Abstract
Cases of Theileria-associated mortality are rarely reported in African wild artiodactyls. Descriptions of lesions are limited, particularly in endangered hippotraginids. Here, we analyzed retrospectively the gross and histologic findings in 55 roan antelope (Hippotragus equinus) with fatal natural theileriosis. The most frequently recorded gross findings in 40 cases were widespread petechiae and ecchymoses (72.5%), probable anemia (67.5%), icterus (60%), splenomegaly (60%), hepatomegaly (52.5%), and pulmonary edema (50%). Histologic lesions in 34 cases were characterized by multi-organ infiltrates of parasitized and nonparasitized mononuclear leukocytes (MLs), and fewer multinucleate giant cells (MNGCs). Liver, lung, kidney, adrenal gland, and heart were most consistently infiltrated, followed by spleen and lymph nodes. Leukocytes were phenotyped in lung, liver, kidney, and heart specimens from 16 cases, using immunohistochemistry to detect CD20, CD3, myeloid/histiocyte antigen (MAC387), IBA-1, and CD204 surface receptors. A roan polyclonal anti-Theileria sp. (sable) antibody was applied to the same tissues to identify intraleukocytic parasite antigens. Similar proportions of intravascular and extravascular IBA-1-, CD204-, and MAC387-reactive putative monocyte-macrophages and fewer CD3-positive putative T-lymphocytes were identified in all organs, especially the lungs in infected roan. CD20-positive putative B-lymphocytes were significantly scarcer than in uninfected controls. Intraleukocytic Theileria parasites labeled consistently in affected tissues. Some parasitized and nonparasitized MLs and the MNGCs failed to label with selected leukocyte markers. Fatal theileriosis in roans may largely be the result of multi-organ monocyte-macrophage activation with associated tissue injury and overwhelming systemic inflammation. The identity of the parasitized leukocytes and characteristics of the lymphohistiocytic response require further clarification in roans.
Theileria is a genus comprising tick-transmitted parasitic protozoa. 57 The taxonomy of the genus at species level has always been complex, and the nomenclature of many recognizable and economically important genotypes is still unresolved. 45 The most well understood of the many species of Theileria that infect domestic livestock are Theileria parva, the causal agent of East Coast fever (ECF) in cattle in eastern and southern Africa, 43 and Theileria annulata, the cause of tropical theileriosis (TT) in cattle in North Africa, southern Europe, and Asia. 40 A number of other named and unnamed species cause significant disease in cattle, sheep, goats, and wild ungulates in Africa, Asia, and Australasia.14,39,41,42,55
Theileriosis in hippotraginid antelope (roan and sable antelope) has been recognized as an important disease that can cause significant mortality in sub-Saharan Africa since 1974.82,83 The causal agent has not been named specifically and is currently known as Theileria sp. (sable) (and possibly Theileria sp. (sable-like)).45,55,59,71,72 Even though Theileria sp. (sable)-infected antelope do not necessarily develop theileriosis, in roans, the disease seems to follow a similar disease trajectory profile to theileriosis in domestic ruminants. 15 The pathogenesis of fatal disease in African wild artiodactyls is not understood, mainly due to the paucity and qualitative nature of the available pathology data (reviewed in Clift et al. 14 ). For instance, the leukocyte tropism of theilerial parasites is not known in wildlife, preventing the identification of disease mechanisms.
Traditionally, Theileria species and genotypes are classified as pathogenic or schizont-“transforming” (whereby parasitic schizonts induce reversible transformation of vertebrate host leukocytes) or benign/non- “transforming” (whereby leukocyte transformation is negligible, but anemia may occur due to piroplasm infection of host erythrocytes).45,80 Although wildlife are frequently asymptomatic carriers of Theileria parasites,7,55,78 stressed and young, immunologically naive wild ungulates are more susceptible to fatal infection.14,32,71,82,84
Theilerial organisms have a complex 2-host life cycle.14,57,66 Extrapolations from ultrastructural studies of T. parva and T. annulata in cattle suggest that the parasites undergo sexual reproduction with the formation of zygotes in the midgut of ticks, followed by asexual sporogony to form infective sporozoites in specialized salivary gland epithelium.57,66 After transmission of sporozoites to mammalian hosts during tick engorgement, asexual reproduction (schizogony and merogony) occurs in mammalian host leukocytes, followed by merozoite infection of erythrocytes to form piroplasms. 66
In mammalian hosts, pathogenic Theileria spp. infect specific MLs, 14 altering their transcriptome and resulting in pseudoneoplastic (reversible, nonmutagenic, incomplete) transformation.21,33,74 This is frequently associated with marked leukocyte atypia,6,14,15,20,81 uncontrolled leukocyte proliferation, multi-organ dissemination, and massive leukocytolysis.14,43,50 Lesions associated with the schizont- “transforming” Theileria spp. typically cause severe systemic disease with consequent high morbidity and mortality rates.1,14
Gross findings that characterize acute fatal theileriosis in domesticated and wild ruminants include the following: carcass pallor (suggestive of anemia), icterus; hemoglobinuria and/or bilirubinuria; widespread petechiae and ecchymoses; edematous, firm, rubbery lungs with a mottled appearance suggestive of acute interstitial pneumonia; watery effusions into body cavities; and cream-colored enlargement, thickening, or mottling of lymphoid and nonlymphoid organs due to leukocyte infiltration which may be associated with foci of parenchymal necrosis and/or mucosal ulceration.14,40,43,55
Typical histologic lesions include intravascular and extravascular accumulations of parasitized and nonparasitized heterogeneous MLs, with associated foci of hemorrhage, necrosis, edema, vasculitis, and thromboembolism.14,24,25,29 Parasitized and nonparasitized MNGCs 14 and enlarged leukocytes with convoluted and multilobulated nuclei 32 have been reported occasionally in wild artiodactyls with fatal theileriosis, but they are not described in fatally infected domestic livestock.
Immunohistochemical analysis of leukocyte surface receptors has been performed in formalin-fixed, paraffin-embedded (FFPE) tissues from cattle with ECF,25,37 TT, 6 and, to a limited extent, in T. taurotragi-associated cerebral theileriosis. 4 Multi-organ intravascular and extravascular leukocytic infiltrates are comprised of monocyte-macrophages and T-lymphocytes, with scarce B-lymphocytes reported.6,25 In experimentally infected cattle with lethal ECF, lymphoblastoid cells were parasitized,25,35 while in calves naturally 6 and experimentally 24 infected with TT, macrophages were parasitized. This mimics the leukocyte tropism of T. parva and T. annulata that has been shown in cell lines infected in vitro.2,8,11,23,70 Most recently, peripheral blood monocytes were subclassified using flow cytometry and their functions were compared in cattle with lethal versus nonlethal ECF. 3 Dysregulated monocyte activation with subsequent impairment of an effective adaptive immune response, and stimulation of proinflammatory and anti-inflammatory pathways seems to characterize lethal theileriosis in cattle.3,25,40
Data on the pathology of theileriosis in hippotraginids are scarce, despite the fact that the disease has a significant negative impact on the success of breeding programs for endangered roan antelope, and the successful introduction of naive animals into Theileria-endemic areas.59,71 In our study, we aimed to describe and analyze the gross and microscopic findings that typify acute, fatal theileriosis in roan antelope. This is the first report of the immunophenotype of the multi-organ leukocytic infiltrates in roan (or other wild ungulate tissues), in an attempt to elucidate the disease mechanisms of severe theileriosis. In addition, we compared our findings with those reported in domestic livestock with lethal theileriosis.
Materials and Methods
Case Selection
Gross and histologic data pertaining to roan antelope that died of acute natural theilerial infection were collected retrospectively from the archives at the Section of Pathology, Department of Paraclinical Sciences (DPS), Faculty of Veterinary Science (FVS), and University of Pretoria (UP) in South Africa. Fifty-five cases with sufficient data were found between 1996 and 2019. There were 20 females, 14 males, and 21 animals with no gender recorded. Ages ranged from 1 day to 5.5 years (mean = 125.42 days; median = 21 days). Gross and histologic data were available for 40 and 34 cases, respectively. Nineteen cases had both gross and histologic data. We selected 16 cases for immunohistochemistry (IHC) to determine schizont and leukocyte immunoreactivity, 11 of which had gross and histologic data and 5 that only had histologic data. Case selection for IHC was based on the availability of tissues that were (1) only mildly autolyzed and (2) most consistently infiltrated with parasitized and nonparasitized leukocytes. No cases with documented concurrent bacterial infections were used for IHC. The work was done with the approval of the UP Animal Ethics Committee (certificate V028-18) and the Department of Agriculture, Land Reform and Rural Development (permit references 12/11/1/1/5 and 12/11/1/1/8).
The diagnosis of theileriosis was based on (1) the observation of schizont-infected MLs and intra-erythrocytic piroplasms in blood smears and, rarely, lymph node and/or spleen impression smears and (2) typical gross and histologic findings and lesions, including the observation of intraleukocytic theilerial parasites (ILP) in hematoxylin and eosin (HE)-stained tissue sections. For 7 of 55 cases, reverse line blot (RLB) hybridization analyses were done on amplified PCR products for the simultaneous detection of Theileria, Babesia, Anaplasma, and Ehrlichia spp., as previously described.54,55 For all 7 cases, infection with at least Theileria sp. (sable) was confirmed. Three animals were coinfected with T. separata and 1 of these 3 was also infected with T. buffeli.
