Abstract
Despite previous detection of Chlamydia pecorum in sporadic ovine abortions, published descriptions of naturally occurring infections with fetoplacental lesions are lacking. This report provides the first descriptions of severe necrosuppurative chorionitis with vasculitis, and fetal pyelonephritis and enteritis in late-term abortions of maiden ewes. Chlamydial infection was detected using a Chlamydia genus-specific qPCR (quantitative polymerase chain reaction) on tissue extracts from 3 fetuses. C. pecorum was identified using a targeted qPCR assay, which also determined infectious load within fetal tissues. The presence of viable C. pecorum in fetal samples was confirmed by cell culture. Multilocus sequence typing (MLST) data indicated that the C. pecorum strains from each fetus were identical and of sequence type (ST) 23. Chlamydia sp. immunohistochemistry showed strong positive immunolabeling of fetoplacental lesions. Other infectious abortigenic agents were excluded with specific testing. This report confirms C. pecorum as a likely cause of ovine abortion and provides the first descriptions of associated fetoplacental lesions in naturally infected sheep.
Chlamydiosis in ruminants is associated with subclinical enteric infection; localized diseases including conjunctivitis, enteritis, and mastitis; and systemic diseases including polyarthritis, encephalomyelitis, and abortion. 7,13,32,39 Ruminants are most commonly infected with Chlamydia pecorum and Chlamydia abortus, less frequently with Chlamydia psittaci, and rarely by Chlamydia suis. 7,32
Chlamydia pecorum is globally distributed and occurs in many species, including sheep, goats, cattle, pigs, chamois, alpine ibex, birds, and marsupials, including the highly susceptible koala. 6,7,17,39 C. pecorum has an estimated prevalence in lambs ranging from 17% to 50% in Australia and 12% to 26% in the United Kingdom. 40,41 A far greater prevalence of 96% was detected in one Australian study, which performed repeated bimonthly sampling in clinically normal lambs from New South Wales. 6 In lambs, C. pecorum infection can manifest clinically as keratoconjunctivitis and arthritis, whereas young cattle can develop sporadic bovine encephalomyelitis. 15,17,39,40
Chlamydial abortion is usually due to infection with C. abortus, except for in Australia and New Zealand where it is considered exotic. 7,13,34,41 In sheep, C. abortus causes ovine enzootic abortion, which is characterized by late-term abortion, stillbirth, and birth of weak neonates, which often die within 48 hours of birth. 7,22,27 Chlamydia abortus manifests similarly in goats, is occasionally reported in cattle, pigs, horses and wild ruminants, and infrequently causes abortion in pregnant women following contact with parturient sheep and goats. 7,13,27
In contrast, comparatively little is known about naturally occurring C. pecorum abortion. While there have been detections in aborting sheep and/or goats in France, Tunisia, Morocco, and Iran; cattle in Iran; and Mediterranean water buffalo in Italy, these studies lack descriptions of associated gross and histological lesions. 2 –4,14,41 There is one published description of fetoplacental lesions associated with naturally occurring C. pecorum abortion, in a 16-month-old Boer doe in the United States with fibrinonecrotic placentitis, also typical of C. abortus infection, and fibrinosuppurative enteritis. 13
To our knowledge, the present report provides the first descriptions of gross and histopathologic fetoplacental lesions occurring with natural C. pecorum ovine infection, with etiological confirmation by PCR (polymerase chain reaction), isolation in cell culture, and immunohistochemistry (IHC).
Materials and Methods
Farm History
In early September 2018, in northwestern New South Wales, Australia, 3 aborted fetuses were observed by a producer among a group of late gestational maiden ewes, aged 12 to 14 months, 1 to 2 weeks prior to normal parturition. Fetuses were sent to the Elizabeth Macarthur Agricultural Institute (EMAI) for postmortem. Fetus 1 (F1), a male, and fetus 3 (F3), a female, were at approximately 126 days of gestation. Fetus 2 (F2) was male and at approximately 112 days of gestation. The property had approximately 1350 sheep, and pregnancy rates, as determined by ultrasound scanning, were 100% in the maiden groups and 159% in the multiparous group. At the conclusion of the lambing season, the producer reported a low rate of lambs weaned per ewe in the maiden ewes (66%), compared to the multiparous ewes (136%), and had noted several stillbirths per day in mid to late September 2018, sometimes associated with dystocia and uterine prolapse. At this time, blood samples were submitted from 6 maiden ewes with previously confirmed pregnancy that were found to be nonlactational, suggesting that they had experienced abortion, stillbirth, or early neonatal loss.
