Abstract
A novel foot disease in free-ranging elk (Cervus elaphus) in southwestern Washington State emerged in 2008 and spread throughout the region. Initial studies showed adult elk had chronic hoof overgrowth, sole ulcers, and sloughed hoof capsules, but no cause was determined. To identify possible causes and characterize the earliest lesions, 9-, 7-, and 3-month-old elk were collected. Nine-month-old elk had sole ulcers (3/9 elk) and sloughed/overgrown hoof capsules (4/9 elk) similar to adults. Histologically, lesions consisted of coronary, heel bulb, and interdigital ulcers with suppurative inflammation, epithelial hyperplasia, deeply invasive spirochetes, and underrunning of the hoof capsule and heel-sole junction. Spirochetes were identified as Treponema via immunohistochemistry and polymerase chain reaction (PCR). Seven-month-old elk had similar underrunning foot ulcers (6/8 elk) with Treponema identified in all lesions but no chronic overgrowth or sloughed hoof capsules. Three-month-old calves had superficial coronary erosions with no inflammation or identifiable spirochetes (3/5 elk) but were culture/PCR positive for Treponema, suggesting possible early lesions. Lesions from 9- and 7-month-old elk included aerobic and anaerobic bacteria, many of which are associated with infectious foot disease in livestock. Antibody enzyme-linked immunosorbent assay of 7- and 3-month-old elk from the enzootic region showed a trend toward increased Treponema antibody titers compared to normal control elk from outside the region, further supporting the significance of Treponema in the pathogenesis of foot disease. Treponeme-associated hoof disease (TAHD) in elk, a debilitating and progressive condition, shares similarities to bovine digital dermatitis and contagious ovine digital dermatitis.
Reports of free-ranging elk (Cervus elaphus) with deformed hooves and severe lameness dramatically increased in southwestern Washington State in 2008. Informal surveys conducted by the Washington Department of Fish and Wildlife (WDFW) revealed that up to 80% of observed elk groups in the Cowlitz river basin (enzootic region) included affected individuals. Within affected groups, between 30% and 90% of elk were lame with or without hoof deformities. Diseased elk have since been identified in groups to the west, east, and, more recently, south of this initial region. 45
Gross foot lesions previously characterized in adult elk with advanced disease included marked overgrowth and deformity of hooves, broken and sloughed hoof capsules, sole ulcers, and underrunning inflammation and necrosis of the epidermal and dermal lamina. Bone lesions, other than secondary pedal osteomyelitis and septic pedal arthritis, were not detected in any elk, and no underlying systemic disease was identified, although most elk had deficient levels of hepatic selenium and copper, minerals that are known to be deficient in this region. Routine aerobic and anaerobic bacteriologic cultures performed on fresh tissue from chronically diseased feet detected numerous diverse species but no obvious primary pathogens. Correspondingly, numerous bacteria were evident histologically in chronic foot lesions, although a specific causative microorganism was not identified. 30 Due to the rapid and concentric spread of hoof disease in this population of elk, an infectious cause spreading within a naive population of animals was suspected. This project was designed to identify the earliest foot lesions and to characterize progressively advanced foot lesions by using different age groups of elk from the enzootic region. Diagnostic tests were performed with the goal of identifying probable pathogens associated with foot disease and to identify common underlying factors that may predispose elk to infection.
Materials and Methods
Study Design
Free-ranging, male and female, age-matched elk were collected within the enzootic region, with individual elk denoted as “cases.” Age-matched elk were also collected from outside the enzootic region in a similar southwestern Washington State habitat or from arid habitat in central Washington State, and elk were denoted as “controls.” Animals were collected based on age and availability, and animals from the enzootic region with clinical lameness were preferentially sought. All elk were killed by gunshot wound to the chest with necropsy and samples collected under the supervision of a WDFW veterinarian and staff with preapproval and strict adherence to WDFW animal handling protocols. Age groups were collected by chronological seasons (winter 2013: 9-month-old elk, summer 2014: 3-month-old elk, late fall 2014: 7-month-old elk). Data are presented from the most established lesions in 9-month-old elk to the earliest discernible lesions in 3-month-old elk for clarity.
Elk collected were as follows:
9-month-old elk
Nine elk (case Nos. 1–9) were collected from affected groups within the enzootic region of southwestern Washington State in late February and March 2013. Seven control elk were collected during this same time period from outside the enzootic region in southwestern Washington State (control Nos. 1–3) and from central Washington State (control Nos. 4–7).
The goal of examining this group was to identify and characterize foot lesions from within the enzootic region, compared to healthy age-matched control elk from a similar habitat and to healthy age-matched control elk from a more arid habitat, and to identify any infectious agents associated with foot lesions in elk from the enzootic region. In addition, we examined the overall health of 9-month-old case and control elk to identify any systemic diseases or conditions, similar to the diagnostic work performed previously on adult elk from the enzootic region. 30
Gross necropsy examinations were performed on all elk. All feet were radiographed to identify any bone involvement, and histopathology of viscera and feet was performed as detailed below. Histochemical staining was performed on representative specimens of healthy and affected feet from case and control elk, with morphological characterization of microorganisms. Selected foot lesions that included spirochetes were tested by immunohistochemistry and flaB2 polymerase chain reaction (PCR) to identify Treponema as described below. Swabs from the feet of affected and control elk were cultured for aerobic and anaerobic bacteria with routine methods as described below. Diagnostic testing to examine overall health was performed on all 9-month-old elk and included the following tests: viral serology, virus isolation of viscera and foot specimens, fecal parasitology with quantitation of parasites, and hepatic trace mineral analysis. Antibody titers to Treponema sp. homogenate were measured for all elk.
7-month-old elk
Eight elk (case Nos. 15–22) were collected from the enzootic region in January 2014. No control 7-month-old elk were acquired because feet from adult, 30 9-month-old, and 3-month-old control elk from southwestern Washington State elk were consistently normal based on gross and histological examination.
The goal of examining this group was to identify and characterize early, established foot lesions in elk from within the enzootic region; to confirm the association of spirochetes with lesions; and to identify bacteria associated with healthy and affected feet. Gross necropsy and histopathology of viscera and feet were performed on all elk as detailed below. Radiographs were not performed as no primary bone lesions had been identified in previously collected groups of elk. Histochemical staining was performed on representative specimens of healthy and affected feet from all elk to identify and characterize morphology of microorganisms. Samples were tested by immunohistochemistry and flaB2 PCR as described below. Aerobic and anaerobic culture of healthy and affected feet was of paramount interest in this group, and culture techniques were modified as described below for enhanced identification of other foot-associated bacteria. 16S ribosomal DNA (rDNA) PCR was performed from bacterial cultures to characterize bacteria, as detailed below. No additional testing for underlying diseases was performed on these elk for reasons mentioned above. Antibody titers to Treponema sp. homogenate were measured for all elk.
3-month-old elk
Five elk (case Nos. 10–14) were collected from the enzootic region in August 2013, and 2 elk (control Nos. 8–9) were collected from outside the enzootic region in a similar southwestern Washington State habitat.
The goal of examining this group was to identify and characterize very early foot lesions in calves from within the enzootic region compared to healthy age-matched control calves from a similar habitat, with a focus to identify spirochetes within lesions. Gross necropsy was performed on all elk. Feet were radiographed to identify any bone involvement, and histopathology of viscera and feet was performed as detailed below. Histochemical staining was performed on representative specimens of healthy and affected feet from case and control elk, with morphological characterization of microorganisms. Samples were tested by immunohistochemistry and flaB2 PCR as described below. Culture to identify other foot-associated bacteria was not performed on this group of elk. No additional testing for underlying diseases was performed on these elk as adult 30 and 9-month-old elk from the enzootic region consistently showed no underlying diseases and predictable trace mineral deficiencies. Antibody titers to Treponema sp. homogenate were measured for all elk.
Tissue Collection
Tissues collected varied between study groups as follows.
All study elk
Elk were killed by gunshot wound to the chest and full necropsies were immediately performed in the field. Gross pathological findings were recorded and the following fresh specimens were collected sterilely: lung, liver, kidney, spleen, mesenteric lymph node, ileum, feces, serum, whole blood, and all 4 limbs, in addition to 5-mm punch specimens of the coronary band, heel, interdigital space, and any macroscopic foot lesions. Foot specimen collection sites were prepared by dry brushing the site and then rinsing with clean water. Several deep 5-mm punch specimens were taken using a sterile biopsy instrument targeting the interface of healthy tissue and the macroscopic lesion, when present. Specimens were placed directly into a sterile cryotube (Fisher Scientific, Hampton, NH). Specimens were sent on wet ice overnight to respective laboratories for processing within 24 to 48 hours after necropsy. Representative specimens of all viscera were collected and preserved in 10% neutral-buffered formalin and processed for routine histopathology. Additional specimens were collected by group as follows:
9-month-old elk
Swabs of normal feet and macroscopic foot lesions were obtained with methods detailed below (see “Aerobic and Anaerobic Bacteriologic Culture 16S rDNA Polymerase Chain Reaction”).
