Abstract
A novel form of neuroaxonal dystrophy is described in 3 Chihuahua pups, 2 of which were from the same litter. It was characterized not only by accumulation of numerous and widely distributed axonal swellings (spheroids) but also by a severe cavitating leukoencephalopathy. The dogs presented with progressive neurological signs, including gait abnormalities and postural reaction deficits. Magnetic resonance images and gross examination at necropsy revealed dilation of lateral ventricles and cerebral atrophy, accompanied by cavitation of the subcortical white matter. Histopathologically, severe axonal degeneration with formation of large spheroids was found in the cerebral and cerebellar white matter, thalamus, and brainstem nuclei. Small-caliber spheroids were observed in the cerebral and cerebellar gray matter. The telencephalic white matter had severe myelin loss and cavitation with relative sparing of the U-fibers. Different from previously reported cases of canine neuroaxonal dystrophy, in these Chihuahuas the spheroid distribution predominantly involved the white matter with secondary severe leukoencephalopathy.
Neuroaxonal dystrophy (NAD) is a progressive neurodegenerative disorder characterized by severe degeneration of neurons and their processes, resulting in the accumulation of abundant focal axonal swellings (spheroids) in the central nervous system (CNS), particularly in the brainstem and spinal cord. 33 Spheroid formation can be an age-related condition (physiological) as well as a primary or secondary phenomenon, developing in diseases that involve neuronal degeneration. 20,32 Primary NAD occurs when the initial pathologic feature is the development of axonal swellings. Secondary NAD occurs as a result of other neurodegenerative and neurometabolic conditions (eg, equine degenerative myeloencephalopathy, feline Niemann-Pick disease type C, vitamin E deficiency), intoxication (eg, delayed organophosphate poisoning), trauma, and tumors. 7
Primary NAD has been reported in several species, including dogs, 9 –11,13,15,16,28 cats, 8,29,30,37 horses, 1 –3 sheep, 14,19,22,27 cattle, 18 and humans. 32 NAD has also been described in mice, both as spontaneous disease 6 and in genetically engineered mouse models. 34 In canine NAD, Rottweilers are a predisposed breed, 4,9,13 and it is also described in the Collie, 10 Chihuahua, 5 Jack Russell Terrier, 31 and Papillon 15 and, more recently, in Spanish Water Dog 17 and Dachshund-cross. 28 Reports regarding spontaneous cases of NAD in dogs have suggested that the disease could be an autosomal recessive disorder resembling the human infantile form in Jack Russell Terriers 31 and the juvenile form in Rottweilers. 33
The axonal swelling starts at the preterminal portion of the axon with distal atrophy and secondary myelin degradation. The axonal degeneration progresses proximally, resulting eventually in death of the neuronal cell body. Most frequent, spheroids are found in the nuclei of gray matter, both in the brain and in the spinal cord. 7,33 According to morphologic studies, spheroids contain tubulovesicular structures, neurofilaments, mitochondria, lysosomes, and membranous bodies. 7 The aim of this study was to describe the clinical, neuroradiologic, and neuropathologic features of a variant of NAD in 3 Chihuahua puppies in which axonal swelling was associated with severe myelin loss.
Two male Chihuahua dogs age 2 months from the same litter (dog Nos. 1 and 2) and a 2-month-old female Chihuahua (dog No. 3) not related to the other two puppies presented with sudden onset and progressive neurological signs of 1-week duration. No history of toxin exposure or drug treatment was reported. All dogs were regularly vaccinated and dewormed. Physical examination revealed poor general condition of dog Nos. 1 and 2, and neurologic examination revealed severe obtundation in all dogs. Dog Nos. 1 and 2 had cerebellar ataxia, intention tremor and hypermetria, especially in the forelimbs with the tendency of falling sideways and back, and postural reaction deficits. Nonambulatory tetraparesis was observed in dog No. 3 with postural reaction deficits in all 4 limbs. Ataxia and bilateral strabismus were also observed. Bilateral menace deficits were observed in all 3 dogs. The other cranial nerve functions and spinal reflexes were normal.