Gross Pathology
Data were tallied from postmortem reports pertaining to 40 roans. Based on descriptions of body condition at the time of death, animals were assigned to 1 of 3 categories: (1) cachexia (moderate-to-severe muscle mass atrophy and serous atrophy of fat), (2) poor condition (imperceptible to mild muscle mass atrophy with depletion of fat reserves, but no evidence of serous atrophy), and (3) normal to good condition (no depletion of muscle mass and adequate fat reserves). The following findings and lesions were scored 0 if absent and 1 if present: watery blood with mucous membrane and carcass pallor (suggestive of anemia), congestion, icterus, hemorrhages (petechiae, ecchymoses, and/or suffusive hemorrhages on serosae, mucosae, parenchymal organs, and/or in subcutaneous tissues), edema (with location), effusions into body cavities, pulmonary edema, interstitial pneumonia, lymphadenomegaly due to parenchymal proliferation and/or edema, splenomegaly due to red and/or white pulp hyperplasia, hepatomegaly with/without cholestasis, myocarditis, myocardial degeneration/necrosis, ulcerative gastroenteritis, proliferative gastroenteritis/gastroenteropathy, hemorrhagic gastroenteritis/gastroenteric hemorrhage, nephrosis (acute tubular injury), interstitial nephritis, renal infarcts, suspected hemoglobinuria, and bilirubinuria. Additional findings and lesions were also recorded.
Histologic Lesions
Tissue sections were fixed in 10% neutral buffered formalin, trimmed, routinely processed, stained with HE, and examined with a light microscope. Available tissues included liver (n = 34), lung (n = 33), spleen (n = 32), heart (n = 29), kidney (n = 29), lymph node (n = 20), gastrointestinal tract (GIT; n = 15), skeletal muscle (n = 12), adrenal gland (n = 11), brain (n = 11), bone marrow (n = 8), thymus (n = 5), thyroid gland (n = 3), eye (n = 2), salivary gland (n = 2), and uterus (n = 1). To ensure selection of tissues that were no more than mildly autolyzed for IHC, autolysis was graded per tissue as mild or moderate depending on tissue integrity and adequacy of HE staining. In mildly autolyzed samples, tissues were well stained, tissue architecture was intact and cells were minimally changed; erythrocytes were still intact. In moderately autolyzed specimens, tissues were architecturally intact but stained paler than normal with HE. Cells were variably swollen and vacuolated (unrelated to a pathological process) with associated autolytic nuclear changes; erythrocytes were often lyzed. Severely autolyzed specimens were automatically excluded from the evaluation.
We specifically evaluated the samples for the following cells and lesions (which we scored 0 if absent and 1 if present): polymorphonuclear leukocytes (neutrophils, eosinophils, basophils), MLs (lymphocytes, monocyte-macrophages), MNGCs (including multilobulated leukocytes), ILP/schizonts, necrotizing vasculitis, thromboembolism, parenchymal necrosis, ulceration, fibrin exudation, congestion, hemorrhage, erythrophagocytosis, edema, cholestasis, and leukocytolysis. Our category of MNGCs included cells with at least 2 distinct nuclei as well as those with at least 2 nuclear lobes because multilobulated leukocytes were often hard to distinguish from MNGCs, unless the nuclei were obviously distinct.
IHC
The preferred tissues were not available for all 5 leukocyte markers for each of the 16 cases selected for IHC. Ultimately, the samples comprised: liver (n = 14–16), lung (n = 15–16), kidney (n = 13–15), and heart (n = 11–13). We applied sequential indirect chromogenic immunoperoxidase assays to (1) detect ILP and (2) phenotype the multi-organ leukocyte infiltrates. For the former, IgG was purified from Theileria sp. (sable)-infected roan sera. These were previously characterized to react with ILP proteins from Theileria sp. (sable) lymph node-derived subcultures in western blots, and in FFPE tissues from infected antelope using IHC. 16 Here, we used antibody B, 1 of 3 purified roan antibodies that consistently detected leukocyte-associated theilerial antigens in roan and sable tissues. 16 Furthermore, this assay could detect ILP antigens in tissues from diverse wild artiodactyls and did not cross-react with potentially confusing nontheilerial organisms or leukocytes in lesions that resemble theileriosis histologically. 16 For immunophenotypic characterization of the leukocyte infiltrates, we used commercially available antibodies to detect the following leukocyte antigens: CD20, CD3, MAC387, IBA-1, and CD204. Details of the primary antibodies used, their target cells, optimal heat-induced epitope retrieval (HIER) methods, antibody dilutions and incubation times, chromogens used, and positive tissue controls are summarized in Table 1.
List of antibodies and immunohistochemical protocols used in tissues from roan antelope with lethal theileriosis.
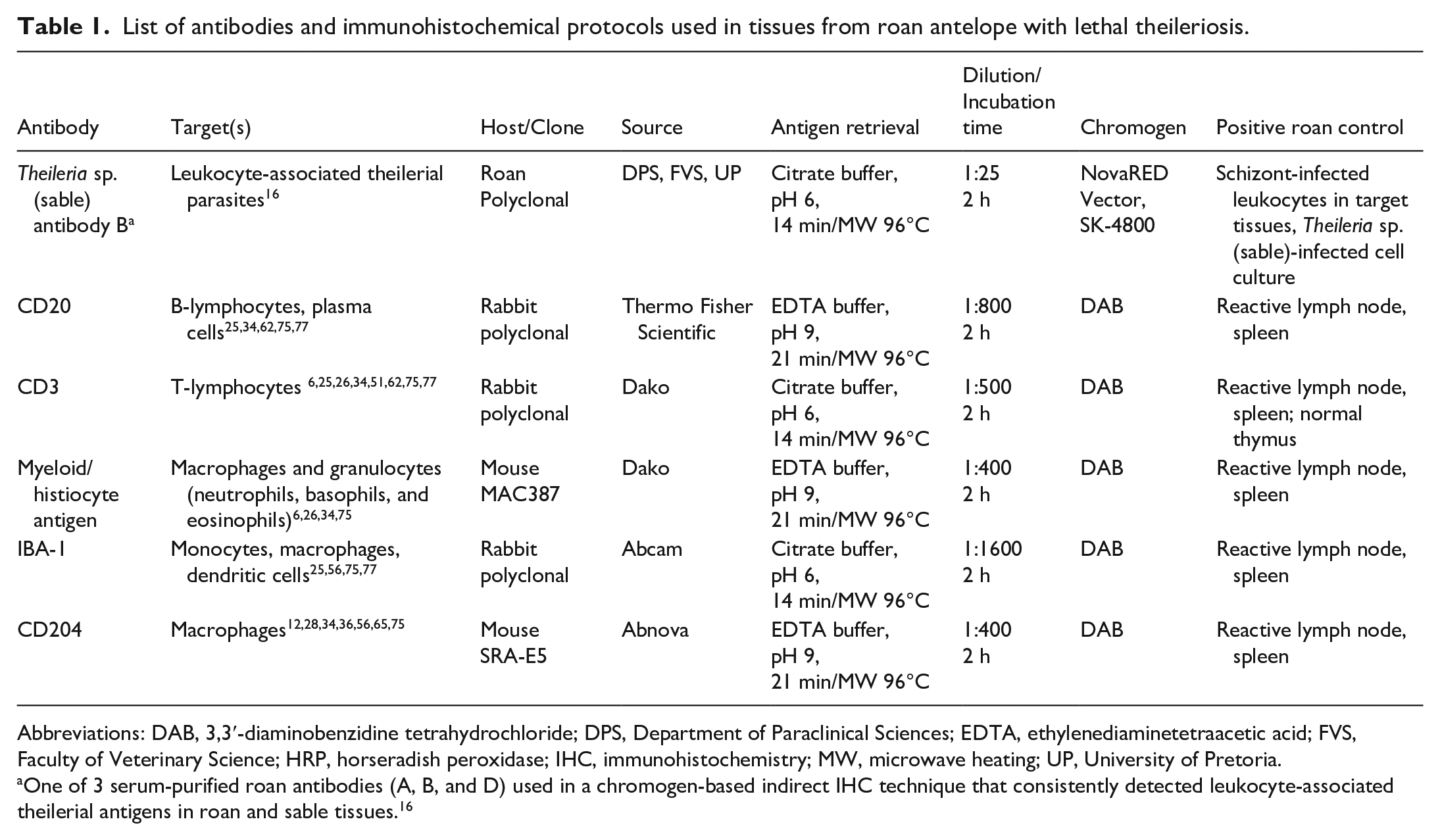
Abbreviations: DAB, 3,3′-diaminobenzidine tetrahydrochloride; DPS, Department of Paraclinical Sciences; EDTA, ethylenediaminetetraacetic acid; FVS, Faculty of Veterinary Science; HRP, horseradish peroxidase; IHC, immunohistochemistry; MW, microwave heating; UP, University of Pretoria.
One of 3 serum-purified roan antibodies (A, B, and D) used in a chromogen-based indirect IHC technique that consistently detected leukocyte-associated theilerial antigens in roan and sable tissues. 16
Briefly, serial sections of 4 μm were cut from paraffin blocks, mounted on positively charged microscope slides (Superfrost Plus, Menzel Gläser), routinely deparaffinized in xylene, and rehydrated through graded ethanol and distilled water. To quench endogenous peroxidase activity, sections were treated with 3% hydrogen peroxide in methanol for 15 minutes at room temperature (RT). Tissues were microwave-heated to 96°C in citrate buffer (pH 6) for 14 minutes or ethylenediaminetetraacetic acid (EDTA) buffer (pH 9) for 21 minutes. Thereafter, the primary antibodies were incubated with the tissue sections for 2 hours at RT. Staining for ILP antigens required some extra steps. 16 Before the application of the anti-Theileria sp. (sable) antibody B, tissues were treated with a 2% solution of skimmed milk powder in distilled water for 15 minutes at RT in order to block nonspecific background staining. 9 After incubation with the anti-Theileria sp. (sable) antibody B, tissue sections were treated with a universal antibody detection reagent (Pierce peroxidase-conjugated recombinant protein A/G, Thermo Scientific) for 30 minutes at RT. A polymer-based detection system (ab236466, Abcam) was applied to all the tissue sections, according to the manufacturer’s instructions. Immunoreactivity was visualized using a 3,3′-diaminobenzidine (DAB) or NovaRED substrate, followed by counterstaining with Mayer’s hematoxylin for 10–20 seconds, rinsing in running tap water for 10 minutes and routine dehydration through increasing alcohol concentrations and xylene. Slides were mounted with entellan (Thermo Scientific) and coverslipped.
