Abstract
Tularemia is a severe disease caused by Francisella tularensis. This bacterium has a major pathogenic potential in countless animal species as well as in humans. Despite the relatively significant body of literature available on this microorganism, many questions are still open concerning its biological cycle in the environment, the pathology and pathogenesis of the disease, the possible routes of infection in animals, and the pathologic and ecological relevance of the distinct phylogenetic clusters of F. tularensis. In order to address these questions, we have thoroughly characterized the pathology and microbiology of terminally ill European brown hares (Lepus europaeus) infected with F. tularensis subsp. holarctica, collected in Switzerland from 2012 to 2014. F. tularensis isolates were typed by defining their phylogenetic clusters. We showed that the pathology associated with F. tularensis subsp. holarctica belonging to the clade B.FTNF002-00 is different from that previously reported to be associated with the clade B.13. In particular, strains of the clade B.FTNF002-00 were almost invariably associated with splenitis and hepatitis and not with the polyserositis affecting pleura, pericardium, and kidney reported in the literature for infections caused by the clade B.13. We describe findings suggesting that the ports of entry for the bacteria might be the respiratory and digestive routes.
Tularemia is a zoonotic disease, the causative agent being Francisella tularensis, which comprises 2 clinically relevant subspecies. F. tularensis subsp. tularensis is responsible for most of the human fatalities and is found essentially in the North American continent, while F. tularensis subsp. holarctica is widespread throughout the whole Northern hemisphere and, while considerably less virulent than F. tularensis subsp. tularensis, may cause severe incapacitating disease in humans. 8,38
Recently, distinct clades of F. tularensis subsp. tularensis with a specific geographical distribution have been associated with different clinical outcomes in humans, virulence in mice, ecological niches, and host species. 26 However, little is known concerning the distinct ecology and pathogenesis of the phylogenetic lineages of the subspecies holarctica, which in Europe is mostly represented by the clade B.FTNF002-00 in the west and the clade B.13 in the rest of the continent. 11
Several forms of tularemia have been described in humans. 38 The most common is an ulceroglandular disease, which is considered the consequence of skin contact with infected material or arthropods bites and consists of a skin ulcer associated with regional lymphadenopathy. 10,38 Additional recognized forms of the disease include the glandular form with regional lymphadenopathy not associated with obvious skin lesions, oculoglandular with primary localization in the conjunctiva, and typhoïdal, a very general clinical presentation, essentially characterized by fever and that may be associated with lesions of internal organs. 9,38 The pneumonic form reported in the literature may occur as primary 9,38 or represent an outcome of any of the forms described previously, but more commonly of the typhoïdal form. 9 Finally, the oropharyngeal form is associated with cervical lymphadenopathy that may occur following ingestion of contaminated food or water. 38
Death in humans may follow bacteremia, 9 whereas death secondary to respiratory disease occurs relatively infrequently. 9 Lung lesions consist mainly of necrotizing bronchopneumonia, likely following inhalation of F. tularensis, while hematogenous spread from other infected organs results in interstitial and necrotizing pneumonia. 9,14,16 In the liver and spleen, the lesions are characterized by disseminated necrosis. 9
In animal hosts, F. tularensis mainly affects rodents and lagomorphs. 18,19 In rodents, necrotic lesions are seen in the liver, spleen, and to a lesser extent in the lung. 6 In naturally occurring tularemia in lagomorphs, liver and spleen are consistently affected while the frequency and severity of the lung lesions may vary. In particular, pulmonary lesions appear to be relatively infrequent in the variable hare (Lepus timidus) 19 and more common in the European brown hare (L. europaeus). 12 In contrast to the situation described in humans, experimental inoculation of F. tularensis, mimicking the human ulceroglandular form, does not appear to lead to a systemic disease in the variable hare. 19 In brown hares, it was shown to be fatal, although not consistently. 3,4 Oral infection in variable hares has also failed to result in systemic disease. 19 Interestingly, in a more recent study focused on natural infection with F. tularensis subsp. holarctica (clade B.13) and associated pathology in the European brown hare, the most commonly affected organs were lung, pericardium, and kidney. This is different from previous observations in lagomorphs. 12 Non-human primates including rhesus monkey (Macaca mulatta), 30,31 grivet (AKA African green) monkey (Chlorocebus aethiops) 5,35 and marmoset (Callithrix jaccus) 20 have also been chosen as experimental models to investigate the pathogenesis of F. tularensis. These animals developed clinical signs and lesions similar to those observed in infected humans at varying degrees of severity, showing a higher sensitivity to F. tularensis subsp. holarctica than displayed by humans. Additionally, during a natural outbreak of tularemia in cynomolgus monkeys (M. fascicularis), clinical signs and lesions were similar to those observed in humans. 17
Several open questions remain, which include the routes of entry of the bacterium in animal hosts and its association with the lesions’ distribution, the definition of the reservoirs for F. tularensis, and the interaction between the virulence factors of the pathogen and the host immune system taking into account the distinct phylogenetic clusters of F. tularensis. 27
Here we report the detailed pathology and bacteriology in terminally ill European brown hares predominantly infected with F. tularensis subsp. holarctica clade B.FTNF002-00 collected in Switzerland from February 2012 to June 2014. Comparison of our results with a previous study describing the pathology and bacteriology in European brown hares infected with F. tularensis subsp. holarctica clade B.13 11 shows that subpopulations of F. tularensis subsp. holarctica may be associated with different pathologic findings in the European brown hare.
Materials and Methods
Animals and Pathology
Fifty-three free-ranging, wild European brown hares were collected by game wardens and/or hunters throughout the entire Swiss territory from February 2012 to June 2014. Since hares were either found dead or culled after being found agonic, this meant under the current Swiss legislation (Federal Animal Protection Law, RS455; Federal Hunting Law, RS922; https://www.admin.ch/opc/fr/classified-compilation/national.html) that no ethical approval or permit for animal experimentation were required. All the animals underwent full necropsy, and a set of tissues was collected from the hares when available. These comprised brain, tongue, esophagus, stomach, small and large intestine, cecal appendix, trachea, lungs, kidney, adrenal glands, urinary bladder, liver, spleen, heart, and gonads. Additionally, ad hoc tissues were collected from animals showing gross lesions in organs other than those described previously (skin, lymph nodes, pericardium, mammary glands, and skeletal muscle). Tissues were fixed in 10% buffered formalin, processed, and stained as previously described. 23
Immunohistochemistry
Tissue sections obtained from examined animals with confirmed infection by F. tularensis (polymerase chain reaction [PCR], see the following) and the presence of histologic lesions underwent immunohistochemical (IHC) staining according to an ad hoc modified established protocol. 22 Briefly, the sections were stained with a mouse monoclonal antibody directed against the F. tularensis LPS (Senova GmbH, Jena, Germany) diluted 1/1000 in PBS in a humid chamber and incubated at 4°C overnight, then treated with a goat polyclonal antibody labeled with horseradish peroxidase (Kirkegaard and Perry Laboratories, Gaithersburg, MD, USA) diluted 1/2000 in PBS. The chromogen (Diaminobenzidine-DAB) (Dako, Baar, Switzerland) was added, and development was carried out according to the manufacturer’s instruction until an unambiguous visible stain was present on positive control tissues but without exceeding 5 minutes. The sections were counterstained with hematoxylin.
Bacteriology
A dedicated set of tissues (Table 1) collected at necropsy was further processed for bacteriological investigations. Lysates were prepared and tested by PCR targeting the fopA gene encoding for the outer membrane protein A for the presence of F. tularensis as previously described. 29,37 Briefly, tissue samples collected with an inoculating loop were transferred in 500 µl of lysis buffer (0.1 M Tris–HCl, pH 8.5, 0.05% Tween 20, 0.24 mg/ml proteinase K) and incubated for 1 hour at 60°C, followed by a 15-minute denaturation step at 95°C. For real-time PCR, 22.5 μl of PCR mixtures were added to 2.5 μl of tissue lysates, and a Ct cutoff value of 40 was used. The same samples were cultivated onto trypticase soy agar with 5% sheep blood (BA) (Oxoid, Basel, Switzerland), chocolate agar with IsoVitaleX (ChocIso) (Becton Dickinson, Allschwil, Switzerland), and F. tularensis selective agar (Ftsel; chocolate agar with brain-heart infusion broth [Becton Dickinson] as base, 1% soluble hemoglobin powder [Oxoid], 0.1% L-cysteine hydrochloride monohydrate, 0.1% D-glucose and Skirrow Campylobacter selective supplement [Oxoid]) for 3 days at 37°C with 5% CO2. All live cultures were manipulated under BSL3 containment. Isolates were further identified at the subspecies level by PCR targeting the RD1 locus and phylogenetically typed using canonical single nucleotide polymorphism (canSNP) as previously described. 21,28,34
Comparison of the Detection of Francisella tularensis by Polymerase Chain Reaction and Culture From Organs of Hares Positive for F. tularensis.