Pathologic Evaluation of Fetoplacental Samples
Postmortem examination of the 3 fetuses (F1–F3) was conducted at EMAI and samples were collected of placenta (chorioallantois and allantoamnion) and umbilicus (available only for F1 and F2), and fetal brain, liver, lung, heart, kidney, spleen, abomasum and small intestine, and pericardial, pleural, and abomasal fluid. Tissues were fixed in 10% neutral buffered formalin for 24 hours, routinely processed into 4-μm-thick sections, and stained using standard methods for hematoxylin and eosin (H&E) and modified Ziehl-Neelsen (MZN). Paraffin-embedded sections were also processed for IHC using a primary mouse monoclonal antibody to Chlamydia lipopolysaccharide (Clone B410F, Thermo Fisher Scientific). Sections were incubated with primary antibody diluted to 1/50 at room temperature for 1 hour, washed and sequentially incubated with anti-mouse serum (EnVisionDako, Agilent) and chromogenic solution (Dakocytomation DAB+, Dako, Agilent). Paraffin-embedded sections of Chlamydia genus-specific qPCR-negative ovine fetoplacental tissue was used as a negative control. Tissue sections were also processed without the primary antibody as an additional control. Additional sections from the brain of F2 were submitted to the University of Sydney Veterinary Pathology Diagnostic Services and processed for IHC detection of Toxoplasma gondii antigen, using rabbit anti–Toxoplasma gondii poylclonal serum (B-282-A1, Thermo Fischer Scientific). 10
Molecular, Bacteriological, and Serological Assays
All fetoplacental samples were tested in a genus-specific qPCR for Chlamydia spp. 11 and a species-specific qPCR for C. pecorum. 28
DNA was prepared by pelleting 500 µl of phosphate buffered gelatin saline transport medium containing swabs of fetoplacental samples at 13 000 rpm for 3 minutes in a benchtop centrifuge, extracting with InstaGene Matrix (Bio-Rad) according to the manufacturer’s instructions. Amplification of DNA by PCR for each assay was performed in a final reaction volume of 20 µl using 10 µl of TaqMan Environmental Master Mix 2.0 (Applied Biosystems), 2 µl of template and primers and probes (Supplemental Table S1). The assays were run on an Applied Biosystems 7500 Fast thermocycler (ThermoFisher) under the following cycling conditions: 95 °C for 10 minutes for 1 cycle, followed by 45 cycles of 95 °C for 15 seconds and 60 °C for 60 seconds. Plasmid standards were generated by cloning C. pecorum amplicons into the TOPO TA cloning vector and transforming into Escherichia coli TOP 10 cells (ThermoFisher), and serially diluted in yeast tRNA (Sigma Aldrich) to determine the limit of detection of each assay (1 gene copy). Standards were run in duplicate in each assay to enable calculation of the number of gene copies detected, and positive and negative controls were included in each run.
qPCR for Chlamydia abortus 29 was performed on all kidney, brain, and fetal membrane samples, and qPCR for Leptospira spp. 38 was performed on all abomasal fluids. qPCR for Coxiella burnetii was performed by the Australian Rickettsial Reference Laboratory using samples of liver and kidney (F3) and fetal membranes (F1).
All abomasal fluid samples were subjected to standard aerobic culture, and abomasal fluids and livers were cultured using selective medium for Campylobacter spp.
Chlamydial species isolation using samples of lung homogenate and abomasal fluid from F1 was attempted in cell culture using monkey kidney cells (BGM) 5 and mouse fibroblast cells (McCoy, ATCC CRL-1696). Both cell lines were grown in Minimum Essential Medium (MEM, Gibco) supplemented with 10% fetal bovine serum (fbs) and antibiotics (amphotericin B 2 μg/ml, gentamycin 5 μg/ml). The cells were grown in 12.5 cm2 tissue culture flasks until the monolayer was approximately 90% confluent, at which time the growth medium was removed for adsorption of the inoculum. The monolayer was washed twice with serum-free MEM medium, which was then discarded and either 10 µl or 100 µl of sample suspension was added to 500 µl of serum-free MEM and pipetted onto the cell monolayer. For inoculation, abomasal fluid was diluted 1/20 in serum-free MEM and lung was prepared as a clarified 10% suspension in serum-free MEM. The flasks were then placed on a rocking platform at 37 °C for 1 hour and then centrifuged at 1000g for 30 minutes at room temperature (approximately 23 °C). Five milliliters of MEM containing 2% fbs, antibiotics, and cycloheximide (1 μg/ml) was then added to each flask, which was incubated at 38 °C for 5 to 7 days. Cultures were examined by light microscopy daily, and after 5 to 7 days the cells were scraped from the surface of the flask with a rubber scraper into 1 ml of residual culture medium, sonicated 3 times for 10 seconds each, and then subcultured onto a new near-confluent monolayer by adding 100 µl of cell suspension to 500 µl of serum-free medium. The adsorption and centrifugation were completed as previously described, new maintenance medium added, and cultures examined for another 5 to 7 days. In the absence of observable cytopathology, the subculturing was repeated twice more. When changes consistent with chlamydial replication were observed, duplicate flasks were prepared, the culture supernatant was carefully removed, and monolayers were fixed in situ with a 1/10 dilution of formalin solution in phosphate-buffered saline. After fixation for 1 hour, the formalin solution was removed and immunoperoxidase staining completed using the primary mouse monoclonal antibody to Chlamydia lipopolysaccharide (Clone B410F, Thermo Fisher Scientific) and peroxidase labelled goat anti-mouse IgG (Dako) with AEC (3 amino 9 ethylcarbazole) substrate. Samples of cells and culture fluid were also tested in a C. pecorum qPCR as described above.
