Abstract
This retrospective study examines pathologic findings in 593 captive cephalopods (340 octopuses, 130 cuttlefish, 33 squid, and 90 nautiluses; 22 species in total) submitted to International Zoo Veterinary Group Pathology between May 2003 and August 2022. Common octopus, European common cuttlefish, hummingbird bobtail squid, and chambered nautilus were the most numerous species from the included orders of Octopoda, Sepiida, Sepiolida, and Nautilida, respectively. Commonly identified conditions included coccidiosis and renal dicyemid mesozoan infection in octopuses, amebiasis in squid, bacterial infections in cuttlefish, and idiopathic multisystemic inflammatory disease in nautiluses. Coccidiosis was most frequent in common octopuses, giant Pacific octopuses, and California 2-spot octopuses (present in 68.4%, 46.3%, and 23.8% of these species, respectively) and was the attributed cause of death in 32.3%, 36.0%, and 60.0% of such cases, respectively. Ulcerative dermatitis (UD) was common, affecting squid, cuttlefish, and octopuses, while many nautiluses exhibited black shell disease and/or UD. Notable differences in the prevalence of UD were detected between laboratory and aquaria-housed decapodiforms; 52.2% for laboratory squid versus 20.0% in aquaria; 51.3% in laboratory cuttlefish versus 11.0% in aquaria. All octopuses and nautiluses in the study were derived from aquaria. Semelparity-associated death in Coleoidea species was identified in 22.4% of octopuses, 11.5% of cuttlefish, and 6.1% of squid. This report aims to provide an overview and reassessment of species-specific disease patterns under aquarium and laboratory management as a starting point for future developments in husbandry and disease investigation.
The Cephalopoda is an ancient molluscan class of primarily marine ocean-dwelling carnivorous animals that arose during the late Cambrian period. 41 It is represented today by over 750 species listed in the International Union for Conservation of Nature (IUCN) Red List, 45 divided into 2 extant subclasses Coleoidea (octopus, squid, and cuttlefish) and Nautiloidea (nautilus). 41 Characteristic features include symmetrical bodies comprised of 2 conjoined parts (a head and foot-like tendrils [eg, arms or tentacles]) 66 and a common ability to move through water by using a tubular siphon, which provides jet propulsion. 6 All cephalopods have both male and female sexes. However, a key life cycle difference between the subclasses is postreproductive programmed death, otherwise known as semelparity or postreproductive senescence. In Coleoidea, individuals reproduce once before geriatric degeneration and subsequent death. 72 In contrast, nautilus are iteroparous species (ie, reproducing multiple times) of markedly greater normal longevity (approximately 20 years, reaching sexual maturity at around 15 years of age, 17 versus a maximal 3-year lifespan for the coleoid cephalopods in this study). Other anatomical and physiological differences in the nautilus include a calcified shell encasing the internal organs, a siphuncular mechanism for buoyancy control, and a recognized sensitivity to temperature and water depth (typically less than 25°C and depths of 100–700 m). 18
Cephalopods are highly intelligent, charismatic, and popular in aquarium exhibits. Despite this, the scientific literature documenting spontaneous diseases of cephalopods, particularly in aquaria, is scattered and poorly reviewed with only limited scale retrospective studies having been recorded to date.26,76,77 This study describes spontaneous disease identified in 593 cephalopods submitted to a private diagnostic service (International Zoo Veterinary Group [IZVG] Pathology) over a 19-year period, placing the findings in context with the previous literature.
Materials and Methods
A retrospective study of cases extracted from the IZVG Pathology database archive (Microsoft 365 Access) was performed for cephalopod submissions of either formalin-fixed whole specimens or various formalin-fixed necropsy tissue sets, submitted by clinicians representing zoological collections/aquaria and private laboratories, between 2003 and 2022. Histologic case records were retrieved using “common name of species” keywords (ie, octopus, squid, cuttlefish, or nautilus) as the primary data entry selector to identify relevant specimens. All cephalopods included in the study had died prior to the submission of whole bodies or specific tissues for diagnostic purposes, and as such ethical approval was not required for this review. When whole specimens were submitted, gross inspection of the carcass was performed prior to selection of tissue samples for histology (Fig. 1a–f). Formalin-fixed paraffin embedded (FFPE) tissues were routinely processed, sectioned at 5 µm, and stained with hematoxylin and eosin (HE). Selected tissues were stained with Twort’s Gram stain (Ollett’s variant), periodic acid–Schiff (PAS), and modified acid-fast (Ziehl-Neelsen) techniques. Cases were examined by board-certified veterinary pathologists and/or Royal College of Veterinary Surgeons (RCVS)-recognized specialists in Veterinary Pathology (Zoo and Wildlife) (A.F.R., M.F.S., D.D., and C.R.S.), and then re-examined by 2 of the authors (A.F.R. and M.F.S.) in cases with uncertainty in the original diagnosis. Data recorded from each individual case included the species, age category, a brief clinical summary, histological descriptions, histopathological diagnoses, report comments, lesioned organs, major disease categories, etiological agents, whether the cause of death/reason for euthanasia was determined, and type (aquarium/laboratory) of submitting collection. Determination of “cause of death” was based upon the presence of either: (1) a severe lesion (eg, florid inflammation with/without infectious agent, marked tissue disruption/necrosis) affecting a single primary organ (ie, brain, gills/branchial hearts, systemic heart, alimentary tract, digestive gland, kidneys); (2) moderate to severe lesions in multiple organs; or (3) marked atrophic changes affecting multiple organs (eg, inanition—digestive gland, salivary glands). Where lesions were only moderate in a single organ, or mild in multiple organs, and in the absence of atrophic changes, the cause of death was labeled “suspected” but not considered definitive.

Gross images of cephalopods. (a) Typical formalin-fixed specimen, common octopus (Octopus vulgaris). (b) Living specimen, chambered nautilus (Nautilus pompilius). Photo courtesy of sea life. (c) Skin, common octopus. Irregular circumscribed foci of ulceration involving the mantle skin (ulcerative mantle disease). (d) Eye, common octopus. Diffuse cloudy lens opacity, indicative of a cataract. (e) Outer shell, chambered nautilus. Numerous linear black encrustations replace normal shell material in the growth lines at the margin of the shell. (f) Inner shell, chambered nautilus. Internal lesion in the shell pictured in Fig. 1e. The siphuncular opening is eroded and enlarged and the shell at the eroding margins is black discolored.
Culture transport medium swabs (charcoal or Amies-liquid/gel; various manufacturers) were submitted with histological samples for a subset of submissions or were taken on gross examination of fresh unfixed carcasses. Routine microbiology was performed by a referral microbiology laboratory, which performed cultures at 22ºC and 37ºC for a minimum of 7 days, using Columbia agar with defibrinated horse blood, MacConkey agar, Columbia blood agar with nalidixic acid, and Sabouraud dextrose agar. Cultured microorganisms were identified using API Microbial Identification (bioMerieux UK Ltd; Basingstoke, Hampshire, UK) or VITEK 2: Healthcare microbial identification system (bioMerieux UK Ltd; Basingstoke, Hampshire, UK). Some identifications were confirmed by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF).
Results
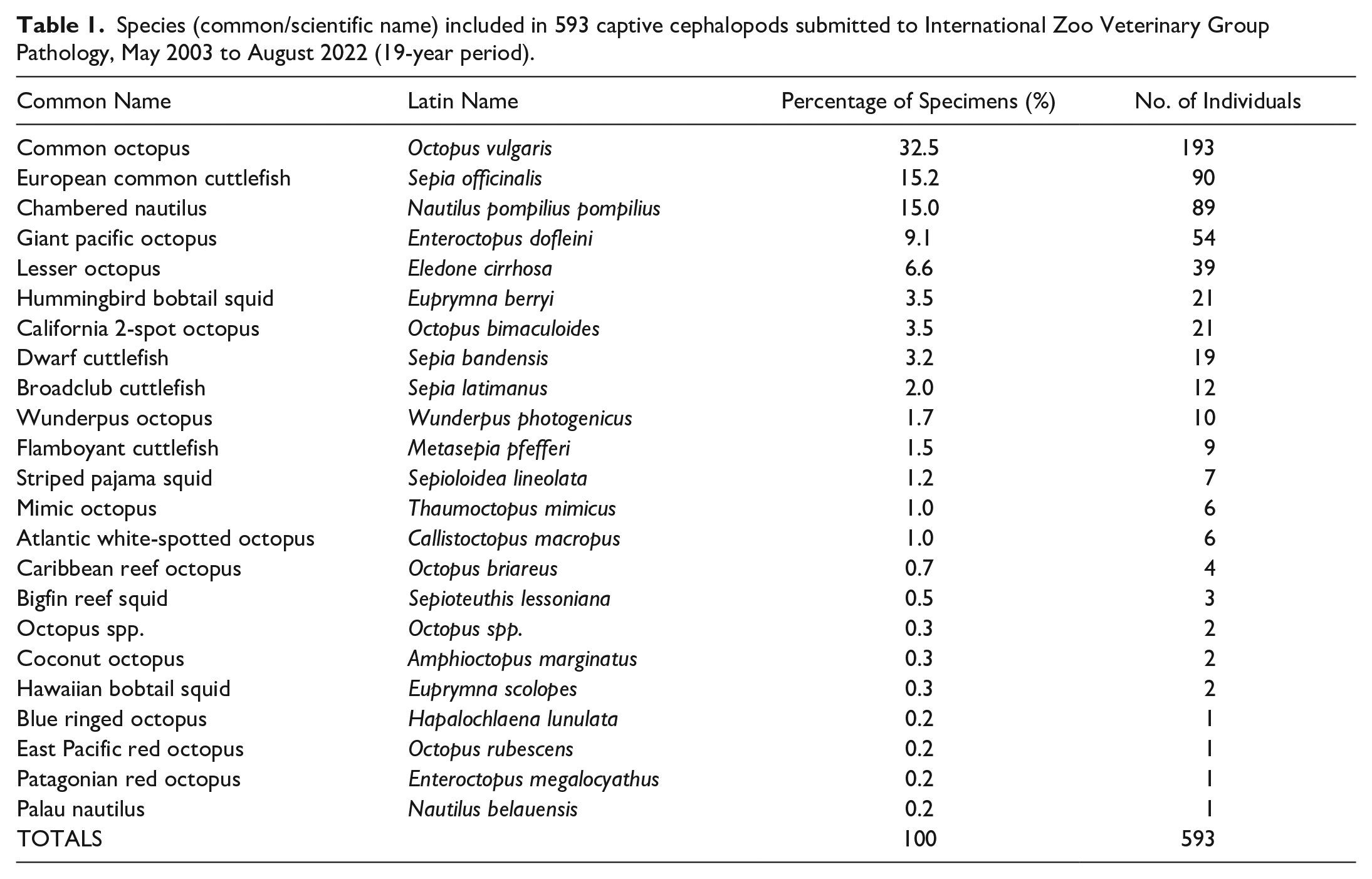
During the period May 2003 to August 2022, 593 cephalopod submissions representing 22 species from 4 orders (Octopoda, Sepiida, Sepiolida, and Nautilida) were submitted. Common octopus (193), European common cuttlefish (90), hummingbird bobtail squid (21), and chambered nautilus (89) were the most numerous species (Table 1 and Supplemental Table S1). Fig. 2a–i demonstrates features of immature, mature, and senescent/atrophic tissues in octopuses used to assist age classifications.
Species (common/scientific name) included in 593 captive cephalopods submitted to International Zoo Veterinary Group Pathology, May 2003 to August 2022 (19-year period).
