Abstract
Feline pulmonary carcinoma (FPC) is an uncommon neoplasm with unique morphological features. We describe the gross, histological, metastatic, and immunohistochemical aspects of FPC, based on postmortem examinations from an 11-year retrospective study. Thirty-nine cases were selected. Predispositions were observed in senior (P < .001) and Persian (P = .039) cats. There were three gross patterns of the pulmonary tumors: (a) a large nodule and additional smaller nodules, (b) a solitary nodule, and (c) small, multifocal to coalescent nodules. Extrapulmonary metastases were present in 22/39 cases (56.4%), mainly in the regional lymph nodes (17/39, 43.5%), skeletal muscles (9/39, 23%), kidneys (6/39, 15.3%), and parietal pleura (4/39, 10.2%). The primary tumor size was correlated with the occurrence of extrapulmonary metastases (P = .002). Histologically, the tumors were classified as papillary adenocarcinoma (19/39, 48.7%), adenosquamous carcinoma (ADS) (8/39, 20.5%), acinar adenocarcinoma (6/39, 15.3%), solid adenocarcinoma (3/39, 7.6%), lepidic adenocarcinoma (2/39, 5.1%), and micropapillary adenocarcinoma (1/39, 2.5%). By immunohistochemistry, 39/39 cases (100%) were positive for pancytokeratin, 34/39 (87.1%) for thyroid transcription factor-1, and 8/39 (20.5%) for vimentin. Immunoreactivity for p40 was detected in the squamous component of all ADSs (8/8, 100%) and occasionally in the glandular component of adenocarcinomas (10/31, 32.2%). Napsin A expression was absent in all feline tissue tested. The results indicate that a modified and simplified histological classification based on current human and domestic animal systems is appropriate for cats. Additionally, this study highlights the utility of p40 as an immunohistochemical marker for the diagnosis of FPC with squamous differentiation.
Keywords
Primary pulmonary carcinomas are malignant neoplasms originating from the epithelial components of the lung. 51 In cats, these neoplasms are infrequent and account for only 0.69% to 0.75% of all postmortem examination diagnoses.10,51 The average age of most affected cats is 12 to 13 years, and Persian cats seem to be overrepresented. 10 Intrathoracic and extrathoracic metastases are expected in up to 80% of the cases, resulting in a poor prognosis and short survival time.10,21 A relatively unusual pattern of metastases to one or more digits that is unique to cats has been referred to as “feline lung–digit (FLD) syndrome.”16,17,43 This aide-mémoire term reflects the fact that metastases to the digits cause noticeable clinical signs. A recent study describes variations on this theme and suggests replacing “FLD syndrome” with “feline muscle/ocular/digit/aorta/lung (MODAL) syndrome,” to remind veterinarians of the frequent involvement of metastases in skeletal muscles, eyes, digits, and aorta in cases of feline pulmonary carcinoma (FPC). 44 Although this information is partially reinforced in the literature, most studies involving sites of FPC metastases are focused only on the digits.16,29,43 There are only occasional case reports with clinical and pathological descriptions of metastases in other sites.2,7,28,35
The first histological classifications of FPC in the literature were based on cell morphology and site of tumor origin (eg, bronchial gland adenocarcinoma and bronchioloalveolar carcinoma).4,20,33 Studies on histological features of FPC in the last decade used previous histological classification for domestic animals, which still recognized tumors of bronchioloalveolar origin.12,50 The current histological classification of pulmonary tumors in domestic animals, 51 derived from human classification criteria provided by the World Health Organization (WHO), is based on cell morphology, without reference to the possible cell of origin. 45 Significant changes include (a) discontinuing the terms mixed adenocarcinoma and bronchioloalveolar carcinoma; (b) adding adenocarcinoma in situ, minimally invasive adenocarcinoma, and invasive adenocarcinoma; (c) replacing the term bronchioloalveolar carcinoma with lepidic adenocarcinoma; and (d) classifying invasive adenocarcinoma according to the predominant subtype. 51 Although the frequency of FPC using part of this classification system has been described in prior studies focused on molecular and clinical alterations,34,36 there are limited histological details to substantiate the appropriateness of this classification scheme for tumors in cats. Therefore, the aim of the present study was to describe the gross, histological, metastatic, and immunohistochemical aspects of FPC.
Materials and Methods
Case Selection
Electronic records of the cats submitted for postmortem examinations between January 2011 and November 2021 to the Department of Veterinary Pathology at the Universidade Federal do Rio Grande do Sul were searched for cases of FPC. Combinations of keywords “feline,” “cat,” “lung,” “pulmonary,” “neoplasm,” and “carcinoma” were used during the digital database search. All cases of FPC found were included in this study, regardless of the cause of death. The animals were from the metropolitan region of Porto Alegre, Rio Grande do Sul, Brazil. When present, information obtained from the records included data of signalment (sex, breed, and age [<1 year, 1–6 years, 7–10 years, and >10 years] 39 ), feline immunodeficiency virus (FIV), and feline leukemia virus (FeLV) status (determined by previous immunohistochemical, serological, or molecular tests), and concomitant comorbidities. Formalin-fixed paraffin-embedded (FFPE) tissue blocks of the selected cases were recovered from the archive. Tissues had been fixed in 10% neutral-buffered formalin for an undetermined period. Cases with poor histological tissue quality (severe artifacts or autolysis), less than one FFPE lung tissue block available for each 2 cm of tumor diameter, or previous history of metastatic extrapulmonary carcinoma were excluded. For comparative purposes, information on the signalment of all domestic cats submitted for postmortem examination during the analyzed period was obtained.
Gross and Histological Evaluation
Original records and corresponding photographs of the selected cases were used to reevaluate the gross characteristics of the pulmonary tumors, including the color, consistency (soft, firm, or hard), maximum diameter (<1 cm, 1.0–1.9 cm, 2.0–2.9 cm, or ≥3.0 cm), distribution (focal, multifocal, multifocal to coalescent, or diffuse), and anatomical location (cranial portion of the left cranial lobe, caudal portion of the left cranial lobe, left caudal lobe, right cranial lobe, right caudal lobe, right middle lobe, accessory lobe, or multiple lobes). Intratumoral cavities (cystic spaces), central area of tumor depression (umbilicated appearance), and additional intrathoracic lesions associated with pulmonary tumors were assessed as present or absent. Similar gross characteristics were reevaluated in the extrapulmonary metastatic sites. All presumptive metastases were confirmed histologically and diagnosed when the neoplastic pulmonary cells invaded and proliferated in extrapulmonary tissue. Organs with neoplastic cells only within a space lined by endothelium were not included as metastatic sites. Diffuse and widespread dissemination of neoplastic cells in the parietal pleura was considered pleural carcinomatosis. Histological evaluation of the eyes was performed in all cases, and of the digits only in cases with gross lesions affecting the paws.
Step sections of the FPCs stained with hematoxylin and eosin (HE), Alcian Blue (pH 2.5) (AB), and periodic acid Schiff (PAS) stains were reexamined and reclassified by 4 veterinary pathologists (I.R.S., J.R., M.B.B., and S.P.P.). A consensus was reached in cases of divergent interpretation among observers. Based on the current publications on pulmonary tumors of human and domestic animal systems,45,51 a modified and simplified histological classification without reference to the possible cell of origin was applied to our cases. This classification included adenocarcinoma, adenosquamous carcinoma (ADS), and squamous cell carcinoma (SCC). Depending on subjective evaluation of the predominant glandular growth pattern, adenocarcinoma was further subclassified into lepidic, acinar, papillary, micropapillary, and solid, in 5% increments. ADS was considered only in cases with both glandular and squamous patterns (≥10% of each pattern). The terms “adenocarcinoma in situ,” “minimally invasive adenocarcinoma,” and “invasive adenocarcinoma,” currently applied in human and canine pulmonary tumors,5,45 were not used because of the lack of well-established definitions (ie, relationship between gross tumor size and histological characteristics/size of foci of invasive lesions) with associated survival implications for cats.