All pleural fluid samples were tested for antibodies against T. gondii by latex agglutination test (ELITex Toxo, Ref 66612, ELITechGroup). Six sera from nonlactating maiden ewes with presumed fetal or neonatal lamb loss were also tested in an agar gel immunodiffusion test for detection of antibodies to ruminant pestiviruses. 21
Multilocus sequence typing (MLST) was performed on DNA extracts from fetal membranes from F1 and F2, and kidney and brain from F3 as fetal membranes were not available. MLST was performed using the scheme of Jelocnik et al with primers targeting 7 different housekeeping genes (gatA, oppA_3, hflX, gidA, enoA, hemN, and fumC); 16 however, all reactions were instead performed with BioTaq polymerase (Bioline). Amplicons were purified using the Qiaquick PCR purification kit (Qiagen). Sequencing of all amplicons was performed at the Australian Genome Research Facility (Westmead, Sydney). MLST analysis of sequence data was performed using the software package, mlst (Seemann T, https://github.com/tseemann/mlst). This publication made use of the Chlamydiales MLST website, the development of which had been funded by the Wellcome Trust (https://pubmlst.org/chlamydiales/) sited at the University of Oxford. 19
Results
Postmortem Findings
Three fetuses designated F1 to F3 were examined. The placenta of F1 had diffusely dark red-brown cotyledons with patchy red-brown surface exudate, and multifocally thickened, reddened, opaque intercotyledonary tissue with markedly injected vasculature. Additionally, the fetal membranes and external body surface were extensively covered in meconium (Figs. 1, 2). The placenta of F2 had diffusely pale red to brown cotyledons, and patchy regions of mildly thickened, dark pink intercotyledonary tissue. The only changes observed in F3 were multifocal mild pulmonary ecchymoses, and no gross lesions were observed in F1 or F2.
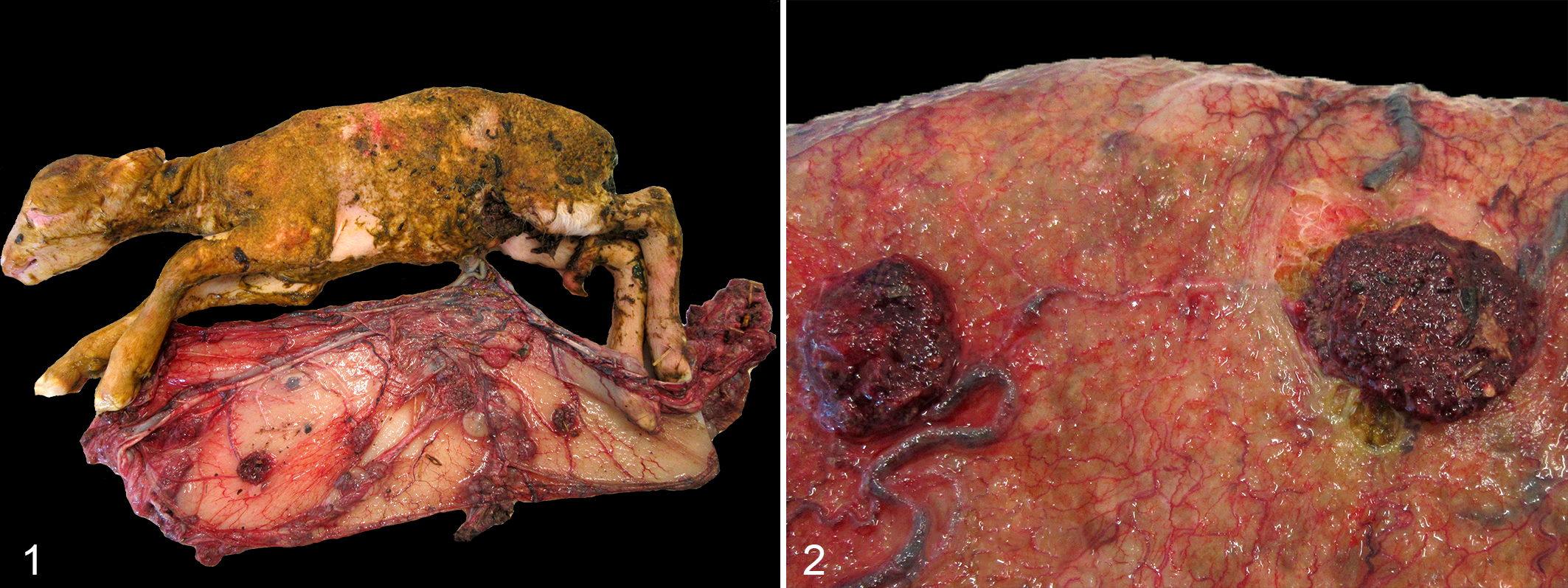
Chlamydia pecorum sporadic ovine abortion, sheep. Fetus 1 with fetal membranes. Cotyledons are covered with red-brown necrosuppurative debris, and intercotyledonary tissue is reddened and thickened with prominent vasculature. The skin surface is diffusely stained with meconium.