Neuroanatomic localization was consistent with an intracranial multifocal or diffuse lesion with predominant cerebellar involvement in all dogs. Blood samples were taken, and complete blood counts and biochemical profiles were performed. These were within the normal ranges for each dog.
Magnetic resonance imaging (MRI) of the neurocranium was performed using a 0.25 Tesla magnet (VetMR Grande®; Esaote, Genoa, Italy) before and after intravenous administration of gadolinium (Magnevist; 0.15 mmol/kg). The MRI studies included T1- and fast-T2-weighted spin echoes (SE T1, SE T2) and fluid-attenuated inversion recovery sequence, performed in all 3 spatial planes. Neuroimages were similar in all dogs. Moderate dilation of the lateral ventricles was detected in dog Nos. 1 and 2, and more severe dilation was observed in dog No. 3. Diffuse atrophy of the cerebral cortex, accompanied by flattening of the cerebral gyri, rupture of the septum pellucidum, and thinning of the corpus callosum were observed in all dogs. Bilateral and symmetrical cavitation of subcortical white matter of the frontal and parieto-occipital lobes was also detected (Fig. 1). No enhancement on postcontrast sequences was observed in all cases. Cerebrospinal fluid examination was unremarkable in all cases.
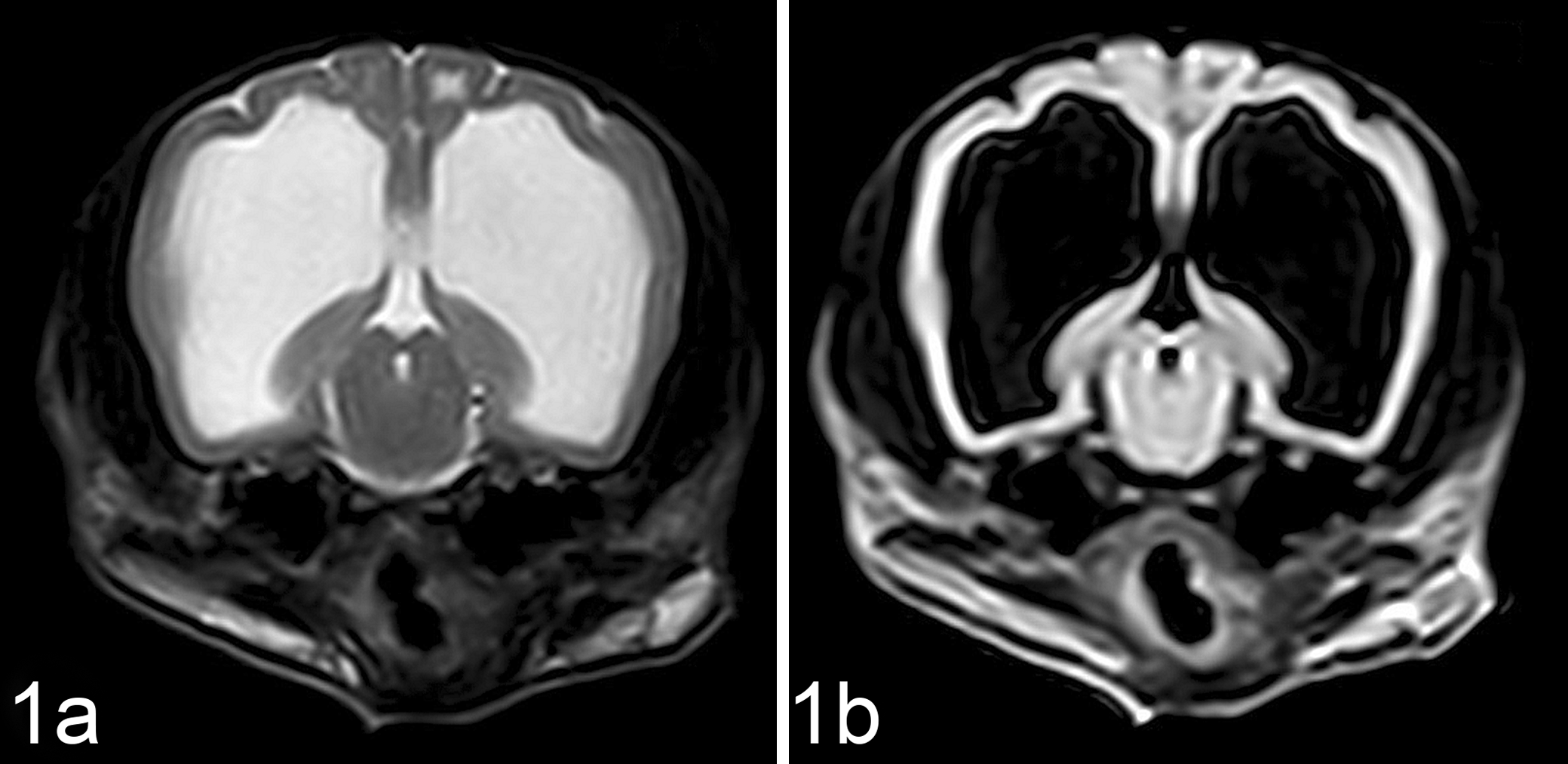
Brain, dog, case No. 3. T2-weighted (a) and fluid-attenuated inversion recovery (b) transverse magnetic resonance images of the brain at the level of mesencephalon. Cerebral atrophy with marked enlargement of the ventricular system is evident. Also note cavitation of subcortical white matter.
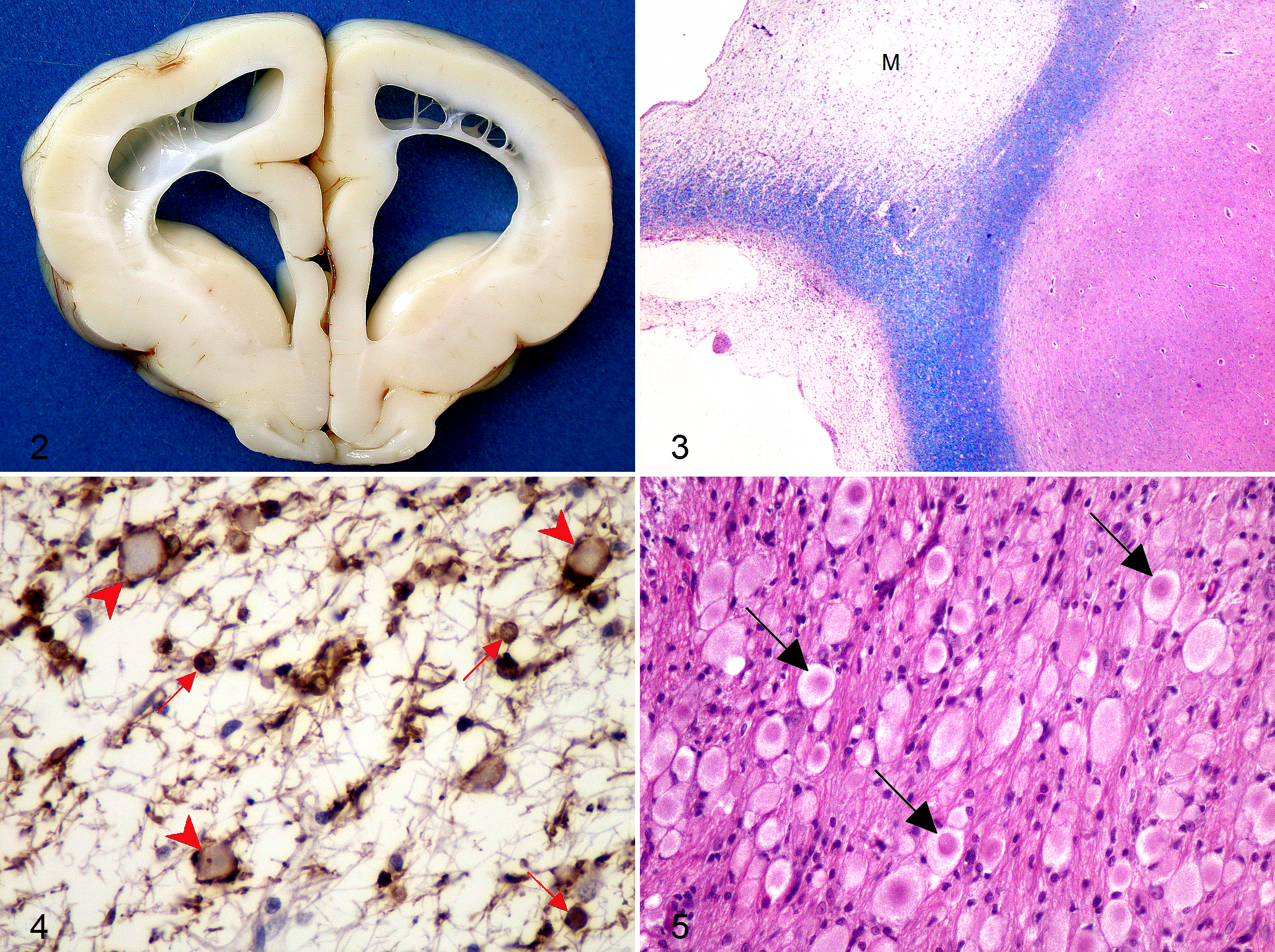
In all cases, because of poor prognosis and progressive worsening of the neurologic condition, owners elected euthanasia. A complete postmortem examination was performed on all dogs and the whole CNS and representative samples of major organs were fixed in phosphate buffered 10% formalin solution. Gross lesions were restricted to the CNS in all dogs. Cortical atrophy with flattening of cerebral gyri and ventricular enlargement, more severe in dog No. 3, was observed. Discoloration of the telencephalic white matter and striking cavitation of the subcortical white matter were also evident (Fig. 2). Moderate to severe thinning of the corpus callosum was observed in all dogs, associated with rupture of the septum pellucidum in dog Nos. 1 and 3. Thalamic, mesencephalic, and rhombencephalic structures were macroscopically normal.
