Abstract
Immunohistochemical (IHC) and fluorescent antibody (FA) techniques were optimized for the detection of Foot-and-mouth disease virus (FMDV) structural and nonstructural proteins in frozen and paraformaldehyde-fixed, paraffin-embedded (PFPE) tissues of bovine and porcine origin. Immunohisto-chemical localization of FMDV was compared with 7 detection systems, 8 primary antibodies, and 11 epitope retrieval techniques. All serotypes tested (O, A, Asia, C [cryosection]; O, A, Asia [PFPE]) were localized in association with mature vesicles. Multi-label FA was used in conjunction with IHC and conventional histopathology to characterize vesicle maturation in 4 steers and 2 pigs experimentally infected with FMDV. At the edge of advancing vesicles, a consistent finding was acantholytic degeneration of basal keratinocytes surrounding dermal papillae with suprabasilar clefts and microvesiculation. Progression of microvesiculation led to coalescence with the expanding vesicle. Cells at the leading edge of vesicles were positive for FMDV antigens by IHC and FA. Cell marker profile of these cells by FA was consistent with keratinocytes (i.e., cytokeratin [CK]-positive, S100-negative, MHC-II-negative). In rare instances, CK-negative, MHC-II-positive, and FMDV-positive cells (presumptive dendritic cells or macrophages) were identified within dermis subjacent to vesicles.
Introduction
Foot-and-mouth disease virus (FMDV; family Picornaviridae, genus Aphthovirus) is the etiological agent of foot-and-mouth disease (FMD), the most economically significant veterinary disease worldwide. FMDV has been microscopically localized in tissues by widefield fluorescent antibody technique (FAT) 31 and FA-confocal microscopy (CFM), 14,17–19 and FMDV RNA has been localized by in situ hybridization (ISH). 3,4,7,14,22,32 However, the only description in the veterinary literature of localization of FMDV antigens by immunohistochemistry (IHC) in domestic species is a recent, limited description from the authors' laboratory. 21 The only other published anti-FMDV IHC technique provides a single image of the technique applied to cervid tissue. 30 In that study, the 2 antistructural protein monoclonal antibodies (mAbs) described are demonstrated to recognize all 7 serotypes of FMDV in an enzyme-linked immunosorbent assay (ELISA); however, positive IHC detection of FMDV antigens is only demonstrated for 1 mAb against 1 FMDV serotype. Anti-FMDV mAbs have been generated against viral structural 2,27,30 and nonstructural proteins 18,29 and have been used in a variety of diagnostic applications.
Immunohistochemistry has been used to identify a variety of viruses in animal tissues including West Nile virus, 5 Bovine viral diarrhea virus, 12 Vesicular stomatitis virus, 6 and African swine fever virus. 11 Advantages of IHC over ISH and CFM, respectively, are the relative simplicity of protocols and the ability to screen far greater quantities of tissues. Historically, most IHC techniques have taken advantage of the affinity of (strept)avidin for biotin (avidin-biotin complex [ABC]) by using either 1) biotinylated primary antibody followed by avidin-reporter enzyme conjugate or 2) nonbiotinylated primary antibody, biotinylated secondary antibody, then avidin-reporter enzyme conjugate. 23 More recently, micropolymer-based assays 24,25 have been developed that claim better sensitivity than conventional ABC systems while avoiding the specificity failures associated with background biotin. 23 However, comparisons across polymer system kits and between polymer and ABC systems are not readily available.
Tissue fixation and epitope (antigen) retrieval are critical components of a successful IHC technique and have recently been thoroughly reviewed. 23 Formaldehyde is the most commonly used tissue fixative in veterinary pathology and is routinely used as 10% neutral buffered formalin (NBF). Paraformaldehyde (PFA) fixation has been used predominantly in research applications when less protein and nucleic acid cross-linking is desired; paraformaldehyde-periodate-lysine (PLP) 15 fixation was originally described for electron microscopy but has been reported to be a superior fixative for IHC techniques owing to its ability to use lower concentration of PFA and thus induce less cross-linking. 13 Detection of FMDV RNA by ISH has been reported in PFA-fixed 22,28 and NBF-fixed 32 tissues. Antigen retrieval techniques from aldehyde-fixed tissues vary widely according to target antigen and operator preference, but generally consist of heat-induced epitope retrieval (HIER), enzymatic antigen retrieval, or a combination of both. 23
The hallmark gross pathological lesions of FMD are the vesicles most commonly occurring in the tongue, interdigital skin, and coronary band, and less commonly associated with the ruminal pillars, snout, and other sites. The histological characteristics of FMD vesicles have been thoroughly described in the veterinary literature. 8,9,16,20,26,31 However, the histogenesis of cells involved in the morphogenesis of vesiculation has not been thoroughly examined. Moreover, elucidation of the mechanism of transfer of FMDV from systemic circulation to lesional epithelium remains elusive.
The current study provides the first thorough description of IHC detection of FMDV antigens in frozen and fixed animal tissues including serotype-specific efficacies of 8 mAbs, comparisons of 4 polymer-based detection systems, and 11 epitope retrieval techniques. Using these optimized techniques, FMD vesicles were characterized by IHC localization of viral antigens and multichannel FAT with the overall conclusion that as vesicles progress laterally within the epithelium, microvesiculation by FMDV-positive dermal papillary basal cells leads to coalescence with, and expansion of, the maturing vesicle.
Materials and methods
Animal inoculations and tissue collection/processing
All work described herein was performed within the biosafety level 3 containment facility at Plum Island Animal Disease Center (Greenport, NY) with adherence to animal use protocols. All animals were included in other experiments in the Plum Island laboratory and will be described in greater detail in subsequent manuscripts. Briefly, cattle were 200–400 kg Holstein steers. Pigs were Yorkshire barrows aged 4–6 months. Two steers were inoculated with FMDV via intra-epithelial lingual injection (IEL); steer B6100 was infected with FMDV strain A24-Cruzero (1.0 × 104 TCID50 [50% tissue culture infective dose] BHK [baby hamster kidney]-21), and steer B732 was infected with FMDV strain O1-Manisa (3.0 × 105 BTID50 [50% bovine tongue infectious dose]). Twenty-four hours after IEL inoculations, each steer was euthanized by intravenous barbiturate overdose. Typical, multifocal, primary injection site vesicles (0.5–2.0 cm in diameter) were apparent at the time of euthanasia. Steers B762 and B7200 were aerosol inoculated via a previously described technique 21 with 1.0 × 107 BTID50 of FMDV O1-Manisa and euthanized 48 hr postinoculation (HPI). At euthanasia, each aerosol-inoculated steer had a single vesicle of the interdigital cleft skin. A grossly detected focal region of pallor of the lingual torus of B7200 was identified microscopically as an early vesicle. Both aerosol-inoculated steers were febrile and viremic at the time of euthanasia.
