Abstract
Brain and spinal cord histopathology findings in male and female 20-month-old mice in a large-scale aging study of 28 inbred Jackson Laboratory mouse strains from 7 genetic families are described. Brain sections from selected strains at 12 and 24 months of age or older were also reviewed. Common lesions include axonal dystrophy in the gracile and/or cuneate nucleus in the sensory tract of the dorsal medulla and in the spinal cord in all strains. Hirano-like bodies were seen in 24/28 strains, and mineralization was observed in the thalamus of 9/28 strains. Less common lesions were also seen in the cerebellum, cerebral cortex, and other brain areas. No brain or spinal cord tumors were found. Evidence of an impairment of the ubiquitin-proteasome system (UPS) and/or suspected autophagy was manifested as medullary axonal dystrophy with intra-axonal granular eosinophilic bodies and LC3B immunohistochemistry in most strains. RIIIS/J, the most severely affected strain, showed moderate axonal dystrophy at 12 months, which progressed to severe lesions at 20 months. Comparative pathology in various species is discussed.
Keywords
Large numbers of inbred laboratory mouse strains are available in repositories worldwide. However, only a few are used regularly in biomedical research. Some strains regularly develop spontaneous diseases that are remarkably similar to human diseases. These conditions are usually caused by mutations in the same gene in both species, such as pseudoxanthoma elasticum in C3H/HeJ, DBA/2J, KK/HlJ, and some 129 strains. 2 These findings make the strains uniquely valuable. Mice of a single inbred strain have no genetic diversity. Comparing lesions in groups of mice from a variety of standard inbred strains provides more variability but it is still limited. Outbred strains can provide variable degrees of genetic diversity, depending on how outbred they are, but result in a high degree of disease variability between individuals. Knowing disease predilection in a large number of inbred strains provides clues on potentially new mouse models for human diseases as well as information on background lesions that might interfere with interpretation of experimental results. Pathology of aging mice has often shown strain-related causes of death and pathologic findings in many tissues.3,7,21,44,52,53,59
Neuropathology in young and aging mice of various strains has been reported.8,10,11,19,22,27,44,45,49,57,66 To expand our understanding of strain-related phenotypes, an aging study was conducted on 31 inbred mouse strains. Median lifespan was usually strain-related 68 and tumors were a major finding. 53 This phenotyping study included histopathologic evaluation of the mice14,53 and reported histopathology findings for many tissues, but did not include neuropathology.14,53,54,67,68 For this report, we reviewed the histopathology in brains and spinal cords of 28 mouse strains in this aging study.
Materials and Methods
Mice
Twenty-eight of 31 inbred mouse strains, which were selected to represent genetic diversity for a large-scale aging project, were studied for brain and spinal cord lesions.4,14,30,52,53,67,68 Strains used were 129S1/SvImJ, A/J, BALB/cByJ, BTBR-T+tf/J, BUB/BnJ, C3H/HeJ, C57BL/10J, C57BL/6J, C57BLKS/J, C57BR/cdJ, C57L/J, CBA/J, DBA/2J, FVB/NJ, KK/HIJ, LP/J, MRL/MpJ, NOD.B10Sn-H2b/J (a congenic strain with the NOD genetic background but with a histocompatibility locus from a diabetes-resistant strain), NON/ShiLtJ, NZO/HlLtJ, NZW/LacJ, P/J, PL/J, PWD/PhJ, RIIIS/J, SM/J, SWR/J, and WSB/EiJ. Mice were obtained at 6 to 8 weeks of age and divided into 3 groups. Two groups of mice were used in a cross-sectional study to define disease onset at defined ages. These groups were euthanized and necropsied at approximately 12 (372–418 days) and 20 (606–663 days) months of age, respectively. A third group was evaluated in a longitudinal study that kept mice until they became moribund or died naturally (composed of 65 females and 35 males, 555–985 days of age). This neuropathology review included 365 male and female mice of 20 months old and selected mice at 12 and over 20 months of age. Those selected included mice with typical lesions noted. For logistics purposes, we could not select more mice for these studies. Strains of the selected mice are noted in the results.