In each pulmonary tumor, anisocytosis, and anisokaryosis (ie, form and size variation of the cytoplasm and nucleus, respectively) were subjectively graded as absent (<1% of the cells), mild (1%–15%), moderate (16%–49%), or marked (≥50%). Mitotic figures (MFs) were manually counted in a 2.37 mm2 area, using consecutive fields of high mitotic density and avoiding intratumoral necrosis, to determine the mitotic count (MC). Cases with lymphovascular invasion (LVI; ie, neoplastic cells invading through a vessel wall and endothelium, neoplastic cells within a space lined by endothelium, or fibrin thrombi adhered to tumor cells within a vascular space) were evaluated in all available sections to determine the number (few, <5 vessels; moderate, 5–10; or many, >10), type of vessels invaded (blood, muscular wall evident; lymphatic, no muscular wall evident; or both), and tumor-related site (intratumoral, peritumoral, or both). Desmoplastic stroma and intratumoral necrosis were estimated based on area as none (<1% of the tumor), small amounts (1%–10%), medium amounts (11%–50%), or large amounts (>50%). Extra- or intracellular mucin (AB- and/or PAS-positive), tumor spread through air spaces (STAS; ie, discontinuous spread of micropapillary clusters, solid nests, or single neoplastic cells from the primary tumor through the air spaces to adjacent or distant pulmonary parenchyma), 24 visceral pleural invasion (ie, neoplastic cells invading any layer of the visceral pleura), and other forms of cellular atypia were assessed as present or absent. Intratumoral inflammatory infiltrates were manually counted in all available sections and classified according to intensity (absent, <1 cell in all tumor area; mild, 1–15 cells; moderate, 16–50 cells; or marked, >50 cells) and cell composition. Available standardized methods of the Veterinary Cancer Guidelines and Protocols were used to evaluate parts of these variables. 30
Immunohistochemical Study
To confirm the origin and histological subtype of the carcinoma, immunohistochemistry (IHC) for pancytokeratin (panCK), thyroid transcription factor-1 (TTF-1), vimentin, and p40 was performed on sections of each pulmonary tumor. An anti-napsin A antibody was also applied in 6 FPC (1 adenocarcinoma of each subtype and 1 ADS). Antibodies details, dilutions, antigen retrieval, detection systems, and chromogens applied in each IHC assay are available in Supplemental Table 1. Harri’s hematoxylin was utilized as counterstain in all cases. The adjacent pulmonary parenchyma was considered as the internal positive controls for panCK, TTF-1, vimentin, napsin A, and p40. As external positive controls, we used normal feline skin for panCK and vimentin, normal feline thyroid gland for TTF-1, normal canine lung and kidney for napsin A, and normal human skin for p40. Primary antibodies were replaced by Universal Negative Control Serum (BioCare Medical, California) in randomly selected sections of FPC as negative controls. Attempting to validate the IHC for napsin A and p40 in feline tissues, we used additional external positive controls from cats (normal kidney and lung for napsin A; and cutaneous SCC and normal skin for p40), fixed in 10% neutral-buffered formalin for 24 to 48 hours. The cell types expected to be positive in the controls for p40 (ie, bronchial basal cells, cutaneous epidermal and adnexal basal cells, and neoplastic cells of the SCC) and napsin A (ie, type II pneumocytes, bronchiolar epithelium, alveolar macrophages, and tubular renal epithelium) were determined according to previous studies.18,19,40
Immunoreactivity for panCK (diffuse cytoplasmic), vimentin (diffuse cytoplasmic), TTF-1 (diffuse nuclear), napsin A (granular cytoplasmic), and p40 (diffuse nuclear) in the pulmonary tumors was subjectively scored based on reaction intensity and percentage of labeled cells by 3 veterinary pathologists (I.R.S., J.R., and S.P.P.). These two scores were assessed in areas with the highest density of positive cells and using a semiquantitative method, without a specific number of cells counted. Areas of intratumoral necrosis were avoided. The reaction intensity in the immunolabeled tumor cells was assessed as 0 (no reaction), 1 (weak), 2 (moderate), or 3 (intense), based on the majority labeling intensity (Supplemental Fig. S1a–d). The percentage of marked tumor cells was scored as 0 (<1% of the cells), 1 (1%–15%), 2 (16%–50%), or 3 (>50%). Cases with values <1 in any score were considered negative. Two final mean scores, one for the percentage of immunolabeled cells, and one for reaction intensity with each antibody, were calculated for all tumor subtypes. In neoplasms with glandular and squamous differentiation, the scores were evaluated in both components. Expected immunoreactivity for p40 and napsin A in the feline internal and external positive controls were also assessed based on the type of positive cells.
Statistical Analysis
General data were evaluated by descriptive statistics, using measures of central tendency for continuous variables and frequency for categorical variables. The correlation between the signalment groups and the occurrence of pulmonary carcinoma was evaluated by Pearson chi-square or Fisher’s exact tests, depending on characteristics of the contingency tables. Pearson chi-square was also used to compare occurrence of extrapulmonary metastases with the pulmonary tumor size. In cases with statistically significant P values (<.05), a pairwise z test with Bonferroni correction was applied to discriminate the differences between the groups. Analyses were performed using IBM SPSS Statistics for Windows, version 22.0 (Armonk, New York). All data analyzed in this study, including individual details of selected cases of FPC, are available as Supplemental Materials or by request to the authors.
Results
Cases Data
Of 1940 cats submitted for postmortem examination during the study period, 42 (2.1%) were diagnosed with pulmonary carcinomas. Three cases were not used in this study based on the exclusion criteria. Selected cats included 27/39 (69.2%) females and 12/39 (30.7%) males, with ages ranging from 3 to 20 years (mean and median of 13.8 and 14 years, respectively). Eighty-nine percent of the cases of FPC were seen in senior (>10 years) cats. The predominant breed was Domestic Shorthair (31/39, 79.4%), followed by Persian (5/39, 12.8%), Siamese (2/39, 5.1%), and Himalayan (1/39, 2.5%). Based on the expected proportion of cats submitted to our department, there were significant statistical differences for the occurrence of FPC in senior and Persian cats (Table 1). Previous retroviral status was available for 30 cats, of which 86.6% (26/30) were FIV and FeLV-negative, 6.6% (2/30) FIV-positive, 3.3% (1/30) FeLV-positive, and 3.3% (1/30) FIV and FeLV-positive. Although all cases had well-established causes of death, only 66.6% (26/39) were associated with pulmonary carcinomas. Comorbidities, seen in 27/39 (69.2%) cats, included lymphoma (9/39, 23%), chronic renal disease (8/39, 20.5%), hypertrophic cardiomyopathy (6/39, 15.3%), thyroid adenoma (3/39, 7.6%), oral SCC (2/39, 5.1%), and splenic mast cell tumor (2/39, 5.1%).
Correlations between signalment and pulmonary carcinoma occurrence in cats. a

P values in bold are statistically significant (P < .05) and indicate a correlation between the groups and the occurrence of pulmonary carcinoma. Numbers followed by the same subscripted letter in each column of a subtable do not differ significantly.
Abbreviation: DSH, Domestic Shorthair.
Submitted to Department of Veterinary Pathology, Universidade Federal do Rio Grande do Sul, from 2011 to 2021.
Pearson chi-square test.
Fisher Exact test.