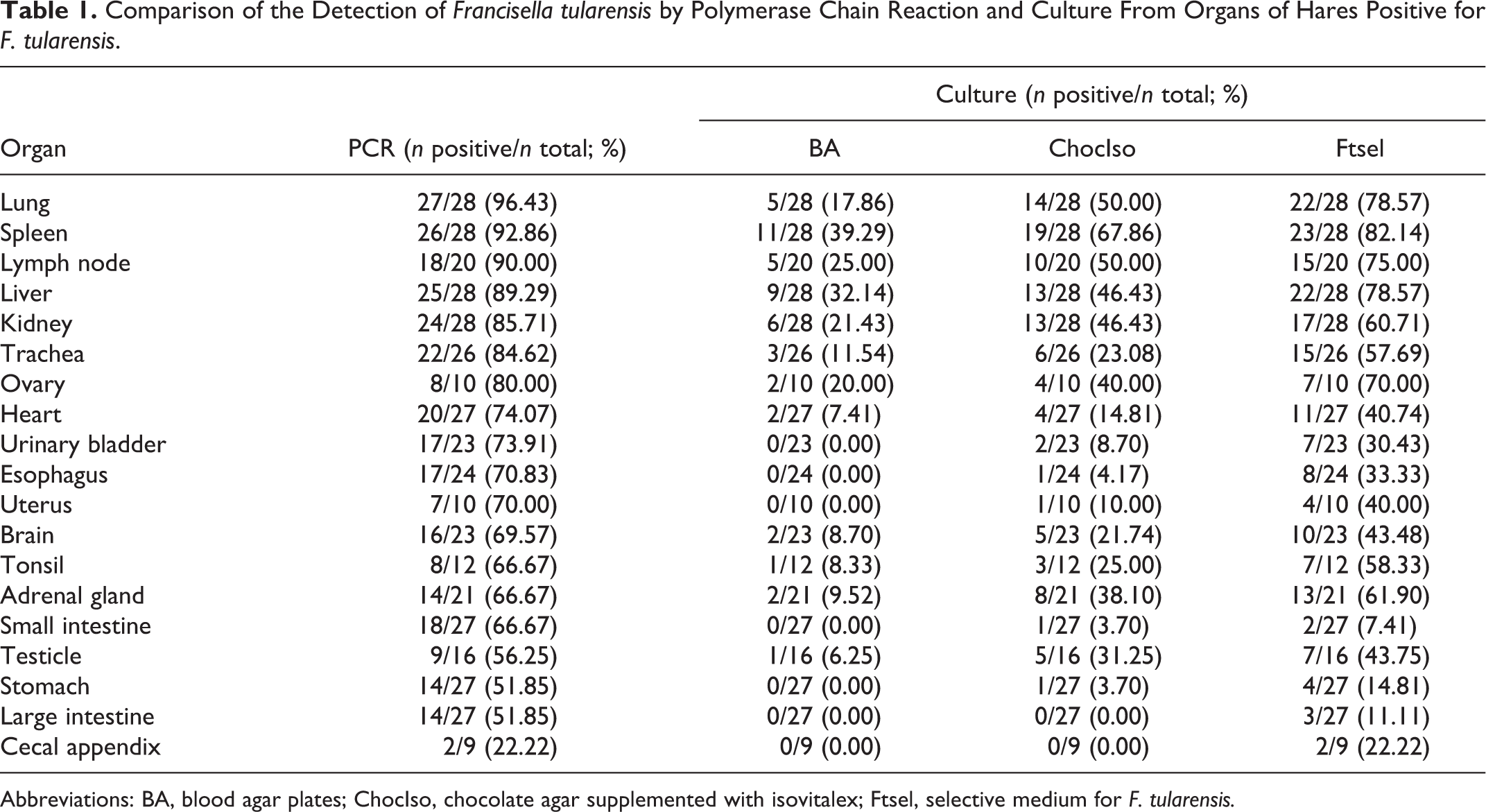
Abbreviations: BA, blood agar plates; ChocIso, chocolate agar supplemented with isovitalex; Ftsel, selective medium for F. tularensis.
Results
Gross Pathological Findings
Of the 28 F. tularensis positive hares, 14 (50%) were found dead, 8 (28%) were culled after being found agonic, while 3 were captured by hunting dogs. For the remaining 3, no specific history was recorded. Additional details are provided in Table 2. Common necropsy findings included a variable degree of splenomegaly (28/28, 100%) (Fig. 1) and lymphadenomegaly (15/28, 53%) (mandibular, tracheal, mediastinal, tracheobronchial, ileocecocolic lymph nodes) with multiple light tan to yellow foci up to 2 mm in diameter in cross section (7/15, 47%) (Figs. 2, 3). Thickening and reddening of the tracheal mucosa was also frequently observed (up to 3 mm thick; 25/28,89%) along with light yellow to light tan 1 to 2 mm in diameter, slightly raised necrotic foci scattered along the mucosa (6/25, 24%) (Fig. 4). Hemorrhages (12/28, 43%) occurred more frequently in the lung. Obvious signs of pneumonia (2/28, 7%) and pleural changes (3/28,11%) were not commonly detected grossly (Fig. 5). Stomach erosions and ulcers were seen occasionally (4/28, 14%). Gross lesions were seen in the kidney in only 2 individuals and consisted of slightly raised light tan to yellow 2 to 3 mm in diameter foci on the kidney capsule in 1 individual (infected with a B.13 strain) and in yellow discoloration of the pelvis in another one. Similar necrotic foci were seen on the testicle of three hares (3/28, 11%). Finally, pericardial lesions were only seen in 1 case and consisted of 2- to 3-mm slightly raised light tan to yellow foci. Similar findings were seen on the serosa of the cecal appendix (Fig. 6) of 1 individual and on the salivary glands of another one.
Gross Findings in Hares Positive for Francisella tularensis.a
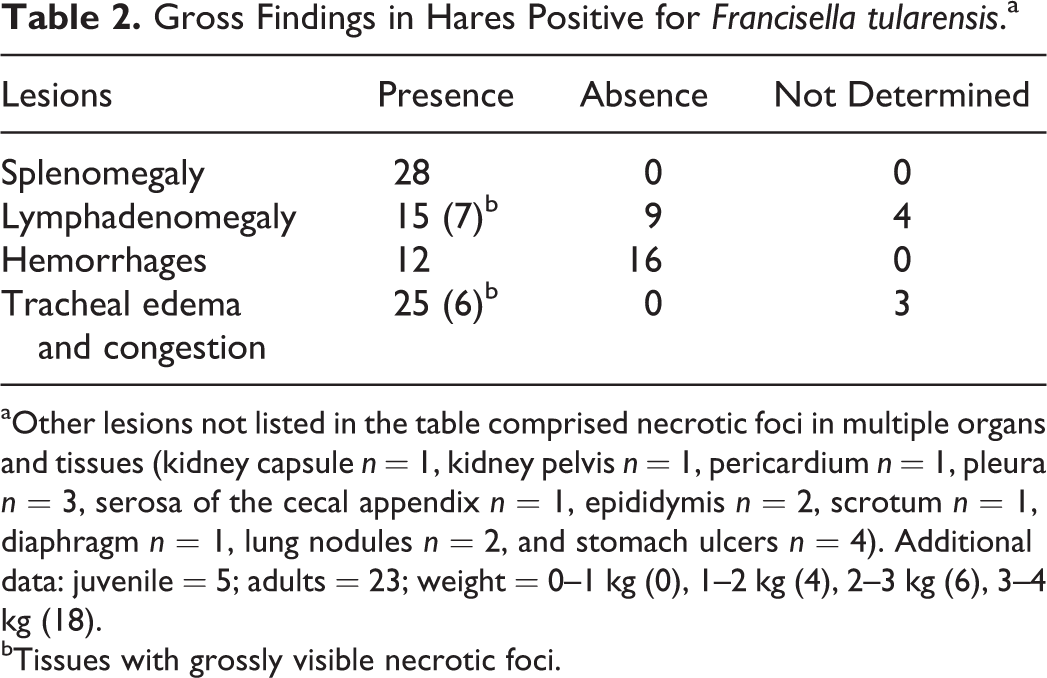
aOther lesions not listed in the table comprised necrotic foci in multiple organs and tissues (kidney capsule n = 1, kidney pelvis n = 1, pericardium n = 1, pleura n = 3, serosa of the cecal appendix n = 1, epididymis n = 2, scrotum n = 1, diaphragm n = 1, lung nodules n = 2, and stomach ulcers n = 4). Additional data: juvenile = 5; adults = 23; weight = 0–1 kg (0), 1–2 kg (4), 2–3 kg (6), 3–4 kg (18).
bTissues with grossly visible necrotic foci.
