary epidermal and dermal lamellae; T, tubular horn; I, intertubular horn. Hematoxylin and eosin (HE). (b) Donkey hoof, keratoma. Abundant degenerate laminar keratin (star). Stunting, fusion, and distortion of the primary lamellae (closed arrow). Regular epidermal hyperplasia (open arrow). HE. (c) Donkey hoof. Normal histomorphology of stratum lamellatum. PEL, primary epidermal lamellae; PDL, primary dermal lamellae; Star, secondary epidermal and dermal lamellae. HE. (d) Donkey hoof. Loss of differential staining (necrosis) of the stratum lamellatum. HE. (e) Donkey, hoof, keratoma. Multiple, strongly Giemsa-positive, wavy spirochete-like bacteria (arrow) within degenerate keratin, Giemsa stain. Inset: similar bacteria, Young’s silver stain. (f) Donkey, hoof, keratoma. Multiple, strongly Giemsa-positive, filamentous and branching fungal hyphae (arrows) within laminar degenerate keratin admixed with nucleated keratinocytes, Giemsa stain. Inset: similar fungal hyphae, Young’s silver stain.
Giemsa, Gram, PAS, and Young’s silver special histochemical stains revealed numerous filamentous, up to 200 µm long, frequently branching fungal hyphae (Fig. 3f) and fewer wavy Spirochete-like bacteria (Fig. 3e) measuring 80–100 µm in length. All microorganisms detected were strongly Giemsa-positive and PAS-positive and weakly positive with Young’s silver histochemical stain (Fig. 3e, f). Interestingly, no fungal hyphae or Spirochete-like bacteria were detected within the healthy hoof wall.