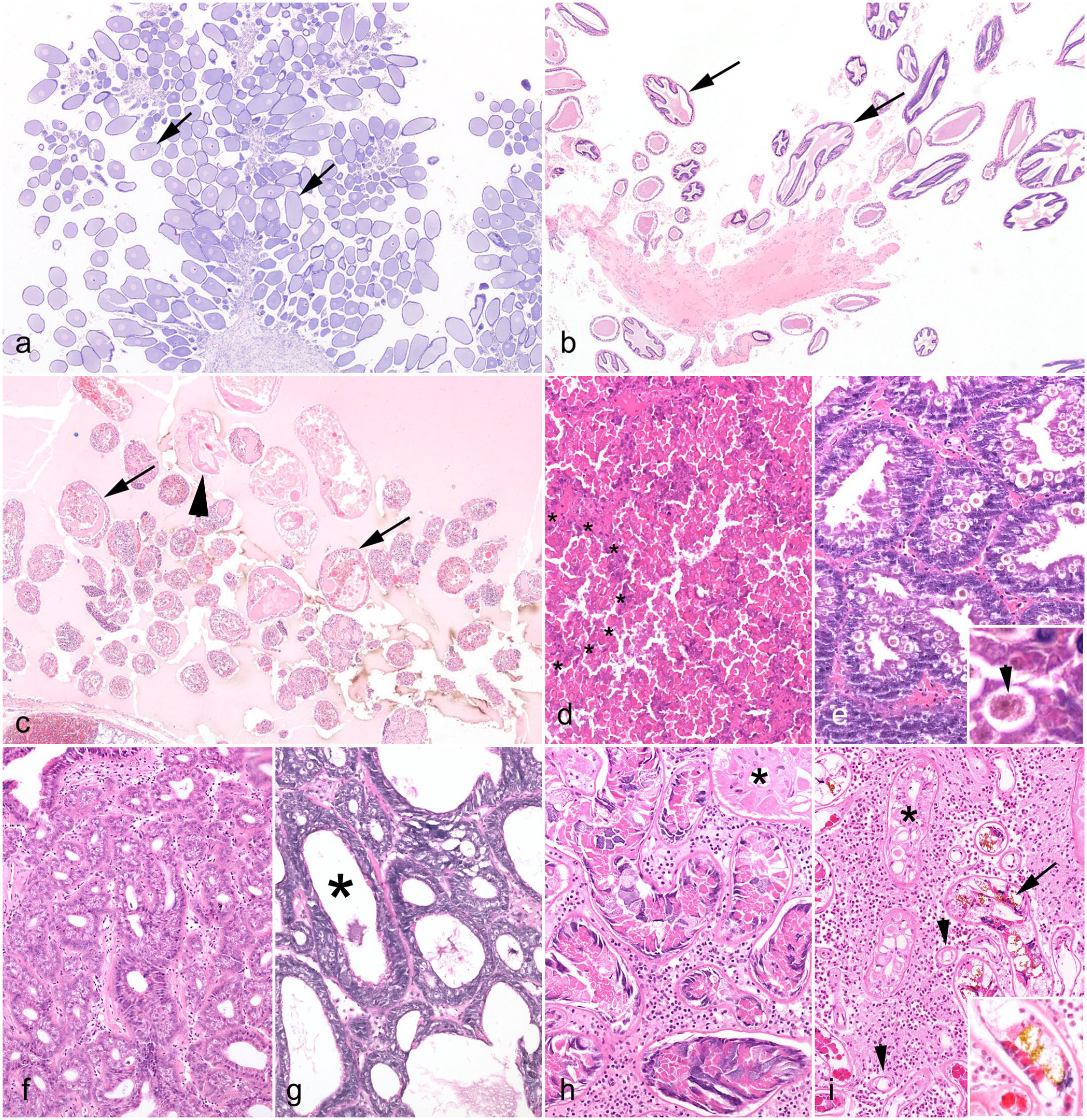
Coleoidea—effects of senescence. Common octopus (Octopus vulgaris). Hematoxylin and eosin (HE). (a–c) Ovary. (a) Immature. Quiescent, with numerous small oocytes (arrows). (b) Postpubescent (mature). Active, with multiple maturing follicles (arrows). (c) Senescent. Diffusely atrophic ovarian tissue including multiple collapsed and degenerating follicles/eggs (arrows), with occasional remnant eosinophilic and convoluted capsular debris (arrowhead). (d–e) Digestive gland. (d) Healthy adult. Abundant eosinophilic densely granular epithelial cytoplasm almost obscures tubular architecture (basement membrane of a single tubule denoted by asterisks, for comparison with 2e). (e) Senescent adult. Dilated tubules and reduced cytoplasm with brown-tinged excretory granules (arrowhead in inset). Note paucity of normal eosinophilic granules. (f–g) First salivary gland. (f) Healthy adult. Plump secretory tubules. (g) Senescent adult. Atrophic and dilated tubules (asterisk). (h–i) Second salivary gland. (h) Healthy adult. Plump tubules and abundant heterogeneous cytoplasmic granules (asterisk, compare with 2i at same magnification). (i) Senescent adult. Tubules with brown excretory granules (arrow and inset), reduced luminal diameter (asterisk, compare with 2h at same magnification), and marked atrophy (arrowheads).
Table 2 lists the broad categories of pathological processes for each cephalopod order.
Prevalence of diseases processes in 593 captive cephalopods submitted to International Zoo Veterinary Group Pathology, May 2003 to August 2022 (19-year period).
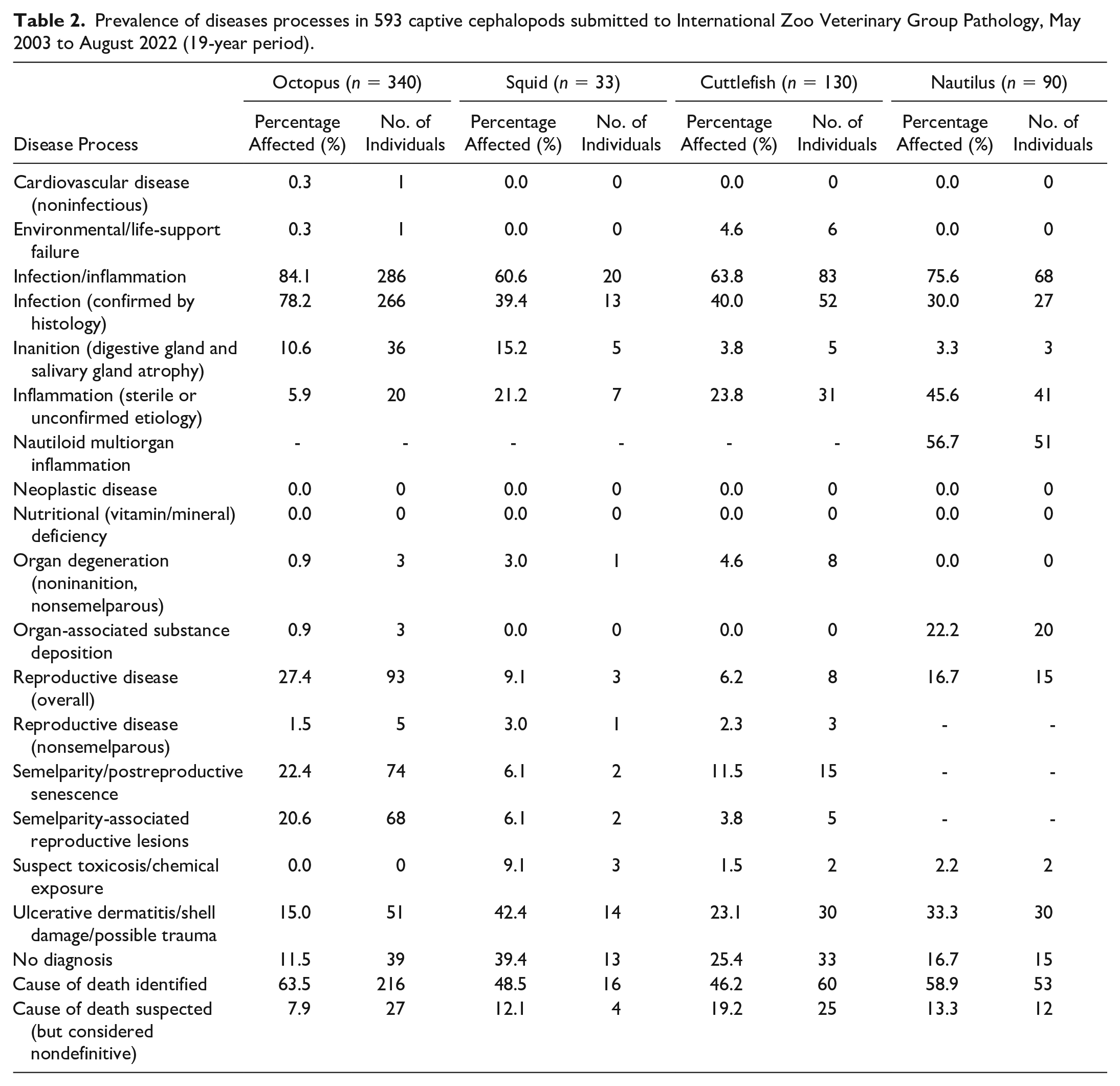
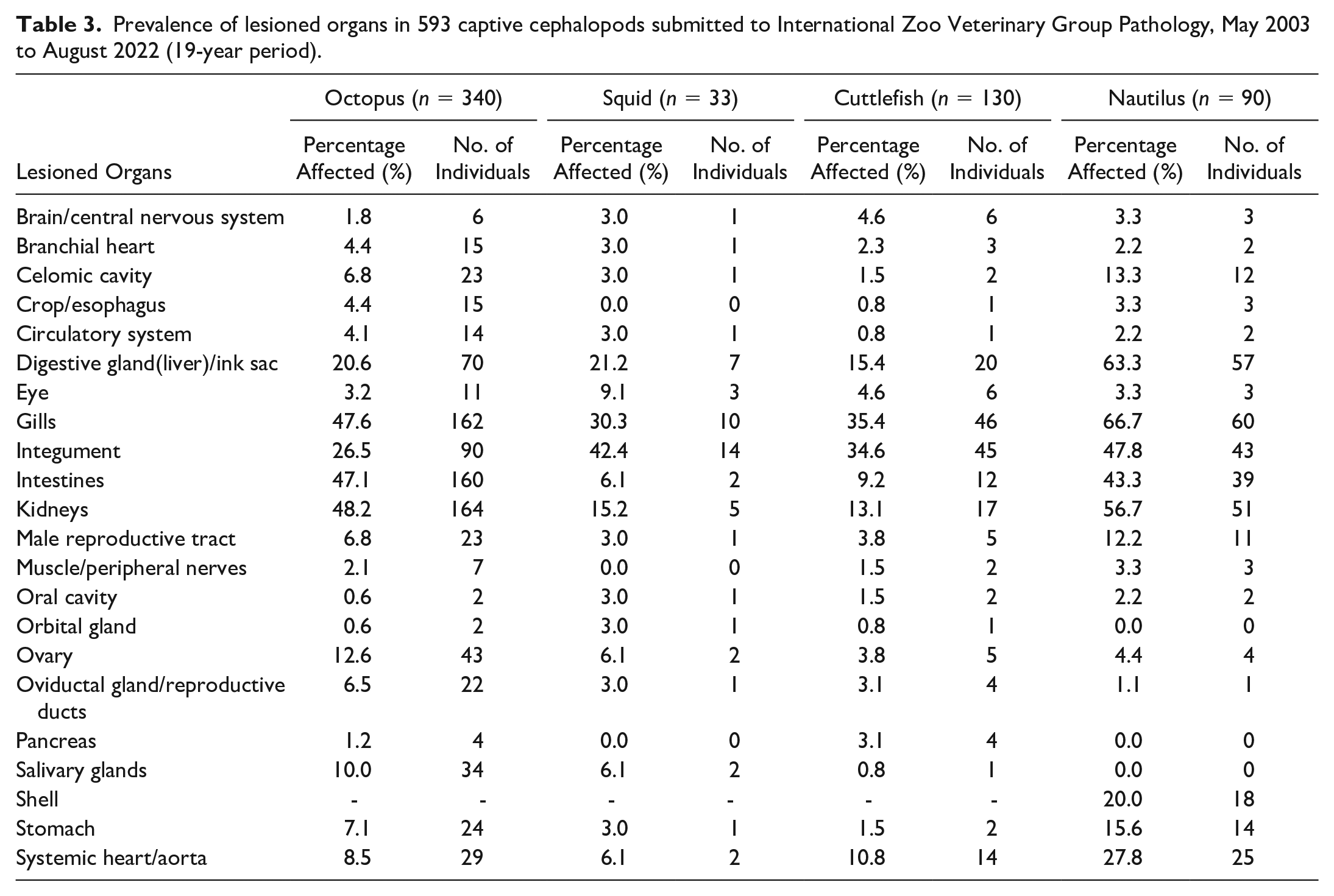
Table 3 lists the prevalence of organs with lesions for each cephalopod order.
Prevalence of lesioned organs in 593 captive cephalopods submitted to International Zoo Veterinary Group Pathology, May 2003 to August 2022 (19-year period).
Infectious/Inflammatory Disease
Table 4 highlights the prevalence of infectious microorganisms for each cephalopod order.
Prevalence of infectious micro-organisms in 593 captive cephalopods submitted to International Zoo Veterinary Group Pathology, May 2003 to August 2022 (19-year period).
Bacteria
Bacterial infections were common in all orders, characterized by histologically visible bacteria accompanied by tissue reactions such as hemocytic infiltrates, necrosis, and ulceration. Sepsis was defined by the presence of similar lesions in multiple organs. They represented the most prevalent confirmed infectious disease in squid (8/33; 24.2%), cuttlefish (47/130; 36.2%), and nautiluses (16/90; 17.8%). Octopuses exhibited a similar prevalence (55/340; 16.2%) but bacterial infections were less numerous than some protozoal infections (eg, coccidia) in this order.