Theileria spp. positive and negative roan tissue controls were stained with every IHC run. Negative tissue controls comprised histologically normal lung, liver, kidney, and heart samples from 2 roans that died due to trauma and hypothermia, respectively. Positive tissue controls were 2-fold. 16 We used formalin-fixed cytospins derived from Theileria sp. (sable)-infected roan leukocytes in continuous culture. 85 We also used FFPE lung, liver, kidney, and heart samples from a fatally infected roan antelope with evidence of parasites in tissue smears, characteristic gross and histologic lesions (with clearly discernible ILP), and PCR-RLB confirmation of Theileria sp. (sable) infection. Specific positive labeling of theilerial antigens was seen as finely stippled red-brown granules in the paranuclear cytoplasm of MLs and MNGCs (Supplemental Figures S1–S2). Conversely, no Theileria-specific positive labeling was identified in the negative tissue controls (Supplemental Figure S3).
We ran validated positive dog tissue controls (lymph node and spleen) together with selected positive roan tissue controls (lymph node and spleen) to demonstrate specific labeling of primary antibodies to selected leukocyte antigens. The positive roan tissue controls were obtained from 3 variously aged roans that died due to bronchointerstitial pneumonia, suspected pyrrolizidine alkaloid toxicity, and hepatorenal microvascular malformation, respectively. None of these roans had conditions or lesions compatible with theileriosis, no hemoparasites were observed in their blood smears, and no Theileria-specific immunoreactivity was detected in FFPE tissues. Labeling of leukocyte antigens was considered specific if the immunoreactive cells were visualized in the expected location in roan-positive tissue controls. Membranous staining was considered CD20-positive, while CD3 positivity was membranous or cytoplasmic (Supplemental Figures S4–S5). Plasma cells labeled CD20-positive in normal roan lymphoid tissues. Nuclear and cytoplasmic staining was considered MAC387-positive (Supplemental Figure S6). IBA-1 immunoreactivity was predominantly membranous and cytoplasmic (Supplemental Figure S7), while CD204-specific labeling was typically cytoplasmic, and only occasionally membranous (Supplemental Figure S8).
In preparation for the quantification and scoring of immunoreactivity in tissues from infected and uninfected roans, whole slides were digitally scanned in bright field at 20× magnification using an Olympus VS120 digital scanner (Wirsam Scientific). Tissues were scored 0 if there was no specific labeling or 1 if there was specific labeling of ILP antigens in association with typical lesions. Subsequently, semiautomatic leukocyte counts were performed using QuPath-0.2-3 open-source digital image analysis software. Where possible, 100 contiguous IHC-labeled and nonlabeled leukocyte-like cells were marked digitally and counted in each of 3 histologic fields per organ. Earnest attempts were made not to count parenchymal, endothelial, and obviously nonhematopoietic connective tissue cells. Only nucleated leukocyte-like cells were counted. Fields that were most populated with immunoreactive cells (so-called “hot spot” areas)30,52,79 were selected subjectively by an observer blinded to the treatment (BMG). Cells were counted from left to right and top to bottom in adjacent grid squares placed over the middle of the “hot spots” until 100 cells were counted. Thus, 300 leukocyte-like cells were counted per tissue for each fatally infected roan. Staining intensity was not scored. Given that very few leukocytes were observed in tissues from the control cases, as many cells as possible (usually far fewer than 100) were counted per “hot spot” per tissue. To obtain a more accurate estimate, the leukocyte-like cells counted in all 3 “hot spots” for each available tissue per antibody were added together. The proportion of immunoreactive cells for each antibody and tissue was defined as [the number of positive leukocytes/total leukocytes counted] x 100.
The location of immunoreactive leukocytes was scored for all antibodies across all tissues as follows: 0 = intravascular; 1 = extravascular (including perivascular, interstitial, and pulmonary intra-alveolar compartments); and 2 = intravascular and extravascular. Labeled leukocyte-like cells were rarely observed within vascular walls, so this category was excluded.
Statistical Analysis
For each leukocyte marker (CD20, CD3, MAC387, IBA-1, and CD204) and tissue (liver, lung, kidney, and heart), we compared the proportions of immunoreactive cells in infected and uninfected roans using Wilcoxon rank sum tests in R 3.5.0 60 (1-tailed test; the Benjamini-Hochberg procedure was used to keep the false discovery rate over the 20 tests at an α = 0.05). Within the infected (positive) cases, we compared the 5 immune cell markers within each tissue to see whether the markers differed significantly from each other using paired Wilcoxon rank sum tests in R 3.5.0 60 (2-tailed test; the Benjamini-Hochberg procedure was used to keep the false discovery rate over the 40 tests at an α = 0.05).
To see whether the location of immunoreactive leukocytes was independent of the tissue and leukocyte marker, we fitted a generalized linear model with quasi-Poisson errors to counts of leukocyte location categories (0 = intravascular, 1 = extravascular, and 2 = intravascular and extravascular) per marker and tissue. The saturated model was count~marker*location*tissue. Terms were dropped starting from higher order interactions using an F-test. 17
Data Availability
The data analyzed in this study are available on Figshare doi:10.25403/UPresearchdata.14601663.
Results
Gross Pathology
Body condition at the time of death was not recorded for 22/40 roans (55%); however, in 13/18 cases, the body condition could be graded as normal to good. The remaining 5 cases included 3 cachectic animals and 2 in poor condition.
The frequency of reported gross pathology findings and lesions was highly variable in the carcasses. The most commonly reported findings and lesions in at least 50% of roans in this cohort are summarized in Fig. 1. From most to least common, findings and lesions included widespread petechiae and ecchymoses, especially on serosae (29/40 cases (72.5%); Figs. 1, 2a, b); watery blood and mucous membrane pallor suggestive of anemia (27/40 cases (67.5%); Figs. 1, 2c); icterus (24/40 cases (60%); Figs. 1, 2a); splenomegaly (24/40 cases (60%); Figs. 1, 2a); hepatomegaly (21/40 cases (52.5%); Fig. 1); and pulmonary edema (20/40 cases (50%); Figs. 1, 2d).
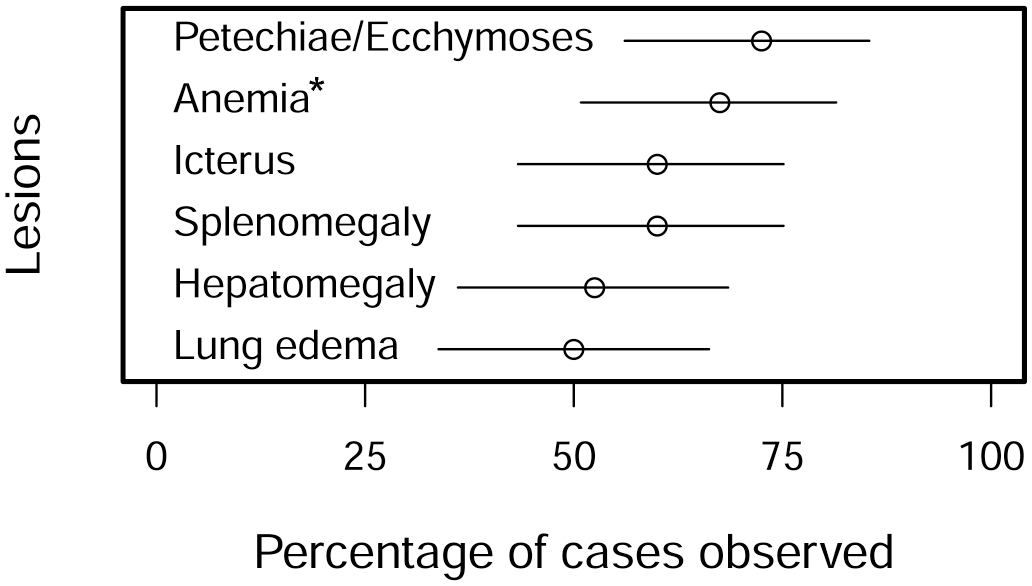
Frequencies of the most commonly reported gross findings in 40 roan antelope (Hippotragus equinus) with fatal theileriosis. The estimate is indicated by the empty circle and the 95% binomial confidence interval (CI) by the line. *Anemia was indicated by carcass pallor and watery blood.
