Abstract
Sebaceous carcinomas of the human ocular adnexa commonly exhibit pagetoid spread, mutations in tumor-suppressor genes, and protooncogene copy number gain. Sebaceous carcinomas are rarely reported in other species, and while the Meibomian gland (MG) represents the most common ocular adnexal structure of the canine eyelid to develop neoplasia, most are clinically and histologically benign. The objective of this study was to compare molecular features of canine MG carcinomas and adenomas. Two retrospectively identified MG carcinomas were subject to immunohistochemistry and qPCR. When compared with normal glands, MYC was upregulated in benign and malignant MG neoplasms. Aberrant p53 expression was restricted to the nuclei of intraepithelial neoplastic cells in MG carcinomas. Adipophilin expression was diminished in MG neoplasms compared with the normal MG. Our findings, if confirmed in a larger cohort of cases, could suggest that MG oncogenesis in a dog may exhibit similar molecular features as their human counterparts.
The canine Meibomian gland (MG) represents the most common adnexal structure of the eyelid to develop cancer, and the majority of MG neoplasms are diagnosed as benign adenomas or epitheliomas.6,11 Meibomian gland adenomas are nodular masses comprised of well-differentiated sebaceous tissue, often with exophytic and papillary features, and are frequently accompanied by lipogranulomas.6,11 Meibomian gland epitheliomas may represent a subset of adenoma that is primarily composed of undifferentiated basal cells and are, thus, more likely to occur deeper in the lid margin than those comprised of more fully differentiated MG acini.6,11 No defined prognostic significance exists between adenomas and epitheliomas, and many agree that adenomas are encountered more commonly in the canine patient. 13 Malignant MG tumors are rare in the veterinary literature. 20 The objective of this study was to compare the molecular features of canine MG carcinomas to adenomas.
We performed a retrospective review of the Comparative Ocular Pathology Laboratory of Wisconsin (COPLOW) archives for cases of canine MG carcinoma from 1988 to 2021. Cases were designated as carcinoma if features of invasiveness (junctional activity and/or pagetoid spread), cellular pleomorphism, or nuclear atypia were documented. Cases were designated as adenoma if corresponding histopathology reported the majority of cells exhibited sebaceous differentiation and the criteria for malignancy were absent. Cases predominantly demonstrating undifferentiated basaloid reserve cells (epitheliomas) were excluded.
Immunohistochemical labeling for adipophilin (Fizgerald Industries; 1:500) and p53 (Leica Biosystems; RTU) was performed using a LeicaBond RXm autostainer platform (Leica). Antigen retrieval was performed using sodium citrate–based ER-1-solution (Leica) at 95°C for 10 min or TE-based ER-2-solution (Leica) at 95°C for 10 min for adipophilin and p53, respectively. Antibody detection was performed using the Bond Refined Detection System (Leica), and positive labeling was visualized using DAB or red chromogen substrate for adipophilin and p53, respectively. Mouse IgG1k Clone P3.6.28 (14-4714-85, 2 mg/mL; Thermo Scientific, Waltham, MA) was included as a negative isotype control. Immunolabeling for MYC (Epitomics, Clone EP121; 1:600) was performed using 5 μm sections and standard protocols as previously described. 10 Positive controls included adrenal cortex, high-grade mammary carcinoma, and colonic mucosa for adipophilin, p53, and MYC, respectively.
RNA purification from grossly-dissected formalin-fixed, paraffin-embedded (FFPE) tissue sections of canine MG adenoma (n = 5; Cases 5, 8, 9, 13, and 14), canine MG carcinoma (n = 2; Cases 16 and 17), and normal canine MG identified from necropsy specimens (n = 3) was performed using the Qiagen RNeasy FFPE kit (Qiagen, Hilden, Germany) per the manufacturer’s instructions. RNA quality and integrity were evaluated in triplicate using the QubitTM 4 Fluorometer (Thermo) and the QubitTM RNA IQ Assay Kit per the manufacturer’s instructions. Reverse transcription and quantitative polymerase chain reaction (qPCR) was carried out using the iTaq Universal Probes One-Step Kit (Bio-Rad, Hercules, CA). MYC expression was quantified using 2_ΔcT normalized to GAPDH using predesigned probes; Cf02697067_s1 (MYC) and Cf04419463_gH (GAPDH) (Thermo). Relative expression was evaluated using an ANOVA with a Dunnett’s post hoc test for multiple comparisons using Prism 8 GraphPad (v. 9.2.0; San Diego, CA) (α = 0.05).
