Abstract
A 14-year-old female alpaca (Vicugna pacos) was presented with a 1-week history of lethargy and anorexia and a 2-day history of recumbency, trembling, and hypothermia. There were no significant gross findings on postmortem examination. Hematoxylin and eosin–stained sections of brain demonstrated the presence of intracytoplasmic crystalline eosinophilic rod-shaped inclusions, mainly in the hippocampal pyramidal cells. Immunohistochemical staining for synuclein, tau protein, ubiquitin, and smooth muscle actin was negative. All inclusions were positive with phosphotungstic acid hematoxylin. Ultrastructurally, the inclusions were multilamellar and filamentous with longitudinal herringbone pattern and cross-sectional latticelike structure. The combination of hematoxylin and eosin appearance, special stains, immunostaining, and ultrastructural findings was consistent with Hirano-like bodies. The Hirano-like bodies were highly unlikely to be the cause of the neurologic signs experienced by this alpaca. To the authors' knowledge, this is the first report of spontaneous cerebral Hirano-like bodies in an alpaca.
Hirano bodies are intracellular eosinophilic crystalline 8 cytoplasmic inclusions seen most often in the hippocampus of human patients with Alzheimer disease and other neurodegenerative diseases, such as amyotrophic lateral sclerosis and Parkinson–dementia complex of Gaum. 7 They were also reported in 50 out of 65 brains of individuals who had no clinical findings. 11 Recently, we observed the presence of Hirano-like bodies in the hippocampus of a 14-year-old alpaca. We currently describe the light microscopic and ultrastructural findings of Hirano-like bodies in this alpaca. This is the first report of the presence of spontaneous cerebral Hirano-like bodies in an alpaca.
A 14-year-old female alpaca (Vicugna pacos) was presented with a 1-week history of lethargy and anorexia and with a 2-day history of recumbency, trembling, and hypothermia. On physical examination, the alpaca was mentally dull, unable to walk, and tachypnic. There was a slight head tilt, bilateral nystagmus, heart murmur, and poor contractility of the left ventricle on cardiac ultrasound. Due to the poor prognosis, the alpaca was humanely euthanized, and a complete necropsy was performed.
Material and Methods
Tissues collected at necropsy were fixed in 10% neutral buffered formalin, routinely processed, and embedded in paraffin according to accepted histologic technique. Five-micron-thick sections were stained with hematoxylin and eosin (HE), phosphotungstic acid and hematoxylin (PTAH), and Bielchovski stain for microscopic examination.
Paraffin blocks of the brain were sectioned, and immunohistochemistry was performed on 5-μm-thick sections derived from these blocks. Immunohistochemistry for smooth muscle actin (BioGenex San Ramon, CA), synculein (Zymed San Francisco, CA), tau (Nova Castra, Norwell, MA), and ubiquitine (Nova Castra) were performed on Techmate 1000 automatic immunostainer (Ventana Medical Systems, Tucson, AZ) using the avidin–biotin complex staining procedure after antigen retrieval and a DBA kit (Ventana Medical Systems) for the color-forming reaction. Samples of formalin-fixed brain (hippocampus) were cut into tissue blocks of 3 mm 2 and transferred into Trump’s Fixative. They were postfixed in 1% osmium tetroxide in sodium cacodylate buffer for 3 hours at 20°C. They were then en bloc stained with 5% aqueous uranyl acetate, dehydrated in graded ethanol series, and embedded in resin (Epon-812). Thick sections (1 μm) were stained with toluidine blue. Thin sections were cut at 50 to 70 nm on an ultramicrotome (LKB8801), stained with uranyl acetate and lead citrate, and photographed with transmission electron microscope (Philips EM 201).
Results
Gross findings included decreased body condition, mild dental malocclusion, and endocardial and myocardial white–tan discoloration of the left ventricle and interventricular septum. Few 1- to 2-cm-long nematode parasites were present in the lumen of the third gastric compartment (C3). These were macroscopically interpreted as Haemonchus contortus. Macroscopic examination of the brain was unremarkable even after formalin fixation and sectioning.