Pigs were inoculated with FMDV via intraepithelial injection of the heel bulb. Pig 18566 received 5.3 × 105 TCID50 of FMDV Asia1-A1 and was euthanized 5 days postinoculation (DPI). Pig 871 received 3 × 104 TCID50 of FMDV C3-Resende and was found dead at 6 DPI. Necropsy and routine histopathology indicated severe myocardial necrosis as the cause of death. Both pigs had extensive, mature, intact and ruptured vesicles of the tongue and all 4 coronary bands and interdigital clefts. Pig 18566 additionally had a 2.0-cm × 1.0-cm intact vesicle of the dorsal aspect of the snout.
Immediately subsequent to euthanasia, vesicles were dissected from each animal and either 1) immediately fixed in 10% NBF or 2% PLP 15 or 2) placed in a cryomold, embedded in optimal cutting temperature (OCT) compound, a frozen in a metal pan over a bath of liquid nitrogen, and stored at −70°C until used for cryomicrotomy. Fixed tissues were processed by routine methods. Fixed and frozen specimens were sectioned at 4–5 μm onto electrostatically charged glass slides. b Cryosections were fixed for 10 min in acetone at −20°C, air-dried at room temperature, and stored at −20°C until use.
Antigen retrieval
Postfixation retrieval of target antigens was performed by HIER and enzymatic techniques (Table 1). Humid heating was performed in a commercially available rice cooker; humid pressurized heating was performed in a typical sterilization autoclave. For both techniques, slides were deparaffinized and hydrated by a series of changes of xylene, ethanol, and distilled water. Slides were then placed in loosely closed Coplin jars containing target retrieval solutions, which were subsequently placed in a water bath during heating. Antigen retrieval solutions evaluated were citrate c (pH 6.0, 6.2, 9.0), Diva Decloaker, d and Reveal d (Table 1). Each heating-system retrieval solution combination was evaluated at durations of 5, 15, and 25 min.
Enzymatic retrieval was performed with pronase, trypsin, and pepsin epitope retrieval kits d and with proteinase K e (Table 1). Kits were used per manufacturers' recommendations with similar variation in digestion time as for HIER. Proteinase K was applied at 20 μg/ml in Tris pH 8.0 and 0.05% Tween-20 at 37°C. For comparison of antigen retrieval techniques, a semiquantitative retrieval quality score (-, +, ++, +++, ++++) was assigned to each technique based upon sensitivity and intensity of staining. Scores were defined as (-) no staining detectable, (+) rare individual positive cells with weak intensity, (++) moderate quantity of individual (or clusters of) positive cells with weak-moderate intensity, (+++) regionally extensive detection with moderate-strong intensity, and (++++) diffuse immunoreactivity within the expected region (vesicle) with strong intensity.
Comparison of epitope retrieval techniques for unmasking of Foot-and-mouth disease virus (FMDV) immunoreactivity in paraformaldehyde fixed bovine tissues.*
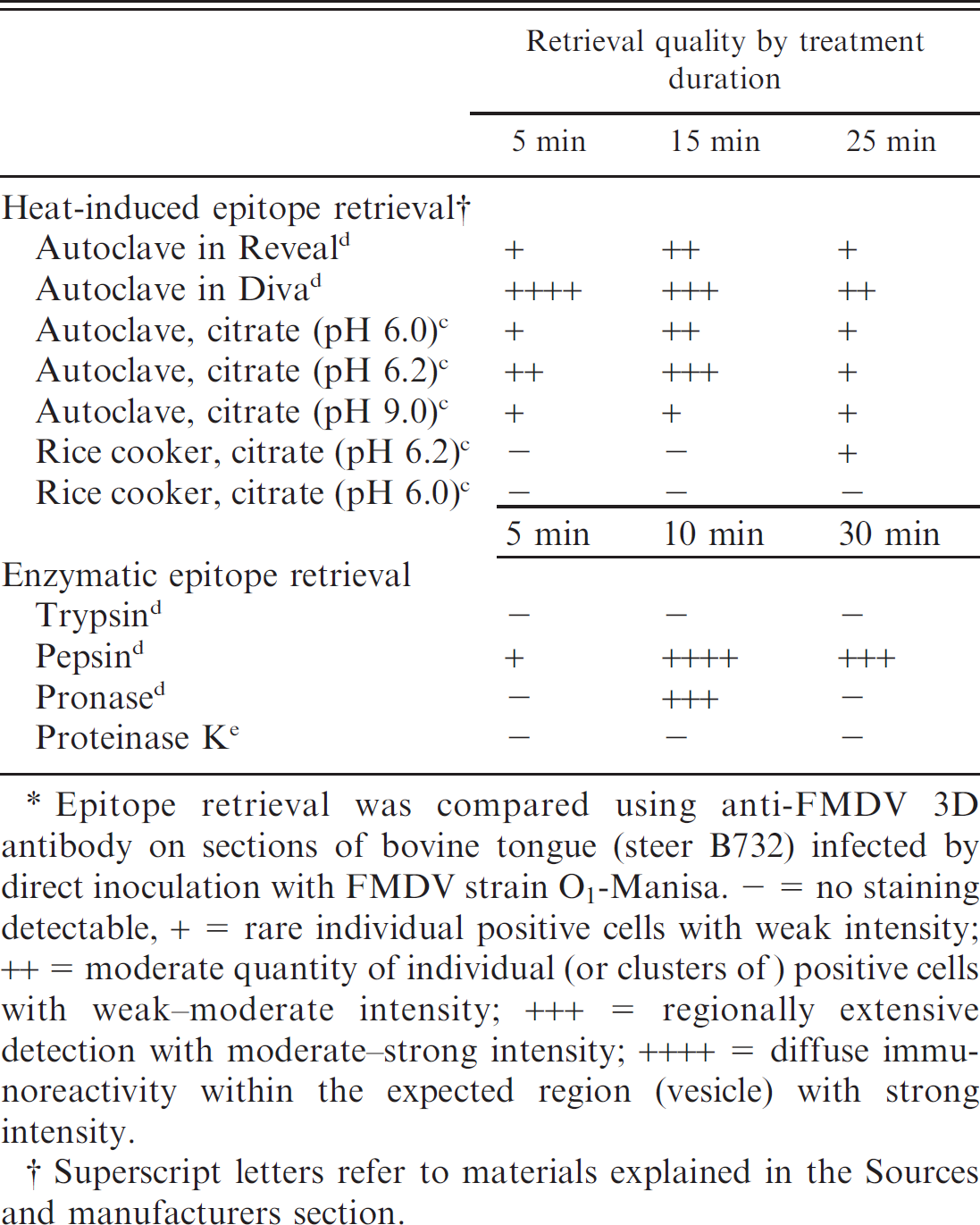
Epitope retrieval was compared using anti-FMDV 3D antibody on sections of bovine tongue (steer B732) infected by direct inoculation with FMDV strain O1-Manisa. - = no staining detectable, + = rare individual positive cells with weak intensity; ++ = moderate quantity of individual (or clusters of) positive cells with weak-moderate intensity; +++ = regionally extensive detection with moderate-strong intensity; ++++ = diffuse immunoreactivity within the expected region (vesicle) with strong intensity.