Mouse rooms were maintained on a 12-hour light-dark cycle and at an ambient temperature of 21-23C. Mice of the same sex (4 per cage) were housed in duplex polycarbonate cages (31 X 31 X 214 cm) on pressurized, individually ventilated mouse racks (Thoran Caging System, Hazleton, PA) with a high efficiency particulate air-filtered supply and exhaust. Mice were fed ad libitum pellets containing 6% fat (LabDiet 5K52; PMI Nutritional International, Bentwood, MO) and given free access to acidified water (pH 2.8–3.2). Mice were maintained in a confirmed pathogen-free environment with routine monitoring for viruses, bacteria, parasites, and microsporidium (https://www.jax.org/jax-mice-and-services/customer-support/customer-service/animal-health). All mice were euthanized by CO2 asphyxiation using methods approved by the American Veterinary Medical Association (
Pathology
At necropsy, the vast majority of brains were fixed in situ in Bouin’s fixative, with the calvarium removed at the time of necropsy and decalcified with Bouin’s. A few brains were fixed in Fekete’s solution and decalcified in Cal-Ex (Fisher, Pittsburgh, PA). One mid-sagittal or one parasagittal section (Supplemental Fig. 1) for the right half of the brain with the skull and coronal sections for the left half of the brain were prepared. The coronal sections were at variable but not well-defined levels. Denominators for each lesion noted were based on the finding of the medullary nuclei examined (the gracile and/or cuneate nucleus). If the gracile or cuneate nucleus was not found in the sagittal or coronal section, the mouse was not included in the frequency counts for all lesions. Three of the original 31 strains evaluated in the large-scale aging project are not included in this report: AKR/J mice had a mean survival time of only 251 days due to a high frequency of lymphomas 67 so the brain and spinal cord sections were not reviewed. SJL/J mice, with a mean survival time of 476 days due to fighting, 53 had only coronal brain sections and no gracile/cuneate nuclei were present in these sections. CAST/EiJ mice developed unusual vertebral and spinal cord lesions that will be reported in another publication.
Brain and spinal cord histopathology slides from 28 strains were reviewed for central nervous system lesions. Most coronal sections did not show any of the 2 medullary nuclei of interest. We tried to get at least 5 males and 5 females from each 20-month-old group, but some strains had fewer than 5 mice of one sex because of the inadequate sectioning. Selected slides of some 12- and 24-month-old mice were also reviewed. However, the brains of mice that lived past 20 months were sectioned coronally so that the gracile nucleus was not usually identified in the sections. The spinal cord with vertebrae was fixed in Bouin’s or Fekete’s solutions and trimmed in cross and longitudinal sections at the thoracic and lumbar regions. Tissues were embedded in paraffin and sections were stained with hematoxylin and eosin stain. Selected sections from various strains of mice were stained by the periodic acid-Schiff (PAS) method, acid fast, and/or von Kossa stains.
For immunohistochemistry, 4 μm sections from 13 mice with representative axonal dystrophy (two 12-month-old RIIIS/J, three 20 month old RIIIS/J, two 2-year-old RIIIS/J, one, 20 month old C57BLKS/6J, three, 2-year-old C57BL/6J, one, 20-month-old C57L/J, and one, 20-month-old BUB/BnJ) showing typical medullary nuclei lesions, were mounted on positively charged glass slides (Superfrost Plus; Thermo Fisher Scientific). Detection of both LC3B (
Autofluorescence of brains and spinal cord sections from mice with representative lesions, including some with neuronal pigments seen in formalin fixed paraffin embedded tissue and H&E stained brain and spinal cord sections, was evaluated for 10 total mice of some strains, using a Keyence (Itasca, IL) BZ-X710 digital fluorescent microscope with a BZ-X GFP filter to detect green autofluorescence, filter wavelength range (470 excitation/525 emission); and a BZ-X710 Texas Red filter to detect red autofluorescence (560 excitation, 630 emission).
Results
General Comments
Most brain sections had few or no areas of dark neuron artifact, especially in the cerebral cortex and cerebellum.19,27 The lack of this neuronal artifact, probably because of fixation in situ with Bouin’s fixative, made it easier to record and score the aging lesions noted. The prevalence of all lesions in a specific anatomical region was dependent on the plane of section. Most of the coronal sections and some sagittal sections did not reveal the gracile or cuneate nuclei and did not allow for the identification of lesions. However, we recorded lesions in coronal sections of these nuclei when they were present. If the gracile/cuneate nucleus was not in any of the brain sections of a mouse, the mouse was not counted in the denominator or numerator for all brain lesions. When Fekete’s fixative was used, the brain sections were only coronal, and vacuoles and crystals, which were considered artifacts, were often seen in white matter.
