Abstract
The published literature on schizont-“transforming,” or pathogenic theileriosis, in African wild artiodactyls is dated and based on limited information. Here the authors review the taxonomy, diagnosis, epidemiology, hematology, pathology, and aspects of control in various species. Molecular studies based on 18S and 16S rRNA gene sequences have shown that African wild artiodactyls are commonly infected with diverse Theileria spp., as well as nontheilerial hemoprotozoa and rickettsia-like bacteria, and coinfections with pathogenic and nonpathogenic Theileria species are often recorded. Although theileriosis is still confusingly referred to as cytauxzoonosis in many species, the validity of a separate Cytauxzoon genus in artiodactyls is debated. The epidemiology of theileriosis is complex; the likelihood of fatal disease depends on the interplay of parasite, vertebrate host, tick vector, and environmental factors. Roan calves (Hippotragus equinus) and stressed animals of all host species are more susceptible to fatal theileriosis. Even though regenerative anemia is common, peripheral blood piroplasm parasitemia does not correlate with disease severity. Other than anemia, common macroscopic lesions include icterus, hemorrhages (mucosal, serosal, and tissue), fluid effusions into body cavities, lung edema, and variably sized raised cream-colored foci of leukocyte infiltration in multiple organs. Histopathologic findings include vasocentric hyperproliferation and lysis of atypical leukocytes with associated intracellular schizonts, parenchymal necrosis, hemorrhage, thromboembolism, and edema. Immunophenotyping is required to establish the identity of the schizont-transformed leukocytes in wild ungulates. Throughout the review, we propose avenues for future research by comparing existing knowledge on selected aspects of theileriosis in domestic livestock with that in African wild artiodactyls.
Studies on pathogenic theileriosis in African wild artiodactyls are heterogenous with inconsistent findings. The reported pathology is dated and based on limited information. Here we summarize and analyze the available literature to identify research gaps in the current state of knowledge.
Theileria parasites belong to the order Piroplasmida. 102,161 They are obligate intracellular hemoprotozoan parasites that are transmitted by ixodid ticks. 129 The Piroplasmida are classified into 3 families based on the vertebrate cells in which they develop 132 : Theileriidae infect mammals, first developing in leukocytes, then erythrocytes; Babesiidae infect mammals and some birds but develop only in erythrocytes; Hemohormidiidae infect fish and reptiles, where they develop in nucleated red blood cells. 132 The word piroplasm derives from the small pyriform stages that develop in vertebrate erythrocytes. 132
Two piroplasmid genera, Theileria and Babesia, are responsible for some of the most economically important diseases of domestic and wild animals. 102,136,137 Theileria species are known to cause theileriosis in a range of domestic and wild ungulates. 102 While morbidity and mortality rates vary across Theileria and host species, 102 theileriosis is the most significant tick-borne disease to affect domestic ruminants in Africa. 129 Theileria parva alone is suggested to be responsible for the death of >1 million cattle annually and a monetary loss of at least US$300 million per year. 111,126 Aside from this loss, the reductions in growth rate and productivity of infected ruminants and the costs of disease control and treatment all significantly limit the expansion of livestock farming and its contribution to the economy of the continent. 80,94,101,129
Historically, theilerial parasites were researched in African wild artiodactyls because they were considered to be likely reservoirs for T. parva infection in cattle.* This is indeed the case with African buffalo (Syncerus caffer), with buffalo-derived T. parva being the causative agent of Corridor disease, an acutely fatal disease of cattle in eastern, central, and southern Africa, especially south of the Zambezi River. 60,93,129,153,198
The impact of theileriosis on the wildlife industry is less well documented. Although difficult to quantify in terms of its exact economic impact in Africa, wildlife tourism currently generates significant government revenue and foreign exchange, which contribute to employment creation on the continent, particularly in eastern and southern Africa. 40,133,136 As a result, a number of countries now target wildlife tourism as a primary growth sector with significant potential for socioeconomic transformation and poverty alleviation. 133 While African wild ungulates carry diverse hemoprotozoa, such as Babesia, Theileria, and Trypanosoma, as well as rickettsia-like bacteria, in particular Anaplasma and Ehrlichia spp., † they are normally asymptomatic, unless stressed. 28,142,145 However, wild artiodactyls may be carriers of potentially pathogenic Theileria spp. 26,28,74,122,123,128,129,169 and may also experience pathogenic theileriosis with resultant decreases in population size; this is especially pertinent in rare and endangered species, such as roan (Hippotragus equinus) and sable (Hippotragus niger) antelope (Tables 1 and 2). 136,137,175
Pathogenic Theileria Species and Strains Reported in African Wild Artiodactyls.a
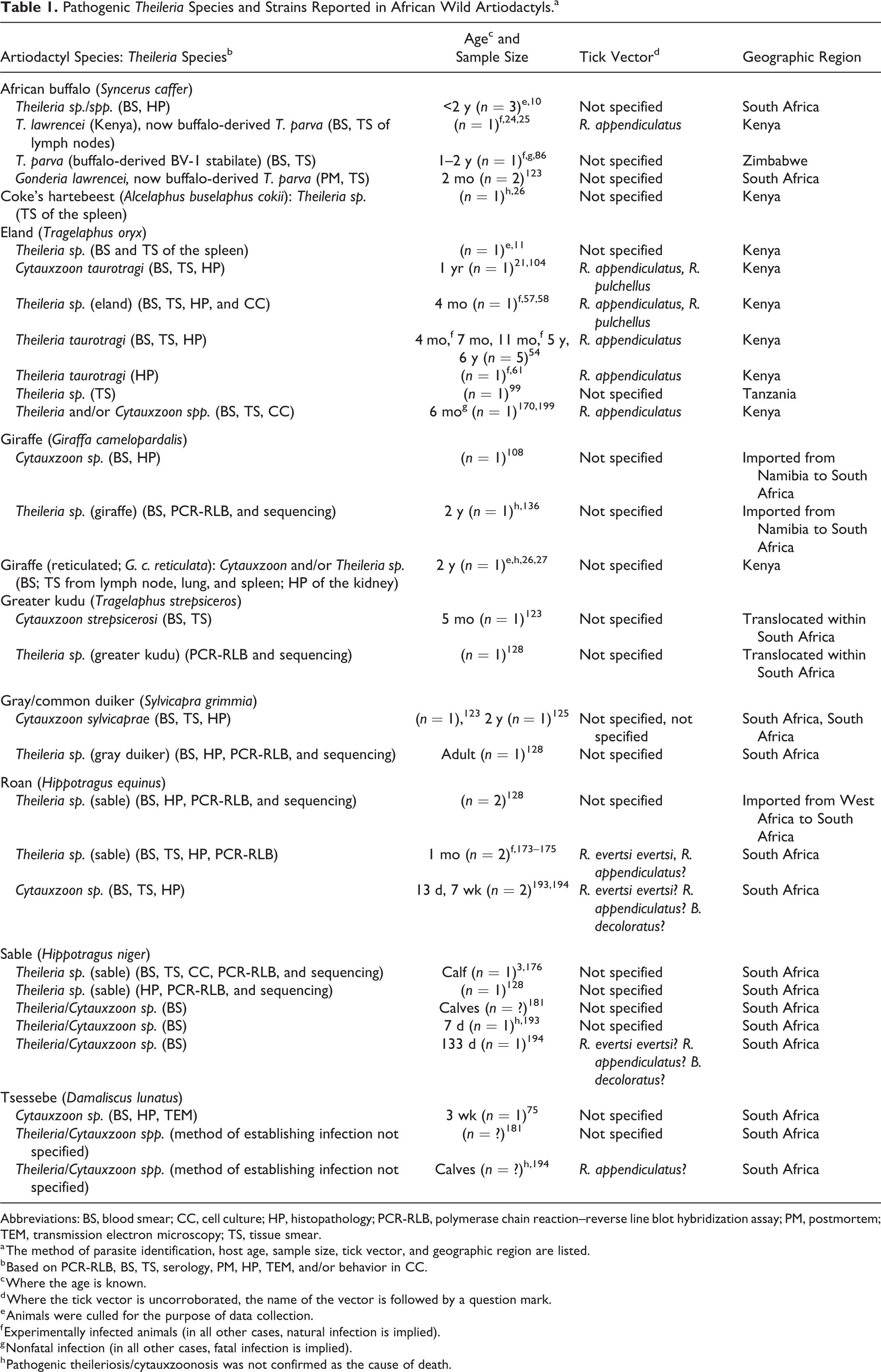
Abbreviations: BS, blood smear; CC, cell culture; HP, histopathology; PCR-RLB, polymerase chain reaction–reverse line blot hybridization assay; PM, postmortem; TEM, transmission electron microscopy; TS, tissue smear.
a The method of parasite identification, host age, sample size, tick vector, and geographic region are listed.
b Based on PCR-RLB, BS, TS, serology, PM, HP, TEM, and/or behavior in CC.
c Where the age is known.
d Where the tick vector is uncorroborated, the name of the vector is followed by a question mark.
e Animals were culled for the purpose of data collection.
f Experimentally infected animals (in all other cases, natural infection is implied).
g Nonfatal infection (in all other cases, fatal infection is implied).
h Pathogenic theileriosis/cytauxzoonosis was not confirmed as the cause of death.
African Wild Artiodactyl Species With Fatal Pathogenic Theileriosis at the Faculty of Veterinary Science, University of Pretoria, South Africa (2007–2018).a

Abbreviations: PCR-RLB, polymerase chain reaction-reverse line blot hybridization assay.
a For which PCR-RLB results are available. The host species, postmortem date, host age, and hemoprotozoa and/or bacteria species identified are listed.
b Dates are given as month.year.
Traditionally, and for the purpose of this review, Theileria species and strains are classified as pathogenic (schizont “transforming”) or benign to mildly pathogenic (“nontransforming”). 102,166 Pathogenic theilerias typically cause disease via schizont-induced alteration of the target mammalian host leukocyte transcriptome to induce blastogenesis, uncontrolled proliferation, widespread leukocyte dissemination, leukocytolysis, acute disease, and death of the vertebrate host. 102,185 Some pathogenic species in cattle have been particularly well researched. Examples include cattle-derived T. parva, which causes East Coast fever (ECF) in eastern, central, and southern Africa, and T. annulata, which causes bovine tropical theileriosis in North Africa, southern Europe, and large parts of Asia. 17,90,94,126 Pathogenic species/strains in African wild artiodactyls include Theileria sp. (sable), 128,175,176 Theileria sp. (gray duiker), 128 and Theileria sp. (greater kudu) 128 (Tables 1 and 2).
In contrast, certain Theileria spp. in cattle (eg, T. velifera and T. sinensis) are considered benign, since they have never been associated with disease. 87,102 Similarly, T. ovis and T. separata are examples of benign, nontransforming species in sheep and goats, although there is significant confusion about the taxonomy of nonpathogenic Theileria species in small ruminants. 89,102,188 These species have also been reported in dogs and diverse wild artiodactyl species (Table 2), 28,102 although the validity of this conclusion is debatable (see Taxonomy section for details).
