Abstract
Oral fibroepithelial polyps (FEPs) are common, benign, nonneoplastic lesions in humans that often develop slowly in sites of local irritation or trauma. This study analyzed 23 oral fibroepithelial polypoid lesions retrieved from 21 dogs (2014–2021). All lesions were pedunculated with usually an irregular/cauliflower-like or rarely smooth surface. FEPs most commonly arose under or lateral to the tongue; other sites included the labial and gingival mucosa, soft palate, and hard palate. All the lesions were characterized by a thick fibrovascular stalk consisting of bundles of fibrocytes and fibroblasts embedded in a collagenous matrix rich in blood vessels. The surface squamous epithelium, when evaluable, was hyperplastic (22/22; 100%) with frequent parakeratotic hyperkeratosis (12/22; 54.5%). Ulceration of variable extent was observed in 13/23 cases (56.5%). Inflammation was associated with 18/23 cases (78.3%), and was mostly lymphoplasmacytic. The connective tissue was consistently immunoreactive for vimentin and generally negative for smooth muscle actin and desmin. All FEPs in cases with available clinical outcome data did not recur after surgical excision. The presence of chronic inflammation and ulceration suggests a causative role of chronic irritation in the pathogenesis of canine oral FEPs. FEPs should be included among the differential diagnoses of proliferative lesions of the oral cavity in dogs.
Oral fibroepithelial polyps (FEPs) are common lesions diagnosed in the oral cavity of humans, and represent 0.3–5% of oral and maxillofacial pathology samples.1,4,13,15 –17 FEPs are benign mucosal lesions, most commonly arising from the tongue, buccal mucosa, labial mucosa, and hard palate, that often develop in response to trauma or frictional irritation.4,17,19,25 In the literature, other synonymous terms are found, such as focal fibrous hyperplasia, irritation fibroma, traumatic fibroma, and chewing granuloma.4,5,19,21,25 FEPs usually appear as sessile or pedunculated, firm, pink masses. Histologically, they are characterized by a core of dense fibrous connective tissue associated with vascular dilatation and proliferation, covered by keratinized squamous epithelium.4,14,17,25 Surgical excision is considered the treatment of choice for FEPs, and recurrence is unlikely unless the inciting trauma is repeated. 4
In dogs, fibroepithelial polyps are well-recognized entities of the skin, as common hyperplastic lesions characterized by a polypoid collagenous core, sometimes richly vascularized, covered by epidermis. 12 In addition to the skin, FEPs have been uncommonly reported in other anatomical sites, such as the vagina, 3 the ureter,6,7,23 and the urethra. 10
This study describes a series of oral proliferative lesions in dogs which share common features with human oral FEPs.
Materials and Methods
The Pathology division archive of the San Marco Veterinary Laboratory was retrospectively screened (2014–2021) for oral polypoid proliferative lesions. Inclusion criteria included canine, exophytic/polypoid oral lesions, histologically characterized by a proliferation of connective tissue and epithelial hyperplasia, for which formalin-fixed paraffin-embedded (FFPE) tissue blocks were available. The used search keywords included the terms “oral,” “oral cavity,” “tongue,” “polyp,” “fibroepithelial,” “fibrovascular,” “granulation tissue,” and “hyperplasia.” The histological reports of the selected cases were reviewed, and cases diagnosed with other known entities, such as pyogenic granuloma, were excluded. The archive screening identified 25 cases, which were histologically reviewed. Two of these cases were excluded because they were considered primary inflammatory lesions (stomatitis/glossitis). Among the 1620 samples of canine oral cavity present in the archive, 23 cases (1.4% of oral samples) proposed to be canine oral FEPs were retrieved.
For each case, the signalment and anamnestic data were collected, including breed, sex, age, anatomical site of the lesion, and type of biopsy (incisional/excisional). The referring veterinarians were contacted by email and/or phone call, and the available outcome data were collected.