In squid, bacteremic sepsis was the presumed primary cause of death, mostly associated with ulcerative dermatitis (UD), in 6 of 8 bacterial infections. Branchitis, nephritis, or male reproductive tract inflammation were seen in single cases. Amongst the organisms grown in aerobic bacterial cultures of the outer mantle, gill, or inner mantle were Aeromonas hydrophila, Shewanella putrefaciens, Pseudomonas alcaligenes, Pseudomonas stutzeri, Rahnella aquatilis, Sphingomonas paucimobilis, and Stenotrophomonas maltophilia. In cuttlefish, bacteremic sepsis was determined to be a contributor/cause of death in 78.7% (37/47) of cases in which sepsis was histologically confirmed in association with tissue lesions. UD was most common (20/47; 42.6%), followed by gill (9/47; 19.2%) and digestive gland (5/47; 10.6%) infections. Infrequent bacterial lesions included enteritis, pancreatitis, reproductive disease, and meningitis/encephalitis (3/47; 6.4% each), with sporadic single cases with infections affecting the heart and kidneys. Agents isolated on routine microbiology included Acinetobacter johnsonii, Aeromonas salmonicida subspecies salmonicida, Alcaligenes faecalis, Alcaligenes species, Bergeyella zoohelcum/Weeksella virosa, Brevundimonas diminuta, Brevundimonas vesicularis, Pasteurella species, Pseudomonas fluorescens, Pseudomonas putida, Pseudomonas stutzeri, Sphingomonas paucimobilis, Vibrio alginolyticus, and Shewanella algae.
Bacteria represented the third most common infectious etiology in octopuses. The most frequently infected organs were integument (15/55; 27.3%), gills (12/55; 21.8%), systemic heart/aorta (8/55; 14.6%), intestines (7/55; 12.7%), peripheral vasculature (6/55; 10.9%), stomach (6/55; 10.9%), and gonads (4/55 7.3%; including ovaries and spermatophore ducts). Bacterial infections were usually associated with other disease processes, especially concurrent coccidial infections (28/55; 50.9%), UD (15/55; 27.3%) (Fig. 3a–d), and postreproductive senescence (11/55; 20.0%). Sepsis was identified histologically in 39/55 cases (70.9%), based on multiple organ infection/inflammation, where fresh tissue or culture-media swabs were unavailable for microbial cultures. In cases where bacteria were isolated, all were rod-shaped, either Gram-positive or Gram-negative, and non-acid-fast on special stains.
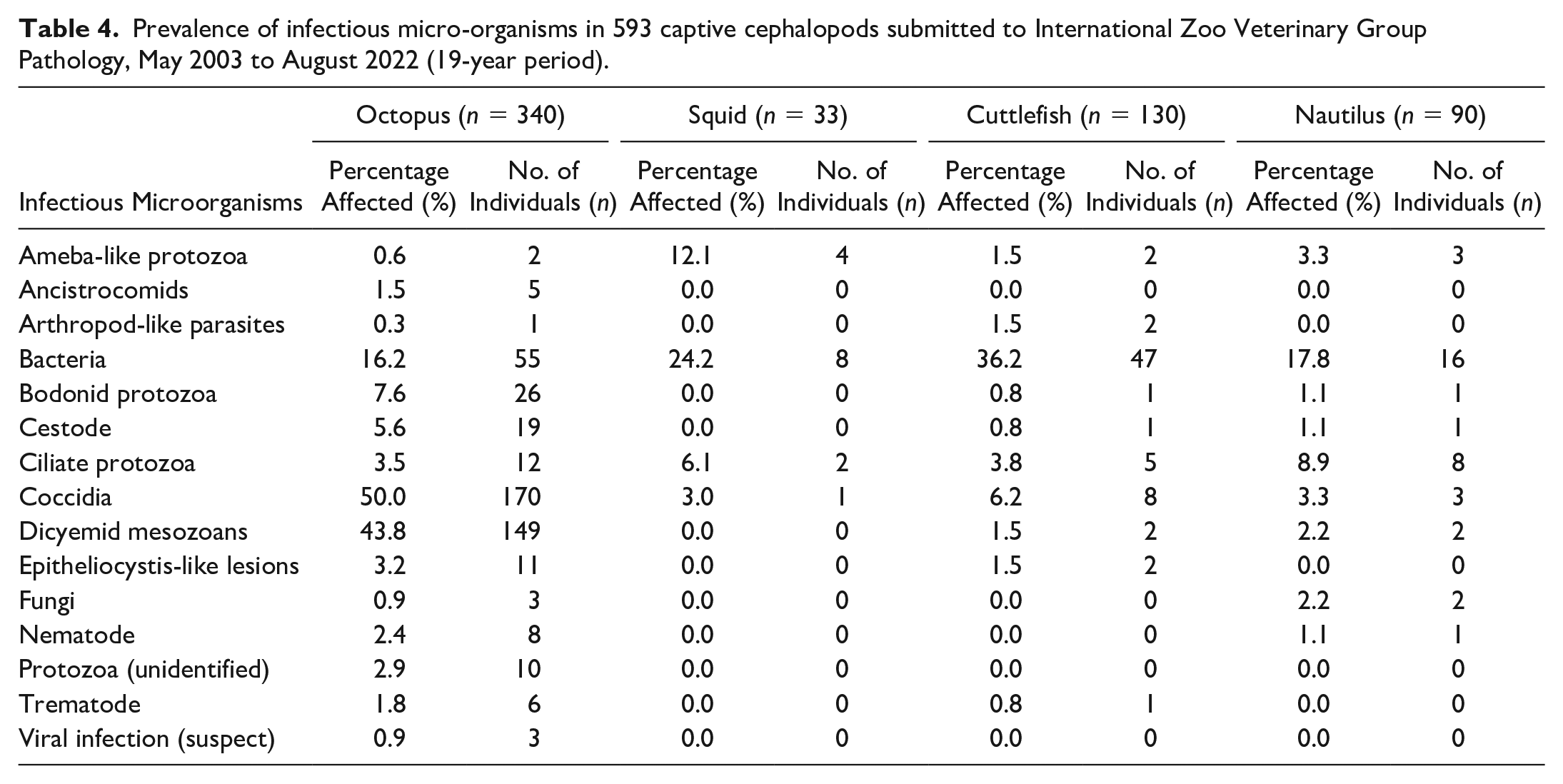
Histopathologic features of UD in Coleoidea. Hematoxylin and eosin (HE). (a) Mantle skin, common octopus (Octopus vulgaris). Marked acute epidermal and superficial dermal ulceration with black pigment dispersion and hemocytic infiltrates (asterisk). (b) Box from 3a. Numerous coccobacillary bacteria (asterisk) colonize the superficial connective tissues beneath the ulcer and underrun the adjacent epithelium. (c) Tentacle skin, common octopus (Octopus vulgaris). Epidermal ulceration, with superficial bacterial invasion and superficial edema. (d) Inset from 3c. Numerous rod-shaped to filamentous bacteria colonize the ulcer and underrun the adjacent epithelium (asterisk). (e) Mantle skin, hummingbird bobtail squid (Euprymna berryi). Epidermal ulceration with invasive presumptive amebic infection. (f) Box from 3e. Intradermal, invading, oval or circular unicellular ameba-like protozoa (arrow), characterized by basophilic cytoplasm with variable cytoplasmic vacuolation and typically distinct eosinophilic outer membrane. Each cell has a single small amphophilic circular nucleus and organisms vary in size up to approximately 40 µm diameter. (g) Mantle skin, European common cuttlefish (Sepia officinalis). UD with hemocytic infiltrates, denuded epithelium, and pigment dispersion. (h) Box from 3g. Mixed intralesional rod-shaped to fine filamentous bacteria (arrowheads) and Ichthyobodo-like protozoa (arrows). Hemocytic sludging within vessel (asterisk).
Infectious microorganisms were less common in nautiluses, with only 41.1% demonstrating histologically confirmed infections (37/90). Of these, bacteria were most frequently detected (16/37; 43.2% of infections). Bacteria primarily affected the gills (6/16; 37.5%), skin (5/16; 31.3%), intestines (4/16; 25.0%), kidneys (4/16; 25.0%), and digestive gland (2/16; 12.5%). All bacteria were rod-shaped, Gram-negative, and non-acid-fast on special stains.
Epitheliocystis-Like Lesions/Rickettsia-Like Organisms
Epitheliocystis-like lesions (ELLs)/Rickettsia-like organisms (RLOs)21,23 were detected in 3.2% (11/340) of octopuses (Fig. 4a) and 1.5% (2/130) of cuttlefish (Fig. 4b). In octopuses, these were found in the gills of all cases with only a single case (coconut octopus) exhibiting a generalized tissue distribution. In this animal, micro-colonies of Gram-negative organisms were present in epithelial cells of the gills and mantle (associated with additional multiple small foci of enlarged [ie, hypertrophied] epithelial cells not containing bacterial inclusions) and in the digestive gland. No other significant lesion was identified in this animal, and infection was considered the cause of death. In cuttlefish, both cases involved the gills, but were considered mild and incidental. ELLs/RLOs were not associated with inflammation in any cephalopod. No molecular diagnostic tests were performed on these cases. ELLs/RLOs were not detected in any squid or nautilus.
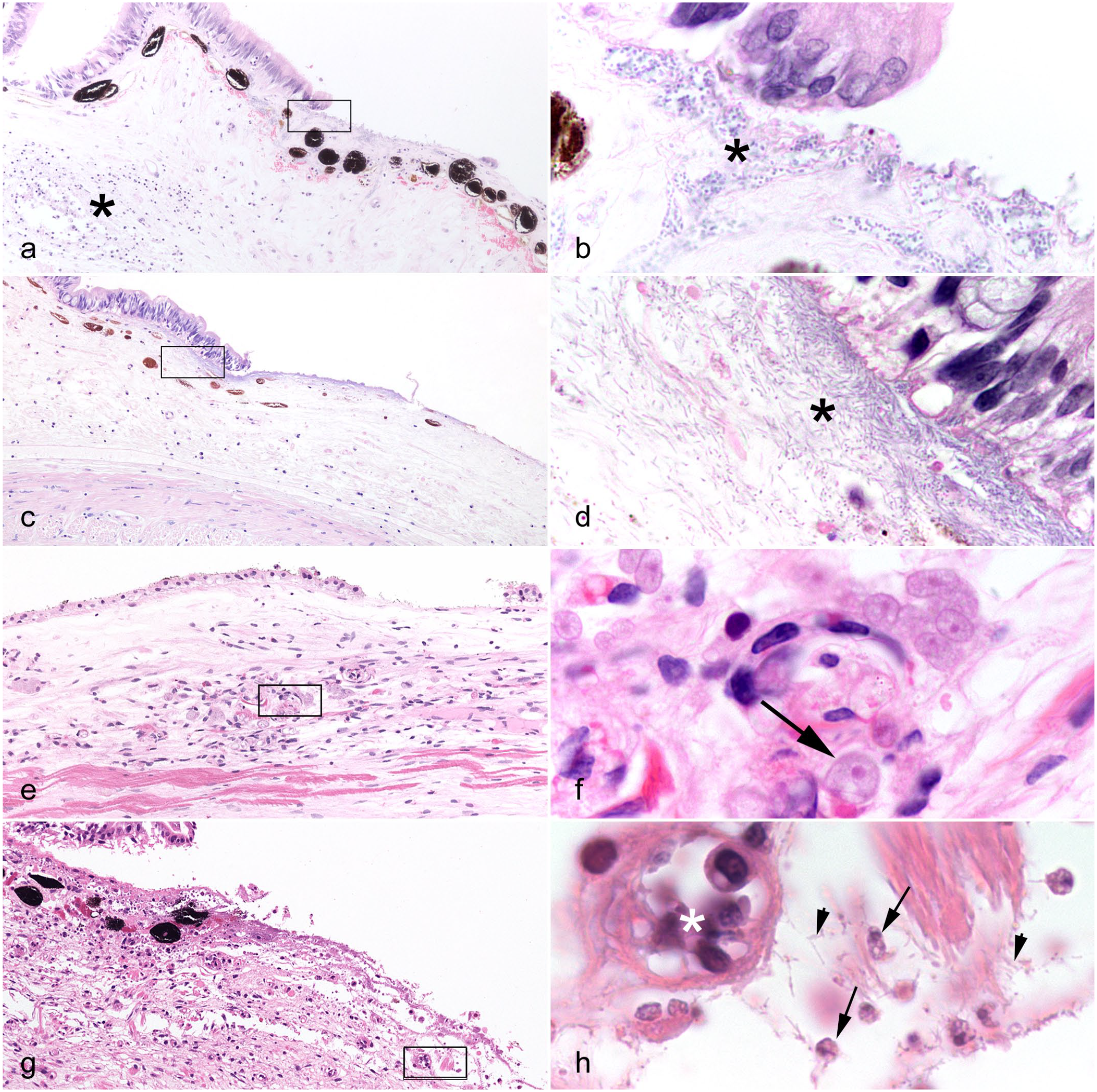
Histopathologic features of gill infections in cephalopods. Hematoxylin and eosin (HE). (a) Common octopus (Octopus vulgaris). ELL/ RLO in gill (arrowhead), comprising (inset) aggregates of coccoid to coccobacillary bacteria (asterisk), distributed within a discrete eosinophilic cytoplasmic rim from the hypertrophic host cell (arrow). (b) ELL/RLO, dwarf cuttlefish (Sepia bandensis). Cyst-like structure in gill (arrowhead), comprising (inset) aggregates of indistinct bacteria (asterisk), distributed within a discrete eosinophilic cytoplasmic rim from the hypertrophic host cell (arrow). (c) Branchial ancistrocomid infection, California 2-spot octopus (Octopus bimaculoides). Moderate numbers of pyriform unicellular protozoa (arrowhead). (d) Higher magnification of 4c. Protozoa (arrowhead) with intensely hyperchromatic ovoid nuclei, basophilic stippled cytoplasm and suctorial tentacle with ciliary rows at the tapering pole attaching to the epithelial surface. (e) Common octopus (Octopus vulgaris). Branchial Ichthyobodo-like protozoal infection. Epithelial surfaces are populated by numerous pyriform bodonid protozoa (arrowheads), which are either free-floating or attached. (f) Higher magnification of 4e. Ichythobodo-like bodonid protozoa (arrow) palisade along the epithelial surface, with their flagella sometimes visible.