Histopathologic Results
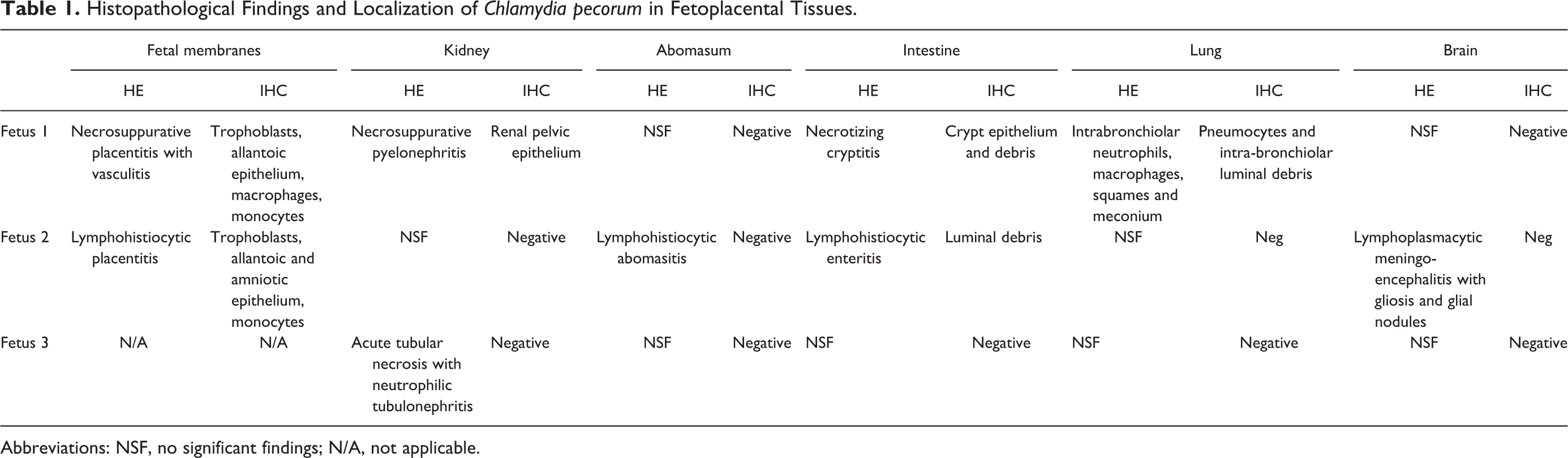
Histopathology and Chlamydia sp. IHC results are summarized in Table 1. In F1 large areas of the cotyledonary and intercotyledonary trophoblastic epithelium were multifocally lost and replaced by cellular debris, hemorrhage, fibrin, neutrophils, macrophages, and occasional lymphocytes (Fig. 3). Similar inflammatory cells diffusely infiltrated the villus stroma and intercotyledonary interstitium, predominantly subjacent to the chorionic surface. There was multifocal fibrinoid vasculitis with thrombosis in intercotyledonary and hilar zones, with fibrinoid necrosis of the tunica intima and media (Fig. 3). Numerous scattered cotyledonary trophoblasts contained intracytoplasmic, acid-fast, circular bodies, interpreted as chlamydial inclusions (inset, Fig. 3). Fetal tissues were also affected, with multifocal marked necrosuppurative pyelonephritis (Fig. 4). In the intestines there was a moderate multifocal necrotizing cryptitis (Fig. 5), and the lungs had bronchiolar intraluminal mucinous material admixed with cellular debris, neutrophils, macrophages, meconium deposits and squames (Fig. 6).
Histopathological Findings and Localization of Chlamydia pecorum in Fetoplacental Tissues.
Abbreviations: NSF, no significant findings; N/A, not applicable.
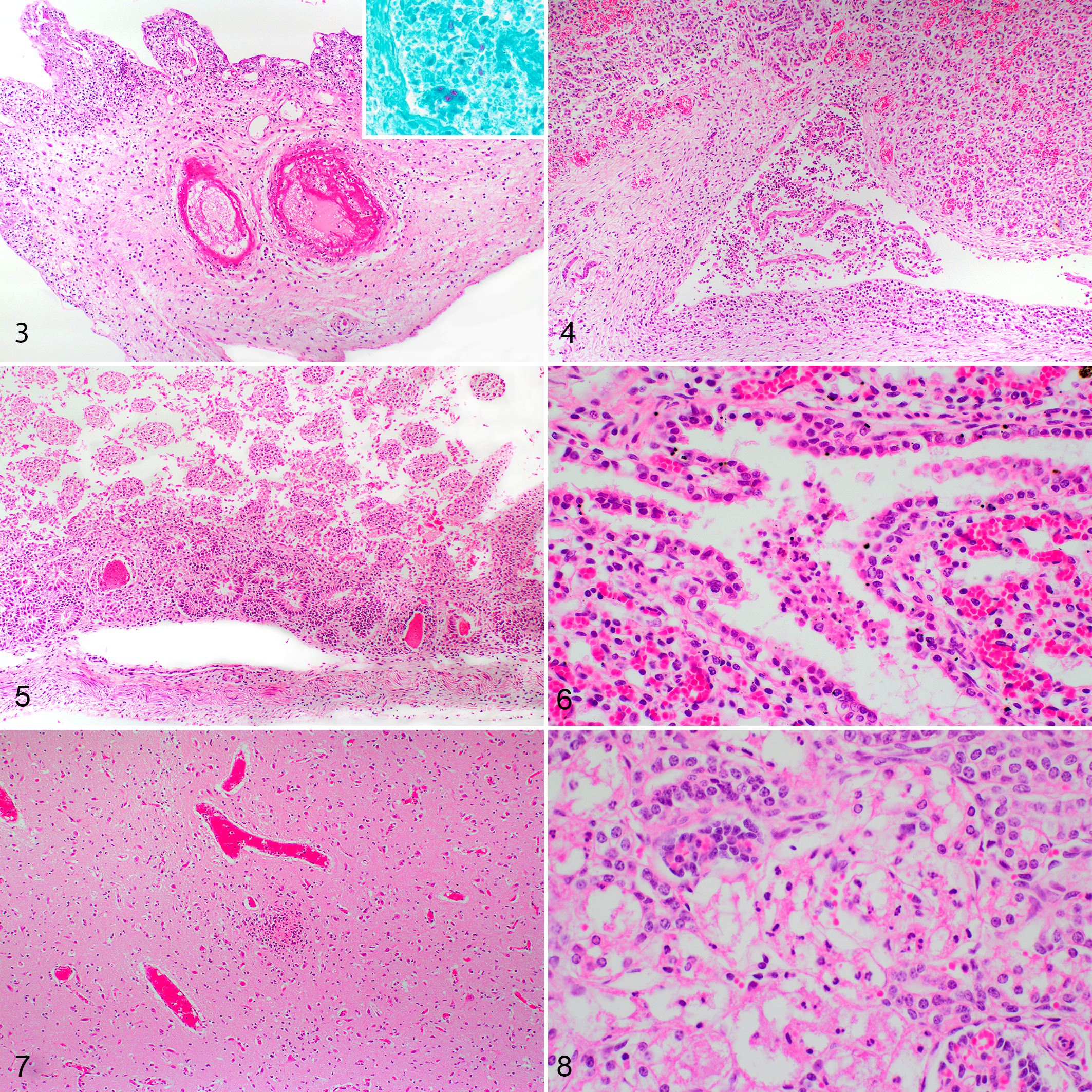
Chlamydia pecorum sporadic ovine abortion, sheep.