Transverse sections of the brains and extraneural tissue samples were routinely processed for histology and sections were stained with hematoxylin and eosin, Luxol Fast Blue, periodic acid-Schiff, and Perls’ iron stain methods. Selected brain sections were stained immunohistochemically with antibodies against glial fibrillary acidic protein (1:1000; DakoCytomation, Carpinteria, CA), ubiquitin (1:1000; DakoCytomation), phosphorylated neurofilaments (clone 2F11, 1:1500; DakoCytomation), and 2’,3’-cyclic nucleotide 3’-phosphodiesterase (CNPase, 1:800; GeneTex, Irvine, CA). Heat-induced epitope retrieval using a pressure cooker was performed for all markers, except for antiubiquitin antibody. Primary antibodies were incubated overnight at 4°C and immunoreactivity was detected by the avidin-biotin-peroxidase complex method (Vectastain® Elite ABC-Peroxidase kit; Vector Laboratories, Inc., Burlingame, CA), using 3,3’-diaminobenzidine as chromogen. Negative controls were obtained by omitting the primary antibody. Small blocks of formalin-fixed subcortical white matter and cerebral cortex were collected and postfixed in osmium tetroxide, dehydrated in graded alcohols, and embedded in epoxy resin. Semithin (1 µm) sections were stained with toluidine blue, and ultrathin sections were cut and stained with uranyl acetate and lead citrate for electron microscopic examination.