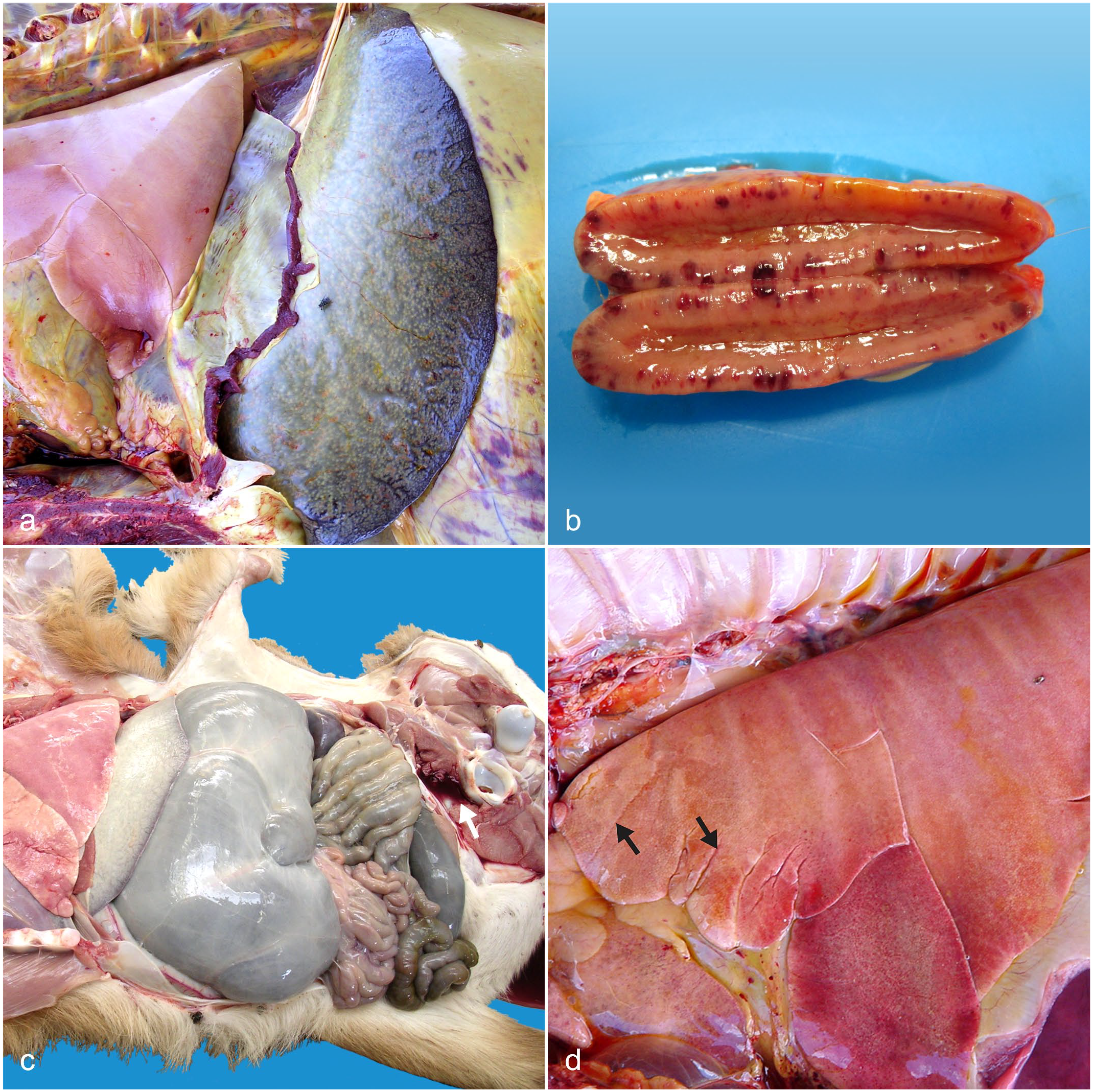
Fatal theileriosis, roan antelope (Hippotragus equinus) (a) Case 5. Icterus, pulmonary edema, and splenomegaly. Petechiae, ecchymoses, and suffusive hemorrhages are visible on serosae (b) Adrenal gland, case 51. Adrenocorticomegaly due to widespread leukocyte infiltration with associated multifocal to coalescing petechiae and ecchymoses (c) Case 4. Anemia (as indicated by watery blood and mucous membrane as well as carcass pallor), unclotted blood (arrow), and mild ruminal impaction. Body condition was poor (d) Lung, case 8. Interstitial pneumonia with diffusely noncollapsed lungs and rib impressions evident on the visceral pleura. Mild interstitial edema is evident in the cranioventral aspect of the left cranial lung lobe (arrows). There are multifocal petechiae and ecchymoses on the parietal pericardium.
Less common lesions included generalized lymphadenomegaly due to parenchymal proliferation (17/40 cases (42.5%)), interstitial pneumonia (12/40 cases (30%); Fig. 2d), and serosanguinous effusions into body cavities (8/40 cases (20%)). Rare lesions included suffusive hemorrhages, especially on serosae and in subcutaneous tissues (5/40 cases (12.5%)), which always occurred with petechiae and ecchymoses; bilirubinuria (5/40 cases (12.5%)); lymph node edema (4/40 cases (10%)); suspected hemoglobinuria (3/40 cases (7.5%)); nephrosis/acute tubular injury (3/40 cases (7.5%)); interstitial nephritis (3/40 cases (7.5%)); generalized carcass congestion (2/40 cases (5%)); anasarca (2/40 cases (5%)), and enteric hemorrhage (2/40 cases (5%)). Four of the 40 animals also had lesions that were obviously associated with bacterial infections including necrotizing hepatitis due to Fusobacterium necrophorum (1 case), limb fracture and septicemia (1 case), and fibrinous enterocolitis with extensive fecal soiling (2 cases), both with positive microbiological cultures for Salmonella spp. (including 1 S. enterica subsp. enterica).
Histologic Lesions
The most commonly observed lesions and the frequency with which they occurred in the available organs are depicted in Fig. 3. In summary, infiltrates of often atypical MLs, both intravascular and extravascular, were observed in multiple organs. Atypical MLs varied in size and had scarce to moderate eosinophilic and occasionally amphophilic cytoplasm. They possessed round to oval, angular, and often irregularly indented or convoluted nuclei, typically with coarsely granular chromatin and variably conspicuous single or multiple nucleoli (Fig. 4). Some, but not all, were parasitized. Both parasitized and nonparasitized MNGCs, in which we included enlarged leukocytes with multilobulated nuclei as it was often difficult to distinguish between them (Fig. 3), were also present in smaller numbers, predominantly in sinusoidal capillaries and the extravascular compartment. Although scattered small lymphocytes were quite commonly seen in multiple tissues, typical polymorphonuclear leukocytes were scarce (usually associated with foci of parenchymal necrosis), as were plasma cells. Intravascular MLs were often aggregated/clumped and/or marginated in capillaries, sinusoids, and small- to medium-caliber veins and lymphatics (Fig. 4).
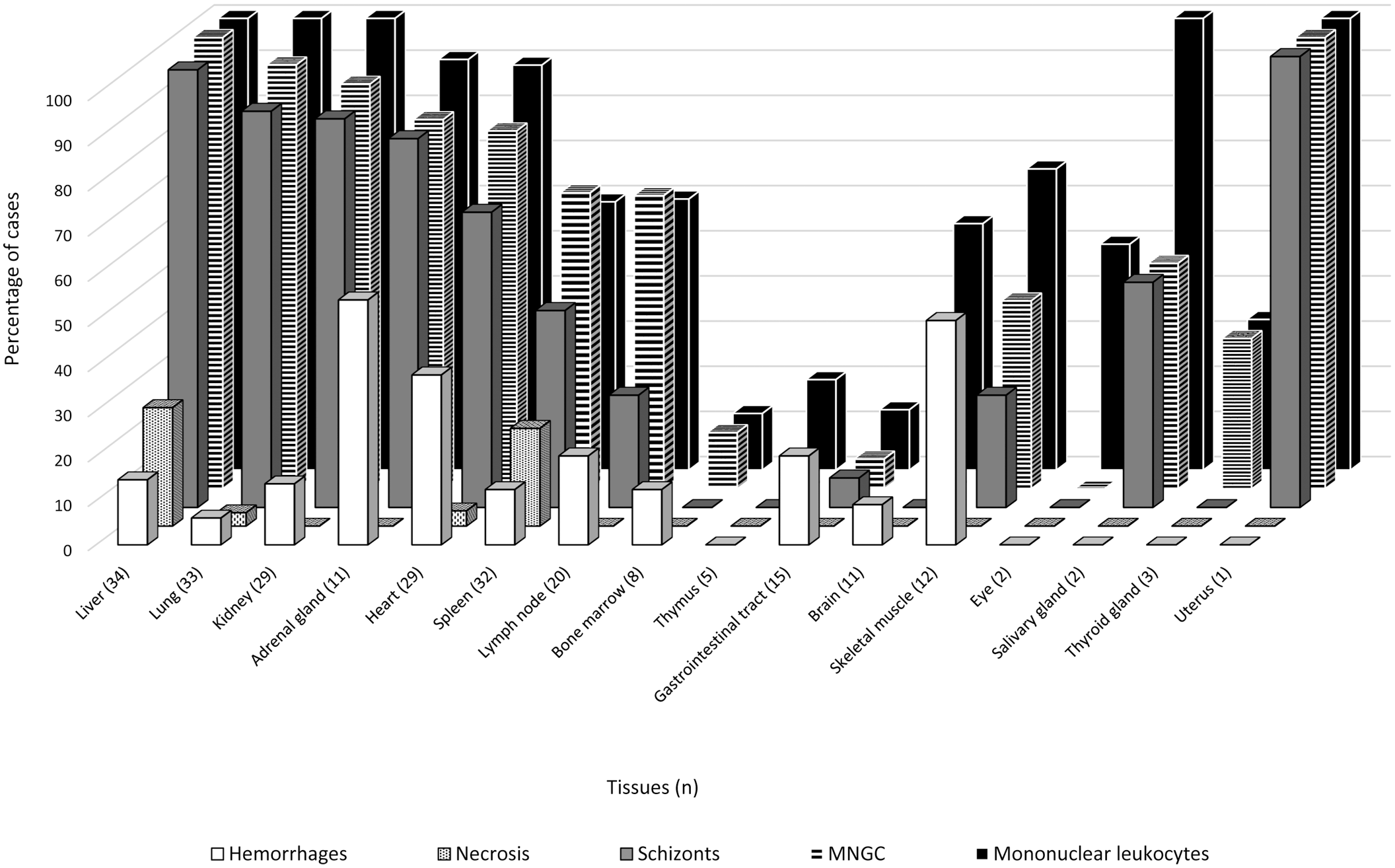
Frequencies of the most common histologic lesions in the tissues that were available for 34 roan antelope (Hippotragus equinus) with fatal theileriosis. MNGC refers to both MNGCs and multilobulated leukocytes that were indistinguishable in the absence of obviously distinct nuclei.