The classification of Theileria species as pathogenic or benign is an oversimplification because some parasites are not consistently pathogenic or benign within and/or among mammalian host species. For example, although generally regarded as benign parasites of cattle, T. taurotragi, T. mutans, and T. buffeli/orientalis are occasionally pathogenic but typically nontransforming. 87,102 In these instances, clinical disease and even rare mortalities occur due to piroplasm-induced acute hemolytic anemia, which is preceded by insignificant intraleukocytic schizogony. 88,91,102
To complicate matters further, the normally benign T. taurotragi (in addition to pathogenic cattle-derived T. parva and T. annulata) is known to cause bovine cerebral theileriosis/turning sickness in young African shorthorn cattle. 14,33,92 This is an atypical acutely fatal form of pathogenic theilerial infection characterized by significant schizont-associated leukocyte transformation, proliferation, and invasion of the brain and spinal cord. 14,33,92,187 Theileria taurotragi also causes pathogenic theileriosis in eland (Tragelaphus oryx), characterized by schizont-induced pathology in diverse organs and tissues (Tables 1 and 2). 21,54,104
The Theileria genus is characterized by a complex 2-host life cycle (Fig. 1). 129 The life cycle of pathogenic Theileria parasites in domestic ruminants has been comprehensively reviewed. 7,90,94,129,162 Specifically, T. parva and T. annulata life cycles have been deduced from the ultrastructural characterization of their developmental stages in vertebrate and tick host cells. 7,112,129,163 In contrast, there are very few ultrastructural studies on Theileria parasites in wild artiodactyls and their tick vectors. 43,75,200,201 As a result, little is known about the life cycle of Theileria species in these hosts. Studies have shown that asexual reproductive stages occur in the vertebrate host and tick, whereas the sexual phase occurs only in the tick midgut. 129,200 The biphasic life cycle in mammalian hosts includes an intraleukocytic developmental phase (pre-erythrocytic schizogony), which is followed by infection of erythrocytes to form piroplasms. 126,185
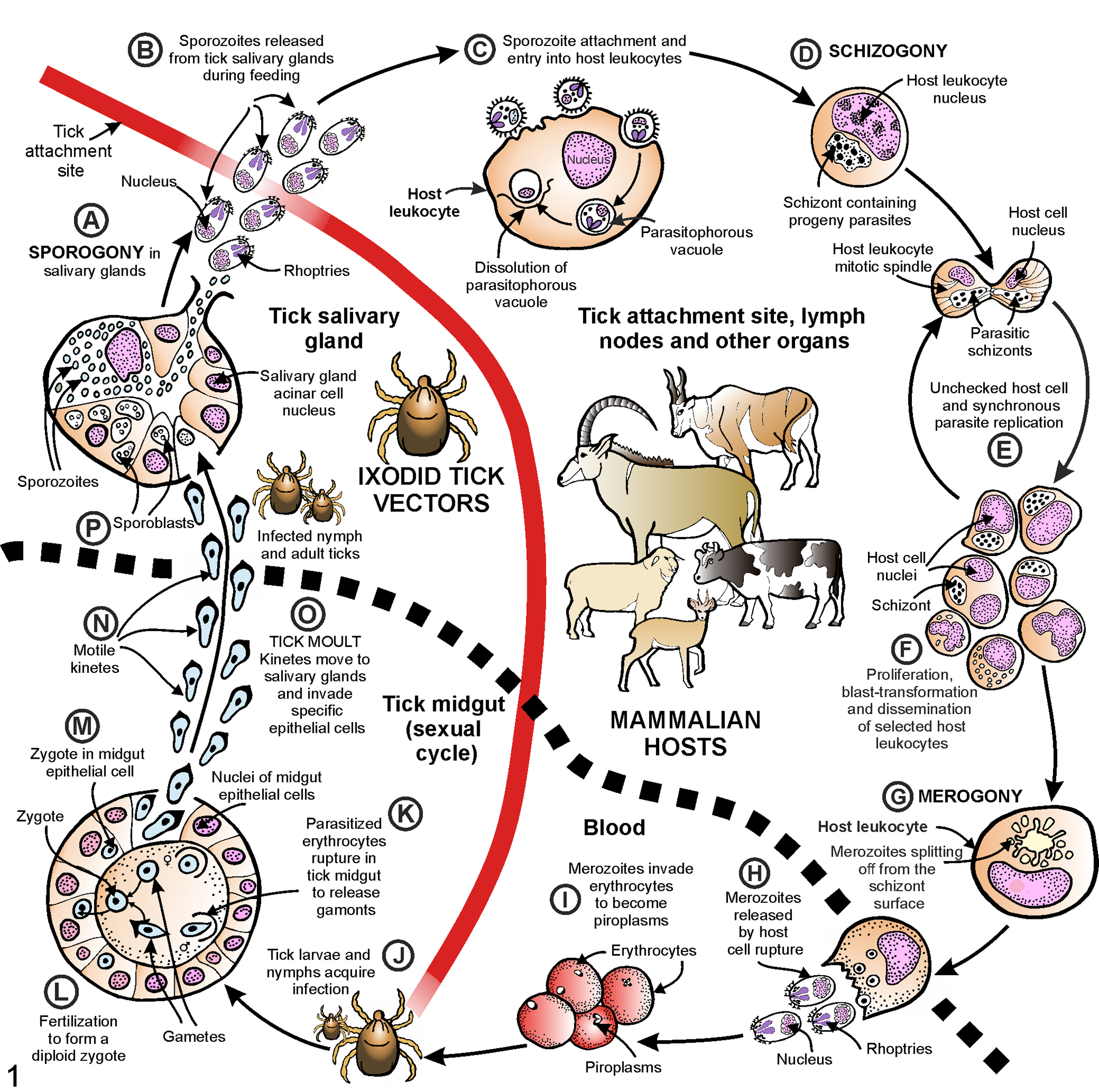
The life cycle of schizont-“transforming” Theileria parasites in domestic ruminants and their tick vectors. (A) During a blood meal, parasite sporogony is completed in specialized salivary gland acinar epithelium of a postmoult nymph or adult tick. (B) After feeding, mature sporozoites are injected into the new vertebrate host. (C) Sporozoites attach to and enter selected host leukocytes within the localized inflammatory reaction of the tick bite. (D) The parasites feed and develop into intracytoplasmic trophozoites, which then undergo schizogony to form multinucleated schizonts, or Koch blue bodies. (E) Schizonts associate closely with the host cell mitotic spindle apparatus, causing uncontrolled host cell and synchronous schizont proliferation with continuous distribution of schizont material to progeny leukocytes. (F) Schizonts induce selected host leukocytes to acquire a pseudo-neoplastic phenotype characterized by reversible blast transformation, self-perpetuating clonal expansion, and dissemination to a variety of lymphoid and nonlymphoid organs and the peripheral blood. These “transformed” leukocytes cause overwhelming systemic inflammation and acute fatal theileriosis. (G) A proportion of intracytoplasmic schizonts undergo merogony with the formation of progeny merozoites. Merozoites radiate in a rosette-like fashion from the surface of schizonts into the host cell cytoplasm, where they mature. (H) Leukocytolysis is induced upon merozoite exit. At this time, leukoproliferation decreases significantly. (I) Merozoites enter host erythrocytes to form pleomorphic intraerythrocytic piroplasms. (J) Tick larvae or nymphs feed on infected vertebrate hosts. (K) Erythrolysis in the tick midgut causes piroplasms to be released, which develop into male and female gamonts. (L) Gametogenesis occurs in the midgut lumen to form male and female gametes, which fuse by syngamy to form a transitory spherical diploid zygote. (M) Zygotes penetrate the intestinal epithelium, where they increase in size over time. Meiotic division occurs in the zygote. (N) In synchrony with the process of tick moulting, zygotes develop into motile kinetes. (O) The kinetes move via the hemolymph to reach the tick salivary glands. (P) In the cytoplasm of specialized acinar epithelial cells, they undergo sporogony to form multinucleated sporoblasts. (A) When the tick engorges on a new host, sporogony is completed and the cycle is repeated. Figure adapted from Bishop et al, 17 Nene et al, 126 and stock image C022/0963. 42
Briefly, after ticks have fed on an infected vertebrate, some piroplasms escape from ingested erythrocytes in the tick midgut. They form female and male gametes, which fuse to produce nonmotile zygotes. The zygotes penetrate the intestinal epithelium, where they undergo meiosis and ultimately transform into motile kinetes. These are transmitted transstadially, ensuring persistence of infection during tick metamorphosis from larvae to nymphs to adults. Kinetes penetrate the gut wall, moving in the hemolymph to reach the salivary glands. Here they produce multinucleated sporoblasts in the cytoplasm of acinar epithelial cells. During feeding, sporogony is completed, and hundreds of infective progeny sporozoites are inoculated with tick saliva into the mammalian host. Sporozoites enter mammalian leukocytes and divide asexually (schizogony) to form multinucleated schizonts. These induce uncontrolled pseudo-neoplastic proliferation of leukocytes, which disseminate to a variety of organs. The ability to induce hyperproliferation and phenotypic atypia in vertebrate host leukocytes sets pathogenic Theileria spp. apart from benign and most mildly pathogenic Theileria spp., as well as other Piroplasmida and apicomplexans. 65,185 Some schizonts undergo asexual reproduction (merogony) to form merozoites. These mature, induce leukocytolysis, and subsequently infect mammalian host erythrocytes to form piroplasms.
Taxonomy
Most of the available information in African wild artiodactyls is dated, and the disease is still confusingly referred to as cytauxzoonosis in many cases (Table 1). 128 Prior to modern molecular assays and methods to identify leukocyte phenotypes, investigators assumed that although Theileria and Cytauxzoon intraerythrocytic piroplasms were indistinguishable, intra- and extracellular theilerial schizonts differed in number and morphology from those of Cytauxzoon species. 7,8,21,26,74,75,108,123,125 It was also assumed that theilerial pre-erythrocytic schizogony was restricted to lymphoid cells (as in ECF) as opposed to lymphoid and other cell types, including histiocytes (or histiocytoid leukocytes) and hepatocytes, as in cytauxzoonosis. 7,21,108,123,125,128 However, since the schizonts of most pathogenic theilerial species induce pseudo-neoplastic transformation of infected and uninfected cells, the prior designation of cellular phenotypes based on morphology alone was simply guesswork. Subsequently, leukocyte phenotyping has shown that certain transforming theilerial species, such as T. annulata and closely related T. lestoquardi in domestic ruminants, are capable of infecting and transforming a variety of leukocytes, including histiocytes. 19,71 As a result, the validity of a separate Cytauxzoon genus in African wild artiodactyl species is uncertain, and some researchers consider Cytauxzoon to be a synonym of Theileria in these species. 23,97,128,181 For the purpose of this review, we refer only to Theileria and theileriosis in African wild artiodactyls.
Phylogeny of Theileria species has largely been elucidated with 18S ribosomal RNA gene sequence analysis, and ever-increasing numbers of Theileria 18S rRNA sequences are available in the public DNA sequence databases. Currently, identification of Theileria parasites in wildlife relies largely on the amplification of full-length or partial 18S rRNA genes and DNA sequence analysis. 47,63,103,128,136,167,184,192 Sequence analyses of nuclear 103 S5 and mitochondrial cytochrome c oxidase 1 genes 146 have been used to distinguish Theileria spp. from closely related to T. parva but have not been widely applied to other members of the genus.
