Abstract
Parasitism of cephalopods is common, including infection with Aggregata spp., Ichthyobodo spp., dicyemids, cestodes of the orders Tetraphyllidea and Trypanorhynchidea, and various crustaceans. Cestodiasis in octopuses is reported, although a full histologic description of lesions has not been previously described. Cestodiasis was identified in 10 octopuses of 4 different species, which included 4 common octopuses (Octopus vulgaris), 3 Caribbean reef octopuses (Octopus briareus), 2 two-spot octopuses (Octopus bimaculoides), and 1 giant Pacific octopus (Enteroctopus dofleini). Larval cestodes were present in the cecum (n = 5), intestines (n = 4), digestive gland (n = 3), chitinous alimentary tract (n = 2), renal appendage (n = 1), and salivary duct (n = 1). In 5 cases, larval cestodes invaded tissue and were associated with hemocytic inflammation and tracts of necrotic tissue in the intestines (n = 3), digestive gland (n = 3), and/or renal appendage (n = 1). When present in the chitinous alimentary tract (esophagus, stomach) or cecum, larval cestodes were in the central lumen and not associated with lesions. One adult cestode was identified in the mantle cavity and was not associated with lesions. Other common concurrent parasitic infections included enteric Aggregata spp. infection, branchial Rickettsia-like organism infection, enteric nematodiasis, and an arthropod-associated branchitis.
Octopuses, together with squid, cuttlefish, and nautiluses, comprise the class Cephalopoda of the phylum Mollusca. Octopus anatomy and microanatomy differ markedly from vertebrate species and include an extensive nervous system that extends into each of the 8 arms, as well as many organs without a vertebrate homolog, such as branchial appendage and siphon. Cephalopods lack adaptive immunity, and hemocytes are the primary immune effector cell, performing functions similar to and beyond that of vertebrate granulocytes, including coagulation, phagocytosis, capsulation, and inflammation. Hemocytes are small ovoid cells with condensed nuclei. Detailed texts on the anatomy and histology of octopuses are available.3,6
Approximately 230 parasitic species, across numerous taxa, have been reported to infect cephalopods. 12 The most commonly reported parasites of cephalopods include the coccidian genus Aggregata, ciliate prostistans of Ancistrocomidae and Opalinopsidae, protozoal flagellates of Ichthyobodo spp., various species of endosymbiont dicyemids, several monogeneans, trematodes such as Derogenes varicus and Lecithochririum sp., larvae of the cestode orders Tetraphyllidea and Trypanorhynchidea, nematode larvae of the ascarid Anisakis simplex and Anisakis pegreffii, and copepod crustaceans such as Cholydia intermedia, lichomolgids, and the larval stages of Pennella varians.6,9,15
Parasites are acquired from the marine environment and can infect cephalopods through ingestion of parasitized food items (eg, crustaceans). Numerous arthropods and helminth species infect both fish and cephalopods with low host specificity; however, cephalopods serve as definitive host to certain metazoan and protozoan parasitic species, such as members of the subfamily Cholidyinae, copepod organisms, and Aggregata spp.12,16There is often consistent organ specificity: Ancistrocomidae parasitize the skin and gills, Chromidina spp. target the renal appendages, Opalinopsis spp. infect the liver and intestines, Ichthyobodo spp. infect the gills, 15 and Aggregata spp. occur most frequently and infect the digestive tract.12,16 Dicyemida, recently classified within Spiralia, are often present in the renal sac of benthic cephalopods.
Cestodiasis in cephalopods is common, and many cephalopod species serve as intermediate or paratenic hosts, and act as vectors for other intermediate or definitive hosts.12,17 Adult cestodes are not frequently reported in cephalopods, but the tremendous diversity of larval and post-larval stages found in cephalopods suggests that they are important intermediate hosts for the development of adult stages that parasitize cartilaginous and bony fish. In cephalopods, larval cestodes most often infect the digestive tract, but may be found free in the mantle cavity or encysted within the mantle musculature. 17 There have been infrequent reports of unidentified adult cestode infection in Octopus cyanea, in which the cestodes were identified in the circulatory system of the arms and throughout the body, and sometimes filled the entire mantle cavity. 9 The most commonly reported cestode to infect cephalopods is Phyllobothrium spp., 12 but cestodes from 3 other genera have also been identified in the common octopus (Octopus vulgaris) and include the onchoproteocephalidean Acanthobothrium spp., the tetraphyllidean Anthobothrium spp., and the trypanorhynch Nybelinia spp. 17 Adult stages of Tetraphyllidea and Trypanorhynchea are found within the gastrointestinal tract of sharks, skates, and rays, and their larval forms are some of the most commonly identified cestodes in cephalopods. 9
Despite the relatively large number of publications describing cephalopod infection with cestodes, there is only a single report describing the histologic findings associated with this infection. The single report histologically examined only the salivary gland, which was found to have inflammation associated with cestode infection. The remainder of the digestive tract and other organs were not examined in the study.7,8 There are no reports characterizing the histopathology of enteric cestodiasis in cephalopods or any reports that systematically study the histopathology of this infection with examination of full tissue sets. Due to infrequent reporting, the clinical and pathologic significance of cephalopod cestodiasis is unknown.