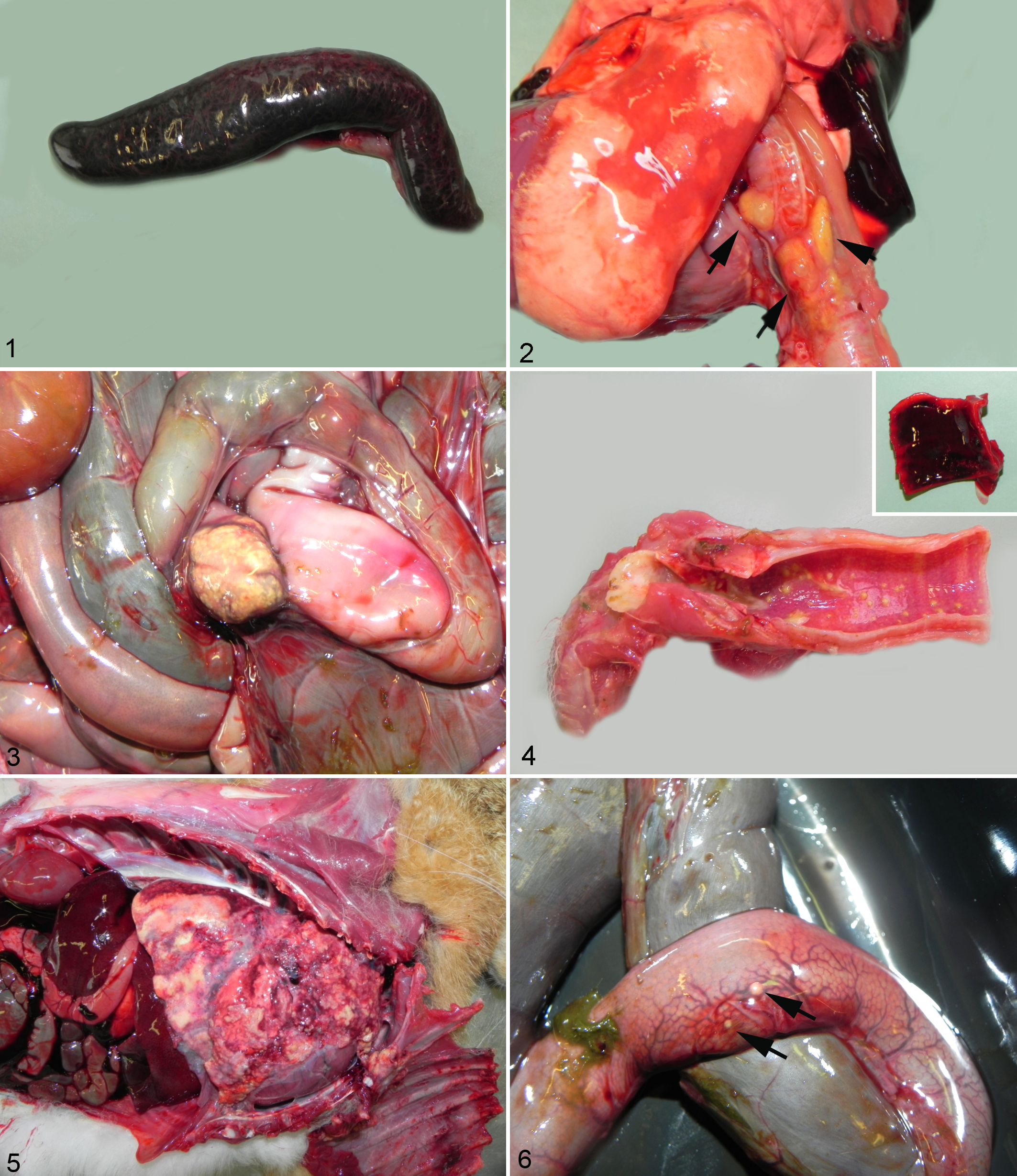
Francisella tularensis infection gross findings.
Microscopic Findings
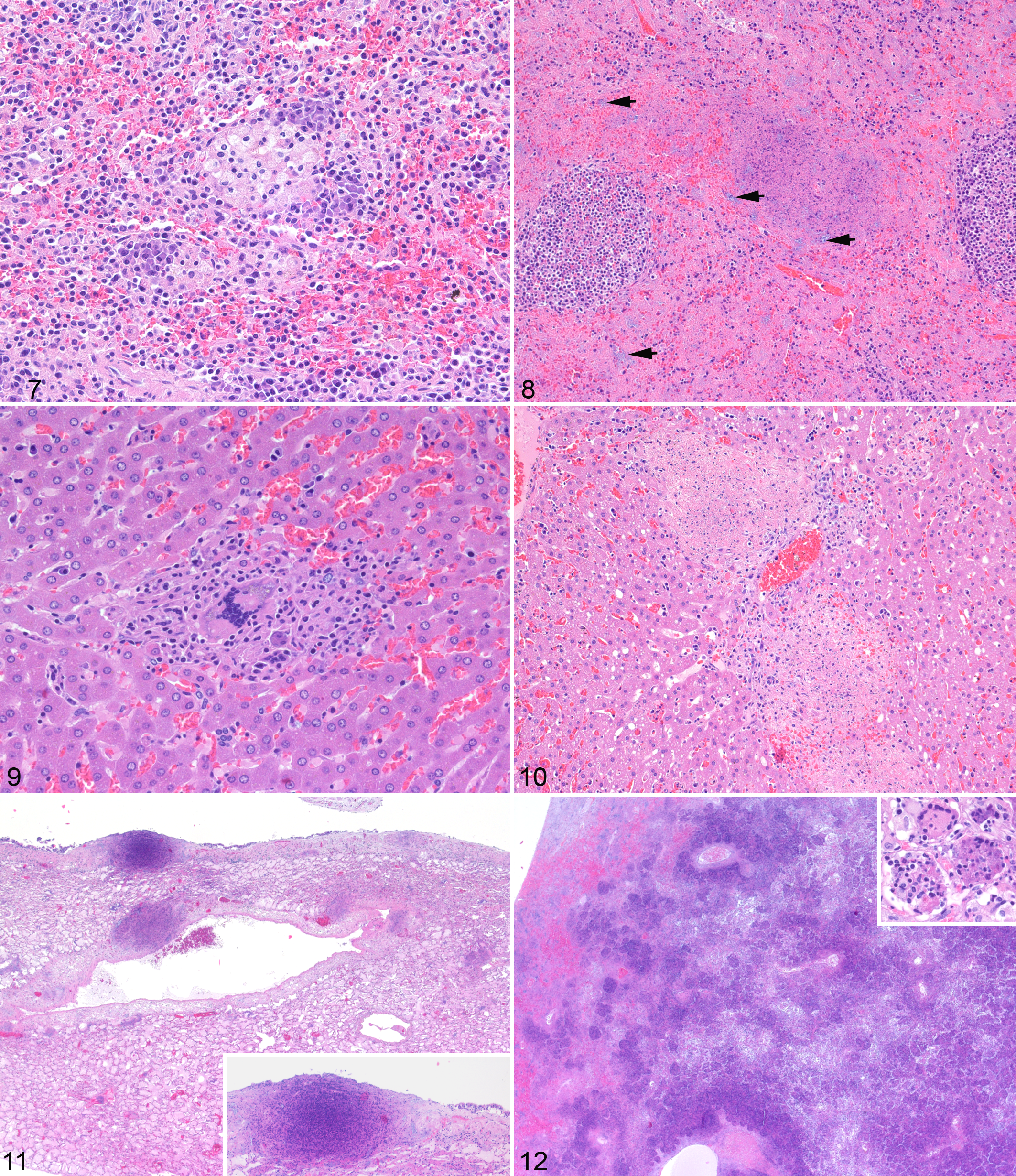
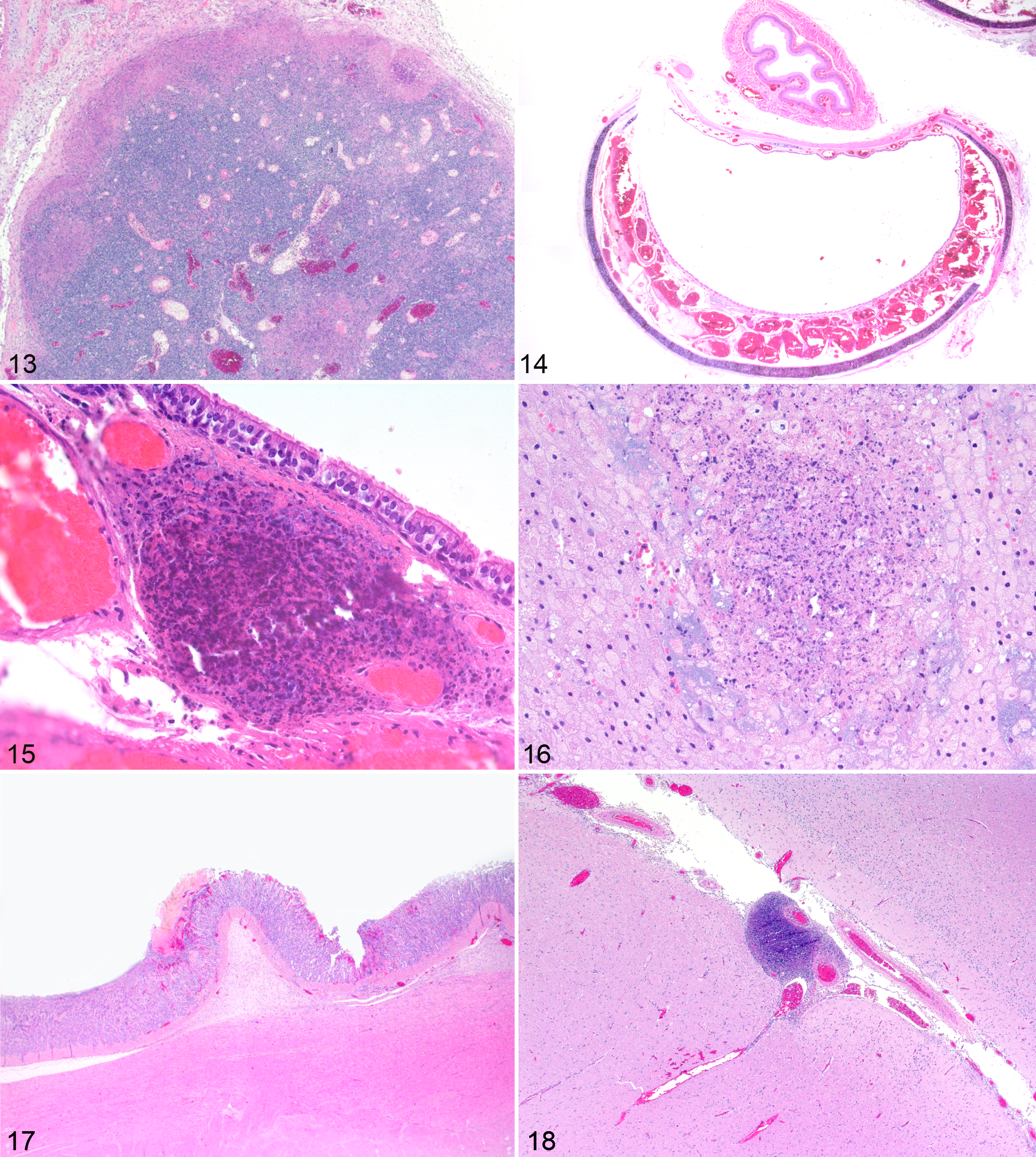
A summary of the histological changes observed in the positive hares is shown in Table 3. Briefly, in the spleen the lesions ranged from granulomatous (Fig. 7) to necrotizing splenitis (10/28, 36%) (Fig. 8) to a more frank necrotizing splenitis (18/28, 64%). Of interest were granulomas composed of foamy and vacuolated macrophages frequently rimmed by small clusters of plasma cells interpreted as expression of earlier stages of bacterial colonization in this organ (Fig. 7). In the liver, the hallmark of the disease was consistent with a necrotizing process (necrotizing hepatitis; 14/27, 51%) and a relatively frequent association either with granulomas and pyogranulomas or remnants of them (granulomatous and necrotizing hepatitis; 13/27, 48%) (Figs. 9, 10) expanding from the sinusoids with accumulation of clusters of mononuclear inflammatory cells and prominent Kupffer’s cells. The presence of multinucleated giant cells was rare (Fig. 9). The lung showed a more extensive range of lesions comprising frank focal necrosis to multifocal necrotizing and granulomatous pneumonia with an interstitial pattern (9/27, 33%), affecting small areas of the lung parenchyma (Fig. 11). Pyogranulomatous and necrotizing bronchopneumonia were more rarely observed (5/27, 18%) (Fig. 12). Only in 2 cases, the bronchopneumonia was very severe and extended over most of the parenchyma, with