Only 2 (0.05%) among the 3697 cases of canine MG tumors identified in the COPLOW database were diagnosed as carcinomas. The 15 most recently diagnosed MG adenomas from the COPLOW archives, which measured at least 5 mm in one dimension at the time of grossing, were also identified. Additional details are provided in Supplemental Table 1 and Fig. 1.
MG tumors in this series varied in gross appearance, ranging from pale pink to dark brown or gray, firm multilobulated or pedunculated masses arising from the eyelid margins, occasionally with foci of ulceration as shown in Fig. 1. Histologic evaluation of the MG adenomas in this cohort generally revealed nonencapsulated, well-demarcated, highly cellular neoplasms expanding the eyelid margin dermis, conjunctival substantia propria, or both. The neoplasms exhibited two morphologic populations as shown in Fig. 1d.
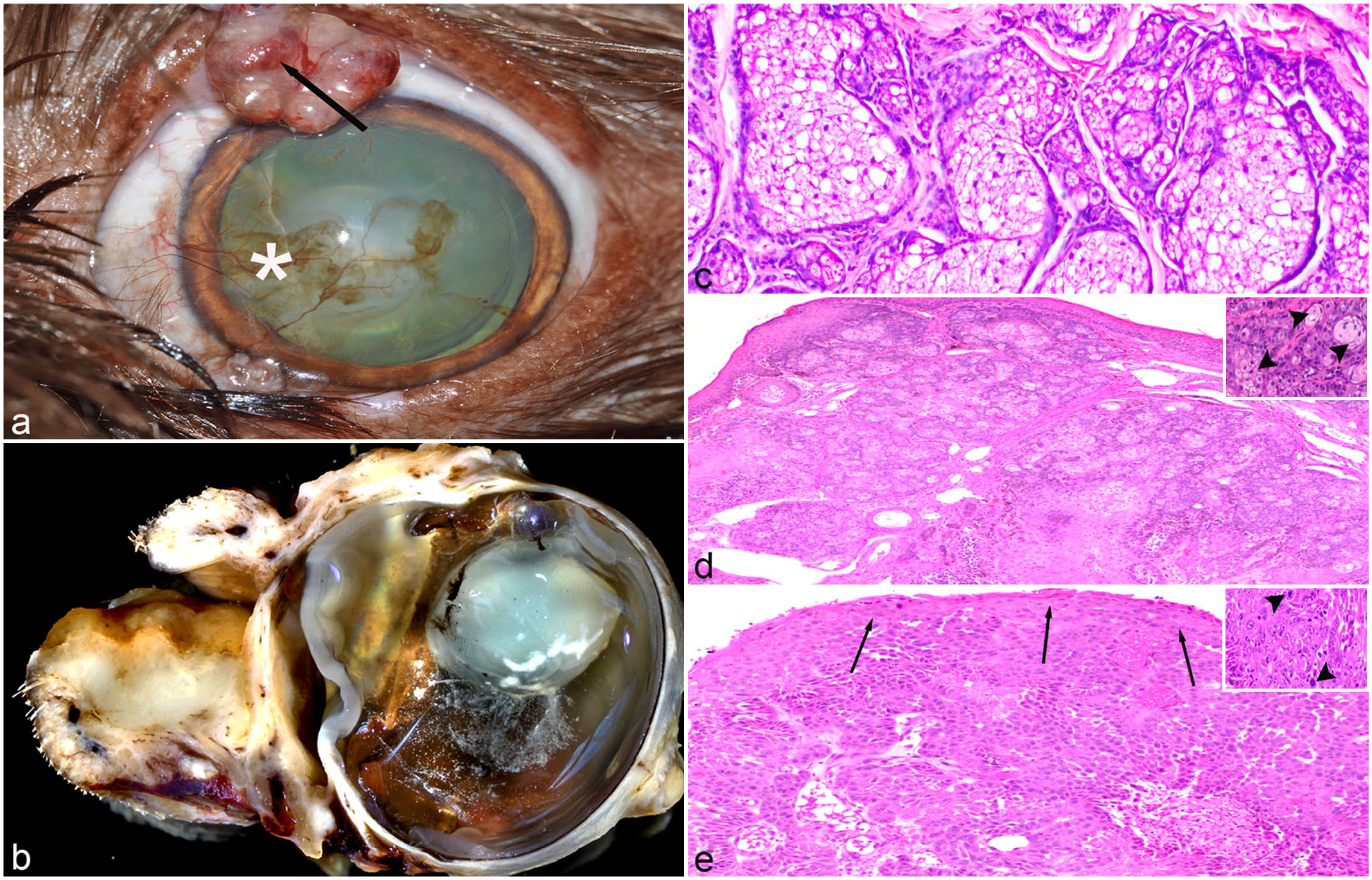
Meibomian gland (MG) tumor, dog. (a) Clinical photograph demonstrating an MG adenoma of the superior palpebral margin with ulceration (arrow), corneal neovascularization (*), and epiphora from Case 2. (b) Gross appearance of sagittally sectioned globe and adnexa with an MG carcinoma effacing and expanding the inferior eyelid from Case 17. (c) Acini of nonneoplastic MG from Case 5. (d) A well-demarcated adenoma expanding the superior palpebral margin from Case 9 with the majority of neoplastic cells exhibiting a large polygonal morphology with abundant foamy cytoplasm (sebaceous differentiation) and a second population of cuboidal cells with scant eosinophilic cytoplasm (basaloid reserve cells) dividing the sebaceous cells into lobules. Inset demonstrating sebaceous cells (arrowheads). (e) An infiltrative MG carcinoma expanding the inferior eyelid and replacing the surface epithelium (arrows) from Case 17. Inset demonstrating clusters of large neoplastic epithelial cells with sebaceous differentiation, marked anisocytosis, and robust mitotic activity (arrowheads).