Slides were stained with H&E and Masson–Goldner trichrome (Bio Optica; Milan, Italy). Immunohistochemistry for vimentin, alpha smooth muscle actin (αSMA), and desmin were performed on tissue sections using an automated immunostainer (Bond RX, Leica; Wetzlar, Germany). Briefly, sections were incubated with anti-vimentin (1:1000; clone V9, Agilent Dako; Santa Clara, CA, United States), anti-αSMA (1:2000; clone 1A4, Agilent Dako; Santa Clara, CA, United States) and anti-desmin (1:200; clone D33, Histo-line, Pantigliate, Italy) antibodies following heat-induced epitope retrieval. The tunica media of blood vessels and a sample of skeletal muscle were adopted as positive controls; negative controls were carried out omitting the primary antibodies. The signal was retrieved through a polymer detection system (BOND Polymer Refine Detection; Leica; Wetzlar, Germany) containing a peroxide block, post primary, polymer reagent, and DAB chromogen, followed by hematoxylin counterstaining.
The sections were jointly examined by 2 ECVP boarded pathologists (LN and SG). Histologically, the lesions were classified as to their subgross appearance (pedunculated, irregular or cauliflower-like, or smooth), and their maximum histological dimension was measured. All lesions were evaluated for fibrous and vascular proliferation, cellular atypia, epithelial ulceration, epithelial hyperplasia, and epithelial hyperkeratosis. The fibrovascular proliferation was evaluated quantifying the fibrous-to-vascular ratio, considering both the areas along the stromal axis and the areas close to the ulcers. The presence of cellular atypia was evaluated in the proliferating spindle cells in areas far from the ulcers as: absent; mild, with uniform or regular nuclei and occasional nucleoli; moderate, with moderate degree of variation in nuclear size and shape, hyperchromatic nuclei, and prominent nucleoli; and severe, with marked variation in nuclear size and hyperchromatic nuclei, with one or more prominent nucleoli. 8 The inflammation was further evaluated as to the cell type (e.g. lymphoplasmacytic, macrophagic, neutrophilic, mastocytic) and site of involvement (stalk-inflammation, ulcer-related inflammation, or both). The inflammatory infiltrate was also semi-quantitatively scored as: 0 (absent), when no increase in inflammatory cells was detected; 1 (mild), when the inflammatory cells obscured less than 10% of the surface of the section; 2 (moderate), when the inflammatory cells obscured 11–50% of the surface of the section; 3 (severe), when the inflammatory cells obscured more than 50% of the surface of the section. The ulceration was semi-quantitatively scored as: 0 (absent), when the mucosal epithelium was intact; 1 (mild), when less than 10% of the mucosal epithelium was interrupted; 2 (moderate), when about 11–50% of the mucosal epithelium was interrupted; 3 (severe), when more than 50% of the mucosal epithelium was interrupted. Epithelial hyperplasia and hyperkeratosis were evaluated as present or absent.
Results
All of the reviewed cases were originally diagnosed as proliferative reactive lesions characterized by newly formed fibrovascular tissue, sometimes with evident granulation tissue in superficial areas (11/23; 47.8%), associated with epithelial hyperplasia and variable ulceration and inflammation.
The 23 cases belonged to 21 dogs (2 dogs had 2 different polypoid lesions in the oral cavity). Dogs ranged from 3 to 15 years of age, with a mean age of 8.9 years. Fourteen dogs were male (66.7%; of which 11 were intact, 2 neutered, and 1 not further specified), seven were female (33.3%; of which 5 were spayed, one intact, and one not further specified). Mixed-breed dogs were the most represented (6 dogs; 28.6%), followed by Dachshund, Maltese, and Pomeranian dogs (2 each; 9.5%) and other pure breeds (one [4.8%] of each of the following breeds: Australian Shepherd, Bichon Frisé, Boston Terrier, Flat-coated Retriever, Lhasa Apso, Pit bull, Rottweiler, Shih Tzu, and Zwergpinscher).
Most of the lesions arose under the tongue (13/23, 56.5%) or on the lateral surface of the tongue (1/23, 4.3%) (Fig. 1a). Of the 13 samples under the tongue, 6 arose on the ventral surface of the tongue; 2 were close to the base of the tongue, and 1 was close to the frenulum. In 4 cases, the anamnestic data only indicated “sublingual” location. Other frequently reported sites were the mucosa of the lip (4/23, 17.4%) and the gingiva (2/23, 8.7%), whereas other sites included the soft palate (1/23, 4.3%) and the hard palate (1/23, 4.3%). For 1 case, the specific site of the oral cavity was not reported. Two dogs had 2 lesions each; one having 2 different FEPs under the tongue and one having a FEP on the labial mucosa and 1 on the gingival mucosa.