The most common brain lesions in 365 mice that are 20-month-old from 28 strains included axonal dystrophy (AD, dystrophic axons, neuroaxonal dystrophy, and spheroids) in the gracile and/or cuneate nuclei of the caudal hindbrain 60 and somatosensory tracts of the spinal cord, Hirano-like bodies in the thalamus, and mineralization in the thalamus. Cerebral or spinal cord tumors were not found in any of the 365 mice. Frequency by strain is summarized (Fig. 1) by genetic group43,67,68 and presented in detail in Supplementary Table 1. Both sexes are combined (Supplementary Table 1) since the frequency of each lesion type was similar in the males and females for all lesions except for a few noted strain differences, but numbers were not sufficient for statistical analysis. The coronal sections of the brains of mice in the longitudinal study often lacked gracile/cuneate nuclei, and thus these mice were not included in the tables.
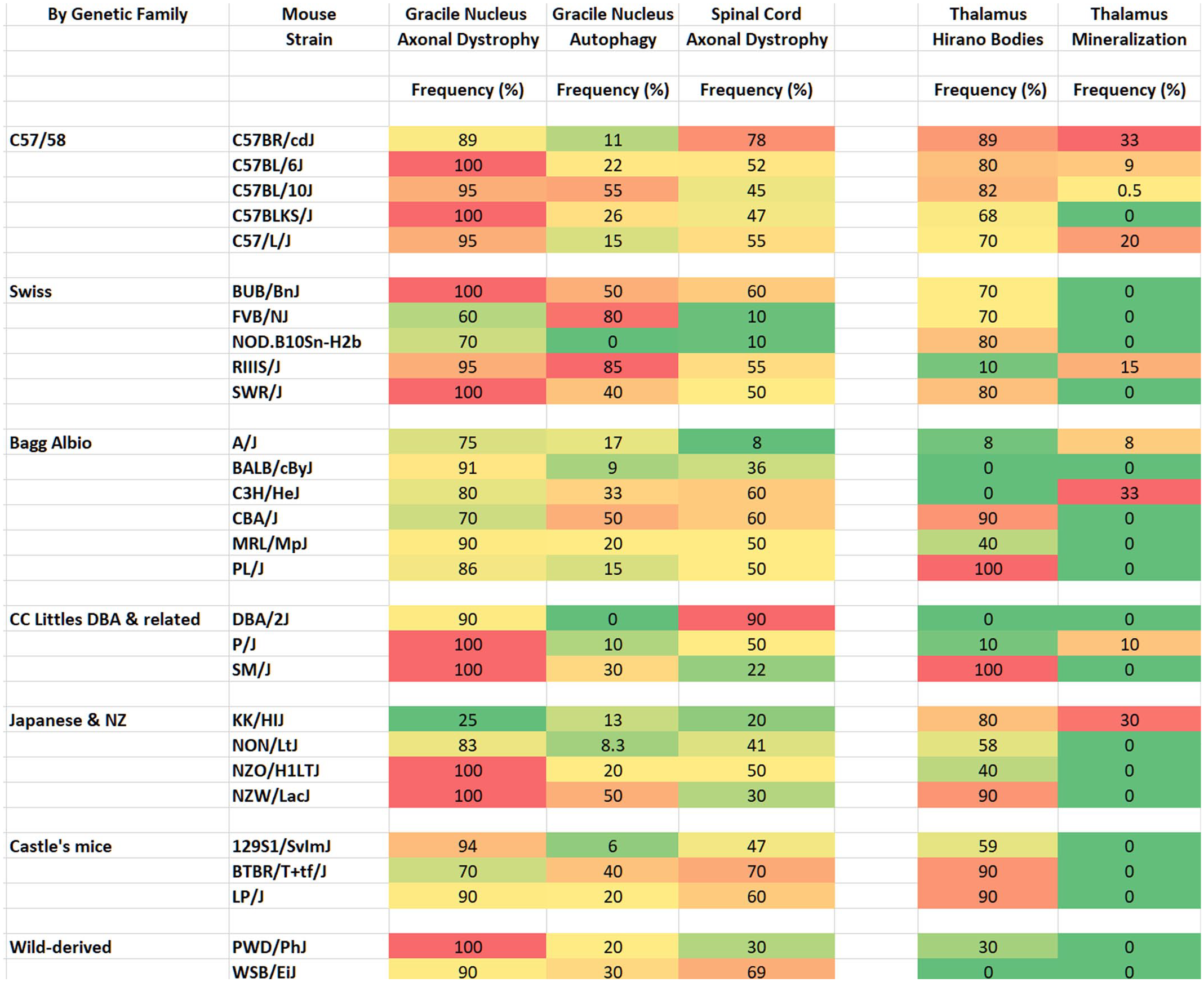
Heat map—The frequency (%) of brain and spinal cord lesions in 365, 20-month-old mice from twenty-eight mouse strains. “Autophagy” is used for “suspected autophagy.” The frequency of each lesion in males and females are combined for each strain. There are 7-23 male and female mice for each strain.
