Abstract
Ocular lesions are uncommonly reported and described in invertebrate species. In this study, cases from 2 diagnostic laboratories, in which lesions were noted in 33 diagnostic specimens from various species of cephalopods, including octopuses, squid, nautiluses, and cuttlefish, were reviewed. Clinical information and gross lesions were described in a minority of cases. The most common lesion was inflammation of varying severity and was most commonly within the anterior uvea (iris and ciliary papilla), followed by the posterior chamber and lens. More than half of the cases with inflammation had concurrent hyperplastic lesions of the iris and ciliary papilla, including posterior iris epithelial hyperplasia, cystic adenomatous hyperplasia, and/or posterior epithelial cysts. The most common clinical observation was cloudy eyes, which correlated histologically to anterior uveitis in all cases where it was documented. Dermatitis and cutaneous ulceration were the most frequent comorbidities in cases where clinical information was available.
Cephalopods are diverse predatory marine invertebrates that comprise a class within phylum Mollusca and consist of 2 distantly related extant subclasses: Nautiloidea and Coleoidea. The subclass Nautiloidea has an external shell, including nautiluses and allonautiluses, whereas Coleoidean molluscs have an internalized or absent shell and include the octopus, cuttlefish, and squid. 2 The word cephalopod is derived from ancient Greek meaning “head-foot,” in reference to their unique anatomy with arms branching directly from their head. 2 Another unique feature that separates cephalopods from vertebrates is their complex ocular anatomy. As in vertebrates, coleoid cephalopod ocular structures include the eyelids, cornea, iris, pupil, lens, and retina (Fig. 1). Their prominent ciliary muscles, which are incorporated into the ciliary papillae, shift the lens forward and backward to focus images on a heavily pigmented retina where the light directly excites photoreceptor cells without traversing other layers.2,7 As a result, the retinal layers of cephalopods are in reverse order compared with those of vertebrates (ie, everted retina). Underlying the photoreceptors is a retinal nerve cell layer with fibers that synapse with nerves of the optic ganglion (ie, “optic nerve rootlets”), in comparison with the confluence of nerve fibers into the optic nerve seen in vertebrates. 1 The eyes of some cephalopods, such as the common octopus, view objects and function independently of each other, which is demonstrated by a lack of consensual pupillary light response. 