Placental lesions were comparatively mild in F2, with no vasculitis. The chorioallantois was moderately autolytic, and chorionic villi were fragmented, mildly hypercellular and multifocally effaced by large amounts of cellular debris. The allantoamnion had multifocal infiltrates of macrophages and occasional lymphocytes and neutrophils, predominantly perivascularly and subjacent to the allantoic epithelium. Within the cerebrum and rostral brainstem of F2 there was multifocal mild to moderate gliosis with occasional glial nodules (Fig. 7), lymphohistiocytic perivascular cuffs up to 3 cells thick, and similar multifocal mild leptomeningeal infiltrates. The abomasal and small intestinal mucosa was diffusely infiltrated by low to moderate numbers of lymphocytes, macrophages and fewer neutrophils, also observed within scattered submucosal lymphatic vessels.
Inflammatory lesions in F3 were only present in the kidney, where the proximal convoluted tubular epithelium was multifocally desquamated, fragmented, lacking nuclear detail, and replaced by cellular debris and low to moderate numbers of neutrophils (Fig. 8).
Immunohistochemistry Results
In F1, there was strong immunolabeling in trophoblast cells, associated with round, intracytoplasmic inclusions, and within necrotic chorionic debris, allantoic epithelial cells and occasional interstitial macrophages (Figs. 9, 10). There was also intense immunolabeling in cellular debris within the chorion and renal pelvis, and in intracytoplasmic inclusions in sloughed epithelial cells in the renal pelvis (Figs. 11, 12), intestinal crypt epithelium (Fig. 13), bronchiolar luminal debris (Fig. 14), occasional pneumocytes, and circulating monocytes and tissue macrophages within fetal membranes. In F2, extensive, florid positive immunolabeling was observed in the small intestinal luminal debris and in low numbers of scattered chorionic, allantoic, and amniotic epithelial cells, and monocytes circulating within placental vessels. In F2, neither chlamydial nor T. gondii antigen was detected in the brain by IHC. No positive immunolabeling was observed in any tissues of F3.
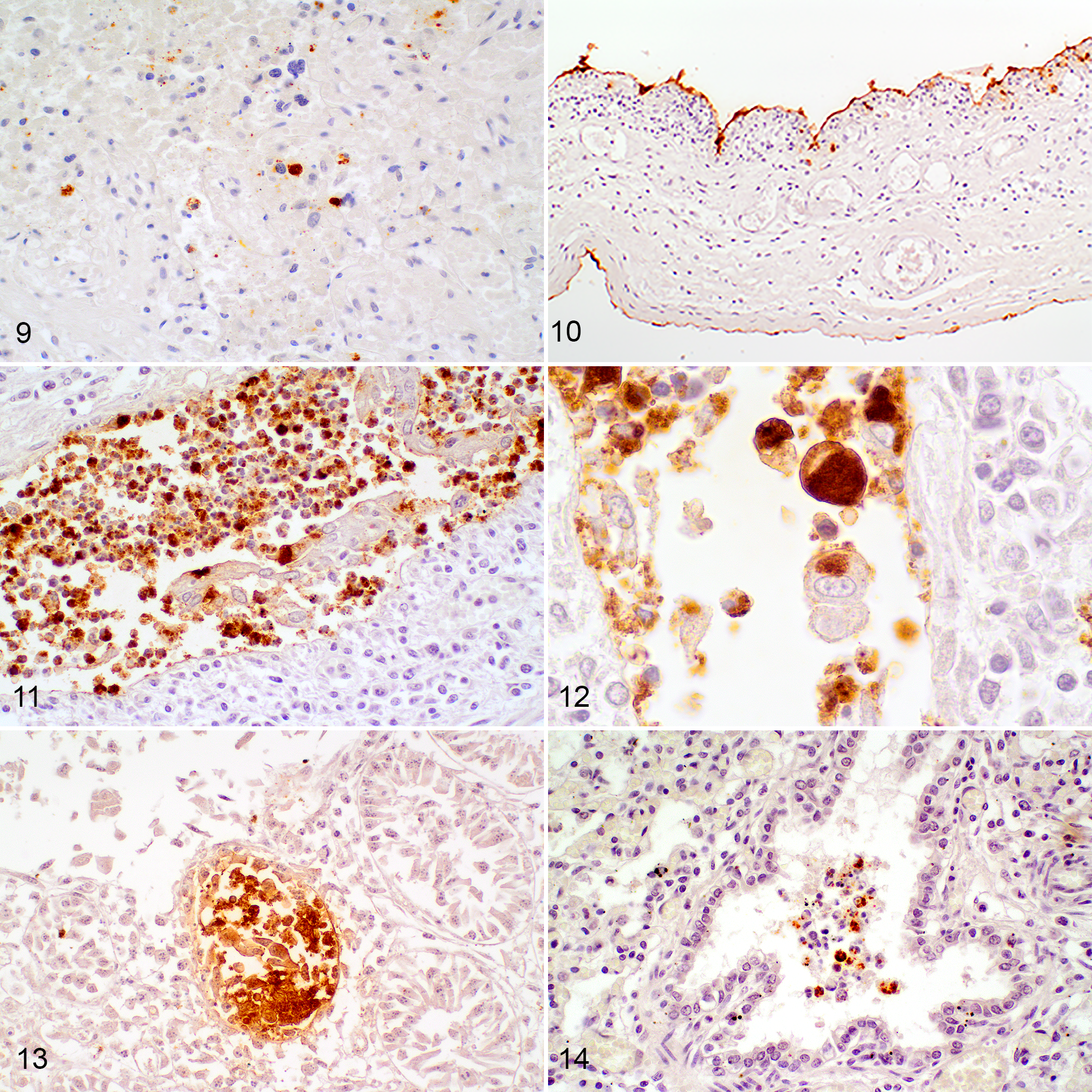
Chlamydia pecorum sporadic ovine abortion, fetus 1. Immunohistochemistry for Chlamydia spp.
Molecular, Bacteriological, and Serological Results