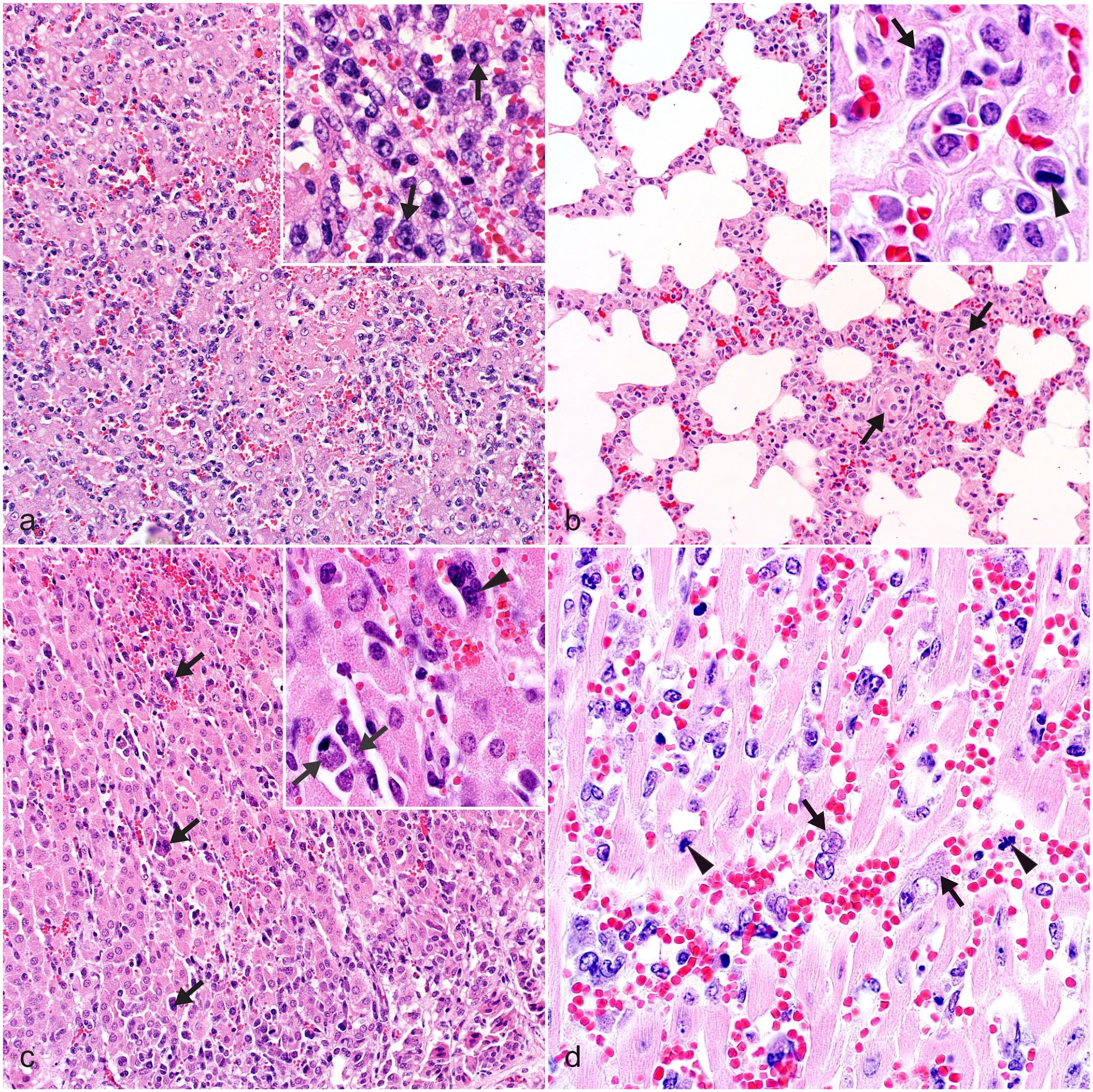
Fatal theileriosis, roan antelope (Hippotragus equinus). HE (a) Liver, case 9. There are multifocal areas of necrosis and hemorrhage. The sinusoids are diffusely congested and contain numerous MLs. Inset: higher magnification. Parasitized and nonparasitized atypical MLs possess irregularly indented nuclei with variably prominent nucleoli and scarce to moderate eosinophilic to amphophilic cytoplasm (arrows) (b) Lung, case 22. Alveolar septa are congested and diffusely thickened due to intravascular (arrows), perivascular and interstitial accumulations of MLs. Inset: higher magnification. Intracytoplasmic parasitic schizonts in an atypical mononuclear leukocyte with a hyperchromatic eccentrically situated nucleus (arrow), and a mitotic figure (arrowhead) (c) Adrenal gland, case 51. Multifocal cortical hemorrhages and numerous MLs with fewer multilobulated leukocytes and MNGCs (arrows) in sinusoidal capillaries. Inset: higher magnification. Intracytoplasmic parasitic schizonts in MLs (arrows) and a multinucleate giant cell (arrowhead) (d) Heart, case 27. Intravascular and extravascular infiltrates of frequently atypical MLs (with euchromatic nuclei and prominent nucleoli), hemorrhages, intracytoplasmic schizonts (arrows), and mitotic figures (arrowheads).
The 5 organs that were most consistently infiltrated with parasitized MLs and MNGCs included liver (34/34 ML; 34/34 MNGC; and 33/34 ILP; Figs. 3, 4a), lung (33/33 ML; 31/33 MNGC; and 29/33 ILP; Figs. 3, 4b), kidney (29/29 ML; 26/29 MNGC; and 25/29 ILP; Fig. 3), adrenal gland (10/11 ML; 9/11 MNGC; and 9/11 ILP; Figs. 3, 4c), and heart (26/29 ML; 23/29 MNGC; and 19/29 ILP; Figs. 3, 4d). A single uterine sample was infiltrated with parasitized ML and MNGC, while both salivary gland samples exhibited ML infiltrates, but only 1 of them also had MNGCs and identifiable ILP (Fig. 3). Less commonly infiltrated organs included spleen (19/32 ML; 21/32 MNGC; and 14/32 ILP), lymph node (12/20 ML; 13/20 MNGC; and 5/20 ILP), and skeletal muscle (8/12 ML; 5/12 MNGC; and 3/12 ILP). This was followed by the brain (6/11 ML; 0/11 MNGC; and 0/11 ILP), eye (1/2 ML; 0/2 MNGC; and 0/2 ILP), thyroid gland (1/3 ML; 1/3 MNGC; and 0/3 ILP), thymus (1/5 ML; 0/5 MNGC; and 0/5 ILP), bone marrow (1/8 ML; 1/8 MNGC; and 0/8 ILP), and GIT (2/15 ML; 1/15 MNGC; and 1/15 ILP), although many of the latter tissues were comparatively rarely sampled (Fig. 3).
The distribution of cellular infiltrates was focal, focally extensive, multifocal, or diffuse. Diffuse infiltrates were typically observed in the liver and lung. In the kidney, cellular infiltrates were observed in glomeruli as well as the cortical and medullary interstitium. In the spleen, infiltrates tended to expand remnant periarterial lymphatic sheaths (PALS) and the red pulp. Cellular infiltrates were usually more prominent in the cortex of adrenal glands, while in the brain specimens, MLs were typically observed within meningeal and, less frequently, parenchymal capillaries.
The cellular infiltrates were periodically associated with foci of hemorrhage (Figs. 3, 4a, c, d). Hemorrhages were observed most commonly in the adrenal glands (6/11 samples [54.5%]; Fig. 4c), followed by skeletal muscle (6/12 [50%]), heart (11/29 [38%]; Fig. 4d), lymph node (4/20 [20%]), liver (5/34 [15%]; Fig. 4a), kidney (4/29 [14%]), GIT (2/15 [13%], excluding the 2 cases with enteric salmonellosis), spleen (4/32 [12.5%]), bone marrow (1/8), brain (1/11 [9%]), and lung (2/33 [6%]). Foci of coagulative necrosis, often admixed with fibrin, were occasionally observed in the liver (9/34 samples [26%]; Fig. 4a) and spleen (7/32 [22%]), and, very rarely, in heart (1/29 [3%]) and lung (1/33 [3%]).
Organ-specific lesions were noted, as follows: mild-to-moderate alveolar and, to a lesser extent, interstitial pulmonary edema with increased alveolar macrophages in all cases; moderate-to-severe depletion of normal lymphoid (especially follicular) tissue in the spleen (28/32 samples (87.5%)) and lymph nodes (16/20 (80%)), with loss of distinction between the splenic red and white pulp and lymph node cortex and medulla, in areas of cellular infiltration. Mild-to-moderate nodular paracortical lymphoid hyperplasia was observed in lymph nodes (6/20 (30%)); leukocytolysis and hemosiderin-laden macrophages were seen in the spleen (8/32 (25%) and 6/32 (19%) samples, respectively), and lymph node (1/20 (5%) and 3/20 (15%) samples, respectively), and mild-to-moderate cholestasis was observed in the liver (17/34 samples (50%)).
IHC
Leukocyte-associated theilerial antigens labeled specifically with anti-Theileria sp. (sable) IHC in all lung (Fig. 5a), liver, kidney, and heart samples that exhibited typical lesions. Many more ILP were identified with IHC than in the HE-stained tissues, but intraerythrocytic parasites labeled inconsistently.