Lesions associated with enteric cestodiasis have not been described, but we hypothesize that this form of cestodiasis in octopuses can cause a host inflammatory response and can be a pathogenic infection. The objectives of this study were to characterize histologic lesions associated with cestodiasis in octopuses and to help pathologists, clinicians, biologists, and aquarists understand the pathogenicity and clinical significance (if any) of this infection in octopuses.
Materials and Methods
Northwest ZooPath archives were searched for cases of octopus necropsy and biopsy specimens with the histologic diagnosis of cestodiasis. Slides were reviewed and cases with cestodes (solid bodied parasites with calcareous corpuscles) were included. Minimal clinical information was collated, specifically the submitting institution and the manner of death (spontaneous death or euthanasia). All animals were maintained in captivity at aquariums in the United States in accordance with the Association of Zoos & Aquariums recommendations for management of octopuses in human care. 2 Animals were either found dead or were euthanized with the following 2-step protocol: the animal is first chemically anesthetized to the point of nonresponsiveness with MgCl2. Subsequently, ethanol is added to reach 10% concentration and until all activity and respiration have stopped for at least 15 minutes. The animal is then removed, and a necropsy is performed. Necropsy tissues were obtained at referring institutions and fixed by submitting veterinarians in 10% neutral buffered formalin. Tissues were processed routinely, embedded in paraffin, sectioned at 5 µm, mounted on frosted glass slides, and stained with hematoxylin and eosin. Slides were examined by multiple pathologists (E.E.B.L., D.K.F.), and histologic findings of parasitism were collated and described.
Results
Cestodiasis was identified in 4 common octopuses (O. vulgaris), 3 Caribbean reef octopuses (Octopus briareus), 2 two-spot octopuses (Octopus bimaculoides), and 1 giant Pacific octopus (Enteroctopus dofleini) (Table 1). The following tissues were evaluated histologically: digestive gland (n = 10), gill (n = 10), branchial appendage (n = 10), salivary gland (n = 9), renal appendage (n = 9), chitinous alimentary tract (n = 8), reproductive tract (n = 8), intestines (n = 7), eyes (n = 7), body wall (n = 6), cecum (n = 6), brain (n = 6), branchial gland (n = 5), branchial heart (n = 5), systemic heart (n = 5), white body (n = 4), and ink sac (n = 3). Cestodes were present in the cecum (n = 5/6), intestines (n = 4/7), digestive gland (n = 3/10), chitinous alimentary tract (n = 2/8), renal appendage (n = 1/9), and salivary duct (n = 1/9). In 5 cases, cestodes were invasive and associated with hemocytic infiltration (ie, inflammation) and tracts of necrotic tissue in the intestines (n = 3), digestive gland (n = 3), and/or renal appendage (n = 1) (Fig. 1a, b).
Cestode and other parasitic infections in captive octopuses.