7-month-old elk
For each elk with macroscopic foot lesions, a specimen was also obtained from the same anatomic location of the contralateral nondiseased foot to serve as a normal control specimen. Specimens were submitted in sterile cryotubes, as noted above, and fresh specimens were submitted in Anaerobic Tissue Transport Medium and Oral Treponeme Enrichment Broth tubes (Anaerobe Systems, Morgan Hill, CA) for anaerobic cultures and Port-a-Cul medium (Becton Dickinson, Franklin Lakes, NJ) for aerobic culture.
3-month-old elk
No additional specimens.
Gross and Histopathology
Upon transport to the laboratory, all feet from each elk were examined grossly and all macroscopic lesions were graded as follows: Grade I foot lesions: Cutaneous only with coronary, interdigital skin, or heel erosions/ulcers with or without hoof capsule overgrowth and deformity Grade II foot lesions: Ulceration with undermining of the hoof capsule or heel-sole junction, with or without hoof capsule overgrowth and deformity Grade III foot lesions: Sole ulceration with inflammation/necrosis of the epidermal and dermal lamina, with or without hoof capsule overgrowth and deformity Grade IV foot lesions: Grade II and/or III lesions with breakage or sloughage of the hoof capsule, with or without hoof capsule overgrowth and deformity
All hooves from each collected elk were processed for histopathology at the Diagnostic Medicine Center at Colorado State University. Feet from 9-month-old and 3-month-old elk were also radiographed in dorsal and lateral positions and evaluated for bone and joint involvement. All feet from each elk were cut in sagittal section toe to heel on a commercial band saw to an approximate thickness of 5 mm and to include the coronary band dorsally and the heel bulb ventrally. Sections were fixed in 10% neutral buffered formalin for 24 to 48 hours and then decalcified in a solution consisting of 5% formic acid and 37% formalin (Cancer Diagnostics, Durham, NC) 7 for 24 to 48 hours until bone was pliable and easily sectioned with a scalpel. Sections were then dekeratinized by immersing in a solution of 17% potassium hydroxide and 10% formalin (StatLab, McKinney, TX) for 16 to 48 hours until keratin was pliable and easily sectioned with a scalpel. Sections of interdigital skin, heel bulb, dorsal hoof capsule/toe tip, sole and heel-sole junction, and all macroscopic lesions were processed from all feet (Fig. 1). Processed sections were paraffin embedded and trimmed to 7 to 10 μm and stained with hematoxylin and eosin (HE) for microscopic examination. Replicate sections of slides were also stained with Brown and Brenn Gram stain as described 44 and with Steiner’s silver stain for spirochetes. 14 Prepared slides were examined by light microscopy. Histopathology of formalin-fixed viscera was performed at the Washington Animal Disease and Diagnostic Laboratory (WADDL) by routine methods 42 and examined by light microscopy.
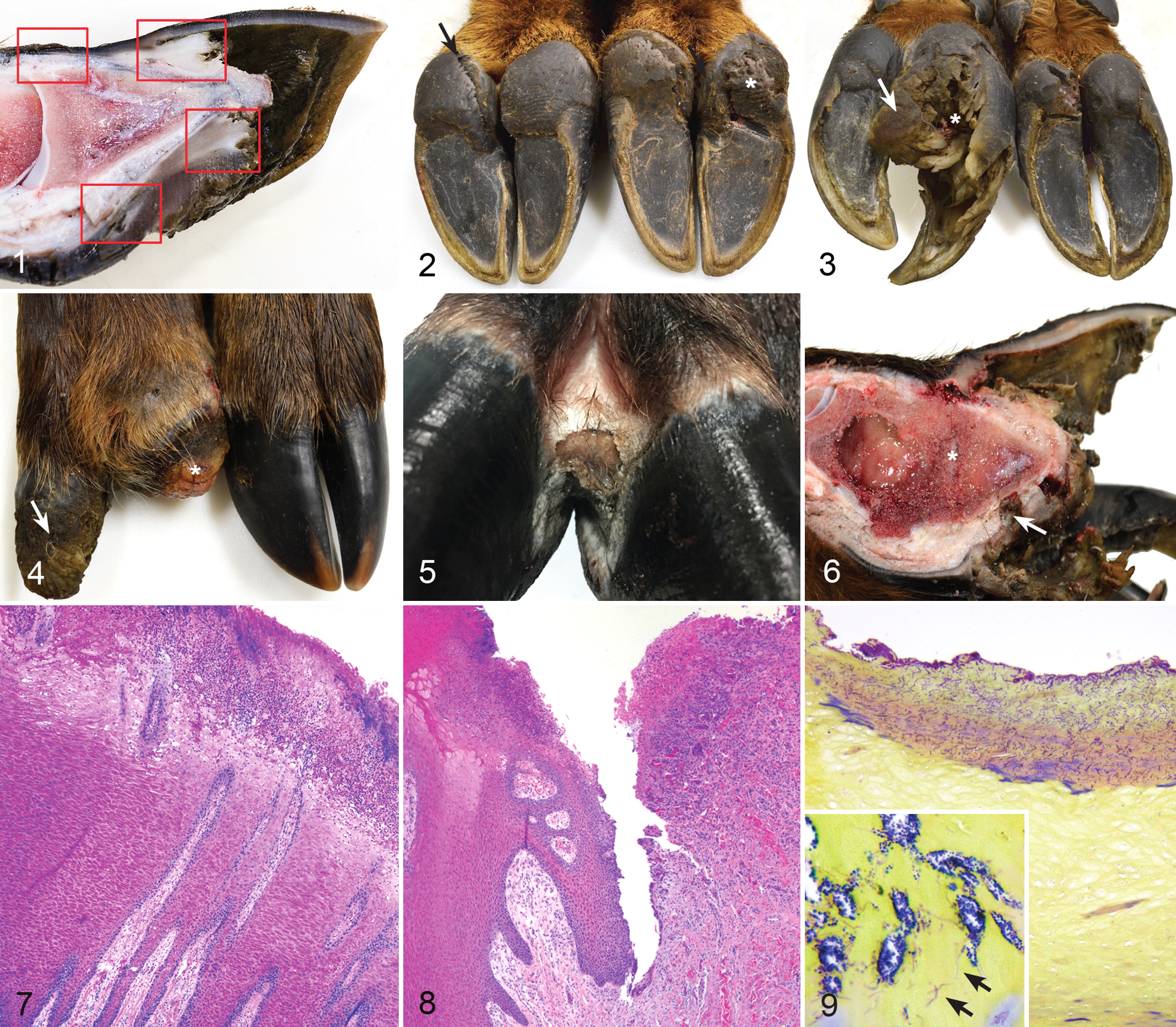
Treponeme-associated hoof disease (TAHD), feet, free-ranging 9-month-old elk (Cervus elaphus) from the enzootic region.
Immunohistochemistry
Formalin-fixed, paraffin-embedded foot lesions were processed as described above and were sectioned to a ∼4-μm thickness, deparaffinized with xylene, and blocked with 10% hydrogen peroxide and distilled water. Slides were bathed in a 0.4% solution of pepsin and 0.1% hydrochloric acid at 37°C for 15 minutes, rinsed, and blocked for 10 minutes with phosphate-buffered saline (PBS) at 37°C. Rabbit anti–Treponema denticola antibody (1:1000 dilution) (California Animal Health & Food Safety Laboratory, San Bernardino, CA) was applied for 1 hour at 37°C and washed with PBS and then probed with a 1:500 dilution of biotinylated secondary antibody conjugated to peroxidase for 20 minutes at 37°C. The sections were then rinsed with PBS, and bound peroxidase was labeled with a chromogen bath (Vector Laboratories, Burlingame, CA) for 30 minutes and counterstained with HE prior to light microscopy. 19 Negative control slides were prepared by incubating unstained slides in diluent consisting of PBS with carrier protein and homologous nonimmune sera. All sequential steps of the immunostaining procedure were performed on negative controls following incubation.