In a phylogenetic tree generated with approximately 1400 bp of the Theileria 18S rRNA gene, Theileria sequences from African wild artiodactyls are found in several clades (Fig. 2). Theileria sp. (sable) is a known pathogen of roan and sable antelope 128,175,176 that groups with theilerias from sheep, African antelope, and dogs. The Theileria sp. (sable) 18S rRNA sequence is most closely related to Theileria species identified in tsessebe (Damaliscus lunatus) 28 and dogs 107 from South Africa. Theileria separata 160 and a Theileria species identified in gray/common duiker (Sylvicapra grimmia) 128 also group closely with Theileria sp. (sable). Full-length 18S rRNA gene sequences are not available for all theilerias that have been identified to date, and more partial Theileria 18S rRNA sequences are available in the public sequence databases. When phylogenetic trees are generated from smaller 18S rRNA sequence fragments, similar groupings are found, although the branching of the clades tends to be slightly different and probably less accurate, as fewer characters are considered. 102 Shorter Theileria 18S rRNA sequence fragments that also group in the Theileria sp. (sable) clade have been identified in red hartebeest (Alcelaphus buselaphus caama), 167 blue wildebeest (Connochaetes taurinus), 192 buffalo, 103 and Rhipicephalus evertsi evertsi ticks collected from gemsbok (Oryx gazella) (Fig. 3). 184
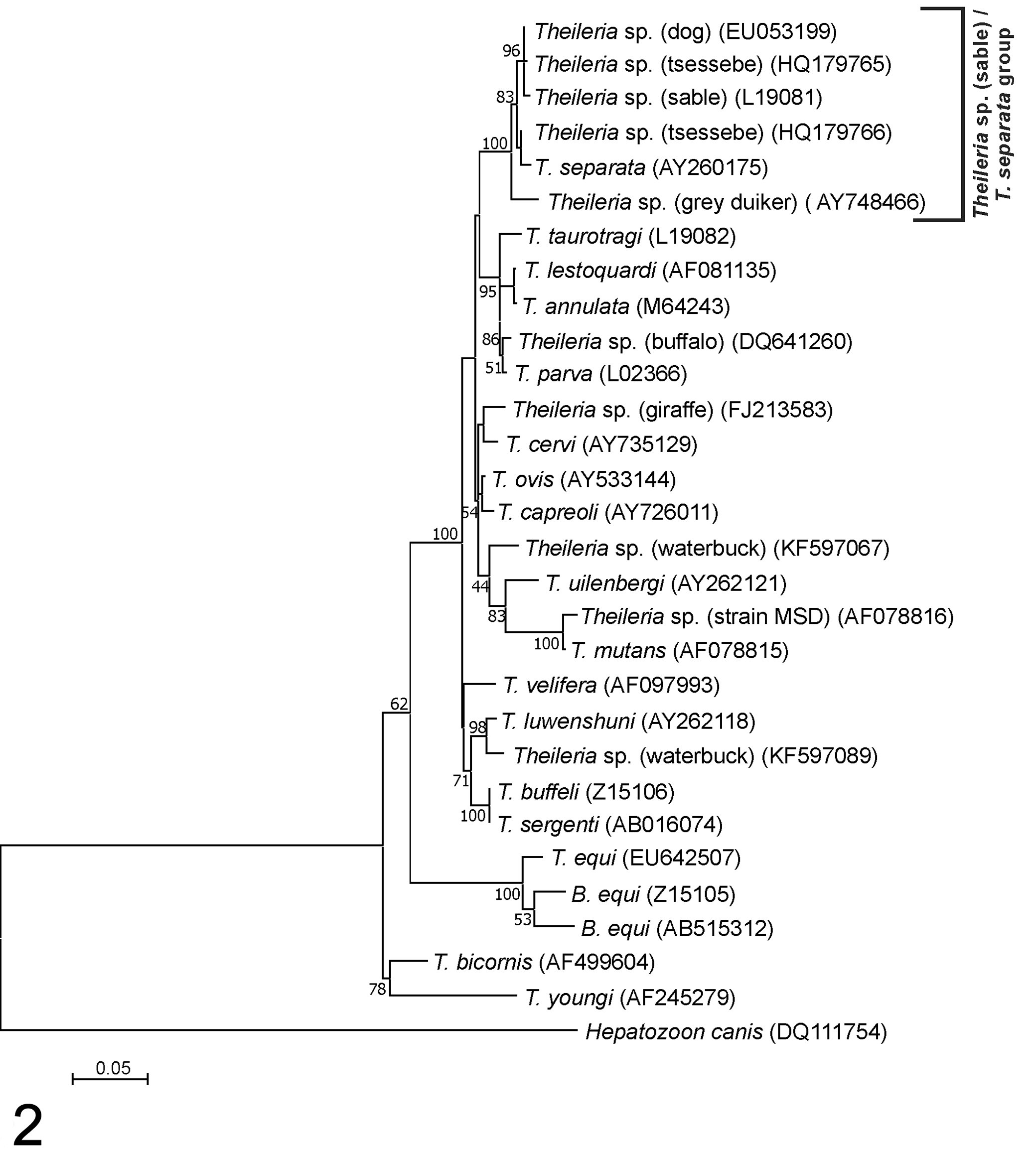
Molecular phylogenetic analysis of Theileria species based on near full-length 18S RNA nucleotide sequence data. The 30 sequences’ accession numbers are shown in parentheses. The evolutionary history was inferred by using the maximum likelihood method based on the Tamura-Nei model. 177 The tree with the highest log likelihood (–5394.93) is shown. The percentage of trees in which the associated taxa clustered is shown next to the branches. Initial trees for the heuristic search were obtained automatically by applying neighbor-join and BioNJ algorithms to a matrix of pairwise distances estimated via the maximum composite likelihood approach and then by selecting the topology with the superior log likelihood value. A discrete gamma distribution was used to model evolutionary rate differences among sites (5 categories; +G, parameter = 0.3259). The rate variation model allowed for some sites to be evolutionarily invariable (+I, 39.51% of sites). The tree is drawn to scale, with branch lengths measured in the number of substitutions per site. All positions containing gaps and missing data were eliminated. There were 1415 positions in the final data set. Evolutionary analyses were conducted in MEGA7. 84
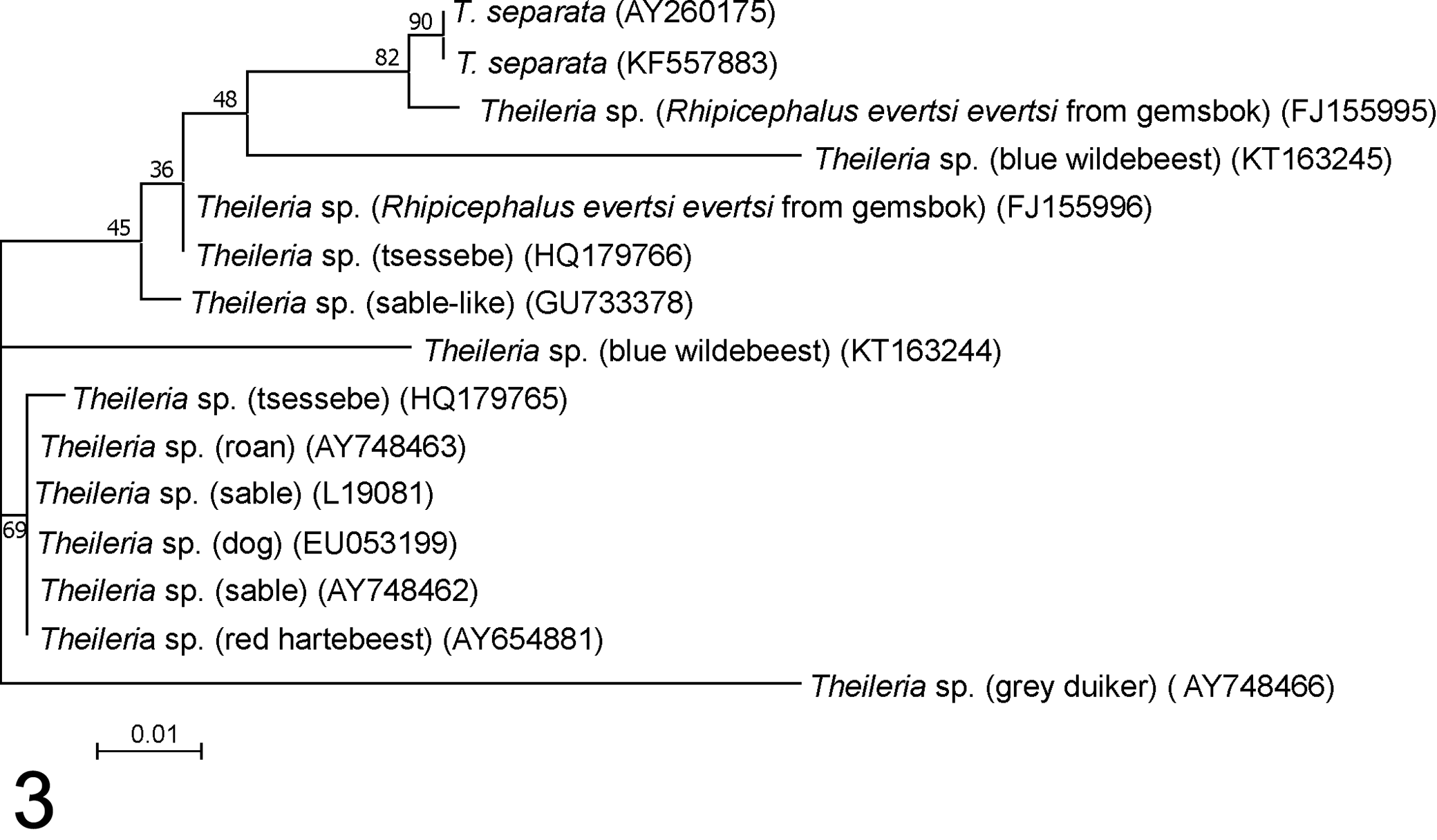
Molecular phylogenetic analysis shows relationships among members of the Theileria sp. (sable) / Theileria separata group based on partial 18S RNA nucleotide sequences. A BLAST search was used to identify 18S sequences in the public sequence databases that are closely related to Theileria sp. (sable); sequence accession numbers are shown in parentheses. The same approach was followed as in Figure 2. Again, the tree with the highest log likelihood (–589.43) is shown. The parameter for the discrete gamma distribution was 0.8124, and 65.88% of sites were invariable. All positions containing gaps and missing data were eliminated from the 15-nucleotide sequences, giving 260 nucleotides in the final data set.
Theileria sp. (sable) has been shown to transform leukocytes 174 –176,204 ; however, T. separata is not known to be transforming, and nothing is known about the transforming capabilities of the other members of this group. Nevertheless, the Theileria sp. (sable) clade is closely related to a clade containing a number of Theileria species that are known to be pathogenic in domestic animals (Fig. 2) and are capable of transforming leukocytes. These include T. parva and T. annulata, as well as T. lestoquardi and T. taurotragi. Another member of this clade, Theileria sp. (buffalo), which infects buffalo, 2,164 is not known to cause disease in domestic ruminants but has been shown to transform lymphocytes. 205 Other well-recognized nontransforming Theileria species that infect cattle and buffalo, T. mutans, T. velifera, and T. buffeli, are more distantly related according to 18S rRNA gene sequence analysis.
Other Theileria 18S sequences that are distinct from Theileria sp. (sable) have been identified in other African wild artiodactyls, including waterbuck (Kobus ellipsiprymnus), 47 greater kudu (Tragelaphus strepsiceros), 128 and giraffe (Giraffa camelopardalis). 136 These are more closely related to nontransforming Theileria spp. such as T. mutans, T. velifera, T. buffeli, and T. ovis from sheep and T. capreoli and T. cervi from deer in the Northern Hemisphere (Fig. 2).
Diagnosis
Differentiation of theileriosis from diseases that cause similar clinical signs, such as pyrexia, anemia, icterus, and enlarged peripheral lymph nodes, requires integration of clinical, epidemiologic, gross pathologic, light microscopic, and molecular data. The most important differential diagnoses for anemia and icterus in African wild artiodactyl species include babesiosis ‡ and anaplasmosis, 4,85,100,120,203 while heavy tick infestations have been associated with blood-loss anemia 113 and nonparalytic forms of tick toxicosis have been associated with pyrexia, anemia, and swollen lymph nodes due to lymphocytotoxicity. 44,121,156,174,180 Animals with nonpathogenic or subclinical infections may have occasional piroplasms in a blood smear and positive polymerase chain reaction (PCR) results. 136,137,142
A reverse line blot (RLB) hybridization assay, originally designed for the simultaneous detection of pathogenic and nonpathogenic Theileria and Babesia species of cattle, 62 was adapted to include probes for the molecular confirmation of Theileria infections, including Theileria sp. (sable), in wildlife. 128 This RLB hybridization assay is based on 2 PCR assays: a Theileria/Babesia primer set and an Anaplasma/Ehrlichia primer set. 28,30,127,128,136,137 The PCR is followed by hybridization of the amplicons to species-specific probes to identify the species. 28,30,127,128 Two conservative probes for each of the Babesia/Theileria and Anaplasma/Ehrlichia groups are added to hybridize any species not identified by the species-specific probe set, so-called catch-all probes (Table 2). 103 A drawback of the RLB is that it will fail to detect novel species if the novel species occurs with a species in the probe set. 103
African ungulates are commonly coinfected with not only a number of theilerial species, both pathogenic and benign, 102 but sometimes also with a variety of nontheilerial hemoparasites (Table 2). 34,102,103,147,167 The significance of these coinfections remains to be investigated, 127 including their potential influence on the sensitivity and specificity of the RLB hybridization assay. 102
To date, Theileria sp. (sable) has been identified via RLB hybridization assays in random blood samples taken from a variety of healthy domestic and wild animals from various regions in southern Africa and Tanzania. 28,128,175,202 However, parasite 18S rRNA amplicons from cattle and buffalo samples that bind to the T. velifera probe also frequently bind to the Theileria sp. (sable) probe, suggesting that the Theileria sp. (sable) probe may cross-react with members of the T. velifera group. 103 Furthermore, 18S rRNA sequence analysis of samples from dogs 107 and tssessebe 28 that hybridized to the Theileria sp. (sable) probe revealed that although they contain a sequence with a 100% match to the Theileria sp. (sable) probe, there are other nucleotide differences that distinguish them from Theileria sp. (sable). A Primer-BLAST 195 analysis revealed that the Theileria sp. (sable) probe also has a 100% match in a Theileria 18S rRNA sequence obtained from a red hartebeest from Namibia 167 and single-nucleotide mismatches with sequences of T. capreoli, T. ovis, and Theileria identified in a variety of other hosts, including spotted deer (Axis axis), sika deer (Cervus nippon), and fox (Vulpes vulpes). Thus, the identification of Theileria sp. (sable) in cases of acute pathogenic theileriosis in African wild artiodactyls other than roan or sable (Table 2) may be due to cross-reactions of the probe with other previously unidentified but closely related Theileria parasites. In such cases, further sequence analysis is recommended to identify the parasite. It is clear that there is a need to develop a more specific Theileria sp. (sable) RLB probe, but until sequence information from closely related parasites from a large variety of wildlife hosts is available, it could be difficult to design a probe that does not cross-react with hitherto unrecognized species.