Coccidia
Coccidiosis primarily affected octopuses and represented the most common infectious disease in this order (170/266 infections; 63.9%) (Fig. 5a–d, f). It was found in 8 octopus species and 1 octopus of unknown species. Squid, cuttlefish, and nautiluses exhibited much lower prevalences at 3.0% (1/33), 6.2% (8/130), and 3.3% (3/90), respectively.
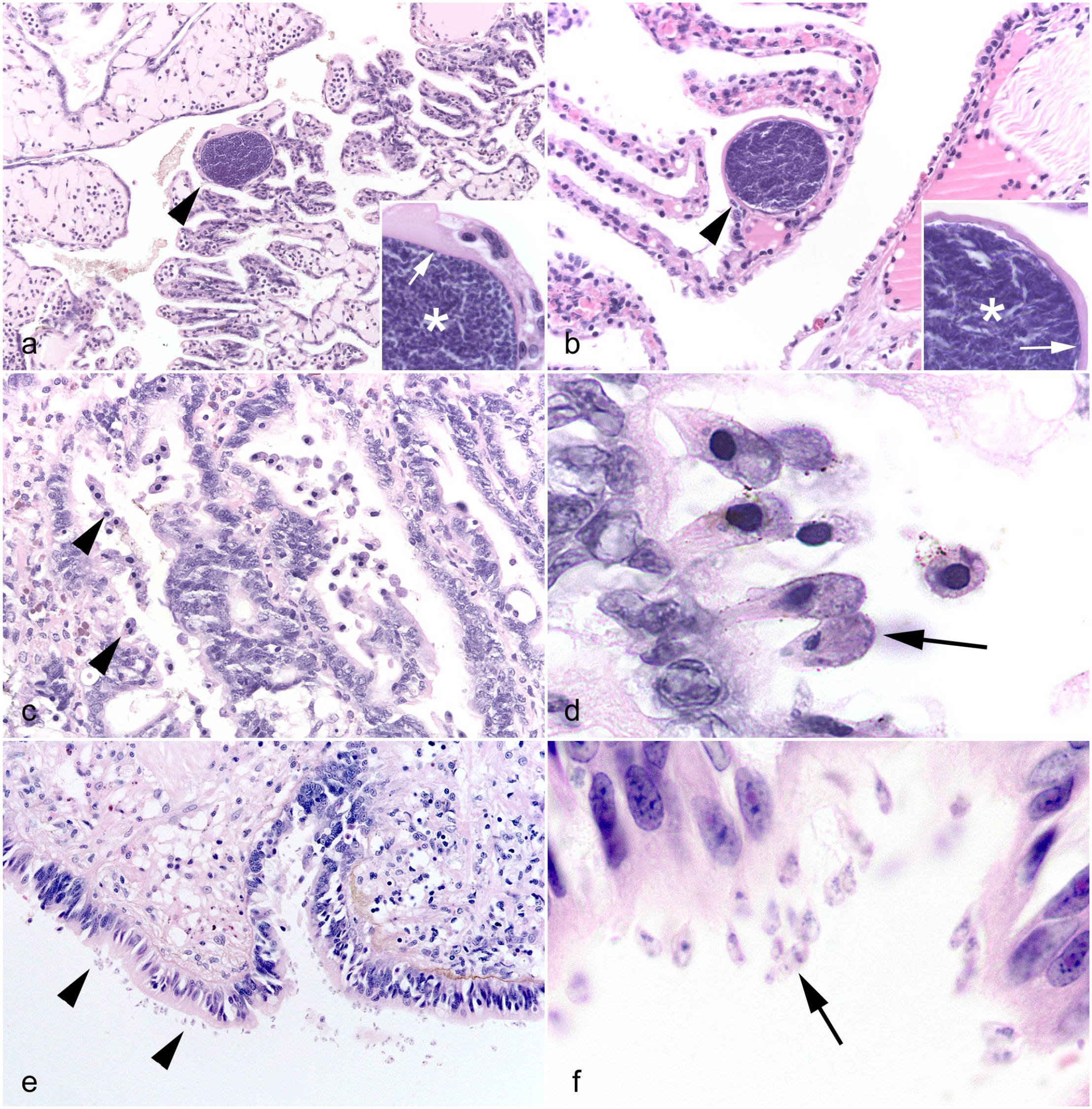
Histopathologic features of coccidial infections in cephalopods. Hematoxylin and eosin (HE). (a) Stomach, common octopus (Octopus vulgaris). Small numbers of discrete submucosal coccidial oocysts surrounded by thin fibrous walls (arrows) with numerous internal sporocysts (arrowheads), consistent with Aggregata octopiana. (b) Coccidial sporocysts, common octopus. Between 4 and 8 sporozoites (arrowheads) are seen within each sporocyst (arrows). (c) Intestine, California 2-spot octopus (Octopus bimaculoides). Marked submucosal infection with numerous coccidial oocysts (arrow) with internal sporocysts (arrowhead, many of which have artifactually dropped out of section). Inset (black box). Sporocysts released by a ruptured oocyst encounter infiltrating hemocytes. (d) Stomach, California 2-spot octopus. Coccidial oocyst (arrow) containing abundant sporocysts (arrowhead), with an adjacent macrogamete (asterisk), representing concurrent developmental stages. (e) Cecal folds, European common cuttlefish (Sepia officinalis). Numerous coccidial oocysts (arrows) within the cecal lamina propria, and a single zygote stage (arrowhead), consistent with Aggregate eberthi. (f) Gills, giant Pacific octopus (Enteroctopus dofleini). Free-floating (arrowheads) and intramucosal (arrow) coccidial gametocytes, consistent with Aggregata dobelli.
The highest prevalences of coccidiosis were found in the common octopus (133/193; 68.9%) (Fig. 5a, b), giant Pacific octopus (25/54; 46.3%) (Fig. 5f), and California 2-spot octopus (5/21; 23.8%) (Fig. 5c, d). Coccidial infection was responsible for the deaths of 32.3% (43/133), 36.0% (9/25), and 60.00% (3/5) of infected individuals from these respective species. Coccidial infection was significantly associated with the cause of death in California 2-spot octopuses compared with common and giant Pacific octopuses, Chi square test, p-valve = 0.0138 (<0.05). Other species affected included the lesser octopus (2/39; 5.1%) and single cases in a Caribbean reef octopus, coconut octopus, east Pacific red octopus, wunderpus octopus, and 1 octopus of unknown species, all of which were deemed incidental infections.
Across these octopus species, coccidial infections affected the following: intestine (139/170; 81.8%) (Fig. 5c, e), gill (41/170; 24.1%) (Fig. 5f), stomach (15/170; 8.8%) (Fig. 5a, b, d), esophagus (12/170; 7.1%), celomic connective tissues (12/170; 7.1%), dermal connective tissues (11/170; 6.5%), digestive gland (9/170; 5.3%), salivary gland (5/170; 2.9%), and systemic heart (2/170; 1.2%). Presumptive identifications of coccidial species were made based on reported host-parasite associations and consistent histomorphologies, but molecular speciation was not performed (ie, Aggregata octopiana in common octopus [Fig. 5b], Aggregata dobelli in giant Pacific octopus [Fig. 5f], and Aggregata polibraxiona in California 2-spot octopus [Fig. 5d]).15,31,69 Oocysts of these coccidial species were ovoid and typically between 600 and 800 µm in diameter. Sporocysts were typically 18 to 30 µm in diameter. Macrogamonts were ovoid and approximately 50 to 70 µm in diameter.15,61 Overall, coccidial infections affected 23 juvenile octopuses (13.5%), 33 octopuses of undetermined age (19.4%), and 114 mature octopuses (67.1%). The latter exhibited concurrent semelparity in 33 cases (33/114; 29.0%). Infected juvenile octopuses included 15 common octopuses, 5 giant Pacific octopuses, 2 California 2-spot octopuses, and a single coconut octopus. There was no difference in coccidial lesion distribution between age classes.
Branchial coccidial gametocytes were seen in a single hummingbird bobtail squid (without an attendant host tissue reaction). This infection was deemed incidental.
Coccidia were uncommon in cuttlefish (8/130; 6.2%) but still represented the second most common infectious agent. Species affected included 6 European common cuttlefish presumed to be infected with Aggregata eberthi (described in Sepia officinalis by Labbe, 1895) 33 and 2 dwarf cuttlefish. Six of 8 cases (all European common cuttlefish) were associated with enteric infection (Fig. 5e), while dwarf cuttlefish exhibited either gill or digestive gland infections. In 4/8 cases (all European common cuttlefish), coccidiosis was considered the cause of death.
Coccidia-like protozoal infections were suspected in 3 nautiluses (3/37; 8.1% of infections) in which the digestive gland (2/3) and intestines (2/3) were the only targeted organs. Moderate numbers of protozoal microorganisms were found within digestive gland tubular epithelial cells, appearing as intracytoplasmic, uniform, round basophilic cells, with distinct cell borders, smooth amphophilic granular-to-vacuolated cytoplasm, and single large round nuclei sometimes surrounded by a clear space. Further internal subdivision was present in a few of these cells, suggestive of coccidial schizonts. Although mild inflammation was seen, the infection was deemed incidental in the presence of more prominent lesions from which these protozoa were absent (eg, multiorgan inflammatory syndrome).
Dicyemid Mesozoans
Dicyemid mesozoans are endosymbiotic parasites of cephalopod renal appendages, which have simple, elongate bodies of 10 to 40 cells largely devoid of differentiated tissues. These were exclusively identified in octopuses, cuttlefish, and nautiluses. No notable epithelial histopathologic changes were associated with these nephridial colonizers in any individual. They were the second most common colonizing organisms in octopuses (149/340; 43.8%) (Fig. 6c, d) but were only seen in 2 co-housed mature male dwarf cuttlefish (2/130; 1.5%) and 2 mature nautiluses (2/90; 2.2%: 1 female and 1 male), from different aquaria.