Aerobic and Anaerobic Bacteriologic Culture and 16S rDNA Polymerase Chain Reaction
9-month-old elk
Swabs from the coronary band, heel-sole junction, interdigital space, and macroscopic foot lesions were collected on each elk, placed in Port-a-cul medium (Becton Dickinson), and transported on ice overnight to WADDL for routine aerobic and anaerobic culture. Swabs were used to streak trypticase soy agar (TSA) plates with 5% sheep blood, MacConkey agar plates, Columbia blood agar (CBA) plates, and Brucella blood agar (BBA) plates, which were then inoculated 48 to 72 hours at 37°C in ambient air (TSA and MacConkey), 5% CO2 (CBA), or in an anaerobic chamber (5% H2, 10% CO2, 85% N2) (BBA). Isolated single colonies on BBA were selected and subjected to DNA extraction using conventional methods, and 16S rDNA sequences were amplified and sequenced as described. 17
7-month-old elk
Five-millimeter punch specimens of macroscopic foot lesions and contralateral healthy feet were collected as noted previously, and specimens were placed in Anaerobic Tissue Transport Medium (Anaerobe Systems) and sent to the National Animal Disease Center (NADC, Ames, IA). The vial was transferred to an anaerobic environment chamber (Coy Laboratories) with 10% H2 10% CO2 and 80% N2 environment. Tissues were disrupted by scraping with a scalpel blade in a 100 × 12 petri dish with approximately 5 ml Oral Treponeme Enrichment Broth (OTEB; Anaerobe Systems). Tissue homogenate was transferred to a 15-ml conical tube and vortexed for 1 minute. Then, 10 μl of homogenate was plated onto Brucella Agar Plates (Difco) supplemented with 5% defibrinated sheep blood, 1 ml (0.1 g/ml EtOH) vitamin K1 (w/v) (Sigma), and 1 ml (10 mg/ml) w/v hemoglobin (equine source; Sigma). Duplicate inoculated plates were incubated in plastic bags to maintain humidity for up to 7 days under aerobic conditions (37°C, ambient O2/CO2) or anaerobic conditions (37°C, 10% H2, 5% CO2, 85% oxygen-free N2). Ten to 20 unique colonies from aerobic and the anaerobic plates were selected for subsequent isolation by repeated streak plating with 24- to 96-hour growth periods depending on the isolate. Two 10-μl loops of selected bacterial colonies were placed in a sterile Eppendorf tube and DNA extracted using a Qiagen DNeasy kit following the protocol for Gram-positive bacteria (Qiagen). Full-length 16S rDNA was amplified using a universal bacterial primer set encompassing the majority of the 16S rDNA gene, resulting in a 1526-bp product. Universal PCR reactions contained 1 μl forward primer 27F (5′-AGA GTT TGA TCC TGG) (10 pmol; IDT, Coralville, IA), 1 μl reverse primer 1492 R (5′-TAC CTT GTT AYG ACT T) (10 pmol; IDT), 2.5 μl Roche 10× PCR Buffer with Magnesium (Roche, Indianapolis, IN), 100 μM dnTPs (Qiagen), 0.5 U Taq (Roche), DNA template to give 5 ng/DNA, and PCR-grade water to give a final volume of 50 μl. PCR conditions were 95°C for 5 minutes, 35 cycles of 94°C for 1 minute, 55°C for 3 minutes, and 72°C for 3 minutes, with a final extension step at 72°C for 7 minutes. Successful PCR reaction was verified by checking the size of the PCR product on 1.5% agarose gel electrophoresis. PCR reaction product was cleaned with a MinElute PCR Purification Kit (Qiagen). PCR amplification products were labeled for sequencing using ABI Big Dye Terminator v3.1 chemistries (Thermo Fisher Scientific, Waltham, MD) and then sequenced using an ABI Prism 3130xl Genetic Analyzer (Applied Biosystems/Hitachi, Forest City, CA). Bidirectional nucleotide sequences with fewer than 10 gaps in 1200 to 1600 base pairs were used for further analysis. Assembled nucleotide sequences were compared with NCBI GenBank entries using the nucleotide BLAST algorithm (http://www.ncbi.nlm.nih.gov) and the Ribosomal Database Project (https://rdp.cme.msu.edu) using Sequence Match function. Sequences were considered matched or identified based on 98% or greater similarity with 16S rDNA sequences of strains/clones in the databases.
Molecular Biology/flaB2 PCR
9- and 7-month-old elk
Five 5-μm-thick scrolls of formalin-fixed, paraffin-embedded foot specimens were selected from 9- and 7-month-old elk and were tested for Treponema by PCR at WADDL. DNA was extracted using a commercial kit (Qiagen DNeasy) with modifications as described. 88 A portion of the flagellin (flaB2) gene was amplified using pan-Treponema primers as previously described. 6 PCR and direct sequencing results were analyzed as described above.
3-month-old elk
Fresh foot specimens were collected as noted previously and shipped on ice to the University of Liverpool, Liverpool, England, for Treponema PCR with methods and results previously published. 10
Treponema Serology
All elk
At the time of death, 60 ml of whole blood was collected from each case and control elk into serum separator vacutainer tubes (BD Vacutainer SST, Franklin Lakes, NJ). Whole blood was allowed to clot and serum was separated by centrifugation (Beckman Coulter Avanti J-E with JS-5.3 rotor, 700 × g, 20 minutes, 4°C) and stored at –20°C until assayed. Treponema phagedenis, Treponema denticola, Treponema vincentii, Treponema medium, and Treponema pedis were grown in Oral Treponeme Enrichment Broth (Anaerobe Systems) and bacteria were washed by centrifugation and antigen prepared from whole-cell sonicates as described previously. 34,87 Treponeme antigen (10–20 μg protein/ml in PBS) was bound to 96-well plates (Nunc Maxisorp; ThermoFisher, Rochester, NY) overnight. Serum was serially diluted, added to plates, and incubated for 1 hour at 37°C and then at 4°C overnight. Plates were washed 3 times with PBS with 0.05% Tween-20. Bound antibody was detected by horseradish peroxidase–conjugated rabbit anti-goat IgG (H&L chain) (KPL, Gaithersburg, MD) incubated 1 hour at 37°C. Substrate was Sure Blue Reserve TMB Microwell Peroxidase Substrate (SeraCare, Milford, MA) and reaction was stopped with TMB Blue STOP solution (SeraCare). Plates were read at 650 nm on a colorimetric plate reader. Titer reported is the reciprocal of the lowest dilution with optical density greater than the threshold determined by the mean plus 2 standard deviations of the negative control (PBS) wells. Assays were performed in duplicate by a technician blinded to the age, health status, and origin of the elk. Results were analyzed and statistical analysis performed on log-transformed data (Log2) using GraphPad Prism 7 software (GraphPad, San Diego, CA) fitting a 1-way analysis of variance (ANOVA) with Tukey’s multiple comparisons to results from normal control elk. Group means were considered significant with a P value <.05.
Additional Diagnostics Performed on 9-Month-Old Elk
Viral serology
Serology was also performed on 9-month-old elk to detect antibodies to viruses known to be associated with vascular disease and hoof or coronary band lesions in cervids. Specifically, antibodies to epizootic hemorrhagic disease viruses (EHDVs) were measured by agarose gel immunodiffusion assay (AGID) as described. 57 Antibodies to bluetongue virus (BTV) were measured by a competitive enzyme-linked immunosorbent assay (ELISA) using a commercially available kit (Veterinary Medical Research and Development, Pullman, WA) performed per the manufacturer’s instructions. A cutoff inhibition value of 50% was used to determine positive results. Bovine viral diarrhea virus (BVDV) types I and II were analyzed by virus neutralization as described. 32 Malignant catarrhal fever (MCF) titers were identified first with a peroxidase-linked assay (National Veterinary Services Laboratories, Ames, IA) to detect ovine, caprine, and alcelaphine herpes virus. Briefly, the test serum was diluted at 1:20 and 1:100 in PBS and mounted on a slide prepared with MCF antigen comprising bovine turbinate cell culture infected with ovine herpes virus II. The assay was incubated 37°C for 30 minutes, rinsed, and conjugated with horseradish peroxidase–rec-Protein G (Life Technologies, Delhi, India) for 30 minutes, rinsed, and probed with a commercial peroxidase substrate kit (Vector Laboratories) per kit instructions for 8 minutes. Slides were rinsed, dried, and read within 24 hours using bovine-positive and bovine-negative controls. Positive results were then tested by virus neutralization for antibodies specific to alcelaphine herpes virus. Briefly, test and control sera were serially diluted in a 96-well plate and mixed with Earle’s cell culture minimal essential medium and alcelaphine herpes virus I, the latter of which was serially diluted 10-fold. Bovine turbinate cell cultures were suspended and incubated until confluent and then mixed with dilutions of serum and virus and incubated for 7 days at 37°C. Titers were interpreted by light microscopy by quantitating cytopathic effect of the cell culture.
Virus isolation
For all 9-month-old elk, 2 separate tissue pools were created at necropsy consisting of fresh lung, liver, spleen, kidney, mesenteric lymph node, and ileum (pool 1) and fresh specimens from the coronary band, interdigital space, heel, and any macroscopic foot lesion (pool 2). Tissue pools were chilled on dry ice for approximately 48 hours before arrival in the laboratory. Once in the laboratory, specimens were chilled at 4°C and cultured for virus isolation (Wyoming State Veterinary Laboratory). Both pools from each elk were used in 3 different tissue culture assays for attempted virus isolation. Tissue culture assays consisted of bovine embryonic testicle, 70 white-tailed deer (Odocoileus virginianus) umbilical endothelial cells, 33 and a commercially available bovine cardiopulmonary arterial endothelium assay (CPAE, No. CCL 209; ATCC Rockville, MD) with methods as outlined by the assays.