3 Nautiloid cephalopods have a more primitive pinhole pupil with a retina, but lack a cornea, ciliary papilla, lens, and ocular chambers. 9 Many of these differences are explained by the evolutionary variation in ocular development between vertebrates and cephalopods, in that the eyes of cephalopods begin as invaginations of skin, whereas those of vertebrates develop from projections of the brain. 5
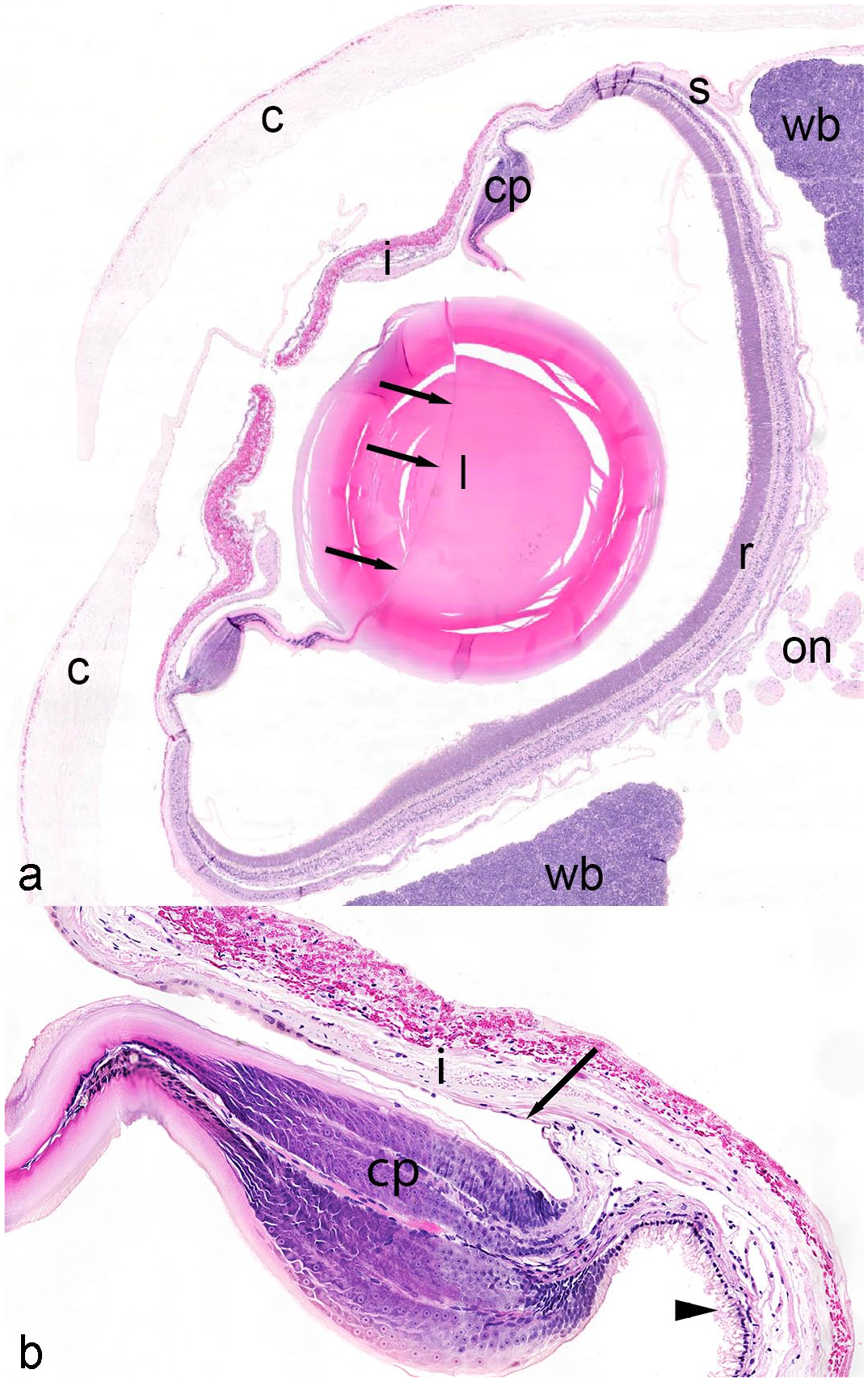
Normal coleoid eye, giant Pacific octopus. (a) Normal ocular structures include the cornea (c; or pseudocornea), iris (i), pupil, ciliary papilla (cp), lens (l) with septum (arrows), optic nerve rootlets (on), sclera (s), retina (r), and white body (wb). (b) The ciliary papillae (cp) is overlain by simple pigmented epithelium that is continuous anteriorly with the posterior iris epithelium (arrow) and continuous posteriorly with the peripheral retina (arrowhead). Hematoxylin and eosin.
Ocular lesions in cephalopods are uncommonly reported with only 3 cases of phakitis and retinitis documented in the literature. 8 This descriptive retrospective study illustrates the prevalence of ocular lesions in cephalopods from diagnostic pathology specimens and characterizes the types and anatomic distributions of histological lesions in these invertebrates.
Materials/Methods
The archives of Northwest ZooPath (1995-2021) and the Connecticut Veterinary Medical Diagnostic Laboratory (2006-2021) were searched for cases of cephalopods with ocular lesions. An additional case was identified from Zoetis Laboratories. Hematoxylin and eosin (HE)–stained paraffin-embedded sections of the eyes were reviewed by both the primary author (KF) and a board certified veterinary pathologist (EEBL for Northwest ZooPath and Zoetis cases and SF for Connecticut Veterinary Medical Diagnostic Laboratory cases). Six cases were stained with Brown and Brenn, Brown and Hopps, and Twort Gram stains and/or Grocott methenamine silver and acid-fast histochemical stains. The anatomic distribution of lesions (ie, cornea, anterior chamber, iris, posterior chamber, ciliary papilla, lens, vitreous, retina, and/or sclera) and associated lesions was recorded and tabulated. Additional case information, including clinical history and gross findings, were collated when available. Each eye was counted as a separate case number (even if from the same animal) as lesions were not always bilateral and not every animal had both eyes submitted for histology (Supplemental Table 1).