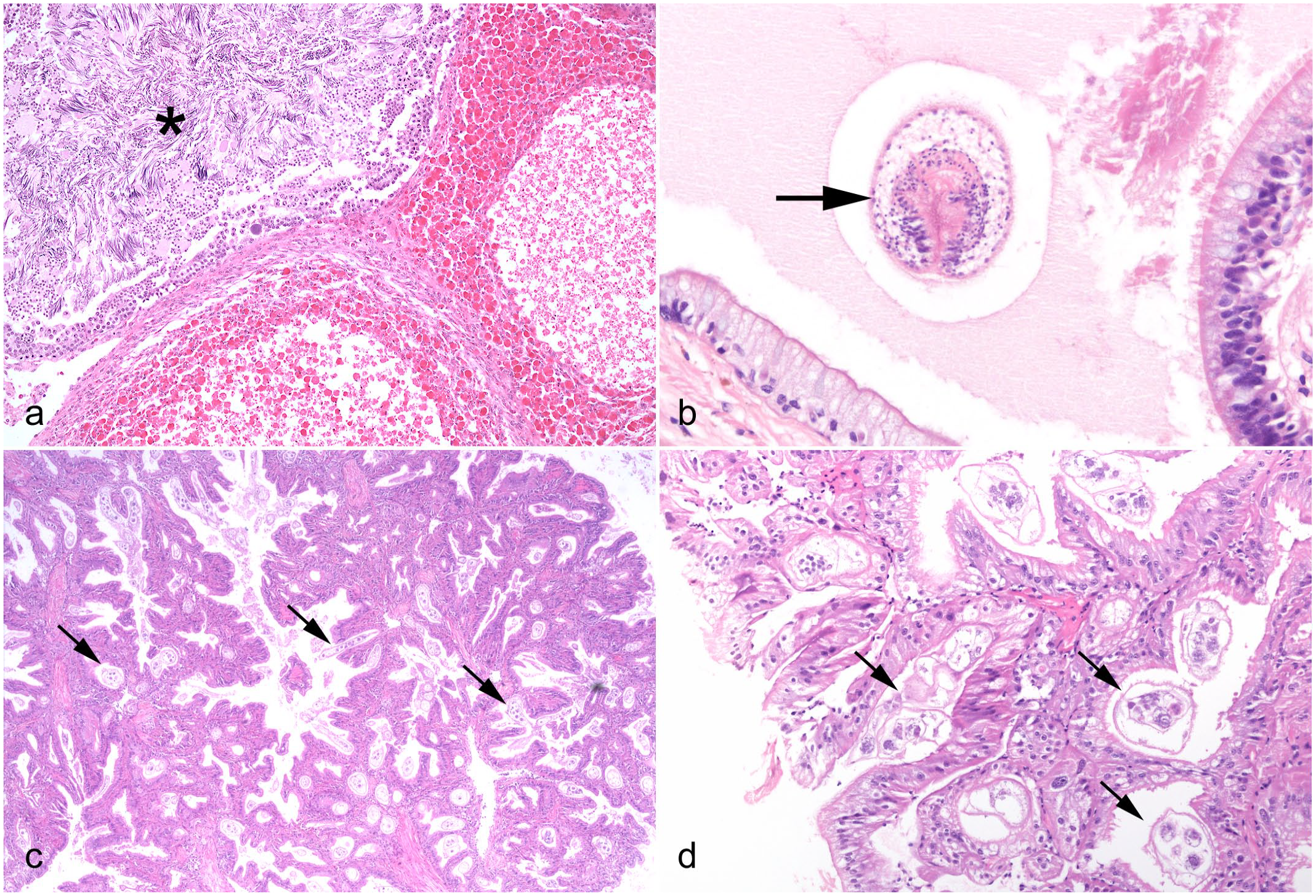
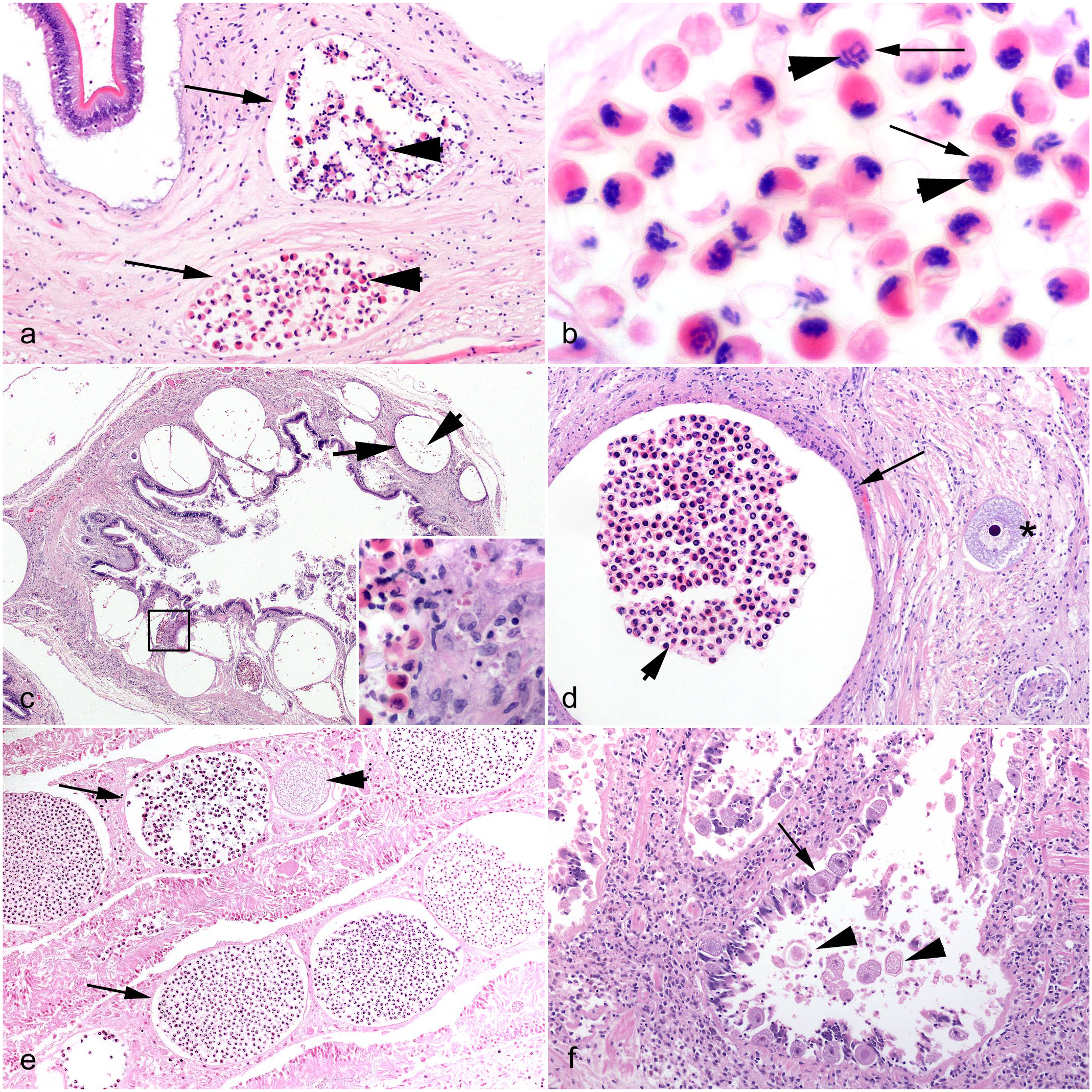
Idiopathic conditions and incidental parasitic infections in cephalopods. Hematoxylin and eosin (HE). (a) Chambered nautilus (Nautilus pompilius), testis, multiorgan inflammatory syndrome. Nodule-forming infiltrates of hemocytes with eosinophilic granules lie adjacent to a seminiferous tubule (asterisk). (b) Lesser octopus (Eledone cirrhosa), intestine. An intraluminal acoelomate helminth parasite, with features consistent with a larval cestode (arrow) including invaginated scolex. (c, d) Giant Pacific octopus (Enteroctopus dofleini), renal appendage, dicyemid mesozoans. (c) Numerous multicellular pale-staining dicyemids (arrows) embedded amongst the invaginations of epithelium. (d) Higher magnification of (c). The simple worm-like multicellular appearance of the dicyemids (arrows) varies with plane of section. Clusters of central axial cells are present within each organism.
Fungal Infections
Fungal infections were rare and only identified in octopuses (3/340, 0.9%) and nautiluses (2/90; 2.2%). In octopuses, 2 cases were associated with UD, while the third presented as testicular and branchial infections. All cases exhibited evidence of vascular invasion and systemic spread by virtue of fungemia. The fungal infections in both nautiluses comprised numerous 4 to 5 µm wide, parallel-walled, septate, nonpigmented fungal hyphae with dichotomous branching, targeting gills in 1 case and tentacle integument in the other. Vascular invasion was detected in both cases and was associated with multiorgan inflammation, suggesting terminal sepsis. Neither the octopus nor nautilus infections had fresh tissues available for definitive fungal identification by cultures. However, the fungal histomorphology in each case was considered consistent with a hyalohyphomycete, such as Fusarium spp., as described in previous literature reports (see later discussion).43,68,78
Noncoccidial Protozoa
Noncoccidial protozoa of various types were detected in all cephalopod orders, including ameba-like protozoa, ancistrocomids, Ichthyobodo-like bodonids, scuticociliates and trichodinids. Other unidentified ciliated protozoa were also seen. Diagnoses were made based on histomorphologic features as molecular speciation could not be performed in any case.
Ameba-like protozoal infections were uncommon across all orders but exhibited greatest prevalence in squid (4/33; 12.1%), followed by nautiluses (3/90; 3.3%), cuttlefish (2/130; 1.5%) and octopuses (2/340; 0.6%). Ameba-like infections in squid were associated with UD (3/4 cases) (Fig. 3e, f), branchitis (2/4 cases) or widespread dissemination affecting multiple (>3) organs (2/4 cases). In the 3 nautiluses, ameba-like protozoa were associated with either UD, ulcerative enteritis, or mixed-etiology branchitis (Gram-negative bacteria, ameba, and nematode). Dermal and enteric infections exhibited notable tissue reactions (ie, hemocytic inflammatory infiltrates and edema) without involvement of other microorganisms, and ameba-like protozoa were considered clinically relevant to the deaths of the 2 individuals with enteric and branchial infections. However, concurrent involvement of Gram-negative bacteria and nematodes may have contributed to the latter. Two affected cuttlefish demonstrated a mild and incidental digestive gland infection and substantial UD, respectively. Only 2 octopuses (1 each of common and lesser octopuses) exhibited amebic infections. Both were gill-associated, and 1 was considered the cause of death.
Substantial gill lesions associated with colonization by Ichthyobodo-like (bodonid) protozoa were identified in 26 octopuses, predominantly giant Pacific and California 2-spot octopuses (9/26; 34.6% each), but also including 4 common octopuses (Fig. 4e, f), 2 Atlantic white-spotted octopuses, and single cases in east Pacific red and wunderpus octopuses. These organisms were 5 × 7 µm, pyriform and flagellate, palisading along the surfaces of host epithelial cells. Tissue changes included epithelial hyperplasia with pseudostratification and papilla-like surface proliferation, accompanied by intra-epithelial and lamellar hemocytic infiltrates and edema. All infected California 2-spot and Atlantic white-spotted octopuses came from the same aquarium and died within a 22-month period with similar lesions of marked branchitis, perhaps suggesting a persistent high-rate of infectivity. 25 Only 2 cases of Ichthyobodo-like protozoal infection had concurrent evidence of postreproductive senescence. Only single cases of bodonid infection in cuttlefish and nautiluses were identified, causing UD with secondary bacterial infection in a mature female European common cuttlefish, and severe branchitis (deemed the cause of death) in a chambered nautilus (unknown age/sex). Bodonids were not found in any squid.
Ciliated protozoa were uncommon but identified in all 4 cephalopod orders, in which prevalences were: 8.9% in nautiluses (8/90), 6.1% in squid (2/33), 3.8% in cuttlefish (5/130), and 3.5% in octopuses (12/340). In most cases, these were histomorphologically described as elliptical ciliates (approximately 30 µm in length) with vacuolated basophilic cytoplasm, eosinophilic granular intracytoplasmic debris, and single hyperchromatic macronuclei; consistent with scuticociliates. However, molecular speciation could not be performed in any case. A single case of infection by trichodinid ciliates (recognized by attachment to ulcerated cutaneous tissues by their adhesive disks) was seen in nautiluses, although most ciliated protozoans in nautiluses were scuticociliates (5/8), invading the skin (4/5), gills (3/5), and eyes (1/5). Unidentified ciliates were seen in the skin or gill of 2 nautiluses. In cuttlefish, scuticociliates were mostly associated with dermal ulcers (4/5 cases), and marked inflammatory reactions were present in only 2 cases.
The only ciliated protozoal infections in octopuses were ancistrocomid protozoa (5/12) and scuticociliates (7/12). Ancistrocomids were characterized as unicellular elliptical protozoa (approximately 25 µm in length) with pale cytoplasm containing a single ovoid hyperchromatic eccentric nucleus and a perinuclear vacuole. Parasites were usually located in the space between gill filaments, or sometimes adherent to the epithelium, with a tapering pole orientated toward the epithelial surface. Such ancistrocomid protozoa were only identified in 5 octopuses including 3 common octopuses, a California 2-spot octopus (Fig. 4c, d), and a wunderpus octopus. In all cases, these protozoa were associated with branchial hyperplasia or hyperplastic branchitis and deemed clinically significant. They were apparently secondary to postreproductive senescence in most cases (4/5 ancistrocomid infections). Scuticociliates were associated with UD (see section Specific entities—Ulcerative dermatitis).
Seven infections involving protozoa that could not be definitively classified were detected in octopuses, affecting the gills (4/7; seen as protozoal cysts), digestive gland (2/7), alimentary tract (2/7), skin and salivary gland (1/7 each). All cases were mild infections and considered incidental. Distinctly different, unidentified ciliated protozoa were found affecting either the gills or deferens/spermatophoric glands/Needham’s sac in 2 squids with nonamebic protozoal infections.
Metazoan Parasites
A range of metazoan parasites were identified across octopuses (34/340; 10%), cuttlefish (4/130; 3.1%), and nautiluses (2/90; 2.2%), including arthropods (3/593; 0.5%), cestodes (21/593; 3.5%), nematodes (9/593; 1.7%), and trematodes (7/593; 1.2%). No squid demonstrated metazoan parasitism.