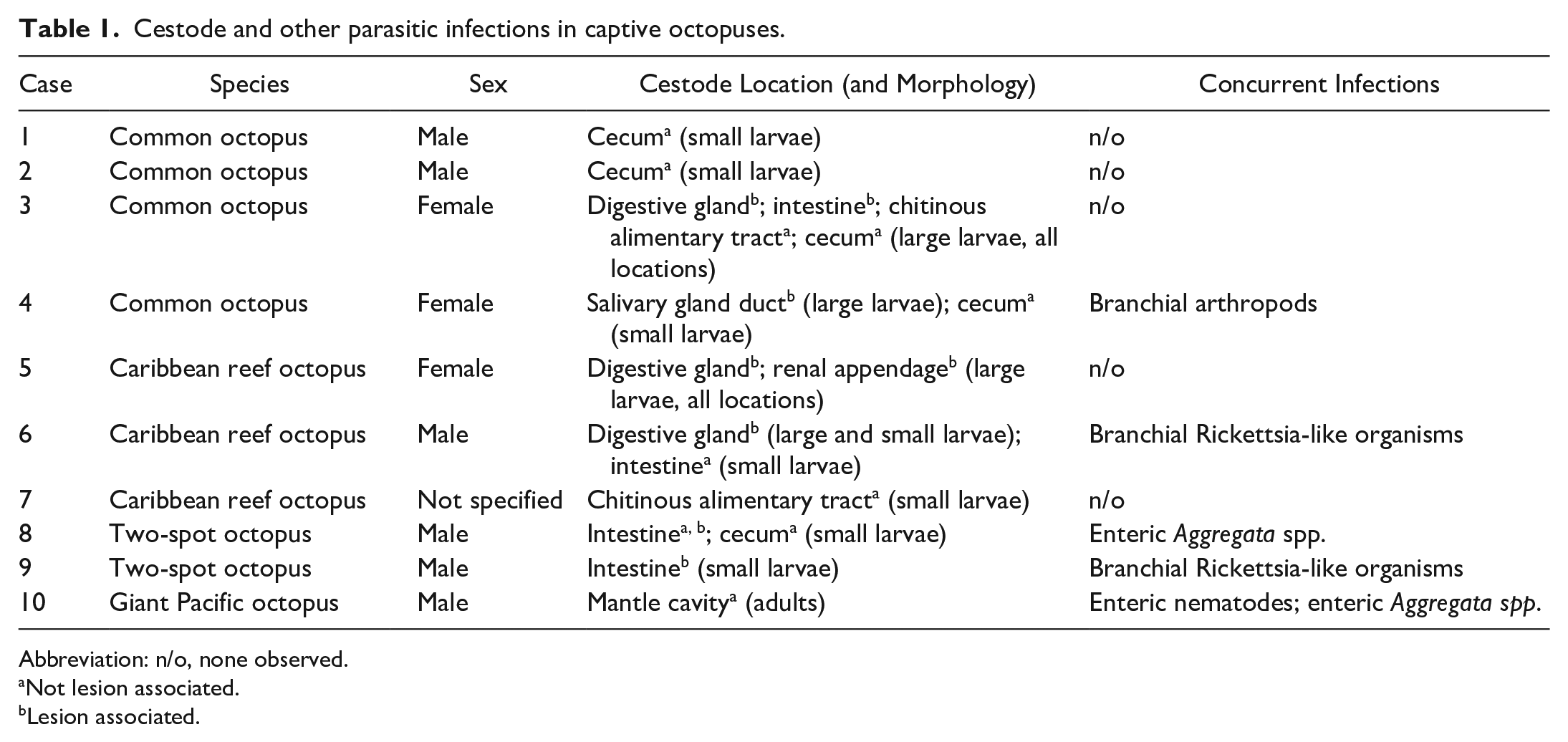
Abbreviation: n/o, none observed.
Not lesion associated.
Lesion associated.