Histologically, CNS lesions were similar in all dogs. Lesions were characterized by widespread severe myelin loss most prominent in the cerebrum and cerebellum with degeneration of descending tracts and relative preservation of the subcortical U-fibers (Fig. 3). Marked reduction of CNPase expression was evident, although CNPase-positive, morphologically normal oligodendrocytes were scattered within the leukodystrophic areas (Fig. 4). Numerous neuroaxonal spheroids were evident in the affected white matter. Spheroids were round, pale, eosinophilic and homogeneous or finely granular and varied in size from 10 to 50 µm. Some spheroids displayed a central hypereosinophilic core structure (Fig. 5). Spheroids were numerous and large (50 µm on average) in the white matter of the cerebrum and cerebellum and several nuclei of the brainstem including lateral cuneatus, spinal tract of trigeminal nerve, olivary, solitary tract, lateral lemniscus, cochlear, trapezoid body, and lateral and medial geniculate. The presence of axonal swellings was moderate in the pontine nuclei, transverse and longitudinal pontine fibers, caudal colliculi, and periaqueductal gray matter. A moderate number of spheroids was also found in the cerebellar nuclei and in the nucleus of vagus. Spheroids of smaller caliber were found in the cerebral and cerebellar cortices, prevalently in the molecular layer (Fig. 6). Scattered spheroids were evident in the reticular substance of the pons, medulla oblongata, and occasionally in the gray matter and all funiculi of the spinal cord. No axonal swellings were found in the basal nuclei, in the hypoglossal nucleus, and in the motor nucleus of the trigeminal nerve. The spheroid distribution is shown in Table 1. Occasional loss of Purkinje cells was observed in all cases, accompanied by moderate cytoplasmic vacuolation in dog No. 1. Thinning of the cerebral cortex was evident, but neuronal loss was not significantly accompanied by gliosis. The neuronal cell bodies, even in the areas in which spheroids were prominent, were well preserved and did not show morphologic abnormalities.