Other methods used for detection of the pathogenic Theileria species, including conventional PCR assays and real-time assays (reviewed in Mans et al 102 ), are designed to be specific for the target parasites, and while they can be used to identify the target parasite in African wild artiodactyls, they cannot be used to detect novel parasites. An oligonucleotide multiplex suspension microarray has been developed for simultaneous detection of some of the Theileria and Babesia species included in the RLB hybridization assay. 157,158 This xMAP Luminex system shows great promise with the ability to multiplex up to 100 targets in a single reaction, but it will suffer from the same limitations as the RLB when it is applied to the detection of novel parasites in wildlife.
Epidemiology
The epidemiology of pathogenic theileriosis in African artiodactyls is complex and closely linked to the parasite’s indirect 2-host life cycle. Disease severity is determined by the interplay of diverse theilerial parasite, tick vector, and mammalian host factors (Fig. 4). 129 To complicate matters further, the environment, which includes numerous geographic, management, and climatic factors, also modulates the outcome of infection. This interplay is dynamic and can result in stable or unstable states. 64,76,94
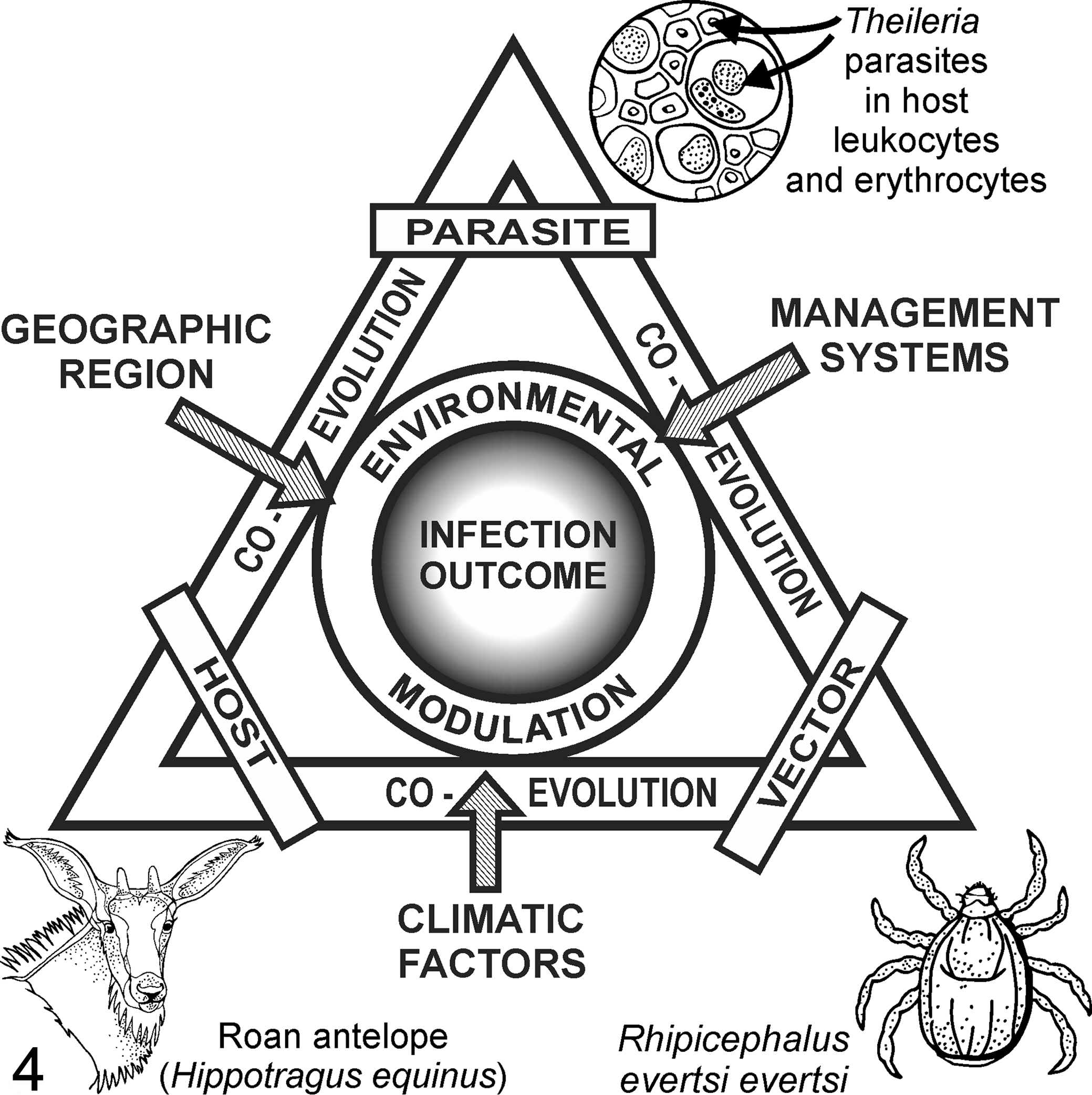
The complex interplay between parasite, vertebrate host, and tick vector factors and diverse environmental factors determines host susceptibility to clinical theileriosis. Parasite factors include variation in virulence among Theileria species and strains. Vertebrate host factors include age and immune status; tick vector factors include the effect of transstadial (vs transovarial) transmission of Theileria parasites on transmission efficiency. Environmental factors include climate (temperature and rainfall), geographic region, and management systems (eg, intensification of wildlife farming).
Endemic stability is a state of host-tick-pathogen interaction where infected ticks with low infection intensity constantly challenge calves and adults in a population, resulting in high levels of protective immunity in the majority of the population and hence low incidence of clinical disease. 64,94 Counterintuitively, anything that reduces the force of infection in such endemically stable areas, including seasonal fluctuations in vector abundance or erratic acaricide application, can lead to a state of endemic instability. 94 This is characterized by sporadic outbreaks of severe clinical disease and fatalities particularly in naïve hosts, such as exotic and crossbreeds of livestock and/or young domestic and wild artiodactyls. 94
There has been a substantial increase in commercial wildlife ranching over the past 2 decades in South Africa. 175,189 The resultant confinement of wild artiodactyls in intensive breeding enclosures has resulted in increased tick burdens and tick-borne diseases, particularly pathogenic theileriosis in roan and sable calves. 128,173,174,189 Roan and sable antelope are rare and valuable, and despite successful breeding under intensive and semi-intensive conditions, calf mortalities due to pathogenic Theileria sp. (sable) infection have negatively affected attempts to establish breeding herds and reintroduce animals into the wild. 174
Parasite Factors
Disease severity is parasite and host associated and depends on a highly specific parasite–host cell relationship that differs among individuals, breeds, and species of hosts and parasites. 1 Although the infective dose of pathogenic Theileria sporozoites is directly related to disease severity in ruminants, 129,175 some Theileria species and strains are more virulent than others, for a variety of reasons that are not yet understood. 102 For example, in cattle, buffalo-derived T. parva causes Corridor disease, a more acutely fatal disease than cattle-derived T. parva, which causes ECF and Zimbabwe theileriosis (January disease). 93 –95,102
Vector Factors
Numerous tick factors influence the transmission success of theilerial parasites in the field. 129,174 Some tick species and strains are better vectors of pathogenic theileriosis than others. 129,131 While a wide variety of hard-bodied ticks (family Ixodidae) have been identified as vectors of diverse theilerial species, 102,129 little is known about the tick vectors of Theileria spp. in African wild ungulates. Specific tick genera that have been identified as vectors of Theileria spp. in domestic ruminants in eastern, central, and southern Africa are Rhipicephalus, Hyalomma, Amblyomma, and Haemaphysalis. 94,129 Rhipicephalus appendiculatus (the brown ear tick) is the most researched of the vectors because it transmits T. parva, the cause of ECF and Corridor disease, both deadly diseases of cattle in Africa. 126,129
Rhipicephalus, Hyalomma, and Amblyomma tick species have also been identified on diverse wild African ungulates, particularly in southern Africa. 68,129,140,186 Of these, R. appendiculatus and R. evertsi evertsi (the red-legged tick), both of which have a wide host range and distribution in Africa, 135 as well as R. pulchellus (the zebra tick, which is largely confined to East Africa 135 ), have been implicated as vectors of benign and pathogenic theileriosis in several wild artiodactyl species (Table 1). These host species include the African buffalo, 9,25,24,53,98,123 eland, 21,57,58,61,104,199 impala (Aepyceros melampus), 73 roan, 173,175,193,194 sable, 194 and tsessebe. 194 However, transmission of Theileria parasites by these Rhipicephalus spp. has been convincingly established only in African buffalo, 25,53,98,123 eland, 21,57,58,61,104,199 and roan antelope. 175
Theileria species are not transmitted transovarially from infected adult ticks to their offspring. 129 Thus, to prevent elimination of theilerial parasites from a tick population, tick vectors need to be in constant contact with a source of parasites to transmit Theileria. 129,173 The source of parasites can be acutely sick, recently recovered, or persistent carrier hosts. 94,129,173
Adult ticks transmit many more Theileria sporozoites than nymphs, 7,129 and a higher infection dose of sporozoites results in more severe disease in domestic and wild artiodactyls. 129,175 Thus, the occurrence of pathogenic theileriosis generally depends on the activity of adult ticks. In southern Africa, adult ticks seek out their vertebrate hosts in the warm, wet summer months (December to March/April). 129 As a result, most cases of theileriosis occur during late summer (Table 2). 173 However, cases of pathogenic theileriosis occur during other times of the year, especially in consistently warmer areas.
Host Factors
Diverse mammalian host factors are important determinants of disease severity. 90,94,129 These factors include age, immunity, genetic susceptibility, and stress. Young ruminants are generally more susceptible than older animals to developing pathogenic theileriosis, 94,173 although transplacental transmission of infection is rare in domestic ruminants and reported but not proven in wild artiodactyls. 89,90,173,175,194 Colostral antibodies are not generally considered to confer protective immunity against Theileria spp. to young ruminants. 90,173 Recently, however, it was shown that colostral transfer of maternal leukocytes including CD8+ cytotoxic T lymphocytes does occur. 182 This should confer a degree of protection to neonates, albeit for an unknown period. Evidence suggests that T. parva and, to an extent, T. annulata infections in cattle and T. lestoquardi infections in sheep (the cause of malignant ovine theileriosis) are largely curbed by an effective cell-mediated immune response. 45,48,50,110,116,154,178,185 Particularly in T. parva infections, protective immunity is principally mediated by parasite-specific major histocompatibility complex class I–restricted, CD8+ cytotoxic T lymphocyte–mediated killing of infected leukocytes. 45,110,154,185 Schizont-infected leukocytes also stimulate a parasite-specific major histocompatibility complex class II–restricted CD4+ T lymphocyte response. 45,110,154
In some antelope species (eg, West African/Western roan antelope [Hippotragus equinus koba]), hypochromic poikilocytic anemia of neonates, which is thought to develop due to delayed or asynchronous perinatal switching over from fetal to adult hemoglobin, may predispose the young of these antelope species to pathogenic theileriosis. 139,173 It has been proposed that this physiologic anemia contributes to the motionlessness of Hippotragini calves during the early neonatal period, when they are sequestered from the rest of the herd for 2 to 4 weeks in tall grass or brush. 139,173 However, whether it does predispose the calves of some species to pathogenic theileriosis requires further investigation.