Axonal Dystrophy
Axonal dystrophy in the gracile nucleus/cuneate nucleus was common (60-100%) in 20-month-old male and female mice of all strains (Fig. 1, Supplemental Table 1) and in selected 12-month-old mice. There was no obvious sex difference. Most sections included portions of the gracile nucleus, but it was not possible to distinguish between the 2 nuclei in all sections as previously described in aging mice. 57 The extent and severity of lesions (numbers of spheroids per nucleus) varied by mouse strain and among mice within a strain. The C57/58 group (C57BL/6J, C57BL/10J, C57BR/cdJ, C57BLKS/J, C57L/J) had the highest frequency, and Castles’s mice group had the lowest frequency. Axonal dystrophy varied from a few dystrophic axons (spheroids) to many per nucleus section (Fig. 2a–e). Spheroids, which were usually uniformly pale eosinophilic, sometimes contained densely eosinophilic bodies often with granular or hyaline eosinophilic substructures, which were variable in size and shape (Fig. 2c–e, Supplemental Fig. 2), noted as autophagy (suspected) in Fig. 1 and Supplemental Table 1. These densely eosinophilic bodies were not always within obvious dystrophic axons. Strains most commonly found with these dystrophic axon substructures include RIIIS/J, BUB/BnJ, C57BL/10J, C57BLKS/J, C57L/J, P/J, SM/J, NZO/HILtJ, 129S1/SvImJ, and PWD/PhJ. Of these strains, RIIIS/J mice had the most severe AD in the gracile nucleus. Often, neuronal nuclei had marginated chromatin and were enlarged or free of heterochromatin (Fig. 2c, Supplemental Fig. 2), or had other abnormal chromatin patterns not seen in nuclei without AD.
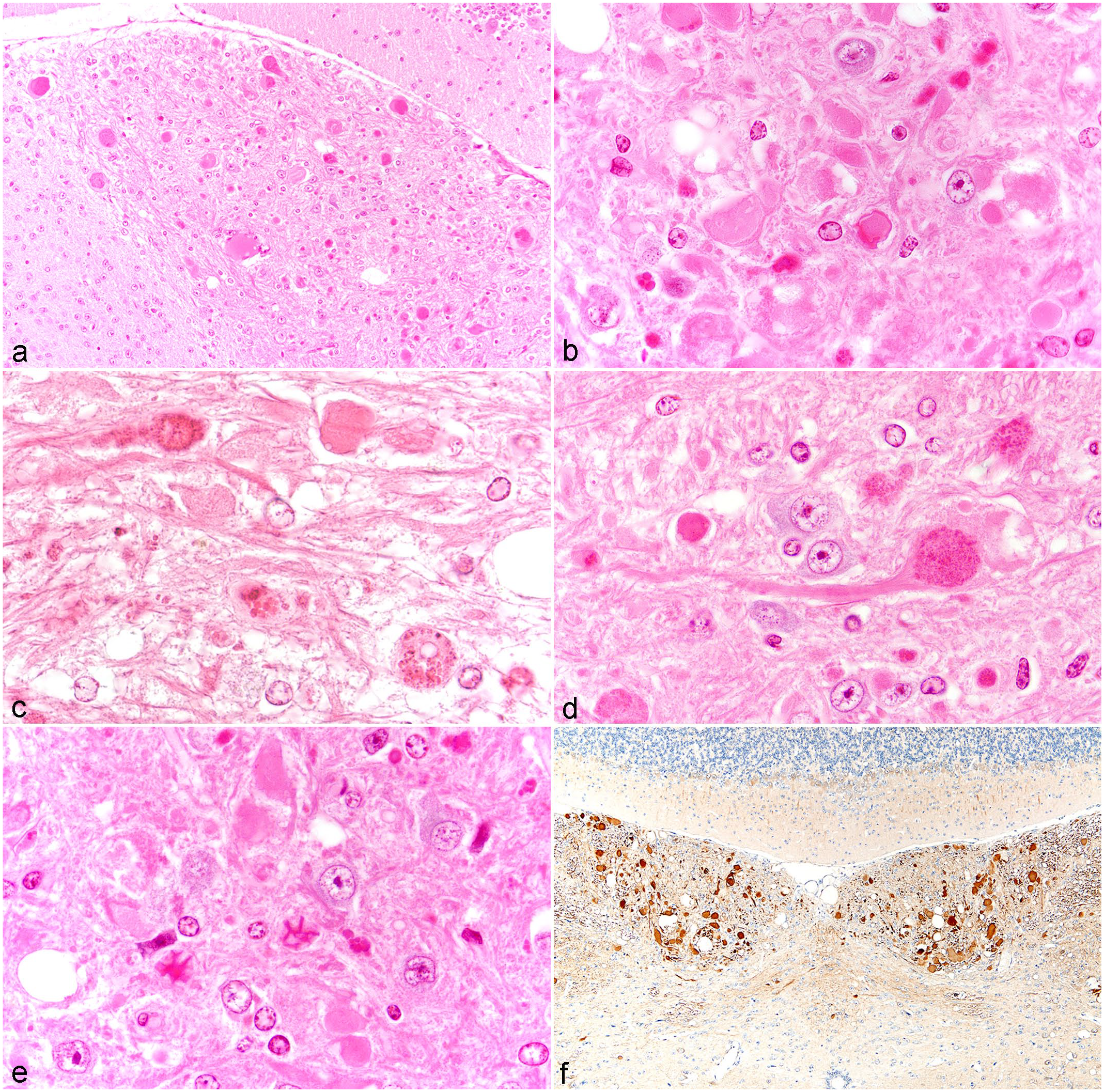
Axonal dystrophy in the gracile nucleus: (a) Severe axonal dystrophy in a 12-month-old RIIIS/J gracile nucleus. Left side is the nucleus of the solitary tract. (b) Spheroids (pale eosinophilic) and more densely eosinophilic bodies in the gracile nucleus of a 20-month-old C57BL/10J mouse. (c) Eosinophilic bodies and granules in spheroids of the gracile nucleus from a 20-month-old BUB/BnJ mouse. Note neuronal nuclei free of