Gross Findings
Gross lesions were observed in all cases, with three clear distribution patterns for the pulmonary tumors. The first consisted of multifocal tumors (24/39, 61.5%), including a large (1–7.5 cm diameter) nodule and additional smaller (0.2–1 cm diameter) nodules in the pulmonary lobes (Fig. 1A). The second was a solitary nodule (1–12 cm diameter) (10/39, 25.6%) (Fig. 1B). In both of these gross distribution patterns, the most commonly affected anatomic locations were the right caudal lobe (13/34, 38.2%) and left caudal lobe (13/34, 38.2%), followed by accessory lobe (5/34, 14.7%), caudal portion of the left cranial lobe (1/34, 2.9%), cranial portion of the left cranial lobe (1/34, 2.9%), and right middle lobe (1/34, 2.9%). The remaining cases (5/39, 12.8%) had diffusely distributed, multifocal to coalescent small (1.5 to 3 cm diameter) nodules in all pulmonary lobes (Fig. 1C). Regardless of the distribution, all tumors had soft to firm consistency and heterogeneous color, varying from white to gray to yellow. Umbilicated appearance was frequent (11/39, 28.2%). On the cut surface, tumors had white to yellow homogeneous color and well to poorly defined borders. Cavities were uncommon (3/39, 7.6%). Additional intrathoracic lesions associated with the pulmonary tumors were observed in 18/39 cases (46.1%) and included atelectasis (10/39, 25.6%), hydrothorax (9/39, 23%), pleural adhesions (5/39, 12.8%), and compression of the adjacent bronchi (1/39, 2.5%) (Supplemental Fig. S2a).
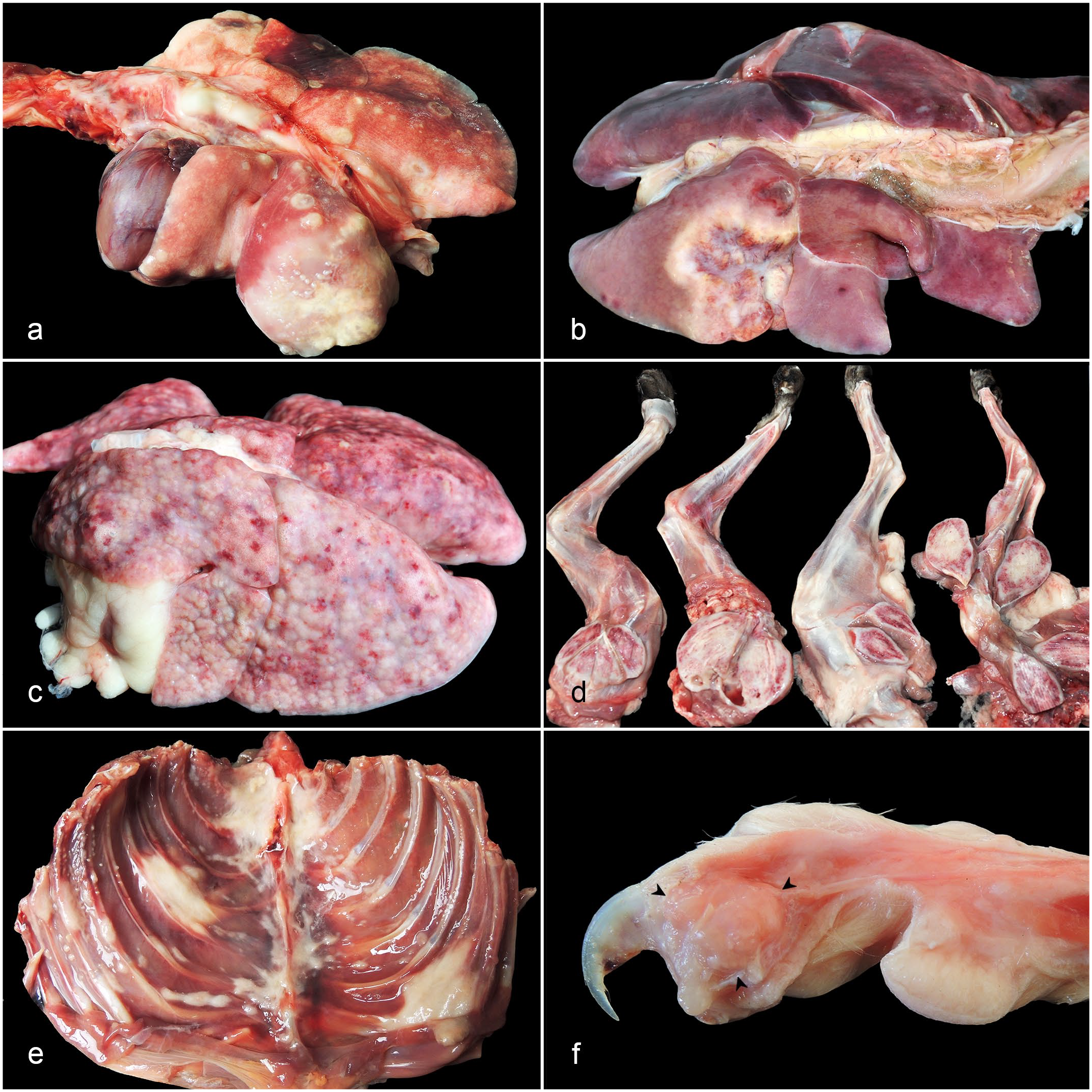
Pulmonary carcinoma, cat. (A) A large nodule in the left caudal lobe with additional smaller nodules in all pulmonary lobes. (B) Large umbilicated nodule in the right caudal lobe. (C) Small, multifocal to coalescent nodules in all pulmonary lobes, mimicking a diffuse pattern. (D) Muscular metastasis. Nodules in skeletal muscles of the hind and fore limbs. (E) Muscular metastasis. Nodules in multiple intercostal muscles. (F) Digital metastasis. A nodule (arrowheads) involving the subcutis and middle and proximal phalanges of the third digit.
Extrapulmonary metastases were found in 22/39 cases (56.4%), and all were readily detected on gross examination. The most common sites were regional lymph nodes (tracheobronchial and/or cranial mediastinal) (17/39, 43.5%), skeletal muscles (9/39, 23%) (Fig. 1D, E), kidneys (6/39, 15.3%), parietal pleura (pleural carcinomatosis) (4/39, 10.2%) (Supplemental Fig. S2b), eyes (3/39, 7.6%) (Supplemental Fig. S2c), and dermis and subcutis (3/39, 7.6%). Gross features of the tumors in these organs are available in Table 2. Uncommon sites comprised the thoracic and abdominal aorta (2/39, 5.1%) (Supplemental Fig. S2d), bones (scapula, femur, and ribs) (2/39, 5.1%), esophagus (2/39, 5.1%), adrenal glands (2/39, 5.1%), digits (1/39, 2.5%) (Fig. 1F), small intestine (1/39, 2.5%), spleen (1/39, 2.5%), mesenteric lymph nodes (1/39, 2.5%), and heart (1/39, 2.5%). Primary tumor size was correlated with occurrence of extrapulmonary metastases (P = .002) (Supplemental Table S2), such that only 16.6% (2/12) of cases with pulmonary tumors measuring less than 1.9 cm had metastasis. On the other hand, metastases were observed in 63.6% (7/11) and 81.2% (13/16) of the pulmonary tumors measuring 2.0 to 2.9 cm and ≥3 cm, respectively. Information on gross features of the pulmonary tumors according to histological subtypes are available in Supplemental Table S3.
Overall gross features and histological distribution of the lesions in the most common extrapulmonary metastatic sites in 39 feline pulmonary carcinomas.

The regional lymph nodes included tracheobronchial and cranial mediastinal.
The affected muscles included the biceps brachii, biceps femoris, brachialis, deltoid, diaphragm, external and internal intercostal, gastrocnemius, gracilis, iliacus, infraspinatus, masseter, peroneus longus, psoas major, sartorius, semitendinosus, semimembranosus, soleus, spinalis, subscapularis, supraspinatus, teres major and minor, thoracic serratus ventralis, and triceps brachii.