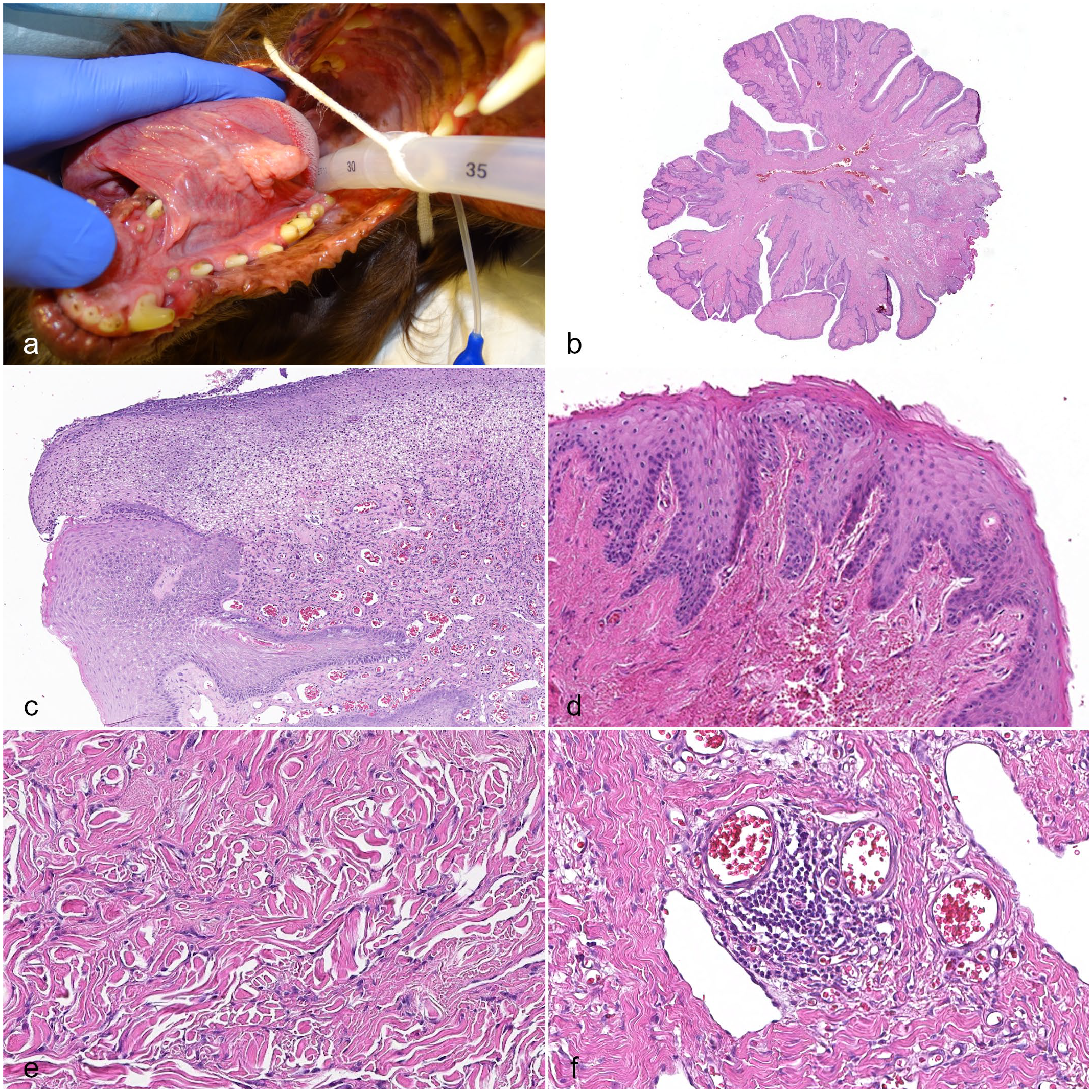
Fibroepithelial polyp (FEP), oral cavity, dog: (a) macroscopic presentation of a FEP on the ventral surface of the tongue, intra-operative view. (b) Pedunculated cauliflower-like subgross appearance, hematoxylin and eosin (HE). (c) Mucosal ulceration obscured by numerous neutrophils and fibrin; this finding has to be interpreted in the context of the lesion to be differentiated from pyogenic granuloma, HE. (d) Irregular epithelial hyperplasia with hyperkeratosis, HE. (e) Collagenous fibrous stalk, HE. (f) Perivascular lymphoplasmacytic inflammation, HE.