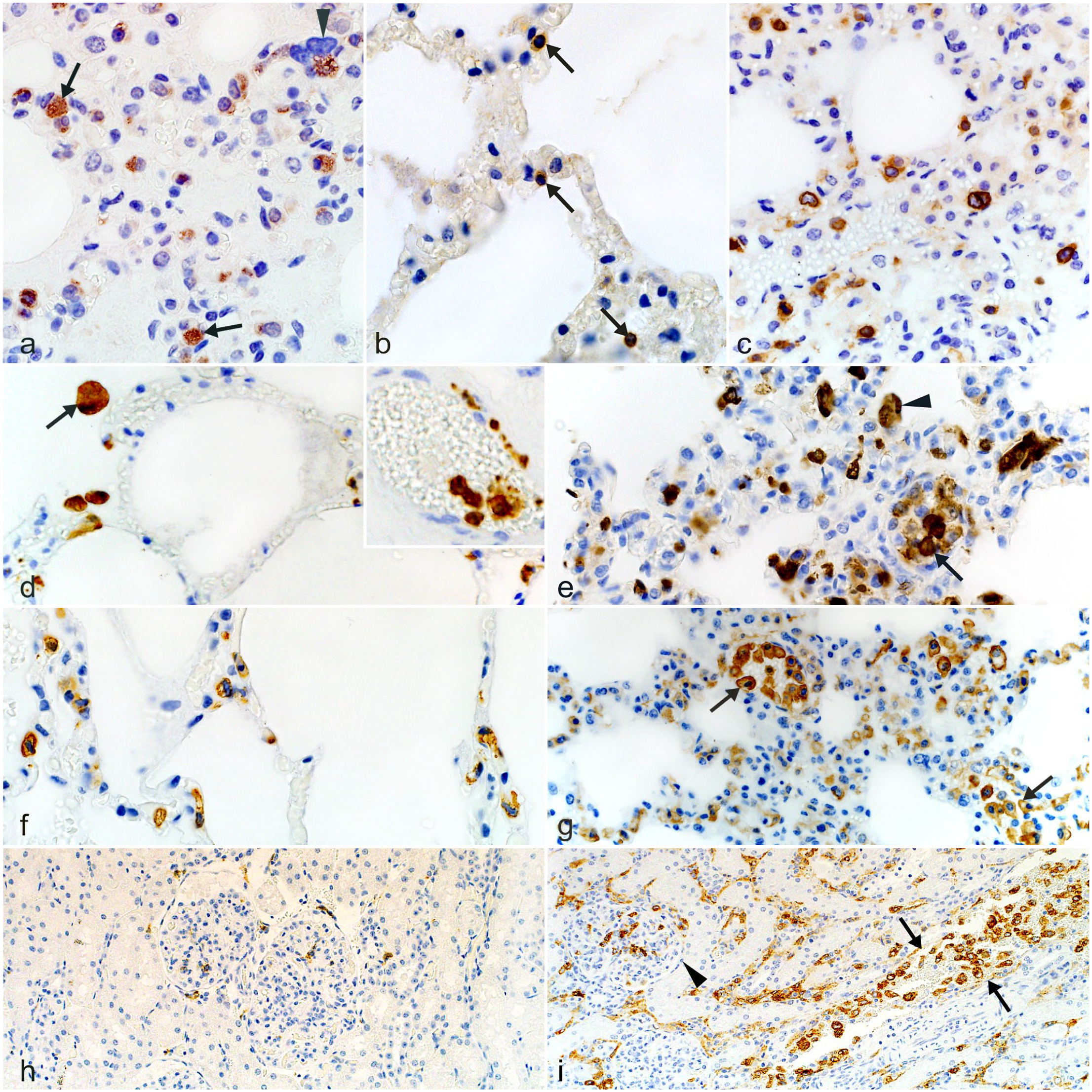
Fatal theileriosis, roan antelope (Hippotragus equinus). IHC (a) Lung, case 27. Immunoreactive parasitic schizonts in the cytoplasm of MLs (arrows) and a multinucleate giant cell (arrowhead) in the interalveolar septa. IHC for Theileria sp. (sable) (b) Lung, uninfected control. Sparse CD3-like immunoreactivity of MLs in the interalveolar septa (arrows). IHC for CD3 (c) Lung, case 27. CD3-like immunoreactivity is significantly increased in the interalveolar septa of the infected animal. IHC for CD3 (d) Lung, uninfected control. MAC387-reactive leukocytes are visible in alveolar lumens and the interalveolar septa. Inset: Intravascular MAC387-positive leukocytes in a different area of the same tissue. IHC for MAC387 (e) Lung, case 39. MAC387-positive intravascular (arrow), interstitial and intraalveolar (arrowhead) mononuclear leukocyte-type cells are significantly increased in the infected animal. IHC for MAC387 (f) Lung, uninfected control. IBA-1-reactive MLs are visible in the interalveolar septa. IHC for IBA-1 (g) Lung, case 9. IBA-1-like immunoreactivity is significantly increased in the infected animal. Aggregates of positively labeled cells are visible in the intravascular compartment (arrows). IHC for IBA-1 (h) Kidney, uninfected control. Singly scattered CD204-positive MLs are visible in the glomeruli and intertubular interstitium. IHC for CD204 (i) Kidney, case 22. CD204-reactive MLs are significantly increased in the glomeruli (arrowhead) and intertubular interstitium, including within the vasculature (arrows), in the infected animal. IHC for CD204.
The proportions of ML-type cells that labeled with specific leukocyte markers in tissues from fatally infected roans compared to uninfected controls are summarized in Table 2. Compared to controls, CD20-positive putative B-lymphocytes were significantly decreased in the liver and lung samples, with nonsignificant decreases seen in kidney and heart tissues. On the other hand, CD3-positive putative T-lymphocytes and MAC387-, IBA-1-, and CD204-positive monocyte-macrophage-like MLs occurred more commonly in tissues from infected roans (Fig. 5b–i). CD3-, MAC387-, IBA-1-, and CD204-positive cells were all significantly elevated in the lung samples (Fig. 5b–g), while CD3-, IBA-1-, and CD204-positive leukocytes were significantly increased in the kidneys (Fig. 5h, i).
Percentage of leukocytes that labeled with each marker per tissue for the control roan (Hippotragus equinus) cases (n = 2) and the roans with fatal theileriosis (11 ≤ n ≤ 16). Values that were significantly decreased or increased between cases and controls are underlined or bolded, respectively.
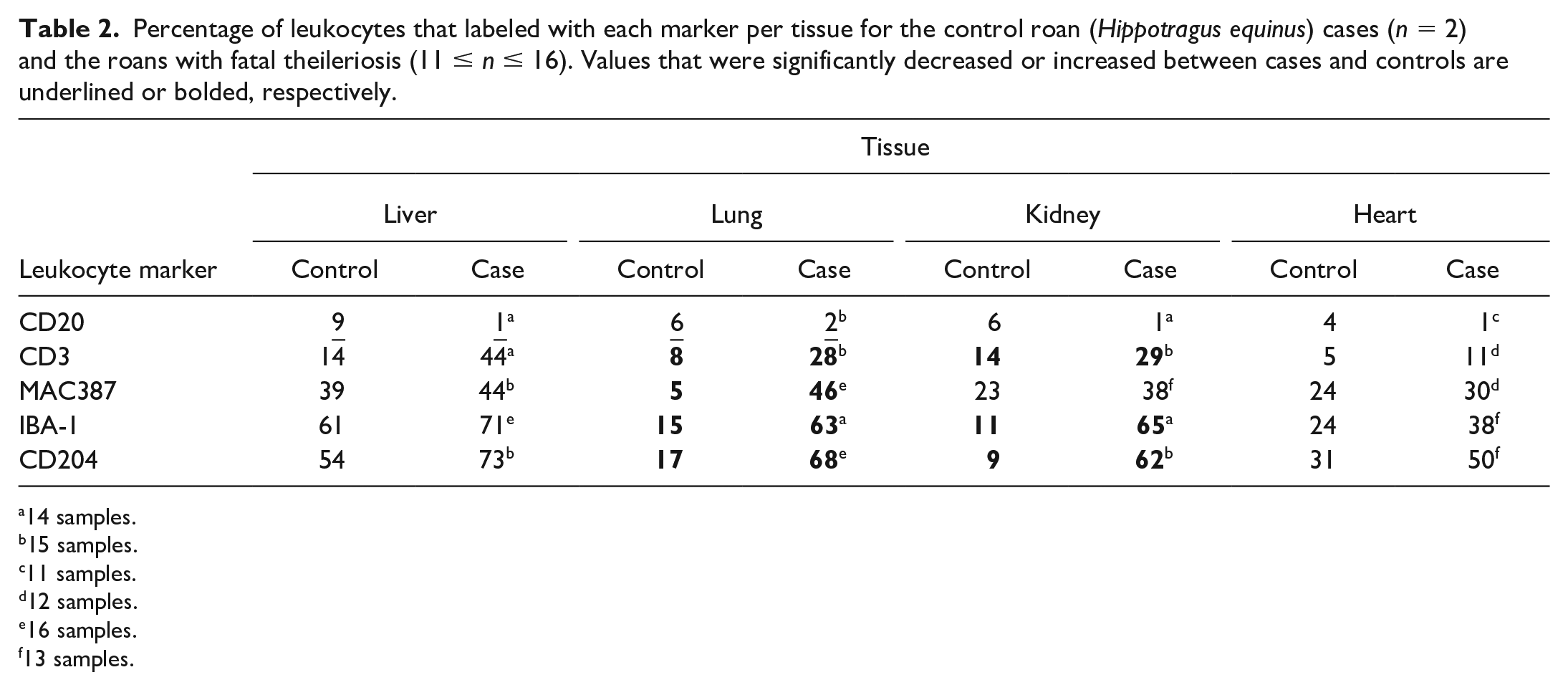
14 samples.
15 samples.
11 samples.
12 samples.
16 samples.
13 samples.
The rank order with which the different markers labeled MLs across the selected tissues was virtually identical (Fig. 6). This indicates that the cellular infiltrates had a similar composition, irrespective of the tissue type. Monocyte-macrophage-like cells (expressing IBA-1 and CD204) and possibly also dendritic-type cells (expressing IBA-1) predominated, followed by MAC387-reactive monocyte-macrophage-like cells, then putative T-lymphocytes (expressing CD3). In all tissues, some MLs and the MNGCs failed to label with the selected markers.