The affected dermis and subcutis included the perilabial, ventral abdominal, lumbosacral, and tail locations.
Pathological details of the ocular metastases from two cases have been published elsewhere. 3
Histological Findings
Most tumors were histologically classified as adenocarcinoma (31/39, 79.4%), of which 19/31 (61.2%) were papillary, 6/31 (19.3%) acinar, 3/31 (9.6%) solid, 2/31 (6.4%) lepidic, and 1/31 (3.2%) micropapillary. Only 10/31 (32.2%) adenocarcinomas had a single glandular pattern of growth; the remaining (21/31, 67.7%) were classified according to predominant pattern. Histological features of ADS were observed in 8/39 cases (20.5%). No SCCs were found. The main histological features of each tumor subtype are detailed below and summarized in Supplemental Table S4. Additional pulmonary lesions, seen in 35/39 cases (89.7%), included alveolar edema (26/39, 66.6%), smooth muscle hypertrophy/hyperplasia of the bronchioles and/or tunica media of arterioles (15/39, 38.4%), type II pneumocyte proliferation (7/39, 17.9%), hemorrhage (6/39, 15.3%), congestion (4/39, 10.2%), reactive pleural mesothelial cells (2/39, 5.1%), and lymphoid hyperplasia of the bronchus-associated lymphoid tissue (1/39, 2.5%).
Papillary adenocarcinoma
The main histological feature of the papillary adenocarcinoma was a nonencapsulated, well to poorly defined neoplastic proliferation of pseudostratified, tall columnar cells arranged in arborizing fronds (papillae), and supported by scant fibrovascular stroma (Fig. 2A). Some cases had mucin in the intraluminal space between the papillae. Neoplastic cells had a small amount of intensely eosinophilic cytoplasm, poorly defined cell borders, and rarely intracellular mucin in the apical pole. Nuclei were round to oval with finely stippled or vesicular chromatin and 1 to 2 nucleoli. Anisocytosis and anisokaryosis were usually moderate, the MC ranged from 3 to 61 (mean of 24), and other forms of atypia (karyomegaly, macronucleoli, and/or bi/multinucleated cells) were frequent. There were small to large amounts of intratumoral necrosis, associated with hemorrhage, cholesterol clefts, and inflammatory infiltrates of neutrophils and hemosiderin-laden macrophages. The presence of STAS, pleural visceral invasion, and desmoplastic stroma varied in this adenocarcinoma subtype. Foci of LVI were seen in most cases (13/19, 68.4%), mainly affecting blood vessels in intratumoral areas. These tumors were commonly infiltrated by small to large numbers of inflammatory cells (lymphocytes, plasma cells, and/or macrophages). The neoplastic cells had occasional polygonal morphology with abundant, pale, finely vacuolated eosinophilic cytoplasm. Foci of other adenocarcinoma patterns (solid, acinar, lepidic, and/or micropapillary) were frequently observed (12/19, 63.1%).
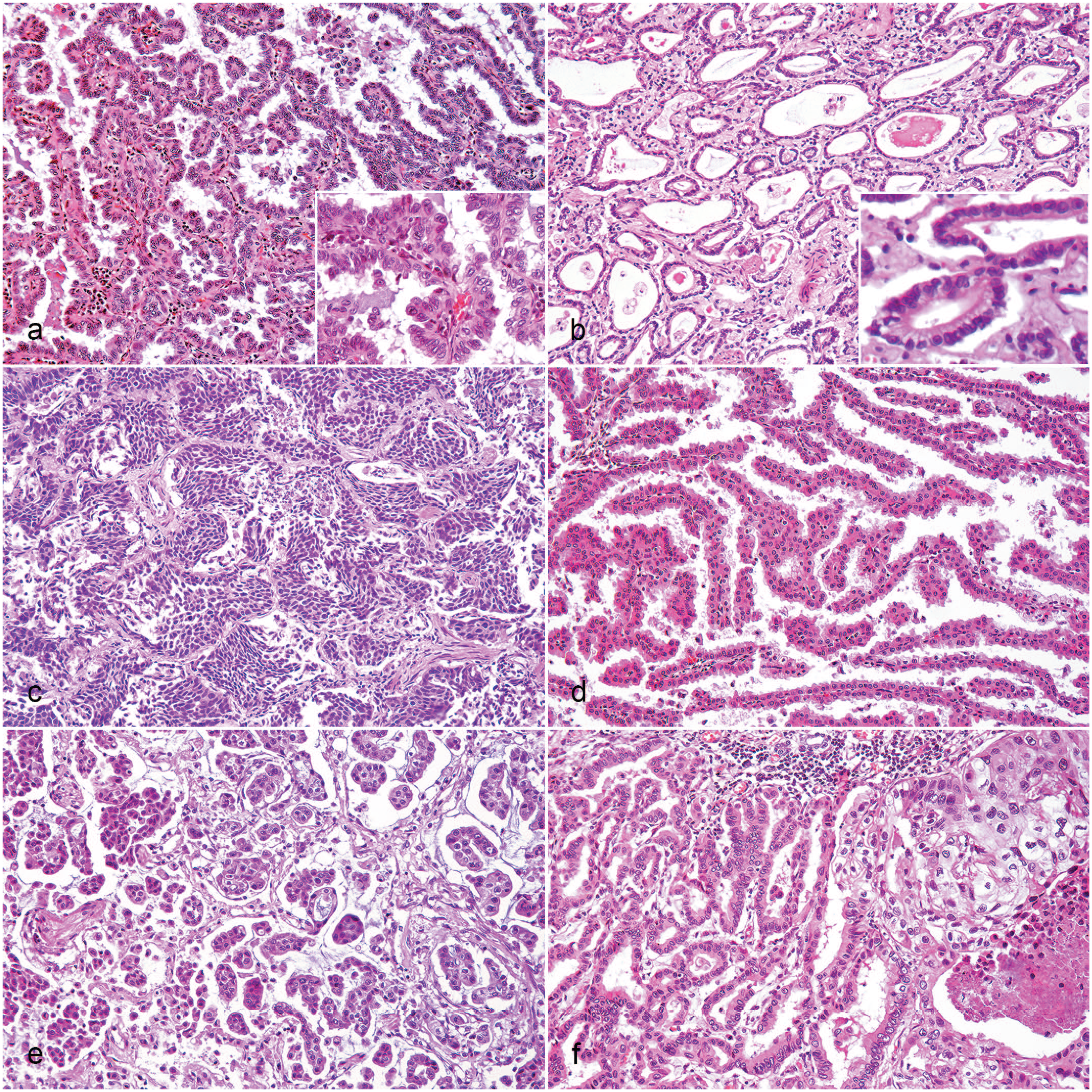
Pulmonary carcinoma, cat. Hematoxylin and eosin. (A) Papillary adenocarcinoma (AD). Arborizing fronds formed by a monolayer of pseudostratified columnar neoplastic cells (inset). (B) Acinar AD. Round to oval glands formed by a monolayer of neoplastic cells (inset). (C) Solid AD. Solid sheets of polygonal neoplastic cells without squamous differentiation. (D) Lepidic AD. Alveolar wall-like fibrovascular structures lined by a monolayer of cuboidal neoplastic cells. (E) Micropapillary AD. Papillary tuffs formed by cuboidal neoplastic cells without stroma. (F) Adenosquamous carcinoma. Mixture of both glandular (left side) and squamous (right side) neoplastic cells.