Hepatic trace mineral analysis
Five grams of fresh chilled liver were processed from 9-month-old elk to analyze for copper, selenium, cobalt, zinc, manganese, sulfur, and molybdenum levels by inductively coupled plasma mass spectrometry (ICP-MS) 1 at WADDL. Results were reported on a wet weight basis and were compared to values reported for cattle, 36 California Tule elk (Cervus canadensis nannodes), 35 and Rocky Mountain elk (Cervus elaphus nelsoni). 93
Parasitology
Feces were collected at necropsy and stored at 4°C. Specimens from 9-month-old elk were processed and examined for parasite ova using the Wisconsin sugar flotation method, lung worm larvae (Dictyocaulus sp.) using the Baermann technique, 5 and liver fluke ova (Faciola sp. or Fascioloides sp.) using sedimentation. 92 Sample preparation and light microscopy were overseen by a parasitologist.
Results
Pathology of 9-Month-Old elk
Nine elk (case Nos. 1–9), 4 females and 5 males, were collected and 5 had macroscopic foot lesions (case Nos. 1, 4, 5, 6, 9) (Table 1) that involved multiple limbs as follows: exclusively the forelimbs (n = 1), exclusively the hindlimbs (n = 3), or both fore- and hindlimbs (n = 1). All 9 elk had mild to marked pitting of the heel bulbs and coronary band with mild or marked clefting of the heel bulbs (Fig. 2). Macroscopic foot lesions included extensive sole ulcers with diffuse epidermal and dermal laminar necrosis (n = 3; a grade III lesion), abnormally long and deformed hooves (n = 2; Fig. 3), sloughed hoof capsules with exposure of the third phalanx (P3) and proliferation of ulcerated granulation tissue that covered the exposed phalanx and extended through sole ulcers (n = 2; a grade IV lesion; Figs. 3, 4), and coronary or interdigital ulcers with and without undermining of the hoof capsule and heel (n = 5; a grade I or II lesion; Fig. 5). Feet with P3 exposure also had secondary pedal osteomyelitis and purulent arthritis of the distal interphalangeal joint (n = 4; Fig. 6). One elk, case No. 9, had a single plaque-like lesion on the lateral coronary band of a hindfoot that was discretely demarcated (2.0 × 1.5 cm), ulcerated, and proliferative with no other foot abnormalities, consistent with a grade I lesion. Radiographs of the 5 affected elk showed mild to marked pedal osteitis in diseased feet only, with no joint involvement other than secondary septic arthritis in association with advanced foot disease that specifically included sole ulcers or sloughed hoof capsules. Foot radiographs of the 4 unaffected elk were normal.
Summary of Gross and Histological Foot Lesions in 9-, 7-, and 3-Month-Old Elk Collected From Elk Groups Within the Enzootic Region.
Of the 4 unaffected elk (case Nos. 2, 3, 7, 8), 1 (case No. 2) had severe septic arthritis of the carpus from a previous injury but no foot disease. No significant visceral lesions were detected in other tissues or organs in 9-month-old elk from the enzootic region.
Histopathology was performed on all 4 feet from each elk, focusing on the coronary band, dorsal hoof capsule/toe tip, sole, heel-sole junction (Fig. 1), and interdigital skin and all macroscopic foot lesions. Macroscopic lesions of the coronary band, interdigital skin, and heel bulb consisted of ulcerative, suppurative, and necrotizing dermatitis (Fig. 7) with underrunning of the hoof capsule or heel-sole junction (Fig. 8). In advanced foot lesions (4 of 5 elk), the dermal lamina was expanded with granulation tissue with abnormal long and arborizing secondary epidermal lamina (rete ridges), lymphoplasmacytic perivasculitis, and mild hypertrophic arteriosclerosis. Granulation tissue was exuberant and coated with suppurative, necrotizing inflammation with areas of reepithelization with acanthotic and eroded squamous epithelium. Most lesion specimens were associated with superficial mixed bacteria evident on HE and Gram stains and consisting of gram-positive cocci, gram-negative rods, and large gram-variable filamentous bacteria (Fig. 9, inset). Similar bacteria were also detected unassociated with inflammation in all crater-like lesions of the heel bulbs and coronary bands (Fig. 9) in diseased and nondiseased elk from the enzootic region. In 4 elk with advanced foot lesions, dense and nearly pure aggregates of fine, tightly coiled spirochetes were detected with Steiner’s silver stain at the deep advancing edge of ulcerated lesions (Fig. 10) (case Nos. 1, 4, 5, 6). Using immunohistochemistry on representative lesions from 3 of the affected elk, spirochetes were positively immunolabeled for Treponema (Fig. 11) (case Nos. 1, 4, 6). Mixed superficial bacteria were found on intact epidermis in the absence of inflammation, but spirochetes were only detected in association with suppurative inflammation and erosive/ulcerative lesions. All specimens had minimal lymphoplasmacytic perivascular inflammation within the dermal lamina (9 elk), similar to age-matched control elk. A plaque-like coronary lesion (case No. 9) consisted of well-demarcated, ulcerated, and proliferative squamous epithelium with suppurative inflammation confined to the epidermis (Fig. 12). No spirochetes could be identified with silver stains or Treponema immunohistochemistry (IHC) or flaB2 PCR within this lesion.
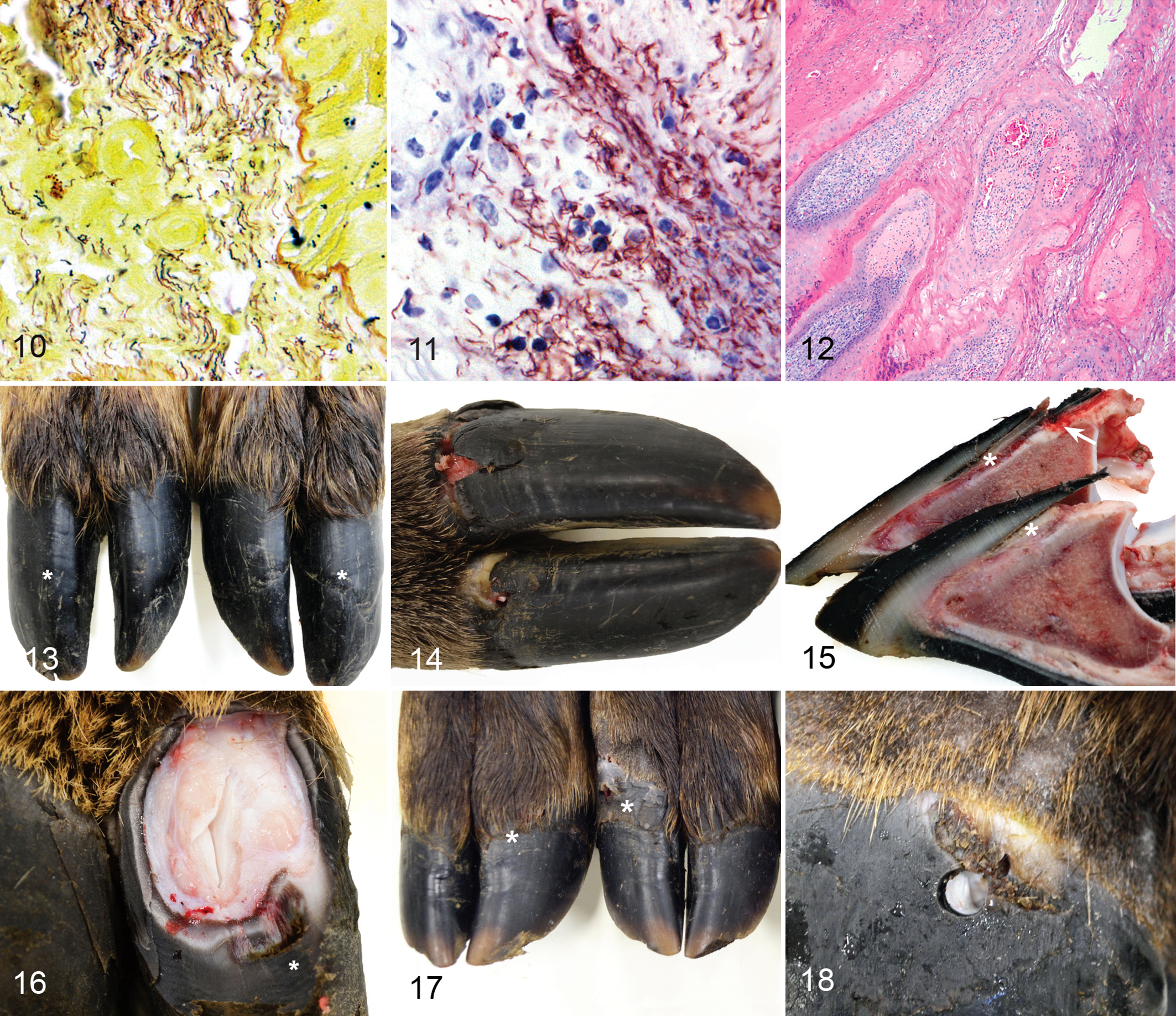
Treponeme-associated hoof disease (TAHD), feet, free-ranging 9-month-old elk (Cervus elaphus) from the enzootic region.