Superscript letters refer to materials explained in the Sources and manufacturers section.
Anti-FMDV antibodies
Antibodies to FMDV used in the current study and their specificities are described in Table 2. Antibodies directed to structural (capsid) proteins were elicited with purified viral particles (140S) or subviral capsid components (12S) and recognize antigens present in the viral particle. 2,27,30 Conventional anticapsid antibodies were predominantly serotype specific and reactive only against the eliciting serotype used. 2,27 Two antibodies were previously generated and characterized with the goal of serotype-independent FMDV detection (serotype-independent, structural antibodies). 30 Antibodies directed to the viral nonstructural protein FMDV 3D (RNA-dependent RNA polymerase) were elicited against purified 3D protein as previously described. 29 Presence of intracellular FMDV 3D reactivity was interpreted as suggestive of active viral replication in infected cells.
Immunohistochemistry
Each antibody was evaluated in cryosections from vesicles from animals inoculated with FMDV strains O1-Manisa, A24, Asia1-A1, and C3-Resende (1 virus strain per animal). Anti-FMDV 3D and serotype-independent, anti-structural antibodies were additionally evaluated in PLP-fixed, paraffin-embedded tissues from animals inoculated with FMDV strains O1-Manisa, A24, and Asia1-A1. Numerous serotype-specific, antistructural antibodies were screened in PLP-fixed, paraffin-embedded tissues, but none were found to have adequate signal-to-noise ratio for use (data not shown). In order to assess the effect of fixative on retrieval of antigen, duplicate specimens from select tissues from the same lesions were fixed in either NBF or PLP and compared through similar epitope retrieval protocols (data not shown).
Frozen and deparaffinized fixed tissue sections were blocked for 2 hr at room temperature with phosphate buffered saline, 0.05% Tween-20, 6% mixed serum, and 2% powdered non-fat milk. Anti-FMDV mouse monoclonal primary antibodies 2,27,29,30 generated as crude supernatants of hybridoma cell lines (Table 2) were diluted in blocking buffer and applied to tissue sections for 18 hr at 4°C.
Anti-mouse immunoglobulin detection kits consisted of a conventional ABC alkaline phosphatase kit, f 2 micropolymer alkaline phosphatase (ALP) kits, g h an ABC peroxidase kit, i and 2 micropolymer peroxidase kits. g j All detection kits were utilized per manufacturers' protocols. Various detection substrates were compared (data not shown); red chromogens provided excellent sensitivity and good contrast to hematoxylin counterstain. A single substrate k consistently provided superior sensitivity and specificity for ALP systems, whereas for peroxidase-based detection, 2 substrates l m had similar properties. Chromogen substrates were applied per manufacturers' suggested protocols with the exception that levamisole i was added to substrate solutions at 5 μl/ml for all slides receiving ALP detection systems. Slides were observed during substrate development, and substrate reactions were terminated when signal was considered to be of adequate strength. Slides were counterstained with Gill's hematoxylin and coverslipped by routine techniques.
For each section examined, a duplicate, negative control slide contained a serial section, which received identical treatment, with the exception that the primary antibody consisted of a mouse, monoclonal anti-Vesicular stomatitis Indiana virus antibody applied at similar concentration. An additional negative control was included with each IHC run, consisting of slides containing similar tissues from a mock aerosol-inoculated steer that was treated identically to the other aerosol-inoculated animals but received a virus-free inoculum; these slides were treated identically to experimental specimens.
Determination of a positive IHC result required the presence of a strong cell-associated signal within the tissue in question with no similar signal present on either of the negative controls from the same run. For antibody and detection system comparisons, sensitivity was graded (-, +, ++, +++, ++++) based upon distribution of antigen detection within the expected region; intensity was similarly graded according to the relative strength of immunopositivity within antigen-positive cells.
Comparison of monoclonal anti-Foot-and-mouth disease virus (FMDV) antibodies for detection of immunoreactivity in frozen and paraformaldehyde-fixed, paraffin-embedded sections of vesicles.*

Tabulated sensitivity, intensity, and background were based on application to cryosections from FMDV strain O1-Manisa-infected tissues, except for antibodies 6HC4 and 7SF3, which were quantitated against FMDV strain A24. - = no staining detectable, + = rare individual positive cells with weak intensity; ++ = moderate quantity of individual (or clusters of) positive cells with weak-moderate intensity; +++ = regionally extensive detection with moderate-strong intensity; ++++ = diffuse immunoreactivity within the expected region (vesicle) with strong intensity.
140S is the intact virion. 12S refers to the acid degradation fraction of FMDV containing structural proteins VP1, 2, 3. VP1-13kd is a VP1 fragment spanning amino acids 55–179.
Positive detection required sensitivity and intensity scores of +++ or ++++ and minimal background for each strain reported positive. Bold text indicates similar finding in formalin-fixed tissue.
Fluorescent antibody techniques
Multi-label FAT was performed on cryosections similarly to IHC with few exceptions. All primary antibodies were collectively diluted in blocking buffer and simultaneously incubated on tissues at 4°C for 18 hr. Mouse mAbs used to label cell markers were anti-pancytokeratin plus (pCK), d anti-cytokeratin-14 (CK-14), d anti-MHC-II, n and anti-S100. e Goat, anti-mouse, isotype-specific, fluorescently labeled secondary antibodies o were diluted collectively in blocking buffer and applied to slides for 30 min at 37°C. For most stains, virus was detected with Alexa Fluor (AF)-594, and cell markers were labeled with AF-488. In some multi-label experiments, conformation to this strategy could not be maintained due to availability of secondary antibodies; in such instances, signal colors were digitally reassigned such that for all images included, virus appeared as a red signal. For double stains, nuclei (DNA) were counterstained with 4′, 6-diamidino-2-phenylindole. p Slides were evaluated with a wide-field epifluorescence microscope, and images were captured with a digital camera. q Individual images were captured in each detection channel and subsequently adjusted for brightness and contrast, and then merged using commercial software. r Isotype controls were included with each experiment and were used to establish camera sensitivity settings. Tissues from mock-infected animals incubated with anti-FMDV antibodies were also included as negative controls.
Results
Cryosection immunohistochemistry
In cryosection staining, the anti-3D antibodies provided excellent sensitivity with minimal background for all tissues and serotypes examined with the exception of poor reactivity of antibody F19-6 against FMDV C3-Resende (Table 2; Fig. 1). Negative control serial sections receiving mouse monoclonal anti-Vesicular stomatitis virus antibodies had no corresponding signal. Similar tissues from mockinfected animals also had no detectable signal. Antibody F19-51 had slightly superior sensitivity in most tissues but, inconsistently, also had greater background at similar dilutions relative to antibody F19-6. Background staining with antibody F19-51 consisted of regional chromogen deposition within vascular basement membranes and smooth muscle and perineurial sheaths. Antibody F19-6 could be used at lower titrations to achieve similar sensitivity without development of background and, overall, provided a superior signal-to-noise ratio. Anti-3D immunoreactivity within vesicles was predominantly limited to the cytoplasm of acantholytic keratinocytes within the stratum spinosum and intact cells of the stratum basale (presumptive basal cells). Rarely, immunoreactivity was identified within cytoplasm of perivascular mononuclear cells in the dermis.