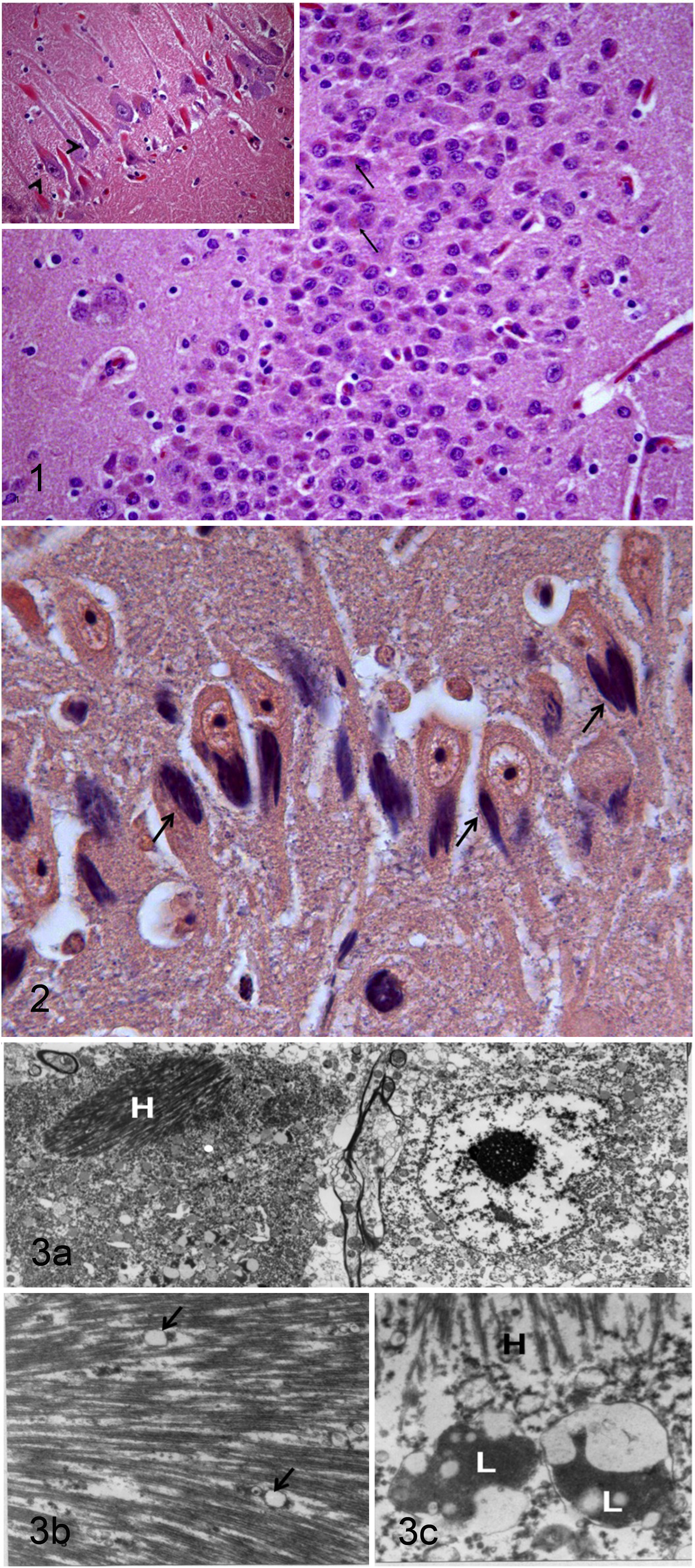
The major histologic finding within the brain was myriad intraneuronal, intracytoplasmic (somatic and axonal) eosinophilic crystalline inclusions bodies, especially within the cerebral cortex, hippocampus, and, to a lesser extent, cerebellar cortex (Fig. 1 ). The affected neurons did not exhibit other signs of degeneration. No inclusions were seen within cerebellar Purkinje cells (Fig. 2). In addition to the inclusions, occasional random foci of neuronal necrosis with gitter cells, neutrophils, and moderate neuropil vacuolation were present within the neuroparenchyma of the brainstem. No etiologic agents were found within these brainstem lesions, but their appearance and distribution were most consistent with migration tracts from nematode larvae such as Parelaphostrongylus tenuis. No known parasiticide treatments were done on this animal. No other changes, such as plaques or tangles, were observed within the examined sections of brain of this old alpaca (lifespan is between 15 and 20 years; R. Melidone, personal observation).
The macroscopically detected areas of white–tan discoloration of the endocardium and myocardium of the left ventricle and interventricular septum corresponded histologically to large bands of fibrous tissue and were likely responsible for the cardiac abnormalities detected clinically.