Grossly, control Nos. 1 to 3 (2 females and 1 male) were collected from adjacent to but outside of the enzootic region and had moderate irregular hyperkeratosis and pitting of the coronary bands and heel bulbs, similar to that described in elk from the enzootic region. Control No. 3 had mild deformity, asymmetry, and deep rings in the hoof capsule of all 4 hooves (Fig. 13), which was not associated with histologic inflammation or degeneration. Feet from control Nos. 4 to 7 (1 male and 3 females), collected from arid central Washington State, were grossly normal with no pitting or clefting of the coronary band or heel bulbs. Foot radiographs from all controls were normal with no underlying bone or joint disease detected. No visceral lesions were detected in any control elk, with the exception of control No. 6, which had nonspecific, chronic interstitial pneumonia. No ulcerative/inflammatory foot lesions and no spirochetes were detected in specimens from any control elk.
Pathology of 7-Month-Old Elk
Eight elk (case Nos. 15–22), 4 females and 4 males, had mild to marked pitting and clefts of the coronary band and heel bulb. Six elk (case Nos. 15, 16, 19, 20, 21, 22) (Table 1) had macroscopic foot lesions that occurred exclusively on the hindlimbs in 1 elk, fore- and hindlimbs in 4 elk, and all limbs in 1 elk. Two elk were grossly normal with no foot disease other than superficial pitting and clefting of coronary band and heel bulbs. Macroscopic foot lesions consisted of deep dissecting ulcers originating at the coronary band, interdigital skin, or heel bulb with underrunning of the hoof capsule and heel-sole junction with tracts containing black odiferous exudate (Figs. 14–16), consistent with grade I and II lesions. No grade III or IV foot lesions were detected in this group. Histologically, lesions were suppurative and necrotizing with local extension into the primary and secondary dermal lamina and proliferation of reactive granulation tissue. Lesion specimens, but also contralateral (healthy) foot specimens, were superficially colonized by gram-positive cocci, gram-negative rods, and gram-variable filamentous bacteria. In 5 of 6 elk with macroscopic lesions (case Nos. 15, 16, 19, 20, 21), dense aggregates of deeply invasive spirochetes were detected within suppurative erosions and ulcers using Steiner’s silver stain. Select lesions were labeled with Treponema IHC, and invasive spirochetes were positively labeled in 3 of 6 elk with foot lesions (case Nos. 16, 19, 20), with the other 3 cases considered nondiagnostic due to high background staining of the specimens. Histopathology of healthy foot specimens from the 2 unaffected elk (case Nos. 17, 18) lacked inflammation, and no spirochetes were detected by silver stain or Treponema IHC. Gross and histopathology of the viscera in all 7-month-old elk were unremarkable with no underlying disease detected. No normal control animals were collected for this group.
Pathology of 3-Month-Old Elk Calves
Five calves (case Nos. 10–14), 3 females and 2 males, were examined and macroscopic foot lesions were detected in 3 (case Nos. 11, 12, 13) (Table 1), involving exclusively the forelimbs (n = 2) or both fore- and hindlimbs (n = 1). Lesions consisted of superficial scaling and pitted lesions of the dorsal and lateral coronary band and proximal hoof capsule (n = 2) (Fig. 17). One calf (case No. 13) had discrete proliferative coronary band plaques on 2 feet (Fig. 18) similar to that described in case No. 9 (9 months old). Plaques were 1 to 2 cm in diameter, well demarcated, and ulcerated. All lesions were grade I. Two calves (case Nos. 10, 14) had grossly normal feet. Foot radiographs of all 3-month-old elk were normal, and no visceral lesions were detected.
Histologically, scaling and pitting coronary lesions were not associated with inflammation or degeneration. In all 5 calves, foot specimens included mild perivascular inflammation of the primary dermal laminae that was lymphoplasmacytic and similar to normal control elk. Coronary plaques consisted of proliferative squamous epithelium with hyperkeratosis and suppurative inflammation confined to the epidermis. No spirochetes were detected in plaques, scaling coronary lesions, or normal foot specimens from any elk in this group using Steiner’s silver stain, and Treponema IHC was negative (3 of 5 elk) or nondiagnostic due to extensive background staining (2 of 5 elk).
Two normal, age-matched control calves (control Nos. 8, 9), 1 male and 1 female, had no macroscopic, radiographic, or histologic foot lesions, and no visceral lesions were detected grossly or histologically.
Bacteriology
9-month-old elk
Aerobic and anaerobic culture were performed on foot swabs of healthy coronary band, heel bulb, and interdigital skin as well as all macroscopic foot lesions. Aerobic and anaerobic cultures from macroscopic foot lesions of five 9-month-old elk (case Nos. 1, 4, 5, 6, 9) identified Streptococcus uberis (n = 1), Actinomyces sp. (n = 1), and Fusobacterium necrophorum (n = 1) along with other mixed unidentified colonies in lower numbers (n = 5). Foot swabs from 4 unaffected 9-month-old elk (case Nos. 2, 3, 7, 8) from the enzootic region also produced cultures of S. uberis (n = 1) and Actinomyces sp. (n = 1) along with other mixed unidentified colonies in lower numbers (n = 3). Foot swabs from three 9-month-old normal control elk from southwestern Washington (control Nos. 1–3) produced cultures of Staphylococcus aureus (n = 2), Clostridium sp. (n = 1), and Peptostreptococcus sp. (n = 1) along with other mixed unidentified colonies in lower numbers (n = 1). Foot swabs from four 9-month-old normal control elk from central Washington (control Nos. 4–7) included cultures of Clostridium sp. (n = 2) and Peptostreptococcus sp. (n = 3), along with other mixed unidentified colonies in lower numbers (n = 2).
7-month-old elk
Aerobic and anaerobic cultures were performed from fresh foot specimens stored in Anaerobic Tissue Transport Media and processed rapidly under specific conditions, with 16S rDNA sequencing of isolates. Specimens included macroscopic foot lesions as well as grossly normal tissues from the heel bulb, coronary band, and interdigital skin. A total of 32 unique bacterial isolates were obtained from 8 foot specimens representing 7 elk and are summarized in Table 2. A complete listing of isolates by specimen and database match results is given in Supplemental Table S1. From healthy specimens, the most common isolates were Streptococcus and Macrococcus sp. from the class Bacilli. From lesion samples, the most common isolates were from the class Bacilli, including Staphylococcus sp., Streptococcus sp., and Macrococcus sp., and from the class Clostridia, including isolates of unidentified Firmicutes, Clostridia, and Peptoniphilus. Additional lesion isolates included bacteria of the classes Actinomycetales and Bacteroides, within which are the pigmented anaerobes Prevotella and Porphyromonas levii.
Bacterial Isolates Obtained From Foot Biopsies of 7-Month-Old Elk From the Enzootic Region.a
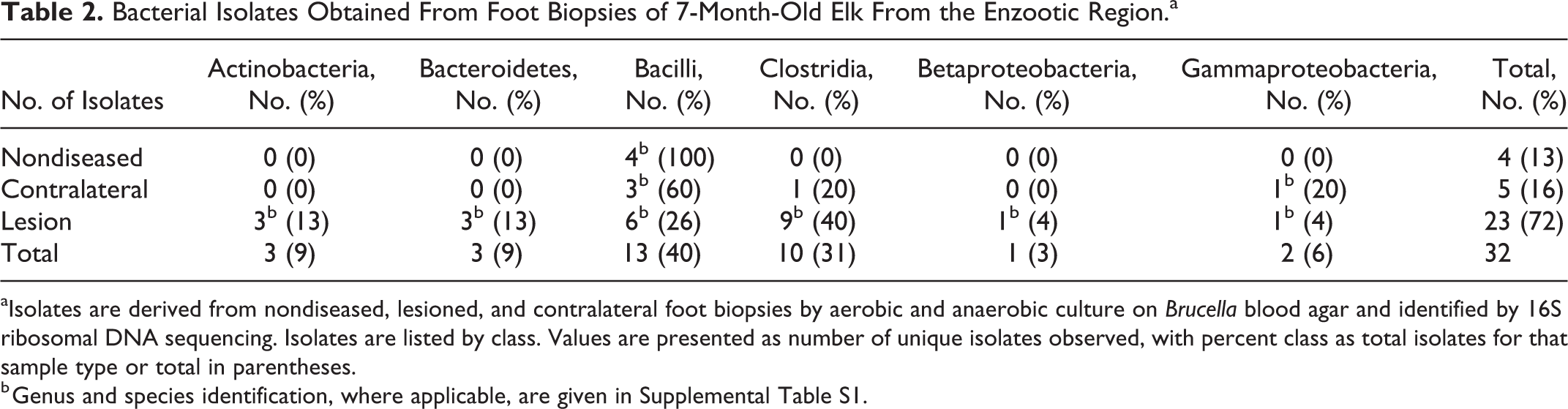
aIsolates are derived from nondiseased, lesioned, and contralateral foot biopsies by aerobic and anaerobic culture on Brucella blood agar and identified by 16S ribosomal DNA sequencing. Isolates are listed by class. Values are presented as number of unique isolates observed, with percent class as total isolates for that sample type or total in parentheses.
b Genus and species identification, where applicable, are given in Supplemental Table S1.