Metazoan parasitic infections in octopuses were represented by larval cestodes (19/340; 5.6%), nematodes (8/340; 2.4%), trematodes (6/340; 1.8%), and arthropod-like parasites (1/340; 0.3). Incidental acelomate helminths with features of larval cestodes (invaginated scolex with suckers) were occasionally found in octopuses (Fig. 6b) without associated tissue inflammation. Organs involved included the intestines (10/19), body wall connective tissues (7/19), gill interstitium (2/19), and single examples in the digestive gland, pancreas, and salivary gland. Nematodes were found in intravascular spaces (3/8; gills in 2 and salivary gland in 1) but also affected the intestines (2/8), integument (2/8), and oviduct (1/8). All parasites were accompanied by either mild or negligible hemocytic reactions and were considered incidental. Incidental trematodiasis affected the body wall/cutaneous connective tissues with metacercaria (4/6) or the intestines with adult parasites (2/6), causing negligible tissue reaction. A single case of an unidentified invasive arthropod-like organism was identified in association with UD; whether this was a primary lesion or secondary to trauma is uncertain.
Arthropod-like parasites were infrequent in cuttlefish (2/130; 1.5%) causing mild gill infections in both cases. Single rare examples of trematodes (periocular connective tissue) and larval cestodes (spermatophore duct) were deemed incidental.
Sporadic single cases of cestodiasis and nematodiasis were identified in nautiluses. Intestinal cestodiasis was seen in a mature male nautilus, characterized by an encysted larval cestode with characteristic hooked mouthparts. This was located within the intestinal wall surrounded by a capsule of hemocytes and compressed connective tissue. A single (most likely incidental) nematode was identified in a juvenile female nautilus associated with mixed-etiology gill lesions (Gram-negative bacteria, amebae, and nematodes).
Inclusion Bodies
Intracytoplasmic inclusion bodies (ICIBs) and intranuclear inclusion bodies (INIBs), were detected in 3 octopuses (2 California 2-spot octopuses and 1 lesser octopus) in association with branchitis and vasculitis. Characteristic lesions in both California 2-spot octopuses were anisocytosis and anisokaryosis in epithelial and mesenchymal cells, cell necrosis, vasculitis, and numerous INIBs and ICIBs. Inclusions were also evident in the lesser octopus with accompanying gill inflammation and necrosis, but vascular changes were not pronounced. Inclusions were not detected in any squid, cuttlefish, or nautilus in this study.
Inflammation (Sterile or Unconfirmed Etiology)
Inflammatory diseases deemed sterile or of unconfirmed/unobservable etiology (following the application of special stains) were seen in 45.6% (41/90) of nautiluses, 23.8% (31/130) of cuttlefish, 21.2% (7/33) of squid, and 5.9% (20/340) of octopuses. A variety of different organs were affected, although UD was more frequently involved overall (see section Specific entities—Ulcerative dermatitis). Nautiluses tended to exhibit multiorgan involvement (see section Specific entities—Nautiloid multiorgan inflammatory syndrome). Most sterile inflammatory cases in nautiluses were the definitive cause of death (28/41; 68.3%), including 12 with UD and/or shell damage (of which 10/12 had multiorgan inflammation). In cuttlefish, 16/31 cases (51.6%) were associated with a definitive cause of death, of which 9/16 (56.3%) displayed UD with multiorgan inflammation. Eight cases had undefined causes of death. Seven exhibited other processes including isolated dermal ulceration (3/7), environmental/life-support failure (2/7), suspect sepsis (1/7), or postreproductive senescence (1/7). Two co-housed bigfin reef squid exhibited branchitis associated with (aquarist-suspected) aluminum residue toxicosis. Octopuses did not show any notable trends in sterile inflammatory lesions.
Specific Conditions/Entities
Ulcerative Dermatitis (Mantle Disease) and Nautiloid Shell Damage (“Black Shell Disease”)
A specific condition of heightened frequency across all cephalopods was ulcerative dermatitis (UD) (Fig. 3a–h) affecting the mantle and/or tentacles of coleoids (colloquially termed “mantle disease”), or the labial tentacles, hood and/or hyponome of nautiloids. Such external lesions were seen in 14/33 (42.4%) squid, 30/90 (33.3%) nautiluses, 30/130 (23.1%) cuttlefish, and 51/340 (15%) octopuses. Intralesional microorganisms were detected in 9/14 (71.4%) squid, 21/30 (70%) cuttlefish, 36/51 (70.6%) octopuses, and 11/30 (36.7%) nautiluses.
Infectious microorganisms in squid UD-lesions (9/14) comprised either bacteria (various species, as described above; 6/14; 42.9%) or amebae alone (3/14; 21.4%) (Fig. 3e, f). There was a notable difference in the prevalence of UD between laboratory squid and aquaria squid, with frequencies of 12/23 (52.2%) for laboratory squid (5 bacterial, 3 amebic, 4 sterile) and 2/10 (20.0%) for aquaria squid (1 bacterial, 1 sterile). Only a single squid with UD was senescent (1/14; 7.1%).
Cuttlefish UD-lesions usually contained visible and/or culturable bacteria (21/30; 70%) (Fig. 3g, h) and concurrent infections included scuticociliates (4/21) and single examples of bodonid (Fig. 3h) and ameba-like protozoa. These were presumed to contribute to epidermal damage. As for squid, there was a notable difference in the prevalence of UD between laboratory and aquaria cuttlefish, with frequencies of 20/39 (51.3%) in laboratory cuttlefish (12 bacterial, 8 sterile), and 10/91 (11.0%) in aquaria cuttlefish (9 bacterial including 4 cases involving scuticociliates and single cases of concurrent bodonid and ameba-like protozoal infections, and 1 sterile). Only 3 cuttlefish exhibiting UD were senescent (3/30; 10.0%).
Infected octopus UD-lesions (36/51) demonstrated a broad range of intralesional microorganisms; namely, Gram-negative bacterial rods (15/36; 41.7%) (Fig. 3a–d), scuticociliates (7/36; 19.4%) Ichythobodo-like protozoa (5/36; 13.9%), coccidia (4/36; 11.1%), fungi (2/36; 5.6%) and single cases of a nonspeciated protozoan, a nematode-like metazoan, and copepod (arthropod) infections. Co-infections were seen.
Nautiluses exhibited a unique presentation of shell damage, characterized by the development of blackened fissures called “black lines” (referred to as “black shell disease,” BSD) (Fig. 1e–f). BSD constituted 60% (18/30) of the external lesions in nautiluses on gross inspection of submitted shells. Multiorgan inflammation (Fig. 6a) was concurrently identified in 22/30 (73.3%) nautiluses with external lesions, including 11 with visible infections and 11 cases with no visible bacteria/organisms. Overall, rod-shaped bacteria were the most prevalent infection (7/11; 63.6%), followed by ciliated protozoans (4/11; 36.4%) including 3 scuticociliates and 1 trichodinid, plus single cases of fungal and ameba-like protozoal infection.
Semelparity (Coleoid Cephalopods)
Features of postreproductive senescence, (ie, noninfected ovarian/testicular degeneration and/or features of digestive gland, salivary gland, and oviductal gland atrophy in postpubescent/mature individuals) were detected in octopuses (76/340; 22.4%) (Fig. 2a–i), cuttlefish (15/130; 11.5%), and squid (2/33; 6.1%). In most of these cases, the predominant findings were the reproductive lesions associated with semelparity (octopuses (70/340; 20.6%), squid (2/33; 6.1%) and cuttlefish (5/130; 3.8%).
Specific senescent reproductive tract changes were identified in 70/76 (92.1%) octopuses identified as postreproductive. The remaining cases were either recorded as having laid eggs/mated or sample sets lacked reproductive tissue but exhibited other consistent degenerate changes. Concurrent disease processes included inflammatory/infectious diseases (63/76; 82.9%), coccidiosis (33/76; 43.4%), UD (16/76; 21.1%), inanition (ie, digestive gland/salivary gland atrophy; 31/76; 40.8%), organ degeneration (5/76; 6.6%) and mineral deposition (2/76; 2.6%). Digestive and salivary glandular atrophy (Fig. 2d–i) were characterized by diffuse epithelial atrophy with increased tubular lumen-to-epithelium ratios, with attenuated epithelial cells exhibiting reduced intracytoplasmic secretory granularity and increased yellow/brown intracytoplasmic granular debris, interspersed by conspicuous interstitial connective tissue.
Two cases of postreproductive senescence (2/33; 6.1%) were identified in aquaria squid (1 male bigfin reef squid and 1 female striped pajama squid), with evidence of reproductive lesions. Each had concurrent inflammatory diseases, either colonizing bacterial and flagellate protozoal infections of the spermatophoric duct and accessory ducts/glands (male) or idiopathic UD (female).
Senescence was recorded in 11 aquarium cuttlefish and 4 laboratory specimens (15/130; 11.5%). The 4 senescent laboratory cuttlefish exhibited bacterial UD (3/4), bacterial pancreatitis (2/4), nidamental gland adenitis (2/4), and multiorgan inflammation (no etiology identified; 2/4). Aquarium cuttlefish were more variable in exhibiting concurrent lesions, with infections detected in 4 cases, sterile inflammation in 3 cases and inanition in 2 cases (digestive gland atrophy).
Nautiloid Multiorgan Inflammatory Syndrome
Multiorgan inflammation was a notable feature affecting 51/90 (56.7%) nautiluses. This was characterized by disseminated perivascular to diffuse interstitial hemocytic infiltrates affecting 3 or more organs (Fig. 6a), in the absence (29/51; 56.9%) or presence (22/51; 43.1%) of an intralesional infectious agent. Infectious microorganisms included bacteria (14/22; 63.6%), ciliated protozoa (ie, scuticociliates and trichodinids; 6/22; 27.3%), coccidia (2/22), fungi (2/22), ameba-like protozoa (1/22), and bodonid protozoa (1/22). Nonlesion-associated dicyemid mesozoans were present concurrent to renal inflammation in 1 of 2 nautiluses colonized by these organisms.
Noninfectious/Miscellaneous Diseases
Noninfectious/miscellaneous disease processes were less common across all cephalopod orders in contrast to infectious or aseptic inflammatory diseases (Table 2).
Environmental/Life-Support Failure
Known environmental/life-support failure was rare but suspected in 6/130 (4.6%) cuttlefish. Four of these dwarf cuttlefish originated from the same aquarium where large temperature declines of 24.7ºC to 18ºC occurred. No histological lesions were detected. Of the 2 other cuttlefish, 1 exhibited interstitial gill edema and branchitis suggestive of nonetiologically specific gill irritation (ie, the result of an aquarist-recognized, but undefined, water quality issue). A mature female common octopus (1/340; 0.3%) died after escaping its aquarium.
Inanition (Digestive Gland and Salivary Gland Atrophy)
Inanition-related atrophic changes were diagnosed in squid (4/33; 12.1%), octopuses (5/340; 1.5%), and cuttlefish (2/130; 1.5%) without postreproductive senescence, and nautiluses (3/90; 3.3%). In squid, 3 of the 4 cases exhibited concurrent UD. The fourth case exhibited ciliated protozoal-associated branchitis. All were of laboratory origin. In octopuses, nonsemelparity-associated inanition was seen with gill disease in 2 mature males (1 with ichythobodiasis), but no common underlying disease processes were detected in the remaining individuals. One aquarium cuttlefish exhibited concurrent lesions (UD, bacterial pancreatitis, and multiorgan inflammation) in contrast to a laboratory specimen with no underlying lesions. Inanition in nautiluses (3/90; 3.3%) was always detected alongside multiorgan inflammation.
Organ Degeneration/Atrophy (Noninanition, Nonsemelparous)
Organ degeneration/atrophy was rare in octopuses (3/340; 0.9%) and included bilateral ocular cataracts in a female senescent common octopus (Fig. 1d), and mantle glandular ectasia or diffuse branchial atrophy in 2 mature (nonsenescent) female lesser octopuses. Such lesions were similarly uncommon in cuttlefish (6/130; 4.6%), comprising ocular cataracts in 2 broadclub cuttlefish, and digestive gland degeneration and renal changes of suspected toxic origin in 2 cases each (see below). A single squid exhibited hepatocellular vacuolar degeneration of unknown relevance or etiology.
Organ-Associated Substance Deposition
Intratubular basophilic granular mineral deposits in the renal appendages were identified in 5 (5/90; 5.6%) nautiluses of variable sexes and ages. Mineral deposition of undefined relevance and etiology in 3 octopuses (3/340; 0.9%) affected salivary glands, gills, or kidneys in each case.
Reproductive Disease
Nonsemelparity-associated reproductive lesions (in females and males) were rarely identified in squid (1/33; 3.0%), cuttlefish (3/130; 2.3%), and octopuses (5/340; 1.5%). These typically involved reproductive organs as part of a systemic disease process.
In iteroparous nautiluses, reproductive lesions were infrequent (15/90; 16.7%) and mostly affected males (12/15), typically associated with multiorgan inflammation syndrome involving the testes (10/23 males, 43.5%).