many multinucleated giant cells in one case (Fig. 12, inset). Prominent necrotizing lymphadenitis was frequent in the diseased brown hares (17/18, 94%), affecting more commonly the nodal subcapsular sinuses and the cortex (Fig. 13). The histological changes in the trachea were very consistent and comprised a prominent edema associated with severely distended blood vessels engorged with erythrocytes (25/25, 100%) (Fig. 14). The tracheal (pyo-)granulomatous and necrotizing lesions (7/25, 28%) were mainly within the submucosa and occasionally extending to the elevated epithelium (Fig. 15). Vascular changes of the trachea involved also the presence of thrombi and emboli (n = 3) or frank hemorrhages (n = 1). In the examined adrenal glands with histological lesions (16/19, 84%), focal to multifocal necrotizing (8/16, 50%) and a more distinct (pyo-)granulomatous adrenalitis (granulomatous and necrotizing adrenalitis; 8/16, 50%) (Fig. 16) were observed. The cecal appendix was the most common section of the gastro-intestinal (GI) tract to be affected by F. tularensis (7/12, 58%), with changes consistent with granulomatous and necrotizing typhlitis. Less frequent lesions comprised gastric ulcers and erosions (4/20, 20%) (Fig. 17) and testicular changes (4/14, 28.5%). F. tularensis–associated inflammatory processes in the kidney (pyelonephritis n = 1; perinephritis n = 1) were rare as well as in the brain (Fig. 18). Of the lesions observed in the heart, frank necrotizing pericarditis consistent with F. tularensis infection was observed in a single case.