Histopathological evaluation of the two MG carcinomas demonstrated hypercellular segments of conjunctiva, some with variable amounts of surface epithelium. The conjunctival substantia propria was effaced and replaced by sheets of neoplastic epithelium which multifocally blended with and replaced the surface epithelium both adjacent (junctional activity) and distant to the tumor mass (pagetoid spread). There were small foci of cells with sebaceous differentiation embedded in the sheets of more basaloid neoplastic cells as shown in Fig. 1e. The neoplastic cells had moderate amounts of pale amphophilic cytoplasm and indistinct cell borders. Nuclei were round with vesiculated chromatin and a single dense nucleolus. Anisocytosis and anisokaryosis were moderate with both neoplastic basaloid and sebaceous cells ranging in size from approximately 6 to 20 μm, and there were up to 55 mitoses in 10 high-power fields (2.37 mm2) in the sample submitted from Case 17 but only 1 mitotic figure in 10 high-power fields in the sample submitted from Case 16. There was variable, multifocal to coalescing lymphoplasmacytic and neutrophilic inflammation throughout the mass lesions.
Membranous adipophilin expression was moderate to strong in the meibocytes of the normal MG; however, expression was weaker in adenomas, and absent to weak in the carcinomas as shown in Fig. 2a–c. Expression of p53 was not observed within the cytoplasm of any normal tissues or of adenomatous MGs; however, both of the MG carcinomas in this series demonstrated moderate to strong nuclear p53 expression within neoplastic MG basal cells and scattered nuclear p53 expression within foci of pagetoid spread as shown in Fig. 2f. MYC expression of the normal MG was weak and restricted to the basal cell nuclei. In MG adenomas, expression of neoplastic nuclei was moderate to strong relative to normal controls, and MYC expression was stronger in MG carcinomas as shown in Fig. 2g–i.