Suspect Toxicosis or Chemical Exposure
Suspected toxicosis or chemical exposure was limited to 3 squid (3/33; 9.1%), 2 cuttlefish (2/130; 1.5%), and 2 nautiluses (2/90; 2.2%). Two co-housed bigfin reef squid exhibited branchitis associated with (aquarist-suspected) aluminum residue toxicosis. A third case of suspected aluminum residue toxicosis was characterized by digestive gland epithelial vacuolar degeneration, but also exhibited features of senescence and sterile UD, which were considered of greater clinical significance.
Two nautiluses from different aquaria exhibited acute diffuse interstitial branchial edema and hemocytic branchitis. One collection was able to provide water quality readings, which revealed high nitrates (100 mg/l; maximum acceptable limit: 50 mg/l) and nitrites (4 mg/l; maximum acceptable limit: 0.1 mg/l), strongly suggestive of toxicity. 55
Two mature male flamboyant cuttlefish (from the same collection as 4 dwarf cuttlefish subjected to marked temperature fluctuations) exhibited renal epithelial cell anisocytosis and anisokaryosis, with additional features of lipid-like vacuolation, autophagocytic vacuoles, and renal epithelial injury. Suspected changes in salinity or exposure to chemical agents were proposed in these 2 cases. No microorganisms were detected.
Discussion
This retrospective review represents the first substantial comparative analysis of pathologic lesions in a large cohort of captive cephalopods of both extant classes: Coleoidea and Nautiloidea. The intention of this discussion is to highlight relevant disease processes that diagnostic pathologists may encounter in such species in aquaria, zoological collections, and laboratories.
Note that for many of the categories analyzed across the data set (except for “no diagnosis”) it was possible for a single animal to have more than 1 concurrent pathologic process. However, any individual animal can only be recorded once in each category describing a pathological entity or feature, and so the category prevalence figure reflects that entity in the whole population. For example, in Table 2, 76/340 octopuses demonstrated semelparity/postreproductive senescence, but this population overlapped with 266/340 octopuses that had infections confirmed by histology. In some cases, the distribution of overlapping entities (eg, confirmed infections within the postreproductive population) was further analyzed and described in the Results section.
Fundamentally, postreproductive senescence is not a disease, but rather a lifecycle stage during which physiological, immunological, and behavioral changes occur. 73 In this study, features of senescence were most common in octopuses in comparison with cuttlefish and squid. It is likely that this is related to the preponderance of decapodiforms (ie, squid and cuttlefish) amongst the laboratory specimens. The latter may not reach their potential full life-expectancies before experimental endpoints are reached. In contrast, aquarium octopuses should be able to live to the end of their natural lives. The over-representation of postsenescent changes in octopuses is therefore understandable but overshadows the issue that 77.6% of aquarium octopuses may have failed to reach their full natural longevity. This may imply occult underlying health and behavioral issues in captive populations.
The most common concurrent disease processes identified in postreproductive/senescent octopuses were general inflammatory/infectious diseases (ie, coccidiosis and UD) and inanition (ie, digestive gland/salivary gland atrophy). Current theories of senescence suggest that the associated changes (eg, immunosuppression, anorexia and potential disorientation leading to traumatic injuries) may increase the likelihood of infections, traumatic skin injuries, and starvation/inanition. Consequently, coccidiosis and UD have been considered early signs of senescence in semelparous cephalopods. 8
It is important to stress that inanition-associated atrophic changes were not limited to senescent cephalopods. 5 octopuses, 4 laboratory squid, 2 aquarium cuttlefish and 3 nautiluses exhibited such changes because of concurrent disease, with UD, gill disease and multiorgan inflammation (nautilus) being most common. Underlying physiological, environmental, or cognitive stressors 63 may have been involved in such cases.
Senescence is accompanied by physiological, immunological, and behavioral changes, which may include anorexia, retraction of periocular skin, corneal edema, mental debilitation/incoordination and eroded and ulcerated skin lesions (likely due to self-mutilation from a lack of co-ordination).3,13,76 Autophagy may also play a role. Consequently, UD is considered common in senescent cephalopods with lesions mostly located over the mantle and tentacles. However, the overall prevalence of UD in senescent coleoid cephalopods was only 21.1% in this study. Therefore, while UD is not uncommon in senescent cephalopods, senescence is not the sole contributing risk factor. This is consistent with the difference between the prevalence of UD in laboratory versus aquaria-reared squid and cuttlefish. UD was more than twice as prevalent in laboratory squid and cuttlefish compared with aquarium specimens. Various infectious microorganisms were identified within these lesions, suggesting that water microbial levels may affect not only the presence of bacteria but also bodonid protozoa, scuticociliates, and ameba-like protozoa. The exact conditions in which laboratory specimens were kept were not disclosed but enclosure size and increased population densities may constitute risk factors (eg, increased organic loading would be a factor in the density and diversity of the water microbiota). In previous studies of octopuses in aquaculture, interanimal contact was the major cause of primary skin damage. UD was rarely observed in individually cultured specimens. 26 Conspecific contact using suckers can induce ulceration, removing the protective epidermal layer and allowing secondary infections, 26 most commonly on the dorsal mantle (“ulcerative mantle disease”). 82 Tentacle lesions have additionally been attributed to injury sustained during the acquisition of live prey food items, eg, crabs and prawns, 26 use of which is limited in many aquaria. Other aquatic or environmental contact/abrasions or self-mutilation may be more likely in laboratory settings. 83 Laboratory octopuses have been recorded to develop single, large, typically idiopathic, chronic lesions on the distal tip of the mantle. It is hypothesized that stress may cause more erratic swimming behavior, resulting in tank wall impact-related mantle injury. 26
Bacteria were found within 29.4%, 42.9%, and 70.0% of UD in octopuses, squid, and cuttlefish, respectively. Using special stains, Gram-negative rods predominated. Various bacteria have been implicated in dermal ulcers in cephalopods and teleosts including Vibrio spp.,9,24,77 (such as V. alginolyticus,42,74 V. damsela, 42 and V. parahaemolyticus) 42 as well as Pseudomonas stutzeri,24,42,82 Aeromonas caviae,24,42 and Bacillus species. 24 In the cephalopods in this study, only squid and cuttlefish had consistent submissions of transport media swabs for microbiological cultures. In this cohort, relevant bacteria (ie, marine-based) isolated from outer and inner mantles (and occasionally gills) included Shewanella putrefaciens, Sphingomonas paucimobilis, and Stenotrophomonas maltophilia in squid and Alcaligenes species, S. paucimobilis, and V. alginolyticus in cuttlefish. As for any aquatic species, microbiological identifications should be treated with caution, particularly when organisms have only been reported once or are not consistent with the type of aquatic environment of the host (ie, freshwater vs brackish vs marine). For example, Aeromonas hydrophila is primarily a freshwater to brackish organism and would not be expected in sea water. The pathologist should be aware of the cephalopod’s preferred environment as well as the limitations of certain biochemical profiling systems that do not reliably distinguish aeromonads and vibrios. The latter are more likely to be associated with marine environments. Most of the bacteria isolated in this study have been associated with active pathogenic infections in aquatic species.1,2,14,19,37,49,50,56,57,60,67,88 However, bacteria discounted as probable freshwater contaminants in this study population included Acinetobacter spp., Aeromonas spp., Pasteurella spp., Pseudomonas spp., and Rahnella aquatilis. Probable spoilage-associated environmental contaminants include Alcaligenes faecalis, Bergeyella zoohelcum/Weeksella virosa, Brevundimonas diminuta/vesicularis, and Shewanella algae. 37 Nonetheless, this study identifies an additional selection of bacterial species, not previously reported in cephalopods, with a potential pathogenic role.
Multiple studies suggest that the immunocompetence of senescent octopuses is reduced or impaired in contrast with juvenile and adult octopuses, proposing postreproductive senescence as a major risk factor for coccidial infection in octopus aquaculture.65,73 Varying inflammatory responses occur in senescent octopuses, ranging from no recognizable responses to mild or moderate infiltrates;65,73 a trend also recognized in this study. Diagnostic certainty diminishes with the consequent lack of inflammatory infiltrates or associated tissue injury in certain diseases. This raises the question of whether substantial coccidial burdens in senescent octopuses are a primary problem or simply incidental in postreproductive animals in which other senescent features should be given greater weight? The observation of coccidiosis in only 43.4% of senescent octopuses is inconclusive either way.
Octopuses exhibited a particular propensity for coccidiosis in this study. Various Aggregata species infect octopus,29,61,65,73,76 cuttlefish,16,33 and squid species, 34 and exhibit complex life cycles involving alternation of hosts between coleoideal cephalopod and crustacean intermediate hosts (eg, crab), with stages of the parasite developing in the intestine of each species. In the definitive cephalopod host, sporocysts develop in the intestinal mucosa resulting in mucosal damage and sloughing of parasitized debris, releasing new infective stages into the feces. Ingestion and schizogony within the intermediate host generates merozoites infective to the cephalopod when it consumes its crustacean prey. 33 As a result, octopuses are unlikely to transmit infection directly to other cephalopods within exhibits.
Coccidial infection was most prevalent in common octopuses, giant Pacific octopuses, and California 2-spot octopuses. The intestines and gills were the main targeted organs across all species, consistent with the current literature,11,29,61,76 in which lesions have been reported in the crop, cuticularized esophagus, intestines, cecum, and digestive gland, plus extraintestinal sites such as the gills, skin, and gonads. Malabsorption syndrome has been associated with intestinal coccidial infections (Aggregata octopiana) in common octopuses, 31 and may underlie the pathogenesis of inanition in coccidia-infected senescent octopuses. Coccidiosis is a substantial problem in young wild-caught octopuses cultured in west Mediterranean seafarms, 59 which subsequently experience reduced growth rates and longevity in captive aquaculture. 32 The same may apply to juvenile octopuses in captivity acquired from wild-caught sources. 65
While common octopuses and giant Pacific octopuses were more likely to be infected, coccidiosis resulted in significantly fewer fatalities in comparison with California 2-spot octopuses. The latter demonstrated lower prevalence but more severe pathology, marked ulcerative and/or necrotizing lesions and inflammatory infiltrates in a greater number of organs (>3). California 2-spot octopuses may be particularly sensitive, may lack innate immunity to coccidial infections, or there may be differences in the pathogenicity of the coccidial species affecting each host.
Coccidiosis was rare in captive decapodiforms (squid and cuttlefish) and nautiluses. This contrasts with the greater reported frequency in captive wild-caught and commercially bred cuttlefish, in which most cases in European common cuttlefish involve Aggregata eberthi (as described in Sepia officinalis by Labbe, 1895). 33 Intestinal coccidia have been recorded in European flying squid (Todarodes sagittatus) and the 7-star flying squid (Martialia hyadesii), which were not represented in this study. Coccidial infections in nautiluses have not been described to date.11,16
Epitheliocystis-like lesions (ELLs) have been infrequently described in octopuses, in which they have been considered to be Rickettsia-like organisms (RLOs) based on histomorphology but have not been characterized at a molecular level.21,27 Similar infections were found in 3.2% of study octopuses and were consistently located in the gill epithelial cells. Comparatively, histomorphologically similar Rickettsia or Chlamydia-like organisms in mollusks have been identified as gammaproteobacterial-Endozoicomonadaceae.10,27,39 Such organisms are not necessarily correlated with disease;21,23 however, heavy infections may contribute to pathology,21,27 and a generalized tissue distribution was identified in a single coconut octopus in this study.
UD lesions were also colonized by nonbacterial organisms. Bodonid protozoa (Ichthyobodo species) are kinetoplastid flagellates, clinically important as ectoparasites of freshwater and marine teleosts in wild and cultured environments.25,70 These parasites can be found both free-swimming and attached to epithelial surfaces such as gills and skin. Ichthyobodo-like organisms are also recognized to be pathogenic in octopuses,70,76 with several reports in cultured 25 and aquarium 76 species. Molecular studies in such cases have suggested that the octopus-associated organisms are closely related to Ichthyobodo species from freshwater teleosts. This study identified 26 cases of such infections in octopuses, and further single cases in an European common cuttlefish and a nautilus.
Previous reports of Ichthyobodo species infection in octopuses have suggested that senescence may be a major contributory factor by reducing immunity to the parasite. 76 In this study, only 2 cases exhibited concurrent senescence (wunderpus octopus and giant Pacific octopus). However, 5 histologically confirmed juvenile octopuses were identified amongst the 26 cases, indicating that Ichthyobodo should not be considered a parasite restricted to geriatric octopuses, and environmental stressors may contribute to outbreaks in younger animals. Nine of the cases of Ichthyobodo-like protozoal infection were associated with UD as previously documented in cultured California 2-spot octopuses, 25 with concurrent bacterial infections in all cases. All infected California 2-spot and Atlantic white-spotted octopuses came from the same aquarium over a period of 22 months suggesting a persistent reservoir for reinfection similar to aquarium scenarios involving teleost fish. A syndrome is recognized in California 2-spot octopuses in which the gills and epithelial surfaces of dying animals are colonized by bodonid protozoa, which may exhibit concurrent infection of the gills by ancistrocomid protozoa. 25 In this study, branchial co-infection of Ichthyobodo-like protozoa and ancistrocomids was only found in a single senescent female wunderpus octopus.