Neuroanatomic Distribution and Features of Axonal Spheroids.a

aWhere the 3 dogs are discordant, this is indicated. Number of spheroids scored from 0 to +3 in a high-power field (400×). 0 = absence of spheroids; +1 = widely distributed, small (10-30 μm) spheroids; +2 = aggregated small spheroids; +3 = mainly large diameter (30-50 μm) spheroids. GM, gray matter; N., nucleus; WM, white matter.
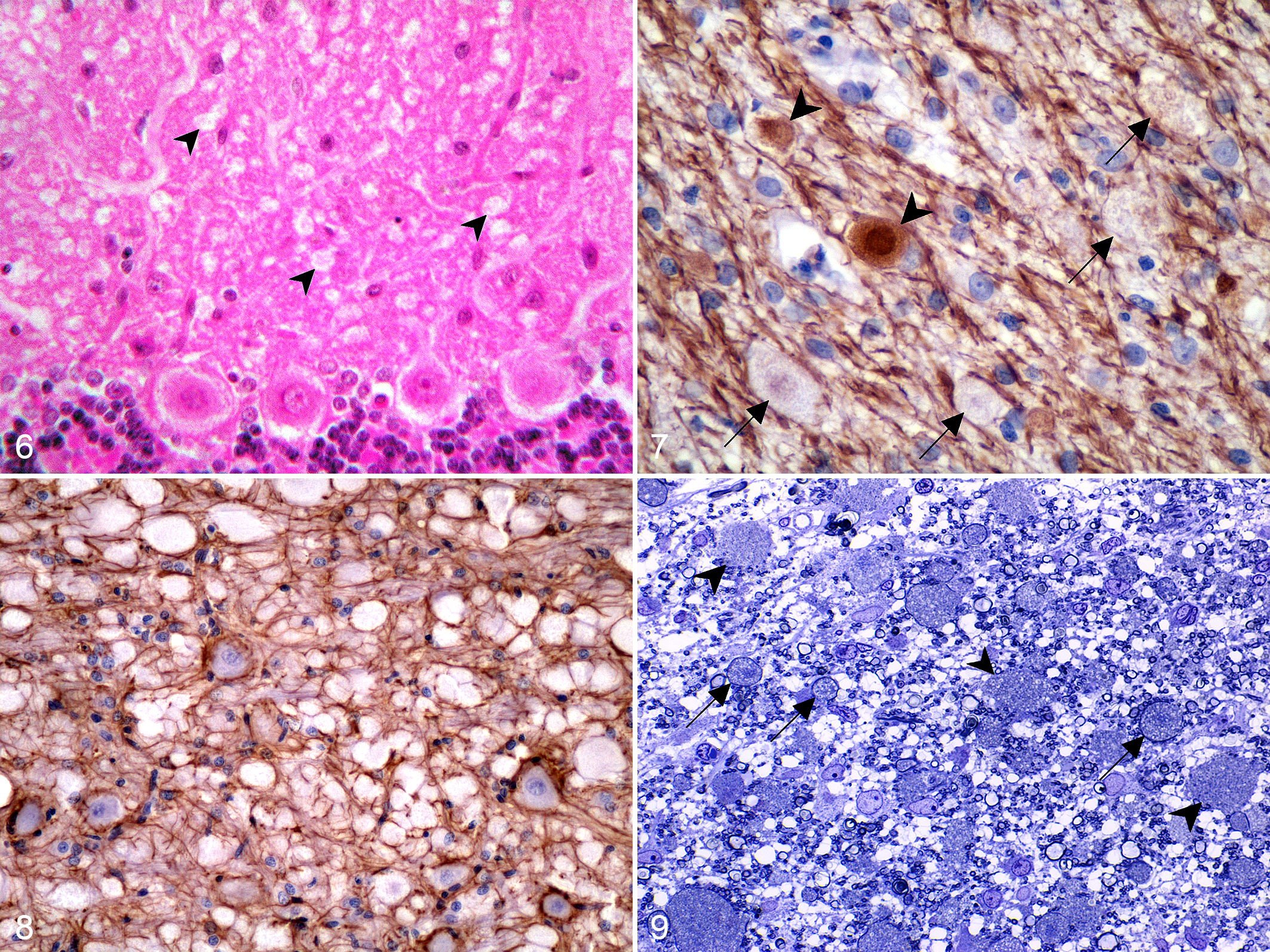
Most spheroids were inconsistently immunolabeled with phosphorylated neurofilaments. Only occasionally, some spheroids were strongly immunoreactive and others showed positive staining restricted to the periphery (Fig. 7). Spheroids were strongly reactive with antiubiquitin antibody and had a vacuolated and granular appearance. Immunostaining for glial fibrillary acidic protein revealed only a mild proliferation of fibrillary astrocytes even within the most severely affected areas (Fig. 8). Neither inflammatory cell infiltrates nor gitter cells were detected.
Ultrastructurally, in medium-size spheroids, myelin was thinner than normal because of severe axonal enlargement, whereas larger spheroids were almost devoid of myelin sheath. Rupture of some axon membranes was evident with consequent dispersion of the accumulated material into the interstitium. Fine vacuolation of the nervous tissue, interpreted as myelin vacuolation, was also observed (Fig. 9). Axonal spheroids contained concentric membranous vacuolated bodies, some morphologically normal mitochondria, and only a few interspersed aggregates of neurofilaments (Fig. 10). There were no ultrastructural differences between large spheroids in the white matter and smaller spheroids found in the gray matter. No relevant lesions were observed in extraneural tissue samples.