Results
Cases
A total of 313 cephalopod biopsy and necropsy submissions were identified in the Northwest ZooPath archives, 12 of which (3.8%) had ocular lesions and in 2 of these submissions, the eyes were bilaterally affected. In a total of 112 cephalopod specimens from the Connecticut Veterinary Medicine Diagnostic Laboratory, 10 submissions (8.9%) had ocular lesions, 7 of which were bilateral. An additional submission with bilateral lesions was identified from Zoetis Laboratories, for a total of 23 affected animals with a total of 33 affected eyes. The most commonly represented subclass were the Coleoidean mollusks, including 8 cuttlefish (Sepia sp.), 7 giant Pacific octopuses (Enteroctopus dofleini), 2 bigfin reef squid (Sepioteuthis lessoniana), 2 common octopuses (Octopus vulgaris), and 1 red octopus (Octopus rubescens). Less commonly, Nautiloidean mollusks were affected with 3 chambered nautiluses (Nautilus pompilius) represented in the study. Lesion location and represented species are summarized in Supplemental Table 1.
Clinical history and necropsy information were available for 21 animals, of which 10 (n = 10/21; 48%) reported ocular issues, including “cloudy eyes” (n = 5/10; 50%), issues with vision (n = 3/10; 30%), “eye injury” (n = 1/10; 10%), and unspecified ocular injury (n = 1/10; 10%). Non-ocular, concurrent diseases identified clinically or at necropsy included cutaneous ulceration (n = 8/21; 38%) and senescence (n = 1/21; 5%). All anatomical parts of the eye were available for histological examination in every eye except for the cornea in 27/33 (82%) eyes and the optic nerve rootlets in 1/33 (3%) eyes. The most common reasons for the lack of cornea in section were that the animals were nautiluses (n = 5/27; 19%), which anatomically lack corneas, 9 or that the corneas were not removed with the globe during biopsy and/or necropsy (n = 22/27; 81%) as the corneas are a continuation of the skin in Coleoidean cephalopods.7,8
Histological lesions were most commonly observed in the iris (n = 29; 88%), followed by the ciliary papilla (n = 26; 79%), lens (n = 24; 73%), posterior chamber (n = 23; 70%), sclera (n = 18; 55%), choroid (n = 16; 48%), retina (n = 14; 42%), anterior chamber (n = 8; 24%), and vitreous (n = 6; 18%), with prevalence calculated based on the total number of eyes examined. Because the optic nerve rootlets and cornea were not present in all examined eyes, the prevalence of lesions in those structures was calculated based on the number of cases for which the structures were present, 56% (n = 18/32) and 67% (n = 4/6), respectively. The most common lesion was inflammation (n = 32; 97%), of which one case had concurrent vasculitis and one case had corneal erosion, followed by cataractous change (n = 12; 36%), iris cysts (n = 11; 33%), posterior iris epithelial hyperplasia (n = 10; 30%), cystic adenomatous uveal hyperplasia (n = 6; 18%), and retinal fold (n = 1; 3%).