Numerous conventional antistructural antibodies were screened, and for each FMDV serotype examined, at least 1 antibody was found to be efficacious (Table 2). Minimal cross-serotype immunoreactivity was occasionally identifiable but was substantially weaker than signal generated from application to homologous serotype viruses. Antibody 6HC4 was unusual in this category in that substantial signal was detected against multiple serotypes. Serotype-independent, antistructural antibodies provided excellent sensitivity and specificity against all serotypes examined with the exception that antibody F21140SO had weak reactivity against FMDV C3-Resende. Distribution of signal from structural and nonstructural antibodies was consistently similar.
Paraffin-embedded tissues
Choice of fixative was found to have minimal effect on overall efficacy of IHC detection of FMDV antigens, with PLP and NBF fixation each having superior sensitivity against some serotypes with some antibodies (data not shown). Tissue morphology and background were also similar with both fixatives. Paraformaldehyde-fixed tissues were utilized for subsequent technique optimization owing to theoretical superiority based upon decreased cross-linking of proteins and nucleic acids.
Epitope retrieval technique had a substantial effect on sensitivity of detection (Table 1). Overall, the best retrieval of FMDV 3D antigen was achieved with the Diva d solution with a 5- or 15-min autoclave treatment. Citrate solution (pH 6.2) with 15-min treatment was only slightly inferior. Heat retrieval at ambient pressure (rice cooker) was consistently ineffective, regardless of retrieval solution, with antigen barely detectable at 25 min. Of the 4 enzymatic epitope retrieval techniques evaluated, pepsin provided the best signal and was only slightly inferior to the optimized HIER. Pronase digestion provided moderate retrieval of FMDV antigens; trypsin and proteinase K were ineffective at all conditions evaluated. Tissue sections were lost from slides less frequently with all enzymatic epitope retrieval combinations relative to HIER. In addition, within these comparisons, enzymatic epitope retrieval induced less distortion of morphology. However, due to the slightly superior sensitivity achievable with the optimized HIER, this technique is favored for most applications.
Detection systems
Multiple detection systems were compared in frozen and fixed tissues (Table 3). In general, the micropolymer-based systems had greater sensitivity and similar or less background relative to ABC systems in both peroxidase and ALP platforms. In paraffinembedded tissues, the one-step peroxidase micropolymer detection kit j was preferred due to excellent sensitivity, specificity, and ease of use. Minimal endogenous peroxidase reactivity was observed in erythrocytes and polymorphonuclear leukocytes but was easily distinguished from the true (specific) signal. By contrast, endogenous peroxidase reactivity in similar cells within frozen sections was substantial enough to be prohibitive for many applications. Peroxidase blocking was attempted with hydrogen peroxide and phenylhydrazine at various reaction conditions (data not shown); however, substantial elimination of background without loss of specific signal was not achieved. Rather, a micropolymer ALP system was adopted for routine use in frozen sections because of favorable signal-to-noise ratio. Phosphatase blocking with levamisole was not necessary in the tissues shown herein but was crucial to decreasing background in other work examining mucosal tissues and lungs in the authors' laboratory.
Histologic and immunohistochemical characterization of vesicles
Lesions chosen for IHC characterization of FMDV antigen distribution were grossly and histologically classic, early-mature vesicles with varying extents of intra- and extracellular edema, keratinocyte acantholytic degeneration, and mixed leukocyte infiltration (Figs. 1, 2). Vesicular cavities contained variable quantities of fibrin and necrotic cellular debris. All vesicles had subjacent superficial perivascular dermatitis characterized by mild to moderate, predominantly mixed mononuclear infiltrates with fewer neutrophils. Selected vesicles were all grossly intact (not ruptured); microscopic rupture was observed only in the coronary band lesion of pig 18566 (Fig. 1J–1L). This lesion also had the unusual characteristic that the vesicle extended beyond the coronary band to the supracoronary haired skin. Multifocally, supracoronary hair follicles had segmental mural folliculitis and furunculosis. This consisted of neutrophilic infiltration and disruption of isthmic follicular epithelium with mural epithelial acantholysis, abscesses, and microvesiculation (Fig. 1J, inset).
Particular attention was focused on dermal papillae and surrounding basal keratinocytes at the periphery of vesicles, as these were considered to comprise regions of progression of vesiculation (Fig. 2). Regional groups of these papillae often had a continuum of pathologic changes that included acantholytic degeneration with cytoplasmic hypereosinophilia of suprapapillary basal cells, segmental-circumferential suprabasilar clefts, and neutrophil infiltration. More severely affected papillae were cavitated with accumulations of necrotic debris and coalesced to become continuous with the leading edge of the vesicle (Fig. 2B).
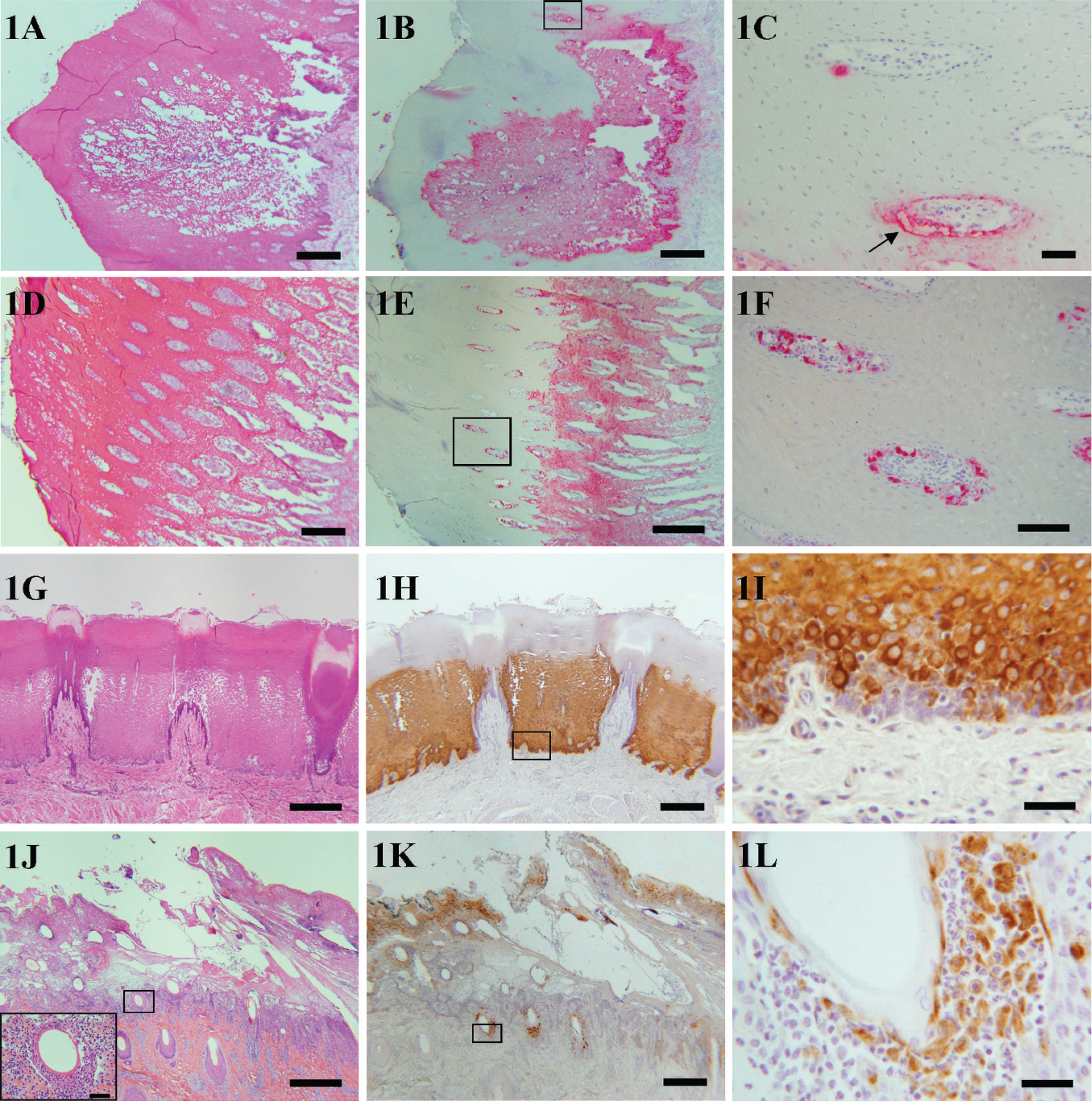
Light microscopy of foot-and-mouth disease (FMD) vesicles. A-F, cryosections; G-L, paraformaldehyde-periodatelysine (PLP)-fixed, paraffin-embedded. Column 1: Hematoxylin and eosin. Column 2: anti-Foot-and-mouth disease virus (FMDV) immunohistochemistry (IHC), low magnification. Column 3: anti-FMDV IHC, high magnification of region of interest identified in column 2.