A geographically separated host population may be naïve to parasites that infect the host in another area. For example, Western roan calves in South Africa are more susceptible to lethal theileriosis (Theileria sp. [sable]) than other roan subspecies (eg, Hippotragus equinus equinus), because H. e. equinus probably evolved with Theileria sp. (sable) in eastern and southern Africa. 173 Similarly, translocated animals may develop disease signs due to exposure to novel Theileria species or strains at the new location. 28,127,128 Furthermore, Theileria epidemics tend to occur when asymptomatic carrier hosts that are persistently infected with Theileria parasites are moved to areas previously uninfected with those particular parasites but where suitable vectors are available to initiate a transmission cycle. 94,173
A number of stressors also increase the susceptibility of ruminants to pathogenic theileriosis. 54,142,194 Stress due to malnutrition, 173,194 translocation, 28,127,128 and high endo- and/or ectoparasite burdens 173 can also influence the outcome of theilerial infections due to altered immune function and response to infection. 67,105
Environmental Factors
Climatic, sociodemographic, economic, and political factors play a role in disease severity, prevalence, and diversity. 46 ECF, bovine tropical theileriosis, and malignant ovine theileriosis in domestic livestock occur in specific geographic regions related to the distribution of suitable tick vectors. 89,90,94 Tick distribution depends on climate (temperature and rainfall), suitable vegetation, and host availability. 94,134 Changes in temperature and rainfall, as well as human population expansion, with associated changes in land use can affect the geographic distribution, density, and diversity of tick vectors and their vertebrate hosts. 28,41,135
Tick habitats are also affected by livestock management systems, including methods of grazing management and tick vector control. For instance, overstocking results in social stress, increased tick burdens, and, occasionally, ensuing tick-induced immunosuppression. 44,79,173 The balance among tick, Theileria, and host can also be disturbed by failure to correctly rotate acaricide formulations and the application of incorrectly diluted formulations, resulting in the evolution of multiacaricide-resistant ticks. 190
The recent explosive growth in the wildlife trade has significantly influenced land utilization, bringing wild and domestic animals into increasingly closer proximity. 41 This has facilitated the transmission of ticks and tick-borne pathogens, including Theileria spp., between domestic and wild ungulate species, threatening the health of both. 13,41 Furthermore, political instability may lead to erratic implementation of tick-borne disease control measures and increased illegal movement of ruminants, their hemoparasites, and ticks. 94 This could result in outbreaks of clinical disease and fatalities in susceptible ungulates. 28
Apart from limited data on tick vector identification in roan and other wild artiodactyls, little is known about the epidemiology of pathogenic theileriosis in wild antelope. 28,174 As a result, most existing strategies for limiting clinical disease in susceptible wild artiodactyls rely on anecdotal evidence and are unsubstantiated.
Hematology and Cytology
Information on the hematology and tissue cytology findings associated with pathogenic or transformative theileriosis in African wild artiodactyls is summarized in Table 3. Typical intraleukocytic theilerial schizonts (Koch blue bodies) have been reported in blood and buffy coat smears stained with Romanowsky stains from a variety of African wild ungulates with natural and experimental theileriosis. 128,174,175,181,193,194 However, there is only a single description of theilerial schizont morphology in a Giemsa-stained blood smear from a sable calf in southern Africa. 181 In this report, schizonts within host leukocytes were similar to those described in cattle with ECF 94,123 and contained variable numbers of chromatin granules/nuclei dispersed in a shared pale blue or neutral cytoplasm (Fig. 5). Similar to ECF, schizont nuclei were smaller but more numerous in the micro- versus the macroschizont stage of parasite development in sable peripheral blood leukocytes. 181
Hematology and Blood Smear Findings in African Wild Artiodactyls With Pathogenic Theileriosis.

Abbreviations: EI, experimentally infected NI, naturally infected.
a Unless indicated differently, infection was natural.
b Nonfatal infection.
c Erythrocyte anisocytosis, basophilic stippling, polychromasia, Howell-Jolly bodies, normoblasts, and/or reticulocytes observed in blood smears.
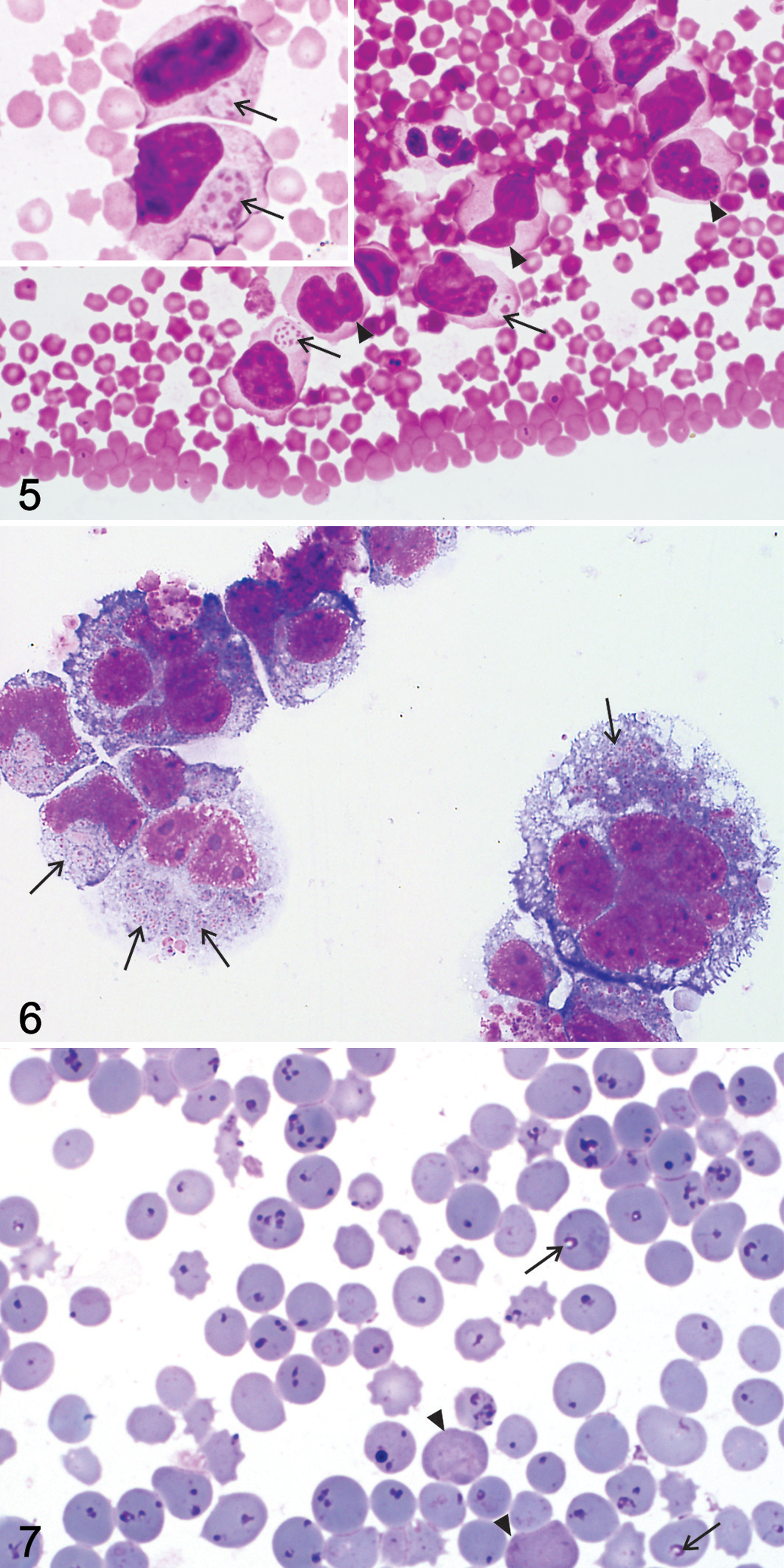
Theilerial schizonts have been well described in organ impression smears and cultured leukocytes (Fig. 6) sourced from peripheral blood and lymph node biopsy samples from diverse healthy and sick artiodactyls.** The reason is that, before the advent of molecular diagnostic techniques, microscopic schizont morphology and infected leukocyte phenotype (without immunophenotypic characterization) formed the basis for differentiation between Theileria and Cytauxzoon genera and among Theileria species. 21,23,75,108,123,125,193,196 The reported average schizont size, number of nuclei per schizont, size of schizont nuclei, and number of intraleukocytic schizonts vary considerably among African wild artiodactyl species with pathogenic theileriosis, but extra- and intraleukocytic schizonts in tissue smears were likened to the schizonts observed in cattle with T. parva (ECF). †† Reasons that have been proposed for the wide variation in domestic and wild artiodactyls include differences in schizont maturity, 123,125 stage of disease (with fewer parasites visible in acutely fatal infections), 93 and differences in theilerial parasite pathogenicity. 93
The identity of the schizont-transformed leukocytes in blood, buffy coat, and lymph node smears from wild ungulates with pathogenic theileriosis has been the subject of debate for decades. These transformed leukocytes have been variably referred to as lymphoblasts, lymphoblastoid cells, or lymphoid cells in African buffalo, 22,197 eland, 54,57,58 and roan antelope (Fig. 5). 128,173 –175 A monocyte-macrophage derivation for these leukocytes was also suggested in the gray/common duiker, 123,125,128 greater kudu, 123 and roan and sable calves. 174,175,193,194 In addition to parasitized mononuclear macrophage-like host cells, infected multinucleated giant macrophage-like cells with ≥17 nuclei were reported in tissue and/or organ smears from a gray/common duiker 123,125 and a greater kudu in South Africa. 123 In roan calves, infected and uninfected leukocytes were often highly atypical and hard to distinguish from neoplastic cells (Fig. 6). 174 They possessed irregularly indented, lobed, and, rarely, multiple nuclei, multiple prominent nucleoli, mitoses, and moderate to ample and occasionally vacuolated basophilic cytoplasm. 174 Schizont-transformed leukocytes in blood and organ smears have not yet been immunophenotyped in any African wild artiodactyl species, so their identity remains unknown.
Several studies have investigated the effects of pathogenic Theileria parasites on the leukocyte count in naturally and/or experimentally infected African buffalo, 9 eland, 54,57,58 greater kudu, 123 and juvenile Western roan antelope. 174 Leukocytosis, leukopenia, and normal leukocyte counts have been reported within and among selected wild ungulate species.
There is a surplus of detailed information on the morphology of theilerial piroplasms and the percentage of piroplasm-infected erythrocytes estimated in blood smears in healthy ‡‡ and sick wild African ungulates.*** In many of these reports, the authors observed marked piroplasm pleomorphism, with shapes varying from round or signet ring shaped to elongated/bacillary, anaplasmoid, comma shaped, and oval/ovoid as well as paired and tetrad/Maltese cross forms (Fig. 7). Theilerial piroplasms occasionally possessed a crescentic or cap-shaped dense nucleus on the margin of round piroplasms and at the wider extremity of ovoid to pear-shaped/pyriform parasites; the cytoplasm was not often visible in the bacillary, comma-shaped, or anaplasmoid forms. 32,35,123,183 In addition, theilerial piroplasms have been described as small, generally measuring 0.5 to 2.5 µm in width or diameter (depending on their shape) and up to 2.5 µm in length. 75,118,123,183 Between 1 and 12 piroplasms were observed within a single erythrocyte in diverse wild artiodactyl species, with 1 to 4 (tetrads) being most commonly observed. 21,75,118,123,125
Percentages of piroplasm-infected erythrocytes that have been reported in sick or fatally infected wild artiodactyls are highly variable within and among species. The erythrocyte infection rate was often <50% but was occasionally as high as 90% or <3%. The level of piroplasm infection may not be related to the severity of disease in wild antelope. In cattle with acutely fatal Corridor disease, parasites are often scarce or even absent in blood and tissue smears. 8,22,26,93,124
Regenerative anemia of variable severity, largely based on the observation of mucous membrane pallor and evidence of a regenerative erythrocyte response in blood smears, 123 has been reported in eland, 54 giraffe, 108 greater kudu, 123 and Western roan antelope 173,174 with pathogenic theileriosis. In experimentally infected eland 54,57 and a naturally infected greater kudu, 123 parameters such as red cell count, packed cell volume, and hemoglobin concentration have been found to decrease after Theileria infection. However, as the algorithm used to separate erythrocyte and platelet populations on cell counters and analyzers may not have been appropriate for the species, the reported red cell counts may not be accurate.