Inflammation
Inflammation appeared histologically as focal, nodular, or diffuse infiltrates of hemocytes. Inflammation (n = 32) was most common in the anterior uvea (n = 29/32; 91%; Fig. 2a), including the iris (n = 29/32; 91%) and ciliary papilla (n = 26/32; 81%), followed by the lens (n = 23/32; 72%), posterior chamber (ie, chamber between the iris and ciliary papilla; n = 23/32; 72%), sclera (n = 18/32; 56%), optic nerve rootlets (n = 18/32; 56%), choroid (n = 16/32; 50%), retina (n = 13/32; 41%), anterior chamber (ie, chamber anterior to the iris; n = 8/32; 25%), vitreous (n = 6/32; 19%), and cornea (n = 4/32; 13%). Bacteria were only identified in eyes with inflammatory lesions, 14/32 (44%) of which had bacteria visible in HE-stained sections. Bacteria observed were typically extracellular in areas of dense inflammatory infiltrates and included rods (n = 5/14; 36%), cocci (n = 4/14; 28%), coccobacilli (n = 1/14; 8%), or both (n = 4/14; 28%). Gram stains were performed in 5 eyes that had bacteria present on HE-stained sections that revealed gram-negative rods (n = 2/5; 40%), gram-negative cocci (n = 1/5; 20%), gram-positive rods and cocci (n = 1/5; 20%), and gram-positive cocci and gram-negative rods (n = 1/5; 20%). In addition to inflammation of ocular tissues, one giant Pacific octopus had concurrent marked necrotizing vasculitis that most severely affected the choroid and optic nerve rootlets, where all layers of the vascular wall were effaced by cellular and karyorrhectic debris, viable and necrotic hemocytes, fibrin, and hemorrhage. Within affected vessels and throughout the adjacent tissues, there were numerous colonies of 1 to 2 µm basophilic cocci. One giant Pacific octopus had unilateral partial thickness loss of the corneal epithelium (erosion), with the subjacent stroma infiltrated by 2- to 3-µm-long basophilic bacterial rods and few hemocytes.
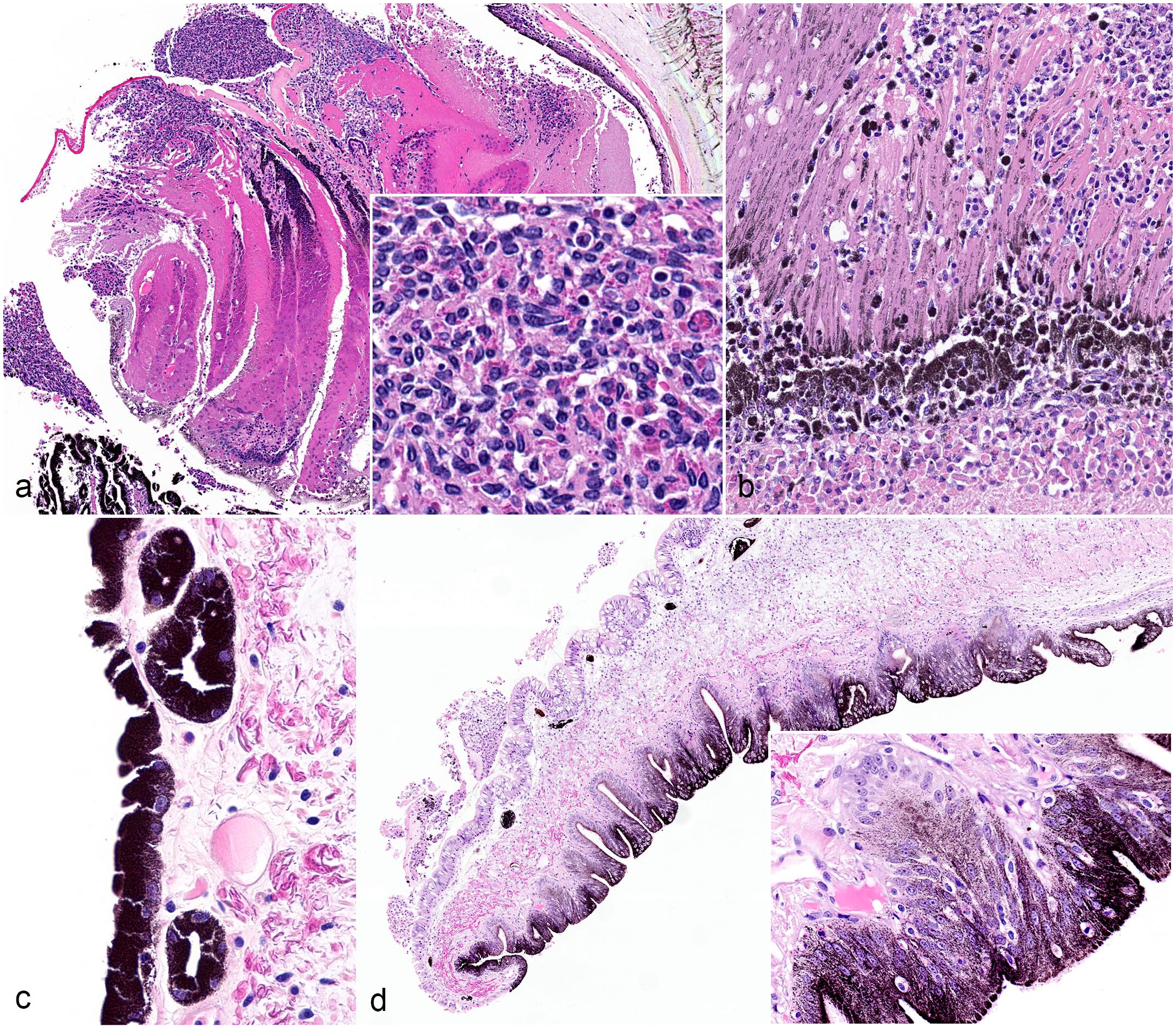