Dermal papillae (DP) at the periphery of foot-and-mouth disease vesicles.
Immunohistochemical detection of FMDV antigens was typically limited to regions of morphologic vesiculation (Figs. 1, 2). For all tissues, distribution of structural and nonstructural FMDV antigens was essentially similar (direct comparison not shown). For tongue (steers B7200 and B6100) and snout (pig 18566), specific immunoreactivity spanned strata basale and spinosum with strongest intensity of staining in basal cells or suprabasal keratinocytes of stratum spinosum (Fig. 1). At the junction of lesional and normal epithelium, immunoreactivity was identified in individual or small clusters of basal cells lining dermal papillae. Rare antigen-positive cells were seen centrally within dermal papillae (Fig. 1C, 1F) and perivascularly within superficial dermis.
Within the coronary band and haired skin junctional lesion of pig 18566, antigen detection was largely limited to superficial stratum spinosum and follicular epithelium. Within the affected follicles of this pig, anti-FMDV 3D immunoreactivity was present in degenerate epithelial cells spanning internal and external root sheaths but not within neutrophils (Fig. 1L).
Fluorescent antibody characterization of vesicles
In order to examine the histogenesis of cells involved in vesicle propagation, FA labeling of FMDV was performed in conjunction with labeling of 1 or more cell markers. Epithelial cells were identified with anti-pCK or anti-CK-14. Within the tissues examined, similar staining with anti-S100 and anti-MHC-II on serial sections were considered specific for dendritic cells or macrophages.
As expected, within vesicles, FMDV predominantly colocalized with CK confirming that FMDV-positive, CK-positive cells were keratinocytes (Figs. 3–9). Tissues from the mock-infected steer and serial sections of vesicles incubated with isotype control antibodies had no corresponding FMDV-specific signal. At the periphery of vesicles, FMDV antigens were distributed in a similar pattern to that seen in IHC staining with few FMDV-positive cells within or surrounding dermal papillae (Figs. 3D, 4B, 4C, 5F). However, in contrast to the IHC staining, FA multilabeling allowed the determination that these FMDV-positive cells within papillary spaces were also CK-positive and were in varying stages of dissociation from overlying epithelium (Figs. 3F, 4B). This finding corresponds to the acantholytic degeneration and microvesiculation detectable by light microscopy. Though basement membrane was not specifically visualized, the regional collapse of epithelial cells into the dermal papillae was interpreted as regional ulceration (ulcer in situ; Figs. 3F, 4B). In addition, comparison of the periphery of lingual vesicles from aerosol-inoculated (Fig. 3A–3F) and IEL-inoculated (Fig. 4A–4C) steers indicated essentially similar morphologic and phenotypic characteristics. Double labeling with structural and nonstructural antibodies indicated that populations of cells containing either structural or nonstructural antigen were identifiable independently, or as double positives (Fig. 5G, 5H).
Cells labeled for S100 and MHC-II had consistent and similar distributions with clusters of positive cells within the deep dermis and dermal papillae (Figs. 5A–5F, 6A, 6B). FMDV antigen rarely localized in close proximity to S100 (Fig. 6B, inset), and, in few additional fields, CK-negative, FMDV-positive cells were identified within the dermis 50–200 μm distant from lesion margins (Figs. 7, 8). Examination of the interdigital vesicle of steer B762 by multi-label FAT indicated multi-segmental discontinuity of the basement membrane at the deep stratum basale (Fig. 9A). In these regions of ulceration, clusters of CK-negative, FMDV capsid-positive, MHC-II-positive cells were identified within the superficial dermis. These cells were morphologically and phenotypically consistent with dendritic cells (Fig. 9B). Similar colocalization of FMDV capsid antigens with MHC-II in (presumed) dendritic cells was observed subjacent to ulcerated vesicles at other sites and was distinct from the failure to identify such colocalization at regions of vesicle progression (dermal papillae).
Comparison of techniques for detection of anti-Foot-and-mouth disease virus (FMDV) immunoreactivity in cryosections of lingual vesicles inoculated by intraepithelial injection with FMDV strain O1-Manisa.*
ABC-ALP = avidin-biotin complex, alkaline phosphatase; ABC-PO = avidin-biotin complex, peroxidase. Grading scheme as described in Table 1.
Superscript letters refer to materials explained in the Sources and manufacturers section.
Discussion
Modern diagnostic laboratories and research institutions have numerous options for detection of FMDV in animal tissues. Though reverse transcription polymerase chain reaction (RT-PCR) and virus isolation (VI) are preferred for many applications owing to their relative ease of standardization and high throughput, these techniques are inadequate for providing information regarding microscopic viral localization to specific anatomic tissue regions or individual cell types. When such data are required or desired, investigators may utilize IHC, FAT, ISH, or CFM. Of these techniques, IHC is the fastest, most readily adaptable, and requires the least sophisticated laboratory equipment. Immunohistochemistry and ISH are similarly advantageous in that, once the slides are generated, they allow the most efficient screening of relatively large regions of tissues. Though light microscopy chromogenic detection of multiple antigens (or nucleic acids) is feasible, fluorescent techniques are more routinely applied for multi-label localization. Wide-field FAT allows nearly similar screening efficiency as IHC and ISH with the added advantage of allowing precise colocation of multiple antigens.