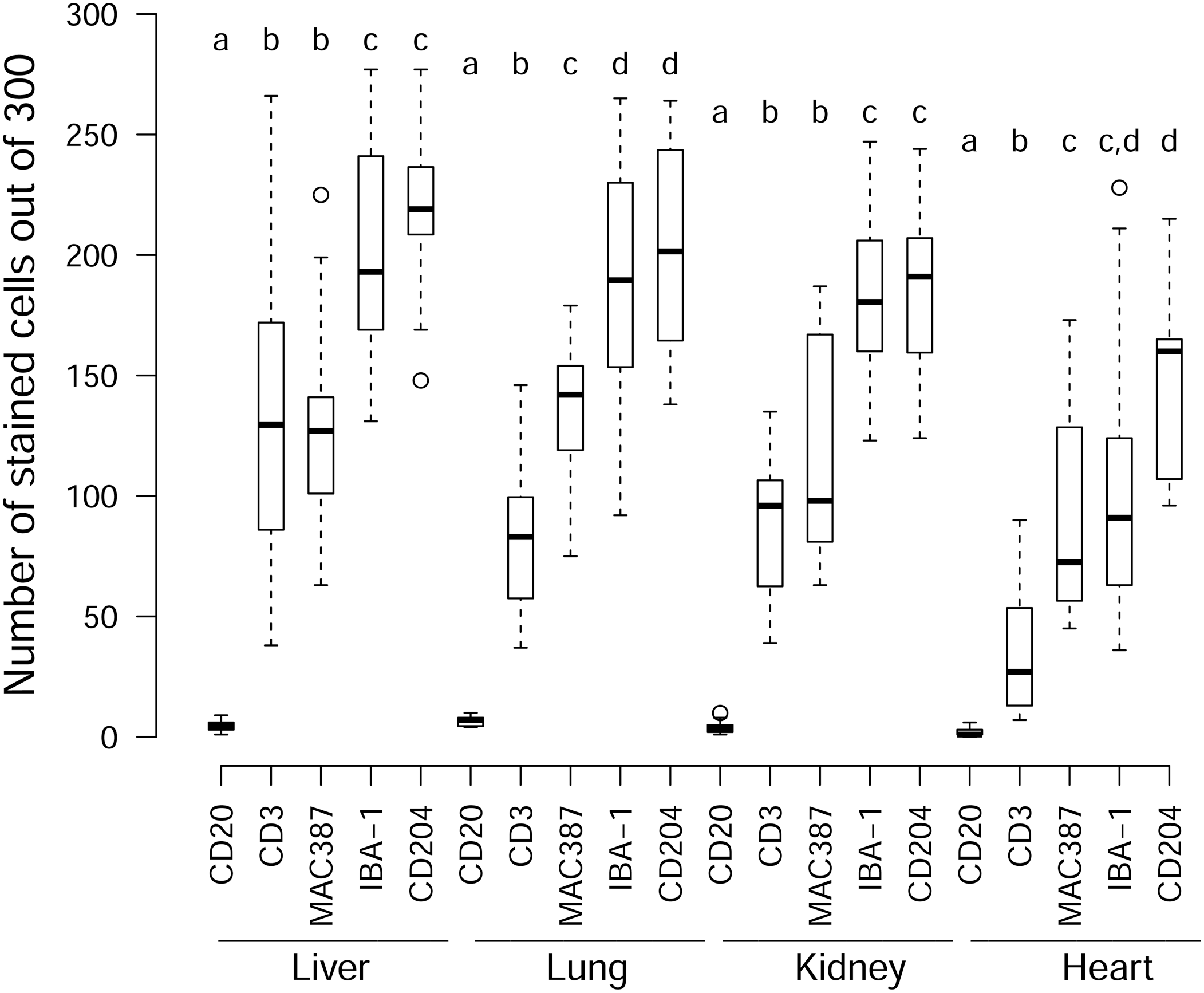
Box and whisker plots of the number of immunoreactive MLs (out of 300) for each tissue per marker. Within each tissue, letters above the boxes indicate if markers are significantly different from one another (different letters) or not (same letter). For example, in the liver, CD20 is significantly different from all the other markers, CD3 is significantly different from CD20, IBA-1, and CD204, but not from MAC387 and while IBA-1 and CD204 are not significantly different from each other, they are significantly different from the other markers. No comparisons were made between tissues.
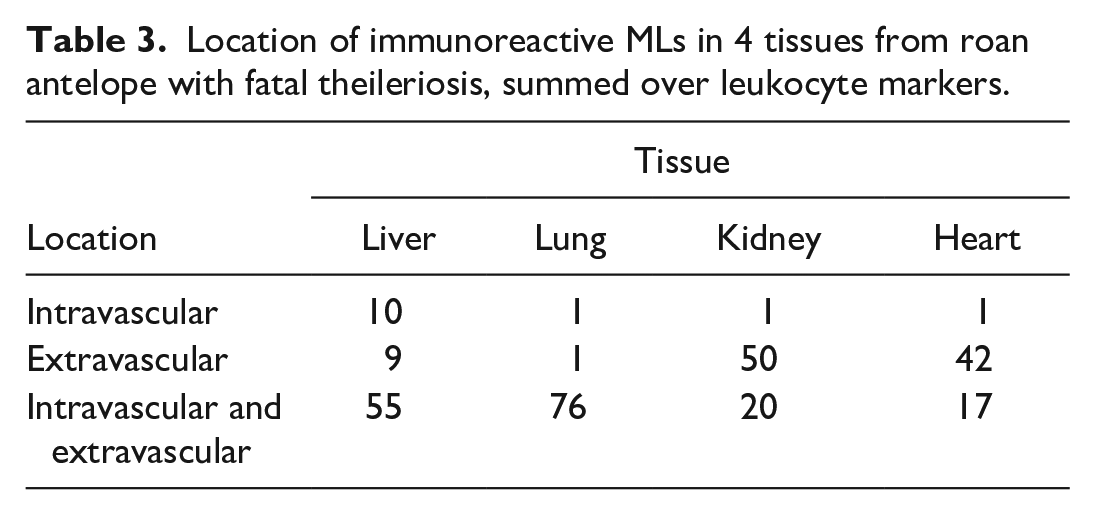
The intravascular and/or extravascular location of immunoreactive MLs in the selected tissues, summed over leukocyte markers, is recorded in Table 3. In testing if the location and tissue had a significant effect, the 3-way interaction (P = .84) and 2 of the 2-way interactions were not significant (marker: tissue, P = 1; marker: location, P = .40). However, the interaction between location and tissue was significant (P = .005). Immunoreactive MAC387-, IBA-1-, CD204-, and fewer CD3-positive MLs occurred within the intravascular and extravascular compartments in lung and liver specimens, whereas they were more commonly observed in the extravascular compartment in heart and kidney samples. Within the intravascular compartment specifically, MAC387-, IBA-1-, and CD204-positive MLs often appeared aggregated and lined the luminal surface of the vascular endothelium, particularly in the lungs (Fig. 5e, g, i). As for the uninfected controls, alveolar macrophages also consistently expressed MAC387, IBA-1, and CD204 antigens in fatally infected roans.
Location of immunoreactive MLs in 4 tissues from roan antelope with fatal theileriosis, summed over leukocyte markers.
Strong cytoplasmic labeling in the phenotyped ML-like cells prohibited the conclusive identification of ILP in these cells. However, occasional ILP were visible in nonlabeled cells per marker in each case. Preliminary attempts to identify the mononuclear leucocytes containing ILP with certainty by colabeling of tissues were unsuccessful, and there was insufficient material left to pursue this investigation. Attempts to label cultured schizont-infected cells derived from lymph node from an infected roan in 2020 also failed, possibly because of loss of cellular antigens in the course of repeated subculturing.
MNGCs failed to label convincingly with any of the lymphocyte and macrophage markers used, and their origin remains unknown.
Discussion
Fatal theileriosis in roan antelope was characterized by multi-organ infiltrates of parasitized and nonparasitized MLs and fewer MNGCs. MLs occurred within the microvasculature and small- to medium-sized veins and lymphatics, as well as in the extravascular compartment, whereas the MNGCs were limited to sinusoids and the extravascular compartment. Many parasitized and nonparasitized MLs appeared atypical, with oval to elongate and irregularly indented or convoluted hyperchromatic nuclei, variably prominent nucleoli, and moderate cytoplasm. Based on phenotypic characterization, the composition of the ML infiltrates was similar across lung, liver, kidney, and heart samples. Most MLs, especially in the lungs, expressed antigens typically associated with activated monocyte-macrophages (MAC387, IBA-1, and CD204) and possibly dendritic cells (IBA-1). There were fewer, but still significantly increased compared to control, numbers of CD3-reactive putative T-lymphocytes, while CD20-reactive putative B-lymphocytes were scarcer in tissues from infected roans than uninfected controls. A few parasitized and nonparasitized MLs and the MNGCs failed to express the leukocyte antigens investigated in this study. Attempts to identify the lineage of the parasitized cells were unsuccessful.
Given that this is the first time that Theileria-associated cellular infiltrates have been phenotyped in any wild ruminant, we cannot be sure that the leukocyte markers we used are transferable to this host species. However, the performance of the selected markers was first assessed in roan-positive tissue controls, and the pattern and location of immunoreactivity for each marker were found to be similar to that observed in the dog positive tissue controls and in the literature pertaining to diverse species (Table 1; Supplemental Figure S1). Therefore, it seems likely that the strong, prolific IBA-1, CD204, MAC387 and CD3 positivity observed in roan tissues represents a significant lymphohistiocytic (macrophage/histiocyte-predominant) response associated with fatal theileriosis in this species, as has been described in cattle with lethal ECF 25 and TT. 6 The MAC387 is also expressed on mature myeloid cells (Table 1); however, granulocytes were scarce in tissues from infected roans in our study.
Bovine TT is also associated with a multi-organ macrophage response,24,40 and is thought to be due to the tropism of T. annulata for macrophages and dendritic cells in particular, with subsequent aberrant T-cell activation and immune evasion.11,70 On the other hand, the widespread macrophage-centric inflammatory response that has been recently described in tissues from calves with ECF 25 is more surprising because of the tropism of T. parva for T-lymphocytes, which has been traditionally associated with lymphoproliferation.1,2,22 Taken together with our observations in roans, it may be worth considering that the macrophage/histiocyte response could be a universal response to Theileria, irrespective of which cell type the schizonts parasitize.
As we only used limited markers that have not yet been properly validated in roan tissues, subclassification of monocyte-macrophage-like leukocytes was not possible. Activated macrophages are thought to differentiate into classically M1 (proinflammatory and microbicidal) or alternatively activated M2 (anti-inflammatory and poorly microbicidal) cells. 47 The data on macrophage M1/M2 profiles in relation to the pathogenesis and outcome of many diseases are contradictory and confusing. 53 This is not altogether surprising considering that the tissue microenvironment is constantly changing, and therefore, macrophage activation is dynamic.53,73 Both Fry et al. 25 and Bastos et al. 3 showed that a heterogeneous proinflammatory and anti-inflammatory macrophage and monocyte response characterizes lethal ECF in calves. In our roan, the CD204 scavenger receptor A protein, which is often upregulated on M2-biased tissue macrophages,10,31,56 was expressed on MLs. MAC387 reactivity was also present and has been tentatively associated with an M1-biased macrophage response in diverse diseases.67,69 This suggests a similar reaction to that in ECF, but further investigation is required using a wider panel of leukocyte markers that have been authenticated in roan tissues.