Treponema flaB2 PCR was performed from selected formalin-fixed, paraffin-embedded specimens of 9- and 7-month-old elk to presumptively identify spirochetes. Four 9-month-old elk from the enzootic region that had macroscopic foot lesions with spirochetes were tested (case Nos. 1, 4, 5, 6). PCR was positive for all 4, and the product sequence matched the Treponema flaB2 gene with 100% identity. PCR for Treponema was negative within a proliferative coronary band plaque from one 9-month-old elk, case No. 9, in which spirochetes were not observed histologically or with IHC.
In five 7-month-old elk, Treponema flaB2 PCR was performed on select specimens using the same formalin-fixed, paraffin-embedded specimen used for Treponema IHC. In 2 cases (case Nos. 19, 20) with foot lesions that included spirochetes and had positively immunolabeled with Treponema IHC, PCR amplified a positive product and the sequences matched Treponema flaB2 with 100% identity. Treponema flaB2 PCR was negative in 1 elk with foot lesions that included spirochetes and had immunolabeled positively with Treponema IHC (case No. 16). Three other elk with lesions and histologically visible spirochetes (with Steiner’s stain) tested negative for Treponema by IHC and flaB2 PCR (case Nos. 15, 21, 22). Two elk with no gross or histologic lesions (case Nos. 17, 18) tested negative by Treponema flaB2 PCR.
Treponema Serology
Serologic titers specific for Treponema antigens were determined for 9-, 7-, and 3-month-old elk from the enzootic region and compared to normal age-matched control elk from outside the enzootic region (Fig. 19). Serum antibody titers to Treponema antigens varied widely. The median titer for control elk (all age groups combined) was 225 (range, 23–875; n = 9). From the enzootic region, 9-month-old elk had a median titer of 555 (range, 23–3628; n = 9), 7-month-old elk had a median titer of 500 (range, 200–6400; n = 8), and 3-month-old elk had a median titer of 3225 (range, 300–3300; n = 5). All affected elk from the enzootic region did mount detectable Treponema titers with a trend toward higher titers compared to nonaffected elk from the enzootic region (Fig. 19). The group median difference between the 7-month-old elk and 3-month-old elk was statistically significant (P = .378 and .0061, respectively) from the control elk, but the median titer for the 9-month-old elk was not statistically different from control elk.
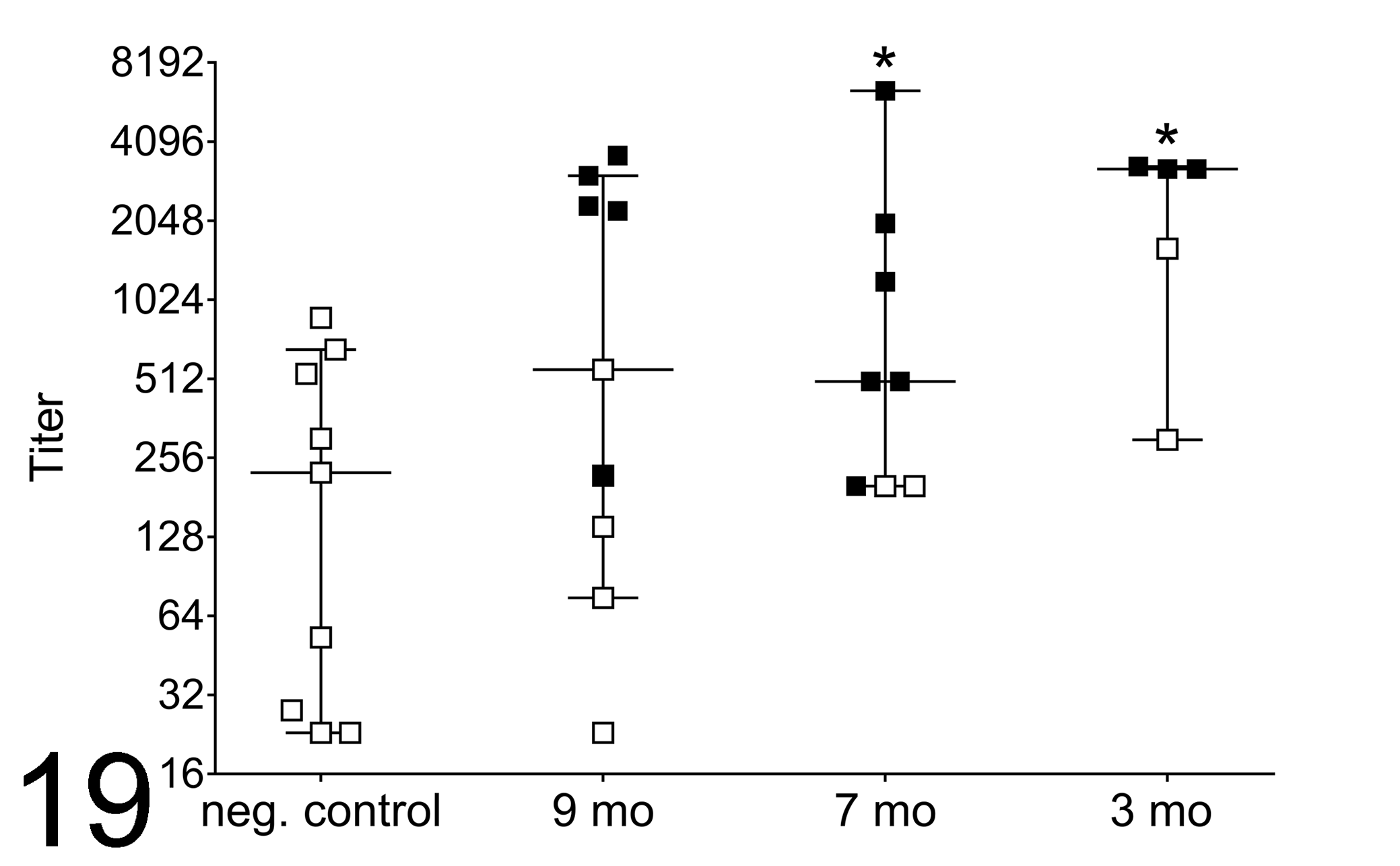
Serum antibody titers to Treponema antigen homogenate, as determined by enzyme-linked immunosorbent assay, from age-matched control elk from outside the enzootic region in southwestern and central Washington State and from 9-, 7-, and 3-month-old (mo) case elk from the enzootic region. Individual titers shown with median (horizontal bars) and 95% confidence intervals (error bars). Open squares represent elk from outside the enzootic region (negative control) and from the enzootic region with no gross or microscopic foot lesions. Black squares represent those with gross or microscopic foot lesions. *Significant differences between case and negative elk (P < .05).
Additional Diagnostics Performed on 9-Month-Old Elk
Virus isolation assays were negative from all tissue pools 1 and 2, with the exception of 2 of 7 control elk, in which an adenovirus was isolated from pool 1 in both elk (control Nos. 6, 7) and adenovirus was additionally identified in pool 2 from control No. 7.
Serologic viral titers examined included BTV, BVDV I/ II, EHDV, and MCF. No elk had detectable titers to BTV, BVDV I/II, or EHDV. Four of 16 elk had 1:20 titers to MCF using an immunoperoxidase assay but were negative for antibody to alcelaphine herpesvirus via virus neutralization assay (control Nos. 1, 3, 6). One of 4 elk with MCF-positive titers was from the enzootic region, case No. 9, an elk with a grade I proliferative coronary band plaque but no other foot lesions; 2 of 4 elk were control elk from southwestern Washington State (control Nos. 1, 3); and 1 of 4 elk was a control elk from central Washington State (control No. 6).