chromatin, some with marginated chromatin and one neuron (lower right corner) with a degenerative clear small nucleus and cytoplasm with eosinophilic granules. More normal gracile nucleus neuronal nuclei are seen in Fig. 2d with a central nucleolus. (d) Densely granular material in a spheroid attached to a long axon in the gracile nucleus in a 20-month-old RIIIS/J mouse. (e) Eosinophilic granules, some with crystalline-like structures in spheroids in the gracile nucleus from a 20-month-old RIIIS/J mouse. (f) Abundant LC3B (Microtubule-associated protein 1 light chain 3 beta,
A review of the brain sections of 12-month-old mice from the selected strains noted below revealed a lower frequency and/or severity of AD than in 20-month-old mice of the same strains. The frequency of the lesions included 16 of 16 RIIIS/J mice (Fig. 1), 6 of 9 NZO/HILtJ mice, 4 of 8 FVB/NJ, 4 of 6 NZW/LacJ mice, 0 of 9 C57BL/6J mice, 2 of 11 C57BL/10J), and 0 of 3 BUB/BnJ mice. RIIIS/J mice had the highest incidence and were among the most severe lesions (Fig. 2a). All gracile/cuneate nuclei of 16, 12-month-old RIIIS/J male and female mice reviewed had AD and 14 had the eosinophilic granular material in the spheroids, as noted below.
Evidence suggestive of autophagy in dystrophic axons was observed in H&E-stained sections of the gracile nucleus associated with AD in most strains at 20 months of age, especially in strains with the highest incidences of AD (Fig. 1, Supplemental Table 1). Dense eosinophilic small or large irregular bodies, granules, and irregularly shaped large axonal inclusions were seen in the pale eosinophilic spheroids (Fig. 2b–d, Supplemental Fig. 2). Rarely, crystalline-like structures were noted (Fig. 2e). These spheroids were sometimes connected to less swollen axons (Fig. 2d), which were nevertheless abnormal due to their presence as swollen axons. Rarely, eosinophilic bodies appeared in degenerating neurons. They were also found in spinal cord spheroids. These eosinophilic granules and bodies were not seen in other nuclei nor in individual neurons in other brain regions.