Inflammatory and proliferative lesions in cephalopod eyes. (a) Anterior uveitis, eye, cuttlefish, eye 20. Hemocytic inflammation (inset) expands the ciliary papilla and extends into the iris and posterior chamber. HE. (b) Chorioretinitis, eye, cuttlefish, eye 20. The choroid and retina are diffusely expanded by hemocytic inflammation with accumulation of cellular and karyorrhectic debris (necrosis), degeneration, and loss of photoreceptors. HE. (c) Posterior iris epithelial cysts, eye, cuttlefish, eye 18. Cysts that are confined to the posterior iris epithelium measure between 10 and 50 µm in diameter and are lined by heavily pigmented iris epithelium. HE. (d) Posterior iris epithelial hyperplasia, eye, giant Pacific octopus, eye 11. The posterior surface of the iris has exophytic papillary ridges (inset) of pigmented epithelium and subtending stroma that protrude into the posterior chamber. HE. HE, Hematoxylin and eosin.
Anterior uveitis, defined as inflammation in the iris and/or ciliary body, was associated with proliferative lesions in 17/29 (59%) eyes, including a combination of posterior iridal pigmented cysts and posterior iris epithelial hyperplasia (n = 6/17; 35%), posterior iridal pigmented cysts without other proliferative lesions (n = 4/17; 24%), and cystic adenomatous uveal hyperplasia without other proliferative lesions (n = 3/17; 18%); a combination of cystic adenomatous uveal hyperplasia and posterior iris epithelial hyperplasia (n = 2/17; 12%), and posterior iris epithelium hyperplasia without other proliferative lesions (n = 1/17; 6%); and a combination of all 3 proliferative lesions (n = 1/17; 6%) (see “Proliferative Lesions” section for more information). All cases diagnosed with “cloudy eyes” grossly (5/5; 100%) had anterior uveitis histologically. Phakitis was associated with cataracts in 11/23 (48%) eyes (see “Other Lesions” section for more information). Retinitis was associated with retinal necrosis in 5/14 (36%) eyes, characterized by marked necrosis, degeneration, and loss of photoreceptors with replacement by cellular and karyorrhectic debris (Fig. 2b). Inflammation in other locations was not associated with concurrent lesions in those locations.