Thrombocytopenia 173 and prolonged bleeding times 128 have also been reported in Western roan calves with terminal Theileria sp. (sable) infection. However, these findings are not validated in roan antelope, and the effects of pathogenic theileriosis on platelets have not been investigated in other wild antelope species. Although hematologic reference values exist for a variety of wild artiodactyl species in captivity and in the wild, 39,149 –152 testing methods need to be standardized to allow for accurate inference over diverse species. 165
Macroscopic Pathology
Gross lesions reported in wild artiodactyls with pathogenic theileriosis are very similar to those in domestic livestock with ECF (T. parva), bovine tropical theileriosis (T. annulata), and malignant ovine theileriosis (T. lestoquardi). 19,35,72,82,89,90,94 Tissue lesions are predominantly the result of focal to diffuse infiltration with hyperproliferative, schizont-transformed leukocytes and associated parenchymal necrosis, hemorrhage, and inflammation (see Histopathology section for details). Table 4 lists the salient macroscopic lesions reported in diverse wild ungulate species.
Macroscopic Pathology Observed in African Wild Artiodactyls With Fatal Pathogenic Theileriosis.a

Abbreviations: EI, experimentally infected; NI, naturally infected.
a Macroscopic lesions are the result of focal to diffuse tissue infiltration with hyperproliferative schizont-transformed leukocytes and associated parenchymal necrosis, hemorrhage, and inflammation.
b For all cases where EI/NI is not specified, natural infection is implied.
Anemia and icterus were commonly observed (Fig. 8), although severity was variable within (eland 21,54 and roan 174,193 ) and among species. ††† Severe carcass congestion was reported only in a gray/common duiker with fatal theileriosis. 123,125
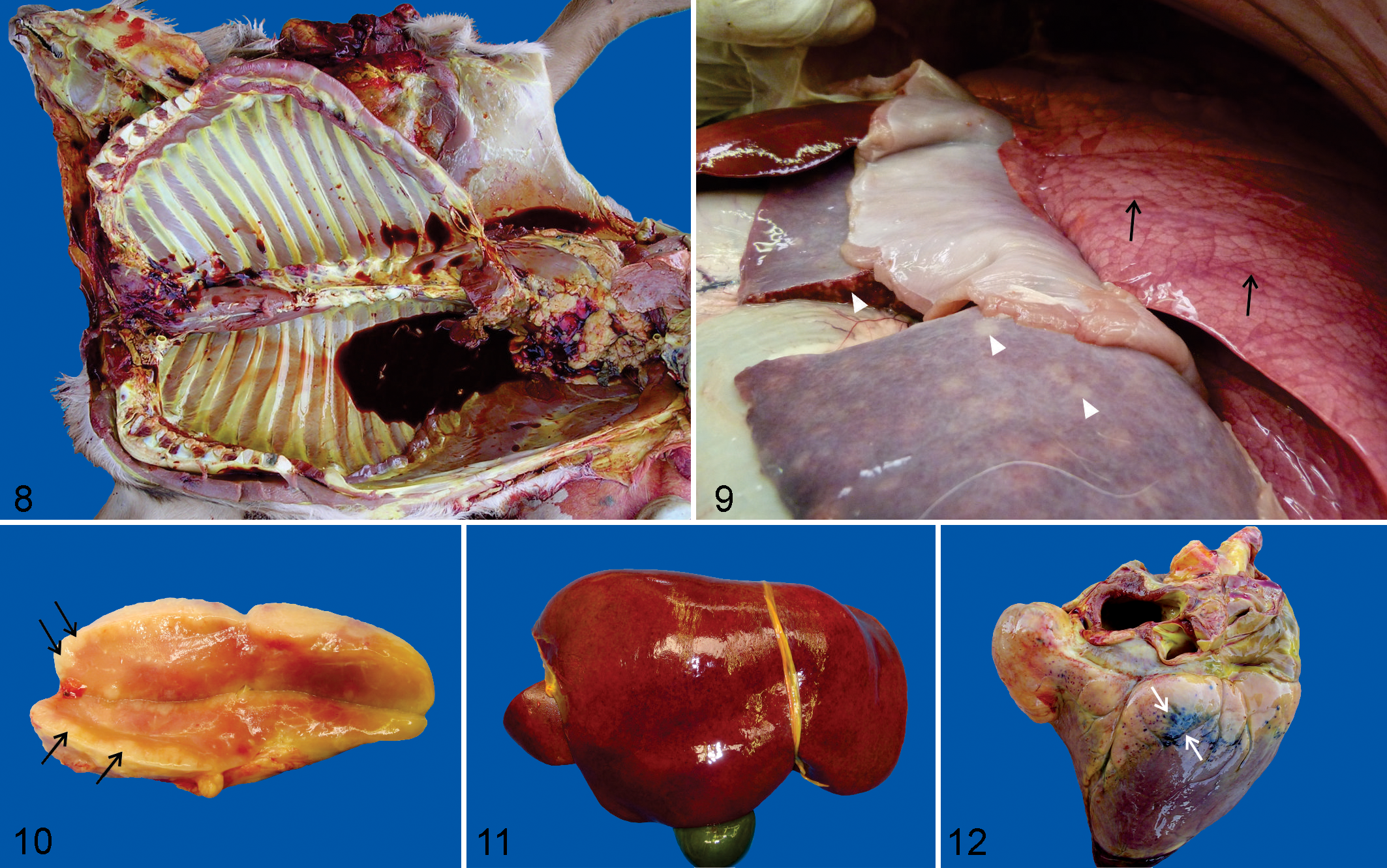
Hemoglobinuria, due to intravascular hemolysis, and bilirubinuria are features of pathogenic theileriosis in domestic livestock, particularly bovine tropical theileriosis and malignant ovine theileriosis. 70,77,89,90 Hemoglobinuria is not a common feature of ECF in cattle 72,94 but was observed in eland, 21,104 giraffe, 108 and roan antelope 193 with fatal pathogenic theileriosis. Bilirubinuria in the absence of hemoglobinuria also occurred in Western roan calves with fatal Theileria sp. (sable) infection. 174 Thus, significant intravascular hemolysis and hemoglobinuria are not consistent features of pathogenic theileriosis in ruminants, although this requires further investigation in African wild artiodactyls.
Splenomegaly and widespread lymphadenomegaly have been frequently reported across diverse species, mainly due to focal/nodular or diffuse leukocyte proliferation (Figs. 9, 10). ‡‡‡ Lymphadenomegaly due to edema was described in a tsessebe, 75 and widespread lymph node edema in the absence of lymphadenomegaly was observed in a giraffe. 108
Prominent lung edema was another frequent but inconsistent 54 observation in diverse species (Fig. 9). 21,54,57,123,125,128,173,175 Diffusely hyperemic, mottled lungs with increased consistency due to widespread atypical leukocyte infiltration and associated hemorrhage and inflammation, typical for interstitial pneumonia at the gross pathology level, has been reported only in a tsessebe. 75
Similar foci of leukoproliferation and hemorrhage in the kidneys, with the macroscopic appearance of interstitial nephritis, which were initially and incorrectly described as renal infarcts in early reports describing the gross appearance of the kidneys in fatal ECF in cattle, 51,81,179 were also reported in eland. 99,123 Affected wild ungulates also commonly showed hepatomegaly due to widespread infiltration of schizont-transformed leukocytes with associated hepatocyte injury and inflammation (Fig. 11). 75,108,123,125,174 Nodular leukocyte infiltration was associated with a multinodular liver (“tumor hepatis”) in an eland and a gray/common duiker. 123
Diarrhea was a common finding in eland with pathogenic theileriosis. 11,54,123 It has been typically associated with marked thickening of the abomasum and small intestine due to extensive leukoproliferation with secondary erosive to ulcerative gastroenteritis and mucosal barrier disruption. 54 Multifocal abomasal erosions were also described in 1 of 2 gray/common duikers that succumbed to pathogenic theileriosis. 123
Serosanguinous effusions into body cavities (pericardium, thorax, and abdomen) were reported in multiple wild ungulate species.**** Anasarca has been reported in eland 21 and a greater kudu. 123
Petechiae and ecchymoses were commonly observed on serosal and mucosal surfaces and the subcutis and in selected organs, including peripheral and visceral lymph nodes, liver (Fig. 11), heart (Fig. 12), lungs, kidneys, urinary bladder, gall bladder, gastrointestinal tract, and skeletal muscles. 21,54,75,108,123,128,173,174 Serosal suffusive hemorrhages have been observed in giraffe 108 and Western roan carcasses 174 ; diffuse enteric hemorrhage was also reported in the latter. 174
In wild artiodactyls with pathogenic theileriosis that lacked other diseases, poor body condition was ascribed to Theileria-associated anorexia and dehydration (Western roan 174 ), diarrhea due to infiltration of the intestinal wall with atypical leukocytes (eland 54 ), and chronic infection (eland 54 and Western roan 174 ). However, in a greater kudu and gray/common duiker that died of schizont-“transforming” theileriosis, other possible causes of weight loss, including the stress of recent translocation, climate (eg, drought), and feed scarcity, were not discussed. 123,125
Poor body condition might predispose wild artiodactyls to severe tick infestation and therefore increase their likelihood of contracting pathogenic theileriosis, 113,194 although this assertion is unproven. Wild ungulates in good and poor body condition that succumbed to pathogenic theileriosis were reportedly variably tick infested. 54,173 Moreover, the number of ticks observed at necropsy may not be a true reflection of the premortal tick burden, because ticks start to drop off carcasses soon after death. 173
Detailed macroscopic descriptions are needed in wild artiodactyls that die due to schizont-“transforming” theileriosis. In particular, the association between pathogenic theileriosis and poor body condition requires in-depth investigation across diverse species. ECF, malignant ovine theileriosis, and, to a lesser extent, bovine tropical theileriosis have all been associated with emaciation and, in rare cases, also chronic infection or a state of partial recovery from initial infection. 89,90,94 These cases are characterized by emaciation, anemia, generalized edema, lymphoid atrophy, and secondary infections due to immunosuppression. 71,89,94 Currently it is not known whether a chronic debilitating form of pathogenic theileriosis exists in wild artiodactyls.
Histopathology
The histopathology of pathogenic theileriosis is well described for ECF in cattle 6,35,45,82,94,123 and bovine cerebral theileriosis/turning sickness, 14,33,155,187 but there are comparatively few descriptions of the histopathology of bovine tropical theileriosis 19,90 and malignant ovine theileriosis 71 in domestic livestock. Similarly, comprehensive histopathologic descriptions of pathogenic theileriosis are scarce in African wild artiodactyls. Lesions have been best described in the African buffalo, 10 eland, 21,54,104 giraffe, 108 gray/common duiker, 123,125 and tsessebe, 75 although, in most cases, only a single animal of each species was necropsied. 21,75,104,108,125
Details of the histologic lesions described in transforming theileriosis across diverse African wild artiodactyl species are listed in Table 5. The following is a summary of key lesions reported. The pivotal lesion is, similar to that in cattle with ECF, multifocal to diffuse infiltrates of target cells in multiple organs. †††† Common secondary lesions include congestion, multifocal disseminated hemorrhages, foci of parenchymal necrosis, and inflammation. For the purpose of this review, target cells refer to host cells that may or may not be schizont infected but are induced to undergo pseudo-neoplastic transformation (as evidenced by cellular atypia), as well as hyperproliferation and widespread dissemination. 31 Target cell phenotype and location in various species are summarized in Table 6. Similar to ECF, widespread lysis of initially proliferative target cells has been reported. 54,75,108,125
Significant Histopathologic Findings in African Wild Artiodactyls With Fatal Pathogenic Theileriosis.

Abbreviations: EI, experimentally infected; GIT, gastrointestinal tract (abomasum and intestine); LN, lymph nodes; MPS, mononuclear phagocytic system; NI, naturally infected.
a For all cases where EI/NI is not specified, natural infection is implied.