Evaluation of the immunohistochemistry of the brain lesions in 13 male or female mice of the strains noted in the methods provided evidence that AD was associated with autophagy. The spheroids and eosinophilic bodies were commonly reactive for LC3B (
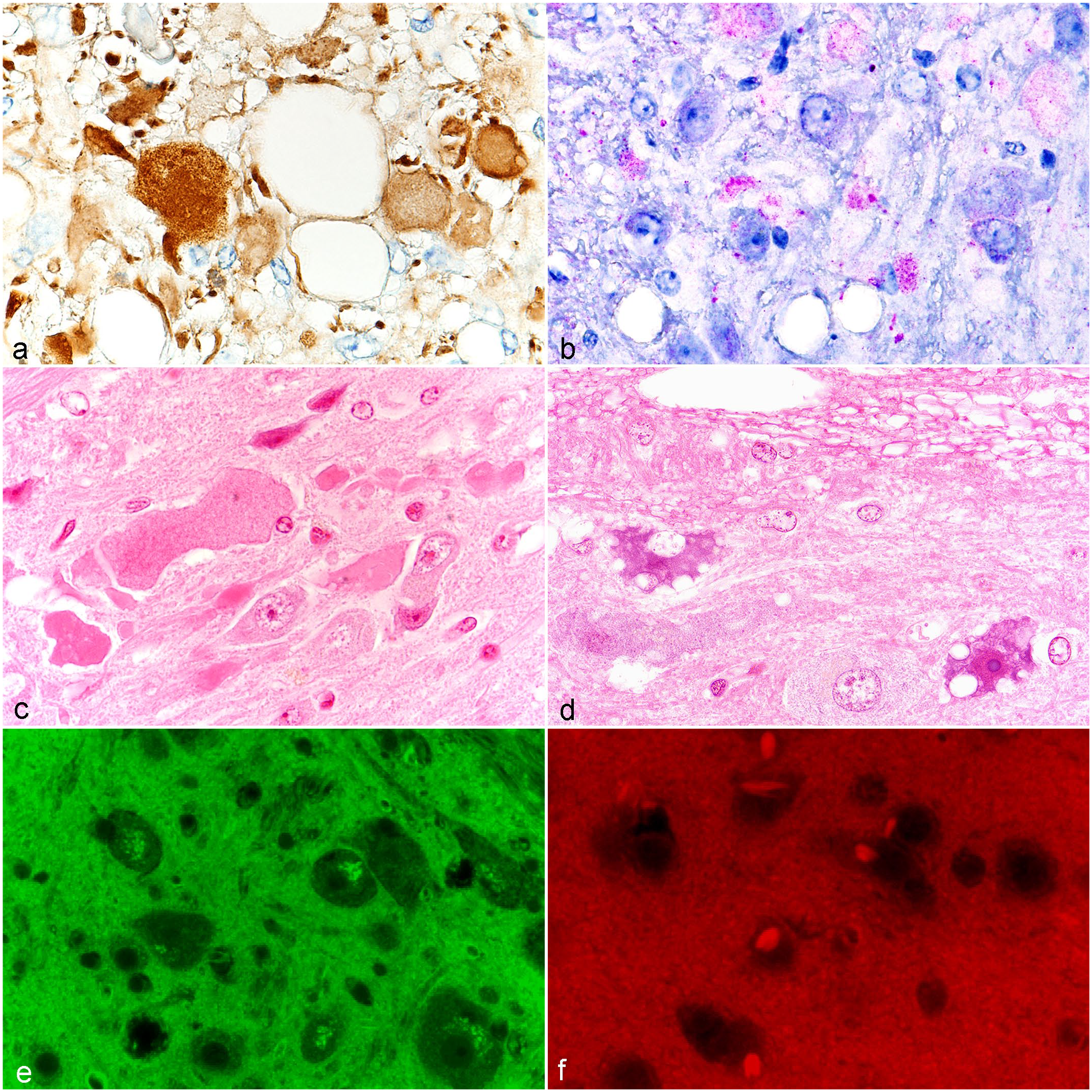
Brain and spinal cord lesions: (a) LC3B (Microtubule-associated protein 1 light chain 3 beta,
The spinal cord also showed focal AD in many mouse strains (Fig. 3c). The lesions tended to be in the same tract in an individual cord section, and were more often seen in longitudinal sections than in cross sections. The higher frequency of spinal cord AD in a strain was not necessarily associated with a higher frequency or severity of AD in the gracile nucleus. Scattered degenerative neurons were often seen in the spinal cord, with vacuoles or pigmented cytoplasm (Fig. 3d). In 3/10 mice studied, the pigmented cells showed green autofluorescent material (Fig. 3e) and were PAS-positive.