Proliferative Lesions
Proliferative lesions were identified in 27/33 eyes (82%) and included iris cysts (n = 11/27; 41%), posterior iris epithelial hyperplasia (n = 10/27; 37%), and cystic adenomatous hyperplasia (n = 6/27; 22%). Iris cysts confined to the posterior iris epithelium, diagnosed in 7 octopuses and 4 cuttlefish, ranged between 10 and 50 µm in diameter; were lined by heavily pigmented iris epithelium; and contained variable amounts of granular pink eosinophilic fluid (Fig. 2c). Posterior iris epithelial hyperplasia, diagnosed in 6 octopuses and 4 cuttlefish, was characterized by exophytic papillary ridges of pigmented epithelium and subjacent stroma that extended into the posterior chamber (Fig. 2d). Cystic adenomatous uveal hyperplasia, diagnosed in 2 octopuses, 1 squid and 3 cuttlefish, was characterized histologically by expansion of the iris and ciliary papilla stroma by multiple cysts up to 3.2 mm in diameter (Fig. 3a, b). These cysts were lined by variably pigmented, cuboidal to columnar epithelial cells up to 6 cell layers thick. Epithelial cells had indistinct cell borders, abundant eosinophilic cytoplasm, and a round to ovoid basally located nucleus with finely stippled chromatin and 1 to 3 distinct nucleoli. There was occasional blebbing of the apical surface or attenuation of the epithelium and cyst lumina was clear or had debris, hemorrhage, amphophilic fluid, and/or hemocytic inflammation. The connective tissues adjacent to iris cysts and regions of cystic adenomatous hyperplasia always contained hemocytic inflammation of varying severity.
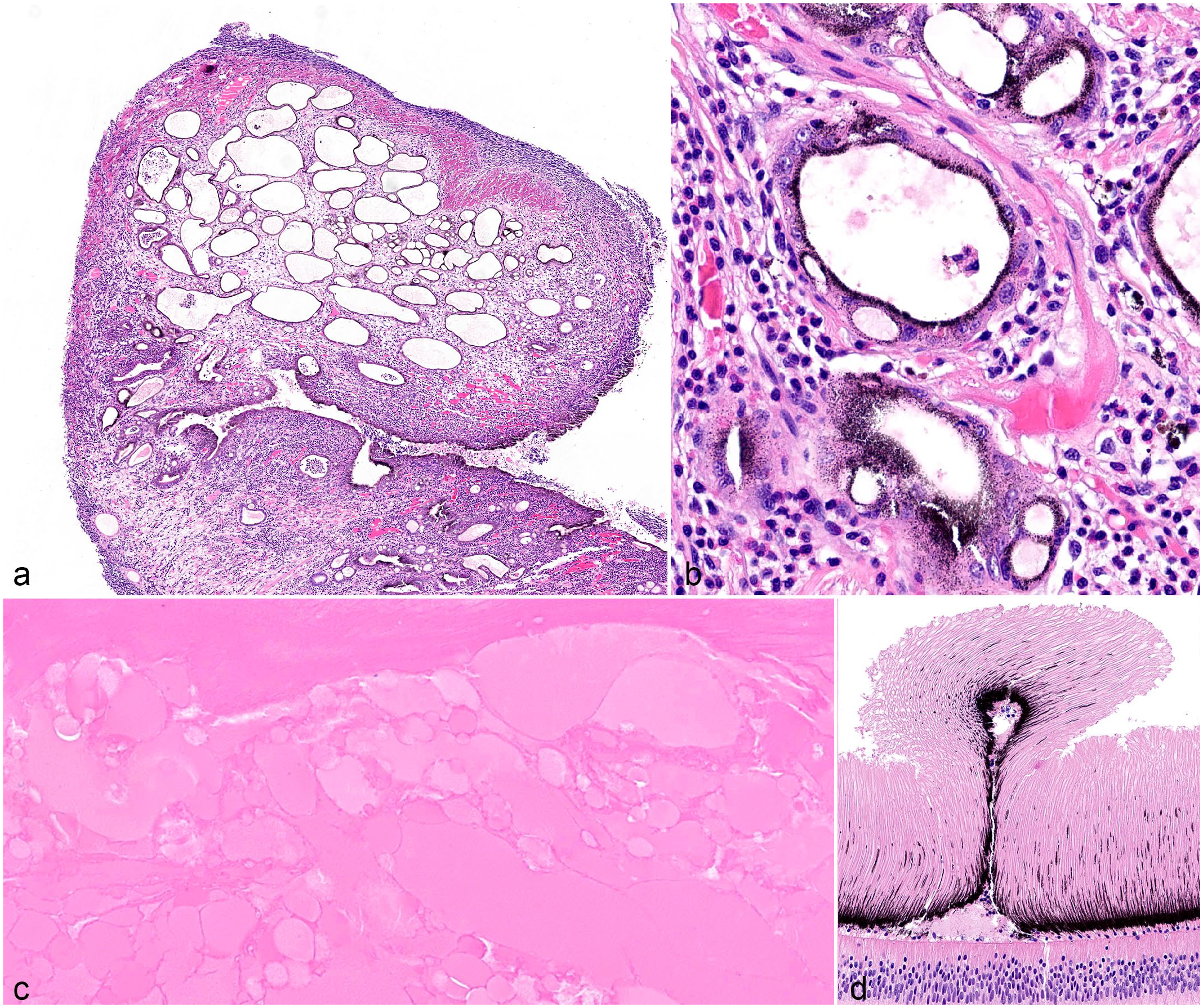
Proliferative and other lesions in cephalopod eyes. (a-b) Cystic adenomatous hyperplasia, eye, cuttlefish, eye 24. (a) There is expansion of the iris and stroma of the ciliary papilla by multiple, variably sized, ovoid cysts up to 500 µm in diameter. The stroma that surrounds cysts has increased clear space (edema) and hemocytic inflammation. (b) Cysts are lined by flattened to cuboidal pigmented epithelium and contain flocculent eosinophilic material, hemocytes, and/or necrotic cellular debris. HE. (c) Cataract, eye, cuttlefish, eye 27. There is focally extensive liquefaction of lens fibers with replacement by discrete, round, eosinophilic globules (Morgagnian globules). HE. (d) Retinal fold, eye, bigfin reef squid, eye 16. A focal retinal fold is composed of the outer segments of the photoreceptors and ommin pigment layers. HE. HE, Hematoxylin and eosin.
Other Lesions