Target Cell Phenotype and Location and Parasitic Schizont Location in Histologic Sections From African Wild Artiodactyls With Fatal Pathogenic Theileriosis.

Abbreviations: EI, experimentally infected; NI, naturally infected.
a For all cases where EI/NI is not specified, natural infection is implied.
b Target cells refer to host cells that may or may not be schizont infected but are induced by parasitic schizonts to undergo pseudo-neoplastic transformation (as evidenced by cellular atypia), as well as hyperproliferation and widespread dissemination. 31
As in tissue smears, large mononuclear target cells that have been described as lymphoid and/or macrophage like in histologic sections were characterized by round to bean-shaped/reniform eccentrically situated nuclei, multiple prominent nucleoli, and ample homogenous to vacuolated amphophilic cytoplasm (Fig. 13). 54,75 Multinucleated giant cells (thought to be macrophage derived) have been inconsistently reported (Fig. 14). 21,75,108,123,125 Parasitized mono- or multinucleated target cells in the liver were thought to be severely hypertrophied hepatocytes. 21,108 Tissue-resident macrophages, including alveolar macrophages, Kupffer cells, and/or splenic macrophages, were commonly identified as target cells. 10,75,108 However, to date, no immunophenotyping has been done on tissue sections to further investigate the identity of the target cells in African wild ungulates.
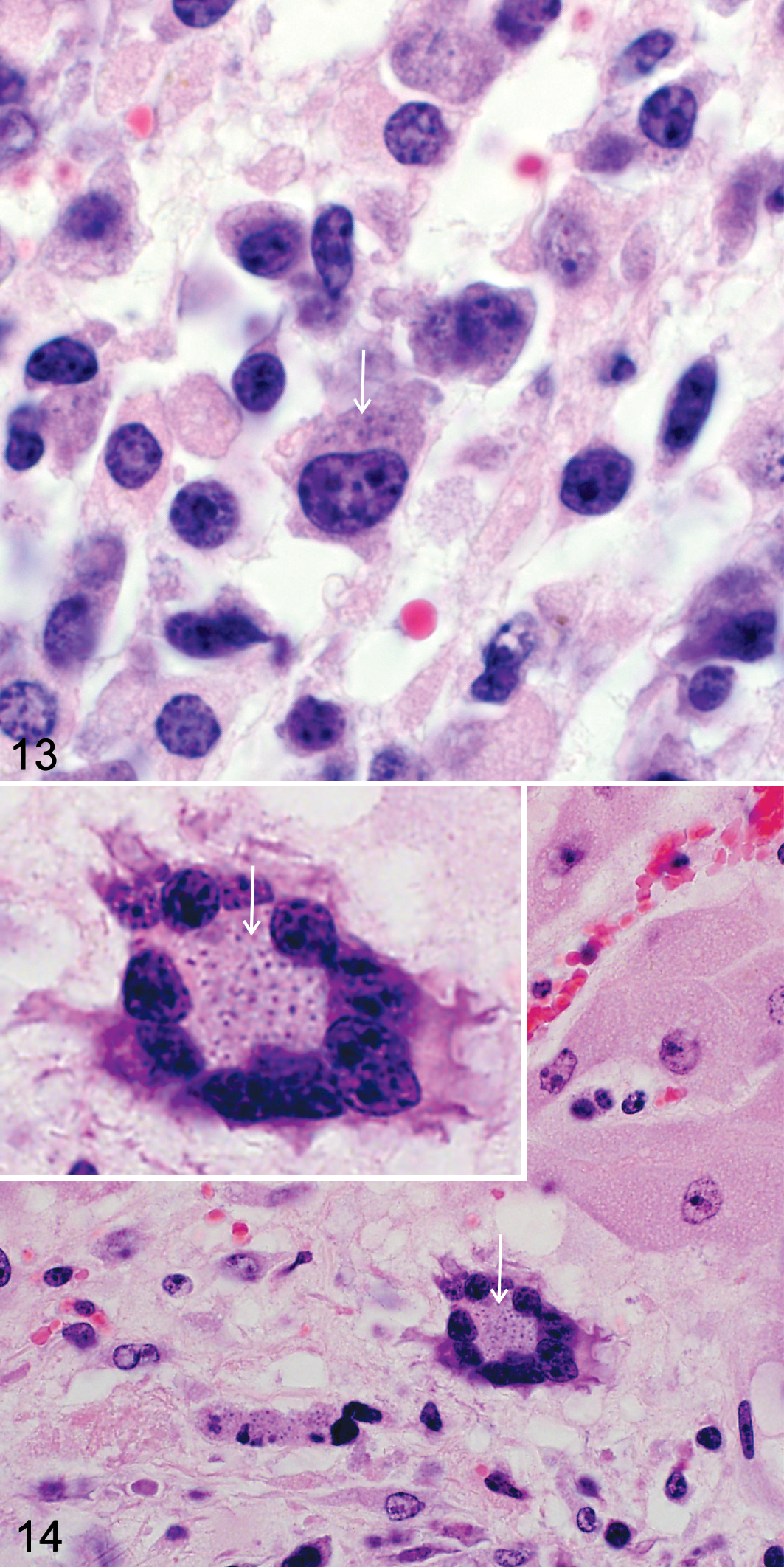
As has been observed in cattle with ECF, 45 target cells infiltrate diverse lymphoid and nonlymphoid organs in wild artiodactyls with pathogenic theileriosis. Affected organs include the following (from most to least commonly infiltrated): lung (Fig. 15), liver (Fig. 16), spleen (Fig. 17), kidney (Fig. 18), heart (Fig. 19), peripheral and visceral lymph nodes (Fig. 20), gastrointestinal tract (abomasum and small intestine; Fig. 21), adrenal glands (Fig. 22), brain (Fig. 23), and skeletal muscles (Table 5). Target cells and/or extracellular theilerial schizonts/merozoites have been reported within and around small- and medium-caliber arteries, arterioles, veins, capillaries, sinusoids, and/or lymphatics. 10,21,75,104,108,123,125,174 Necrotizing vasculitis was an inconsistent finding. 123,125 Vascular distension and occlusion by target leukocytes and/or fibrin, often associated with foci of parenchymal necrosis, has been described (Figs. 16, 18, 22, and 23). 54,123,125 Necrotic foci were most frequently reported in the liver, followed by the heart (Fig. 19), spleen, skeletal muscle, gastrointestinal tract, and brain. 10,21,54,75,108,123,125 Further investigation is needed to determine whether these are all due to vascular occlusion. Foci of hemorrhage and/or congestion was commonly described in the lungs (Fig. 15) and, to a lesser extent, the liver (Fig. 16), kidney, and brain. 54,75,108,123,125,174
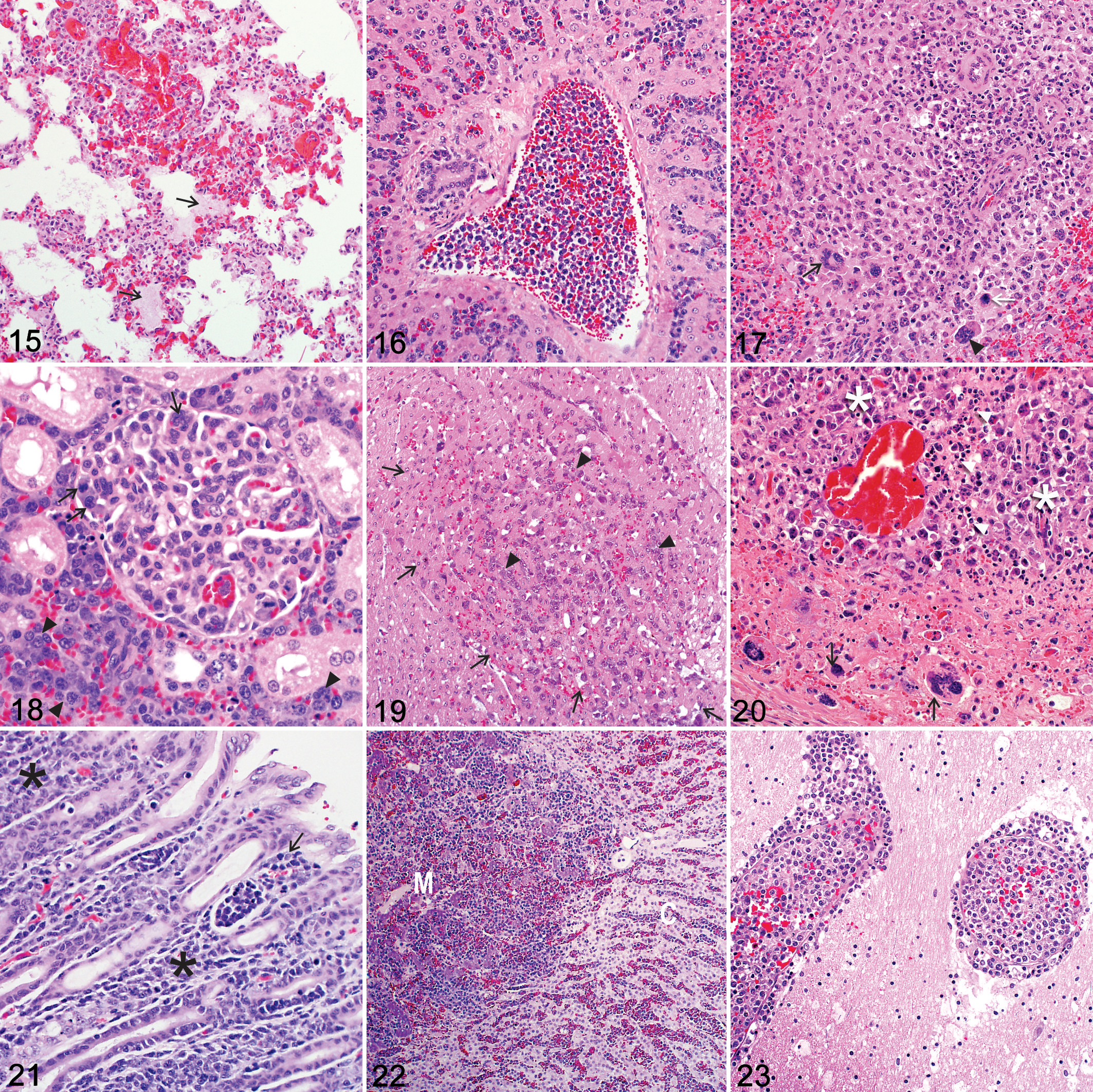
As might be expected, parasitic intracytoplasmic schizonts/merozoites were most frequently identified in those organs most commonly infiltrated by target cells (Table 5). Schizonts have been identified as clusters of distinct pleomorphic, darkly basophilic chromatin granules/nuclei embedded within a neutral-staining parasitic cytoplasm that may displace target cell nuclei to one side (Figs. 13, 14). 54,75,94,108 Their size, number, internal structure, occurrence, and distribution in different tissues and organs vary considerably within and among wild artiodactyl species. ‡‡‡‡
Similar to ECF, 45 widespread pulmonary intra- and perivascular mononuclear target cell infiltrates have been described with diffuse congestion, proteinaceous edema (Fig. 15), multifocal disseminated hemorrhages, and multifocal alveolar septal necrosis associated with thrombi. 10,54,75,108,123,125,174 Parasitic schizonts were observed in alveolar macrophages 75 and multinucleated target cells. 21,75,123,125 Reference was made to interstitial pneumonia in a tsessebe 75 and mild pneumonitis in a giraffe. 108 Pulmonary histopathology in the tsessebe was characterized by diffuse congestion and widespread lymphocyte and macrophage infiltration, although there was no mention of leukocyte atypia. Pneumonitis in the giraffe referred specifically to multifocal disseminated foci of interstitial and intra-alveolar fibrin exudation and hemorrhage. Infiltrating parasitized and nonparasitized leukocytes were observed only in a focal area of giraffe lung.