Ancistrocomidae are ectoparasitic protozoans associated with disease in cephalopods. 80 In European studies, they are mostly seen as gill infections of common octopuses, with occasional skin involvement. There are no reports in decapodiforms or nautiluses. 80 Five cases of ancistrocomid protozoa were identified in this study, in the common octopus, California 2-spot octopus and wunderpus octopus, and were associated with clinically significant branchial hyperplasia or hyperplastic branchitis; 4 accompanied postreproductive senescence. This is more consistent with a syndrome of Ancistrocomidae infection in geriatric/dying California 2-spot octopuses, in contrast with primary ichthyobodiasis. 25 No cases were detected in decapodiforms or nautiluses, consistent with the current literature. 80 In contrast, the main ciliate infection of European common cuttlefish and common squid (Loligo vulgaris) is Chromidina species, which instead targets internal tissues such as the digestive tract and renal appendages. 79 No Chromidina species infections were seen in this cohort.
Ameba-like protozoa are another major group of pathogens in squid from this study, which have not been previously documented in a similar context. The cases exhibited variable pathogenicity, including integumental and systemic/celomic invasion with minimal to marked inflammatory reactions, but the ameba-like protozoa were not further speciated. Amebae were previously reported in 3 giant Pacific octopuses, 2 of which were presumed pathogenic. The exact species involved were not molecularly identified. 76
Scuticociliate-like protozoa were occasionally detected in octopuses, nautiluses, squid, and cuttlefish. Recently, molecular speciation has been performed on similar cephalopod infections, with Miamiensis avidus (syn Philasterides dicentrarchi) (Ciliophora: Scuticociliatida) identified as a causative agent of skin ulcers in reared pharaoh cuttlefish (Sepia pharaonis). 84 The identification of Miamiensis avidus in cuttlefish is of particular importance to aquarium collections housing cephalopods and fish in the same closed systems. Miamiensis avidus is a major pathogen of marine fish,47,54,81 which likely represents a source of infection for cephalopods, and vice versa. Of this cohort, skin ulceration was a common feature in octopuses, cuttlefish, and nautiluses. Ciliates were morphologically consistent with scuticociliates such as Miamiensis-like species (elliptical ciliates; approximately 30 µm in length with vacuolated basophilic cytoplasm, eosinophilic granular intracytoplasmic debris, and single hyperchromatic macronuclei); however, genetic identification would be ultimately required for further speciation. Gills were the other main target organ in octopuses, nautiluses, and a single squid.
Literature on nautiluses and their diseases is limited.5,38,75 Mucodegeneration (a systemic ocular and tentacular bacterial infection causing pronounced mucus production) has been considered the most common disease syndrome in captive nautiluses but was not identified in clinical histories or detected on histological examination in this study. Lack of clinical awareness or recognition may be a consideration. For further information, see the references.4,5
Black shell disease was a key presentation in this cohort of captive nautiluses. Literature reports are limited but describe irregular shell formation and black lines, developing on the outside of adult nautilus shells at aquaria and zoos shortly after acquisition.48,62 This has also been observed in wild animals with broken shells, but such animals appeared to self-resolve by continued growth. 62 In contrast, aquarium specimens rarely recover. The black material has been characterized as a mixture of excess copper, zinc, and bromine, apparently unrelated to the nautilus’ diet and presumably derived from the environment. 48 Further study of the chemical composition and growth characteristics of nautilus shells in aquaria and natural environments is required. This may determine whether key environmental deficiencies or chemical imbalances impair aquarium specimens’ ability to grow and repair their shells to survive long term. 62
Nautiloid multiorgan inflammatory syndrome was the most notable disease trend in the nautiluses, affecting 56.7% of animals. In these cases, infectious microorganisms were identified histologically in less than half of the cases (41.2%). These cases included bacteria (66.7%), ciliates (scuticociliates or trichodinids; 28.6%), coccidia (9.5%), fungi (9.5%) and single examples of ameba-like protozoa, bodonid protozoa, and incidental dicyemid mesozoans. It is unclear whether this is a genuine multisystemic inflammatory response or a widespread reactive hematopoietic response. In either case, it may suggest that these animals are mounting an aggressive inflammatory reaction to a generalized process such as septicemia. However, no infectious microorganisms were observed in 30 cases in which multiorgan inflammation was diagnosed (58.8%), despite use of special stains. While systemic iridoviral infection has been reported in a single nautilus, 38 none of the study cases exhibited cellular hypertrophy or inclusions to raise suspicion of viral involvement.
Multiorgan inflammatory syndrome was identified in 73.3% of the 33% of nautiluses with external lesions (ie, UD and/or shell damage). These findings may imply that the presence of external lesions increases the likelihood of the development and concurrent detection of multiorgan inflammatory syndrome. This highlights the importance of thorough clinical examination and recording of shell damage and skin lesions by aquarists prior to submission for histological examination or submission of the whole animal (including shell) for postmortem examination.
Dicyemid mesozoans were frequently identified in octopuses. These are commensal/symbiotic, host-adapted, tiny marine parasites that only occur in the kidneys of cephalopods and are phylogenetically related to flatworms. 28 Large numbers may be found in an individual adult host. Cephalopods have a complex excretory system including branchial hearts (not present in Nautiloidea), pericardial/branchial heart appendages (Nautiloidea/Coleoidea, respectively), pancreatic (or digestive duct) appendages and finally renal appendages generating urinal fluid that collects in the renal sac. This provides an optimal habitat for various endosymbionts.28,44 Dicyemid mesozoans are not considered pathogenic and may assist nitrogen/ammonia metabolism by contributing to urine acidification. 28 It is suggested that colonization approaches 100% in wild cephalopod populations. 28 In this study, captive octopuses retained a moderate prevalence of 43.8%. No dicyemids were seen in squid, and only occasionally occurred in cuttlefish and nautiluses. Multiple octopus species are recognized to be hosts for 1 or more host-specific dicyemid species. Squid and cuttlefish are also recognized, more rarely, to have dicyemid populations. With a single exception, dicyemids are not described in nautilus.
Fungal infections were rare, consistent with the literature.43,58,64,68 Septicemic fungal infections of similar morphology (pale eosinophilic, 4- to 5-µm wide, parallel-walled, septate, nonpigmented fungal hyphae with dichotomous branching) were identified in 3 octopuses and 2 nautiluses, affecting the skin and gills with evidence of angioinvasion, plus multiorgan inflammation in the nautiluses. Fresh tissue was unavailable for culture and molecular diagnostic speciation was not performed. Fusarium species are an example of infectious marine fungal pathogens, which may infect cephalopods and fish.68,78
Inclusion bodies raising suspicion of viral involvement were seen in 2 California 2-spot octopuses and a single lesser octopus. To date, only a handful of viruses and viral-like lesions have been described in cephalopods.20,38,71 Material for PCR testing was unavailable, and electron-microscopy of these cases was not performed.
In this study, metazoan parasitism in cephalopods was sporadic. Cephalopods act as intermediate hosts in the life cycle of anisakine nematodes, pennellid copepods, and numerous cestodes. In general, metazoans appear to exhibit low host specificity in cephalopods worldwide.30,36,85 Only rare examples in this study exhibited any form of inflammation associated with parasitic invasion. This reinforces the hypothesis that such infections should generally be considered incidental, although a recent publication has drawn attention to associated inflammation and necrosis in some cases of cestodiasis in octopuses. 23
Ocular cataracts were seen in 2 cuttlefish and a single common octopus, which could have impaired vision and potentially affected feeding. A recent study of cephalopod ocular pathology identified inflammatory lesions, particularly anterior uveitis, as a frequent presentation, with cataracts in a substantial proportion (36%) of the 33 diagnostic specimens included from mixed cephalopods. 22
Idiopathic intratubular basophilic granular mineral deposits were described in the renal appendage of 5 nautiluses (of variable sex and age). These deposits (of uncertain chemical composition) were considered incidental but may be related to mineral-homeostasis associated with shell turnover. Their rarity in this study contrasts with the experience of others (LaDouceur, pers. comm.) who consider mineral aggregates to be a normal finding in these tubules. This may reflect differences in husbandry, fixation, or histological processing between institutions.
While laboratory and aquarium conditions are heavily studied topics in the captive husbandry of cephalopods,7,52 reports of toxicoses or environmental/life-support failure are few.12,40,87 Concentrations of dissolved inorganic nitrogenous compounds, such as nonionized ammonia (NH3-N), nitrite (NO2-N), and nitrate (NO3-N), are critical in closed systems housing cephalopods. Nonionized forms (NH3-N) are highly toxic to many marine species, even in very small concentrations. 86 In juvenile cuttlefish experimentally exposed to 6 mg/L of dissolved ammonia over 96-hour periods, such exposure resulted in bright red, swollen and friable digestive glands. 12 Histologic lesions included digestive gland epithelial swelling, anisokaryosis and intracytoplasmic vacuolation (hydropic degeneration) with multifocal to coalescent areas of lytic necrosis and sinusoidal dilatation. 12 Mild branchitis was observed.
Suspected toxicosis/chemical exposure or environmental/life-support failure can rarely be definitively diagnosed by diagnostic histopathology and may be considered diagnoses of exclusion in combination with relevant clinical histories. There are very few published studies specifically characterizing lesions of chemical insult in cephalopods and the authors advise caution in diagnosing toxicosis and chemical exposure based on histological examination alone. Water quality analysis with a focus on dissolved inorganic nitrogenous compounds, heavy metal concentrations, and salinity are warranted in cases exhibiting either: (1) severe acute diffuse interstitial branchial edema with sterile branchitis; or (2) renal epithelial cell intracytoplasmic vacuolation with pronounced anisocytosis and anisokaryosis. Further toxicological studies are needed to clarify toxin-related pathology.
Neoplastic diseases or specific nutritional deficiencies were not identified in any cephalopod in this study. To date, only rare unverified reports limited to tumor-like lesions of the ventral mandible 46 and an iridophoroma 75 are described in cuttlefish, while neoplastic disease in nautiluses are not described in the literature. Proliferative lesions are described in the eyes of cephalopods. 22
In conclusion, it is important that veterinary pathologists approaching cephalopod cases understand the life cycle differences between Coleoidea and Nautiloidea and how issues such as semelparity, life stage, physiology, and longevity may impact normal and abnormal findings. Pathologists are heavily reliant on clinical veterinarians or aquarists to undertake careful gross examination of integument and shell (where present) if harvesting tissues for histology, and to collect full tissue sets including reproductive organs. Recognition of alterations associated with environmental and toxic insults remains particularly difficult, and high-quality information about husbandry and environment, including water quality and life-support system parameters, should be requested to support disease investigations. Resources to assist the veterinary pathologist with cephalopod cases remain limited, scattered, and in unfamiliar locations. However, this study contributes to an increasing body of information describing clinical examination, normal macroscopic and microscopic anatomy, and their alterations in disease.35,51,53
This review offers a compilation of pathologic findings in captive cephalopods across a large, multicollection-based cohort of predominantly aquarium-housed cephalopods. Its findings provide additional insight into previously reported diseases and further evidence for hypotheses and presumed knowledge scattered widely in the cephalopod pathology literature. It identifies novel lesions in previously undescribed species and highlights disease entities prevalent in captive aquarium populations. It provides a framework for future clinicopathologic research, treatment, and husbandry refinement for these charismatic creatures.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231186306 – Supplemental material for A retrospective study of pathologic findings in cephalopods (extant subclasses: Coleoidea and Nautiloidea) under laboratory and aquarium management
Supplemental material, sj-pdf-1-vet-10.1177_03009858231186306 for A retrospective study of pathologic findings in cephalopods (extant subclasses: Coleoidea and Nautiloidea) under laboratory and aquarium management by Andrew F. Rich, Daniela Denk, Cheryl R. Sangster and Mark F. Stidworthy in Veterinary Pathology
Footnotes
Acknowledgements
The authors thank submitting aquaria and laboratories for their continued support and supply of cephalopod cases to IZVG Pathology, and IZVG’s veterinary and AHEF teams for their involvement in case management. Current and past technical staff at IZVG Pathology deserve mention for their histological expertise, as do staff at Nationwide Laboratories for their assistance in the microbiological examinations. Two former veterinary students, Helena Brewer and Victoria Lauren, were involved in student projects that collated information in earlier years, providing background for the current study. Thanks to Dr Imogen Payne BVSc MRCVS for her skilled assistance with postmortems and octopus photographs included in the figures.
Supplemental material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