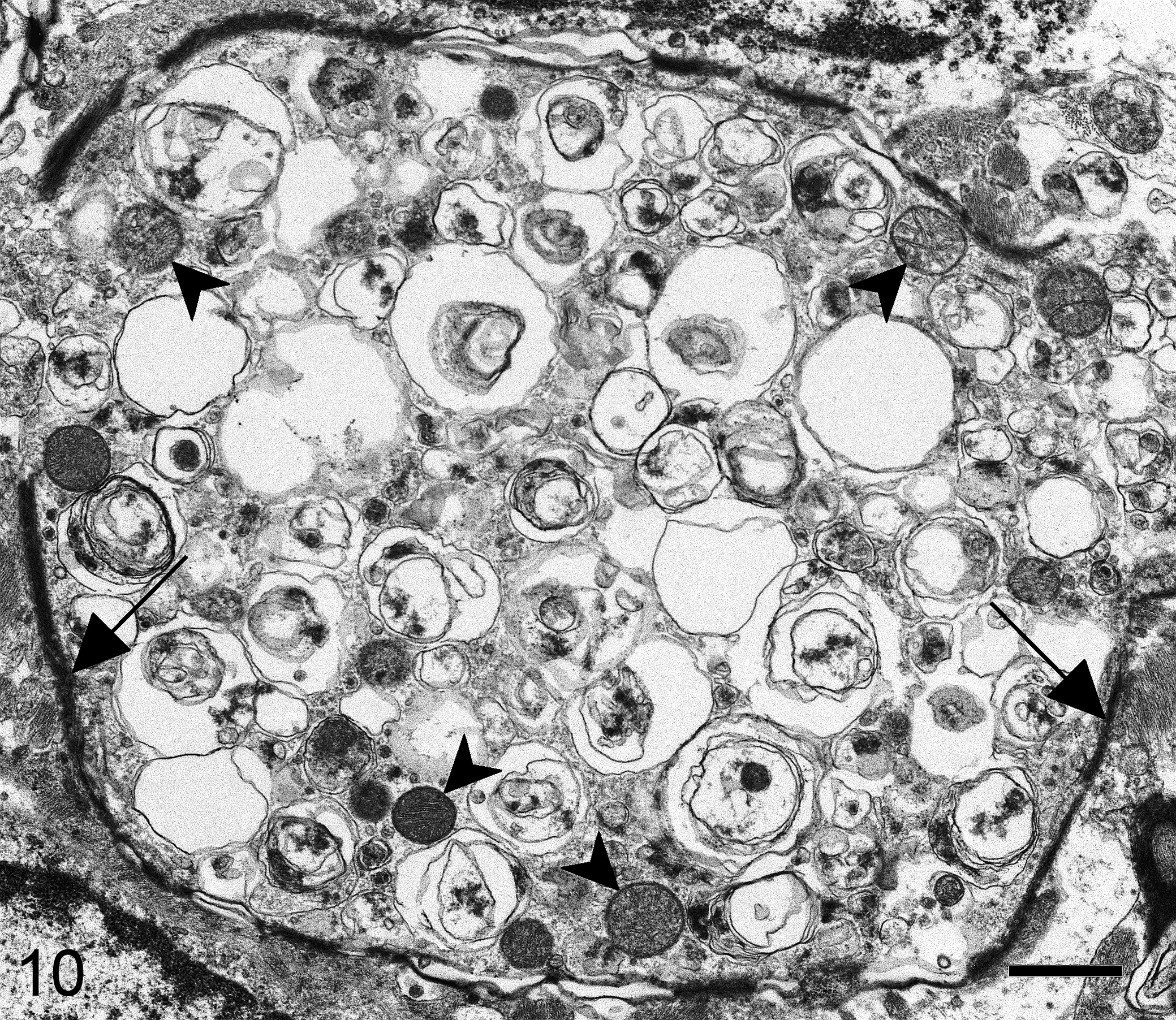
Brain, dog, case No. 2. A spheroid with thin, discontinuous myelin sheath (arrows) contained concentric membranous vacuolated bodies, peripherally located mitochondria (arrowheads), and inconspicuous aggregates of neurofilaments. Transmission electron microscopy; bar = 1 μm.
Histopathologic findings in our dogs were consistent with a form of NAD associated with marked leukoencephalopathy. Clinical and neurological presentation as well as progression of signs in dogs affected by NAD were similar to those shown by our dogs. Progressive neurologic abnormalities included severe obtundation, gait abnormalities with postural reaction deficits, and absence of menace response. 5,11 –13,24,25,28,35 The progression of signs was often very rapid and dogs with NAD had short survival. The early age of onset in our dogs is similar to that reported in other cases of NAD in Chihuahua and Papillon puppies. 12,24,35 The development of the disease in at least 2 puppies from the same litter suggests a familial background.
The use of MRI for detection of specific features of NAD in dogs is lacking. Neuroimages in our cases showed dilation of the lateral ventricles with diffuse brain atrophy and cavitation of subcortical white matter. Reported MRI features of canine NAD vary from no abnormalities 12 to severe atrophy of the cerebellum. 24 Different from previous reports in dogs, in our cases cerebellar atrophy was not detected. Dilation of lateral ventricles with cerebral atrophy was described in a Papillon puppy, 35 whereas cavitation of subcortical white matter of the cerebrum as observed in our cases was not previously reported in canine NAD.