In the liver, target cells were observed in sinusoids and portal and hepatic veins as well as in the perivascular interstitium of the portal tracts (Fig. 16) frequently associated with intra- and extracellular theilerial schizonts. 54,75,108,123,125,174 Parasitized Kupffer cells occasionally surrounded foci of hepatocyte necrosis and hemorrhage. 75,108 Parasitized multinucleated giant cells of indeterminate origin 21,75,108,123,125 were rarely and inconsistently associated with necrotizing vasculitis. 123,125
Target cells have been described predominantly within the splenic red pulp, but the white pulp was also occasionally infiltrated (Fig. 17). 10,54,75,108,123,125,174 Foci of hemorrhage and necrosis variably rimmed by schizont-infected macrophages were observed. 54,108 Multinucleated giant cells were rarely and inconsistently observed in the red pulp and within necrotic trabecular and white pulp arterioles. 123,125 Descriptions of lymph node histopathology are relatively scarce in wild artiodactyls with pathogenic theileriosis. 10,54,174 Variable numbers of pleomorphic, mono-, and/or multinucleated target cells were associated with thrombi and variable degrees of necrosis (Fig. 20). 10,54,123,174 Edema and congestion were rare. 123
In the kidney, intravascular target cells were observed, especially in larger arteries, 54,123,125 with foci of necrosis and hemorrhage in some cases. 75,123,125 Glomerular 54 and perivascular (interstitial) target cell infiltrates have also been seen (Fig. 18). 10,75,123,125,174
Target cells were reported within and/or surrounding the myocardial 10,54,75,123,125,193,194 and skeletal muscle 75 vasculature. Foci of myofiber degeneration and necrosis with or without associated hemorrhages have been described in cardiac (Fig. 19) and/or skeletal muscle. 10,75,123,125 Plugging of the myocardial vasculature with target cells and associated vasculitis was a rare and inconsistent finding. 123,125
The gastrointestinal histopathology in wild artiodactyls with pathogenic theileriosis has been well described in gray/common duiker, eland, and giraffe. 54,108,123,125 Intra- and perivascular target cell infiltrates have been reported predominantly in the superficial abomasal mucosa (Fig. 21), where they were variably associated with erosions and ulcers, hemorrhage, and edema. 54,108 Transmural infiltration of target cells has been rarely described in the abomasum. 54 Foci of necrosis and hemorrhage were also observed in the small intestinal mucosa. 54,108 Arteritis and thrombosis was rarely and inconsistently associated with intravascular multinucleated target cells. 123,125
Adrenal glands were not often sampled in wild artiodactyls that died of pathogenic theileriosis. 54,173,193,194 Reported lesions included intra- and perivascular target cells in the cortex and medulla (Fig. 22), 54,173,193,194 diffuse congestion, 123 associated foci of necrosis, 54 and multifocal hemorrhages. 123 Adrenal lesions were reportedly severe enough to cause adrenal dysfunction in 2 eland. 54
Clinical neurologic abnormalities were not often reported, and the brain was uncommonly sampled, so central nervous system lesions have been rarely documented in wild artiodactyls with pathogenic theileriosis. 54,123,125 Mono- and/or multinucleated target cells were described in dilated arteries and arterioles, particularly in the choroid plexus, meninges, and midbrain (Fig. 23). 54,123,125 Congestion, 54,123,125 fibrin thrombi, 54 as well as foci of malacia/infarction 123,125 and hemorrhage 54,123,125 have been reported. These lesions are similar to those described in the brain and spinal cord of cattle with cerebral theileriosis/turning sickness caused by T. taurotragi and, rarely, also by T. annulata and T. parva. 14,33,187 However, due to the small sample size in wild artiodactyls, meaningful conclusions can be drawn only if the central nervous system histopathology is investigated in a greater number of wild ungulates, especially eland with fatal pathogenic theileriosis due to T. taurotragi infection.
Additional reported findings that may or may not be specific to theileriosis include variable sublethal vacuolar injury of hepatocytes, 75,108,123,125,174 splenic and lymph node lymphoid depletion with no discernible lymphoid follicles, 54,75 and stunting and fusion of intestinal villi. 54
Detailed descriptions of the histopathology associated with pathogenic theileriosis are required in larger numbers of different wild artiodactyl species. The most detailed histopathologic descriptions are dated, and, in most cases, there was no molecular confirmation of theilerial infection. In addition, the target cell phenotype has not yet been established in organ sections from diverse species. As a result, the pathogenesis of transformative theileriosis is largely unknown in African wild artiodactyls.
An important histologic differential diagnosis for pathogenic theileriosis in wild artiodactyls is alcelaphine herpesvirus 1– or ovine herpesvirus 2–associated malignant catarrhal fever. 94 This disease has been reported in free-ranging and captive African buffaloes in South Africa, as well as in a variety of other captive and semicaptive wild artiodactyls. 144 Wild artiodactyls that die due to malignant catarrhal fever exhibit widespread lymphoid hyperplasia, multisystemic necrotizing vasculitis, and variable lymphoplasmacytic and histiocytic perivasculitis, often associated with parenchymal necrosis or mucosal ulceration. 144 Commonly affected organs include the kidneys, lymph nodes, spleen, hepatic portal tracts, brain, heart, gastrointestinal tract, trachea, and lungs. 144 Histopathology of the lesions and PCR (including RLB assays) on fresh spleen or lymph node samples is required to differentiate these diseases.
Leukocyte Phenotype and Pathogenesis
The transforming theilerias in domestic ruminants induce changes in a range of pivotal immune cells, resulting in an impaired immune-inflammatory response to the parasites, 31,38,45 which is fundamental to the proliferation, dissemination, and survival of target leukocytes in the host. 45,90,94 Target leukocytes differ in their phenotypic expression among theilerial species in domestic ungulates. 5,37,49,90,94,96,168 Early research used the differences in phenotypic expression between T. parva– and T. annulata–transformed leukocytes in cell culture to explain differences in pathogenesis and immune response. 49,96
In vitro and ex vivo studies show that T. parva infects and transforms T and B lymphocytes but preferentially transforms T lymphocytes expressing CD4 or CD8 and either αβ or γδ T-cell receptors. 5,37 In contrast, T. annulata and closely related T. lestoquardi preferentially transform monocyte-macrophages, B lymphocytes, dendritic cells, and, to a lesser extent, T lymphocytes in cell culture. 31,37,89,90,96,168,172
Recent studies that have investigated the leukocyte tropism of pathogenic Theileria species in vivo in domestic cattle suggest that the in vitro findings may be slightly misleading. For instance, in bovine tropical theileriosis 19 and ECF, 45 the proliferative foci in tissues consist largely of T lymphocytes and macrophages. In bovine cerebral theileriosis/turning sickness in Tanzanian cattle, histologic lesions in the brain were found to resemble lymphomatoid granulomatosis in humans, a nodular to diffuse angiocentric and angiodestructive granulomatous disease in the lungs, characterized by lymphocytes, macrophages, and atypical multinucleated giant cells. 14
By using specific cell markers such as CD163, it was possible to show in ECF-infected cattle that macrophages likely play a significant role in the downregulation of a functional T-cell response and that key clinical signs and lesions may in fact be the result of a macrophage activation syndrome. 45 This syndrome has been implicated in the pathogenesis of diverse neoplastic, autoimmune, and infectious diseases in humans and animals, including bovine trypanosomiasis. 45
Thus, recent in vivo studies indicate that subpopulations of T lymphocytes and macrophages/dendritic cells likely play a pivotal role in the pathogenesis of schizont-“transforming” theilerial diseases in domestic ruminants. The relationship among these leukocyte populations requires further study. Therefore, characterization of target leukocyte phenotypes in tissues from African wild artiodactyls with pathogenic theileriosis would contribute to our understanding of pathogenesis.
Aspects of Control
Currently, the “infection and treatment” method of immunization is the only available method of vaccination against ECF in cattle in Africa. 141 Cattle are vaccinated with a mixture of tick-derived T. parva stabilates (referred to as the Muguga cocktail), and they are treated concurrently with long-acting oxytetracycline, which prevents clinical disease but allows immunity to develop. 29,117,141 However, since oxytetracycline treatment is not protective against higher sporozoite doses in cattle, each new batch of sporozoite vaccine needs careful titration to establish the effective dose. 115 In addition, cattle that fail to respond effectively to oxytetracycline treatment may succumb to clinical disease unless treated with theilericidal drugs, such as buparvaquone, a naphthoquinone compound that is the drug of choice in domestic livestock with pathogenic theileriosis. 89,90,94
Recently, a crude “infection and treatment” method with a tick-derived Theileria sp. (sable) stabilate was developed and used successfully to artificially expose captive-bred roan calves to infection. 175 However, the development of clinical disease in the calves meant that therapeutic intervention was required, which is a significant constraint to the practical application of this method under field conditions, especially in wild artiodactyls. 12,174 Postinfection treatment requires immobilization of sick calves, which is costly, logistically onerous, and could negatively affect their recovery. In addition, buparvaquone, the most effective treatment for pathogenic theileriosis in roan calves, 175 is not commercially available in South Africa because it induces a carrier state in Corridor disease, which is a controlled disease. 93
Alternative, less invasive methods of controlled exposure in susceptible wild artiodactyls that do not require therapeutic intervention are needed. 12 One such method, which requires further investigation, is the inoculation of antelope calves with schizont-infected in vitro–cultured leukoblasts. Research shows that attenuation of T. annulata and T. parva occurs after multiple schizont-infected leukocyte replication cycles in cell culture. 115 In addition, inoculation of live attenuated T. annulata schizont-infected cell cultures confers protective immunity in cattle without causing clinical disease and therefore bypasses the requirement for treatment. 148 However, for reasons that are unclear, transfer of T. parva to cattle via inoculation of in vitro live attenuated cell cultures is not as effective. 18 A Theileria sp. (sable) schizont-infected leukocyte cell line exists with successful propagation of the parasite in vitro. 204 Preliminary studies on very few animals show that theilerial infection can be safely transmitted to roan calves via inoculation of an infected cell line and that calves appear to develop a degree of protective immunity. 12 However, this technique requires significant refinement and testing on a greater number of animals. Limiting factors include the scarcity and value of roan and sable antelope in particular and the possibility of incurring severe, potentially fatal disease.
Conclusion
It is likely that a variety of Theileria species infect the diverse wild ungulate species on the African continent. While several Theileria species have been identified in African wild artiodactyl species by phylogenetic analysis of the parasite 18S rRNA genes, many more probably remain to be discovered. Known Theileria species are commonly classified as pathogenic (schizont “transforming”) or benign to mildly pathogenic (“nontransforming”). Normally a dynamic balance exists among wild mammalian hosts, tick vectors, Theileria parasites, and their environment, and as a result, the majority of animals in a population develop only subclinical infection. However, any perturbation of this balance can increase the susceptibility of wild ungulates to clinical disease and death. Similar to ECF in cattle, pathogenic theileriosis in wild artiodactyls is characterized by parasitic schizont-induced pseudo-neoplastic transformation, hyperproliferation, and dissemination of host leukocytes to multiple organs and the bloodstream. Histologically, leukoproliferative foci occur intra- and perivascularly in multiple organs and are associated with fibrin thrombi, necrosis, hemorrhage, and edema. More research is required to clarify the epidemiology and pathogenesis of theileriosis in African wild artiodactyls to prevent fatalities in susceptible species, particularly roan and sable antelope.
Footnotes
**
Acknowledgements
We thank Professor Christo Botha, head of the Department of Paraclinical Sciences, Faculty of Veterinary Science, University of Pretoria, for funding this study. We also thank Sue Clark at Equilibrium Studio and Estelle Mayhew in the Department for Education Innovation, Faculty of Veterinary Science, University of Pretoria, for their help with the figures and Dr June Williams in the Section of Pathology, Department of Paraclinical Sciences, Faculty of Veterinary Science, University of Pretoria, for her contribution of Figure 9 and cases for ![]() . We acknowledge other colleagues and students at the Faculty of Veterinary Science, University of Pretoria, including Ilse Vorster, Milana Troskie, Antoinette Lourens, Naomi Timmerman, Muhammad Seedat, and Caitlin Holyoake for their assistance with data collection for this project. In addition, we wish to thank Professor Jaco Greeff for editorial assistance.
. We acknowledge other colleagues and students at the Faculty of Veterinary Science, University of Pretoria, including Ilse Vorster, Milana Troskie, Antoinette Lourens, Naomi Timmerman, Muhammad Seedat, and Caitlin Holyoake for their assistance with data collection for this project. In addition, we wish to thank Professor Jaco Greeff for editorial assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received internal university funding for this review.