Immunohistochemical staining revealed that the previously described intracytoplasmic, intraneuronal crystalline inclusion bodies were negative for ubiquitine, smooth muscle actin, synculein, and tau. Negative immunohistochemical staining of the intraneuronal inclusions for synuclein and tau protein excluded the possibility of Lewy bodies and neurofibrillary tangles, respectively.
Hirano bodies are paracrystalline F-actin-rich intracellular aggregates of actin-associated proteins1,3,10 and tau proteins. 4 The presence of actin and actin-associated proteins could explain the positive staining with PTAH. It is likely that the negative immunohistochemical staining for smooth muscle actin and tau is due to the difference in the antigen expression between human and alpacas. However, all the inclusions stained positive for PTAH and negative for Bielchovski stain. Ultrastructural examination revealed that the inclusions were multilamellar and filamentous (Fig. 3A) with longitudinal herringbone pattern (Fig. 3B) and cross-sectional latticelike structure (Fig. 3C).
The results of HE, immunohistochemistry, PTAH stain, and ultrastructural examination are consistent with Hirano-like bodies.
Discussion
Hirano bodies were initially reported as highly organized structures in neurons 8 and other cell types both in vivo and in vitro 1 as well as in the eukaryote organism Dictyostelium amoebae. 10 They are occasionally found in the hippocampal/pyramidal cells in some human neurodegenerative disorders, such as Alzheimer disease, amyotrophic lateral sclerosis, Parkinson–dementia complex of Guam, Pick disease, prion diseases, hepatic encephalopathy, and chronic alchoholism. 1 They have been reported in diabetic rats.1,2 Hirano bodies have been also reported in oligodendrocytes, Schwann cells, and astrocytoma5,11,15 and within the skeletal muscles of a human patient affected by adult glycosinosis II, 2 as well as within neurons of the urinary bladder of normal dogs 16 and in canine testicular interstitial Leydig cell. 14 Hirano bodies have been experimentally observed in cultures of 6 different cell lines, some of which are not of neural origin 1 as well as in the eukaryote organism D. amoebae. 10 They appear as rodlike eosinophilic, highly refractile, structures on HE-stained sections. Ultrastructurally, they are characterized by osmiophilic crystalloid arrays of interlacing filaments forming latticelike structures or herringbone configuration with electron-dense material between the fibrillary structures.7,9
It has been speculated that Hirano bodies are a cell response to an aberrant function of the actin cytoskeleton. 10 However, little is known about the exact mechanism that leads to the formation of Hirano bodies. 13 The eukaryote organism D. discoideum has been used as a live cell model to study the mechanisms of the formation of Hirano bodies. 13 It has been suggested that unregulated protein fragments play a major role in initiating cell toxicity and, ultimately, cell death in Alzheimer disease.6,12 However, the underlying mechanisms that lead to protein aggregation, Hirano bodies formation, and their role in neurodegeneration are unclear. 13 The results of another study conducted on a cell culture model demonstrated that although Hirano bodies are considered to be associated with a large number of pathologic conditions, they are not necessarily cytotoxic. 1 Hirano bodies have also been reported in several normal tissue specimens from humans and animals.10,17 It has been observed that neurites of neuronal cells with Hirano bodies appear normal, 1 implying that Hirano bodies may not affect the neuronal cell function. It has also recently been observed that Hirano bodies form by the aggregation of numerous smaller inclusions. 13
In conclusion, it is unclear whether the neurologic signs expressed by this alpaca were specifically related to one of the previously described central nervous system lesions detected in this animal or to both; however, according to the current literature, it is highly unlikely that the Hirano-like bodies detected in the brain of this alpaca contributed to the neurologic clinical signs.
The exact mechanism of the formation of Hirano bodies is still unclear, as well as their exact role in the pathogenesis of many of the conditions they have been associated with. Furthermore, Hirano bodies have been found in normal human and animal tissues, supporting the hypothesis that Hirano bodies are not necessarily a primary pathologic lesion but may be present secondary to other processes. Based on our experience—specifically, 1 to 2 alpacas necropsied each week on average, with complete gross examination of all brains of all animals and with histologic examination of representative sections of all brains—Hirano-like bodies are not a normal finding in the brain of alpacas. To the best of our knowledge, this is the first report of cerebral Hirano-like bodies in an alpaca, and as previously stated, their exact significance and role are uncertain.
Footnotes
Acknowledgements
We thank Annette Shepard-Barry and Inna Lomakin for their technical support.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