Acinar adenocarcinoma
Acinar adenocarcinomas were nonencapsulated, well-defined tumors, comprised of a neoplastic proliferation of simple tall columnar cells arranged in round to oval glandular structures with a central luminal space (acini) (Fig. 2B), surrounded by moderate fibrovascular stroma. Acini varied in size and were frequently filled by cellular debris, extracellular mucin, and neutrophilic and histiocytic exudate. Neoplastic cells had well-demarcated cell borders, moderate pale eosinophilic cytoplasm, and occasional intracytoplasmic mucin in the apical pole. Nuclei were basal and round to oval with finely stippled chromatin and 1 nucleolus. Anisocytosis and anisokaryosis were mild to moderate, and the MC varied from 3 to 35 (mean of 17). Multinucleated cells and karyomegaly were rarely seen. Some cases had STAS, visceral pleural invasion, and large amounts of intratumoral necrosis, associated with hemorrhage and inflammatory infiltrates of neutrophils and hemosiderin-laden macrophages. LVI was variably present (4/6, 66.6%), with all types of vessels affected in intra- and peritumoral areas. Mild to moderate, intratumoral, lymphoplasmacytic, and/or macrophagic inflammation was seen in all cases. Rarely, acini had pluristratified cuboidal epithelium and the stroma contained small to large amounts of desmoplasia. Foci of solid and papillary growth were present in almost all cases of this subtype (5/6, 83.3%).
Solid adenocarcinoma
The solid adenocarcinoma subtype was histologically composed of a nonencapsulated, well-defined neoplastic proliferation of polygonal cells arranged in solid sheets and supported by scant fibrovascular stroma (Fig. 2C). The cells had scant, pale eosinophilic cytoplasm, and poorly demarcated cell borders. Nuclei were central and round to oval with condensed to finely stippled chromatin and 1 nucleolus. Anisocytosis and anisokaryosis were usually moderate, and the MC varied from 22 to 25 (mean of 23). Karyomegaly and multinucleated cells were commonly present. Few to many foci of invasion of blood vessels in intra- and/or peritumoral areas were seen in most cases (2/3, 66.6%). There were small to large amounts of intratumoral necrosis associated with neutrophilic inflammation and cholesterol clefts. Mild lymphocytic inflammation was rarely present in intratumoral areas. STAS and visceral pleural invasion were present in 66.6% (2/3) of the cases. Desmoplastic stroma was absent, except in one case. Occasionally, acinar and papillary growth patterns were also seen in all cases. There was no extracellular or intracellular mucin.
Lepidic adenocarcinoma
Lepidic adenocarcinoma was characterized by a nonencapsulated, well-defined neoplastic proliferation of simple cuboidal to low columnar cells lining alveolar wall-like fibrovascular structures (Fig. 2D). Neoplastic cells had poorly demarcated cell borders and small amounts of intensely eosinophilic cytoplasm. Central nuclei were round with condensed to finely stippled chromatin and 1 nucleolus. There was mild anisocytosis, mild to moderate anisokaryosis, and 3 to 32 MFs (mean of 17) per 2.37 mm². Karyomegaly was present in rare neoplastic cells. Visceral pleural invasion and small amounts of intratumoral necrosis, associated with neutrophilic inflammation, cholesterol clefts, and dystrophic calcification, were present. Only mild intratumoral lymphocytic inflammation was seen. Occasionally, there were foci of papillary and micropapillary differentiation amidst the lepidic growth. One case had extracellular and intracellular mucin in the foci of other glandular growth patterns. Desmoplastic stroma, LVI, and STAS were absent in this adenocarcinoma subtype.
Micropapillary adenocarcinoma
The single case diagnosed as micropapillary adenocarcinoma was characterized by a nonencapsulated, poorly defined neoplastic proliferation of cuboidal cells arranged in papillary tufts forming florets and morula-like structures that lacked fibrovascular cores (Fig. 2E). Papillary tufts appeared detached from alveolar walls and often floated in extracellular mucin. Neoplastic cells had distinct, intensely eosinophilic cytoplasm. Nuclei were central and round to oval with condensed to finely stippled chromatin and 1 nucleolus. There was moderate anisocytosis, marked anisokaryosis, a MC of 33, and occasional macrocytosis and bi/multinucleated cells. The neoplastic cells frequently invaded the visceral pleura. Many foci of blood vessel invasion were seen in both intra- and peritumoral areas. Mild neutrophilic inflammation was observed intermixed with the neoplastic cells. It was not possible to evaluate STAS due to micropapillary growth being a characteristic of this pattern. Desmoplastic stroma and intratumoral necrosis were absent.
ADS
ADS cases formed a nonencapsulated, poorly defined neoplastic proliferation with 2 distinct components (Fig. 2F). The first, a glandular growth, had histological features similar to papillary and acinar adenocarcinoma. The second was a squamous component characterized by solid sheets of polygonal cells with evidence of intercellular bridges and/or individual keratinized cells. The squamous cells had abundant, glassy eosinophilic cytoplasm, and well-demarcated cell borders. Nuclei were round to oval with finely stippled or vesicular chromatin and 1 to 2 nucleoli. Anisocytosis was moderate, anisokaryosis marked, and the MC ranged from 22 to 103 (mean of 53). Frequently, neoplastic cells had karyomegaly, macronucleoli, and multiple nuclei. There were small to medium amounts of desmoplastic stroma and medium to large amounts of intratumoral necrosis, associated with neutrophilic inflammation, cholesterol clefts, and dystrophic calcification. Mild to moderate lymphoplasmacytic inflammation and occasional visceral pleural invasion were observed. Many blood vessels in both intra- and peritumoral areas were invaded by neoplastic cells. In foci of glandular growth, there was extra- and intracellular mucin. STAS and keratin pearl formation were not seen.
Extrapulmonary metastatic sites
Regardless of the primary pulmonary tumor classification, extrapulmonary metastatic sites were histologically characterized by the formation of round to oval glandular structures or rare solid sheets. There were small to large amounts of desmoplastic stroma, mainly in the muscular tissues (Fig. 3A). Details of the histological distribution of the neoplastic cells in the main metastatic sites are available in Table 3.