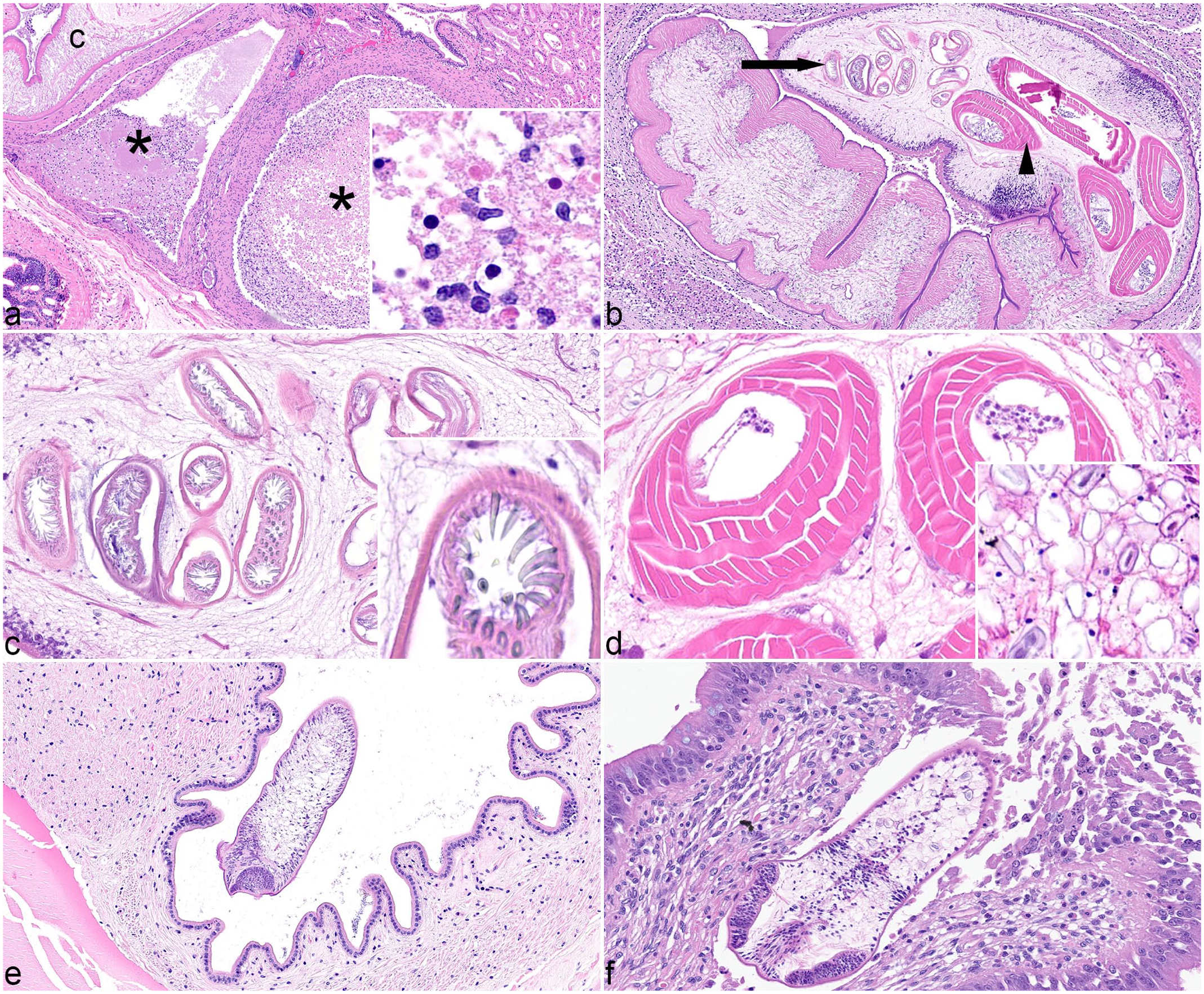
Cestodiasis, octopuses. All images hematoxylin and eosin. (a) Digestive gland, Caribbean reef octopus, case 6. Large necrotic tracts (asterisks) are adjacent to a large cestode larva (c). Necrotic tracts contain flocculent eosinophilic material and hemocytes (inset). (b) Digestive gland, common octopus, case 3. A large cestode larva with hooks (arrow) at the anterior end that are attached to muscular bulbs (arrowhead) positioned posteriorly. The cestode compresses the surrounding, inflamed tissue. (c) Large cestode larva, common octopus, case 3. Tentacles armed with hooks are inverted within sheaths. Inset: Hooks are uniform size and shape at this location of the tentacle. (d) Large cestode larva, Caribbean reef octopus, case 6. Four muscular bulbs comprise concentric layers of muscle. Calcareous corpuscles (inset) are in the outer spongy parenchyma. (e) Esophagus, Caribbean reef octopus, case 7. A small cestode larva is free in the lumen. (f) Intestines, two-spot octopus, case 9. A small cestode larva invades the mucosa and submucosa with associated ulceration and inflammation.