Though anti-FMDV IHC and FAT have been used as tools for pathogenesis investigation, they could also be effective ancillary diagnostic procedures, particularly when postmortem tissues might be the only available samples. Precise diagnosis of FMD is of utmost importance because of the severe economic impact associated with detection of the disease. Definitive diagnosis is rarely made with a single modality of testing. Rapidity of diagnosis is crucial as small delays in detection can lead to rapid geographic dissemination through movement of animals and aerosolization of infectious droplets. The cryosection IHC technique described herein is a sensitive, specific, and extremely rapid modality of detection of FMDV, and thus could be an extremely useful ancillary diagnostic test. The entire protocol may be completed within 2 hr of obtaining fresh tissue specimens. Application of the technique with anti-FMDV 3D or serotype-independent, anti-FMDV structural antibodies further enhances the utility of this technique as a simple, single method to detect numerous strains of FMDV.
The IHC efficacies of conventional antistructural antibodies described herein were generally limited to the serotype against which antibodies were elicited. This is consistent with virus neutralization data published as part of the original characterization of the antibodies. 2,27 The ability of antibody 6HC4 to recognize FMDV serotypes A, Asia 1, and C suggests that this antibody is specific for an epitope conserved across these serotypes. The serotype-independent, antistructural antibodies had excellent efficacy of FMDV detection by IHC across all serotypes tested; the conserved epitopes for which these antibodies are specific have been desdcribed. 30 The broad serotype efficacy of the anti-FMDV 3D antibodies was not surprising given that the amino acid sequence of this protein is highly conserved across serotypes. 29
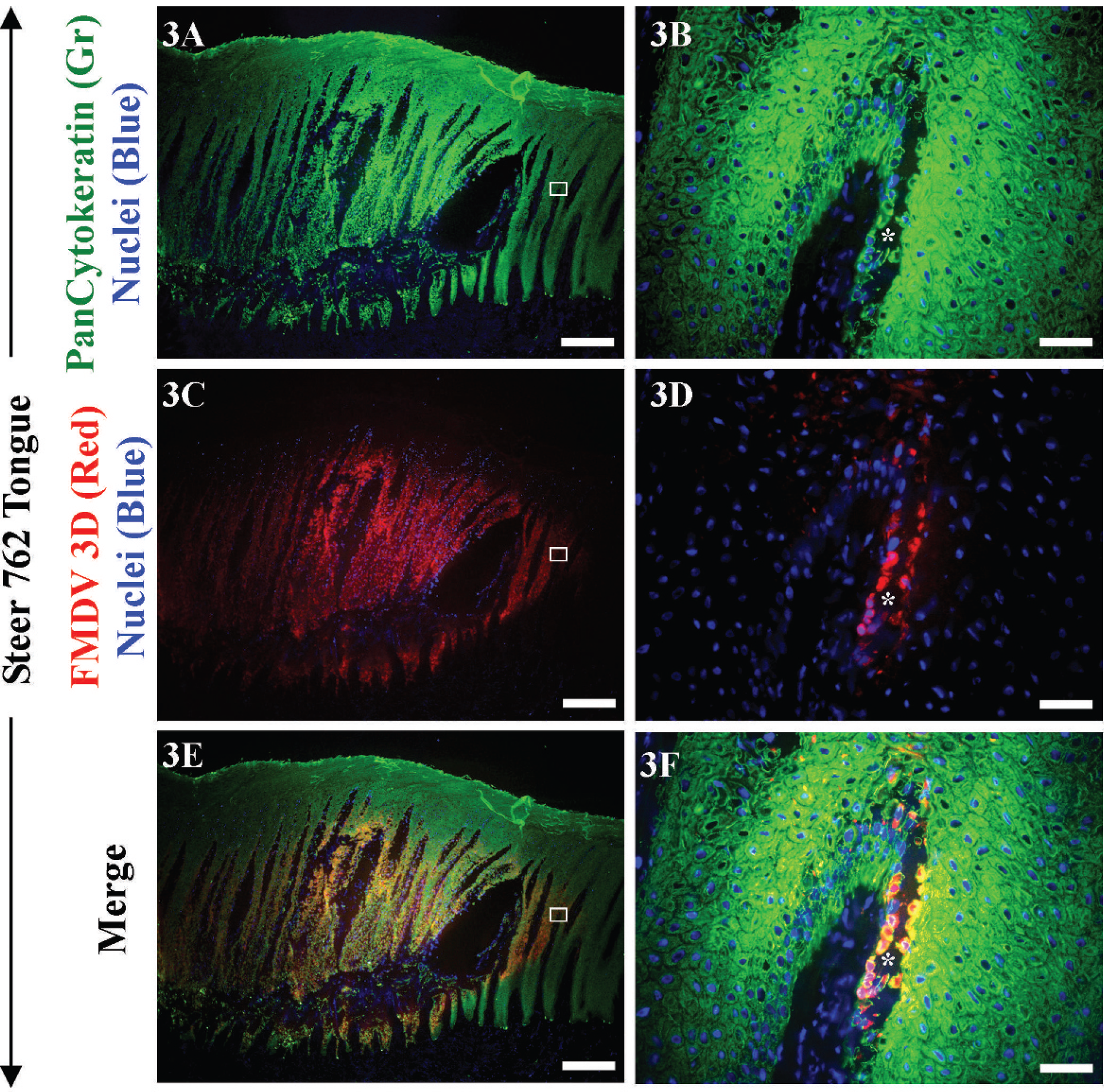
Steer 762, fluorescent antibody technique of lingual vesicle. Forty-eight hours after aerosol inoculation Foot-and-mouth disease virus (FMDV) strain O1-Manisa. Pancytokeratin (pCK) green, FMDV 3D red, colocalization yellow in merge, nuclei blue. Right column higher magnification of region of interest identified in left, low magnification image.