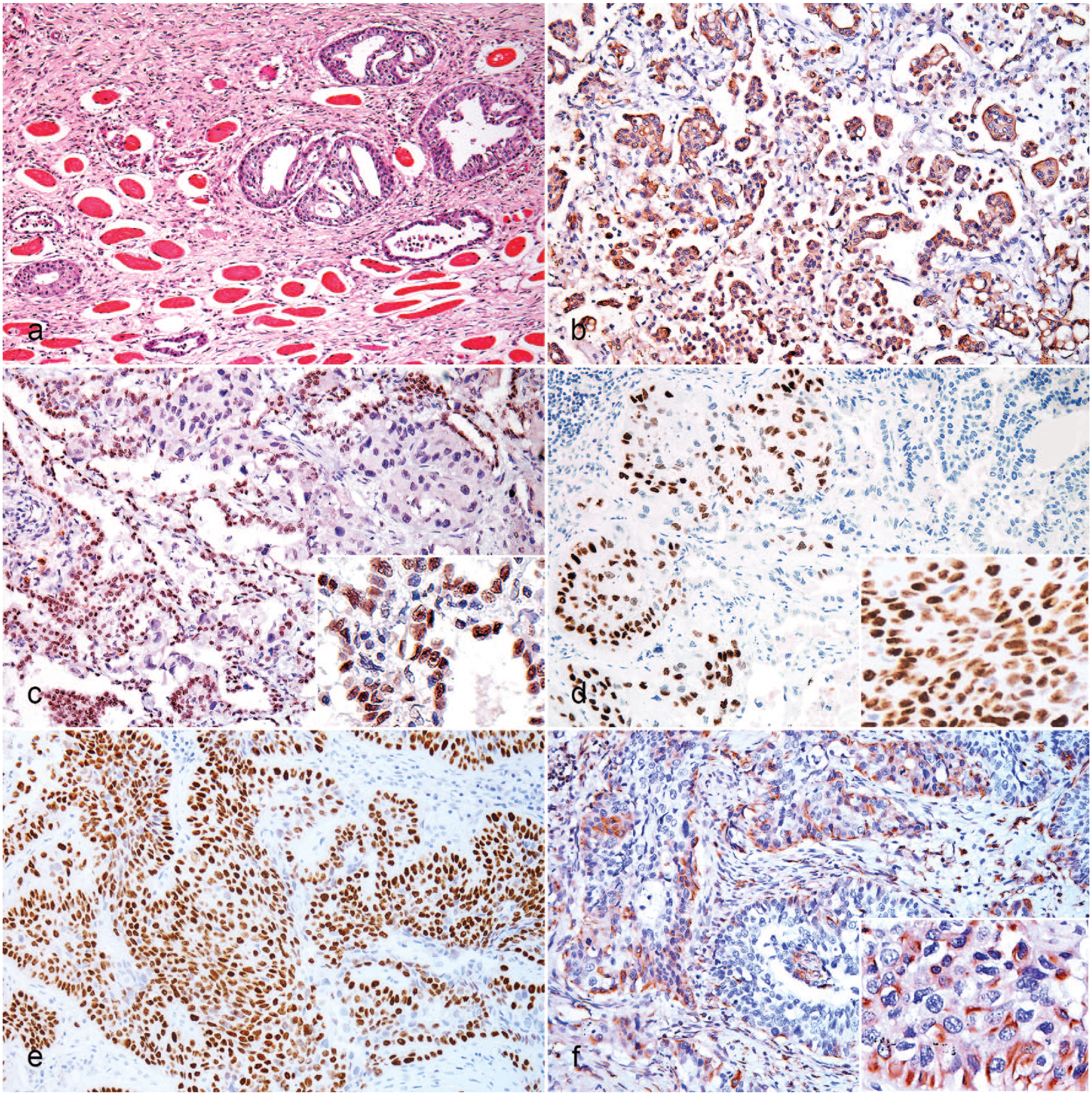
Pulmonary carcinoma, cat. (A) Muscular metastasis. Round to oval glandular structures within large amounts of desmoplastic stroma. Hematoxylin and eosin. (B) Micropapillary adenocarcinoma. Diffuse, intense cytoplasmatic immunoreactivity in all neoplastic cells. Immunohistochemistry (IHC) for pancytokeratin. (C) Adenosquamous carcinoma. Diffuse, intense nuclear immunoreactivity (inset) in the glandular component. IHC for thyroid transcription factor-1. (D, E) Adenosquamous carcinoma. Diffuse, intense nuclear immunoreactivity (inset) in the squamous component. IHC for p40. (F) Adenosquamous carcinoma. Intense cytoplasmic immunoreactivity (inset) in squamous neoplastic cells. IHC for vimentin.
Immunoreactivity for pancytokeratin, vimentin, thyroid transcription factor-1, and p40 in 39 feline pulmonary carcinomas.
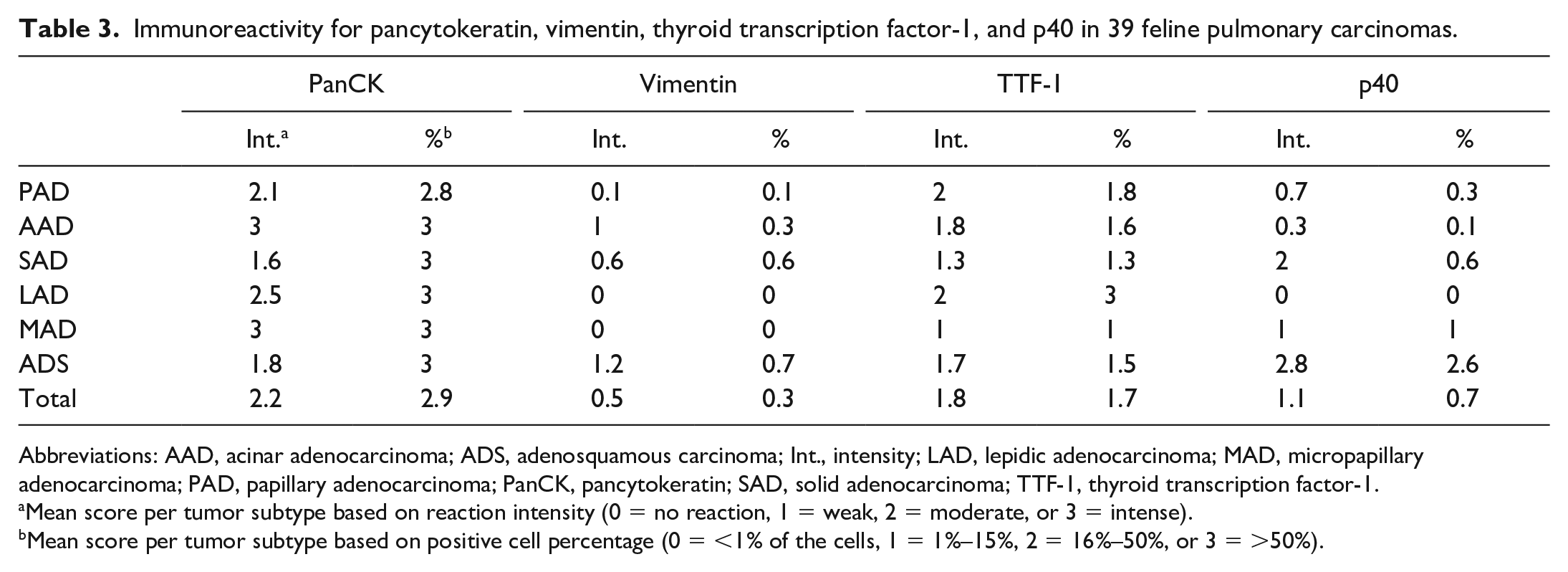
Abbreviations: AAD, acinar adenocarcinoma; ADS, adenosquamous carcinoma; Int., intensity; LAD, lepidic adenocarcinoma; MAD, micropapillary adenocarcinoma; PAD, papillary adenocarcinoma; PanCK, pancytokeratin; SAD, solid adenocarcinoma; TTF-1, thyroid transcription factor-1.
Mean score per tumor subtype based on reaction intensity (0 = no reaction, 1 = weak, 2 = moderate, or 3 = intense).
Mean score per tumor subtype based on positive cell percentage (0 = <1% of the cells, 1 = 1%–15%, 2 = 16%–50%, or 3 = >50%).
Immunohistochemical Characterization
The expected immunoreactivity for p40 was observed in all feline internal and external positive controls. It was characterized by diffuse nuclear labeling in the bronchial basal cells, cutaneous epidermal and adnexal basal cells, and neoplastic cells of the cutaneous SCC (Supplemental Fig. S3a–b). However, the normal feline lung and kidney and pulmonary internal controls adjacent to the tumors did not have immunoreactivity for napsin A, although the canine external controls were positive (Supplemental Fig. S3c–d).
In the FPCs, the panCK, TTF-1, p40, and vimentin had the expected immunoreactivity and variable scores of intensities and percentage of positive cells among the histological classifications (Table 3). For panCK, all cases (39/39, 100%) were positive with a high percentage of positive cells (>50%) and moderate to intense reaction with diffuse distribution (Fig. 3B). Thirty-four (87.1%) of the cases were positive for TTF-1 in the glandular component, usually characterized by <50% of positive cells and weak to moderate reaction with diffuse (Fig. 3C) or random distribution. Immunoreactivity for TTF-1 was not seen in areas of squamous differentiation. In contrast, there was a higher percentage of positive cells and moderate to intense reaction for p40 with diffuse distribution in the squamous component of the ADSs (8/8, 100%) (Fig. 3D, E). Minimal immunoreactivity for p40 was also observed in occasional adenocarcinoma subtypes (10/31, 32.2%), characterized by 1% to 15% positive cells and weak to moderate reaction with basal-like distribution in solid nests or scattered cells in random distribution. Immunoreactivity for vimentin was detected in 8/39 cases (20.5%), mainly in ADSs (Fig. 3F) and occasional adenocarcinoma subtypes. This vimentin immunoreactivity had variable percentage of positive cells and moderate to intense reaction with random distribution in both squamous and glandular cells, which also expressed panCK.