Hirano-Like Bodies and Other Lesions
Hirano-like bodies in the thalamus were found in most but not all strains (Fig. 1, Supplemental Table 1). C57/58 and Castle’s mice strains had the highest frequencies. Hirano-like bodies appeared as uniformly eosinophilic, round to oval-shaped or elongated structures (Supplemental Fig. 3). They were mostly homogeneous eosinophilic, but some had substructures. They were not PAS-positive, but were autofluorescent with 560 nm excitation, (Fig. 3f) but not with 470 nm excitation. No other neuronal or neuropil lesions were found in the brain areas reviewed. Bilateral focal mineralization associated with arterioles in the thalamus was only seen in 9 strains (Fig. 1, Supplementary Table 1).
We performed PAS staining on 9 mice from representative strains (FVB/NJ, C57BL/6J, PWD/PhJ, BUB/BnJ, CBA2/J) and confirmed that the mice had PAS-positive granules in the neuropil of the hippocampus of all strains reviewed.13,26,32,35,57 Many neurons in the cerebral cortex, cerebellum (Purkinje cells), medulla, thalamus, and spinal cord often had small PAS+ granules which usually were not present as pigmented granules in H&E stain nor autofluorescence with a GFP green filter. The cerebellar Purkinje cells had pigmented cytoplasm occasionally but were also usually not autofluorescent. The medial vestibular nucleus was not often seen in sections, but when present it occasionally had neurons with clustered eosinophilic cytoplasmic granules that were PAS+.
Discussion
The frequencies of common brain and spinal cord lesions in aging mice from 28 Jackson Laboratory mouse strains revealed some differences by genetic groups and strain, but we found no consistency within the groups. The lesions included axonal dystrophy in the gracile/cuneate nuclei and spinal cord, Hirano-like bodies in the thalamus, and mineralization in the thalamus. The C57/C58 group generally had higher frequencies of axonal dystrophy in medullary nuclei. Hirano-like bodies were not found in some families, while thalamic mineralization was seen sporadically in most families. Genetic causes of each of these 3 common age-related central nervous system lesions would require large numbers of individuals to generate detailed quantitative strain differences.
The finding of axonal dystrophy in the medullary nuclei has been reported previously in some lines of aging inbred strains of mice,8,11,57,63 including the ubiquitin carboxy-terminal hydrolase L1; gracile axonal dystrophy (Uchl1gad) mutant,38,40,64 and other lines of mutant mice.5,20,23,24,28,37,51,58 At least 43 mouse mutants have shown axonal spheroids in the brain (http://www.informatics.jax.org/mp/annotations/MP:0010047).
Axonal dystrophy has also been described in these medullary nuclei and in nerves in other species, including humans, 6 rats,17,19,36 dogs,42,66 and nonhuman primates.12,16 Loss of neurons in the cuneate nucleus of aging mice has been reported. 50 It was reported in C57BL6/J mice and 3 other mouse genotypes that axonal dystrophy is not affected by dietary restriction in aging studies. 9 The high frequencies and lesion severity for AD were not associated with obvious clinical signs, although neurobehavioral phenotyping was not performed.
This appears to be the first report suggesting autophagy involvement29,56,62 in medullary nuclear axonal dystrophy in strains of aging mice, based on morphology and IHC. Eosinophilic inclusions, bodies, or dots were often seen in the spheroids with ubiquitin-positive AD 15 and eosinophilic bodies were LC3B-positive as markers for autophagy29,56,62 and in the spontaneous neuroaxonal dystrophy (nad) mutant mice. 5 The localization of ubiquitin and LC3B in dystrophic axons suggests that there is impairment of the ubiquitin-proteasome system (UPS) and/or autophagy. Unfortunately, the genetic basis of the spontaneous nad mutant mice was never investigated, and the colony has been lost.