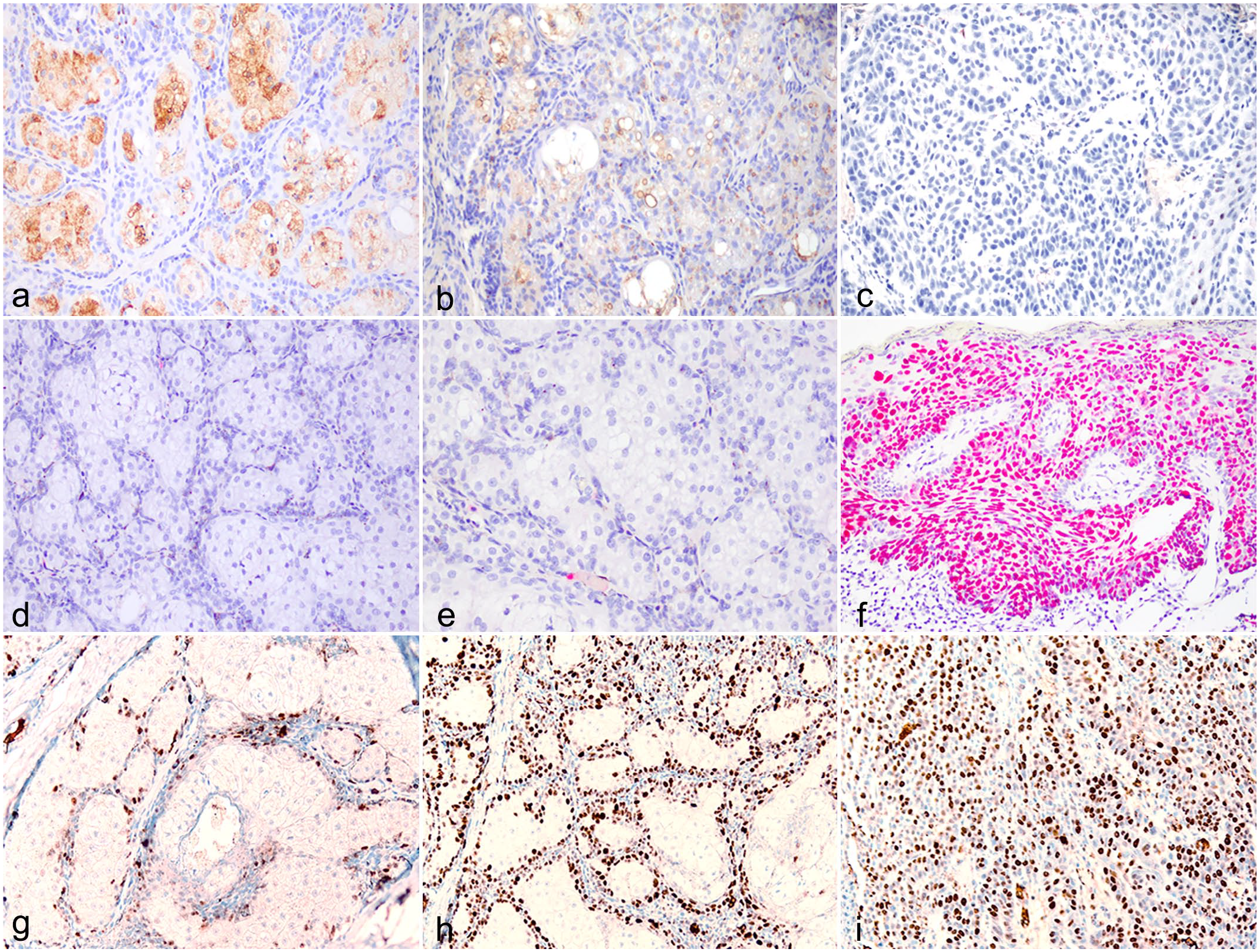
Molecular features of the Meibomian gland (MG), canine. Adipophilin expression in (a) normal MG, (b) MG adenoma, (c) MG carcinoma. Expression of aberrant p53 in (d) normal MG, (e) MG adenoma, and (f) MG carcinoma. MYC expression in (g) normal MG, (h) MG adenoma, and (i) MG carcinoma.
Relative MYC expression was significantly higher in MG carcinomas (9.6 ± 1.2; p ≤ 0.0001) compared to normal tissue (1.0 ± 0.3) as shown in Fig. 3. There was no significant difference between MG adenomas (2.1 ± 0.9; p = 0.2123) and normal glands.