Hepatic mineral analysis was performed on all case and control 9-month-old elk, and adequate hepatic copper levels were identified in 1 of 9 elk from the enzootic region (case No. 1) and 1 of 7 control elk (control No. 4), compared to values (20–120 μg/g) reported for domestic cattle, free-ranging Rocky Mountain elk from Idaho, and Tule elk from California. 35,36,93 Hepatic copper deficiency was identified in 8 of 9 elk from the enzootic region, with levels ranging from 3 to 19 μg/g (mean, 7.9 μg/g), and 6 of 7 control elk, with levels ranging from 3.6 to 13 μg/g (mean, 7.2 μg/g). All 9-month-old elk were deficient in hepatic selenium compared to values reported for domestic cattle and free-ranging elk (reference range, 0.45–0.75 μg/g). 35,36,93 Elk from the enzootic region had hepatic selenium levels ranging from 0.07 to 0.1 μg/g (mean, 0.085 μg/g), and control elk had levels that ranged from 0.1 to 0.18 μg/g (mean, 0.14 μg/g). Other mineral abnormalities identified included mild elevations in hepatic molybdenum levels (reference range, 0.14–1.4 μg/g) 35,36,93 in 4 of 9 elk from the enzootic region (case Nos. 1, 4, 5, 6, 9), with a range of <1 to 1.7 μg/g (mean, 1.4 μg/g), and 2 of 7 control elk (control Nos. 5, 6), with a range of <1 to 1.6 μg/g (mean, 1.2 μg/g), as well as a mild elevation in hepatic sulfur levels (reference range, 1500–2600 μg/g) 35,36,93 in 5 of 9 elk from the enzootic region (case Nos. 1, 3, 4, 5, 6, 7), with a range of 2300 to 2900 μg/g (mean, 2611 μg/g). Seven of 7 control elk had hepatic sulfur levels within the reported reference range, 2300 to 2600 μg/g (mean, 2425 μg/g). All 9-month-old elk had hepatic levels of cobalt, zinc, and manganese within the reference ranges published for domestic cattle and free-ranging elk. 35,36,93
Parasitological examination of the feces from case and control 9-month-old elk detected parasites in low numbers in all elk and included the following: Dictyocaulus viviparous (12 of 16 elk), 1 to 47 larvae/g feces (mean, 16 larvae/g); Eimeria sp. (11 of 16 elk), 4 to 38 oocysts/g feces (opg) (mean, 11 epg); and strongyles (9 of 16 elk), 1 to 267 epg (mean, 77 epg). All 16 elk had low fecal numbers of eggs from the following parasites: Nematodirus sp., 1 to 25 epg; Trichuris sp., 1 to 10 epg; and Capillaria sp., 1 to 4 epg. A single elk from the enzootic region, case No. 2, was more heavily infected with D. viviparous (254 larvae/g feces) but had no foot disease. This animal was emaciated and incapacitated with septic carpal arthritis from prior trauma.
Discussion
The pathology of chronic and severe foot disease in elk from southwestern Washington State was previously characterized. 30 In this preliminary work, foot lesions were necrotic and heavily contaminated with no probable primary pathogens isolated using routine methods. Due to the relatively rapid spread of this disease originating from a discrete region of the Cowlitz River Basin, an infectious agent spreading among a naive population of elk was considered the most likely scenario. This second phase study was aimed at identifying infectious agents associated with foot disease and characterizing the gross and histopathology of early lesions by collecting groups of 9-, 7- and 3-month-old elk from known affected herds within the enzootic region.
Nine-month-old elk were, surprisingly, found to have advanced foot pathology, including grade III and IV foot lesions, similar to chronic disease of adult elk. 30 Lesions were strongly associated with invasive spirochetes as characterized by Steiner’s silver stains and identified as Treponema by specific IHC and flaB2 PCR. Spirochetes were, importantly, never identified in normal foot specimens of elk from the enzootic region or in specimens from control elk. This finding of an invasive spirochete highly associated with foot lesions is consistent with a true infectious agent, and we suggest the disease be referred to as treponeme-associated hoof disease (TAHD) of elk. No underlying systemic disease was detected in 9-month-old elk, similar to digital dermatitis of livestock, indicating that TAHD of elk is not secondary to other disease processes.
Seven-month-old elk from the enzootic region were found to have grade I and II lesions with ulceration and underrunning of the hoof capsule and sole, and they lacked advanced grade III and IV lesions as were detected in 9-month-old elk. Lesions were also strongly associated with invasive spirochetes identified with Steiner’s stain, although Treponema IHC and flaB2 PCR from formalin-fixed, paraffin-embedded lesions in this group were less reliable. Importantly, lesion and normal specimens included large numbers of mixed bacteria, which are thought to play an important role in the development and progression of foot disease, as it does in digital dermatitis of domestic livestock. 61,86 Decreased success of IHC and flaB2 PCR may be due to fewer numbers of Treponema in these earlier and polymicrobial lesions. Interestingly, as part of a prior study, Clegg et al 10 performed PCR and culture of fresh foot specimens from all macroscopic foot lesions in 7-month-old elk and successfully identified Treponema in most lesions, characterized to be a mixture of up to 4 phylogroups that included T. pedis, T. medium/vincentii-like, T. denticola, and T. phagadenis, as detailed in their article. Isolates were found to be genetically similar to those found in bovine digital dermatitis (BDD) and contagious ovine digital dermatitis (CODD). 9
Three-month-old calves from the enzootic region had grade I noninflammatory scaling of the coronary band, grade I proliferative coronary band plaques, and no identifiable spirochetes within lesion and healthy foot specimens using Steiner’s stains and Treponema IHC. However, fresh specimens of foot lesions and healthy foot tissues were included in the prior study of Clegg et al, 10 where Treponema culture and PCR were performed and showed that coronary lesions were indeed positive for Treponema, and organisms were determined to be highly genetically similar to the same 4 mixed Treponema phylogroups detected in 7-month-old elk, while healthy foot specimens were negative for detection of Treponema. It is speculated that these superficial lesions may indeed represent the earliest grossly detectable stage of TAHD in elk calves.
Treponema sp. are highly associated with infectious foot disease in domestic livestock, including BDD, 20,62,64 CODD, 2,3,18,75,76 and severe CODD-like disease of dairy goats, 15,77 with which TAHD of elk shares many gross and histological features. BDD was originally diagnosed in Europe 8,65 and is now globally enzootic in dairy 13 and beef cattle. 74,86 Unlike TADH of elk, in BDD, the most common lesions include proliferative and ulcerated plaques and wart-like masses, termed hairy heel warts, originating between the heel bulbs of the hindlimbs, although forelimbs may be affected as well, 62 with less common ulceration of the dorsal coronary band and sole, 16,19 the latter of which was more commonly observed in TAHD. Affected cattle are markedly lame with loss of body condition, decreased milk and meat production, and decreased fertility. 4,62,66 Similar to TAHD, histologic BDD lesions consist of suppurative and ulcerative dermatitis and acanthosis 62,64 with invasive spirochetes detectable with silver stains and Treponema IHC. 62 PCR of BDD-associated Treponema includes a mixture of multiple phylogroups, including T. pedis, T. medium/vincentii-like, T. denticola, T. phagadenis, and Treponema brennaborense, 20,37,38,52,55,83 similar to that detected in TAHD of elk. 10
CODD was first described as a severe debilitating foot disease of sheep in the United Kingdom in 1997. 31 CODD is highly associated with deeply invasive Treponema sp. consisting of similar mixed phylogroups as described in cattle, including T. pedis, T. medium/vincentii-like, T. denticola, and T. phagadenis. 3,18,37,68,75,76 CODD-like disease has recently been described in domestic dairy goats with severe progressive lesions similar to CODD and TAHD and included the same mixed phylogroups of Treponema. 15,27,77 CODD lesions are grossly and histologically nearly identical to TAHD of elk and manifest early as ulcers of the coronary band, interdigital space, and heel bulb with underrunning of the hoof capsule and heel-sole junction, 2,3,18 with chronic lesions that include severe sole ulcers and sloughing of the hoof capsule with abnormal regrowth of a distorted and partially avulsed hoof capsule. 2,3,18
Through this current study, we have developed a system of lesion grading for TAHD of elk from I to IV. Grade I foot lesions are cutaneous only with coronary, interdigital skin or heel erosions/ulcers with or without hoof capsule overgrowth and deformity (Fig. 5). Grade II lesions include undermining of the hoof capsule or heel-sole junction, with or without hoof capsule overgrowth and deformity (Figs. 14–16). Grade III lesions include inflammation/necrosis of the epidermal and dermal lamina with sole ulceration, with or without hoof capsule overgrowth and deformity (Figs. 3, 6). Grade IV foot lesions include grade II and/or III lesions with breakage or sloughing of the hoof capsule, with or without hoof capsule overgrowth and deformity (Fig. 4). The exact development and progression of TAHD lesions remain unknown, although this study would suggest grade II lesions may occur as early as 7 months of age with rapid progression to grades III and IV by 9 months of age. Development of TAHD lesions from incipient, as seen in 3-month-old elk calves, to more advanced grade II and higher, occurred in a timeframe consistent with that observed for lesion development in BDD, in which naturally infected adult dairy cattle progressed from incipient stages to clinical lesions in 133 days (mean time of 4.5 months). 39
Antibody ELISA was used to demonstrate that TAHD-affected elk were mounting a significant humoral immune response specific for Treponema antigens. High titers in 3-month-old elk calves from the enzootic region were thought to also reflect immune status of the dam during this active nursing phase. As observed with BDD, there is considerable individual variability in measurable titers within the control groups, which illustrates the limitations of using serology alone as a diagnostic tool. 86 Interestingly, since the 2013 and 2014 sampling, groups of elk from the control region outside of the enzootic region have been confirmed with TAHD (Mansfield, Han, and Wilson-Welder, unpublished data), and elevated Treponema antigen titers of control elk found during this study may indicate that TAHD was actually present but not clinically detected in these presumed negative regions.