Discussion
FPCs are uncommon, sporadic tumors. In the present case series, the frequency of pulmonary carcinomas in the feline population evaluated was slightly higher than the frequency described in the literature (ie, 2.1% versus 0.6%–0.7%, respectively),10,51 but is still considered low. Previously reported predispositions for pulmonary carcinomas in senior (>10 years) and Persian cats 10 were confirmed. However, based on the relatively small number of cases included in our study, the strength of these predispositions in cats is unknown and should be further analyzed in a larger population. Interestingly, even though pulmonary carcinoma is considered an aggressive disease in cats, this neoplasm was considered the cause of death in just over 65% of our cases. Most of these cats also had other diseases commonly diagnosed in old age (eg, chronic renal disease and intestinal lymphoma), reinforcing FPC as a geriatric disease. Similar to other published studies,10,21,26 there was no apparent sex predisposition. In humans, although pulmonary cancer incidence is higher in men, a sex-specificity in lung cancer risk for women is associated with certain epidemiological, hormonal, and molecular factors. 42
Detailed pathological characterization of gross patterns of FPC is limited in the literature. Most studies are focused on thoracic radiologic and tomographic results, which often use a different methodological approach.1,4,26 Similar to our findings, the gross distribution of a focal tumor involving the caudal pulmonary lobes, regardless of the presence of additional smaller tumors, has been reported in up to 79% of the cases. 1 Tumors in multiple pulmonary lobes without an obvious primary nodule are uncommon and present only in 3.5% of FPCs, 1 in contrast to the 12.8% of multifocal to coalescent tumors observed in the current study. The pulmonary tumor size was correlated with the occurrence of extrapulmonary metastases. This result parallels previously reported data, in which metastasis has not been observed in cats with pulmonary tumors smaller than 1 cm in diameter. 10 Furthermore, as observed in our cases, common gross features of pulmonary tumors in cats include umbilicated appearance and irregular margins.1,51 Only cavities, described as a frequent finding in FPCs, 51 had a lower frequency than expected. Additional intrathoracic lesions associated with pulmonary tumors were atelectasis, pleural adhesions, bronchial compression, and hydrothorax, validating previous surveys.1,51
A modified and simplified histological classification system, derived from current human and domestic animal systems, was applied to the FPCs of this study. The most significant change was the exclusion of the terms “adenocarcinoma in situ,” ‘minimally invasive adenocarcinoma,’ and “invasive adenocarcinoma,” which are currently used for pulmonary tumors in humans and dogs.5,45 The histological diagnostic criteria that differentiate these subtypes are based on tumor size, growth patterns, and presence/size of invasive lesions. 46 The main problem with the application of this subclassification to FPCs is the size criteria of the human classification. Considering the huge size discrepancies between human and feline lungs, tumors ≤3 cm, and invasive foci ≤5 mm may have different clinical implications for cats. Of the cases with pulmonary tumor ≤3 cm in our study, 39.1% had extrapulmonary metastases and had at least 1 invasive component (ie, any histological subtype other than a lepidic pattern and neoplastic cells infiltrating vessels, stroma, air spaces, or visceral pleura). On the other hand, considering these histological definitions without reference to tumor size, all our cases of adenocarcinoma were tentatively classified as “invasive adenocarcinoma.” The single case with only lepidic growth pattern was likely to be classified as lepidic adenocarcinoma rather than minimally invasive adenocarcinoma due to the presence of intratumoral necrosis, visceral pleural invasion, and minimal cytoplasmatic and nuclear atypia.
The vast majority of the FPCs examined were diagnosed as adenocarcinoma and the minority as ADS (ie, 79.4% and 20.5%, respectively). Similar frequencies of feline pulmonary adenocarcinoma, including the obsolete bronchioloalveolar carcinoma, and ADS are reported in published studies (79.5%–92.3% and 15.4%–19.4%, respectively).10,20,27,34 In decreasing order, solid and lepidic patterns have been reported as the most common histological subtypes of feline pulmonary adenocarcinoma. 34 These data are inconsistent with our results since the papillary adenocarcinoma accounted for almost half of our FPCs. Five per cent of the cases (2/39) had histological characteristics of lepidic adenocarcinoma, a subtype that is morphologically compatible with the bronchioloalveolar carcinoma and represents 13.6% to 38.8% of FPCs in prior studies.20,27 Interestingly, there was a case of micropapillary adenocarcinoma, a highly aggressive subtype described only once in cats. 34 Although SCC is reported in up to 11.7% of FPCs, 4 this subtype was not diagnosed in our cases. It is possible that the classification criterion of ≥10% of both glandular and squamous patterns for the ADS diagnosis could have affected comparisons. In previous surveys,10,20,27,34 the use of a minimal percentage of each component is not entirely clear. Moreover, there were numerous similarities in the histological features between the subtypes found in this study and corresponding subtypes described in humans and dogs.45,51
The classically established mechanisms of tumor spread from pulmonary carcinoma are hematogenous, lymphatic, and transcoelomic routes. 45 In humans, STAS has recently been recognized as a route of tumor spread and associated with poor prognosis, 24 resulting in its inclusion as a microscopic invasion criterion. 45 Histological lesions compatible with these mechanisms of tumor spread were seen in this study. Although aerogenous spread has been cited in veterinary literature,31,33,51 studies using the established criteria for the diagnosis of STAS 24 have not been previously described. Visceral pleural invasion in humans is classified based on the affected layer using elastic stain, resulting in the determination of tumor staging and therapeutic strategy. 47 This histological assessment was not performed in our cases because of the lack of associated clinical implications for cats. Furthermore, the routes of tumor spread can lead to the involvement of specific organs in the metastatic process (eg, regional lymph nodes and intrapulmonary sites in lymphatic spread; liver, bones, brain, and adrenal glands in hematogenous spread; and pleural carcinomatosis in transcoelomic spread).14,45 These patterns could not be determined in our cases due to the low number of cases and overlap of histological lesions and spread routes in metastatic cases.