PCR Detection for Potential Bacterial Pathogens and Fungal Culture
PCRs for bacterial pathogens were performed in 10 keratomas and 9 healthy controls. All donkey samples were negative for both of the ovine scald/footrot-associated pathogens D. nodosus–specific and F. necrophorum–specific PCR assays (Tables 1, 2).
The PCR detection of treponemes, Dichelobacter nodosus, and Fusobacterium necrophorum in donkey healthy (control) hooves.
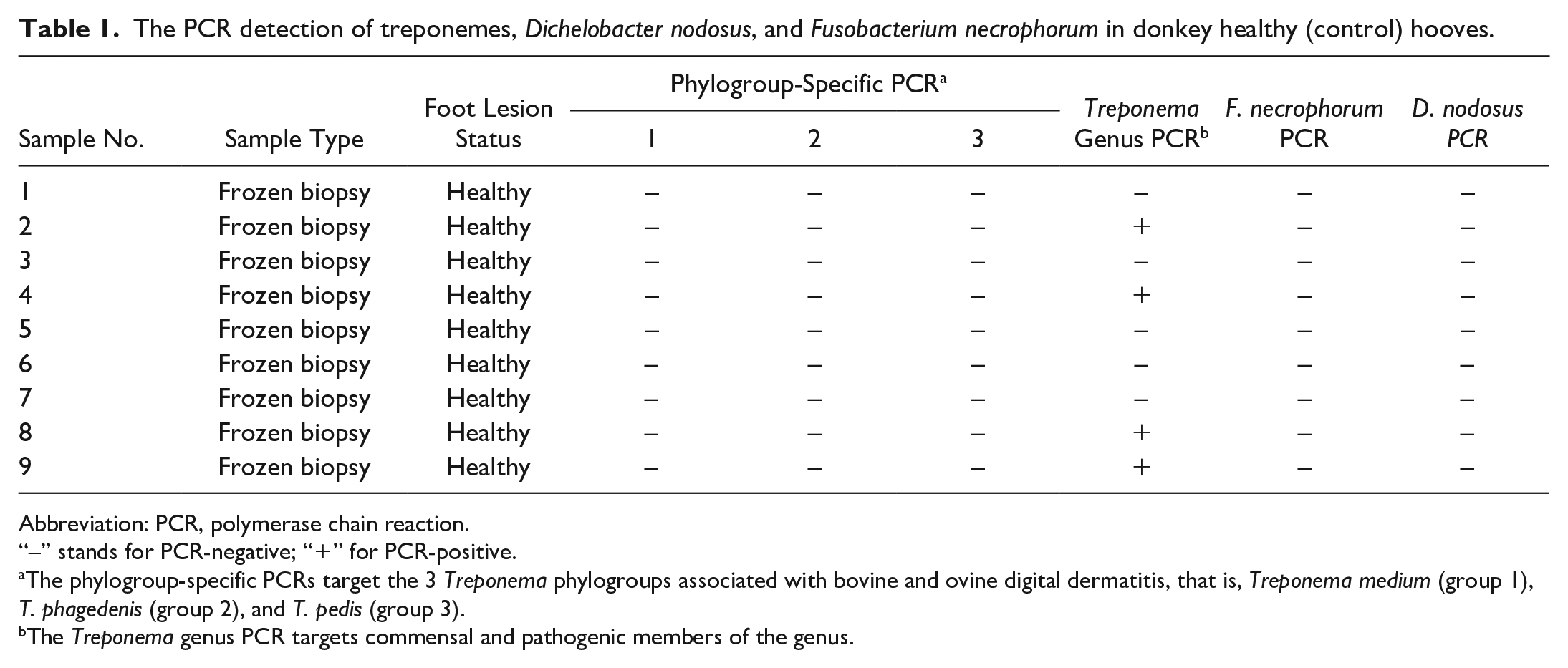
Abbreviation: PCR, polymerase chain reaction.
“–” stands for PCR-negative; “+” for PCR-positive.
The phylogroup-specific PCRs target the 3 Treponema phylogroups associated with bovine and ovine digital dermatitis, that is, Treponema medium (group 1), T. phagedenis (group 2), and T. pedis (group 3).
The Treponema genus PCR targets commensal and pathogenic members of the genus.
The PCR detection of treponemes, Dichelobacter nodosus, and Fusobacterium necrophorum in donkey keratomas.
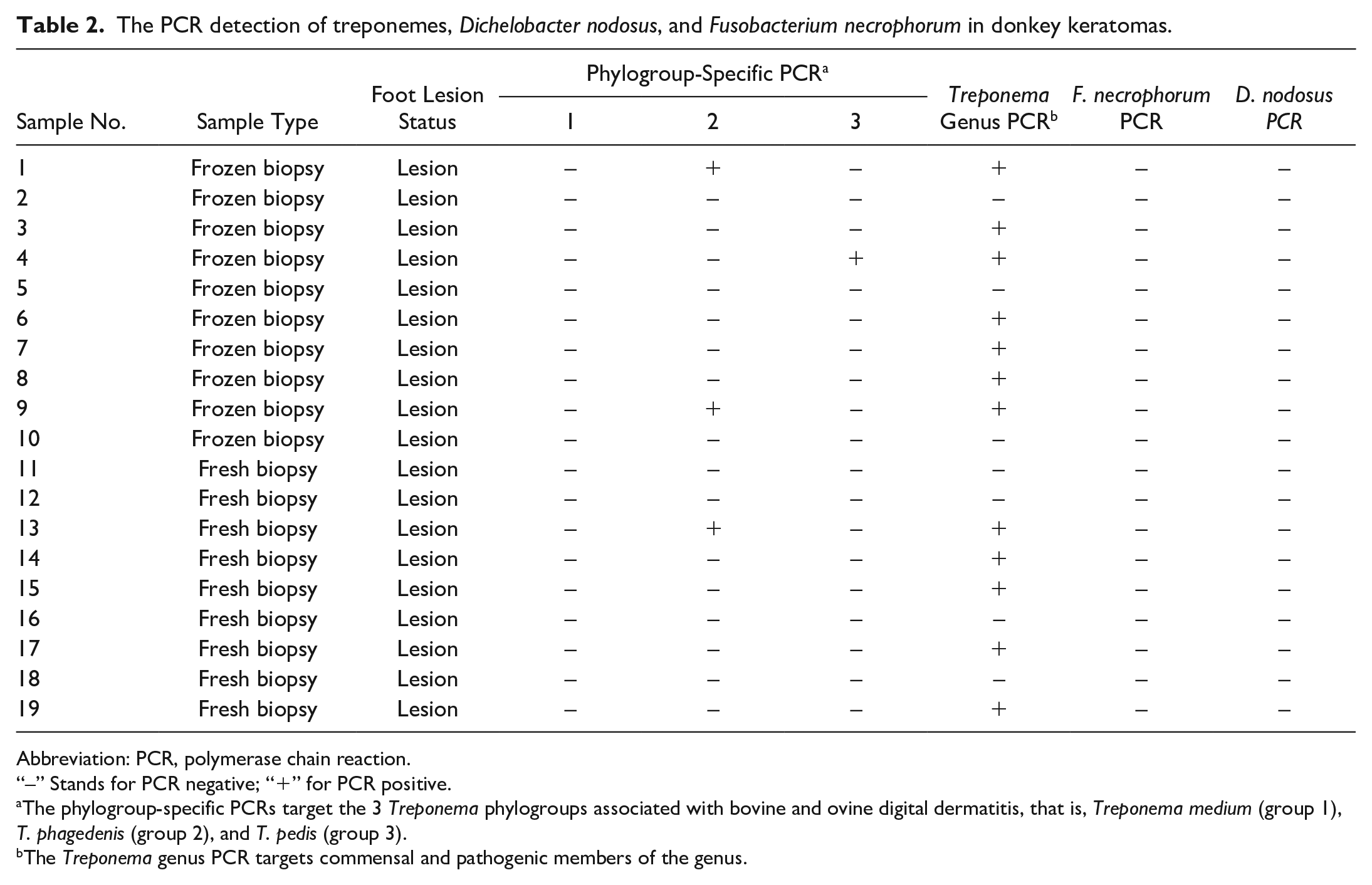
Abbreviation: PCR, polymerase chain reaction.
“–” Stands for PCR negative; “+” for PCR positive.