Multiple fetal tissues of all 3 fetuses had positive results in the genus-specific Chlamydia qPCR, with the highest number of gene copies detected in the fetal membranes and kidney of F1 and in the fetal membranes of F2 (Fig. 15). Additionally, all tissues that were positive in the genus-specific Chlamydia qPCR were also positive in the C. pecorum–specific qPCR (Supplemental Table S2). Negative results were obtained by qPCR for C. abortus, Leptospira spp., and C. burnetii. Chlamydiales MLST analysis, using sequences directly amplified from tissue extracts, identified C. pecorum ST23 in all 3 fetuses. The sequences were identical in all samples including the kidney and brain samples from F3. Sequences for all MLST loci have been deposited in GenBank under accessions MT757677-83. Standard aerobic cultures and selective Campylobacter cultures did not yield any significant growth, and T. gondii and pestivirus serology were negative.
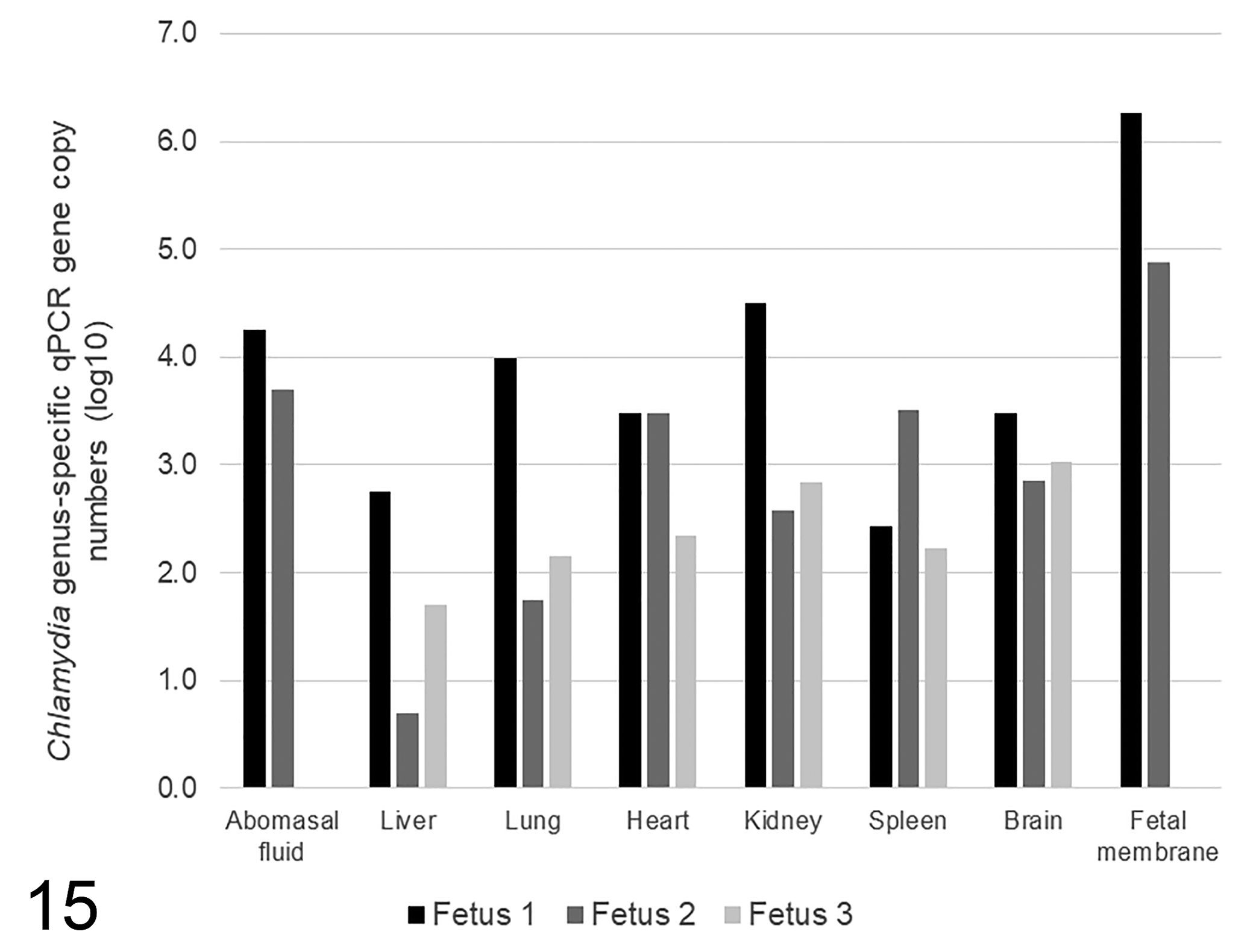
Chlamydia genus-specific qPCR gene copy numbers (log10) in fetal tissues and fluids from fetuses 1 to 3.
Chlamydial culture was attempted on samples of lung homogenate and abomasal fluid from F1. These samples were selected on the basis of high concentrations of chlamydial DNA, with Ct values less than 25. When McCoy cells that had been inoculated with the supernatant of homogenized fetal lung were examined by light microscopy at the third passage, there was evidence of the accumulation of refractile bodies after 3 to 4 days. Immunoperoxidase staining of these monolayers after 7 days revealed intensely stained pin-point foci throughout the inoculated cultures with no staining in uninoculated control monolayers. Culture supernatant collected at day 8 gave a Ct value of 18.9 in the C. pecorum qPCR compared to a Ct of 24.9 from the original sample of lung homogenate, confirming the replication of organisms in the cell culture. After 2 further passages and expansion of the cell culture, a Ct value of 17.1 was obtained from the culture supernatant. No replication was detected in the BGM cells. There was no evidence of replication of organisms from the sample of abomasal fluid after 3 passages in cell culture.
Discussion
Since the 1960s there have been occasional reports of sporadic ovine abortion in New South Wales associated with necrotizing placentitis, with intratrophoblastic inclusions and isolation of Chlamydia in embryonated chicken eggs, without species identification. 37 Australia and New Zealand differ from other sheep-producing countries in that C. abortus is an exotic pathogen, making C. pecorum a more likely cause of ruminant abortion where chlamydial infection is suspected. 