The distribution pattern of axonal spheroids in our dogs was dissimilar to previous reports of canine NAD, in which spheroids were mostly found in the nuclei of the gray matter in the brain and spinal cord. 11,12,17,25 In Papillons, spheroids were described in the white matter, although, also in this breed, neuroaxonal changes primarily affected the gray matter. 12,25 In 2 Chihuahuas, 5 spheroids were mainly located throughout the white matter of the brain and thalamus. In our cases, a consistent involvement of cerebral and cerebellar white matter with marked and widespread accumulation of large spheroids was accompanied by severe cerebral atrophy and cavitating leukoencephalopathy, not previously reported in canine NAD. In most forms of NAD, the axonal swellings are presynaptic and hence are usually observed in the nuclei of the gray matter. The diffuse distribution of spheroids in the white matter, with relative preservation of the subcortical U-fibers, suggests that neuroaxonal degeneration of long projecting fibers underlies this disease entity. Ultrastructural findings showed that myelin loss was more severe with the increase of axonal swelling, and hence, it is reasonable that myelin sheath abnormalities can be considered as a consequence of axonal dystrophy. Moreover, the detection of morphologically normal oligodendrocytes within unaffected and leukodystrophic areas can allow the ruling out of a possible hypomyelinating process. The fact that these pups did not show clinical signs at birth might also tend to exclude hypomyelinogenesis.
Numerous spheroids of smaller caliber, compared with those present in the white matter areas, were found in both the cerebral and cerebellar cortices. The fact that there were no ultrastructural differences between spheroids of different sizes and locations could reinforce the hypothesis that they were of 1 type (ie, primary), as there are reported differences in fine structural spheroidal composition between primary and secondary forms of NAD. 6 The differing sizes and staining intensities of spheroids with HE staining could reflect the fact that they were at different stages of temporal evolution and/or of different aggregated protein composition.
The immunohistochemical findings in our cases partially differed from those described in dogs by other studies. Nibe et al 25 reported neurofilaments as 1 of the major components of spheroids in canine NAD. However, in our cases, spheroids showed only occasional positivity for phosphorylated neurofilaments and were composed of layered vesicular membranous material, similar to that described in 2 Chihuahua puppies. 5 Differences in immunoreactivity could reflect variable axonal disruption at different sites and/or reflect some fiber dissolution. The strong reactivity of spheroids with antiubiquitin antibody and the mild proliferation of reactive astrocytes were consistent with previous studies of canine NAD. 12,24,25 These findings suggest a perturbation of retrograde axonal transport with accumulation of proteins that possibly exceeds the capability of degradation by the ubiquitin-proteasome system. 25
The combination of leukoencephalopathy and neuroaxonal spheroids in the abnormal white matter is a novel finding in veterinary neuropathology, whereas it has been described in rare complex syndromes of humans, such as dermatoleukodystrophy with neuroaxonal spheroids, 23 polycystic lipomembranous osteodysplasia with sclerosing leukoencephalopathy (Nasu-Hakola disease), 36 and hereditary diffuse leukoencephalopathy with spheroids. Both neuroradiological and neuropathological features of our dogs only in part resemble this latter condition. Hereditary diffuse leukoencephalopathy with spheroids is an adult-age onset, autosomal dominant disease due to mutations in the colony-stimulating factor 1 receptor (CSF1R) gene. 26 Microscopically, diffuse demyelination of the subcortical white matter with sparing of the U-fibers, accompanied by numerous spheroids, is a finding consistently characteristic of hereditary diffuse leukoencephalopathy with spheroids. 21
The homogeneity of the clinical picture and histopathologic findings suggests that the disease in our dogs is a distinct variant of canine NAD, resembling the disease described by Blakemore and Palmer. 5 At this time, it appears that the disease in Chihuahuas could be an inherited neurodegenerative disorder. As for other leukoencephalopathies, the possible contribution of neuroimaging to the in vivo identification of the white matter changes in this NAD variant is remarkable, but the differential diagnosis based on the imaging features alone is very difficult. Awareness and recognition of additional cases and their pedigree analysis might help to investigate the genetic base of the disease.
Footnotes
Acknowledgements
We thank Prof Ian D. Duncan and Dr Abigail Radcliff (Department of Medical Sciences, University of Wisconsin–Madison) for acquiring and interpreting the electron microscopy images, and Mrs Lisa Baroncini for expert technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