Evidence of autophagy (large autophagosomes, dense eosinophilic bodies or granules in dystrophic axons) was not seen in a large study of aging C57BL/6J mice with brains fixed in formalin or paraformaldehyde. 57 The reason is not known since we found it commonly in our C57BL strains (Supplemental Table 1). Perhaps Bouin’s fixation may have played a role in the morphological appearance. Autophagy has been reported in damaged axonal mitochondria in some mutant mice, 1 the brainstem of tectonin beta-propeller repeat containing 2 (Tecpr2em1Wei) mutants, 55 and in various neurons in dogs with a mutation in the autophagy related 4D, cysteine peptidase (ATG4D) gene 31 and mutant Atg4bGt(OST264114)Lex mice. 46 In human brain, granular bodies and granulovacuolar degeneration, with evidence of autophagy, have been found in hippocampal neurons in degenerative diseases15,18,25,61 and eosinophilic crystalline bodies in the external cuneate nucleus. 25 There are some morphological similarities between the human eosinophilic bodies and the mouse medullary nuclei lesions, which are found mostly in dystrophic axons.
Lipofuscin has been reported in aging brains of many species.48,66 Lipofuscin was reported in aging mouse brains,8,9,41,47,57 based on histological appearance, PAS stain, and/or electron microscopy, but often without green autofluorescence. We saw many PAS-positive granules in cerebral cortical and other neurons, but autofluorescence was limited to some cerebellar and spinal cord neurons. Autofluorescence appears to be helpful for the diagnosis of lipofuscin. 48 It was suggested that different types of lipofuscin may occur in mice. 41
Several other aging mice brain lesions were not reviewed in this study in detail for various reasons. We did not use GFAP IHC to characterize gliosis, an apparently common finding in some strains of aging mouse brain.10,34,57,59 PAS stains of aging mouse brain have shown common PAS-positive granules in the hippocampus and other regions of many mouse strains.13,26,32,35,57 We confirmed its common occurrence in a few strains. Focal thalamic mineralization, the frequency of which was strain-related, was found to be blood vessel associated and highest in C3H mice. 39 C3H mice (as well as DBA/2J, KK/HlJ, and some 129 strains) have the same hypomorphic mutation in the Abcc6 gene that causes systemic ectopic mineralization and is a model for human pseudoxanthoma elasticum. 2 It was anticipated that strains prone to ectopic mineralization33,53 would also exhibit focal thalamic mineralization, but this was not consistent. Some strains, such as C3H/HeJ and KK/HIJ, had thalamic mineralization, while others did not, notably DBA/2J and BALB/cByJ. It is likely that a higher incidence of mineralization would be found with more step sections of the thalamus. Hirano-like bodies57,65 were also strain-related and were not found more commonly in 2 families. These bodies, associated with the actin cytoskeleton, are found in various human aging neurodegenerative diseases, but not in experimental mouse diseases.
Most of the strains in this study are readily available in The Jackson Laboratory’s Biorepository, which will enable more detailed studies on these lesions. It was disappointing to find that the RIIIS/J, a strain with the most severe and diverse lesions, is now listed as being extinct (https://www.jax.org/strain/000683); however, the diversity of lesions and distribution among strains provides a unique insight into genetic predisposition to these neurological abnormalities.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221120009 – Supplemental material for Brain and spinal cord lesions in 28 inbred strains of aging mice
Supplemental material, sj-pdf-1-vet-10.1177_03009858221120009 for Brain and spinal cord lesions in 28 inbred strains of aging mice by Jerrold M. Ward, Peter Vogel and John P. Sundberg in Veterinary Pathology
Supplemental Material
sj-xlsx-2-vet-10.1177_03009858221120009 – Supplemental material for Brain and spinal cord lesions in 28 inbred strains of aging mice
Supplemental material, sj-xlsx-2-vet-10.1177_03009858221120009 for Brain and spinal cord lesions in 28 inbred strains of aging mice by Jerrold M. Ward, Peter Vogel and John P. Sundberg in Veterinary Pathology
Footnotes
Acknowledgements
We thank Jim Miller, Kathleen Silva, Michael McCluskey for necropsy support, Beth Sundberg for data support, and Nicholas Gott and Elaine Bechtel for histotechnology support (The Jackson Laboratory) and Meifen Lu (St Jude) for immunohistochemistry. We thank Rod Bronson for neuropathology protocol guidance during the study and Bob Garman, Jimmy Tarrant, and Enrico Radaelli for discussions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Ellison Medical Foundation, the National Institutes of Health (AG25707), and the American Lebanese Syrian Association (ALSAC).
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