13 Chlamydia pecorum has been previously detected in ovine abortion cases in the United States, France, Tunisia, and Morocco, but without associated lesions. 13,31 The present study describes and characterizes cases of naturally occurring C. pecorum abortion in sheep, with associated fetoplacental lesions. The likely etiological role of C. pecorum as a naturally occurring ovine abortigenic agent was confirmed on the basis of pathological findings, immunolabeling of fetoplacental lesions for Chlamydia sp., detection of C. pecorum by qPCR and MLST, and isolation of viable organisms in cell culture.
Historically considered the diagnostic gold standard, chlamydial isolation is no longer routinely performed as it requires specialist expertise and culture facilities, and is more time-consuming and less sensitive and specific than nucleic acid amplification tests. 7,13,32,34 However, when asserting the etiological importance of a suspect pathogen in a disease process, demonstration of viable, replicating organisms is required for greater confidence. 7 Additionally, bacterial isolation enables detailed molecular characterization, allowing for unambiguous identification of species and subtype. 34 There is little published information available for isolation of C. pecorum in cell culture; in this study, McCoy cells were used successfully, although human colonic adenocarcinoma cells (CaCo-2) have also been used in the past. 34
MLST has demonstrated that multiple genetically distinct C. pecorum genotypes are present in both healthy and diseased sheep and cattle, sometimes varying by anatomical site. 15,17,18 The genotype of C. pecorum found in all 3 fetuses was determined to be ST23, which has been previously associated with multiple disease states in different hosts, including ovine polyarthritis and conjunctivitis, and sporadic bovine encephalomyelitis in cattle in Western Australia, the United States, and England. 15 –18,39
Although not pathognomonic, gross placental lesions were consistent with findings in experimental ovine C. pecorum abortion following intravenous inoculation, 30 and gross and histologic findings with natural caprine C. pecorum abortion. 13 Other abortigenic pathogens were excluded using both direct qPCRs (Chlamydia abortus, Leptospira spp., and Coxiella burnetii), IHC for T. gondii, bacterial culture using standard aerobic and selective Campylobacter cultural methods, indirect serological methods (T. gondii and pestivirus), and on the basis of their exotic status within Australia (Brucella abortus).