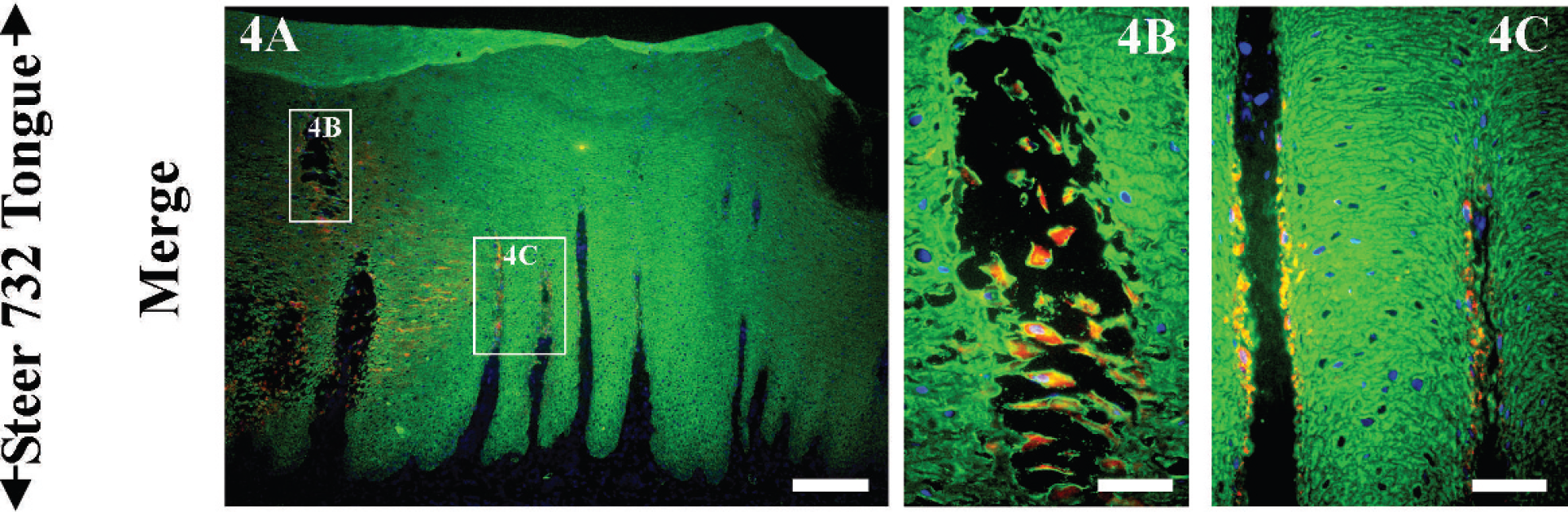
Steer 732, fluorescent antibody technique of periphery of lingual vesicle. Twenty-four hours postinoculation intraepithelial lingual injection of Foot-and-mouth disease virus (FMDV) strain O1-Manisa. Pancytokeratin (pCK) green, FMDV 3D red, colocalization yellow, nuclei blue. Right column higher magnifications of regions of interest identified in left, low magnification image. A, FMDV antigen localizes to regions of early cavitation (vesiculation). Bar = 200 μm.
Pig 18566, fluorescent antibody technique of snout vesicle. Five days postinoculation intraepithelial heel bulb injection of Foot-and-mouth disease virus (FMDV) strain Asia1-A1. Right column higher magnification of area of interest identified in left, low magnification image.
Steer 7200, fluorescent antibody technique of lingual vesicle. Forty-eight hours after aerosol inoculation Foot-and-mouth disease virus (FMDV) strain O1-Manisa. Pancytokeratin (pCK) green, FMDV 3D red, S100 blue, colocalization of FMDV with pCK is yellow-orange within vesicle acantholytic keratinocytes.
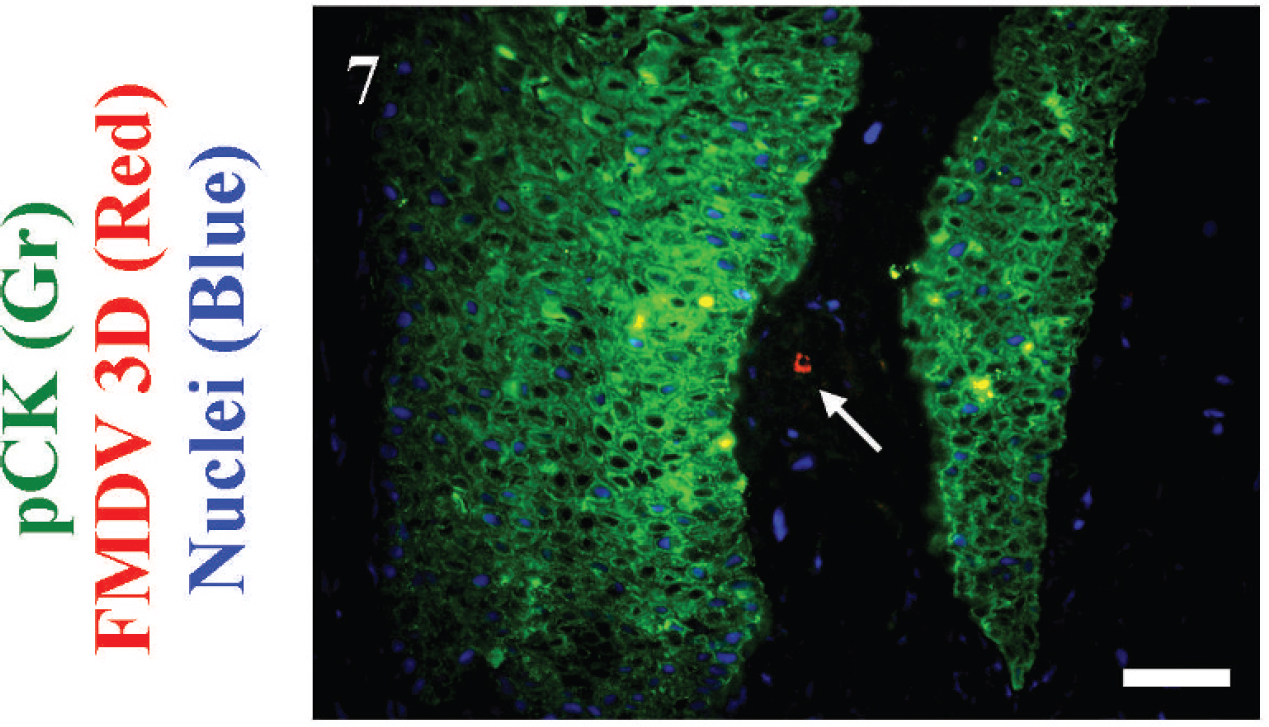
Pig 871, fluorescent antibody technique of tissue subjacent to interdigital vesicle. Six days postinoculation of Foot-and-mouth disease virus (FMDV) strain C3-Resende. Pancytokeratin (pCK) green, FMDV 3D red, nuclei blue. FMDV-positive, pCK-negative cell within dermis (arrow). Bar = 50 μm.
Steer 732, fluorescent antibody technique of deep aspect of lingual vesicle. Twenty-four hours postinoculation intraepithelial lingual injection of Foot-and-mouth disease virus (FMDV) strain O1-Manisa. Pancytokeratin (pCK) green, FMDV 3D red, colocalization yellow, nuclei blue. FMDV-positive, CK-negative cell within dermis (arrow). Bar = 20 μm.
Steer 762, fluorescent antibody technique of interdigital vesicle. Forty-eight hours after aerosol inoculation Foot-and-mouth disease virus (FMDV) strain O1-Manisa. Pancytokeratin (pCK) green, FMDV-capsid red, MHC-II aqua, nuclei blue.