An important question is the role of other bacteria in the pathogenesis of TAHD of elk. Digital dermatitis of domestic livestock is a known polymicrobial disease. While many phylogroups of Treponema are associated with the lesions, aerobic and anaerobic bacteria are also routinely found. 61,86 In 16S rDNA sequencing microbial profile studies of BDD, the phyla Spirochaetes, Bacteroidetes, Actinobacteria, and Firmicutes are consistently associated with digital dermatitis lesions, 40,47,53,67,90,94 while Firmicutes and Proteobacteria tend to be dominant in healthy foot specimens. In the present study of TAHD, foot specimens from 7-month-old elk in the enzootic region were most commonly associated with the black-pigmented Porphyromonas, P. levii, Prevotella, Fusobacteria, Bacteroides, and Peptoniphilus, all of which are strongly associated with BDD foot lesions 51,53 and are important cofactors in other tissue-destructive diseases such as foot rot, 78,84 soft tissue infections/abscesses, and periodontitis. 21,43,54,69,82 Interestingly, these organisms are also part of the normal rumen microflora of cattle, 28,46 and it is thought that Treponema pass through feces, contaminate feet, and ultimately result in deep and underrunning foot lesions. Some of the other isolates, such as Luteococcus, which were found in lesions but not in healthy tissue, are also associated with opportunistic infections. 11,41,48 Streptococcus uberis was among the most common isolates from both healthy and lesion foot specimens. This opportunistic pathogen is typically associated with mastitis in dairy cattle; 59,91 however, the presence of S. uberis in this study and in other observations of chronic foot disease of wildlife (lameness in white-tailed deer from Kansas with enzootic hemorrhagic disease lesions complicated with foot rot; Wilson-Welder, unpublished data) suggests that S. uberis is widespread in the environment and is generally associated with animal skin. 85 While the initiating event remains elusive, the polymicrobial colonization of foot lesions in TAHD of elk and BDD most likely contributes to further tissue destruction and failed healing, as seen with polymicrobial infection of other chronic wound conditions. 50,60,80
The mode of transmission of TAHD between elk is not known and would likely require controlled laboratory studies to elucidate. BDD and CODD are assumed to be transmitted via environmental and limb contamination spread by diseased feet and fecal slurry. 38,86 Attempts to reproduce BDD in healthy cattle have been successful with provision of a moist anaerobic environment and inoculation of foot lesion material, composed of mixed Treponema sp. phylotypes and secondary polymicrobial infection. Attempts to induce BDD with pure culture of a single Treponema species have been less successful. 26,40,52,63,75,86 Importantly, elk from southwest Washington are subject to wet conditions year-round as this maritime region receives close to 60 inches of precipitation per year 23 (National Oceanographic Atmospheric Administration, unpublished data). Particularly during the wettest and coldest seasons, large groups of elk gather in valley bottoms, where they share pasture land with domestic cattle in which BDD and other microbes causing infectious foot disease are known to occur. In addition, elk within the enzootic region use mountainous and densely forested timberlands interspersed with commercial clear-cuts that collectively consist of slash piles, downed trees and limbs, and coarse woody debris, all of which are subjectively thought to cause additional foot trauma. Macroscopic pitting and clefting of the coronary band and heel bulb were detected in most elk from the enzootic region and control elk from southwestern Washington State habitats adjacent to the enzootic region. Importantly, control elk from the more arid central Washington region did not have pitting and clefting lesions of the feet. Pits and clefts in the coronary band and heel bulb were histologically associated with mixed superficial bacteria but not inflammation and may represent damage due to maceration of the feet, which could be an important risk factor for the initiation and development of TAHD.
As with most wildlife diseases, the susceptibility of elk from southwestern Washington to TAHD is likely multifactorial. Importantly, elk in the enzootic region were documented to be in poor nutritional condition compared to other groups of elk throughout the state, 12 with food limitation likely being a function of elk density within this habitat. 49 Poor overall health may have been an important risk factor for infection and transmission of TAHD in this particular group of elk. As determined in this and our prior study, elk from southwestern Washington are also deficient in hepatic selenium and copper, 30 which may have important consequences for cellular and humoral immune response, skin and wound healing, 24,58,73,79,89 control of oxidative stress and neutrophil function, and integrity of the hoof capsule, leading to susceptibility to a variety of foot diseases as described in domestic cattle, sheep, and free-ranging moose. 22,25,29,56,71,72,81 Interestingly, selenium and copper deficiency in affected elk from the enzootic region was similar to that detected in normal control elk from central Washington State, where TAHD has not been detected to date. As an additional consideration, the first cases of TAHD were recognized in early 2008 within the Boistfort Valley of the Cowlitz River Basin in the heart of the enzootic region, following a period of severe flooding. During these floods, much habitat for both domestic livestock and wildlife was destroyed, with floodwater and mud killing hundreds of cattle (The Seattle Times, printed December 13, 2007). Although speculative, this may have been a significant opportunity for introduction of a novel digital dermatitis-like disease (TAHD) into a naive and stressed free-ranging elk population.
In conclusion, this study shows that severe foot disease in free-ranging elk from southwestern Washington State is associated with an infectious agent consisting of an invasive Treponema sp. spirochete in combination with a diversity of aerobic and anaerobic bacteria similarly described in digital dermatitis of domestic livestock. Superficial macroscopic foot lesions were detected in 3-month-old calves, with deep invasive foot lesions manifesting at 7 months of age and advanced chronic foot lesions, similar to adult elk, noted at 9 months of age. Better understanding of the dynamics and pathogenesis of TAHD within affected groups of elk and better understanding of risk factors predisposing elk to TAHD are paramount to future monitoring and management of this disease.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818798108 - Treponeme-Associated Hoof Disease of Free-Ranging Elk (Cervus elaphus) in Southwestern Washington State, USA
Supplemental Material, DS1_VET_10.1177_0300985818798108 for Treponeme-Associated Hoof Disease of Free-Ranging Elk (Cervus elaphus) in Southwestern Washington State, USA by Sushan Han, Kristin G. Mansfield, Dan S. Bradway, Thomas E. Besser, Deryck H. Read, Gary J. Haldorson, David P. Alt, and Jennifer H. Wilson-Welder in Veterinary Pathology
Footnotes
Acknowledgements
A sincere thank you to the hardworking employees of Washington Department of Fish and Wildlife for assistance with elk collection and sample processing, specifically Patrick Anderson, Stefanie Bergh, Dan Chadwick, Brooke George, Scott Harris, Daren Hauswald, Brock Hoenes, Ted Holden, Eric Holman, Bret Hopkins, Dyanna Lambourn, Aaron Marthaller, Lance Martin, Warren Michaelis, Pat Miller, William Moore, Anthony Novack, Katie Perez, Ella Rowan, Anna Sample, Scott Schroeder, Nicholle Stephens, and Bob Weaver, as well as Sandra Jonker, Jerry Nelson, and Dave Ware for administrative and logistical support; Julia Burco from the Oregon Department of Fish and Wildlife for assistance with field necropsies; microbiology technicians at USDA-ARS; and the Colorado State University histology laboratory, specifically Todd Bass, whose dedication helped prepare every hoof slide. Many thanks to the researchers of the Evans laboratory at the University of Liverpool for sharing their expertise in digital dermatitis and for their hard work at rapidly identifying and typing Treponema within these complex foot lesions.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Mention of trade names or commercial products in this study is solely for providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Washington Department of Fish and Wildlife as well as a grant from the Rocky Mountain Elk Foundation, USA. Portions of this work were completed by US Department of Agriculture employees in the course of their assigned duties in relation to project number 5030-32000-223-00-D.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