The most severe fetoplacental lesions occurred in the chorioallantois and consisted of diffuse necrosuppurative placentitis with vasculitis (Figs. 1–3). This lesion is typical of many blood-borne bacterial pathogens, which enter maternal hematomata at the cotyledonary hilus and extend into adjacent chorionic villi and peripheral intercotyledonary tissues. 25,26,35
The precise mechanisms of ovine chlamydial abortion are unknown, although it is likely the result of a combination of immunological and physiological changes associated with pregnancy and stress, direct placental damage by chlamydial invasion, and subsequent impairment of maternal-fetal exchange of oxygen and nutrients, disruption of placental endocrine functions, fetal infection and the fetal inflammatory response, and thrombotic vasculitis. 9,23,32,33 Fetal stress in response to infection may also play a role in the pathogenesis of abortion and was suggested in this case by the presence of meconium on the body surface and within the bronchioles of F1. 35
Fetal enteritis with cryptitis, which is not recognized with C. abortus infection, has been suggested as a possible distinctive pathologic feature of C. pecorum abortion in natural caprine infection. 7,13 Our findings support this hypothesis, with intense immunolabeling in the intestines, which in F1 correlated with areas of cryptitis (Fig. 13). Enteric colonization with C. pecorum also occurs in postnatal ruminants, either causing persistent subclinical infection or enteritis and diarrhea. 13,32
The severe necrosuppurative pyelonephritis observed in F1 (Fig. 4) has not been previously described in experimental or naturally occurring ruminant abortion associated with either C. pecorum or C. abortus. Our results suggest that renal pelvic epithelium can be an additional site of chlamydial replication and cellular damage, given the presence of intraepithelial chlamydial inclusions and necrotic luminal debris within the renal pelvis (Figs. 4, 11, 12). In koalas, a species in which C. pecorum is a common cause of urogenital disease, IHC has also been used to demonstrate the presence of chlamydial antigen within the renal pelvis, bladder, endometrium, and prostate gland. 8 In this study, chlamydial antigen was also detected in allantoic epithelial cells (F1), which could suggest the possibility of an ascending urogenital infection. Additionally, in F1 the amniotic epithelium, intraluminal bronchiolar debris and intestinal crypt epithelium were positively immunolabeled, possibly suggesting fetal inhalation and ingestion of C. pecorum suspended in amniotic fluid. This hypothesis is supported by high levels of C. pecorum DNA detected by qPCR in F1 and F2 fetal membranes and abomasal fluid, and F1 lung and kidney, compared to other fetal tissues.
Fetal lesions were also observed in the brain of F2, including mononuclear perivascular cuffing and patchy gliosis, most pronounced in the brainstem, and a lymphoplasmacytic meningitis (Fig. 7). Similar neurologic lesions have been described with sporadic bovine encephalomyelitis in 12- to 16-week-old calves in Western Australia. 15,20,40 The failure to detect chlamydial antigen by IHC in any of the fetal brains, despite all brains being qPCR positive for C. pecorum, likely reflects the differences in sensitivity of these methods. An indirect cause of brain injury may also be considered, such as fetal hypoxia secondary to compromise of maternofetal circulation associated with placentitis, as postulated in some C. abortus experimental abortion trials in which multifocal leucomalacia and microgliosis were observed. 1,9,23,24,32,36 Hypoxic injury is also considered as a differential diagnosis for the acute renal tubular necrosis observed in F3.
In this study, the maiden group had a low rate of lambs weaned per ewe (66%) compared with 2018 New South Wales averages, which was in contrast to the excellent rate (136%) observed in the multiparous group. 12 This disparity surpasses the normal difference seen between maidens and multiparous ewes, and given our results, chlamydiosis is strongly suspected to have contributed to lower rate of lambs weaned per ewe in the maiden group. This suspicion is consistent with the farmer’s observations of abortions and still births, and the absence of other factors likely to have caused neonatal loss, such as extreme weather conditions, predation, or poorly managed nutrition. 12
In addition to causing abortion, C. abortus placentitis, particularly in ewes with low levels of placental colonization, can be subclinical, or can be associated with weak neonates experiencing poor survival. 1,26,33 It is possible that C. pecorum placentitis causes a similar spectrum of disease, given that experimentally inoculated pregnant sheep have produced live-born lambs with normal gestation lengths and birth weights, despite having macroscopic placental lesions. 30
Conclusion
This report confirms the likely abortigenic effect of C. pecorum in naturally infected sheep, with placental lesions similar to those described in experimental C. pecorum and natural C. abortus infections, fetal enteric lesions as observed in a naturally occurring C. pecorum caprine abortion case, and the first descriptions of fetal pyelonephritis associated with chlamydial abortion in a ruminant.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Ostfeld_et_al - Chlamydia pecorum–Associated Sporadic Ovine Abortion
Supplemental Material, Combined_supplemental_materials-Ostfeld_et_al for Chlamydia pecorum–Associated Sporadic Ovine Abortion by Thomas Westermann, Cheryl Jenkins, Emily Onizawa, Sarah Gestier, Justine McNally, Peter Kirkland, Jing Zhang, Daniel Bogema, Leah K. Manning, Keith Walker and Pedro Pinczowski in Veterinary Pathology
Footnotes
Acknowledgements
We would like to thank the North West Local Land Services New South Wales district veterinarian, Moree, for their excellent assistance with sample collection, and the landowners for their generous cooperation. EMAI pathology, virology, bacteriology, and serology teams, specimen receival and customer service officers are gratefully acknowledged.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