The main metastatic sites of FPC described by previous reports are the intrathoracic organs, including the pulmonary lobes, regional lymph nodes, and parietal pleura.10,20 The diagnosis of intrapulmonary metastases based only on pathological features (ie, tumor size, anatomical location, and histological patterns) is difficult. Clonality assessment of a single or separate lineage would be ideal to confirm presumed intrapulmonary metastasis and differentiate it from synchronous primary tumors, 11 although a comprehensive histologic evaluation can have similar accuracy. 14 In our cases, the additional small tumors with multifocal distribution shared similar histological findings with the respective larger tumor. Therefore, we assumed that these cases corresponded to intrapulmonary metastases. Digital metastasis was found in only one case, which was unexpected data considering the reports on “FLD syndrome.”16,17,43 Although skeletal muscle was the main site of extrapulmonary metastasis, lesions in this tissue are less commonly discussed in the FPC literature. 28 Curiously, skeletal muscle can be a metastatic site easily identified during clinical physical examination. 28 These results reinforce the use of the term “feline MODAL syndrome,” even if not all the highlighted organs (ie, skeletal muscle, eyes, digits, and aorta) were major metastatic sites in this study. Secondary neoplastic lesions in the kidneys, parietal pleura, and dermis and subcutis were more frequent than in the aorta and digits. Rare metastatic sites previously described and not seen in our cases include trachea, omentum, mesentery, liver, salivary glands, and brain.1,7,29,32
IHC is an important diagnostic tool for pulmonary carcinomas, mainly to differentiate primary from secondary tumors. In human and veterinary medicine, the most frequent pneumocyte marker for pulmonary carcinoma is TTF-1,5,10,45 a nuclear tissue-specific protein expressed in normal type II pneumocytes and bronchiolar epithelial cells. 41 TTF-1 expression in this study (87.1%) was considerably higher than previously described in FPC (58%),10,13,34 although it was mainly characterized by <50% positive cells and weak to moderate labeling. Differences in the frequency of positive cases may be partly explained by the different antigen retrieval methods used in the studies. In human pulmonary adenocarcinoma, areas with papillary and lepidic pattern are known to have more extensive TTF-1 immunoreactivity than solid-predominant areas. 23 Decreased TTF-1 expression in less-differentiated tumor cells has also been reported for FPC, 27 while our negative cases included a heterogeneous group of histological subtypes (3 papillary adenocarcinomas, 1 solid adenocarcinoma, and 1 ADS). According to previously published data, prolonged fixation does not significantly alter TTF-1 immunoreactivity; 41 therefore, this common cause of IHC failure was not considered to be a problem in the present case series.
Napsin A is a functional aspartic proteinase, detected in normal type II pneumocytes, bronchiolar epithelial cells, and alveolar macrophages. 40 This marker is considered another important pneumocyte marker for human pulmonary carcinoma, with expression in up to 100% of the adenocarcinoma subtypes. 22 A high sensitivity of 92% is also reported for canine pulmonary carcinoma. 5 Based on these results and the lack of immunohistochemical characterization of napsin A in feline tissues, we initially hypothesized that the application of this marker could increase the accuracy of the FPC diagnosis. However, unexpectedly, there was no expression of napsin A in any of the feline normal tissues and pulmonary carcinomas tested. The IHC protocol used in this study has been validated in normal bovine (unpublished data) and canine tissues. Negative results with the same napsin A clone are documented in only one case report of feline sarcomatoid renal cell carcinoma, 52 while details of the IHC protocol and positive controls were not included. Moreover, a prior study reported no reduction in immunoreactivity for napsin A in tissues fixed in formalin ≤5 weeks, 40 and it is unlikely that all our tested cases were fixed longer than 6 weeks. We also attempted to investigate this possible explanation for these results using feline normal tissues with controlled fixation time as external positive controls. Therefore, it cannot be determined whether our negative results reflect a lack of reactivity of this napsin A clone in feline tissues or an issue with the IHC protocol.
IHC for cytokeratin and vimentin have been used to differentiate epithelial from mesenchymal neoplasm origins, except for neoplasms that express both markers (eg, mesothelial tumors). In the present case series, the epithelial origin of all FPCs was confirmed by IHC for panCK. A fifth of the cases (8/39) also showed immunoreactivity for vimentin, including 4 ADSs, 2 acinar adenocarcinomas, 1 solid adenocarcinoma, and 1 papillary adenocarcinoma. In the prior studies of FPC, cytokeratin and vimentin co-expression has been reported in only 4 lepidic adenocarcinomas. 49 A similar phenomenon is reported in 9.4% to 38% and 38% of human and canine pulmonary carcinomas, respectively.9,15,37 Aberrant expression of vimentin in pulmonary carcinoma is associated with several mechanisms of the tumor initiation and progression, mainly epithelial-to-mesenchymal transition and metastatic spread. 25 To date, the potential relevance of this co-expression in FPC is unknown. Additionally, in the diagnostic work-up, alternative markers are recommended to distinguish mesothelial tumors and primary pulmonary adenocarcinomas. 37 Herein, TTF-1 immunoreactivity in 7 of the 8 cases positive for vimentin confirmed the pulmonary origin. In the single case negative for TTF-1 and positive for vimentin, the histological pulmonary findings and lack of pleural lesions ruled out the diagnosis of pleural mesothelioma.
Another important role of IHC in human pulmonary carcinoma is to differentiate the cellular composition of the tumor subtypes, specifically adenocarcinoma, ADS, and SCC. 45 A minimal panel of two-antibodies containing pneumocyte and squamous markers is effective for confirming these subtypes 38 when the tumor does not allow confident morphologic classification. Currently, the most popular squamous-specific marker for human pulmonary carcinoma is p40 (ΔNp63), an isoform of p63. 8 In the veterinary literature, to our knowledge, p40 expression has been reported only in canine mammary tumors and a salivary neoplasm of a black-tailed prairie dog.6,48 In this study, immunoreactivity for p40 was observed in squamous neoplastic cells from all pulmonary ADSs and the cutaneous SCC external control. Rarely, human pulmonary adenocarcinoma can express p40 reactivity in the peripheral basal-like layer of tumor nests or in random foci, 8 similar to the pattern observed in some of this study’s adenocarcinomas. These positive cells had no morphological evidence of squamous differentiation. Also, the distribution patterns of positive cells in the adenocarcinomas were readily distinguishable from the diffuse reactivity for p40 in squamous neoplastic cells. In normal human tissues, p40 is expressed by bronchial basal cells and cutaneous epidermal and adnexal basal cells,18,19 which was consistent with our observations. These results suggest the usefulness of p40 in the immunohistochemical diagnosis of FPC and probable specificity for feline squamous and basal epithelial cells. However, additional studies using other pulmonary and extrapulmonary tumors and normal tissues will be needed to better characterize the expression of this marker in cats.
As all FPCs selected in this study were diagnosed at the time of postmortem examination, determination of the clinical implication and importance of the pathological findings found was not possible. Future studies will need to focus on the analysis of the prognostic significance associated with the gross patterns, histological subtypes, and IHC scores reported. Other inherent limitations were the low sample size and non-standardization in sample collection and fixation. Despite the limitations, we described detailed features of FPC. Pulmonary gross lesions were mainly characterized by a large focal nodule in the caudal lobes and additional small nodules. Metastases were found in most cases, with a distribution pattern that corroborated the use of the term “feline MODAL syndrome.” Histological subtypes were similar to human and canine pulmonary carcinomas, indicating that a modified and simplified classification system is appropriate for cats. Furthermore, the utility of p40 as a squamous cell marker in the diagnosis of FPC was highlighted.
Supplemental Material
sj-pdf-2-vet-10.1177_03009858221122517 – Supplemental material for Feline pulmonary carcinoma: Gross, histological, metastatic, and immunohistochemical aspects
Supplemental material, sj-pdf-2-vet-10.1177_03009858221122517 for Feline pulmonary carcinoma: Gross, histological, metastatic, and immunohistochemical aspects by Igor R. Santos, Jacqueline Raiter, Éryca C. Lamego, Marcele B. Bandinelli, Tainah P. Dal Pont, Kalvin F. Siqueira, Bruno A. Almeida, Welden Panzeira, Luciana Sonne, David Driemeier and Saulo P. Pavarini in Veterinary Pathology
Supplemental Material
sj-xlsx-1-vet-10.1177_03009858221122517 – Supplemental material for Feline pulmonary carcinoma: Gross, histological, metastatic, and immunohistochemical aspects
Supplemental material, sj-xlsx-1-vet-10.1177_03009858221122517 for Feline pulmonary carcinoma: Gross, histological, metastatic, and immunohistochemical aspects by Igor R. Santos, Jacqueline Raiter, Éryca C. Lamego, Marcele B. Bandinelli, Tainah P. Dal Pont, Kalvin F. Siqueira, Bruno A. Almeida, Welden Panzeira, Luciana Sonne, David Driemeier and Saulo P. Pavarini in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) supported this study.
ORCID iDs
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