Microscopic Findings in Hares Positive for Francisella tularensis. a
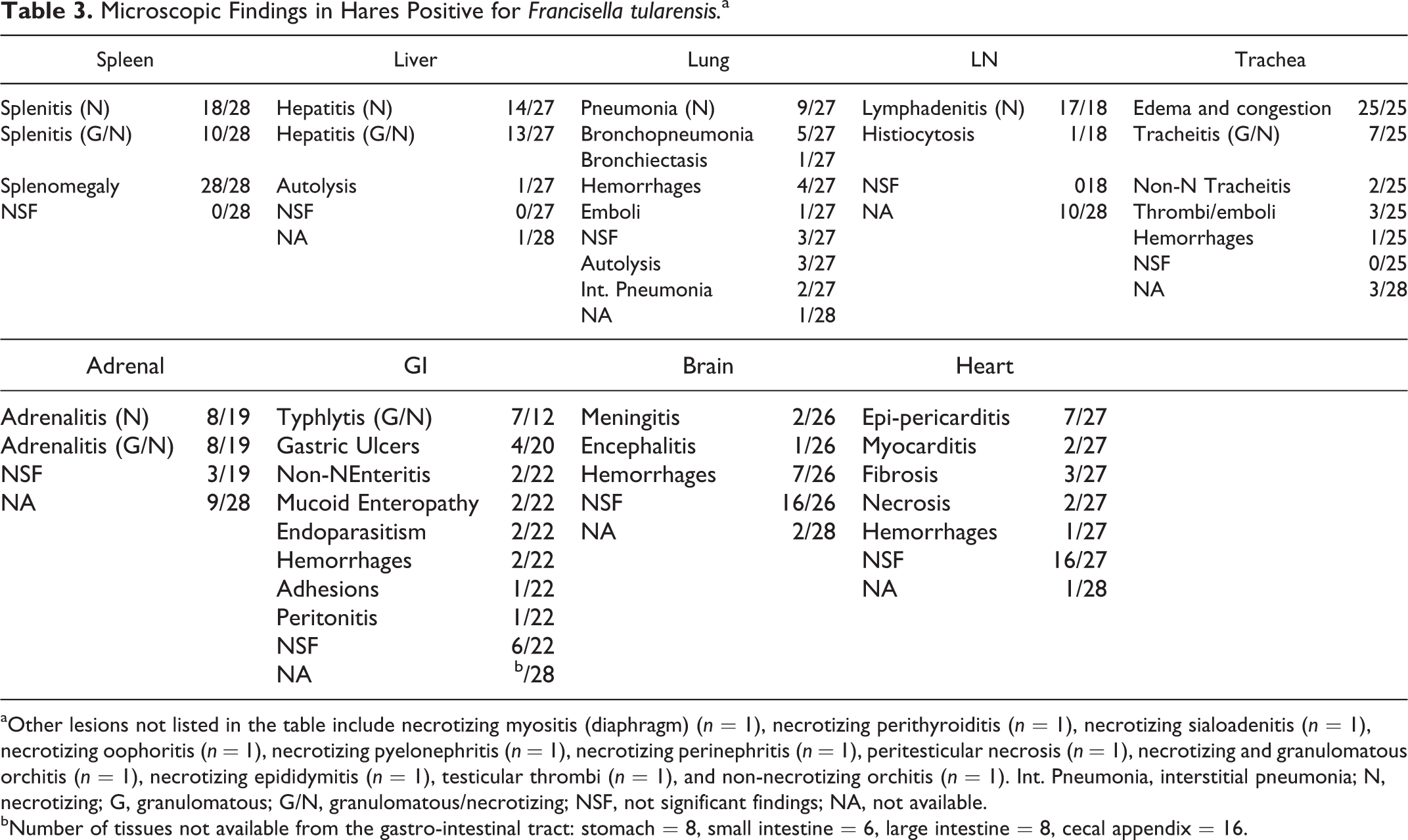
aOther lesions not listed in the table include necrotizing myositis (diaphragm) (n = 1), necrotizing perithyroiditis (n = 1), necrotizing sialoadenitis (n = 1), necrotizing oophoritis (n = 1), necrotizing pyelonephritis (n = 1), necrotizing perinephritis (n = 1), peritesticular necrosis (n = 1), necrotizing and granulomatous orchitis (n = 1), necrotizing epididymitis (n = 1), testicular thrombi (n = 1), and non-necrotizing orchitis (n = 1). Int. Pneumonia, interstitial pneumonia; N, necrotizing; G, granulomatous; G/N, granulomatous/necrotizing; NSF, not significant findings; NA, not available.
bNumber of tissues not available from the gastro-intestinal tract: stomach = 8, small intestine = 6, large intestine = 8, cecal appendix = 16.
Francisella tularensis infection, brown hare.
Francisella tularensis infection. Brown hare. HE.
Immunohistochemistry
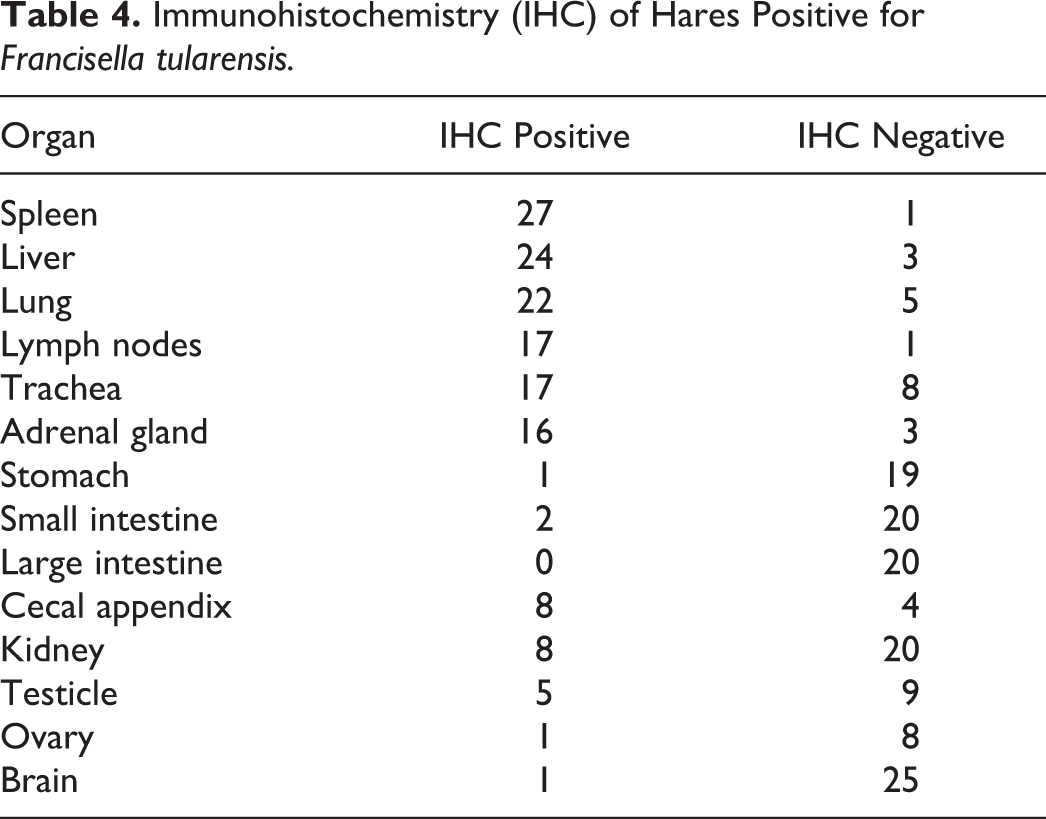
The tissues collected from all the F. tularensis–positive hares were also investigated by IHC in order to better localize the bacterial antigens within the lesions and additional features of the Francisella-associated tissue changes that could not be assessed by conventional HE staining. The results are summarized in Tables 4 and 5. The spleen was almost invariably positive by IHC (27/28, 96%), and the stain was observed to be more frequently associated with the granulomas and necrotic foci (22/27, 81%) and more rarely scattered in the parenchyma (13/27, 48%) (Fig. 19). The stain was seen both intra- and extracellular. The only IHC-negative spleen showed the granulomas interpreted as suggestive of early lesions following a late colonization of the spleen with an amount of bacterial antigen below the limit of detection of the IHC. In the liver, the immune stain was detected in 24 animals (24/27, 89%). The positive stain highlighted more frequently the granulomatous and necrotic foci (23/24, 95%). Immunohistochemical reaction was observed less consistently lining the sinusoids (8/24, 33%), in Kupffer cells (5/24, 21%) (Fig. 20), and rarely in the hepatocytes (n = 1), with prominent cytoplasmic vacuolization (Fig. 20, inset). The lung of 22 (22/27, 81%) hares stained positive and showed the most variable distribution of the chromogen. Interalveolar septa (9/22, 41%), scattered single or small clusters of cells (9/22, 41%), perivascular tissue (9/22, 41%), alveolar macrophages (4/22, 19%), and alveolar epithelium (4/22, 19%) stained positive. The stain was only occasionally observed in the airways within the mucosal epithelium (3/22, 14%) (Fig. 21), the submucosa (n = 1), and the peripheral connective tissue (n = 1). The lymph node parenchyma was often stained (17/18, 94%), consistently highlighting the cortical portion, including the subcapsular sinuses (17/17, 100%). The trachea was also frequently stained (17/25, 68%), mostly within the vascular spaces with highlighted endothelial lining and associated thrombi/emboli (13/17, 76%). The submucosa was the second most affected tissue component of this organ (8/17, 47%). The epithelium was more marginally affected (5/17, 29%) and often not primarily but in contiguity with the underlying necrotic submucosa (Fig. 22). The serosa was rarely affected (n = 1). The cortex of the adrenal gland was consistently stained (16/19, 84%). The most prominent stain observed in the GI tract was observed in the cecal appendix (8/12, 66.7%), highlighting the central portion of the lymphoid follicles (Fig. 23) or associated with clusters of macrophages infiltrating the follicles. In the other tracts of the GI, the positive stain was rarely seen (stomach: 1/20, 5%; small intestine: 2/22, 9%; large intestine other than cecal appendix: 0/20, 0%; serosa/peritoneum: n = 1) with the exception of the blood vessels, where it was a relatively common finding, likely reflecting deposition of antigen along the vessels more than a primary lesion. In the kidney, the detection of positive stain was relatively frequent (8/28, 28%) but mainly focused on the vascular portion (7/8, 87%) of the organ (glomerular tufts). Immunohistochemical staining was seen also in the testicle (5/14, 35%). In parallel with the histological findings, positive staining of the brain was uncommon. Of interest was the frequent occurrence of positively stained monocytes observed within the lumina of several blood vessels (Fig. 24), including those of tissues that did not show any positive stain elsewhere.