In all 10 cases, cestodes were characterized by a 4- to 6-µm-thick, homogeneous, eosinophilic tegument, spongy internal parenchyma, and numerous peripheral calcareous corpuscles. Three different cestode morphologies were identified: adult (n = 1), large larvae with hooks and tentacles (n = 4), and small larvae with suckers (n = 7). Two cases (cases 6 and 4) had both small and large larvae. It is unknown whether the small larvae with suckers represented the anterior end of large larvae with hooks. Adult cestodes were up to 1 mm in width, too long to measure histologically, and had evident proglottids and elaborate suckers. Large cestode larvae were up to 650 µm in diameter with bilaterally symmetrical bothria with multiple tentacles that were anchored by prominent bulbs encircled by striated retractor muscles that attached to everted or invaginated hooks (Fig. 1c, d). 5 Small larval cestodes were 150-250 µm in diameter with prominent suckers; proglottids, tentacles, and hooks were not observed (Fig. 1e, f). In 8 cases, larvae or adults were present in the lumen of at least one organ without evident mucosal attachment or lesions (Fig. 1e); in 3 of those cases (3/8), larvae were also present in another organ where they were associated with histologic lesions. Luminal larvae without associated lesions were in the cecum (n = 5), intestines (n = 2), chitinous alimentary tract (n = 2), salivary duct (n = 1), and free in the mantle cavity (n = 1).
In the intestines, larvae were attached to the mucosa or submucosa (Fig. 1f) with mucosal epithelial necrosis and erosion or ulceration, necrosis of the cells in the lamina propria, and infiltration by low numbers of hemocytes, some of which were necrotic. In 2 cases of intestinal infection, there was segmental inflammation with low numbers of hemocytes and a small amount of necrosis of the tunica muscularis. In the digestive gland and/or accessory digestive gland, there was subtotal effacement of up to 60% to 80% of the accessory digestive gland (n = 2) and 10% to 60% of the digestive gland (n = 2) by nodules up to 650 µm in diameter comprised of inflammation and necrosis centered around cestode larvae. In the third case of digestive gland cestodiasis, the entirety of the gland was not present, but an estimate of 10% glandular effacement was associated with larvae. Foci of necrosis were characterized by amorphous amphophilic to eosinophilic granular material admixed with cellular debris and fragmented hemocytes. There was multifocal degeneration of surrounding tubular epithelium characterized by swollen and vacuolated cytoplasm, and necrosis characterized by loss of cellular membrane integrity with pyknotic to karyorrhectic nuclei. The mass effect of cestode larvae and inflammation often compressed adjacent structures, such as the ink sac complex, intestines, and/or other structures. In the renal appendage, cestode larvae were surrounded by sloughed and exfoliated renal tubular epithelial cells and compressed or degenerative renal tubular epithelial cells. Renal tubular lumina often contained amorphous eosinophilic, granular to flocculent extracellular fluid. In all, 1 to 6 cross sections of larvae were seen per organ.
Concurrent parasitism was present in 5 cases and included Aggregata spp. (n = 2), Rickettsia-like organisms (n = 2), nematodes (n = 1), and arthropods (n = 1). Animals infected with Aggregata spp. had multifocal cystic cavities in the gastrointestinal submucosa and tunica muscularis that contained various life stages including sporozoites, immature sporocysts, macrogamonts, and microgamonts.6,11 The Aggregata spp. infection was associated with infiltration of low to moderate numbers of hemocytes that effaced portions of intestinal mucosal epithelium and expanded the tunica muscularis and connective tissue of the intestines. Two cases had Rickettsia-like organisms in the gills with no associated inflammation. One case had a nematode infection in the tunica muscularis of the intestines that was associated with tissue compression, but no inflammation. One case had arthropod parasitism of the gills associated with infiltration by moderate numbers of hemocytes and a small amount of necrosis.