Among the 23 cases, 20 cases were complete excisional biopsies and 3 cases were incisional biopsies. For the excisional biopsies, the lesions ranged from 0.4 to 2.5 cm in maximum dimension in histological sections, with an average of 1.1 cm. All the excised lesions were pedunculated (20/20, 100%), most commonly with an irregular (12/20, 60%) to a cauliflower-like surface (4/20, 20%), or less commonly with a smooth surface (4/20, 20%) (Fig. 1b). All lesions were in contiguity with the adjacent oral mucosa, which was raised by the FEP without a clear demarcation.
On histopathology, all lesions consisted of a broad fibrovascular stalk covered by stratified squamous epithelium. The stalk was characterized by interlacing and parallel bundles of well-differentiated fibrocytes and fibroblasts, variably embedded in an extracellular matrix (Fig. 1e) that stained deep green with Masson–Goldner trichrome, consistent with collagen (Fig. 2b). Significant atypia was not detected. These bundles were admixed with numerous small- and medium-caliber blood vessels, which were often mildly congested. The vascular proliferation was observed both along the fibrovascular axis of the polyp and in areas close to the ulcers. The fibrous component was constantly prevalent over the vascular component, representing about 60-90% of the stroma, with a fibrous-to-vascular ratio that approximately ranged from a minimum of 3:2 to a maximum of 9:1. The ratio was lower in areas close to the ulcers, which were characterized by a densely cellular proliferation of small-caliber blood vessels, lined by plump endothelium, and frequently associated with fibroblasts, consistent with immature granulation tissue. These areas blended with the more heavily collagenous areas of the stalk . Multifocally, in 12/23 cases (52.2%), the interstitium was expanded by scant extracellular amorphous material (edema).
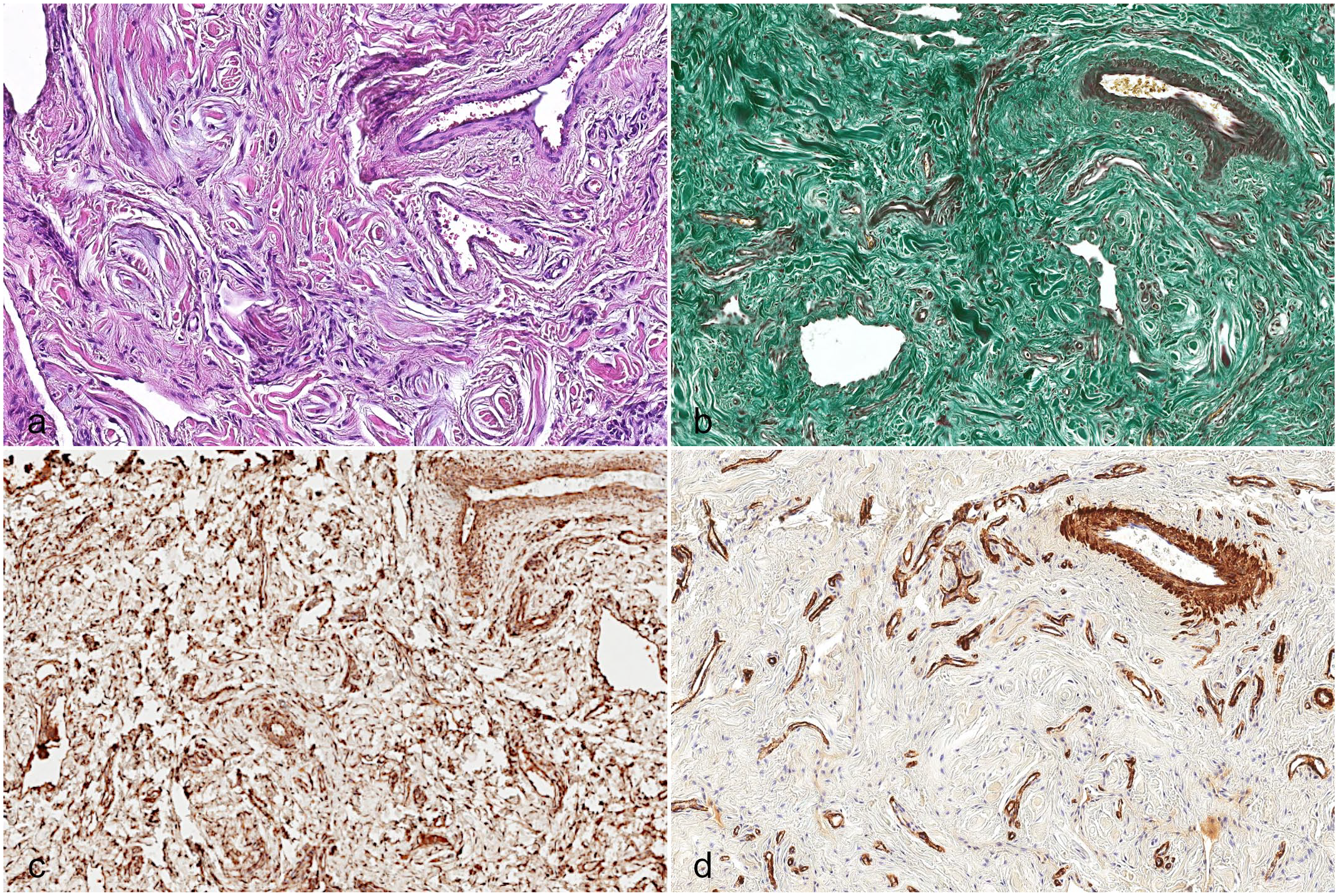
Fibroepithelial polyp, oral cavity, dog. (a) Hematoxylin and eosin (HE); (b) extracellular matrix is stained deep green, consistent with collagen. Masson–Goldner trichrome. (c) Spindle cells in the supporting stroma are consistently vimentin-positive, immunohistochemistry for vimentin. (d) The tunica media of blood vessels is alpha-smooth muscle actin (αSMA) positive, whereas the stroma is negative, immunohistochemistry for αSMA.