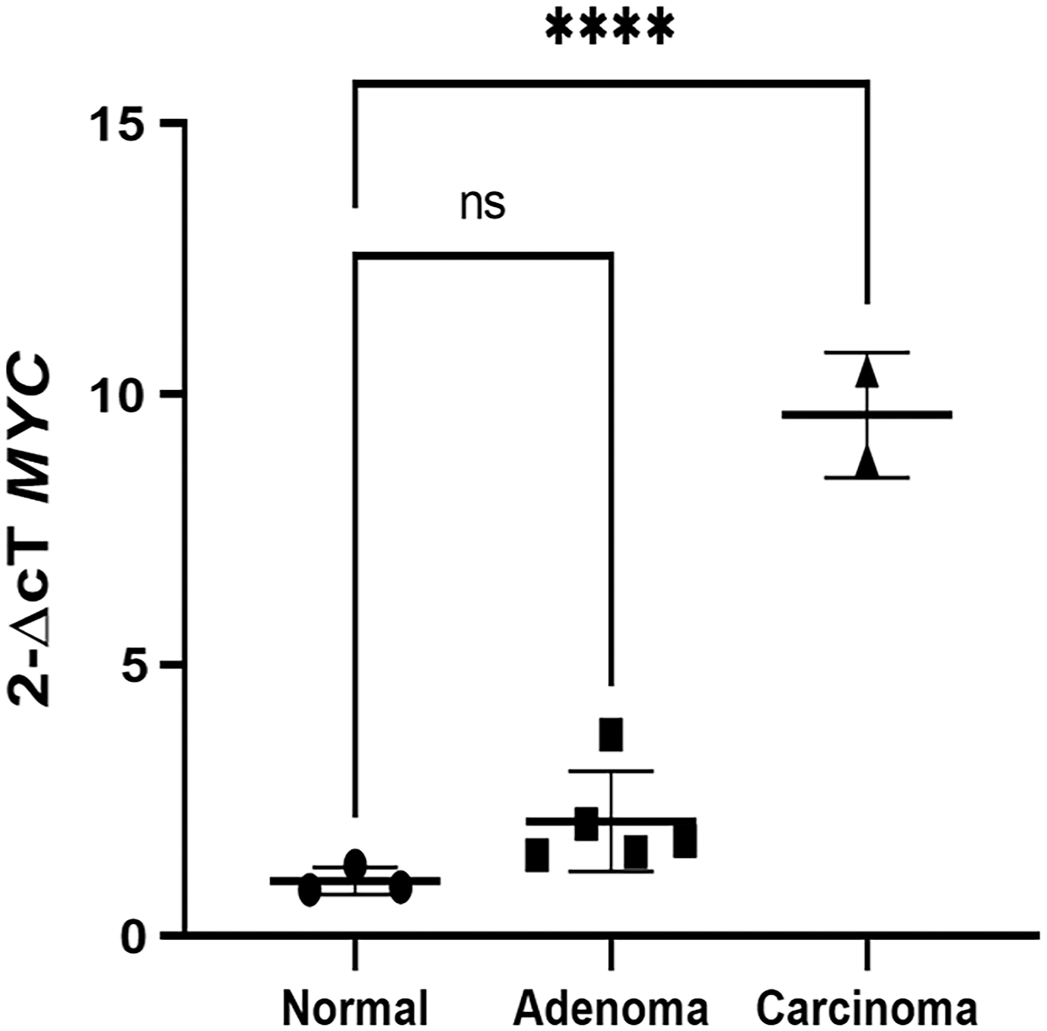
MYC Expression of the Meibomian gland (MG), canine. Relative MYC expression was significantly greater in carcinomas than in the normal gland (****p ≤ .0001).
Sebaceous neoplasms of the eyelid are well-documented in human medicine, with carcinomas exhibiting more aggressive behavior and higher rates of metastasis and recurrence than those of extraocular sites. 14 The molecular drivers of these tumors are incompletely elucidated; however, somatic mutations and human papillomavirus have been implicated in oncogenesis of these neoplasms.14,15 Sebaceous adenomas are more common than either sebaceomas or sebaceous epitheliomas and can be associated with syndromic microsatellite instability or mutations in genes encoding DNA mismatch repair proteins in affected Muir Torre patients.14–16 Immunohistochemistry is often used to differentiate sebaceous carcinomas from benign lesions of the eyelid and other epithelial malignancies including basal and squamous cell carcinomas.1,16
Here, we describe the features of the rare canine MG carcinoma and compare these features to the adenoma, which occurs more commonly in both man and dog. The classic histomorphologic features of human sebaceous carcinomas, particularly invasive growth patterns with pagetoid spread, but also sebaceous differentiation, cellular pleomorphism, and nuclear atypia, foci of necrosis, and high mitotic activity, were observed in cases of canine MG carcinoma, suggesting similar biologic behavior as their human counterparts.12,17
The most striking finding in this study was consistently robust MYC expression in the neoplastic canine MG. The protooncogenic role of MYC in physiologic epithelial differentiation and pathologic development of carcinomas has been well-documented, and copy number gain of the c-MYC locus has been recently described in sebaceous carcinomas of the ocular adnexa.5,15 While, to the authors’ knowledge, there are no reports of MYC expression in canine sebaceous or MGs, upregulation of MYC has been reported in canine prostatic and mammary neoplasms.7,8
Expression of p53 in the canine MG was also explored, as somatic TP53 mutations are among the most frequently reported in human sebaceous carcinomas.3,18 Here, no intracytoplasmic expression of aberrant p53 was observed in the normal MG or within the adenoma samples; however, strong, scattered expression within the nuclei of neoplastic intraepithelial cells was observed in both cases of MG carcinoma. Expression of variant p53 has been documented in numerous canine neoplasms with correlation to higher proliferation indices, and strong labeling for aberrant p53 in MG carcinomas suggests inactivation of tumor-suppressor complexes and the resulting protein overexpression may be a discerning feature of malignancy in this tissue.2,19 The paucity of cytoplasmic expression in the neoplastic MG suggests that loss of function mutations do not involve domains affecting nuclear localization or nuclear exclusion sequences; however, these postulations would require DNA sequencing to confirm.