Immunohistochemistry (IHC) of Hares Positive for Francisella tularensis.
Immunohistochemical Localization of Francisella tularensis Antigen in the Tissues of Hares.a
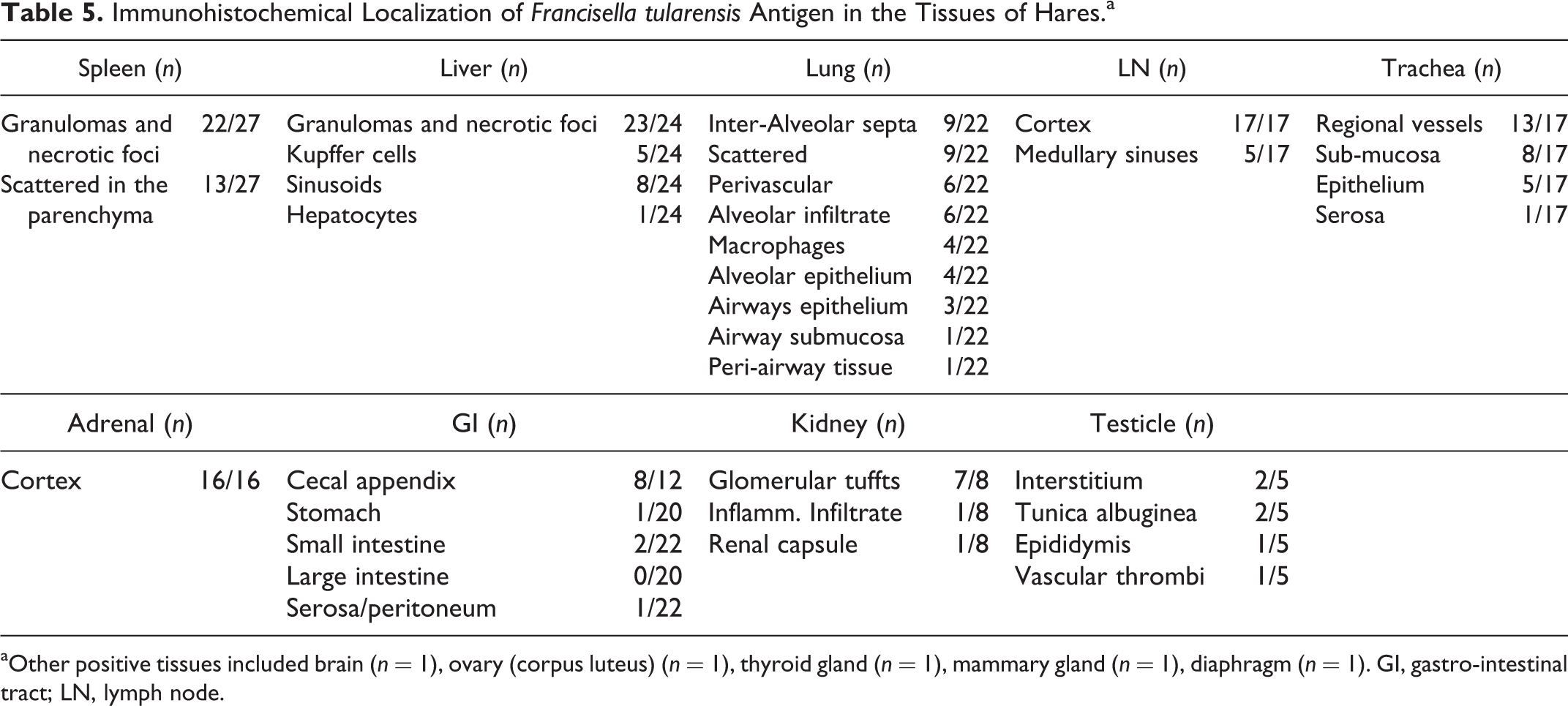
aOther positive tissues included brain (n = 1), ovary (corpus luteus) (n = 1), thyroid gland (n = 1), mammary gland (n = 1), diaphragm (n = 1). GI, gastro-intestinal tract; LN, lymph node.
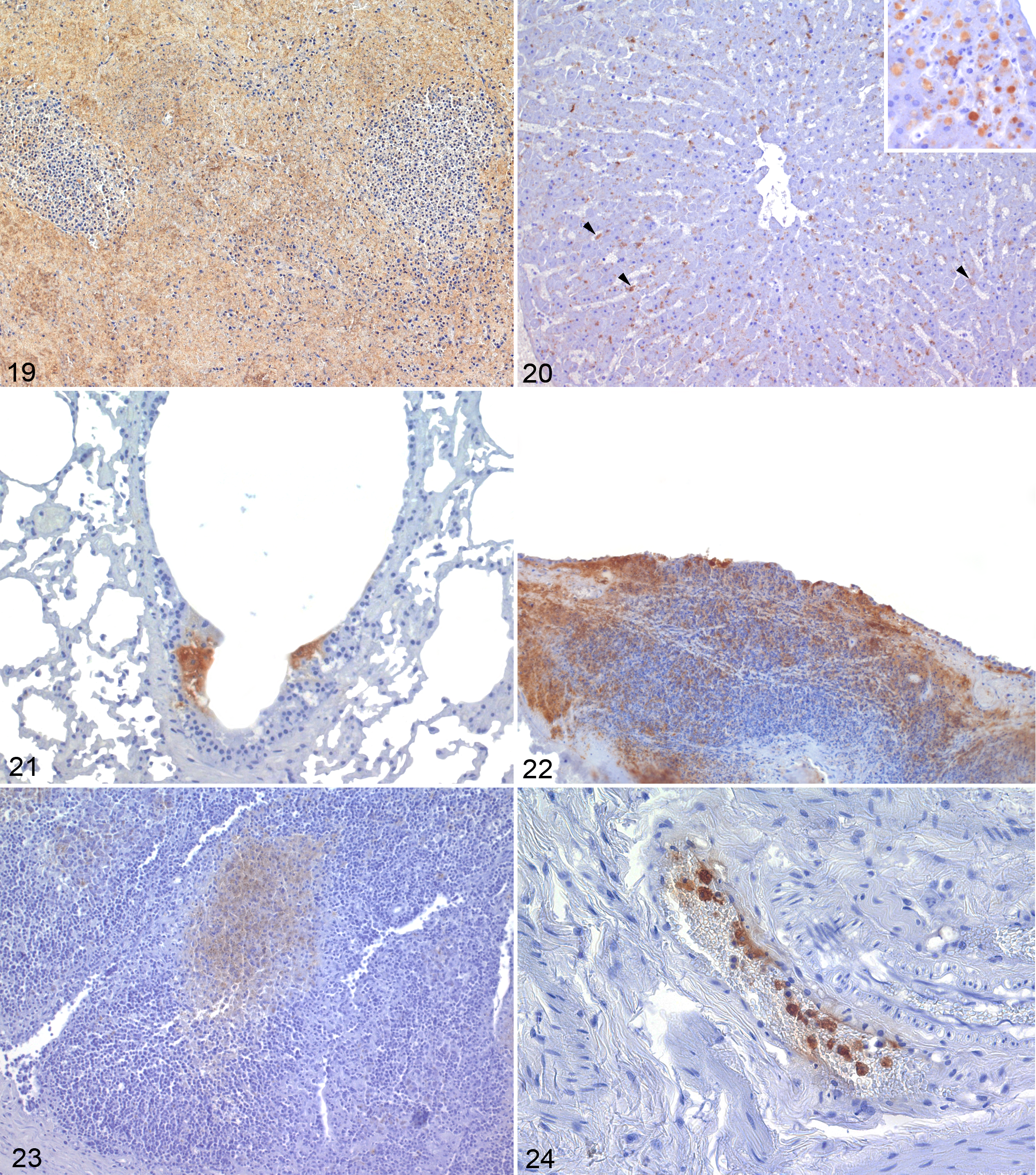
Francisella tularensis infection, Brown hare. Immunohistochemistry for F. tulanensis.