The surface epithelium was ulcerated in 13/23 cases (56.5%) (Fig. 1c). The degree of ulceration was evaluated as 1 (mild) in 3 cases, 2 (moderate) in 4 cases, and 3 (severe) in 6 cases. The intact epithelium, evaluable in 22 cases, was always moderately to severely hyperplastic (22/22 cases, 100%) with associated hyperkeratosis, mostly parakeratotic, in 12 cases (54.5%) (Fig. 1d). No viral inclusions were detected. The presence of an inflammatory infiltrate was observed in 18 cases (18/23, 78.3%). The inflammatory infiltrate was strictly associated with the area of ulceration in 3 cases (3/23, 13%); in 15 cases, it involved the stalk of the polyp (15/23, 65.2%), either in the absence (7 cases) or presence of ulceration (8 cases). The inflammation of the core of the polyp ranged from mild to moderate, was mainly perivascular to interstitial, and consisted of lymphocytes and plasma cells (15/15), sometimes accompanied by fewer macrophages (in 6 cases) or few neutrophils (in 1 case) (Fig. 1f). The inflammation associated with the ulcers was moderate to severe and involved mostly neutrophils and macrophages with smaller number of lymphocytes and plasma cells.
In summary, the nonulcerated areas of the polyps were characterized by a mature, variably dense fibrovascular tissue, multifocally accompanied by a mild, perivascular, lymphoplasmacytic inflammatory infiltrate; the ulcerated areas were more frequently characterized by a densely cellular, immature granulation tissue associated with a more severe inflammatory infiltrate, which was mostly neutrophilic. The clinical and histologic data are summarized in Supplemental Table S1.
Immunohistochemistry revealed that the supporting stroma was composed of consistently vimentin-positive (23/23, 100%) spindle cells. These cells were mostly negative for αSMA and desmin, except for mild, multifocal positivity for αSMA in two cases and desmin in 1 case. In addition, αSMA labeled the tunica media of scattered blood vessels in all samples (Fig. 2).