The characterization of efficacies of various antigen retrieval and secondary detection systems will be useful to investigators developing IHC protocols for FMDV or other antigens in bovine tissues. Such comparisons are scarcely available in the published literature. The finding of the current study, that HIER and proteolytic retrieval techniques are effective for IHC of FMDV antigens, will allow flexibility in the adaptation of the techniques described.
Observations in the current study that FMD vesicles progress peripherally via infection of basal cells surrounding dermal papillae is consistent with earlier work, 31 which concluded that these regions serve as the sites of initiation of lesions. A recent report 7 that indicated that these regions are strongly (FMDV) ISH-positive in mature lesions also reinforced findings of the present study. However, the notion that dermal mononuclear cells serve as “virus-carrying cells,” transporting FMDV from the blood to the epithelium, presented in a previous work, 31 is not supported by the present study. The rarity with which virus-positive, cytokeratin-negative cells were identified in the papillae suggests that these cells (presumptive dendritic cells) do not have a significant or consistent role in vesicle maturation. The disparity of results between the current study and that earlier work 31 may be explained by technical and/or study design differences between the 2 investigations. Aspects of the earlier work that could explain this disparity include the different strain of FMDV used and the strategy within that study of examining lesion predilection sites before any lesions were grossly detected. 31
The IHC and FA techniques in the current study identify virus-positive cells within the dermal papillae. However, the double and triple FAT stains indicate that these cells are of epithelial origin and are, in fact, acantholytic keratinocytes rather than dermal mononuclear cells. Examination using a single, virus-only label visualization system might have led to misidentification of these cells as dermis-derived. Overall, the conclusion of the present study is that under the conditions used, FMDV moves from the vasculature to basal keratinocytes in a cell-free manner. Furthermore, the detection of microvesicles surrounding dermal papillae distant from developing vesicles suggests that a single vesicle is often formed through multiple transfer events of virus from blood to epithelium. The rare findings of FMDV 3D and capsid antigen positivity in dermal CK-negative cells support the notion that a population of nonepithelial cells becomes infected as lesions progress. However, the role of these cells is unclear. It is possible that despite the rarity of detection of such cells, they do play a significant role in vesicle maturation. In addition, since these cells are identified near mature lesions, it is not possible to discern whether the FMDV antigens contained within these cells came from the vasculature or the adjacent vesicle.
As dermal papillary microvesiculation proceeds to cavitation, the basal lamina is necessarily breached exposing the dermis; hence, each such occurrence, technically, is considered an ulceration (ulcer in situ). However, this process is morphologically distinct from the true ulceration of the deep basal lamina occasionally seen in advanced vesicles. The finding of relative abundance of FMDV, MHCII-double-positive cells subjacent to disrupted basement membranes in regions of true ulceration also is distinct from the characteristics of vesicle maturation described above (ulcer in situ). It is likely that, in such instances, the observed viral antigens are contained within dermal phagocytic cells, which are engaged in the elimination of foreign material and necrotic cellular debris. These cells are more likely involved in resolution of the lesion rather than progression. However, it has been suggested that in early FMDV infection, the major systemic viral amplification occurs in various epithelial sites. 1 This hypothesis is not definitively ruled out by the current work.
In the characterization of FMD vesicles, authors commonly describe affected regions of skin and oral cavity using nomenclature that is appropriate for typical epithelia with uniformly lamellar strata (i.e., strata basale, spinosum, granulosum, corneum). However, the typical FMD lesion sites (interdigital skin, coronary band, tongue, and snout) are composed of epithelia that are anatomically complicated by the interdigitation of long dermal papillae with epithelial pegs. This arrangement presumably provides greater resistance to abrasion through a greater surface area of adhesion between basal cells and dermis. The complication arises in that the dermal papillae insert dermal tissue outwardly to regions that are superficial to subjacent regions of epidermis. This leads to the similarly awkward arrangement of basal cells surrounding dermal papillae, which (in 2 dimensions) are superficial to apparently subjacent spinous keratinocytes. In the current study, this concept is more than just semantic in that the observation that vesicles propagate initially through infection of peripapillary basal cells suggests that these cells have increased susceptibility to FMDV infection relative to adjacent spinous cells. This may be a biological vulnerability due to differential expression of virus receptors 17,19 or other factors, or simply a physical characteristic purely associated with proximity to virus exiting dermal capillaries.
The finding of supracoronary hair follicle mural micro-abscesses with identification of FMDV antigens in the current study is novel, but not surprising. Subgross 9 and grossly detectable 20 lesions of haired skin have been reported in FMDV-infected livestock, and FMDV has been isolated from the nonlesional haired skin of cattle. 9,10 Localizing FMDV to porcine follicular epithelium has implications to the understanding of FMD pathogenesis, as it suggests that these cells have biological qualities that confer a susceptibility to infection similar to keratinocytes of classic lesion sites.
The IHC and FA techniques described in the current study could facilitate various aspects of FMDV investigation. With more thorough examination of sensitivity and specificity, rapid and effective diagnostic tests could easily be validated, which could readily be utilized by laboratories lacking resources for diagnosis through PCR or VI. As research tools, these techniques have allowed precise localization of FMDV in various stages of infection, which will be described in additional manuscripts forthcoming from the authors' laboratory.
Acknowledgements
The authors wish to thank Dr. Juan Pacheco for assistance with animal inoculations and monitoring, Dr. Vivian O'Donnell for consultations on fluorescence microscopy techniques, Meghan Tucker for histotechnological support, and Michael LaRocco for assistance with growing hybridoma cell lines. This work was funded by USDA-ARS CRIS project 1940-32000-052-00D.
Footnotes
a.
Sakura Finetek USA Inc., Torrance, CA.
b.
SuperFrost® Plus, Thermo Fisher Scientific Inc., Waltham, MA.
c.
Dako North America Inc., Carpinteria, CA.
d.
Biocare Medical, Concord, CA.
e.
Sigma-Aldrich, St. Louis, MO.
f.
VECTASTAIN® ABC-AP, Vector Laboratories Inc., Burlingame, CA.
g.
EnVision™ G‖2 System, Dako North America Inc., Carpinteria, CA.
h.
MACH 3 Mouse AP Polymer, Biocare Medical, Concord, CA.
i.
Invitrogen Corp., Carlsbad, CA.
j.
ImmPRESS™ Anti-Mouse Ig (peroxidase), Vector Laboratories Inc., Burlingame, CA.
k.
Vector® RED Substrate Kit, Vector Laboratories Inc., Burlingame, CA.
l.
ImmPACT™ DAB Substrate, Vector Laboratories Inc., Burlingame, CA.
m.
Vector® NovaRED™ Substrate Kit, Vector Laboratories, Burlingame, CA.
n.
Catalog No. CAT82A, VMRD Inc., Pullman, WA.
o.
Molecular Probes Inc., Eugene, OR.
p.
Vector Laboratories Inc., Burlingame, CA.
q.
Olympus DP70, Olympus Imaging America Inc., Center Valley, PA.
r.
Adobe® Photoshop® CS2, Adobe Systems Inc., San Jose, CA.