Pathology of F. Tularensis–Negative Hares
Twenty-five of the 53 investigated brown hares were negative for F. tularensis. Of these, 18 were males and 7 were females, 17 were adult and 8 juvenile. Their weight ranged between 0.8 to 4.3 kg, with animals between 3 and 4 kg overrepresented (n = 15). Three out of 25 were emaciated. Twenty-one hares out of 25 were found dead because of blunt trauma (traffic accidents), 3 were culled, and 1 died in a wildlife rehabilitation facility after being found agonic. Significant findings included tracheal edema in 13 of the hares found dead and in 1 of those culled. Other findings in F. tularensis–negative hares included mild interstitial pneumonia (n = 7), granulomatous pneumonia with intralesional lungworms (Protostrongylus sp) (n = 2), parasitic hepatitis with intralesional trematodes (n = 1), and intestinal coccidiosis (n = 2). Finally, pyogranulomatous cellulitis, myocardial degeneration, glossitis, proliferative arteropathy, myocarditis, and preputial adenitis were observed as a single occurrence in distinct individuals.
Bacteriological Findings
All samples were tested by direct PCR and by culture. Twenty-eight hares out of 53 were positive by bacteriological analysis (by PCR and/or culture). PCR was the most sensitive method to detect F. tularensis in organs (Table 1), although in 1 single case the PCR failed to give a positive result, whereas cultures of F. tularensis were recovered from the organs. Results were obtained in less than 4 hours due to lysis of bacteria making DNA extraction redundant. The most appropriate organs to test for F. tularensis were determined to be the major organs, including lung, spleen, liver, and kidney (Table 1). Finally, the in-house selective medium Ftsel enabled twice as many bacteria to be recovered than the routine medium used in diagnostic laboratories (ChocIso medium). According to the RD1 and canSNPs typing systems, all isolates belonged to F. tularensis subsp. holarctica subgroup B.FTNF002-00 11,21,36 ; however, 2 hares harbored isolates of the clade B.12, subgroup B.13, more specifically B.33/34 and B.34/35. 11,21
Discussion
Tularemia appears to be a relevant infectious disease for European brown hares in Switzerland. The relatively high number of infected hares among those collected during our investigation from February 2012 to May 2014 parallels the increased number of human infections reported during the same period, 2 consistent with the situation observed previously in other European regions and the US. 18
European brown hares infected with F. tularensis belonging to the B.FTNF002-00 cluster developed lesions with different morphology and distribution than those infected with the B.13 cluster. 12 In particular, the B.FTNF002-00 cluster was associated with the most classic tularemic lesions in the brown hare, whereas a polyserositis was often observed in B.13 infected hares (Table 6), 12 a difference that might depend on a different cluster-associated tropism besides a potential sampling bias related to the stage of the disease (terminally ill hares in our study and hunted hares in the study by Gyuranecz and colleagues 12 ). Experimental transmission studies with a more balanced sample size are needed to validate and better characterize these findings. Curiously, the only hare in our study showing gross lesions affecting the external surface of the kidney, a common change observed in brown hares infected with B.13 strains, 12 was 1 of the 2 hares infected with a B.13 strain. However, given the very small number of hares in our study infected with B.13 strains, this finding might represent a coincidence.
Distribution of Lesions in European Brown Hares Naturally Infected with Francisella tularensis Subsp. holarctica clades B.FTNF002-00 and B.13.
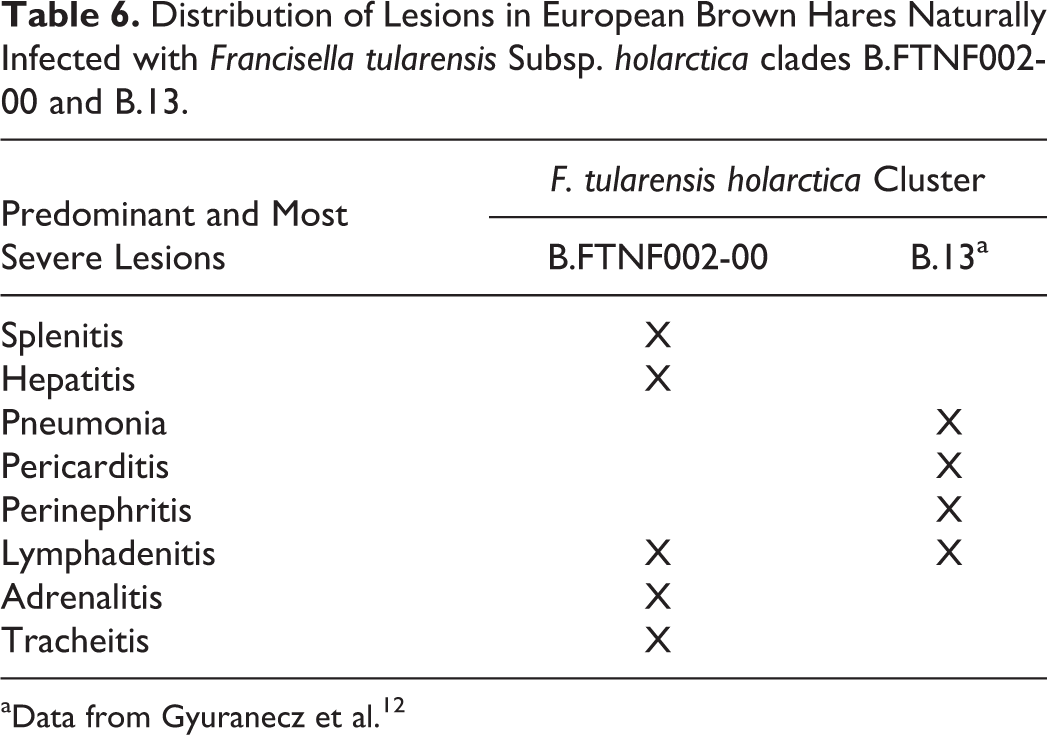
aData from Gyuranecz et al. 12
The bacteriology results paralleled the pathology findings with consistent recovery of the bacterium from the major organs (Table 1). However, F. tularensis was also detected in other organs (ovary, 8/10, 80%; heart, 20/27, 74%; urinary bladder, 17/23, 74%; tonsil, 8/12, 67%; testicle, 9/16, 56%) also when no or only marginal F. tularensis–associated lesions were observed, suggesting the presence of this bacterium mainly in the vascular system of these organs. This hypothesis was confirmed by the IHC results. Additionally, our findings revealed that the in-house selective medium was more sensitive than routine growth media for isolation of F. tularensis.
The thorough pathological investigation of the infected hares allowed us for the first time to gain an insight into the pathogenesis of tularemia in brown hares infected with the B.FTNF002-00 cluster of F. tularensis subsp. holarctica. This is the first characterization of what we interpret as early lesions in the spleen consistent with variably extensive granulomas composed of foamy macrophages associated with small clusters of peripheral plasma cells in the absence of obvious necrosis (Fig. 7) and with likely low bacterial load as suggested by the negative PCR and bacterial culture.
The liver lesions were generally characterized by inflammatory infiltrates expanding from the sinusoids and compressing the surrounding hepatic cords, suggesting a primary access to the liver through Kupffer’s cells in lagomorphs. In rodents, F. tularensis was reported to infect the hepatocytes first. 7
The lymph nodes were consistently affected, similar to what was reported in the infection of the cluster B.13, 12 and were associated with lesions in multiple organs. As is the situation reported from human 16 and animal 32 studies, the most severely affected portions of the parenchyma were those of the subcapsular sinuses and more in general the cortical areas, suggesting that F. tularensis would gain access to lymph nodes through the lymphatic system, likely draining infected organs.