Clinical outcome data were available for a subset of 14 dogs. Eleven dogs were alive at the time of writing, but no further information on the status of the oral cavity of 2 of them was provided. Three dogs died of unrelated causes in a period of 2 months to 4 years after the diagnosis of FEP. Eleven of these dogs had no recurrence of the polyp in a follow-up period, which ranged from 2 months to 4 years after diagnosis. One of the dogs with concurrent FEPs (dog no. 21, having undergone the removal of 2 different sublingual FEPs at the same time) developed a new polypoid lesion on the contralateral side of the tongue at the opening of the salivary duct 6 months later. The surgery to remove the first FEPs revealed that the lesions arose at the opening of the mandibular salivary duct, which appeared blocked. Many veterinarians reported that polyps were incidental findings. In 2 dogs, FEPs were noticed when the dogs were brought in for exam because of halitosis (case 20) or chewing difficulties (case 16). In 2 cases, the owners noticed the presence of the oral neoformations (cases 17 and 18). In the other cases, FEPs were reportedly incidental findings in dogs undergoing medical procedures or anesthesia for various reasons such as castration, diagnostic imaging, or dental cleaning/removal (cases 3, 7, and 20).
Discussion
All the described FEPs consisted of a proliferation of fibroblasts and fibrocytes often associated with vascular proliferation, epithelial hyperplasia, and ulceration. Significant atypia was not detected, and the newly formed fibrovascular tissue was consistent with a reactive change. The multifocal, mild immunoreactivity to αSMA suggests the presence of reactive myofibroblastic cells in the stroma.
The common presence of ulceration and inflammation, together with the epithelial hyperplasia and hyperkeratosis, support the hypothesis that trauma could be the eliciting cause of FEPs. In fact, the most common sites of the FEPs were friction points of the oral cavity or sites prone to direct self-induced trauma, especially the mucosa under the tongue. Traumatic lesions may be secondary to masticatory trauma or malocclusion, fractured or displaced teeth, or self-induced occlusal trauma associated with mucosal swelling. 21 However, the underlying cause of the trauma may not be evident in the surgical biopsy; therefore, good medical anamnesis and careful evaluation of the oral cavity are recommended. Surgical excision of traumatic lesions and the resolution of the triggering cause are generally considered resolutive.4,21 For example, in case 21, a dog that developed multiple lesions on both sides of the tongue, reportedly associated with a blocked opening of the mandibular salivary gland duct, was cured after surgical removal of the FEPs.
Indeed, oral FEPs in the veterinary field have historically been colloquially referred to as “chewing granulomas,” a misnomer referring to proliferative granulation tissue arising in sites of “chewing” trauma, such as the vestibular buccal mucosa and lateral lingual/sublingual mucosa,5,21 and not to granulomatous inflammation. The ulcerating/traumatic primary event may result in acute neutrophilic inflammation accompanied by edema, vasodilation, and congestion, followed by a fibrovascular proliferative response forming exophytic nodules of exuberant granulation tissue. Exuberant granulation tissue may occur with unsuccessful attempts to re-epithelialize ulcerated areas, which are too large, or when the eliciting injury is persistent or repetitive. 21
Altogether, the morphological features (fibrovascular and epithelial reactive proliferation with expansile growth and lack of atypia) and the clinical outcome (with no reported recurrences or aggressive behavior) suggest that oral FEPs in dogs, similar to FEPs in humans, are benign reactive lesions.