The phylogroup-specific PCRs target the 3 Treponema phylogroups associated with bovine and ovine digital dermatitis, that is, Treponema medium (group 1), T. phagedenis (group 2), and T. pedis (group 3).
The Treponema genus PCR targets commensal and pathogenic members of the genus.
All healthy donkey foot tissues (n = 9) were negative for the 3 recognized DD treponeme phylogroups, whereas 3 of 10 (30%) donkey keratoma samples were positive for one of the DD treponeme phylogroups (Tables 1 and 2). No lesions contained multiple of the recognized DD treponeme phylogroups. The phylogroup-specific PCR for T. medium, T. phagedenis, and T. pedis DD spirochetes showed they were present in 0 of 10 (0%), 2 of 10 (20%), and 1 of 10 (10%) donkey keratomas, respectively.
Of the healthy donkey tissues, 4 of 9 samples (44.4%) were positive with the general Treponema PCR, whereas 7 of 10 (70%) keratomas were positive for the presence of general treponemes (Treponema genus–specific PCR). Culture for treponemes was not attempted.
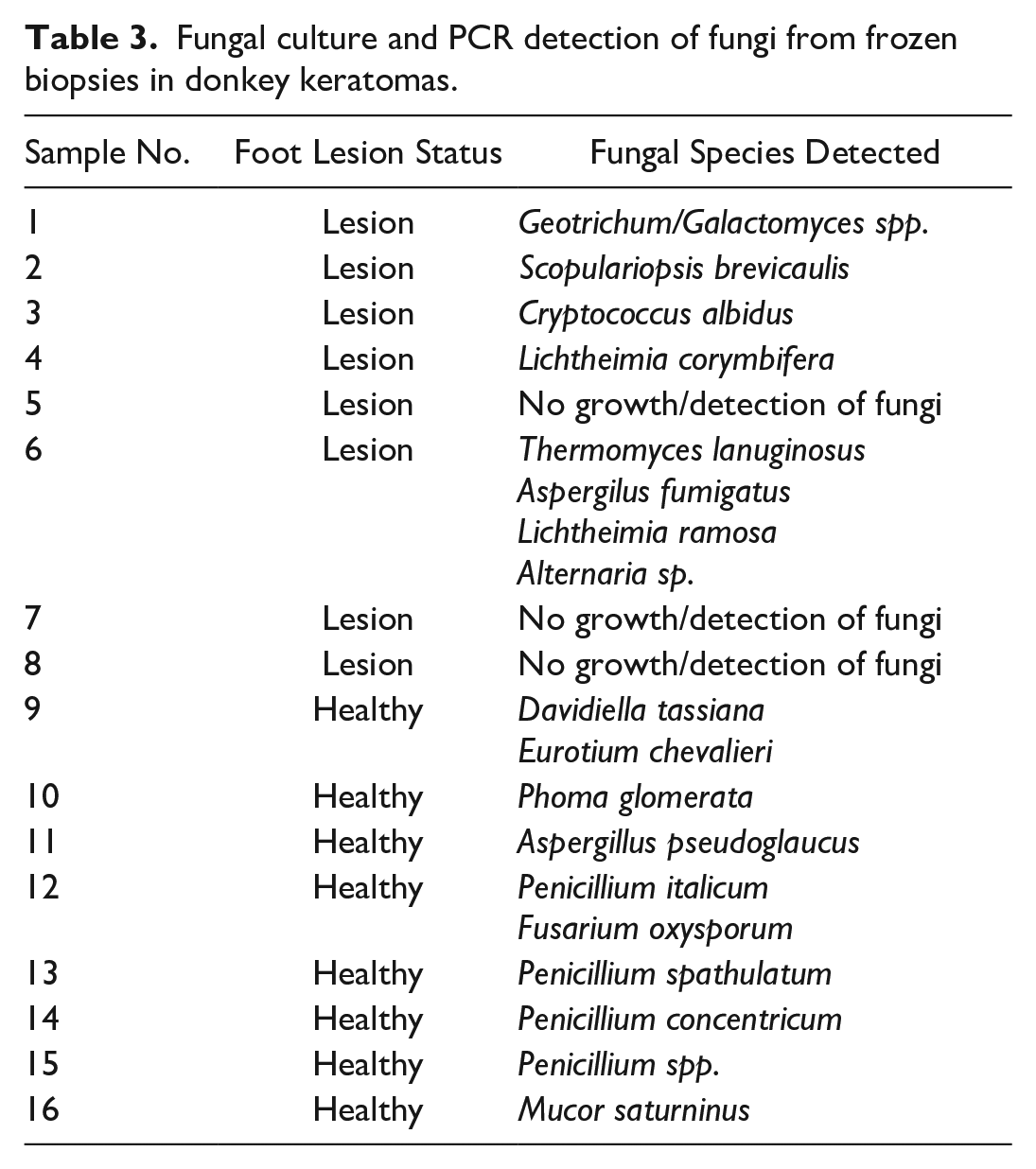
Fungal cultures were performed in 8 keratomas. In 1 of 8 (12.5%) keratomas, the keratinopathogenic fungus Scopulariopsis brevicaulis was detected (Table 3). 24 In 3 of 8 (37.5%) keratoma lesions, 4 fungi of unknown keratin pathogenicity (Lichtheimia corymbifera, Lichtheimia ramosa, Alternaria spp., and Geotrichum spp.) were detected. 24 However, Lichtheimia corymbifera produces keratinases, and Lichthemia ramosa has been involved in cutaneous infections in humans.1,5 In 3 of 8 (37.5%) keratomas, there was no growth of fungi. In 1 of 8 (12.5%) keratomas, a non-keratinopathogenic fungus (Cryptococcus albidus) was detected. In all healthy hoof samples, only environmental, non-keratinopathogenic fungi were detected.
Fungal culture and PCR detection of fungi from frozen biopsies in donkey keratomas.
Discussion
Keratomas are rare lesions of horse’s hooves but should be included in the differential diagnosis in foot-oriented lameness cases.17,23,29,36,42,46,48 Although keratomas have been recognized as a condition in donkeys, the associated literature is sparse. 47 To the authors’ knowledge, this study comprises the largest number of donkeys with keratomas. A potential reason for this is the fact that donkey hooves evolved to absorb a vast amount of moisture in order to stay hydrated and flexible in an arid environment. 7 When exposed to long periods of high environmental humidity, as in the United Kingdom, donkey hooves absorb excessive moisture, and as a result, they are prone to recurrent abscess formation, one of the possible causes of keratomas.