In the GI system, the involvement of the lymphoid follicles of the cecal appendix was not uncommon, suggesting that F. tularensis might also gain access to the lymphatics by this route, likely carried by macrophages or/and dendritic cells. F. tularensis might use this portal to become systemic following infection of the host by the oral route. 15 The association between F. tularensis and gastric ulcers and erosions might suggest that the bacterium contributes to the pathogenesis of this lesion, although the dynamic of this phenomenon is not clear. These lesions might explain in part the severe gastro-intestinal hemorrhages observed in variable hares infected by F. tularensis in winter 19 and also occasionally observed by the authors of this investigation in the brown hare in previous years.
Surprisingly, the adrenal gland, an organ not reported to be affected in brown hares infected with the B.13 cluster, 12 was very commonly affected in hares infected with the B.FTNF002-00 cluster. Similar lesions were described in naturally infected Japanese hares with F. tularensis subsp. holarctica biovar japonica, 24 in non-human primates experimentally infected with F. tularensis subsp. “tularensis”, and in humans that died of tularemia. 25
Lung lesions can be described as either very severe or relatively marginal. The reason for these 2 main distinct patterns (severe bronchopneumonia or mild to moderate interstitial and necrotizing pneumonia) with very different severity is not known, but it does not appear to depend on the bacteria as the distinct forms were observed in hares infected with the same subgroup of F. tularensis (B.FTNF002-00). Our hypothesis is that the different lesions observed in the lung might reflect 2 distinct mechanisms of pathogenesis resulting in alternatively a “primary” or a “secondary” lung complex, the former occurring secondarily to a strong primary local inflammatory (histiocytic) response and the latter secondary to a rapid spread of the bacteria from the airways to the bloodstream/lymphatics and to a later lung involvement following systemic replication and septicemic spread of F. tularensis. In this scenario, the aerosol infection in hares would generally be followed by a rapid systemic spread of F. tularensis using alveolar macrophages as “Trojan horses” that would cross the blood barrier with lesions detectable in the lung only later on after the septicemic spread of F. tularensis (secondary complex). However, in rare cases, the alveolar macrophages might congregate in the airways leading to a blockade, at least temporary, of the bacteria with the development of severe bronchopneumonia (primary complex). This alternate pathogenesis would then reflect on the different F. tularensis tissue colonization and ultimately in the different tissue lesions. As a matter of fact, the hare that showed the most severe bronchopneumonia had very limited lesions in other organs, including the spleen, suggesting an overall, likely temporary containment of the bacteria at its putative port of entry. Individual differences might also clearly contribute to the development of the 2 predicted lung pathological outcomes.
The pathology findings of the lung confirm the involvement of this organ in the disease and its role as one of the possible ports of entry for the bacterium. However, its role as the main route of entry for F. tularensis is questioned by our results. A primary aerosol infection would be more consistent with a bronchopneumonia, but this form of lung disease is detected in only about a third of the Francisella-associated lesions in the lung. Conversely, the majority of the lung lesions are consistent instead with a blood-borne disease. These findings suggest that either the lung does not serve as the primary port of entry for F. tularensis in brown hares, or if it does, then the infection is likely to occur without major local lesions, which would then occur later on with the systemic spread of the bacteria. A transmission study is necessary to clarify this point.
Edema and congestion of the tracheal submucosa were very common in both infected and uninfected brown hares, although generally more severe in the infected animals where necrotic foci scattered along the mucosa could be observed. Interestingly, these lesions were only very rarely seen in the epithelium and generally in contiguity with the deeper lesions, suggesting that the localization of F. tularensis in the trachea is due to systemic vascular spread and not to inhalation in accordance with the features of the lung lesions. The frequent involvement of the trachea was consistent with the bacteriology findings with more than 80% detection of F. tularensis.
The presence of a relatively frequent number of lesions, especially in the cecal appendix (Figs. 6, 23), may also support the hypothesis of a digestive route of infection as is known for humans and other animals. 13,15,19 The presence of multiple IHC-positive lymphoid follicles (Fig. 23) might be compatible with the recruitment of antigen presenting cells containing or carrying F. tularensis antigens within the lymphoid follicles of the cecal appendix followed by their draining to the regional lymph nodes. Experimental infections in mice administered F. tularensis per os showed that this port of entry is compatible with the development of the classic F. tularensis–associated lesions, although with a relatively minor involvement of the lung, which is consistent with our findings. 15 This infection route would allow a relatively easy access to the liver through the mesenteric veins first and ultimately through the portal vein. A direct access to the spleen through the blood stream from a presumptive gastro-intestinal bacterial access would be relatively unusual given the indirect venous flow, but it cannot be ruled out because of a possible retrograde flow to the spleen from the intestinal tract as already postulated for colon cancer metastases in the spleen of humans. 1 Alternatively, the spleen would be affected only later when the systemic spread of the bacterium already occurred.
It is tempting to speculate on the existence of 2 putative routes of infection in the brown hare comprising of the respiratory and the digestive path, opening up completely new epidemiological scenarios for F. tularensis. Independently from the infection route, it appears evident that F. tularensis seeds mainly within the spleen and the liver where it probably multiplies. The presence of lesions in relatively “unusual” organs (eg, the adrenal gland) might represent the beginning of a more systemic and relatively uncontrolled spread of the bacteria massively entering into the bloodstream in the later stages of the disease, causing septic shock and death.
The pathology and presumptive pathogenesis of F. tularensis in brown hares is consistent with the classic lesions observed in infected humans and other animals. Similar to humans, the lesions affect mainly spleen, liver, lymph nodes, and lung. Infection appears to occur either through aerosol and/or the alimentary route, while in contrast to human cases, the ulceroglandular form does not appear to be relevant in brown hares in Switzerland. The presumptive, predominantly respiratory and digestive infection routes, which likely occur in brown hares, make this species an ideal candidate as an animal model for human infection. Additionally, the fact that authorities consider these 2 routes the most likely to occur following a terrorist attack and that they lead to more severe forms of tularemia demonstrates further the potential as an alternative animal model. The relatively lower sensitivity of the brown hare to F. tularensis compared to mice and its similarity to the situation in humans is another ideal feature of F. tularensis infection in brown hares. 4 Additionally, the lung lesions in mice are mostly associated with blood vessels with prominent vasculitis, while in human lung disease, the most common findings are areas of necrotizing and granulomatous pneumonia, similar to what we have observed in brown hares. Brown hares, like humans and different from mice, develop lymphadenopathy. 33 Rabbits are relatively more resistant than brown hares to F. tularensis subsp. holarctica. 8 Finally, although primates develop an even more similar physiopathology to humans than hares, their use is limited due to ethical and practical reasons associated with the large sample sizes required. Brown hares therefore represent an alternative model to mice and could be used as a second animal species in regulatory studies.
In summary, we have presented the pathological and bacteriological data concerning terminally ill, F. tularensis naturally infected brown hares collected in Switzerland. We have shown that (1) tularemia is a relevant cause of death in free-ranging, wild brown hares; (2) the lesions associated with F. tularensis subsp. holarctica belonging to the B.FTNF002-00 cluster are different from those associated with the B.13 cluster; (3) the most likely ports of entry for the bacteria are the respiratory and digestive routes; (4) the lesions observed in brown hares resemble several aspects of those seen in humans infected with F. tularensis; (5) the brown hare represents an alternative experimental animal for modeling the most threatening forms of human infection with F. tularensis; and (6) the in-house selective medium for isolation of F. tularensis is more sensitive than routine growth media. Further studies carried out with experimental infections in European brown hares using both B.FTNF002-00 and the B.13 clusters including oral, respiratory, and subcutaneous inoculations are warranted to conclusively clarify these important aspects of F. tularensis–associated pathology in lagomorphs.
Footnotes
Acknowledgments
We would like to thank the Swiss game wardens who provided the cases investigated in this study; Drs Samoa Giovannini, Mainity Batista Linhares, Ezgi Akdesir, and Roman Meier for their precious help; and Dr Fabio Aloisio for a critical review of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Swiss Federal Office for the Environment (Grant No. 12.0003.KP/L121-0964) and by the Swiss Expert Committee for Biosafety (Grant No. 04.1240.PZ/L064-7521).