Clinical differential diagnoses for oral FEPs include both inflammatory and proliferative nonneoplastic and neoplastic lesions. FEPs should be differentiated from pyogenic granuloma, gingival hyperplasia, peripheral odontogenic fibroma, nonodontogenic fibroma and fibrosarcoma, squamous papilloma, and viral papilloma. Pyogenic granuloma is a reactive inflammatory lesion which has been described in detail in cats and has also been sporadically reported in dogs.22,24 Pyogenic granuloma macroscopically presents as a reddish mass arising most commonly on the buccal mucosa close to a mandibular molar, frequently associated with dental malocclusion. Histologically, it is densely vascular granulation tissue with edema and neutrophilic inflammation associated with epithelial ulceration and necrotic debris.2,21 It can be either dominated by an inflammatory pattern, with abundance of lymphocytes and plasma cells and lesser neutrophils, or by hyperplastic tissue composed of granulation tissue with neovascularization and activated fibroblasts. 9 Grossly, pyogenic granulomas appear redder than FEPs, and usually involve the buccal mucosa adjacent to a mandibular molar tooth, while the lateral aspect of the tongue is seldom affected. 21 Compared with FEPs, pyogenic granulomas are histologically characterized by prominence of the inflammatory component and vascular proliferation, which can be extensive enough to mimic a hemangioma of the granulation tissue/lobular capillary subtype. Moreover, the newly formed vessels and collagen fibers in pyogenic granulomas frequently have a perpendicular orientation, with respect to the mucosal surface (granulation tissue), or lobular orientation. 21
Gingival hyperplasia and peripheral odontogenic fibroma typically arise in the gingiva, mostly in association with teeth. The former is histologically characterized by the proliferation of stromal connective tissue, covered by a proliferating epithelium and coarse, dense, mature collagen arranged in bundles, with few fibrocytes. 20 Peripheral odontogenic fibroma has distinguishing features of ectomesenchyme derivation, including the presence of large vessels—reminiscent of those of the periodontal ligament—and often variable amounts of odontogenic epithelium;18,20 peculiar morphological features that easily differentiate it from FEPs.
Fibromas are expansile and well-circumscribed growths, characterized by interlacing bundles of collagen associated with few well-differentiated fibrocytes.
Fibrosarcomas are infiltrative and locally destructive. 20 The neoplastic cells can have variable degrees of atypia. Although scattered mitotic features can be found in reactive fibroblasts, including FEPs, additional features of malignancy, such as infiltrative growth, nuclear hyperchromatism, multinucleation, atypical mitotic figures, and necrosis, can be observed in fibrosarcomas. 12 Nevertheless, since it may be difficult, especially in incisional biopsies, to morphologically distinguish trauma-associated fibrosis from low-grade fibrosarcoma, it is useful to recognize which sites in the oral cavity are predisposed to trauma and which sites and canine breeds are predisposed to fibrosarcoma. 21
Squamous papilloma and viral papilloma are considered gross differential diagnoses of FEPs, since they are sessile to pedunculated 0.5–1 cm masses; however, histologically, they are epithelial tumors characterized by papillary projections, sustained by thin fibrous septa, lined by hyperplastic and hyperkeratotic epithelium that, in case of viral papilloma, may show cytopathic effects, such as giant keratohyalin granules, koilocytosis, or inclusion bodies. 11 None of these features have been described in FEPs.
The anatomical site of onset, clinical presentation and biological behavior, together with distinctive histomorphology, can generally rule-out other differential diagnoses and direct suspicion toward FEP. Although the term “chewing granuloma” is frequently used clinically to refer to these lesions, we suggest, in a histological diagnostic setting, the term “fibroepithelial polyp” as the morphological diagnosis.
Conclusions
Canine oral FEPs are benign reactive proliferative lesions characterized by a fibrovascular stalk covered by hyperplastic, often ulcerated, epithelium, and variably associated with inflammation. Chronic irritation may play a role in the pathogenesis of these lesions; surgical excision and resolution of the eliciting trauma can be curative. FEPs should be included in the differential diagnosis for oral proliferative lesions in the dog.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221144708 – Supplemental material for Oral fibroepithelial polyps (“chewing granulomas”) in 21 dogs: Histomorphology, immunohistochemical characterization, and clinical outcome
Supplemental material, sj-pdf-1-vet-10.1177_03009858221144708 for Oral fibroepithelial polyps (“chewing granulomas”) in 21 dogs: Histomorphology, immunohistochemical characterization, and clinical outcome by Laura Nordio, Cristiano Stefanello and Stefania Gasparini in Veterinary Pathology
Footnotes
Acknowledgements
Authors thank all the colleagues in the Pathology division, and especially Dr Anderson Gaglione, Dr Marta Orazi, and Rebecca Pietrobelli for the technical support. Authors are grateful to all the referring veterinarians for submitting their cases and collaborating with their precious help.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