The observation of intravascular aggregated and marginated monocyte-macrophage-like MLs (which far exceeded intravascular CD3-reactive T-lymphocytes) in small to medium-caliber thin-walled vessels, especially in the lungs, was a consistent finding in roans, and is similar to calves with lethal ECF 25 and to eland (Tragelaphus oryx) with fatal theileriosis. 29 It is noteworthy that the intravascular MLs consistently expressed both IBA-1 and CD204, which are traditionally markers of mature tissue macrophages (Table 1).28,36,75 In addition, MAC387 expression on intravascular MLs, as was observed in these roans, is associated with bone marrow–derived circulating monocyte-macrophages. 75 Thus, the weight of evidence suggests that the intravascular MLs in roans are likely to be monocyte-macrophages. A possible role for pulmonary intravascular macrophages (PIMs), which reliably express MAC387 in horses, 58 also needs to be investigated in roans and other ruminants with fatal theileriosis. PIMs are mature phagocytes, which function similarly to Kupffer cells in the liver, and are firmly adhered to the capillary endothelium in the normal lungs of cattle, sheep, goats, pigs, horses, cats, and deer.5,13,64 They can be identified readily by transmission electron microscopy and have been shown to play a significant proinflammatory role in acute lung injury induced by endotoxins and infectious agents.19,64,68
We established that CD20-reactive B-lymphocytes were significantly reduced in tissues from fatally infected roans compared to uninfected controls. This finding is similar to cattle with fatal theileriosis, where the B-lymphocyte response is significantly diminished.6,25,37 The depletion of putative B-lymphocytes in roans may be linked to the marked atrophy of follicular lymphoid tissue in lymph nodes and spleen. This has also been reported in fatally infected cattle6,25 and common eland 29 with theileriosis. Similar findings are reported in other hemoprotozoal diseases, including Plasmodium falciparum malaria in humans27,76 and Babesia rossi infection in dogs.34,61 It may be that B-cell activation, migration, and maturation processes are compromised in severe theileriosis and other hemoprotozoal diseases. 27 So far, mechanisms for the B-cell depletion have not been elucidated, but it is possible that the cytokine milieu associated with the dysregulated T-cell and hyperplastic macrophagic responses in fatal theileriosis suppresses the differentiation of antibody-producing cells.
Similarly to theileriosis in livestock25,40,43 and nonhippotraginid African wild artiodactyls, 14 hemorrhages, particularly petechiae and ecchymoses, were commonly seen in infected roans in our study. Suffusive hemorrhages and diffuse enteric hemorrhage were rarely seen. Two roans with diffuse enteric hemorrhage also had widespread suffusive hemorrhages as well as petechiae and ecchymoses, with no evidence of underlying GIT pathology. This suggests that the enteric hemorrhage was due to a hemorrhagic diathesis in these cases.
The hemorrhages in livestock with lethal theileriosis have been ascribed to macrophage-derived cytokine-mediated vasculitis, 25 thrombocytopenia due to disseminated intravascular coagulation, 48 bone marrow suppression,49,81 and platelet degranulation and lysis, 38 as well as liver dysfunction-associated coagulopathy. 63 In our roans, hemorrhages were not obviously associated with necrotizing vasculitis. Mononuclear leukocyte and MNGC infiltration of bone marrow were only observed in 1 of 8 roans, unlike in ECF,20,49,81 and, although histologic evidence of liver damage was consistently present in our roans, the size of the hemorrhages in roan carcasses was not typical for liver dysfunction-associated coagulopathy. It was concluded that the hemorrhages were probably the result of vascular endothelial injury and/or a platelet disorder(s).
Icterus was frequently reported in fatally infected roans and may be associated with hepatic dysfunction. However, the simultaneous occurrence of icterus and probable anemia in most cases, with frequent splenomegaly and occasional reports of bilirubinuria and hemoglobinuria, suggests that an acute hemolytic syndrome might play a role in this disease in roans, as has been reported in cattle with TT, 40 and, to a lesser extent, wild artiodactyls with theileriosis.14,32,84 However, the cause of the watery blood and mucous membrane pallor (suggestive of anemia) and icterus could not be established and requires further investigation.
Despite the consistent, often diffuse infiltration of infected and uninfected MLs in the lungs, with histologic edema and multiple hemorrhages, pulmonary edema and interstitial pneumonia were reported grossly in only 50% and 30% of cases, respectively. On the other hand, severe lung edema is frequently observed macroscopically in cattle with fatal ECF and is thought to cause the severe respiratory distress that characterizes the late stage of infection.25,43 It is possible that the gross lung pathology was underreported in roans with fatal theileriosis or that the roans died before the lungs became severely edematous. Proinflammatory cytokine-induced vascular endothelial injury has been implicated in the pathogenesis of the pulmonary edema in a variety of infectious and immune-mediated conditions, including ECF, that result in hypercytokinemia or the “cytokine storm syndrome.”25,43,44,46 Although we did not observe obvious necrotizing vasculitis in the lungs in roans in our study, the distension and occlusion of the pulmonary vasculature with monocyte-macrophage-like cells and their association with the vascular endothelium are likely to be pivotal in the pathogenesis of the lung edema in roans, similar to domestic livestock with theileriosis. 25 The degree of vascular endothelial activation and/or injury may simply be more subtle in roans.
Histologic renal lesions consistently revealed variably hypercellular glomeruli, leukostasis, and multifocal, mild-to-moderate interstitial infiltrates of parasitized and nonparasitized leukocytes. These lesions were often subtle at the microscopic level, which is probably why macroscopic kidney lesions were reported so rarely in roans in this study. Similarly, subtle histologic renal lesions were reported in eland with fatal theileriosis. 29 However, white-spotted kidneys, which are traditionally interpreted as interstitial nephritis due to leukocyte infiltration in theileriosis, are commonly reported in cattle with ECF 43 and, to a lesser extent, with TT. 40 It is possible that the course of the disease is longer incattle with ECF, allowing more time for large aggregates of MLs to develop.
Diarrhea, and GIT erosions and ulcers (thought to be associated with vascular occlusion by MLs and/or thrombi) were not specifically associated with theileriosis in roans in this study. Only 2/15 roans with histologic sections of GIT had evidence of Theileria-associated leukocytic infiltration in the wall of the abomasum and small intestine. In both cases, the infiltrates were mild, multifocal, and not associated with macroscopic thickening of the wall or with obvious fecal soiling of the carcasses. This contrasts with the often extensive vasocentric infiltration of parasitized and nonparasitized leukocytes in the wall of the GIT commonly associated with macroscopic thickening of the wall and/or erosions and ulcers in cattle40,43 and eland 29 with fatal theileriosis. In our study, fecal soiling was only recorded in 2 roans with fibrinous enterocolitis associated with Salmonella coinfection. In both cases, leukocytic infiltrates (some with obvious theilerial parasites) were observed in multiple organs except the GIT, indicating that these animals died of theileriosis and concomitant enteric salmonellosis.
It was not possible to perform IHC on lymphoid organs in our study because of the moderate degree of autolysis in many spleen and lymph node samples. As these are the initial target tissues in ruminants with theileriosis,24,40,43 it would be important to include them in future IHC studies on Theileria-infected roans.
We only compared leukocyte antigen expression in tissues from roans that died of theileriosis to uninfected controls that died of noninfectious causes. Rather than look at a snapshot of the end of the disease process, it would be more informative to evaluate changes in leukocyte phenotype during the course of disease, as was recently done in steers with experimental ECF. 3 However, temporal sampling would require experimental infection of animals, and roan antelope are scarce and expensive, making this impractical.
In summary, the multi-organ vasocentric putative lymphohistiocytic reaction in roan antelope with fatal theileriosis is similar to that in cattle with TT and ECF. The possibility of an immune deregulatory macrophage activation-like syndrome (MAS), as was recently described by Fry et al. 25 in cattle with ECF, which might also be responsible for the findings and lesions observed in cattle with TT,6,24,40 is worthy of further investigation in roans. The MAS is characterized by a particularly exuberant multisystemic macrophagic (histiocytic) response, with resultant tissue damage, systemic inflammation (hypercytokinemia), and multiple organ failure.18,25
Future studies in roan will require validation of leukocyte markers for IHC, including markers of the CD163 scavenger receptor antigen, which is often, but not exclusively expressed by alternatively activated (M2-polarized) macrophages that are also associated with the MAS.18,25 Furthermore, schizont-infected leukocytes in roans require phenotyping using double labeling IHC capable of colocalizing intracytoplasmic theilerial antigens and leukocyte receptor antigens to identify whether they are monocyte-macrophages/histiocytes or T-lymphocytes.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221120011 – Supplemental material for Theileriosis in naturally infected roan antelope (Hippotragus equinus)
Supplemental material, sj-pdf-1-vet-10.1177_03009858221120011 for Theileriosis in naturally infected roan antelope (Hippotragus equinus) by Sarah J. Clift, Bernat Martí-Garcia, John A. Lawrence, Emily P. Mitchell, Jeanni Fehrsen, Jorge Martínez, June H. Williams and Johan C. A. Steyl in Veterinary Pathology
Footnotes
Acknowledgements
We thank the skilled histopathology and immunohistochemistry technologists, particularly Rephima Phaswane and Naomi Timmerman at the Faculty of Veterinary Science (FVS), University of Pretoria (UP). We thank Caitlin Holyoake, recently graduated veterinarian (FVS, UP), for her invaluable assistance with data curation and Jaco Greeff (UP) and Geoffrey Fosgate (FVS, UP) for statistical support. Thanks to the Faculty pathologists and residents who contributed cases to the study; Eric Liebenberg from the University of the Witwatersrand, for whole slide imaging, and Estelle Mayhew in the Department for Education Innovation, FVS, UP for her assistance with the figures.
Authors’ Note
The manuscript has been prepared in the Uniform Requirements format.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Department of Paraclinical Sciences at the Faculty of Veterinary Science, University of Pretoria, and the Health and Welfare Sector Education and Training Authority (HWSETA) in South Africa.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