Histopathologic examination is the gold standard for diagnosing keratomas; however, they have not been studied extensively. In this study, we aimed for a concise and thorough histopathologic description of keratomas including pathologic changes of the stratum medium, stratum lamellatum, the laminar corium, and the third phalanx, which can be used as a guide by pathologists presented with hoof masses.
Partial or complete surgical excision, which is the treatment of choice, will typically result in complete recovery. However, in one report, there was evidence that postoperative complications such as excess granulation tissue formation, hoof crack formation, and recurrence occurred more often in keratomas subject to complete resection than the ones subject to partial resection (71% vs 25%). 6
The cause of keratomas is unknown, but it is thought that chronic focal irritation, focal infection, such as hoof abscesses, or focal trauma is commonly associated with keratoma pathogenesis.40,48 In this study, 61 of 167 (37%) donkeys with keratoma lesions had a history of recurrent hoof abscess or other trauma on the affected foot. Interestingly, in this study, both keratoma and healthy samples were negative for both the ovine scald/footrot-associated pathogens D. nodosus–specific and F. necrophorum–specific PCR assays. Similar PCR assays in one paper investigating the same anaerobic bacteria in horses with equine hoof thrush revealed the presence of F. necrophorum in 1 of 14 control healthy hooves and 5 of 14 hooves with thrush, while D. nodosus was not isolated in any of the control or affected hooves. 39 The literature lacks in similar studies in donkeys. Therefore, to date, it is unknown if those bacteria are part of the normal hoof flora in donkeys. The involvement of Treponema spp. and keratinopathogenic molds as a primary or secondary cause of keratoma has not been previously investigated. BDD-associated Treponema phylogroup spp. have been isolated in 3 of 10 keratomas, but not from 10 healthy control hoof walls. Keratinopathogenic molds were isolated in 1 of 8 keratomas while only non-keratinopathogenic, environmental fungi were detected in 8 healthy control hoof walls. It is unknown how those microorganisms penetrate to the keratoma. It is believed that BDD-Treponema spp. and keratinopathic molds are found on the soil.14,24,25 Although donkeys at The Donkey Sanctuary farms do not cograze with any ruminants, sheep graze in some fields when there are no donkeys. Shedding of Treponema spp. by those sheep may be a source of soil, and subsequently donkey hoof contamination. It has been demonstrated that the bovine gut is an important reservoir of microbes involved in BDD pathogenesis.15,51 This assumption remains to be proven in equines. In terms of the treponemal species identified here, both are considered serum-dependent and to cluster closely on phylogenetic analysis to the agent of human syphilis Treponema pallidum, differentiating them from treponemes typically considered as commensal.13,43 Both species have been implicated in the digital dermatitis of cattle, sheep, and American elk, with T. pedis recently reported to be most highly associated with equine canker samples.10,12,30 Of note, recent data suggest that digital dermatitis T. phagedenis strains exhibit genetic evidence of pathogenicity islands including a type IV secretion system that differentiates it from human nonpathogenic strains. 43 While further studies are needed to establish the contribution of Treponema spp. and keratinopathogenic molds to the pathogenesis of keratomas, the findings of this study are strongly indicative that there is at least secondary involvement in the pathogenesis and prognosis.
Conclusion
To the author’s knowledge, this is the most detailed and conclusive histopathologic description of keratomas in equines and could be used as a guide for the diagnostic workup of equine hoof masses where keratoma is included in the differential diagnosis. This is the first time BDD-associated Treponema phylogroup and keratinopathogenic fungi have been detected in keratomas. Further studies are required to assess the significance of this finding.
Footnotes
Acknowledgements
We would like to thank Stuart Norris, Laura Kubasiewicz, and Nikki Bell, senior statistician, senior researcher, and senior researcher at The Donkey Sanctuary, respectively, for their invaluable help with data collection and process, and the histopathology laboratories and the histology technicians at the diagnostic laboratory, Royal Veterinary College, London, UK, and Abbey Veterinary Services, Newton Abbot, UK, for their endless patience while processing the hoof samples.
Declaration of Conflict of Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