Other ocular lesions included cataracts (n = 12/33; 36%) and a retinal fold (n = 1/33; 3%). Cataracts were characterized by lens fiber liquefaction and Morgagnian globules (Fig. 3c). Almost all eyes with cataracts were associated with phakitis (n = 11/12; 92%). Within the retina of one bigfin reef squid, there was a focal retinal fold composed of layers of the outer segments of the photoreceptors and the ommin pigment layer (Fig. 3d).
Discussion
In this retrospective study, we describe and categorize the ocular lesions in 33 eyes from 23 cephalopods. Ocular lesions were generally uncommon, with lesions occurring in 3.8% of all cephalopod specimens submitted to Northwest ZooPath and 8.9% of those submitted to the Connecticut Veterinary Medicine Diagnostic Laboratory during the study period. The most commonly reported clinical observation was cloudy eyes, which is a common gross observation in vertebrates. 12 The histopathologic correlate for cloudy eyes in all cephalopod cases in this study was anterior uveitis, which was noted in 91% of eyes in this study.
The most common histological ocular lesion of cephalopods was inflammation. The preponderance of inflammatory lesions in this study is similar to the high prevalence of ocular inflammatory lesions reported in vertebrates.6,12 Factors that likely contributed to inflammatory ocular lesions in cephalopods include intraocular bacterial infection, environmental causes, unique ocular anatomy, and comorbidities. Bacteria were observed in 13/32 cases (41%) with inflammation, suggesting bacterial infection is an important contributing factor to ocular inflammatory conditions in cephalopods. It is unclear whether the observed bacterial populations were primary pathogens or opportunistic invaders. Senescence can result in immune dysfunction and possibly predispose these animals to bacterial infection. 11 Senescence was indicated in the clinical history in only one case, which had a bacterial infection. Additional clinical information, specifically bacterial culture and presence or absence of senescence, as well as bacterial genetic sequencing and determination of virulence factors may aid in determining the relevance of ocular bacterial infections. Bacterial culture can be obtained from a corneal biopsy (in those species that have them) or centesis of the anterior chamber, although access may be challenging. Culture of aquatic organisms can be challenging given the difficulty of replicating bacterial growth in vitro (chemical and physical conditions) to optimize growth and a laboratory specializing in aquatic bacterial cultures is recommended.