Adipophilin immunolabeling is commonly used to confirm sebaceous differentiation in anaplastic human sebaceous carcinoma specimens, and its use in canine neoplasms to highlight intracytoplasmic lipids of sebaceous cells has also been reported.4,9 Here, we demonstrated strong cytoplasmic vacuolar membrane expression of adipophilin in normal MGs with attenuated expression in MG adenomas and weak to absent expression in carcinomas. These results correlate well to the histologic findings in these cases in which malignant MGs demonstrated increasing anaplasia, often exhibiting a more basaloid phenotype, indicating similar utility for this marker in epithelial tumors of the canine eyelid.
This study is limited by the small number of MG carcinomas (n = 2); however, queries of the COPLOW archives, which contains tens of thousands of veterinary ocular pathology cases, and additional veterinary anatomic pathologists with substantial private practice and academic diagnostic ocular pathology caseloads yielded only these two MG tumors with features of malignancy. Exploring multi-institutional international collaboration may be a potential avenue to identify additional specimens. In addition, while care was taken to demarcate and exclude normal tissue prior to RNA isolation, sensitive techniques including laser microdissection were not pursued, and inclusion of nontumoral tissue in transcriptional evaluation of MYC is possible. However, induced expression in adjacent nonneoplastic tissue would be an interesting subject for future study. Comparing expression of the markers used here to expression in MG epitheliomas may provide additional insight into tumorigenesis, and interrogating these tumors for mismatch repair mutations or microsatellite instability may reveal additional contributors to carcinogenesis, relevant to both human sebaceous adenomas and carcinomas.
In summary, we have described the pathologic features of canine MG carcinomas, a rare malignancy with similar invasive and proliferative features of human ocular adnexal sebaceous carcinomas.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221143400 – Supplemental material for Upregulated MYC expression and p53 mutations may contribute to the oncogenesis of canine Meibomian gland carcinomas
Supplemental material, sj-pdf-1-vet-10.1177_03009858221143400 for Upregulated MYC expression and p53 mutations may contribute to the oncogenesis of canine Meibomian gland carcinomas by Cornelia Peterson, J. L. Hicks, A. M. De Marzo, A. A. Campbell, C. G. Eberhart, R. R. Dubielzig and L. B. Teixeira in Veterinary Pathology
Footnotes
Acknowledgements
The authors thank Dr. Heidi Clark for assistance in locating archived paraffin blocks, Dr. Elizabeth Adkins for her clinical photograph of the Meibomian gland adenoma used in ![]() , Dr. James Foster for his expertise in optimizing the iTaq One-Step protocol, and Ms. Riley Richardson and Ms. Shana Lee for their technical support in initial IHC optimization and processing the histopathology slides, respectively.
, Dr. James Foster for his expertise in optimizing the iTaq One-Step protocol, and Ms. Riley Richardson and Ms. Shana Lee for their technical support in initial IHC optimization and processing the histopathology slides, respectively.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: While the authors received no financial support for the authorship and/or publication of this report, CP was supported by NIH T32 OD011089 (PI: Mankowski) and AAC was supported by the King Khaled Eye Specialist Hospital Grant 134839.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