Discussion
Larval cestodiasis can be a pathogenic infection in cephalopods, but was typically only associated with mild inflammation and necrosis. As such, it is unlikely that larval cestodiasis in cephalopods is a lethal disease. Infection with adult cestodes was not associated with any lesions and may be incidental; that said, only one octopus was infected with an adult, and hence definitive conclusions cannot be drawn due to the low case number. The presence or lack of pathology associated with larval cestodiasis clustered closely with the anatomical location of infection. When present in the cecum and chitinous alimentary tract, cestodes were consistently confined to the lumen without tissue invasion. The lack of tissue invasion in these areas may be in part due to a physical barrier to infection or reaction. The chitinous alimentary tract includes the esophagus and gizzard, which are lined by a thick layer of chitin that may be impermeable or inhospitable to cestodes, as with other invertebrate species. 4 When larval cestodes were present in the digestive gland, intestines, and renal appendage, tissue invasion and an inflammatory response were identified. Assuming cestodes infect octopuses via the digestive tract, it is not clear why some of these cases had tissue invasion and others did not. 9 One possibility is that these captive animals are exposed to parasites that they do not encounter in the wild (perhaps through food sources), and as such lack tolerance to novel infections. 19 Another possibility is that the animals with an inflammatory response had some immunosuppression or modulation, allowing for tissue invasion. Senescence is a common cause for immune depression/modulation in octopuses, which is the period of time following sexual maturity, with or without reproduction, wherein octopuses go through a sort of “controlled death.” The period is incompletely understood, but often highlighted by inappetence and associated weight/body condition loss, nonhealing wounds, and varying degrees of lethargy. 13 Another common cause of stress and immune modulation includes the capture, collection, and transport of these animals from the wild. 6 Captivity-associated stressors are often multifactorial, with some documented factors including poor water quality, high stocking density, self-induced trauma, and inappropriate tank size, all of which can contribute to a compromised immune system.6,14
Inflammation associated with larval cestodiasis was characterized by an accumulation of hemocytes, which are the primary immune effector cell of cephalopods. Inflammation has been demonstrated in many parasitic infections of octopuses. The inflammatory response to cestodes is similar to the response to other tissue-invading parasitic infections and includes various hemocytic defenses such as phagocytosis of foreign debris, encapsulation, and fibrosis. 6 In 2 cases of digestive gland cestodiasis, necrotic tracts were present in the same region as cestode larvae. The extent of the host response in these cases exceeded that of mild inflammation and may be attributed to specific host-parasite interactions, the absolute burden of disease, or the extent of larval migration.
Based on the size and morphologic features, the large larval cestodes in all cases likely originate from the order Trypanorhynchidae. There are 254 species within Trypanorhynchidae, and species identification is based on morphologic characteristics, such as scolex shape, number of bothria, tentacle characteristics, genital organs, and bothrial pits. These characteristics are typically evaluated by examination of whole specimens rather than histologic sections. 10 Tentacles were not present in the small cestode larvae, and these larvae may represent a section without structures of the Trypanorhynchidae scolex, or they may represent cestodes of a different order. Further identification of the cestodes in these cases was not attempted.
The most common concurrent diseases included enteric Aggregata spp. infection, branchial Rickettsia-like infection, enteric larval nematode infection, and branchial arthropod infection. Aggregata spp. is one of the most common Apicomplexan parasites of cephalopods.6,9,11,18 While Aggregata spp. is not typically a lethal infection in octopuses, it can severely weaken the immune system of octopus and may have been a major contributing factor in the death of multiple animals in this series. 6 Branchial Rickettsia-like organisms were not associated with any inflammation or necrosis. This is consistent with previous reports of these organisms, which are generally considered incidental, but may cause some respiratory compromise if present in large numbers or combined with stressors such as overcrowding or poor water quality. 6 Coinfection with multiple species of parasites was relatively common in this cohort, the cause of which is unknown, although dietary and environmental factors, as well as senescence, may have contributed. 13 The state of reproduction and senescence for the animals in this series is unknown and it is not possible to determine to what extent their susceptibility to infection was affected. When multiple species of closely related parasites coinfect a host, there may be cooperation that results in increased virulence. However, parasites may also modulate the effects of other parasitic species to prolong the life of the host. Interference competition is distinct from exploitative competition but may be necessary for a parasite’s evolution. 1
This case series demonstrates that larval cestodes can be pathogenic in octopuses, particularly in the digestive gland, intestines, and renal appendage, where infection is often associated with inflammation and necrosis. Larval or adult cestode infection in other portions of the alimentary tract was not associated with inflammation or tissue invasion. Cephalopods, and coleoids specifically, have been the focus of research for more than a century due to their many unique anatomical and physiologic features. While cephalopods continue to be integral research animals, pathologists likely will continue to identify various diseases.
Footnotes
Acknowledgements
We are grateful to the individuals and institutions who contributed case material, including Drs Brian Joseph (Sea Life Kansas City) and Jeff Proudfoot (Indianapolis Zoo). We also thank Leroy Brown (Histologic Consulting Services) for slide preparation, Dr John Trupkiewicz (Northwest ZooPath) for his histopathologic diagnoses, Cathy Minogue (Northwest ZooPath) for data retrieval, and the aquarists at Monterey Bay Aquarium.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