Adverse environmental factors such as temperature, toxins, lighting conditions, pH, and other water quality parameters such as mineral content and osmolality are common contributing causes to debilitation in invertebrates. 10 The ocular anatomy of cephalopods, particularly their unique corneal anatomy, causes intraocular structures to be in direct communication with the surrounding aquatic environment.2,7 While Nautiloids lack a cornea altogether, the cornea of Coleoid cephalopods is sometimes called a “pseudocornea” and has a dorsal opening that allows the anterior chamber to communicate directly with the surrounding environment.7,8 One of the primary functions of the cornea in vertebrates is the exclusion of pathogenic organisms and the preservation of a controlled, immune-privileged intraocular environment. 4 Without the protective corneal barrier, it is possible that infectious organisms and/or suboptimal environmental conditions may more readily lead to intraocular compromise and inflammation in cephalopods.
The anterior uvea (iris and ciliary papilla) was the most commonly affected portion of the cephalopod eye. A previous report of ocular lesions in 3 octopuses reported that inflammation in all 3 animals was most pronounced in the ciliary papilla, lens, and retina. 8 The report did not describe changes in the iris, which is the predominant component of the anterior uvea in cephalopods, and as such the distribution of inflammation in those 3 cases is difficult to compare with the present study. A likely explanation for the anterior distribution in the present study is the increased vulnerability of anterior ocular structures to environmental insults, including trauma, infectious agents, and derangement in water quality, as these anterior structures are in direct communication with the external environment. Examination of the cornea was limited in this study as it was available for evaluation in the minority of cases. The cornea of Coleoid cephalopods is not connected to the sclera and it cannot be removed with the underlying ocular tissues.7,8 As such, it was likely not sampled with the globe during necropsy.
A particularly interesting histological finding was epithelial hyperplasia of the iris and ciliary papilla. Hyperplasia ranged from papillary to adenomatous and occurred exclusively in regions of inflammation. In addition, these hyperplastic lesions were present in over half of the cases of anterior uveitis and occurred across a range of cephalopod species. It is suspected that hyperplasia of the anterior uvea is a common reactive lesion due to chronic anterior uveitis in cephalopods. Neoplastic proliferation was not seen and may be an unlikely sequel.
The most common comorbidity was dermatitis and/or cutaneous ulceration. It is unknown whether this finding is due to a relatively high degree of cutaneous lesions in captive cephalopods or due to an association between cutaneous and ocular lesions. 1 If the latter is true, these cutaneous lesions may have predisposed cephalopods to ocular lesions through bacteremia or may have been due to some degree of visual compromise resulting in trauma to the skin.
Based on the previous report and current study, inflammatory conditions of the eye, particularly in the anterior uvea and lens, are the most common intraocular lesions in cephalopods. With the limitations to this study, including paucity of clinical history and ancillary diagnostics, as well as the limitations to evaluation and treatment of intraocular lesions in cephalopods, future research will be needed to understand predisposing conditions and infectious agents that may influence the development of these lesions.
Supplemental Material
sj-docx-1-vet-10.1177_03009858221133079 – Supplemental material for Ocular lesions of captive cephalopods
Supplemental material, sj-docx-1-vet-10.1177_03009858221133079 for Ocular lesions of captive cephalopods by Kelsey Fiddes, Mike Murray, Salvatore Frasca, Michael M. Garner and Elise E. B. LaDouceur in Veterinary Pathology
Footnotes
Acknowledgements
We thank individuals and institutions who contributed case material, including Aquarium at Moody Gardens, Cornell University, Long Beach Aquarium, Monterey Bay Aquarium, National Aquarium in Baltimore, New England Aquarium, Steinhart Aquarium, Waikiki Aquarium, Tennessee Aquarium, New York Aquarium, Houston Zoo, Adventure Aquarium, and Minnesota Zoo. We also thank Rebecca Dunham and Alyssa Osier of Zoetis Reference Laboratories, Cathy Minogue of Northwest ZooPath, and Vickie Weidig of the Connecticut Veterinary Medical Diagnostic Laboratory for collation of case materials.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The views expressed in this abstract are those of the authors and do not reflect the official policy of the Department of Army/Navy/Air Force, Department of Defense, or U.S. Government. The identification of specific products or scientific instrumentation does not constitute endorsement or implied endorsement on the part of the author, DoD, or any component agency.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
